6
Bringing New Products to Market and Ensuring Their Reach
Antimicrobial medicines are miracle drugs: highly effective (at least in the absence of resistance), relatively inexpensive, easy to use, and often with a broad spectrum of activity. The goal of the antimicrobial stewardship and preventive measures described in the previous chapter is to minimize the need for these medicines, prolonging the useful life of those already on the market. But the nature of resistance means that new antimicrobials will always be needed, as will diagnostic tools to correctly target therapy. There are also a range of promising nontraditional therapies, “ways to influence disease beyond inhibiting or killing pathogens through small molecules” (Theuretzbacher and Piddock, 2019). Examples include chemicals that act by reducing bacterial virulence instead of bacterial growth, and bacteriophages, the viruses that infect bacteria (Czaplewski et al., 2016; Kirienko et al., 2019).
The market for new medical products needed to combat antimicrobial resistance includes novel antimicrobials. While these products are at the center of much of the public discourse on antimicrobial resistance, new antimicrobials are not the only innovative products needed. Recognizing that there are finite resources to direct to this problem, the committee acknowledges that some trade-offs will be necessary. There is a need for an integrated investment across different product types, some preventive and some therapeutic—including, but not limited to, new antimicrobial medicines.
This chapter discusses some of the barriers to bringing new medical products to market and ensuring their reach. Challenges unique to human medicine and diagnostic markets in the United States are presented first. The last section describes problems that cut across multiple product lines
and countries. The recommendations in this chapter reflect the committee’s judgment that some special programs are needed to bring new antimicrobials to market and to ease the burden they place on diagnostic laboratories. At the same time, there is a need for a more explicit discussion of a balance of investments in a range of products that decrease use and preserve the life of antimicrobials. The concluding section describes a One Health portfolio of preventive and therapeutic products for both humans and animals and the importance of a holistic strategy for making these investments.
MEDICINES
At the center of the problem of the antimicrobials market is that the medicines are complicated and costly to develop and manufacture and have a relatively small market both in terms of duration of use, usually only a few days, and need (Chapman, 2020). Demand for these drugs is further constrained by the public health imperative to hold new antimicrobials in reserve (Ardal et al., 2020; McKenna, 2020). Given these constraints, it can more than 10 years of on-patent sales for a new antimicrobial to achieve profitability (McKenna, 2020). There is also a chance, depending on the pace of resistance in the target pathogen, that by this time the drug may no longer be an effective treatment. Although these medicines are essential to the future of clinical medicine, the present demand for them complicates their market viability.
For example, although there are over 2.8 million resistant infections every year in the United States, causing 35,000 deaths, infections with any particular resistant pathogen are rare (CDC, 2019a). A recent study of resistant gram-negative infections at 134 U.S. hospitals found that difficult-to-treat, gram-negative infections with no or poor treatment options were relatively rare (Strich et al., 2020). Across almost 3 million patient encounters, only 39 to 138 would be candidates for a novel antimicrobial against gram-negative infections (Strich et al., 2020). As long as the prevalence of gram-negative infections not susceptible to available treatments is substantially lower than the prevalence of infections susceptible to them, the market incentive alone is not likely to motivate new drug development (provided the prices are constrained) (Fitzpatrick, 2020).
The same pattern holds with more common resistant infections. Methicillin-resistant Staphylococcus aureus (MRSA) is one of the more common, serious resistant infections. By 2019 estimates, there are about 323,700 MRSA cases a year, causing 10,600 deaths, exceeding the National Institutes of Health (NIH) threshold for a rare disease by about 30 percent (CDC, 2019a; NCATS GARD, 2021). As with difficult-to-treat, gram-negative infections, there are still several effective treatment options available for MRSA (CDC, 2019a). While new treatments for MRSA, as for all resistant pathogens, are needed and valuable, good stewardship
would require older drugs be used first, even if there were no difference in price. When multiple antimicrobials can treat the same pathogen with similar effectiveness, the new medicine has more in common, economically, with a commodity, meaning it is largely interchangeable with similar products, than with a branded, niche product (Spellberg et al., 2013).
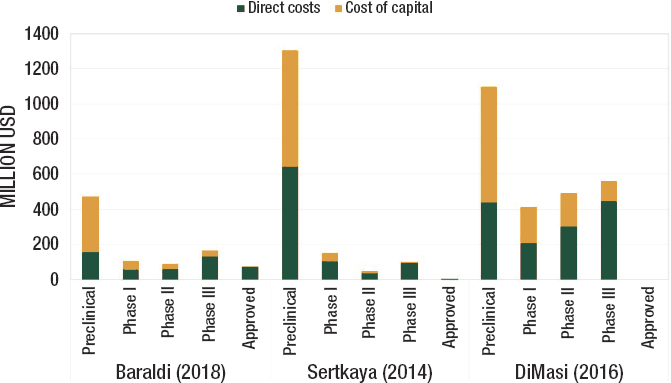
But unlike other commodities, the cost, time, and expertise needed to bring a new antimicrobial to market is extremely high. Estimates of the median development price for antimicrobials are between $673 million and $1.86 billion (Towse et al., 2017; Wouters et al., 2020).1 These estimates do not account for significant postmarket expenses. After the drug is approved, there are regulatory requirements such as pharmacokinetic studies in children and special adult populations (e.g., overweight or obese patients), routine pharmacovigilance and postmarket surveillance, susceptibility testing for diagnostic devices (discussed in the next section), and manufacturing (Krause, 2021a). As with most pharmaceuticals, manufacturing antimicrobials is complicated. Sourcing raw materials for them can take 8 months and manufacturing another 14 months, so some manufacturing expense is incurred more than a year before the drug is sold (Krause, 2021a).
Against all these expenses is the reality that new antimicrobials will not sell well. There are public health reasons to use them sparingly and even in the absence of a public health reason, the drugs have to compete on price with older, cheaper medicines, often of comparable clinical value. Furthermore, regardless of the drug’s activity in vitro against various pathogens, its use is largely, practically limited to those indications the regulatory agency approved based on clinical trial data. Finding trial participants with suspected resistant infections is challenging in the United States, where these infections are relatively rare, a topic discussed later in the chapter. Box 6-1 discusses how the limited approved indications for the drug plazomicin contributed to its commercial failure.
The bankruptcy of Achaogen described in Box 6-1 and those of several other similar small antibiotic developers led to widespread calls of a market failure for antimicrobials (Daniel et al., 2013; Gotham et al., 2021; Jacobs, 2019; Jit et al., 2020; O’Brien and Chu, 2020). Others have argued that, strictly speaking, this is not a market failure because,
drugs with limited clinical benefit over existing treatments (which plazomicin was for urinary tract infections in the United States) provide smaller financial return. As such, Achaogen’s bankruptcy is not necessarily a good example of a broken market for antibiotics, nor should the company’s collapse serve as a justification to pressure governments to establish large-scale pull incentives for the multinational pharmaceutical industry. (Aagaard et al., 2021)
___________________
1 After accounting for the cost of failures and the cost of capital.
Furthermore, although Achaogen’s bankruptcy might deter other firms from entering the market, plazomicin is still available to patients. The key public health goal of making a new antimicrobial medicine available was met.
It is possible that while the global market for antimicrobials has failed, national markets in the high-income countries responsible for most new drug development are, ironically, performing. At the same time, the U.S. government, like other governments and international organizations, recognizes that, market failure or not, there is a serious mismatch in the
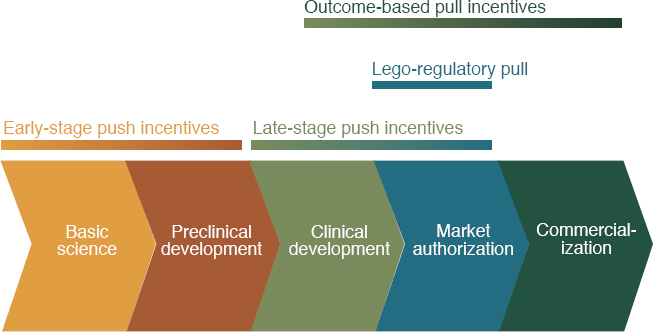
need for new antimicrobials and the willingness of industry to invest in them. To this end, the government encourages antimicrobial development with assistance to reduce the cost of research and development (i.e., push incentive) and to transition the products to market and sustain them (i.e., pull incentives) (Simpkin et al., 2017). Figure 6-1 shows how these various incentives work on the drug development timeline, showing also where push and pull incentives can overlap.
Because the different types of incentive programs work at different stages of the development timeline, some quite distal, it is difficult to estimate the effectiveness, let alone the relative cost-effectiveness, of any one incentive program. Furthermore, the different incentives tend to work together as bundles of initiatives. The committee recognizes that the success of an incentive program is partly predetermined by earlier success of different programs or incentives. With this in mind, the next sections review the ways different push and pull incentives contribute to the antimicrobial drug pipeline. This is not an exhaustive discussion of the hundreds of incentive programs working around the world to stimulate the antimicrobials market and is heavily, though not exclusively, weighted to programs in the United States.
Push Incentives
Push incentives work early in the drug development timeline. They aim to reduce the costs of research and development to any one entity by spreading these costs across a range of interested parties (Renwick et
SOURCE: Renwick, M., and E. Mossialos, Fostering R&D of novel antibiotics and other technologies to prevent and treat infection, The European Journal of Public Health, 2020, Vol. 30, Supplement 5, by permission of Oxford University Press.
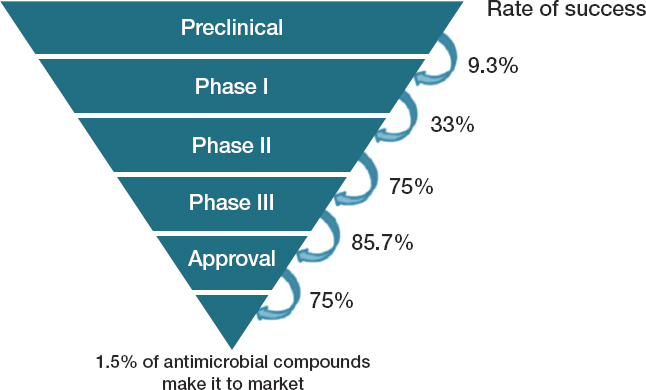
al., 2016). As Figure 6-2 shows, these early costs are high and the risk of failure is great. As a product moves into later development stages the risk of failure declines (see Figure 6-3). This high-risk, preclinical research is funded almost entirely by taxpayers (Aagaard et al., 2021).
SOURCES: Aagaard et al., 2021 ReAct Report: Ensuring sustainable access to effective antibiotics for everyone everywhere. Figure based on TLV report.
SOURCE: OECD et al., 2017.
The goal of push incentives is to make drug development more attractive to firms by minimizing early costs, encouraging cooperation of a diverse pool of experts from academia and research institutes, nongovernmental organizations, and private industry. Push incentives work through three main pathways: increasing access to research by putting more tools and information in the public domain, investing in scientific training needed for antimicrobial development, and direct research funding (Mossialos et al., 2010; Renwick et al., 2016).
The first two of these strategies are not specific to antimicrobial development, but are sometimes forgotten in considering the mix of government tools encouraging new antimicrobials. For example, the Community for Open Antimicrobial Drug Discovery, launched in 2015, uses high-throughput screening to test drug compounds for antimicrobial activity (CO-ADD; Desselle et al., 2017).2 The service is free and aims to include academic chemists in the drug discovery process. The CDC and the Food and Drug Administration (FDA) Antibiotic Resistance Isolate Bank also works to increase access to research by making resistant organisms available to researchers; the National Database of Antibiotic-Resistant Organisms discussed in Chapter 4 works at a similar, early stage in the pipeline through increasing access to standardized data on resistance genes, bacterial genomes, and antibiotic susceptibility (CDC, 2020a; NLM, 2021). The Pew Charitable Trust’s Shared Platform for Antibiotic Research and Knowledge works at a similar point in drug discovery, providing a consolidated, publicly available database of results (some previously unpublished) and insights garnered from scientists studying gram-negative bacteria (Pew, 2021; Thomas et al., 2018).
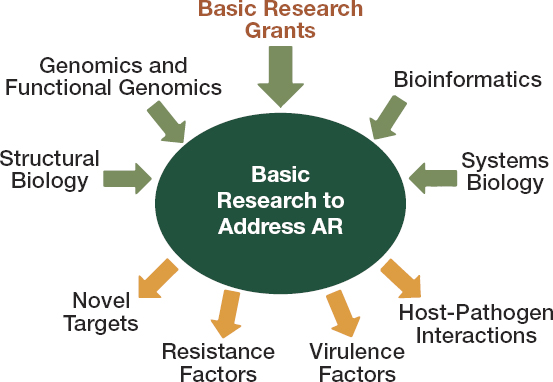
Grant funding for basic and applied scientific research is part of the earliest phase of push incentives (Årdal et al., 2018). Grants for training young scientists and other support to academia and research institutes would all fall into this category of early-stage push incentive (Ardal et al., 2020). Figure 6-4 shows how the NIH’s National Institute of Allergy and Infectious Diseases (NIAID) extensive portfolio of basic science research informs understanding of host–pathogen interactions, virulence, resistance mechanisms, and novel drug targets, for example.
While it is not always counted as part of the investment in developing medical products, this early-stage research can be some of the most helpful in the long run. Resistance can emerge rapidly and in unpredictable ways. The multidrug-resistant fungal pathogen Candida auris, for example, was unheard of before 2009 (CDC, 2019b). In a span of just a
___________________
2 High-throughput screening is a drug discovery tool that relies on robotics and advanced computing to test a sample for a pathway or activity against an organism in millions of combinations in a short time (ScienceDirect, 2021b).
SOURCE: Knisely, 2020.
few years, it has become a CDC urgent threat, with cases increasing over 300 percent between 2015 and 2018 (CDC, 2019a; Chiller, 2017). Though information about this pathogen is limited, there is reason to suspect a case fatality rate of 30 to 60 percent for patients with invasive infection (CDC, 2019b). C. auris is a valuable reminder that it will be difficult to predict what pathogen will be of greatest public health threat even 5 or 10 years in the future. The strong, adaptable research base described in Figure 6-4 is an investment in the response to future threats.
Preclinical and Early Clinical Development
There are also many push incentives providing direct funding for preclinical research and early clinical trials, sometimes called the midstage of antimicrobial development (Årdal et al., 2018). A recent review found that, as of mid-2019, there were 314 institutions around the world active in discovery and preclinical development of antibacterials, supporting a combined 407 projects (Theuretzbacher et al., 2020). Most institutions working at these stages (81 percent) are small and medium-sized firms in North America and Europe (Theuretzbacher et al., 2020). As Figure 6-5 shows, this research is heavily weighted to direct-acting, small-molecule antibiotics.
In the United States, the NIH and the Biomedical Advanced Research and Development Authority (BARDA) provide multiple grants and awards to support research and development into new antimicrobials (GAO, 2020;
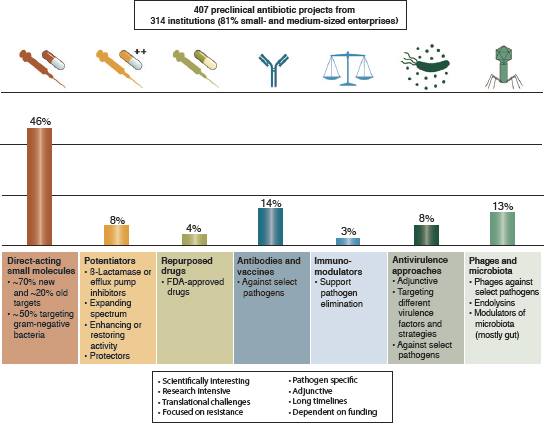
SOURCE: Reprinted by permission from Springer Nature: Springer Nature, Nature Reviews Microbiology, The global preclinical antibacterial pipeline, Theuretzbacher et al., COPYRIGHT 2019.
Simpkin et al., 2017). The Joint Programming Initiative on Antimicrobial Resistance provides similar research funding in Europe (JPIAMR, n.d.). Private foundations, including the Wellcome Trust and the Bill & Melinda Gates Foundation, also support development of new antimicrobials, either individually, or through public–private partnerships such as the Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator (CARB-X), (CARB-X, 2021b; Knox, 2020). Many of these programs work at the preclinical stage and earlier, though the lines are not always clear. NIH grants can provide funding up to the point of regulatory approval, and BARDA’s activity is more concentrated in middle- and late-stage clinical trials (Årdal et al., 2018; Simpkin et al., 2017). The nonprofit Global Antibiotic Research and Development Partnership (GARDP), described in Box 6-2, works across all phases of drug development, though most heavily in clinical development and postmarket stages (Balasegaram, 2021).
Combating Antibiotic Resistant Bacteria Biopharmaceutical Accelerator
One of BARDA’s main contributions to the research and development of new antimicrobials, as well as other therapeutics, diagnostics, and pre-
ventive products, was the 2016 creation of the nonprofit CARB-X (2021a) partnership. Other CARB-X contributors, either financially or in kind, include the Wellcome Trust, the German government (via the Federal Ministry for Education and Research), the Bill & Melinda Gates Founda-
tion, the British government’s Global Antimicrobial Resistance Innovation Fund, and NIAID (CARB-X, 2021a). The program aims to speed the development of a range of antimicrobial products providing product developers with technical support and nondilutive funding (meaning the funding is added to other revenue streams but is not contingent on the owner selling a piece of the company) (CARB-X, 2020). CARB-X (2020) works mainly in preclinical development and early clinical trials for WHO and CDC priority pathogens.3 With $500 million of funding for its first 5 years, CARB-X is the world’s largest early development investor in new antimicrobials and related products (Alm and Gallant, 2020).
Figure 6-6 shows how, across its three funding cycles to date, CARB-X has invested heavily in the riskiest stages of product development. For therapeutic and preventive products this includes the process of refining an active compound (i.e., a hit) to a chemical prototype (i.e., a lead) and establishing the compound’s in vitro activity as well as other important chemical properties such as solubility, stability, and permeability through structure-activity relationship studies (lead optimization), steps that occur before preclinical development (Bleicher et al., 2003). (To give a sense of
SOURCE: Alm and Gallant, 2020. https://pubs.acs.org/doi/10.1021/acsinfecdis.0c00026. Further permissions related to this material should be directed to ACS Publications.
___________________
3 CDC urgent threat pathogens are Clostridioides difficile, carbapenem-resistant Acinetobacter, carbapenem-resistant Enterobacterales, Candida auris, and drug-resistant Neisseria gonorrhoeae (CDC, 2019a). The WHO critical threat pathogens are carbapenem-resistant Acinetobacter baumannii, carbapenem-resistant Pseudomonas aeruginosa, and carbapenem-resistant extended spectrum beta-lactamase-producing Enterobacterales (OECD et al., 2017).
the CARB-X scope of work, Figure 6-6 also shows investments across the analogous stages in diagnostics development.)
CARB-X funds product development all over the world, but most of its funding goes to small firms in Europe and North America (Alm and Gallant, 2020; CARB-X, 2020). As a condition of receiving funding, product developers are required to develop plans for stewardships and access to their products within 3 months of entering phase 3 trials (CARB-X, 2021e). At this point, however, most products would no longer be part of the CARB-X portfolio, but possibly handed off to BARDA for phase 2 and 3 clinical trials (Singer et al., 2020).
Mid- and Late-Stage Clinical Development
While preclinical stages of antimicrobial research are the most financially risky, later-stage trials have increasing technical and regulatory demands (Ardal et al., 2020; Ventola, 2015). Even effective drugs can sometimes fail to demonstrate their value at these stages because of problems with trial design, its cited endpoints, or a lack of statistical power (Fogel, 2018). As projects advance from the preclinical and early clinical trials stages, different funding and technical support may be needed. In the United States, BARDA funding supports this stage of research and development (Buckmon, 2020). Through partnership with various private companies, BARDA supports the largest portfolio of antibacterial drug development in the world (Buckmon, 2020).
BARDA’s Project BioShield and the Broad Spectrum Antimicrobials Program
Project BioShield Act was signed into law in 2004 with the objective of accelerating the research, development, acquisition, and availability of medical countermeasures, those medical tools that the government would need to respond to biological or chemical weapons (HHS, 2019; Russell, 2007). The act created a special reserve fund from which the secretary of health can, with presidential approval, access up to $5.6 billion over 10 years to develop and procure medical countermeasures for which the government is the main market (MedicalCountermeasures.gov, 2019b; Coller, 2006). Through its provisions for emergency use, it allows FDA to give temporary authorization to unapproved medicines or unapproved use of approved ones (ASTHO, 2021). BARDA manages the development and purchasing of countermeasure products under Project BioShield (Houchens and Larsen, 2017). To this end, BARDA provides advanced research and development contracts as well as market commitments to qualified pharmaceutical and biotechnology companies (Larsen and Disbrow, 2017). These contacts are intended to reduce the risk of developing products and improve their return on investment (Larsen and Disbrow, 2017).
In 2010, BARDA established the Broad Spectrum Antimicrobials Program to encourage research and development of novel antimicrobial drugs with broad-spectrum activity against pathogens that threaten national or global security (MedicalCountermeasures.gov, 2019a). The program funding, like CARB-X funding, is nondilutive (Albrecht and Swenson, 2020). Should the product gain FDA approval the company would be free to sell it on the commercial market (Albrecht and Swenson, 2020).
Initially, the Broad Spectrum Antimicrobials Program had to employ a so-called dual utility approach, meaning that drug candidates had to treat a clinically prevalent infection and be useful against one or more of the biodefense threats listed in the Department of Homeland Security (DHS) material threat list (Billington, 2015; Eichberg, 2015). The DHS material threat list did not, however, overlap with the CDC list of antimicrobial resistance threats (Billington, 2015; Eichberg, 2015) (see Table 6-1). The 2014 executive order Combating Antibiotic Resistant Bacteria gave BARDA the ability to target CDC priority pathogens (Billington, 2015). Further, the first National Action Plan for Combating Antibiotic-Resistant Bacteria, released in March 2015, called for BARDA to partner with at least one drug or biotechnology company to speed the development of antibacterial medicines (Billington, 2015; PCAST, 2015). By 2018 the agency was to have 12 candidate
TABLE 6-1
Comparison of CDC and BSA Priority Bacterial Threats
| CDC Urgent or Serious Antibiotic Resistance Threats | DHS High-Priority Bacterial Threats |
|---|---|
| Clostridioides difficile | Burkholderia mallei (glanders) |
| Carbapenem-resistant Enterobacterales | Burkholderia pseudomallei (melioidosis) |
| Drug-resistant Neisseria gonorrhoeae | Francisella tularensis (tularemia) |
| Multidrug-resistant Acinetobacter | Rickettsia prowazekii (typhus) |
| Drug-resistant Campylobacter | Yersinia pestis (plague) |
| Fluconazole-resistant Candida (a fungus) | Bacillus anthracis (anthrax) |
| Extended spectrum ß-lactamaseproducing Enterobacterales (ESBLs) | |
| Vancomycin-resistant Enterococcus (VRE) | |
| Multidrug-resistant Pseudomonas aeruginosa | |
| Drug-resistant nontyphoidal Salmonella | |
| Drug-resistant Salmonella Typhi | |
| Drug-resistant Shigella | |
| Methicillin-resistant Staphylococcus aureus (MRSA) | |
| Drug-resistant Streptococcus pneumoniae | |
| Drug-resistant tuberculosis |
SOURCE: Adapted from Billington, 2015. The publisher for this copyrighted material is Mary Ann Liebert, Inc. publishers.
antibiotics in development, with at least two of these products submitted for FDA approval by 2020 (Billington, 2015; PCAST, 2015).
Figure 6-7 presents BARDA’s antimicrobial drug candidate portfolio as of fall 2021. Along with supporting development of medicines for priority biothreats, such as plague and anthrax, the agency supports the development of treatment for CDC priority pathogens (Kadlec, 2019). Four of its antimicrobial candidates received FDA approval (Albrecht,
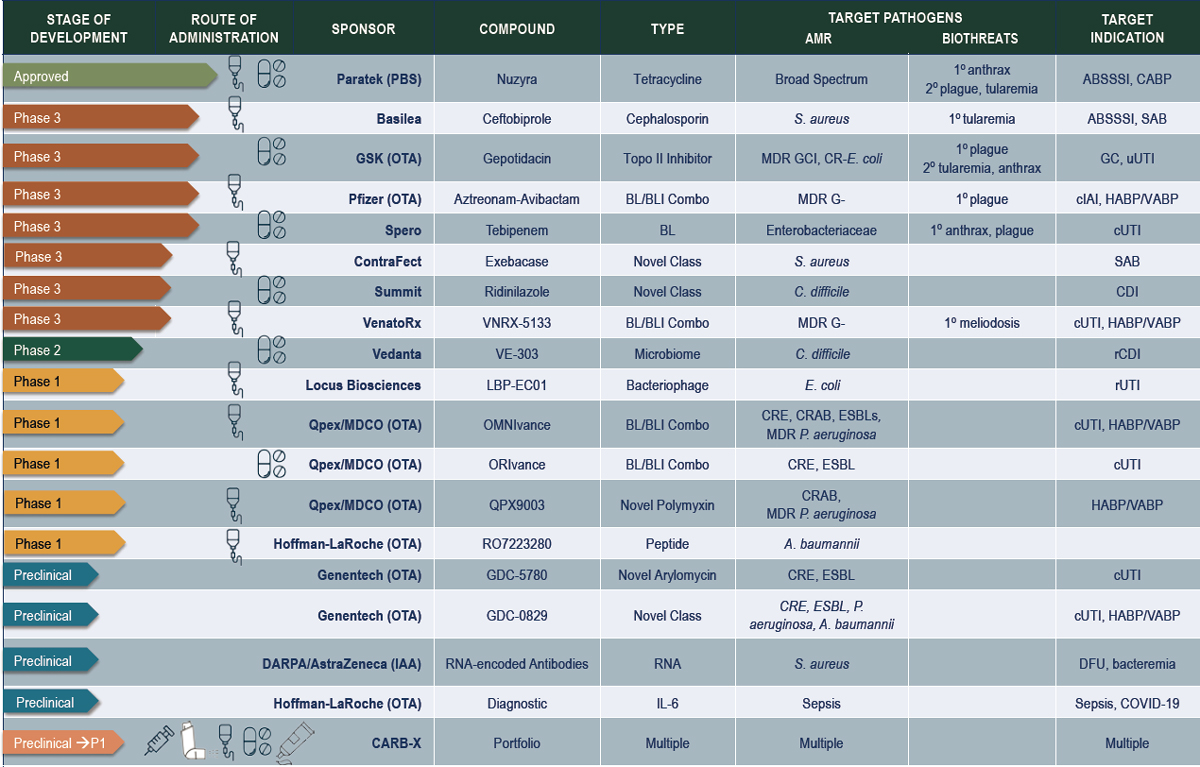
Figure legend:

NOTE: ABSSSI = acute bacterial skin and skin structure infections; BL = beta-lactam; BL/BLI = beta-lactam/beta-lactamase inhibitor; CABP = community-acquired bacterial pneumonia; CDI = Clostridioides difficile infection; cIAI = complicated intra-abdominal infection; CRAB = carbapenem-resistant Acinetobacter baumannii; CRE = carbapenem-resistant Enterobacterales; cUTI = complicated urinary tract infection; DFUI = diabetic foot ulcer infections; ESBL = extended spectrum beta-lactamase; GCI = gonococcal infection; HABP/VABP = hospital-associated bacterial pneumonia/ventilator-associated bacterial pneumonia; MDR = multidrug resistant; MDR G– = multidrug-resistant gram-negative bacteria; rCDI = recurrent Clostridioides difficile infection; rUTI = recurrent urinary tract infection; SAB = Staphylococcus aureus bacteremia; uUTI = uncomplicated urinary tract infection.
SOURCE: Images were provided courtesy of the Biomedical Advanced Research and Development Authority (BARDA), a division within the Assistant Secretary for Preparedness and Response of the Department of Health and Human Services.
2020; Kadlec, 2019).4 As Figure 6-7 shows, another seven are in phase 3 clinical trials (Kadlec, 2019).
Broad Spectrum Antibacterial program contracts end when the sponsoring company receives new drug approval from FDA (Larsen and Disbrow, 2017). While other types of BARDA contracts include advance market commitments, there are no such provisions for broad-spectrum antimicrobials (Kadlec, 2019). BARDA can, however, arrange advance market commitments for qualified products needed for the national stockpile (Albrecht, 2018).
Increasing Attention to Push Funding
While push incentives are helpful and necessary for antimicrobial development, they are not tied to results, so there is less of a direct relationship between the incentive and its intended goals (Dutescu and Hillier, 2021). For example, early-stage research funding cannot be readily contingent on ensuring access to new medicines or antimicrobial stewardship. Individual companies and scientists benefit from push incentives, but they are also seen to have broad, indirect value to society. These indirect benefits and the relatively straightforward implementation make push incentives attractive tools for governments and other funders (Dutescu and Hillier, 2021). For these reasons, government and private incentive programs may be proportionately overinvested in early-stage and preclinical research (Simpkin et al., 2017). At the same time, the high-risk early stages may be the most appropriate place for public spending on drug development as it is the riskiest. Especially with antimicrobial medicines, the later stages of drug development have far less risk of failure.
In 2017 the Organisation for Economic Co-operation and Development (OECD) estimated that governments spent over $546 million a year on push funding for antimicrobial research and development (OECD et al., 2017). At the time, this amounted to 64 percent of research and development funding and 95 percent of the total incentive funding for antimicrobial development (OECD et al., 2017). The mix of incentives for antimicrobial development has changed more recently. There may be relatively less venture capital available today for small- and medium-sized biotechnology firms (Dall, 2020; Nielsen et al., 2019). On the other hand, growing international attention to the problem has brought new funders to early stage development. The Novo Nordisk Foundation’s REPAIR Impact Fund, for example, has a $165 million budget to invest in about 20 different therapies for antimicrobial resistance (Novo Nordisk Foundation, 2018).
___________________
4 Vabomere® (a combination of meropenem and vaborbactam) by Melinta Therapeutics, Zemdri® (plazomicin by Achaogen [now Cipla]), Xerava® (eravacycline) by Tetraphase Pharmaceuticals, and Nuzyra® (omadacycline by Paratek Pharmaceuticals) (Albrecht, 2020).
The AMR Action Fund, announced in 2020, will supply about $1 billion in funding, as well as technical support, from a group of innovator pharmaceutical companies via their trade association to small biotechnology companies developing treatments for CDC or WHO priority pathogens (AMR Action Fund, 2021). This push funding will be available “across all stages of clinical development” (AMR Action Fund, 2020, 2021). Though financed mainly by pharmaceutical companies, private foundations are also involved, and the fund’s groundwork was set by the European Investment Bank, the Wellcome Trust, and the WHO (Beyer et al., 2020).
There is some imprecision in estimating the effects of push incentives, partly because some early-stage incentives work so broadly that they could not be reliably counted against any research and development funding total (Årdal et al., 2018). While push funding will be invaluable to maintaining a strong pipeline for antimicrobials and other products needed to fight resistance, there is growing consensus that they need to be paired with pull and hybrid incentives to maximize their usefulness and to compensate for the lack of a vigorous market for new antimicrobials (Årdal et al., 2018; Dutescu and Hillier, 2021; WHO, 2021).
Pull Incentives
While push incentives are geared toward reducing the cost of research and development, pull incentives are designed to facilitate higher market returns for product developers (Bhavnani et al., 2020; Renwick et al., 2016). Pull incentives can be divided into two categories: ones that provide direct monetary reward, sometimes called outcome-based incentives, or ones that act through legal and regulatory channels to indirectly increase a company’s returns (called lego-regulatory incentives) (Renwick and Mossialos, 2020). Outcome-based pull incentives include lump-sum payments and cash rewards for sales and regulatory milestones. Advanced commitment to buy a certain amount of a drug or to license the patent for a set sum are also considered outcome-based pull incentives. Box 6-3 describes one such advance commitment program recently introduced in Congress.
In contrast, legal and regulatory pulls are designed to increase financial returns indirectly using strategies such as extension on market exclusivity or accelerated regulatory review (thereby reducing the time to bring a drug to market). By only rewarding successful development, both types of pull incentives aim to shift the risk associated with bringing a medicine to market from the developer to the payer.
France and Germany have instituted a number of legal and regulatory pull incentives that allow for more flexible pricing and accelerate the regulatory review process for antimicrobial drugs. Both programs work through attention to list price and sales incentives, though the French one puts more emphasis on rewarding added therapeutic value (Gotham et
al., 2021). These incentives have been in effect since 2015 in France and 2017 in Germany (Gotham et al., 2021).5 Formal outcome evaluations for these programs are not yet available, but it is reasonable to expect that they have increased company revenues from sales of their qualified antimicrobial drugs in those markets. The United States also uses several legal and regulatory pull incentives, Box 6-4 describes some of the more common ones employed as of midyear 2021. Box 6-4 does not include some outcome-based pull incentives such as the purchase of a new antimicrobial for the national stockpile or an advanced purchase commitment. Since new antimicrobials have relatively small patient populations, it is difficult to envision such purchases being of sufficient volume to be a meaningful incentive.
___________________
5 There is an additional law that was passed in 2020 in Germany that exempts certain antimicrobials from the health technology assessment process used for making reimbursement decisions. The exemption is similar to that granted to orphan drugs, meaning those drugs aimed to treat rare diseases and conditions (Gotham et al., 2021).
There are also two bills pending in Congress that include additional legal and regulatory incentives for antimicrobial manufacturers. First is the 2018 Re-Valuing Anti-Microbial Products Act (REVAMP). This act amends the Food, Drug, and Cosmetic Act to enable developers of priority antimicrobial medicines designated as “qualified infectious disease products” to receive transferable extensions on market exclusivity for up to a year.6 The act’s provisions include a committee of FDA, CDC, and BARDA representatives as well as other experts from medicine, public health, economics, and related fields of research.7 The fact that Congress has taken no action on REVAMP since 2018 suggests it is not a priority and may not be revisited.
The 2019 Congress saw proposed changes to Medicare’s system of
___________________
6 The authorizing legislation of the modern FDA (2018a).
7 REVAMP Act, HR 6294, 115th Cong., 2nd sess., Congressional Record 164, no. 109, daily ed. (June 28, 2018): H 5977.
bundled payments, the paying of hospitals or other providers for multiple, related services for a predetermined flat fee (Hardin et al., 2017). Bundled payments are meant to make medicine more efficient and remove the incentive to overtreat inherent in a fee-for-service system (Hardin et al., 2017). One of the earliest forms of bundled payment, in places since the 1980s, are “fixed payments for inpatient services associated with specific diagnoses and procedures” or Diagnosis Related Groups (Cortese et al., 2018). By paying a flat fee for related services and medicines, the rule creates a disincentive for hospitals to use newer, expensive antimicrobials and related diagnostics (Gotham et al., 2021). Revisions to the Developing an Innovative Strategy for Antimicrobial Resistant Microorganisms (DISARM) Act would allow the Centers for Medicare & Medicaid Services (CMS) to reimburse antimicrobials qualified by FDA separately, in addition to the bundled payments determined by diagnosis.8 Supporters of this change point to the limited patient pool and short treatment duration for novel antimicrobials. By reimbursing more for these medicines in hospitals, they maintain, CMS would improve outcomes for patients with resistant infections, slow the emergence of resistance, and contribute to the market viability of companies making these drugs (Coukell, 2019; IDSA, 2019; Segerman, 2019).
At the same time, research has shown that, however well-intentioned, recent pull incentives have not brought about the changes intended. To start, eligibility as a qualified infectious disease product, a determination on which many of the proposed incentives hinge, appear to be overly broad (Darrow and Kesselheim, 2020). Many serious, life-threatening infections already have good treatments on the market, yet the qualification process “disproportionately rewards modifications to existing drugs rather than the creation of novel drugs” (Darrow and Kesselheim, 2020). To put it another way, new antimicrobials that do not necessarily satisfy an unmet need can qualify for fast-track approval and extended exclusivity protections afforded as a qualified infectious disease product.
Ironically, uniform extensions on market exclusivity are most valuable to the least innovative products. At the heart of the questions is the time value of money, the idea that money is worth more in the present than the same amount in the future because of forgone investments it could have been used for. In deciding whether to invest in a given drug development project, an investor calculates the net present value of that project in which future revenues are discounted by the cost of capital. (Due to this discounting, revenues in out-years are worth less than those realized in the shorter term.) Therefore, as Figure 6-8 shows, a 5-year extension to exclusivity is worth more than half the net present value of a
___________________
8 DISARM Act of 2019, S 1712, 116th Cong., 1st sess. (June 4, 2018).
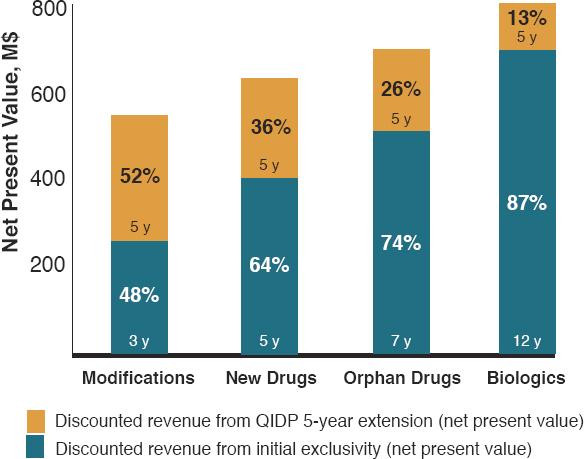
NOTE: Biologics are not eligible for exclusivity under the act and are included for illustrative purposes only.
SOURCE: Darrow, Jonathan J; Kesselheim, Aaron S, Incentivizing Antibiotic Development: Why Isn’t the Generating Antibiotic Incentives Now (GAIN) Act Working?, Open Forum Infectious Diseases, 2020, Vol 7, Issue 1, by permission of Oxford University Press.
drug’s revenue for modification to an existing drug, something otherwise determined to warrant a 3-year period of market exclusivity, but only about a quarter of the present value for an orphan drug product guaranteed 7-year market exclusivity. Baseline differences in market exclusivity reflect the relative value different new medical products bring society. Blanket extension on market exclusivity undermines that calculation, disproportionately rewarding the least valuable products (Darrow and Kesselheim, 2020).
CMS’s attempts to increase payment for novel antimicrobials have also met with roadblocks. To qualify for New Technology Add-On Payments staff in the hospital pharmacy have to apply to CMS for reimbursement, expending significant, unbillable time and effort on the application (Bhavnani et al., 2020). To complicate the matter, although the expense of the new medicine and the staff time to file for an add-on payment are incurred at the pharmacy level, CMS reimburses the highest organizational level, the hospital (Bhavnani et al., 2020). This lump-sum pay-
ment is not broken out with details about what technologies are being reimbursed, information that could at least give the hospital executives a sense of which divisions’ work had contributed to the reimbursement (Bhavnani et al., 2020). Furthermore, even after reimbursement of 75 percent of a new antimicrobial’s launch price (what the CMS New Technology Add-On Payment would allow) the new drugs are still considerably more expensive than generics (Bhavnani et al., 2020).
Nevertheless, the add-on payments do offer hospitals additional payment for expensive, new antimicrobials. While there are problems with the tracking of these payments and with the administrative burden they put on staff, especially on pharmacy staff, these are not, strictly speaking, problems with the incentive, but rather with the way it is managed.
It is difficult to gauge the effect of the 2016 Limited Population Pathway for Antibacterial and Antifungal drugs on stimulating the market, as only two drugs have been approved under it to date (FDA, 2020d). One treats lung disease caused by Mycobacterium avium complex, the other a type of highly resistant tuberculosis (FDA, 2020d). Unlike the other pull incentives described in Box 6-4, this pathway does not hinge on qualification as an infectious disease product but on FDA’s judgment that the medicine will affect “such factors as survival, day-to-day functioning, or the likelihood that the condition, if left untreated, will progress from a less severe condition to a more serious one” (FDA, 2020d).
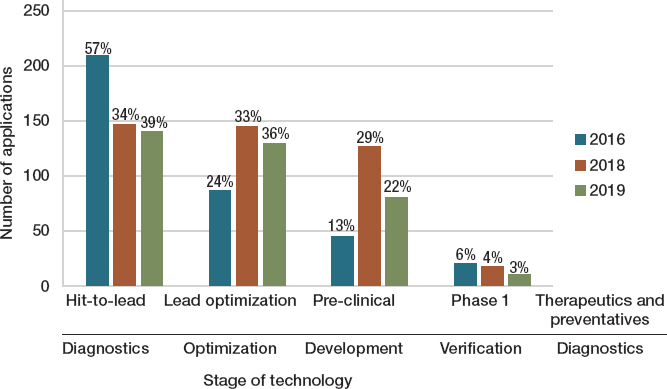
The Strength of the Pipeline
Overall, the push and pull incentives in effect modestly improved the number of products in the antimicrobial drug pipeline. After a drop in the 1990s and early 2000s, the number of new antimicrobials FDA approves every year has risen recently (Spellberg, 2021; Spellberg et al., 2013). In a 2019 paper, Cunha and colleagues (2019) estimated that the number of drug candidates in the pipeline has increased more than 10 percent between 2014 and 2019 (see Figure 6-9). Moreover, during the same period, FDA approved a total of 20 new antimicrobial drugs, 17 of which had activity against the so-called ESKAPE pathogens, pathogens designated as urgent threats by the CDC or the WHO.9 Of these 17 new antimicrobials, 12 qualified as infectious disease products, thereby
___________________
9 ESKAPE pathogens include Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species (CDC, 2019a).
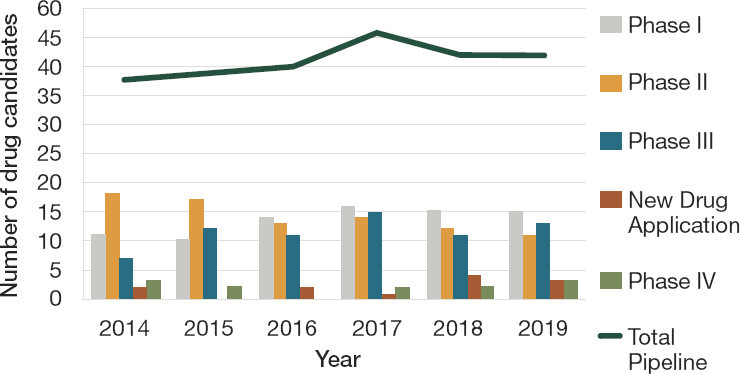
SOURCE: Cunha et al., 2019.
earning priority regulatory review and extended exclusivity protections (Berger et al., 2021; FDA, 2021c).10
However, these figures only tell a partial story about the antimicrobial drug pipeline. Of the 43 new antimicrobials currently in clinical development, only 10 are of a novel class or target (Pew, 2014). Of the 15 new antimicrobials in phase 3 trials or granted new drug approval in the last quarter of 2020, only 4 had expected activity against CDC urgent threats or WHO critical, priority pathogens (Pew, 2014). In short, the majority of the pipeline drugs are not very different from existing antimicrobial medicines, nor do they have activity against those pathogens that are the most worrisome, mainly multidrug-resistant, gram-negative bacteria (WHO, 2019b). Only 6 of the 50 antibiotics currently in the pipeline meet even one WHO criteria for being innovative,11 only two target multidrug-resistant, gram-negative pathogens (WHO, 2019a).
Further, most of the recently approved drugs appear to offer little to no added clinical value over existing treatments (Schulz et al., 2019; WHO, 2019b). A 2019 WHO review commented on “a visible mismatch between the few newly approved antibiotics and the WHO priority pathogen list” (WHO, 2019a). The review concluded that overall the newly approved
___________________
10 FDA aims to take action within 6 months on an accepted new drug application that is designated as Priority Review rather than the 10 months under standard review (OECD et al., 2017).
11 These criteria are no known cross-resistance with existing medicines, a new drug class, a new target, or a new mode of action (WHO, 2019b).
products’ “lack of differentiation against existing treatments, their non-inclusion in clinical guidelines, and their higher prices in comparison to existing generic treatments make it difficult to predict their place in the treatment landscape” (WHO, 2019a). While the existing incentives have helped revitalize research and development in antimicrobials and increased the number of antimicrobial drugs in the pipeline and on the market, they have not succeeded in bringing to market innovative new antimicrobial drugs for serious and life-threatening infections caused by pathogens of concern.
It is difficult to say how many drug candidates or other products in development constitutes a strong pipeline. As Figure 6-9 showed, many candidates fall away in clinical trials before reaching the New Drug Application phase. Market entry rewards that fully or partially delink product revenues from quantity sold are one promising strategy. Such additional incentives could improve the expected net present value of antimicrobial drug projects compared to other therapeutic areas and entice large pharmaceutical companies to reenter the antimicrobial market, goals that existing mechanisms have not yet achieved (Daniel et al., 2018; PACCARB, 2019; WHO, 2019b).
Participation of large, multinational companies in antibiotic development would also help ensure the viability of new antimicrobials. These companies are immensely profitable, with cumulative profits of over $8 trillion between 2000 and 2018 (Ledley et al., 2020). Their diverse product lines generate sufficient revenue to offset the manufacturing and postmarket expenses associated with new antimicrobials during the roughly 2 decades before the drugs become profitable (McKenna, 2020). But the same economic factors that drive small antimicrobial manufacturers to bankruptcy apply to large pharmaceutical companies as well. Publicly traded companies are not supposed to lose money on purpose, even if they have a lot of money to cover the losses. This is why 15 of the 18 largest pharmaceutical companies have quit antimicrobial development in the last 20 years (Council of Canadian Academies, 2019; Talbot et al., 2019). In their place are small and medium-sized biotechnology firms, which account for over 95 percent of the antimicrobials in development today (The Pew Charitable Trusts, 2020b). These companies do not have the capital reserves to withstand the time between launch and profitability, nor do they have comparable infrastructure, laboratories, or depth of staffing (Talbot et al., 2019; The Pew Charitable Trusts, 2020b; WHO, 2019b).
Optimal Incentives for Antimicrobial Development
The optimal incentives for antibiotic development would improve the drug’s net present value and facilitate cooperation of both large pharmaceutical companies and small biotechnology firms (Renwick et al., 2016). Market entry rewards are one promising tool to this end. A recent systematic review found market entry rewards to be the most frequently suggested incentive for antimicrobial development (Dutescu and Hillier, 2021). There is, however, no consensus on the appropriate size of the reward, eligibility criteria, or the implementation method.
It is also difficult to judge the success of some incentives discussed in this chapter simply because they are so new. For example, the widely criticized 2019 revisions to the Inpatient Prospective Payment System may, in some ways, be seen as a failure (Outterson, 2019; The Pew Charitable Trusts, 2020a). At the same time, most impact evaluations for economic programs give longer than a year between program implementation and evaluation. Hospital administrations can move slowly, but it may be rash to conclude that they will not move at all to adjust to a new incentive.
The Amount of Market Entry Reward
The Infectious Diseases Society of America (IDSA) has proposed a reward of “at least $500 million” for new antibiotics that address unmet needs (Talbot et al., 2019). The O’Neill report and the European public–private partnership Drive-AB have suggested that a market entry payment of $1 billion for each new antibiotic approved may be appropriate (Årdal et al., 2018; O’Neill, 2016). After the British government announced a plan to pay over $140 million for each new antibiotic approved, proposals for a similar, proportionate payment from G20 countries led to calls for a pooled payment of $4 billion for each new drug (Mullard, 2020; Rex and Outterson, 2020). Given the United States accounts for almost half of global pharmaceutical sales, it is safe to conclude that the U.S. share of a $4 billion payment would be close to $2 billion per drug (Mikulic, 2021). In its 2017 recommendations on this question, the President’s Advisory Council on Combating Antibiotic-Resistant Bacteria recommended the use of market entry rewards, pointing to recent analyses suggesting $1 to $2 billion or more may be needed (HHS, 2017). This represents a significant outlay of taxpayers’ money. To put it in perspective, $1 billion is comparable to FDA’s entire 2020 budget for food safety; the CDC’s total budget request for fiscal year 2020 was $6.6 billion (CDC, 2020b; FDA, 2020c).
The amount of the reward should be sufficiently large to entice entry but not so large that it results in what economists describe as rent seek-
ing, the practice of companies asking the government for financial protections not proportionate to their value (Henderson, 2019). The reward amounts cited in the literature are often based on economic models with high parameter uncertainty. The O’Neill report’s $1 billion figure, for example, was based on a “broad estimate” of a $40 billion cost of inaction over 10 years (O’Neill, 2016). This estimate was in turn influenced by the report’s predictions of the future burden of resistance, a prediction of questionable reliability and based on uncertain methods (de Kraker et al., 2016; Harbarth, 2018). Drive-AB’s similar estimate was derived from economic modeling, though with more clearly stated assumptions (Årdal et al., 2018). One such explicit assumption of this model was that large pharmaceutical companies would not enter the market for profitability below a $50 to $500 million threshold (Årdal et al., 2018). This may reflect company expectations based on highly successful product launches.
However, across the pharmaceutical market as a whole, both launch year and peak year sales for most drugs have been on the decline (Berndt et al., 2015). For example, from 2011 through 2015, 64 percent of all new drug launches garnered less than $100 million in annual sales within the first 5 years, and 23 percent earned less than $10 million per year (Aitken and Kleinrock, 2017). The average peak-year sales for new drugs have decreased by more than 50 percent from 2010 through 2018, from $816 million to $316 million per annum (Steedman and Taylor, 2019). Given these trends, it may be more prudent to benchmark the market entry reward to independently reported industry averages, thereby reducing the likelihood of under- or over-incentivizing the market.
Full or Partial Delinking of Sales and Revenues
One of the main questions in offering a market entry reward for novel antimicrobials is the delinking of the drug’s revenues from sales. Originally proposed as a way to encourage development of medicines for neglected diseases, delinking essentially pays the development costs of a new drug up front, rather than gradually through sales (Aagaard et al., 2021). Antimicrobials are well suited to some delinking of revenues, as the best interests of society are served not by selling the drug but by holding it in reserve to use only when needed.
The delinking of sales and revenues is not necessarily an all-or-nothing proposal, however. In some models the market entry reward is paid in addition to sales revenue, meaning that the link between sales and revenues is partially delinked. The Duke-Margolis Center and Drive-AB consortium have both proposed partially delinked rewards, seeing them as more flexible, responsive to unpredictable changes in demand for the drug, more adaptable to different countries’ national reimbursement models,
and easier to pilot (Årdal et al., 2018; Schneider et al., 2020a). The Duke-Margolis model specifically pointed to an impartial antibiotic manager that would adjust payments (in this case annual subscription fees) in response to ebbs and flows in demand, or to reflect its relative value to public health (Schneider et al., 2020a,b). The partially delinked model can also make payments contingent on meeting goals for stewardship or investments in the drug supply chain (Hillock et al., 2020).
Other models advocate for full delinking of drug revenues from sales, citing concerns that, should manufacturers earn any sales revenues, their incentive to oversell remains (Aagaard et al., 2021). The full delinking of sales from revenues also allows for better controls on the drug’s price, assuring its affordability (Aagaard et al., 2021). Full delinkage requires the drug company to refrain from marketing or promoting the drug in any way (Sciarretta et al., 2016). In short, fully delinked reward payments are an alternative to sales revenues; partially delinked payments are a supplement to them (Okhravi et al., 2018).
Especially when applying rewards internationally, partial delinking might seem more fair; high-income countries can and arguably should pay more for medicines than low- or middle-income ones. Some scholars have argued, however, that when the manufacturer’s incentive to sell in lucrative markets remains, it can aggravate inequities in access, giving companies an incentive to concentrate on rich-country markets, regardless of their relative need (Outterson et al., 2016).
Payment Eligibility
Suggested eligibility for a reward payment also varies. Some guidelines emphasize rewarding only novel antimicrobials that target the highest-priority pathogens (WEF, 2018). Others allow additional rewards for products with a novel mechanism of action, but do not make these criteria strict eligibility requirements for the payment (Talbot et al., 2019). A sizable group, including the President’s Advisory Council on Combating Antibiotic-Resistant Bacteria and the Duke-Margolis Center, have advocated for a reward payment benchmarked to objective determination of the drug’s value to public health (Daniel et al., 2019; HHS, 2017).
Despite good consensus that rewarding added clinical value is the best use of a market entry reward, this is something that is difficult to discern. FDA approval indicates that a new drug provides benefits that outweigh its known and potential risks for specific indications. Regulatory approval is not, however, an endorsement of meaningful or added clinical value over existing treatments. This distinction is informed, in part, by late-stage clinical trials, trials that are classified, depending on the regulatory agency, as noninferiority (establishing the new drug is no
worse than old drugs), equivalence (neither better nor worse than existing treatment), or superiority (establishing added clinical value over the old drug) (CPMP, 2000). This classification is based on criteria that are not always clear and are influenced by sample size and statistical power (Dunn et al., 2018). In some situations, companies may go in to the trial intending to establish superiority, but give an a priori margin for noninferiority if that result would be sufficient for licensing (CPMP, 2000).
Superiority in clinical trials is a clear indicator of added clinical value, but such trials are not often feasible for antimicrobial medicines. Since the use of a placebo control would be unethical, only patients suspected to have an infection caused by a pathogen susceptible to both the conventional and test drug are eligible (IDSA, 2012; Rex et al., 2017). There are also logistical challenges to recruiting patients with a specific, resistant pathogen within hours of their presentation for treatment, especially without rapid diagnostics (IDSA, 2012; Rex et al., 2017). These patients, especially those suffering from serious infections with resistant, gram-negative pathogens, may be too mentally or physically deteriorated at intake to give informed consent (IDSA, 2012). Given these constraints on trial design, it is unsurprising that antimicrobial drugs approved by FDA between 2014 and 2019 used noninferiority pivotal trial designs (FDA, 2021c).
Superiority trials cannot replace the current system by which new antimicrobials are evaluated. Such trials are neither feasible nor necessary for new antimicrobial agents against resistant pathogens with limited or no treatment options (Rex et al., 2017). Furthermore, patients can have multiple infections with different pathogens; it is not always clear which one is the primary cause of disease, making it difficult to judge a patient’s suitability for a trial. In other cases, the diagnosis of infection may be based on clinical presentation rather than culture results, leading to a situation where the more seriously ill patients are more likely to be considered for the new treatment, making the treatment’s superiority hard to judge (Stafford et al., 2014).
At the same time, superiority trials can be useful. Additional therapeutic value can be hard to discern in noninferiority trials, which are intended to show that the difference, if any, between the new treatment and its comparator is small (Gotham et al., 2021; HHS et al., 2016). A recent study of antimicrobial prescription guidelines from 70 hospitals in 12 countries and regional standards from seven academic societies found that preferred antimicrobial treatment classes for the same infections varied widely, concluding “the lack of consensus seemed to emanate from a dearth of studies designed to determine superior treatment options, leaving the possibility for standards to vary when interpreting the same
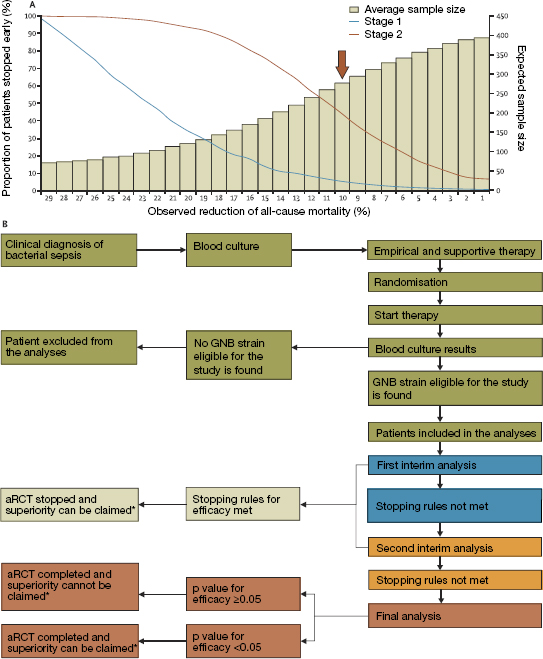
(A) The probability of early stopping of the aRCT (lines) and expected sample size (bars) for observed reduction in all mortality between the control (assumed at 30 percent) and experimental group (variable between 1 percent and 29 percent). Brown arrow represents sample size (n = 278) for the main aRCT assumption, including power of 80 percent, α = 0.05, and efficacy (risk difference) of 10 percent. (B) Different phases of the aRCT, including participant enrollment and selection (blue), first interim analysis (green), second interim analysis (yellow), binding decision on early aRCT termination (gray), and final analysis (red). aRCT-adaptive, postmarketing randomized clinical trial. GNB gram-negative bacteria. * As one or multiple aRCTs are completed, their results can be added to the results of other existing trials in cumulative meta-analyses that provide new, comprehensive views of the developing evidence.
SOURCE: Reprinted from The Lancet Infectious Diseases, Vol. 19, No. 12, Simone Lanini, John P. A. Ioannidis, Francesco Vairo, Michel Pletschette, Gina Portella, Virginia Di Bari, Alessia Mammone, Raffaella Pisapia, Stefano Merler, Boniface Nguhuni, Martin Langer, Antonino Di Caro, Sarah J. L. Edwards, Nicola Petrosillo, and Alimuddin Zumla. Non-inferiority versus superiority trial design for new antibiotics in an era of high antimicrobial resistance: The case for post-marketing, adaptive randomised controlled trials, e-444-e451, Copyright (2019), with permission from Elsevier.
literature base” (Rost et al., 2021). Evidence of clinical superiority could do much to harmonize clinical guidelines.
Emerging approaches, such as data exchange and adaptive clinical trial designs, may enable conduct of superiority trials for antimicrobial drugs (Gatti et al., 2020; Lanini et al., 2019; Paul et al., 2021; Rubin, 2016; Trusheim et al., 2016). An adaptive clinical trial design allows for prospective modifications to the trial design based on the accumulated data (HHS et al., 2019). As Figure 6-10 shows, adaptive randomized controlled trials require fewer participants, largely because stopping criteria are revisited at multiple points; the risk difference between groups calculated in interim analyses can influence the statistical power and needed sample sizes for later stages (Lanini et al., 2019). In other words, study parameters are carefully modified while the adaptive trial is in progress on the basis of a review of interim data (Lanini et al., 2019). Such trials are better indicators of added clinical utility and should not be abandoned during pre- or postapproval evaluations. The amount of any market entry payment should be proportionate to the quality of evidence provided in the clinical data submitted for regulatory approval.
A Deliberative Process for Establishing Added Value
The amount of market entry rewards, options for delinking revenues, and eligibility criteria all stand to affect the programs’ viability and its effects—both intentional and unintentional—on the global antimicrobials market. The varied experiences with legal and regulatory pull incentives described here point to a need for deliberation and piloting of possible market entry reward programs, as both Sweden and the United Kingdom are currently doing (Gotham et al., 2021). The Swedish program combines partially delinked rewards and a minimum guaranteed annual revenue amount for a qualifying drug that has efficacy against a WHO critical, priority pathogen and an acceptable safety profile (Gotham et al., 2021). The British program uses fully delinked fixed annual payments in the range of $40 to $140 million regardless of volume (Gotham et al., 2021).12 Participation in both pilot programs is voluntary and requires companies to apply for consideration. The Swedish pilot program will run through 2022 (Gotham et al., 2021). The British program launched, after some delays, in June 2020 (Mahase, 2020). Later that year, NHS England announced the selection of Fetroja®, a treatment for drug-resistant, gram-negative bacteria in patients with limited treatment options, and Zavicefta®, a combination antibacterial used against serious, gram-negative infections (Bassetti
___________________
12 The amount represents 2 percent of the $2 to $4 billion valuation for a new antimicrobial. The percentage is based on the UK share of global pharmaceutical sales (Gotham et al., 2021).
et al., 2021; BusinessWire, 2017; Perkins and Glover, 2020). Fetroja® is made by Shionogi and Zavicefta® by Pfizer; both products should be available to patients in late 2022 (Perkins and Glover, 2020).
Market entry rewards require significant investment of taxpayer dollars. While the threat of antimicrobial resistance is real and more antimicrobial drugs are needed in our arsenal, it is important to ensure the best possible design and execution of reward payments to minimize the risk to taxpayers. Before funding any market entry reward, the government needs to be clear that it is rewarding a truly novel and useful antimicrobial.
Recommendation 6-1: A Department of Health and Human Services (HHS) interagency committee should establish well-targeted, objective criteria to identify novel antimicrobials with high potential for filling a critical, unmet need. HHS should then support trials to establish the additional clinical benefit and optimal use of these drugs.
The importance of independent, objective criteria for determining eligibility for payment cannot be overstated. The success of any future market entry reward program depends on these criteria against which a product’s true value would be assessed. For this reason, the committee recommends such criteria be set by an independent panel (Daniel et al., 2018). Recognizing that there will be differences of opinion as to what constitutes a product of meaningful value to public health, the deliberative process for setting criteria should be open and the relative weight given to competing criteria made public (Schneider et al., 2020b).
An arbiter on what constitutes critical unmet need
Pending legislation in Congress makes attention to the eligibility criteria for market entry rewards especially urgent. One of the main criticisms of the GAIN Act is its reliance on overly broad criteria to qualify as an infectious disease product (i.e., products for which there is an effective alternative are not excluded) (Gatti et al., 2020; Rubin, 2016). What is more, the designation as a qualified infectious disease product may be misunderstood to be a reflection of value, something that could justify excessive spending on a drug with limited to no added benefit to the public health (Darrow and Kesselheim, 2020). The DISARM Act, currently introduced in the Senate, carries forward GAIN’s flawed eligibility criteria in its plan to raise CMS reimbursement on qualified antimicrobials. Therefore, the pending DISARM legislation gives some urgency to the need to narrow the eligibility criteria for market entry rewards. One important role for the proposed committee would be
to identify those products with greatest potential for clinical value to avoid continued reliance on the GAIN Act’s criteria.
The Pioneering Antimicrobial Subscriptions to End Up-surging Resistance (PASTEUR) Act proposes that governments pay for new antimicrobials by subscription, similar to the model NHS England is currently piloting.13 Antimicrobials determined to meet a critical need would earn annual contracts of between $750 million and $3 billion a year, paid out over a period of up to 10 years or length of patent exclusivity.14 This model would fully delink drug sales from revenues, as the award would be independent of quantity of drug sold. Eligibility for payments includes but is not limited to “treating infections for which there is unmet need; improving clinical outcomes for patients with multidrug-resistant infections; being a first-approved drug that treats certain multidrug-resistant infections, and, to a lesser extent, second and third drugs that treat such infections; addressing an infection located in an organ or other location that is challenging to treat; or addressing a multidrug-resistant infection through a novel chemical scaffold or mechanism of action, especially through oral administration.”15 It also calls for regulatory measures to establish the relative weight assigned to each of these desired characteristics.16 Recent revisions to the PASTEUR Act make it clear that the subscription payments would end if the drug developer fails to submit a plan for registering it in low- and middle-income countries. Purchased drugs would also be available to Medicare, Medicaid, and Veterans Health Administration beneficiaries in the United States; the revisions also allow for smaller contracts with new developers and a requirement that the list of high-priority microbes for which medicines are needed be updated every other year.17
The PASTEUR Act aims to reward drugs that improve clinical outcomes in drug-resistant infections. It contains similar provisions for the establishment of an interagency Committee on Critical Need Antimicrobials to identify products that meet a real clinical need.18 This group is an essential feature not just of the PASTEUR Act, but of any public effort to reward novel antimicrobials. For this reason, the committee recommends HHS convene this panel regardless of how or when Congress votes on the PASTEUR Act.
A new antimicrobial’s real clinical value is not usually obvious at the
___________________
13 The PASTEUR Act, HR 8920, 116th Cong., 2nd sess., Congressional Record 166, no. 208, daily ed. (December 9, 2020): H 7111.
14 The PASTEUR Act, S 4760, 116th Cong., 2nd sess. (September 30, 2020).
15 The PASTEUR Act, S 4760, 116th Cong., 2nd sess. (September 30, 2020).
16 The PASTEUR Act, S 4760, 116th Cong., 2nd sess. (September 30, 2020).
17 The PASTEUR Act, S 4760, 116th Cong., 2nd sess. (September 30, 2020).
18 The PASTEUR Act, S 4760, 116th Cong., 2nd sess. (September 30, 2020).
time it is approved, however. The key challenge is to keep the drug on the market and used sparingly for long enough to establish its value and extend its label indications (Clift et al., 2015). It may be possible to use a drug’s early postmarket years, traditionally intended for surveillance of infrequent side effects and assessment of cost-effectiveness, for adaptive trials to this end (Lanini et al., 2019). These trials could be integrated into infection control programs in places that see considerable incidence of drug-resistant infections, including the long-term acute care hospitals and dialysis centers described in the previous chapter (Lanini et al., 2019).
Public funding for trials that establish value
One of the main advantages the proposed strategy would bring to the discussion of incentives for antimicrobial development is the public funding for late-stage trials. This would be a major incentive for drug developers, as clinical trials are costly to run. It would also benefit prescribers who may be reluctant to use a drug outside of its approved indications. Even those who are willing to authorize such use, infectious disease specialists, for example, have difficulty using new medicines off label because of lack of clarity on the dose or duration of treatment. By identifying the most promising antimicrobials and supporting their late-stage trials, HHS could help bridge a crucial gap preventing use of new antimicrobials.
Having the government support the label extension and clinical value trials for promising antimicrobials has several advantages. The first is cost. The exact costs of clinical trials are confidential, and industry estimates may be padded to justify high drug prices (Aagaard et al., 2021). In any case, roughly half the expense of the trial is driven by the cost of capital, the company’s lost opportunity to invest money used in the trial (Aagaard et al., 2021). The cost of capital is not an expense that would apply to a government funder, making the total cost of trials considerably lower.
There would also be logistical advantages. As this chapter has explained, finding trial participants for antimicrobial studies is difficult. However, as COVID-19 has shown, trials working across multicenter consortiums can quickly enroll the patients they need and report trials results (Li Bassi et al., 2020). As with COVID-19, it may be wise to include international centers in the network both to ease the licensure of novel antimicrobials abroad and to speed the process of establishing added clinical value (Trusheim et al., 2016).
The HHS interagency committee would select antimicrobials for which HHS would fund the additional studies on clinical value. At this time, the drug sponsor could also receive a milestone payment, which may be necessary to keep the company in business. This split approach to market entry rewards also controls the risk to the taxpayer, as the pay-
ment would be smaller than the single, lump-sum payments that have been proposed as market-entry rewards. It is also possible that the proposed interagency committee could conclude that, in certain cases, some kind of fully delinked lump sum reward would be warranted.
This strategy is deliberative, and critics may find it too time consuming in a market where new antimicrobial developers routinely go bankrupt in their drug’s first year on the market (Jacobs, 2019; Lepore and Kim, 2021; Plackett, 2020). At the same time, it is not financially or politically feasible for Congress to authorize payments of a billion dollars or more without significant deliberation on the value of the investment. HHS is well positioned to advise on this value, so Congress can properly target the taxpayers’ investment in novel antimicrobials.
A Nonprofit Model
By providing trial funding and identifying candidate medicines that need it, the recommended strategy is essentially a public–private partnership for drug development. There is precedent for this kind of partnership in antimicrobial development, including CARB-X and the BARDA partnerships discussed earlier in this chapter, as well as many similar partnerships in Europe (Desselle et al., 2017). It is also possible, however, that drugs with a very small market may be natural nonprofits (i.e., it is not possible to profit from their sale). If so, one alternative to public spending in the form of market entry rewards is to invest the same amount of money (or less) in a nonprofit drug development institute.
A nonprofit model may be better suited to development of medicines with small markets and low peak sales (Nielsen et al., 2019). It would also be in a better position to promote judicious use of the drug if the developer were a nonprofit as the imperative to sell the drug, often at odds with good stewardship, would be removed (Nielsen et al., 2019). New compounds could be introduced sequentially and over fairly long intervals, promoting good drug stewardship (Spellberg, 2021). For reference, four new antimicrobials targeting extremely drug-resistant, gram-negative bacilli have been introduced since 2015; they compete for a small market of carbapenem-resistant Enterobacterales infections (Nielsen et al., 2019).
Another point in favor of a nonprofit model is that it may require less financial support from the taxpayer than the other incentive programs suggested. A $1 billion investment has been suggested as sufficient seed capital to create such an institute, and this money would be invested only once, making it more sustainable than long-term subsidies for drug companies (Nielsen et al., 2019; Spellberg, 2021). This type of institute might also be able to draw from expertise in government and academia (Desselle et al., 2017). It would likely use the same contract manufacturers and
contract research organizations as the biotechnology firms engage today to manage the trial and manufacturing steps in the drug development process. One major difference, however, is that a nonprofit developer would not have the same expectation to recoup the costs of development with sales (Aagaard et al., 2021). This is an advantage with antimicrobial development as society benefits from the drugs being held in reserve. It is also not clear that the cost of developing a new antimicrobial even could be recouped through sales. Recent research indicates that the majority of new antimicrobials approved in the 2010s were accessible in only three countries (the United States, the United Kingdom, and Sweden) (Outterson et al., 2021). It is possible that the expected sales of these medicines are not sufficient incentive for companies to outweigh the costs of seeking authorization in other markets (Outterson et al., 2021).
At the same time, entry of a nonprofit antimicrobial drug developer could alter antimicrobial market dynamics and has the potential to crowd out private investment. In the committee’s judgment there is too much uncertainty to accurately assess whether this change would result in a net societal benefit to recommend this strategy. Such a change may be necessary in the future, however, and is an important topic for ongoing public discussion.
Ensuring the global reach of new products
Some of the precedent for nonprofit drug development comes from products intended largely for low- and middle-income country markets. The GARD-P and DNDi examples discussed in Box 6-2 are evidence that nonprofit drug development is valuable, especially in developing products for patients who will not be able to pay for them. GARD-P and DNDi give considerable attention to the registration of new medicines in low- and middle-income countries, as does the nonprofit Medicines for Malaria Venture (DNDi, 2021; GARDP, 2021b; MMV, 2021).
The push and pull incentives described earlier in this chapter are evidence of the considerable sums of money the United States and other high-income countries are willing to spend to fight antimicrobial resistance. When the taxpayer spends a billion dollars or more to bring a new product to market, the government may rightly have a say in how and where that product is deployed. This logic underlies a condition of accepting CARB-X funds. Projects supported with the Global Antimicrobial Resistance Innovation Fund via the British government’s development assistance must produce a stewardship and access plan detailing how the product will be made available and affordable in low- and middle-income countries (CARB-X, 2021d). CARB-X stewardship and access guidelines
clarify that these products are intended to “primarily and directly benefit” patients in low- and middle-income countries (CARB-X, 2021d).19,20
The emphasis on a primary and direct benefit to patients in low- and middle-income countries may be somewhat arbitrary, however. Given the vastly higher burden of antimicrobial resistance in these parts of the world and problems with access to safe, affordable medicines, it would seem that almost any product, at least any novel antimicrobial, rapid point-of-care diagnostic, or preventive product would be disproportionately beneficial in low- and middle-income country markets, markets these products do not currently reach. For this reason, the ReACT network, an international group dedicated to mitigating antimicrobial resistance, has proposed that any product developer that takes government money for product development should enter a patent pool facilitating global procurement (Aagaard et al., 2021).
DIAGNOSTICS
The process of culturing bacteria does not provide susceptibility information fast enough to inform the first choice of medicine (Okeke et al., 2011). For this reason, diagnostic testing is a rate-limiting step in the optimal use of antimicrobials. Slow or expensive diagnostic tests influence providers to use empiric treatment and contribute to considerable misuse in human and animal health. A lack of rapid diagnostic tests also holds back the development of new antimicrobial medicines. With rapid diagnostics, researchers could identify participants for narrow-spectrum drug trials faster, removing a serious logistical hurdle in the new drug approval process (Okeke et al., 2011).
Antimicrobials are underpriced because the price does not include the future cost of resistance (Okeke et al., 2011). One option to adjust the value calculation for using antimicrobials might lie with subsidizing the cost of the diagnostic tests that inform the decision to use antimicrobials in the first place. Making up-to-date diagnostic testing easier would advance the goal of antimicrobial stewardship and the correct use of new medicines. New drugs pose challenges to diagnostic laboratories, however.
___________________
19 The same stewardship and access guidelines ask all product developers to describe plans for sublicensing the product in low- and middle-income countries via the Global Antibiotic Research and Development Partnership (GARDP), as well as compassionate use or equitable pricing plans (CARB-X, 2021d).
20 This text has changed since the prepublication release of this report to clarify CARB-X sponsorship requirements around licensing drugs for use in low- and middle-income countries.
Barriers to Keeping Diagnostic Tests Up to Date21
As the previous chapter discussed, antibiotic susceptibility testing is one of the mainstays of diagnostic microbiology. Susceptibility results allow providers to tailor antimicrobial treatment. These results can also prompt de-escalation from the broader-spectrum antibiotics often selected for empiric therapy to a narrower, more targeted antibiotic, thereby curtailing the selection pressure that drives emergence of resistant pathogens (van Belkum et al., 2020). When resistant pathogens are involved, susceptibility testing gives insight into the mechanism of resistance. It can also identify asymptomatic patients infected with resistant pathogens, allowing for them to be isolated if necessary to control a resistant outbreak (Burnham et al., 2017). In cases of multi- and pan-resistant bacteria, there are generally few treatment options, and a newer antibiotic with activity against the target pathogen will be indicated. For this reason, susceptibility testing for novel antibiotics is necessary. These tests ensure the drug can be used and establish the dosage appropriate to treat the infection.
When clinical microbiology laboratories cannot test a pathogen’s susceptibility to a new antimicrobial, then clinicians will not feel comfortable using it, seriously limiting the use of the new medicine. Another possibility is that the drug would be misused, triggering the development of resistance before the new antimicrobial even sees wide, appropriate clinical use (Burnham et al., 2017). But there are multiple barriers to susceptibility testing for new antimicrobials. First is the inclusion of the microbe–drug combination in automated testing device panels (Krause, 2021b). As the previous chapter described, these are essentially flat test plates holding wells, or microtubes, each containing different concentrations of medicines. The test plates are read by machine; breakpoint changes are automatically updated as part of routine software updates. The number of wells the plates can hold is fixed, so adding a new medicine to the plate usually means removing something else and forfeiting the associated diagnostic information. Removing an old medicine in favor of a new one is not something most device companies are inclined to do, particularly when there is no strong demand for new antibiotics since these medicines are meant to be used only infrequently.
In the absence of an automated testing option, the susceptibility of a pathogen to a new drug can be established with manual testing methods. The drug manufacturer will, for example, supply discs or strips (i.e., E-tests®) saturated with the drug, which diffuses onto a culture plate
___________________
21 This section deals with the challenges of susceptibility testing in human medicine. Veterinary antimicrobial susceptibility testing is done with broth micro dilution; disk diffusion is less common (Bowden and Burbick, 2020). Challenges relating to the need for animal-specific test breakpoints were discussed in Chapter 5.
inoculated with the target pathogen (ScienceDirect, 2021a). The drug’s potency against the target pathogen can be inferred from the diameter of growth it inhibits in the culture (ScienceDirect, 2021a). Using such manual methods is difficult for most clinical laboratories in the United States because they use automated methods. First of all, these methods are extremely time-consuming (Benkova et al., 2020). Culturing pathogens requires an incubation of at least 16 hours, sometimes several days, while automated tests usually yield results in 6 to 12 hours (Benkova et al., 2020). Manual testing requires more staff time, the redirection of staff disrupts routine workflow, and test results do not automatically integrate with the hospital information system (Humphries et al., 2018c). These methods are used less frequently so fewer technicians practice them regularly enough to remain proficient (Humphries et al., 2018c; van Belkum et al., 2020).
The disks and test strips manufacturers provide with new antibiotics are often designated as “research use only” until they receive regulatory clearance, a process that can take months or years. FDA does not authorize the use of research-only tests to inform clinical care as the manufacturer’s tests have not been through regulatory clearance to establish their clinical performance (Humphries and Hindler, 2016). Research-only tests are meant to inform surveillance and to give clinical microbiologists better information about patterns of susceptibility in the organisms they are seeing (Humphries and Hindler, 2016). As a condition of requesting reagents for research-only tests, the requesting scientist has to attest in writing that the results will not be reported to a physician or used to guide treatment; the clinical laboratory cannot bill for the tests (Humphries and Hindler, 2016). The associated liability concerns are enough to prompt many hospitals to prohibit the use of research-only diagnostics (Humphries and Hindler, 2016).
Most clinical laboratories in the United States today use only automated methods for susceptibility testing (Humphries et al., 2018b,c). Therefore, the inclusion of new antimicrobials on automated testing systems is a practical requirement for use, but is not, strictly speaking, essential for the safe and effective use of the medicine. If it were, FDA would require the medicine and diagnostic be developed and reviewed simultaneously, eliminating any lag time between the availability of the drug and associated diagnostics (FDA, 2018c). Since the regulatory approval for the medicine and diagnostic are separate, but related, the drug and device companies have to cooperate on the development of the automated test. This involves significant expense on both sides.
The device company, for its part, must be regularly reevaluating the time needed to bring a new test through regulatory review against obligations to support testing changes for drugs already in wide use. The
volume of breakpoint changes alone creates an overwhelming amount of work when incorporating them into automated susceptibility testing devices (Brasso, 2017). Emerging resistance to a medicine that is heavily used has, after all, more immediate implications for public health than the introduction of a new one (Krause, 2021b).
Developing tests for new antimicrobials can be slowed when actual breakpoints are lower than what the manufacturer predicted, as often happens when the wild-type pathogens’ resistance mechanisms render it less susceptible to the drug than was initially assumed (Carpenter and Brasso, 2016). Changes in the indications for use can also present a barrier. Indications for a new antimicrobial are usually narrow during drug development (Theuretzbacher et al., 2020). Early on, the range of pathogens the new molecule has activity against is not always clear or may be described only generally (e.g., active against gram-negative bacteria) (Theuretzbacher et al., 2020). Because of the demands of clinical trials and participant recruitment, the drug developers usually apply for approval using only one indication. Many pathogens against which the drug shows activity, especially rare ones or uncommon clinical presentations, are not included on the FDA approved drug label (Boucher et al., 2017). Such regulatory changes make it impossible for the device manufacturer to start developing the susceptibility test before the drug company has at least filed a new drug application containing the proposed drug label and indication with FDA (Carpenter and Brasso, 2016). Even then, only those pathogens and indications included in the final FDA-approved drug label can be included in the regulatory application for the device (Shawar, 2016).
After any changes to automated susceptibility tests, either the addition of new drugs or updating of breakpoints, the testing device and software all need to be updated (Humphries et al., 2018c). This has long presented a challenge to clinical microbiology labs. Even after the Clinical and Laboratory Standards Institute (CLSI) had gone through the research to revise a breakpoint, it could take years for these standards to be included in automated devices. A 2016 survey of California clinical laboratories found that only 72 percent of the state’s 128 clinical microbiology laboratories used the most recent carbapenem breakpoints for Enterobacterales and that implementing the new breakpoint took a median of 4.5 half years (Humphries et al., 2018b). The use of an outdated breakpoint can cause clinicians to make incorrect treatment decisions, to say nothing of downstream effects such as failing to implement contact precautions and allowing an outbreak to spread.
The mismatch of regulatory timelines for drug and diagnostic developers contributes to the delay in bringing new antimicrobials into automated test panels. More recent changes in the FDA review for automated
susceptibility test devices has brought the timelines into better alignment, but there is still a lag time of up to several years between an antimicrobial being introduced into clinical practice and routine diagnostic testing of pathogens’ susceptibility to it gaining regulatory clearance (Burnham et al., 2017). This delay limits the market viability of the medicine.
The validation and trials necessary to bring a new automated susceptibility test to market are time consuming and costly (van Belkum et al., 2020). Regulatory approvals are separate for each new antimicrobial and each new indication (Carpenter and Brasso, 2016). Still, much has improved in the last 5 years, especially for manual test methods such as disk diffusion, which can be included in the data the company submits with the new drug application. For example, because of close and early collaboration among the drug developer, device company, and FDA, three manual susceptibility tests for the new drug delafloxacin gained regulatory clearance in only 44 days (compared to the more typical lag time of several years) (FDA, 2019a; Humphries et al., 2018a).
FDA’s 2016 draft guidance publication, Coordinated Development of Antimicrobial Drugs and Antimicrobial Susceptibility Test Devices, encouraged collaboration between drug and device companies in relatively early stages of drug development (FDA, 2019a). Collecting clinical isolates, previously a major bottleneck in test development, improved with the creation of the FDA and the CDC Antibiotic Resistance Isolate Bank, which provides isolates to companies developing diagnostic tools and for validation studies (CDC, 2020a; Shawar, 2016).
The 21st Century Cures Act, signed into law in 2016, also helped by ensuring a more efficient process for updating breakpoints. Rather than having FDA-recognized breakpoint criteria on drug labels, the act moves this information online (FDA, 2020a; Humphries et al., 2018a). Two websites, one for antifungals and one for antibacterials, lists all the current FDA-recognized breakpoints from drug labels (FDA, 2017, 2020a). Every 6 months, FDA has to update the breakpoint websites, ensuring more timely addition of new or revised breakpoints into clinical practice; the agency can also recognize any new or updated breakpoints not included on a drug’s label. Therefore, the automated device companies can use any breakpoints from these websites in their applications for regulatory clearance. Removing breakpoints from drug labels also decouples the drug’s label indications from susceptibility testing, allowing device manufacturers to use breakpoints for microbe–drug combinations with demonstrated in vitro activity, even if there are not necessarily in vivo studies establishing the same (FDA, 2020a; Humphries et al., 2018a). The act also recognizes some but not all CLSI breakpoints (Humphries et al., 2018a).
Despite significant recent progress, driven in large part by the 21st Century Cures Act, there are still regulatory restrictions that stand in the
| Challenge | Addressed by 21st Century Cures Act? |
|---|---|
| Test devices cleared after 2007 can only test antimicrobials against organisms for which there are clinical indications listed in drug label | Yes |
| Current breakpoints are not available on all test devices used by clinical laboratories | Partially |
| Lack of test devices for new drugs | No |
| Comments |
|---|
Progress:
|
Progress:
|
Progress outside 21st Century Cures Act:
|
NOTE: CDER = Center for Drug Evaluation and Research; CDRH = Center for Devices and Radiological Health; CLSI = Clinical and Laboratory Standards Institute; FDA = Food and Drug Administration.
SOURCE: Humphries, R. M., and J. Hindler, Twenty-first Century Cures Act and Antimicrobial Susceptibility Testing: Clinical Implications in the Era of Multidrug Resistance, Clinical Infectious Diseases, 2018, Vol. 67, Issue 7, by permission of Oxford University Press.
way of prompt regulatory clearance of antimicrobial susceptibility test devices (see Table 6-2). One important limitation is that automated test devices are still limited to only the microbe–drug combinations approved by FDA (Humphries et al., 2018a). FDA strongly discourages testing or interpretation of pathogens not included in the approved drug label, even if, as sometimes happens with antimicrobials, the off-label indication is widely used (Humphries et al., 2018a).
Furthermore, FDA still retains full authority to accept or reject breakpoints proposed by CLSI. There are still many CLSI breakpoints not recognized by FDA, sometimes because the agency recognizes an older or higher breakpoint (FDA, 2021a). FDA’s willingness to recognize some CLSI breakpoints is related to the agency’s formal recognition of CLSI as a standards development organization (FDA, 2021a). This recognition hinges on CLSI meeting statutory requirements for transparency, scientific rigor, vetting its volunteers for conflicts of interest, and soliciting public input on technical decisions (FDA, 2021a). Because automated susceptibility test manufacturers can use the recognized breakpoints from the FDA websites in their test development, it streamlines the manufacturer’s process to keep the devices up to date (FDA, 2021a).
As antimicrobial resistance continues to emerge and more data are available, breakpoint changes will only be needed more frequently. Every investment in keeping automated testing devices up to date is an investment in keeping clinical practice more responsive to antimicrobial resistance and protecting public health (Humphries, 2018). By recognizing all CLSI breakpoints, FDA could allow for more widespread use of breakpoint criteria for many additional microbe–drug combinations. This in turn would allow susceptibility test manufacturers to report minimum inhibitory concentrations for the antibiotics that currently do not have FDA recognized breakpoints. This would speed the regulatory clearance by widening the number of recognized breakpoints without putting a burden on the manufacturer to develop them. This process would also lessen some burden on clinical laboratories to resort to manual testing to report these susceptibility results.
Recommendation 6-2: To reduce regulatory hurdles in bringing automated susceptibility tests to market, the Food and Drug Administration should coordinate the review of new antimicrobials with the review of their automated susceptibility tests and work with the Clinical Laboratories Standards Institute to issue and update breakpoints for microbe–drug combinations.
Ideally, automated susceptibility testing devices would include new antimicrobials immediately upon market entry and revised breakpoints
for older drugs as they are approved. This would mean the new drug and device approvals work simultaneously, not sequentially, as they currently do (i.e., the device application begins after the new drug approval). Since 2012, FDA has expedited the review process for novel antimicrobials (FDA, 2018d). A similar fast track approval option is necessary for automated susceptibility testing devices. This process would be akin to Operation Warp Speed for vaccine development and emergency use authorization granted to diagnostic tests during the COVID-19 pandemic (FDA, 2021b).
To this end, FDA and the susceptibility test device manufacturers should work together to define a less restrictive pathway for validation studies and new ways to assess device performance. This accelerated review would not compromise the quality of the devices cleared for diagnostic use, as recent experience with COVID-19 has shown. For example, allowing multiple antimicrobials or multiple indications to be included in one submission (called bundling) could ease the application burden and fees for industry. Current FDA policy allows for “one drug, one method of reading, and one method of inoculation” in susceptibility test submissions, although the manufacturer is able to bundle gram-positive and gram-negative claims provided the same procedure is followed (FDA, 2018b).
Furthermore, the label indications for new antimicrobials are usually restrictive. If CLSI has established breakpoints for other microbe–drug combinations, it would greatly ease clinical practice to recognize them. But FDA’s process for assessing CLSI breakpoints and their timeline for doing so is not clear (Humphries et al., 2018a). Antimicrobial drug labels are not all encompassing. The nature of the drug’s biological activity against microbes means that there will be demonstrable in vivo activity against organisms not included in the initial regulatory review.
A consequence of FDA not recognizing all CLSI breakpoints is that automated test device manufacturers are not allowed to report out the minimum inhibitory concentration for any drug without breakpoints recognized by FDA (Zimmer, 2021). Device companies should also be able to share the minimum inhibitory concentrations for microbe–drug combinations, even when the breakpoint interpretation is not recognized by FDA. Including minimum inhibitory concentrations in the automated test would allow clinical laboratories to interpret the susceptibility pattern for organisms even when only non-FDA recognized breakpoints exists. For example, currently FDA only recognizes meropenem breakpoints for gram-negative bacteria such as Enterobacterales, Pseudomonas aeruginosa, and Acinetobacter species. Despite existing meropenem breakpoints established by CLSI years ago, FDA still does not recognize breakpoints for Burkholderia cepacia complex and non-Enterobacterales. In the absence of FDA acceptance for these other organisms, manufacturers are prohibited
from reporting meropenem inhibitory concentrations for the latter two organism groups (CLSI, 2021; FDA, 2020e). If clinical laboratories had this information, they could avoid the need for manual test methods that few labs can support, thereby broadening drugs’ usefulness in clinical practice.
Successful implementation of this recommendation will remove some of the barriers automated susceptibility test manufacturers face in developing tests for new antimicrobials and updated breakpoints. This updating is a voluntary process for the companies. Even with the assistance that the Antibiotic Resistance Isolate Bank and streamlining of breakpoint recognition has provided, the trials and validation involved are expensive and time consuming. The companies have little incentive to go through this process, except the moral incentive to protect public health.
The Reinvigorating Antibiotic and Diagnostic Innovation (READI) Act, introduced to Congress in 2015, aimed to encourage research and development on new antibiotics and rapid diagnostics for antimicrobial-resistant pathogens by providing a 50 percent tax credit against clinical testing expenses to companies that create these products.22 This tax credit might be an even more meaningful incentive for the manufacturers of automated susceptibility devices to offset their clinical trial expenses incurred accommodating breakpoint revisions (Humphries et al., 2018a).23
Recommendation 6-3: Congress should make automated susceptibility test manufacturers eligible for tax incentives to bring new automated susceptibility tests to market.
Tax incentives and streamlining regulatory processes could do much to reduce the lag time in bringing automated susceptibility tests to market, but there are some drugs for which there will simply never be sufficient demand to warrant inclusion in an automated susceptibility panel. The decision to add a new drug to these panels is influenced by local and national epidemiology of resistance patterns as well as customer demand for the test. For those drugs that will not be included in the automated test panel, manual diagnostic testing will be necessary. Such tests pose challenges to clinical labs. The next section discusses a strategy to mitigate these challenges.
___________________
22 Reinvigorating Antibiotic and Diagnostic Innovation Act of 2017, HR 1840, 115th Cong., 1st sess., Congressional Record 163, no. 56, daily ed. (March 30, 2017): H 2601.
23 Reinvigorating Antibiotic and Diagnostic Innovation Act of 2015, HR 3539, 114th Cong., 1st sess., Congressional Record 161, no. 134, daily ed. (September 17, 2015): H 6137.
The Antibiotic Resistance Laboratory Network
New antimicrobials and breakpoint changes also pose significant challenges to clinical laboratories. The logistics of manual testing make it unrealistic for most clinical labs. While clearance of automated susceptibility test panels would remove a major hurdle, it is still likely that the testing for new antimicrobials will not be a priority if the drug is not commonly used or not on the hospital formulary. The CDC Antibiotic Resistance Laboratory Network (ARLN) aims to fill this gap by funding 55 public health laboratories, as well as seven regional labs, and the National Tuberculosis Molecular Surveillance Center to test pathogens that are beyond the capacity of clinical microbiology laboratories (CDC, 2021d). The committee commends the CDC for this valuable service. At the same time, there is room to improve the network’s ability to support public health and clinical labs.
The ARLN offers expanded susceptibility testing for hard-to-treat infections with Enterobacterales that carry metallo-beta-lactamases, enzymes that make bacteria resistant to beta-lactam antibacterials, including the carbapenems (CDC, 2021d; Palzkill, 2013). This service is free of charge, but all samples must be sent with confirmation that they are not susceptible to all the beta-lactam medicines tested, “including either ceftazidime/avibactam or meropenem/vaborbactam,” and send confirmatory molecular testing that the isolate has at least one metallo-beta-lactamase gene (CDC, 2021a). These inclusion criteria are difficult for some hospital laboratories to meet, especially if they do not have the means to test these broad-spectrum medicines. The other services the network provides (colonization screening, whole genome sequencing, molecular testing for resistance genes, culturing for carbapenemase, and identification of pathogens) have similar requirements for submission that can be challenging for clinical laboratories (CDC, 2021d).
Between 2017 and 2019, 42,423 carbapenem-resistant Enterobacterales and nearly 15,000 carbapenem-resistant Pseudomonas aeruginosa isolates were tested through the ARLN (CDC, 2021c; Vallabhaneni et al., 2021). This represents a significant investment in public health laboratories, but one that has not necessarily reached clinical laboratories or other microbiology labs. For example, in fiscal year 2020 the CDC invested over $6.9 million in resistance programming in California alone, much of it through the ARLN, only $609,000 of which went to universities or health care partners, and that to only two universities (CDC, 2021b). This investment stands to grow: the CDC’s fiscal year 2022 budget included $672 million for expanding the ARLN domestically and internationally (IDSA, 2021).
Expanding the ARLN would be helpful, but this expansion would ideally be done in a way that extends the reach of the services offered in the most efficient and economical way possible. To this end, inclusion of
all broad-spectrum drugs in the expanded susceptibility testing service would be helpful, including cefiderocol and others not yet offered. In a larger sense, it would help to have the network put at least as much of an emphasis on clinical diagnostics as on surveillance. The ARLN emphasis on surveillance means that many of the results are reported to the CDC but not back to the clinical laboratories sending the samples (Vallabhaneni et al., 2021). There is a difference between the public health laboratories’ responsibility for surveillance and the need for support for challenging clinical testing. Relative to the emphasis on surveillance, the ARLN’s support for clinical testing is less.
Recommendation 6-4: The Centers for Disease Control and Prevention (CDC) should expand the capacity of the Antibiotic Resistance Laboratory Network by offering expedited, expanded susceptibility testing of all broad-spectrum antibiotics via certain CLIA-certified laboratories.24 The CDC should also promote this service to clinical laboratories.
It is not reasonable to expect hospital clinical microbiology laboratories to be able to test microbe–drug combinations they see only once or twice a year. Laboratories need a backstop for these tests. The ARLN provides this service to clinical laboratories struggling with diagnostic testing and test interpretation for resistant bacteria. At the same time, there is room to improve the efficiency of the service, the turnaround time on results, and the amount of testing offered. Currently, the ARLN’s extended susceptibility testing offers results within 3 business days (CDC, 2021a). Clinical laboratories may be able to turn these results around more quickly; 48 hours would be the ideal response time for most cases, allowing that some microbes are slow growing and may take longer.
The Clinical Laboratory Improvement Amendments of 1988 (CLIA) are a set of federal regulations for laboratories that work with human specimens for the diagnosis, treatment, or prevention of disease (CDC, 2018). Before any laboratory can accept human samples for diagnostic testing, the Centers for Medicare & Medicaid Services have to certify that the lab meets CLIA regulations (FDA, 2020b). CLIA certification would be a minimum criteria for supporting the expanded susceptibility testing program recommended.
CDC could take advantage of CLIA-certified laboratories to expand susceptibility testing. Academic medical centers, for example, have laboratory technologists and clinical microbiologists with expertise in diag-
___________________
24 The Clinical Laboratory Improvement Amendments (CLIA) regulate testing and are required for laboratories handling human samples.
nostic testing. These laboratory scientists routinely use these skills and may even be available for consulting on challenging cases.
Most of the CLIA-certified laboratories best placed to offer expanded susceptibility testing will be at tertiary care, teaching hospitals. Major medical centers often have an infrastructure in place to do broad-spectrum antimicrobial susceptibility testing. Many of these laboratories are already offering in-house testing similar to the ARLN extended panel. It would generally be less of a burden on staff in academic laboratories to do these tests than it might be for the public health system. Furthermore, some academic medical centers already serve as reference laboratories, so they have systems in place to receive and process isolates from external laboratories. The challenge would be ensuring they have protected time to do them, which a formal contract with the CDC could provide. Furthermore, many of these laboratories already have community support programs in place, sometimes necessitated by a surge in demand for diagnostic testing because of COVID-19 (Tsai et al., 2021; Warrington et al., 2021).
It is not enough for the CDC to expand the ARLN’s capacity for susceptibility testing if the service is not thoroughly communicated to the clinical laboratory managers who would need to use it. The CDC could promote the service through state and local health offices and with regular targeted outreach in clinical laboratories.
INVESTING IN ONE HEALTH SOLUTIONS
As this chapter has discussed, there are serious difficulties bringing needed antimicrobial medicines to market in the United States and other high-income countries. There are also barriers to product development that cut across countries as well as some that are far more pronounced in low- and middle-income countries. In general, less attention is paid to products for animal health. No drug developer will bring a new antimicrobial to market specifically for use in animals, for example. A One Health approach to product development takes a broader view of the need for new products—both therapeutic and preventive. A One Health model is helpful in guiding countries’ support for products intended for crop and animal agriculture, aquaculture, and the environment, and as such is called out, at least in principle, in many countries’ national action plans for antimicrobial resistance (GCOA and IDSA, 2021).
While not ignoring the pressing need for improving the market for small-molecule antimicrobials and diagnostics, proper management of antimicrobial resistance in humans, animals, and the environment will require attention to a larger range of products. Given the global transmission of resistance and a shared, global vulnerability to resistant patho-
gens, some product development initiatives would be most valuable if undertaken with a goal of shared global access to novel products.
Need for Innovative Products
This section discusses how international cooperation could stimulate development of some important tools for fighting resistance. New antimicrobial medicines are obviously one type of essential and needed product. It is also possible that attention to novel delivery mechanisms could do much to improve the antimicrobial activity of existing or repurposed medicines. Nanostructured materials, for example, can be used to deliver antimicrobials, and some nanoparticles have antimicrobial activity on their own (Baptista et al., 2018). Advances in materials science have brought about new biomaterials to deliver antimicrobials and antibacterial polymers with preventive uses such as catheters that resist infection (Kalelkar et al., 2021).
There is a need for a variety of new and innovative products to combat antimicrobial resistance. Factors unique to the development of novel small molecule medicines are discussed earlier in this chapter; this section will give more attention to the need for new diagnostics and preventive tools for human and animal health. This is not an exhaustive discussion of all preventive products needed, however. Promising anti-virulence and phage therapies, for example, are not discussed in detail, neither are nanostructures. Rather, this section will highlight some important needs and challenges in the market for point-of-care diagnostics and some widely used preventive products.
Point-of-Care Diagnostics
The diagnostic tests currently considered rapid are those feasible in one microbiologist’s 8-hour shift (van Belkum et al., 2019). Especially with gram-negative infections, starting antibiotic treatment with a properly targeted treatment in the first 6 to 12 hours is crucial for the patient’s recovery prospects (Burnham et al., 2017). There is good evidence that antibiotic use declines with increasing use of rapid diagnostics (Goossens et al., 2005; van de Sande-Bruinsma et al., 2008). Rapid tests are also essential to support clinical trials for new antimicrobial medicines. Such tests enable the identification of the appropriate patients, thereby reducing the total number of enrolled patients in a trial (Okeke et al., 2011).
As this chapter has discussed, the true value of diagnostics can be difficult to determine. The problem of lack of funding and low return on investment (real or perceived) is a special barrier to bringing rapid diagnostics to market, especially in the low- and middle-income countries
TABLE 6-3
Clinical Needs for Rapid Point-of-Care Diagnostics for Respiratory Tract Infections
| Technology Requirements | Purpose | Desired Characteristics | Technological Innovation and Current Stage of Development | |
|---|---|---|---|---|
| Viral respiratory infections | Point-of-care (eg, primary care office, outpatient clinics, accident and emergency) | To distinguish viral and bacterial infections and inform antiviral therapy. Infection control and bed management allowing patients with different viruses to be separated; outbreak tracing |
|
|
| Community-acquired pneumonia | Near-patient, rapid response (eg, in larger outpatient clinic or laboratory adjacent to accident and emergency) | To diagnose cause of infection and recommend effective and proportionate antimicrobial therapy, to asses whether patient should be admitted |
|
|
| Technology Requirements | Purpose | Desired Characteristics | Technological Innovation and Current Stage of Development | |
|---|---|---|---|---|
| Hospital-acquired pneumonia and ventilator-associated pneumonia | Rapid response (near intensive care unit/in clinical microbiology laboratory with good transport and communication systems) | To diagnose cause of infection and recommend effective and proportionate antimicrobial therapy |
|
|
| Tuberculosis | Point of care (eg, doctors office, tuberculosis clinic) | To identify those with acute tuberculosis and needing therapy |
|
|
NOTE: NAAT = nucleic acid amplification techniques.
SOURCE: Reprinted from The Lancet Infectious Diseases, Vol. 14, No. 11, Alimuddin Zumla, Jaffar A. Al-Tawfiq, Virve I. Enne, Mike Kidd, Christian Drosten, Judy Breuer, Marcel A. Muller, David Hui, Markus Maeurer, Matthew Bates, Peter Mwaba, Rafaat Al-Hakeem, Gregory Gray, Philippe Gautret, Abdullah A. Al-Rabeeah, Ziad A. Memish, Vanya Gant, Rapid point of care diagnostic tests for viral and bacterial respiratory tract infections—needs, advances, and future prospects, 1123-1135, Copyright (2014), with permission from Elsevier.
that bear the highest burden of resistant infections (Okeke et al., 2011). For the tests to achieve the needed reach in these settings, they need to be easily usable and not dependent on clean water, electricity, or specialty training (Moeller et al., 2007). Models suggest that rapid diagnostics for respiratory tract infections alone could avert over 150,000 child deaths a year and far more unnecessary courses of antimicrobial (Okeke et al., 2011). Yet the diagnostic tests needed for respiratory infections shown in Table 6-3, and published as part of a 2014 Lancet series, are, for the most part, still needed today.25
Products for Human Health
The purpose of antimicrobial chemotherapy is to treat infection. Antimicrobials are also used injudiciously when infections are suspected but not actually present. Therefore, tools that can prevent infections or lower the burden of viral illness have the potential to reduce both judicious and inappropriate antimicrobial use. Vaccines can do this. They can also prevent healthy, vaccinated people from being colonized by resistant bacteria (Anderson et al., 2018; Bloom et al., 2018; Lipsitch and Siber, 2016). As the previous chapter discussed, these benefits can extend beyond those who receive the vaccine by way of population effects.
More infectious diseases today are vaccine-preventable than in any other time in history. Vaccines that prevent human infections caused by bacteria, particularly those bacteria that are resistance prone, such as pneumococci, Haemophilus influenzae, cholera, and typhoid fever, may also prevent the emergence of resistance (Kaufhold et al., 2019; Lewnard et al., 2020; Lipsitch and Siber, 2016; Moore et al., 2015; Okeke, 2009). Viral vaccines such as influenza vaccine, rotavirus and, more recently, respiratory syncytial virus and COVID-19 vaccines prevent syndromes for which antimicrobials are commonly misused, and can therefore also lower selective pressure for antimicrobial resistance (Buckley et al., 2019; Lewnard et al., 2020; Vekemans et al., 2021). The formidable net effect that vaccines could have on antimicrobial resistance in humans is generally under-appreciated and grossly under-exploited (Vekemans et al., 2021).
Vaccines specifically targeted at resistant bacteria, particularly those that have been highlighted as urgent or serious threats by the CDC, and as critical or high-priority resistant pathogens by the WHO, remain to be approved. Some promising candidates are in development; there are four in the CARB-X portfolio, for example. But it could take years before they are available to prevent resistant infections (Vekemans et al.,
___________________
25 The committee recognizes some changes since this table’s publication, including the introduction of rapid, highly multiplexed nucleic acid amplification tests.
2021). Such “anti-resistance” vaccines would be valuable for high-risk groups such as residents in nursing homes or women with recurrent urinary tract infections. They could also be used to vaccinate patients upon hospital admission to protect them against antimicrobial resistant nosocomial pathogens. Outside hospitals and other care facilities there is also a pressing need for Neisseria gonorrhoeae and Shigella vaccines as multiple drug-resistant pathogens belonging to these species are spreading worrisomely.
When resistant bacterial infections cannot be prevented through hygiene or contact precautions or with the use of specific vaccines, resistant organisms and the resistance genes they carry can spread. Therefore, vaccination is one area where inadequate deployment in other parts of the world contributes to the burden of resistance in the United States (as does inadequate deployment in the United States). Global travel is easy, and most travel vaccines are not required even when good options exist (McAteer et al., 2020). Therefore, antibiotic-resistant organisms and resistance genes can be imported via travel and trade (D’Souza et al., 2021; Frost et al., 2019).
Preventive Products for Animal Health
Intensive agriculture in high-income countries depended on antimicrobials well before developments in biosecurity, and selective breeding made these interventions less necessary. Farmers in low- and middle-income countries need not repeat this pattern. Preventive tools will be essential to reducing agricultural use of antimicrobials. Even if new antimicrobial medicines were coming to market frequently, none would likely be authorized for veterinary use (Laxminarayan et al., 2015). It is therefore important to look to preventive measures to control the emergence of resistance and reduce the need for antimicrobials in agriculture and aquaculture.
Infection prevention strategies for animals, as for humans, include access to safe water, improved sanitation and biosecurity, selective breeding, and vaccines. They also include therapeutic alternatives to antimicrobials such as antibodies, probiotics and fecal transplant therapy, bacteriophages, and antimicrobial peptides, among many others (Czaplewski et al., 2016; Ghosh et al., 2019). Another promising alternative tool to directly fight antimicrobial resistance is the use of oligonucleotides for silencing resistance genes or other approaches still in research stages (Czaplewski et al., 2016). Many of these strategies show great promise but will require further experimental and translational expertise to bring them to market if they are to deliver clinical benefit (Czaplewski et al., 2016).
While new antimicrobial medicines are sorely needed, in the long
term, preventive tools and alternative therapies may do more to break the cycle of resistance (Roope et al., 2019). The next sections discuss two commonly used tools to avoid antimicrobial use in agriculture: vaccines and probiotics.
Agricultural vaccines
Reducing the use of antimicrobials in animal production demands alternatives that can be used to maintain animal health and welfare and sustain the productivity of animal agriculture. Vaccines have been successfully used for the prevention and control of infectious diseases and represent promising alternatives for antibiotics (Hoelzer et al., 2018). In Norway, for example, aggressive vaccination, in conjunction with good management, has eliminated the need for antibiotics in salmon production (WHO, 2015). Vaccination has also successfully resulted in less use of antibiotics in terrestrial animal species such as swine and poultry (Hoelzer et al., 2018). Despite the demonstrated benefits of vaccination, there is still a significant shortage of efficacious and economically affordable vaccines for animal agriculture (Hoelzer et al., 2018). There are too few vaccines, and those that are approved tend to be less affective against polymicrobial infections, which often occur under natural conditions (Chae, 2016; Chamorro and Palomares, 2020). The cost of vaccine production and difficulty in administration further limit their use (Hoelzer et al., 2018).
Vaccines and other innovative products for preventing infection in animals are especially needed in low- and middle-income countries. This need motivated the World Organisation for Animal Health (known by the historical acronym OIE), to convene two expert groups to set priorities for agricultural vaccine development (Erlacher-Vindel, 2019). Their choices highlighted diseases for which there is no available vaccine or the existing vaccine is impractical to use (e.g., every fish in a pond has to be individually vaccinated) or cost prohibitive (OIE, 2015, 2018). In response, animal medicines producers, through their industry association Health for Animals, committed $10 billion to research and development for these vaccines and other preventive products to reduce the need for antimicrobials in animals (Health for Animals, 2020). To this end, the industry committed to developing 100 new animal vaccines, 20 new diagnostic tools, and at least 50 other nutritional or immune-boosting products (Health for Animals, 2020). This is valuable work and an area where government support at relatively modest levels could help ensure the products reach their intended markets. Cooperative regulatory review, for example, would facilitate prompt licensing of the new vaccines. International cooperation, harmonized review, and a common application form could also do much to improve the reach of new animal vaccines, much as the regulatory cooperation via the International Council for Harmonization of Technical
Requirements for Pharmaceuticals for Human Use has done for human medicine.
In promoting livestock and finfish vaccines for use in low- and middle-income countries, it will be important to ensure the products are affordable and can be easily administered on farms. Without an economic reason, producers are unlikely to consider the use of vaccines in lieu of antimicrobials, thus losing the benefits of vaccines as an alternative to antimicrobials.
New vaccines could control considerable antimicrobial use in agriculture. The committee recognizes that it is not possible to produce vaccines against every pathogen in every animal species. Nevertheless, an OIE ad hoc group looking at this topic developed a list of priority diseases and pathogens in poultry, swine, and finfish (OIE, 2015). Similarly, the American Veterinary Medical Association’s Committee on Antimicrobials has identified host and species-specific pathogens of concern in both food-producing and companion animals (Scheftel et al., 2020). These lists are a good starting point for research and development efforts in animal vaccines.
Microbiome strategies
Probiotics are “live micro-organisms that, when administered in adequate amounts, confer a health benefit on the host” (FAO and WHO, 2002). Bacteria, yeast, and microalgae can all act as probiotics. Prebiotics are feed ingredients, such as complex carbohydrates, that modulate the microbiome of the host, promoting the growth of beneficial organisms (Davani-Davari et al., 2019).
Pre- and probiotics are relatively inexpensive, easy to use, accessible, and an environmentally friendly option for disease management. When
TABLE 6-4
Regulation of Directly Fed Micro-Organisms (Probiotics) by FDA
| Claim or Intended Use | Legal Status | Regulated as | Regulated by |
|---|---|---|---|
| Cure, mitigate, treat, or prevent disease | New animal drug | Drug | FDA |
| Affect the structure and function of the body | New animal drug | Drug | FDA |
| Without any therapeutic or structure/function claim (micro-organisms listed in AAFCO official publication) | Food | Food | State government |
| Without any therapeutic or structure/function claim (micro-organisms not listed in AAFCO official publication) | Food additives | Food additives | FDA |
SOURCE: FAO, 2016, reprinted with permission.
SOURCE: FAO, 2016, reprinted with permission.
used in animal feeds, they have an FDA designation of “Generally Recognized as Safe,” meaning they are not subject to premarket review, given a recognition among qualified experts that the product has been shown to be safe under its intended condition of use (FAO, 2016; FDA, 2019b). But, as Table 6-4 shows, there are different regulatory requirements if the probiotic is intended as a treatment or preventive product for disease.
Probiotics can work as immune modulators to enhance growth and prevent disease (FAO, 2016). For animals, these are mainly enteric diseases, but for aquatic species there is evidence that probiotics work against a variety of diseases and enhance reproduction, maintain water quality, inhibit pathogenic bacterial growth, and aid in nutrient metabolism (FAO, 2016; Martinez Cruz et al., 2012). Most probiotics need to be used daily to be beneficial to the host (Chauhan and Singh, 2018).
Probiotics can be a powerful tool to manage the risks of antimicrobial treatment on the gut microbiota that can lead to secondary infections (Ghosh et al., 2019; Schmidt et al., 2017). They have also been shown to improve animal growth, meat yield, and quality; to decrease zoonotic pathogens; and to increase survival to bacterial and viral challenge in a variety of animals used in agriculture, from chickens to shellfish (FAO, 2016; Hasan and Banerjee, 2020; Hoseinifar et al., 2018). Though the mechanism through which probiotics work is not always clear, there is evidence that they can alter stress and inflammatory response; reduce the permability of the gut walls; change the microbial flora of the gut or other tissues; promote the production of digestive enzymes and their metabolites; compete for space and nutrients with pathogenic bacteria; and, in aquaculture, improve water quality (FAO, 2016; Hasan and Banerjee, 2020; Hoseinifar et al., 2018). The relative contribution of any one of these mechanisms may differ between probiotic strains (even between those closely related), so there is an advantage to mixing probiotics with complementary mechanisms of action (FAO, 2016; Hasan and Banerjee, 2020; Hoseinifar et al., 2018).
Despite extensive research on the benefits of probiotics in animal agriculture, there are relatively few commercially available probiotic and prebiotic products consistently used in clinical practice or animal husbandry (FAO, 2016; Hasan and Banerjee, 2020). One challenge lies with screening candidate probiotic strains, a technique that relies heavily on in vitro identification of bacterial strains with antimicrobial activity, especially activity against the most common target pathogens. Figure 6-11 shows the main questions in the safety screening of these products. The next step is an evaluation of activity in small-scale cultures. This approach does not account for other potential mechanisms of action, nor does it capture the complex microbe, host, and ecosystem interactions (de Souza Vandenberghe et al., 2017). Therefore, many probiotic candidates with good laboratory poten-
SOURCE: FAO, 2016.
tial show inconsistent performance in the field (Day et al., 2019; Terpou et al., 2019). There are also barriers related to cost. Some micro-organisms that show promise in laboratory and small-scale commercial trials cannot be easily and cheaply grown at large scales (e.g., through fermentation) or formulated into stable products that can be easily transported, stored, and delivered (Cunningham et al., 2021; Fenster et al., 2019).
These are not unsurmountable obstacles. Research to develop high-throughput screening for probiotics could allow for investigation of the mechanisms of action in the target environmental conditions, facilitating discovery of effective probiotics. The same tools are already being used in antimicrobial development and animal breeding. The challenge is directing more attention to this problem in industry, academia, and nonprofits and establishing a product development and regulatory environment conducive to research and commercial development in the field.
As Table 6-4 showed, probiotics that avoid therapeutic label claims face considerably less regulatory scrutiny. Reports of uneven quality are common, especially among probiotics for human use (Drago et al., 2010; Jackson et al., 2019). Supplements not containing the labeled organisms are common; a 2013 study found less than a third of commercial, human
probiotics tested met label claims for micro-organisms listed and their viability (Drago et al., 2010). Increasing research and industry interest in probiotics would need to be mindful of this potential pitfall, possibly contributing to third-party certification to ensure product quality and confidence in the market (Jackson et al., 2019).
Facilitating Cooperation on Product Development
Insufficient exchange of information between the public and private sectors holds back the development of new medicines, diagnostics, and preventive products for antimicrobial resistance (van Belkum et al., 2019). The recent success of the product development partnerships for COVID-19 vaccines, diagnostics, and therapeutics depended on steady funding and collaboration among industry, government, and academia. The NIH Rapid Acceleration of Diagnostics program, for example, worked to speed the development of point-of-care and laboratory diagnostics for COVID-19 (NIH, 2021). Such partnerships can make faster action possible and limit redundancy of effort, especially when political will for action is high.
There is some indication that political will for action against antimicrobial resistance has reached a tipping point. A recent statement from finance ministers and central bank governors of the G7 countries pledged to “work together with our health colleagues … including with industry, to explore proposals for strengthening market incentives for antibiotic drug development” (USDT, 2021). The analogous meeting for health ministers produced a statement about cooperation in the implementation of clinical trials for therapeutics and vaccines (G7 Research Group, 2021). The health ministers commented on the need for internationally coordinated testing and the sharing of test materials in response to “pandemic threats” (G7 Research Group, 2021). Their statements are evidence of growing global commitment to cooperation on product development.
The international product development partnerships put in place for COVID-19 have transferrable elements especially relevant to product development for other infectious threats. This is the ideal framework upon which to build a coordinated product development partnership for antimicrobial resistance. A partnership of this scale could help make the U.S. investment in antimicrobial resistance more of a One Health effort, with coordinated action on the human, animal, and environmental fronts.
Recommendation 6-5: The Department of Health and Human Services should establish a public–private partnership similar to ACTIV for antimicrobial resistance, bringing together the Biomedical Advanced Research and Development Authority, the
National Institutes of Health, the U.S. Department of Agriculture, the Environmental Protection Agency, and the Department of Defense and interested academic, industry, and nonprofit organizations. The partnership would have working groups on diagnostics, alternatives to antibiotics, and prevention, with a goal of supporting a diversified and balanced portfolio of tools to reduce antimicrobial resistance using a One Health approach.
Public–private partnerships are well suited to medical product development, as Figure 6-12 illustrates. Such partnerships are able to draw on a range of needed expertise and have the benefit of a relatively long time horizon. These types of collaborations are well known in antimicrobial resistance. The CARB-X public–private partnership discussed earlier in this chapter is clearly important to development and market shaping for new medical products. A public–private partnership was able to bring the Xpert MTB/Rif assay, a rapid diagnostic for rifampicin-resistant tuberculosis, to
SOURCE: Andrew M. Davis, Ola Engkvist, Rebecca J. Fairclough, Isabella Feierberg, Adrian Freeman, and Preeti Iyer, Public-private partnerships: Compound and data sharing in drug discovery and development, SLAS Discovery (vol. 26, no. 5) pp. 604-619, copyright © 2021 by SAGE Publications. Reprinted by permission of SAGE Publications.
116 countries with a high burden of tuberculosis (Albert et al., 2016; Bill & Melinda Gates Foundation, 2012; CDC, 2016). Another public–private partnership, the Foundation for Innovative Diagnostics, works with various governments and private-sector partners to develop diagnostic tests and bring them to market in low- and middle-income countries (FIND, 2021b). The foundation’s road map emphasizes developing diagnostics for resistant infections, particularly those that cause gonorrhea and chlamydia, as well as neonatal sepsis and severe infections in hospitals (FIND, 2021a).
The committee is not suggesting that the government replicate these efforts. Rather, the needed partnership would complement these and other programs from development banks, multilaterals, and various regional cooperatives (Okeke et al., 2011). As with ACTIV, this would require multisite, multi-arm trials of different medical products simultaneously in different countries (Murray et al., 2021). This kind of coordinated global attention will be essential in trials on resistant pathogens, as the major burden of these infections is in low- and middle-income countries.
There is also already good evidence that the private sector would be amenable to joining the type of partnership suggested. The AMR Industry Alliance (2019), for example, is a 5-year old coalition of private-sector partners working on solutions to prevent and mitigate antimicrobial resistance. This coalition includes generic and innovator pharmaceutical companies as well as diagnostics companies and small biotechnology firms (AMR Industry Alliance, 2019). Such collaboration is essential to One Health progress. For example, reducing manufacturing discharge has been an AMR Industry Alliance priority, and something its members have collaborated on to identify best practices (AMR Industry Alliance, 2019). The AMR Industry Alliance progress report gives attention to access, especially access to affordable medicines and diagnostics in low- and middle-income countries, as well as access to vaccines and preventive products (AMR Industry Alliance, 2020). The Health for Animals industry coalition has a similar commitment to the development of animal health vaccines, medicines, immune boosters, and development of best practices (Health for Animals, 2017). These industry coalitions would be good targets for inclusion among the proposed partnership’s private-sector contributors.
The ACTIV model, with its collaborative working groups and strategies for streamlined trials, is the best model to coordinate our national investment in antimicrobial resistance (Collins and Stoffels, 2020). The model is also helpful in avoiding duplication of effort both within the United States and internationally. It is easier for one large collaborative body to work in close connection with counterpart organizations in other parts of the world, including multilaterals and foundations (Collins and Stoffels, 2020). This is an area where the NIH and BARDA have
developed considerable expertise over the last 18 months, and one that could be adapted to speed the development of needed medical products to fight antimicrobial resistance. The NIH and BARDA also have valuable relationships with foreign research efforts, including the European Commission’s Joint Programming Initiative on Antimicrobial Resistance (JPIAMR, 2021a,b). JPIAMR (2021a,b) aims to coordinate research and encourage collaborative action against antimicrobial resistance among its member countries, mostly in Europe, and internationally. Continuing and building on such collaborative relationships would be an important role for the partnership envisioned in this recommendation.
There is also a need to balance investments in antimicrobial resistance across new medicines, diagnostics, and preventive products. Some products have considerable market potential that the private sector will recognize; not all products need the same level of government investment in development. Determining the right balance of investments across product types is challenging and would benefit from explicit public discussion of the sort a prominent public–private partnership could engender. BARDA and the NIH have experience managing this discussion as they have perspective on what society’s relative investment is in small-molecule therapeutics. The Department of Defense (DOD) would also provide valuable perspective, drawing from its experience in medicines, diagnostics, and vaccines to treat infectious diseases (USAMRIID, 2021). The DOD has experience with some of the nontraditional therapies for resistant infections. Phage therapy, for example, has received relatively greater attention in military medicine than in other practice settings, including recent funding for clinical trials of phage therapy (Clevenger, 2020; Gelman et al., 2018; Trudil, 2015).
It will also be important to the One Health orientation of this strategy to include the Environmental Protection Agency (EPA) and the U.S. Department of Agriculture (USDA) among the partnership’s convening agencies. The EPA has programs to fund small business to produce innovative technologies for use in environmental monitoring, water remediation, and viral decontamination; it also provides for cooperative research and development agreements and technology transfer (EPA, 2021). It would also be important to involve the EPA in the development of any product that might be used on crops or in water. Similarly, USDA (2014) has an agency action plan on antimicrobial resistance that calls for the development of alternatives to antimicrobials and other mitigating technologies. When the discussion turns to the optimal balance of spending across a range of innovative products, the involvement of government experts from the range of One Health disciplines will be crucial.
Global, coordinated efforts were helpful in streamlining supply chains and procurement for COVID-19 diagnostics (Peplow, 2020; The
Rockefeller Foundation, 2020). There were transferable lessons learned in responding to COVID-19 that would apply to the problem of antimicrobial resistance. For example, rapid portable tests that do not rely on laboratory infrastructure similar to those developed for COVID-19 would be essential for fighting infections in low- and middle-income countries (WHO, 2020).
A global, cooperative approach to product development could also have the advantage of easing regulatory review. This could be important for animal preventive products for which the regulatory barriers among countries can vary widely, and for alternative therapies (e.g., bacteriophages) that may require novel regulatory review (Czaplewski et al., 2016; Hauser et al., 2016; Nwokoro et al., 2016; Rex et al., 2014). Licensing and deploying new diagnostics are also serious challenges in low- and middle-income countries, where the regulatory systems and product distribution chains are not necessarily designed to handle these products (Peeling and Mabey, 2010). The ACTIV partnership had strategies to speed regulatory review across its international collaborating centers, mainly by sharing regulator’s questions and coordinating regulatory submissions (Murray et al., 2021). The experience with COVID-19 prompted a statement from the International Coalition of Medicines Regulatory Authorities (ICMRA, 2020) stating its commitment to aligning regulatory requirements and collaborating on accelerated approvals, taking advantage of the opportunity “to advance regulatory understanding and convergence.”
REFERENCES
Aagaard, H., R. Malpani, and A. Zorzet. 2021. Ensuring sustainable access to effective antibiotics for everyone, everywhere—how to address the global crisis in antibiotic research and development.” ReAct - Action on Antibiotic Resistance. https://www.reactgroup.org/wp-content/uploads/2021/09/ReAct-Report-Ensuring-sustainable-access-to-effectiveantibiotics-for-everyone-everywhere-How-to-address-the-global-crisis-in-antibiotic-Research-and-Development-March-2021.pdf (accessed June 3, 2021).
Aitken, M., and M. Kleinrock. 2017. Lifetime trends in biopharmaceutical innovation—recent evidence and implications. Parsippany, NJ: QuintilesIMS Institute.
Albert, H., R. R. Nathavitharana, C. Isaacs, M. Pai, C. M. Denkinger, and C. C. Boehme. 2016. Development, roll-out and impact of xpert mtb/rif for tuberculosis: What lessons have we learnt and how can we do better? European Respiratory Journal 48(2):516-525.
Albrecht, M. 2018. Incentivizing antibacterial research and development. https://www.medicalcountermeasures.gov/BARDA/Documents/BID2018_Presentations/Albrecht_BID18_Antibacterials.pdf (accessed October 29, 2020).
Albrecht, M. 2020. BARDA’s achievements under the national actionplan. Committee on the Long-Term Health and Economic Effects of Antimicrobial Resistance in the United States, Webinar. January 5, 2021. https://www.nationalacademies.org/event/01-05-2021/examining-the-long-term-health-and-economic-effects-of-antimicrobialresistance-in-the-united-states-meeting-3.
Albrecht, M., and J. Swenson. 2020. BARDA’s reflections on antibiotic resistance and the path forward. ASPR Blog: Office of the Assistant Secretary for Preparedness and Response. https://www.phe.gov/ASPRBlog/pages/BlogArticlePage.aspx?PostID=399 (accessed June 24, 2021).
Alm, R. A., and K. Gallant. 2020. Innovation in antimicrobial resistance: The CARB-X perspective. ACS Infectious Diseases 6(6):1317-1322.
AMR Action Fund. 2020. New AMR Action Fund worldwide launch—press release. AMR Action Fund. https://www.amractionfund.com/resource/new-amr-action-fund-worldwide-launch-press-release (accessed September 22, 2021).
AMR Action Fund. 2021. Our investments. https://amractionfund.com/ourinvestments/#page-section-0 (accessed September 22, 2021).
AMR Industry Alliance. 2019. Introducing the AMR industry alliance. https://www.amrindustryalliance.org/wp-content/uploads/2019/03/AMR_Alliance_Membership_Brochure.pdf (accessed September 22, 2021).
AMR Industry Alliance. 2020. 2020 progress report executive summary. AMR Industry Alliance. https://www.amrindustryalliance.org/progress-report (accessed July 13, 2021).
Anderson, E. J., M. A. Daugherty, L. K. Pickering, W. A. Orenstein, and R. Yogev. 2018. Protecting the community through child vaccination. Clinical Infectious Diseases 67(3):464-471.
Årdal, C., D. Findlay, M. Savic, Y. Carmeli, I. Gyssens, R. Laxminarayan, K. Outterson, and J. H. Rex. 2018. Revitalizing the antibiotic pipeline—stimulating innovation while driving sustainable use and global access. Drive-AB. http://drive-ab.eu/wp-content/uploads/2018/01/CHHJ5467-Drive-AB-Main-Report-180319-WEB.pdf (accessed June 3, 2021).
Årdal, C., M. Balasegaram, R. Laxminarayan, D. McAdams, K. Outterson, J. H. Rex, and N. Sumpradit. 2020. Antibiotic development—economic, regulatory and societal challenges. Nature Reviews: Microbiology 18(5):267-274.
ASTHO (Association of State and Territorial Health Officials). 2021. Project bioshield act fact sheet. https://www.astho.org/Programs/Preparedness/Public-Health-EmergencyLaw/Emergency-Use-Authorization-Toolkit/Project-BioShield-Act-Fact-Sheet (accessed June 11, 2021).
Balasegaram, M. 2021. Global antibiotic research and development partnership (GARDP): A model for public-health driven R&D and access. Committee on the Long-Term Health and Economic Effects of Antimirobial Resistance in the United States, March 16, 2021, Webinar. March 16, 2021. https://www.nationalacademies.org/event/03-16-2021/examining-the-long-term-health-and-economic-effects-of-antimicrobial-resistance-in-the-united-states-meeting-4.
Baptista, P. V., M. P. McCusker, A. Carvalho, D. A. Ferreira, N. M. Mohan, M. Martins, and A. R. Fernandes. 2018. Nano-strategies to fight multidrug resistant bacteria—“a battle of the titans.” Frontiers in Microbiology 9:1441.
Bassetti, M., R. Echols, Y. Matsunaga, M. Ariyasu, Y. Doi, R. Ferrer, T. P. Lodise, T. Naas, Y. Niki, D. L. Paterson, S. Portsmouth, J. Torre-Cisneros, K. Toyoizumi, R. G. Wunderink, and T. D. Nagata. 2021. Efficacy and safety of cefiderocol or best available therapy for the treatment of serious infections caused by carbapenem-resistant gram-negative bacteria (credible-cr): A randomised, open-label, multicentre, pathogen-focused, descriptive, phase 3 trial. Lancet Infectious Diseases 21(2):226-240.
Benkova, M., O. Soukup, and J. Marek. 2020. Antimicrobial susceptibility testing: Currently used methods and devices and the near future in clinical practice. Journal of Applied Microbiology 129(4):806-822.
Berger, C., A. Sertkaya, A. Jessup, C. Sullivan, A. Kolbe, Eastern Research Group Inc., and HHS Office of Inspector General. 2021. Integrating existing metrics to assess comparative real-world clinical effectiveness of recently approved antimicrobial drugs. Washington, DC: Academy Health Annual Research Meeting.
Berndt, E. R., D. Nass, M. Kleinrock, and M. Aitken. 2015. Decline in economic returns from new drugs raises questions about sustaining innovations. Health Affairs 34(2):245-252.
Beyer, P., T. B. Cueni, J. Knox, and F. Riedl. 2020. A research and development fund for new treatments for bacterial infections. Bulletin of the World Health Organization 98(12):822.
Bhavnani, S. M., K. M. Krause, and P. G. Ambrose. 2020. A broken antibiotic market: Review of strategies to incentivize drug development. Open Forum Infectious Diseases 7(7):ofaa083.
Bill & Melinda Gates Foundation. 2012. Public-private partnership announces immediate 40 percent cost reduction for rapid TB test. https://www.gatesfoundation.org/ideas/mediacenter/press-releases/2012/08/publicprivate-partnership-announces-immediate-40-percent-cost-reduction-for-rapid-tb-test (accessed September 15, 2021).
Billington, J. K. 2015. The ABCs of the US broad spectrum antimicrobials program: Antibiotics, biosecurity, and Congress. Health Security 13(6):349-354.
Bleicher, K. H., H. J. Bohm, K. Muller, and A. I. Alanine. 2003. Hit and lead generation: Beyond high-throughput screening. Nature Reviews Drug Discovery 2(5):369-378.
Bloom, D. E., S. Black, D. Salisbury, and R. Rappuoli. 2018. Antimicrobial resistance and the role of vaccines. Proceedings of the National Academy of Sciences of the United States of America 115(51):12868-12871.
Boucher, H. W., P. G. Ambrose, H. F. Chambers, R. H. Ebright, A. Jezek, B. E. Murray, J. G. Newland, B. Ostrowsky, J. H. Rex, and Infectious Diseases Society of America. 2017. White paper: Developing antimicrobial drugs for resistant pathogens, narrow-spectrum indications, and unmet needs. Journal of Infectious Diseases 216(2):228-236.
Bowden, R., and C. Burbick. 2020. Learning about veterinary ast from lady glittersparkles. https://clsi.org/about/blog/learning-about-veterinary-ast-from-lady-glittersparkles (accessed September 16, 2021).
Brasso, B. 2017. Industry perspective on the challenges of ast methods and the role of STMA. Public Workshop—Antimicrobial Susceptibility and Resistance: Addressing Challenges of Diagnostic Devices, September 13, 2017, Silver Spring, MD. https://www.fda.gov/media/100293/download (accessed June 4, 2021).
Brozak, S. 2018. An unlikely biotech investor: The government. https://www.forbes.com/sites/stephenbrozak/2018/06/08/merck-and-achaogen-two-companies-working-withbarda-to-fight-emerging-health-threats/?sh=3b5762e64fd0 (accessed June 10, 2021).
Buckley, B. S., N. Henschke, H. Bergman, B. Skidmore, E. J. Klemm, G. Villanueva, C. Garritty, and M. Paul. 2019. Impact of vaccination on antibiotic usage: A systematic review and meta-analysis. Clinical Microbiology and Infection 25(10):1213-1225.
Buckmon, K. 2020. BARDA’s reflections on antibiotic resistance and the path forward. In ASPR Blog: Medical Countermeasures. https://www.phe.gov/ASPRBlog/Lists/Posts/Post.aspx?ID=399 (accessed June 10, 2021).
Burnham, C. D., J. Leeds, P. Nordmann, J. O’Grady, and J. Patel. 2017. Diagnosing antimicrobial resistance. Nature Reviews: Microbiology 15(11):697-703.
BusinessWire. 2017. Pfizer launches Zavicefta™ (ceftazidime-avibactam) in the U.K. and Germany, a new antibiotic to treat complicated infections caused by gram-negative bacteria. BusinessWire. https://www.businesswire.com/news/home/20170314005570/en/Pfizer-Launches-Zavicefta%E2%84%A2-ceftazidime-avibactam-in-the-U.K.-and-Germany-a-New-Antibiotic-to-Treat-Complicated-Infections-Caused-by-Gram-Negative-Bacteria (accessed December 29, 2021).
CARB-X (Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator). 2020. Antibiotic resistance: A global health preparedness crisis. Boston, Massachusetts: CARB-X.
CARB-X. 2021a. About CARB-X. https://carb-x.org/about/overview (accessed June 10, 2021).
CARB-X. 2021b. Funding partners. https://carb-x.org/partners/funding-partners (accessed June 10, 2021).
CARB-X. 2021c. Portfolio-product developers. https://carb-x.org/portfolio/portfolio-companies (accessed June 28, 2021).
CARB-X. 2021d. Stewardship & access plan (SAP) development guide. https://carb-x.org/about/stewardship-and-access (accessed June 10, 2021).
CARB-X. 2021e. Stewardship and access. https://carb-x.org/about/stewardship-and-access (accessed June 10, 2021).
Carpenter, D., and B. Brasso. 2016. How to develop & obtain regulatory approval of commercial antimicrobial susceptibility tests in a timely manner. Public Workshop—Coordinated Development of Antimicrobial Drugs and Antimicrobial Susceptibility Test Devices, September 29, 2016, Silver Spring, MD. https://www.fda.gov/media/100293/download (accessed July 13, 2021).
Carroll, J. 2019. Once picked as a $500m winner, bankrupt achaogen auctions off its antibiotic for a fraction of that. https://endpts.com/once-picked-as-a-500m-winner-bankrupt-achaogen-auctions-off-its-antibiotic-for-a-fraction-of-that (accessed June 10, 2021).
CDC (Centers for Disease Control and Prevention). 2016. A new tool to diagnose tuberculosis: The Xpert MTB/RIF assay. https://www.cdc.gov/tb/publications/factsheets/testing/xpert_mtb-rif.htm (accessed September 22, 2021).
CDC. 2018. Clinical Laboratory Improvement Amendments (CLIA). https://www.cdc.gov/clia/about.html (accessed July 1, 2021).
CDC. 2019a. Antibiotic resistance threats in the United States, 2019. Centers for Disease Control and Prevention, Department of Health and Human Services. https://stacks.cdc.gov/view/cdc/82532 (accessed April 8, 2021).
CDC. 2019b. General information about candida auris. https://www.cdc.gov/fungal/candidaauris/candida-auris-qanda.html (accessed June 10, 2021).
CDC. 2020a. Antibiotic Resistance Isolate Bank. https://www.cdc.gov/drugresistance/resistance-bank/index.html (accessed July 1, 2021).
CDC. 2020b. CDC—budget request overview: FY20 president’s budget request | $6.594 billion. Centers for Disease Control and Prevention. https://www.cdc.gov/budget/documents/fy2020/cdc-overview-factsheet.pdf (accessed June 3, 2021).
CDC. 2021a. About exast. https://www.cdc.gov/drugresistance/laboratories/AR-labnetwork-testing-details/expanded-ast.html (accessed June 29, 2021).
CDC. 2021b. Antibiotic resistance investment map. https://arinvestments.cdc.gov (accessed June 29, 2021).
CDC. 2021c. Carbapenem-resistant enterobacteriaceae (CRE): An urgent public health threat. https://arpsp.cdc.gov/story/cre-urgent-public-health-threat (accessed June 29, 2021).
CDC. 2021d. Expanded antimicrobial susceptibility testing for hard-to-treat infections (ExAST). https://www.cdc.gov/drugresistance/laboratories/AR-lab-network-testing-details/expanded-ast.html (accessed June 28, 2021).
Chae, C. 2016. Porcine respiratory disease complex: Interaction of vaccination and porcine circovirus type 2, porcine reproductive and respiratory syndrome virus, and mycoplasma hyopneumoniae. Veterinary Journal 212:1-6.
Chamorro, M. F., and R. A. Palomares. 2020. Bovine respiratory disease vaccination against viral pathogens: Modified-live versus inactivated antigen vaccines, intranasal versus parenteral, what is the evidence? Veterinary Clinics of North America: Food Animal Practice 36(2):461-472.
Chapman, R. 2020. It’s time to fix the antibiotic market. https://wellcome.org/what-we-do/our-work/drug-resistant-infections/its-time-fix-broken-antibiotics-market (accessed June 10, 2021).
Chauhan, A., and R. Singh. 2018. Probiotics in aquaculture: A promising emerging alternative approach. Symbiosis 77(2):99-113.
Chiller, T. 2017. The unexpected and troubling rise of candida auris. https://www.medscape.com/viewarticle/884470 (accessed June 10, 2021).
Clevenger, T. 2020. Adaptive phage therapeutics awarded initial contract with Department of Defense. https://www.aphage.com/adaptive-phage-therapeutics-awarded-initial-contract-with-department-of-defense-2 (accessed September 21, 2021).
Clift, C., U. Gopinathan, C. M. Morel, K. Outterson, J.-A. Røttingen, and A. So. 2015. Towards a new global business model for antibiotics: Delinking revenues from sales. London, England: Chatham House.
CLSI (Clinical and Laboratory Standards Institute). 2021. Performance standards for antimicrobial susceptibility testing. https://clsi.org/media/3481/m100ed30_sample.pdf (accessed June 29, 2021).
CO-ADD (Community for Open Antimicrobial Drug Discovery). Sending compound library for high throughput screening. https://co-add.org/content/sending-compound-library-for-high-throughput-screening (accessed June 10, 2021).
Coller, S. 2006. Office of Public Health Emergency Preparedness; draft HHS Public Health Emergency Medical Countermeasures Enterprise (PHEMCE) strategy for chemical, biological, radiological and nuclear (CBRN) threats. Federal Register. 71 FR 53097. Washington, DC: Department of Health and Human Services, Office of Public Health Emergency Preparedness.
Collins, F. S., and P. Stoffels. 2020. Accelerating COVID-19 therapeutic interventions and vaccines (ACTIV): An unprecedented partnership for unprecedented times. JAMA 323(24):2455-2457.
Cortese, D. A., N. Landman, and R. K. Smoldt. 2018. Making bundled payments work: Leveraging the CMS DRG experience. NEJM Catalyst 4(3). https://catalyst.nejm.org/doi/full/10.1056/CAT.18.0179 (accessed June 3, 2021).
Coukell, A. 2019. (DISARM) act of 2019 letter—The Pew Charitable Trusts. https://www.pewtrusts.org/-/media/assets/2019/08/08-01-19-disarm-support-letter.pdf (accessed June 11, 2021).
Council of Canadian Academies. 2019. When antibiotics fail. Ottawa, Canada: The Expert Panel on the Potential Socio-Economic Impacts of Antimicrobial Resistance in Canada, Council of Canadian Academies.
CPMP (Committee for Proprietary Medicinal Products). 2000. Points to consider on switching between superiority and non-inferiority. London, England: The European Agency for the Evaluation of Medicial Products.
Crunchbase. n.d. List of Achaogen’s 2 lead investors. https://www.crunchbase.com/search/principal.investors/field/organizations/num_lead_investors/achaogen (accessed June 10, 2021).
Cunha, B. R. d., L. Fonseca, and C. Calado. 2019. Antibiotic discovery: Where have we come from, where do we go? Antibiotics (Basel) 8(2):45. https://doi.org/10.3390/antibiotics8020045.
Cunningham, M., M. A. Azcarate-Peril, A. Barnard, V. Benoit, R. Grimaldi, D. Guyonnet, H. D. Holscher, K. Hunter, S. Manurung, D. Obis, M. I. Petrova, R. E. Steinert, K. S. Swanson, D. van Sinderen, J. Vulevic, and G. R. Gibson. 2021. Shaping the future of probiotics and prebiotics. Trends in Microbiology 29(8):667-685.
Czaplewski, L., R. Bax, M. Clokie, M. Dawson, H. Fairhead, V. A. Fischetti, S. Foster, B. F. Gilmore, R. E. Hancock, D. Harper, I. R. Henderson, K. Hilpert, B. V. Jones, A. Kadioglu, D. Knowles, S. Olafsdottir, D. Payne, S. Projan, S. Shaunak, J. Silverman, C. M. Thomas, T. J. Trust, P. Warn, and J. H. Rex. 2016. Alternatives to antibiotics-a pipeline portfolio review. Lancet Infectious Diseases 16(2):239-251.
Dall, C. 2019. Medicare payment changes aim to boost antibiotic development. https://www.cidrap.umn.edu/news-perspective/2019/08/medicare-payment-changes-aim-boost-antibiotic-development (accessed August 5, 2020).
Dall, C. 2020. New pharma-led fund aims to boost antibiotic development. https://www.cidrap.umn.edu/news-perspective/2020/07/new-pharma-led-fund-aims-boost-antibiotic-development (accessed June 11, 2021).
Daniel, G. W., H. Colvin, and S. Mayer. 2013. Antibiotic development and market failure: No quick fix. https://www.brookings.edu/blog/up-front/2013/09/20/antibiotic-development-and-market-failure-no-quick-fix (accessed June 3, 2021).
Daniel, G. W., M. Schneider, M. H. Lopez, and M. B. McClellan. 2018. Implementation of a market entry reward within the United States. Journal of Law, Medicine and Ethics 46(1 Suppl):50-58.
Daniel, G. W., M. B. McClellan, M. Schneider, J. Qian, G. Lavezzari, and E. D. Graffenreid. 2019. Value-based strategies for encouraging new development of antimicrobial drugs. Washington, DC: Duke Margolis Center for Health Policy.
Darrow, J. J., and A. S. Kesselheim. 2020. Incentivizing antibiotic development: Why isn’t the generating antibiotic incentives now (GAIN) act working? Open Forum Infectious Diseases 7(1):ofaa001.
Davani-Davari, D., M. Negahdaripour, I. Karimzadeh, M. Seifan, M. Mohkam, S. J. Masoumi, A. Berenjian, and Y. Ghasemi. 2019. Prebiotics: Definition, types, sources, mechanisms, and clinical applications. Foods 8(3)92.
Davis, A. M., O. Engkvist, R. J. Fairclough, I. Feierberg, A. Freeman, and P. Iyer. 2021. Public-private partnerships: Compound and data sharing in drug discovery and development. SLAS Discovery 26(5):604-619.
Day, R. L., A. J. Harper, R. M. Woods, O. G. Davies, and L. M. Heaney. 2019. Probiotics: Current landscape and future horizons. Future Science OA 5(4):FSO391.
de Kraker, M. E., A. J. Stewardson, and S. Harbarth. 2016. Will 10 million people die a year due to antimicrobial resistance by 2050? PLoS Medicine 13(11):e1002184.
de Souza Vandenberghe, L. P., L. M. B. Garcia, C. Rodrigues, M. C. Camara, G. V. de Melo Pereira, J. de Oliveira, and C. R. Soccol. 2017. Potential applications of plant probiotic microorganisms in agriculture and forestry. AIMS Microbiology 3(3):629-648.
Desselle, M. R., R. Neale, K. A. Hansford, J. Zuegg, A. G. Elliott, M. A. Cooper, and M. A. Blaskovich. 2017. Institutional profile: Community for open antimicrobial drug discovery—crowdsourcing new antibiotics and antifungals. Future Science OA 3(2):FSO171.
DNDi (Drugs for Neglected Diseases Initiative). 2021. Registration & access. https://dndi.org/research-development/registration-and-access (accessed July 13, 2021).
Drago, L., V. Rodighiero, T. Celeste, L. Rovetto, and E. De Vecchi. 2010. Microbiological evaluation of commercial probiotic products available in the USA in 2009. Journal of Chemotherapy 22(6):373-377.
D’Souza, A. W., M. Boolchandani, S. Patel, G. Galazzo, J. M. van Hattem, M. S. Arcilla, D. C. Melles, M. D. de Jong, C. Schultsz, C. Consortium, G. Dantas, and J. Penders. 2021. Destination shapes antibiotic resistance gene acquisitions, abundance increases, and diversity changes in Dutch travelers. Genome Medicine 13(1):79.
Dunn, D. T., A. J. Copas, and P. Brocklehurst. 2018. Superiority and non-inferiority: Two sides of the same coin? Trials 19(1):499.
Dutescu, I. A., and S. A. Hillier. 2021. Encouraging the development of new antibiotics: Are financial incentives the right way forward? A systematic review and case study. Infection and Drug Resistance 14:415-434.
Eichberg, M. J. 2015. Public funding of clinical-stage antibiotic development in the United States and European Union. Health Security 13(3):156-165.
EPA (Environmental Protection Agency). 2021. EPA’s SBIR and homeland security programs: A decade of success. https://www.epa.gov/sbir/epas-sbir-and-homeland-security-programs-decade-success (accessed July 13, 2021).
Erlacher-Vindel, E. 2019. Prioritization of vaccines to reduce antibiotic use in animals. PACCARB Meeting, Washington, DC. https://www.hhs.gov/sites/default/files/erlacher-vindel-paccarb.pdf (accessed May 24, 2021).
FAO (Food and Agriculture Organization of the United Nations). 2016. Probiotics in animal nutrition—production, impact and regulation. Rome, Italy: Food and Agriculture Organization of the United Nations.
FAO and WHO (World Health Organization). 2002. Guidelines for the evaluation of probiotics in food. Geneva, Switzerland: World Health Organization.
FDA (Food and Drug Administration). 2017. Antifungal susceptibility test interpretive criteria. https://www.fda.gov/drugs/development-resources/antifungal-susceptibility-test-interpretive-criteria (accessed June 4, 2021).
FDA. 2018a. 80 years of the Federal Food, Drug, and Cosmetic Act. https://www.fda.gov/about-fda/fda-history-exhibits/80-years-federal-food-drug-and-cosmetic-act (accessed June 4, 2021).
FDA. 2018b. Antimicrobial susceptibility test (AST) systems: Class II special controls guidance for industry and FDA. https://www.fda.gov/medical-devices/guidance-documentsmedical-devices-and-radiation-emitting-products/antimicrobial-susceptibility-test-astsystems-class-ii-special-controls-guidance-industry-and-fda (accessed June 4, 2021).
FDA. 2018c. Companion diagnostics. https://www.fda.gov/medical-devices/in-vitrodiagnostics/companion-diagnostics (accessed June 4, 2021).
FDA. 2018d. FDA in brief: FDA reports on its progress advancing policies for developing next generation antibiotics. https://www.fda.gov/news-events/fda-brief/fda-brief-fda-reports-its-progress-advancing-policies-developing-next-generation-antibiotics (accessed June 4, 2021).
FDA. 2019a. Coordinated development of antimicrobial drugs and antimicrobial susceptibility test devices: Guidance for industry and food and drug administration staff. Washington, DC: Center for Devices and Radiological Health and Center for Drug Evaluation and Research, Food and Drug Administration, Department of Health and Human Services.
FDA. 2019b. Generally recognized as safe (GRAS). https://www.fda.gov/food/food-ingredientspackaging/generally-recognized-safe-gras (accessed).
FDA. 2020a. 21st Century Cures Act. https://www.fda.gov/regulatory-information/selectedamendments-fdc-act/21st-century-cures-act (accessed June 4, 2021).
FDA. 2020b. Clinical Laboratory Improvement Amendments (CLIA). https://www.fda.gov/medical-devices/ivd-regulatory-assistance/clinical-laboratory-improvementamendments-clia (accessed July 1, 2021).
FDA. 2020c. Fact sheet: FDA at a glance. https://www.fda.gov/about-fda/fda-basics/fact-sheet-fda-glance (accessed June 3, 2021).
FDA. 2020d. Limited population pathway for antibacterial and antifungal drugs—the LPAD pathway. https://www.fda.gov/drugs/development-resources/limited-population-pathway-antibacterial-and-antifungal-drugs-lpad-pathway (accessed June 11, 2020).
FDA. 2020e. Meropenem—injection products. https://www.fda.gov/drugs/developmentresources/meropenem-injection-products (accessed June 4, 2021).
FDA. 2021a. FDA-recognized antimicrobial susceptibility test interpretive criteria. https://www.fda.gov/drugs/development-resources/fda-recognized-antimicrobial-susceptibility-test-interpretive-criteria (accessed June 4, 2021).
FDA. 2021b. In vitro diagnostics EUAs. https://www.fda.gov/medical-devices/coronavirusdisease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitrodiagnostics-euas (accessed June 4, 2021).
FDA. 2021c. Orange book: Approved drug products with therapeutic equivalence evaluations. https://www.accessdata.fda.gov/scripts/cder/ob/index.cfm (accessed June 29, 2021).
Fenster, K., B. Freeburg, C. Hollard, C. Wong, R. Ronhave Laursen, and A. C. Ouwehand. 2019. The production and delivery of probiotics: A review of a practical approach. Microorganisms 7(3):83.
FIND. 2021a. Antimicrobial resistance. https://www.finddx.org/amr (accessed June 28, 2021). FIND. 2021b. Partners & donors. https://www.finddx.org/partners-donors (accessed June 28, 2021).
Fitzpatrick, M. A. 2020. Real-world antibiotic needs for resistant gram-negative infections. Lancet Infectious Diseases 20(10):1108-1109.
Fogel, D. B. 2018. Factors associated with clinical trials that fail and opportunities for improving the likelihood of success: A review. Contemporary Clinical Trials Communications 11:156-164.
Frost, I., T. P. Van Boeckel, J. Pires, J. Craig, and R. Laxminarayan. 2019. Global geographic trends in antimicrobial resistance: The role of international travel. Journal of Travel Medicine 26(8)1–13.
G7 Research Group. 2021. G7 therapeutics and vaccines clinical trials charter. http://www.g7.utoronto.ca/healthmins/G7-clinical-trials-charter.pdf (accessed June 29, 2021).
GAO (Government Accountability Office). 2020. Antibiotic resistance: Additional federal actions needed to better determine magnitude and reduce impact. Washington, DC: Government Accountability Office.
GARDP (Global Antibiotic Research and Development Partnership). 2021a. About GARDP. https://gardp.org/who-we-are/about-gardp (accessed July 13, 2021).
GARDP. 2021b. Access & stewardship. https://dndi.org/research-development/registration-and-access (accessed July 13, 2021).
Gatti, M., M. Giannella, L. Scudeller, and P. Viale. 2020. Adaptive trials of new antimicrobials for infections with carbapenem-resistant, gram-negative bacteria. Lancet Infectious Diseases 20(1):24.
GCOA and IDSA (Global Coalition on Aging and Infectious Diseases Society of America). 2021. 2021 AMR preparedness index. https://globalcoalitiononaging.com/wp-content/uploads/2021/06/GCOA-AMR-Preparedness-Index_FINAL.pdf (accessed July 12, 2021).
Gelman, D., A. Eisenkraft, N. Chanishvili, D. Nachman, S. Coppenhagem Glazer, and R. Hazan. 2018. The history and promising future of phage therapy in the military service. Journal of Trauma and Acute Care Surgery 85(1S Suppl 2):S18-S26.
Ghosh, C., P. Sarkar, R. Issa, and J. Haldar. 2019. Alternatives to conventional antibiotics in the era of antimicrobial resistance. Trends in Microbiology 27(4):323-338.
Goossens, H., M. Ferech, R. Vander Stichele, M. Elseviers, and ESAC Project Group. 2005. Outpatient antibiotic use in europe and association with resistance: A cross-national database study. The Lancet 365(9459):579-587.
Gotham, D., L. Moja, M. van der Heijden, S. Paulin, I. Smith, and P. Beyer. 2021. Reimbursement models to tackle market failures for antimicrobials: Approaches taken in France, Germany, Sweden, the United Kingdom, and the United States. Health Policy 125(3):296-306.
GSA (General Services Administration). 2020. Federal procurement data system—next generation. https://www.fpds.gov/fpdsng_cms/index.php/en (accessed July 1, 2021).
Harbarth, S. 2018. Will 10 million people die a year due to antimicrobial resistance by 2050? Risk Talk on Antibiotic Resistance, Rüschlikon, Zurich, Switzerland. https://www.swissre.com/dam/jcr:bfb80df4-b8f7-43a4-b0c3-ef3b6370e2af/amr_stephan_harbarth.pdf (accessed June 3, 2021).
Hardin, L., A. Kilian, and E. Murphy. 2017. Bundled payments for care improvement: Preparing for the medical diagnosis-related groups. Journal of Nursing Administration 47(6):313-319.
Hasan, K. N., and G. Banerjee. 2020. Recent studies on probiotics as beneficial mediator in aquaculture: A review. The Journal of Basic and Applied Zoology 81(1).
Hauser, A. R., J. Mecsas, and D. T. Moir. 2016. Beyond antibiotics: New therapeutic approaches for bacterial infections. Clinical Infectious Diseases 63(1):89-95.
Health for Animals. 2017. Responsible antibiotic use: Five ways the animal health industry is tackling AMR. Health for Animals Newsletter. https://www.healthforanimals.org/resources-and-events/newsletter-repository/6-responsible-antibiotic-use.html?q=20 (accessed July 13, 2021).
Health for Animals. 2020. Our contribution. https://healthforanimals.org/roadmap/contribution.html (accessed July 13, 2021).
Henderson, D. R. 2019. Rent seeking. https://www.econlib.org/library/Enc/RentSeeking.html (accessed June 11, 2021).
HHS (Department of Health and Human Services). 2017. Recommendations for incentivizing the development of vaccines, diagnostics, and therapeutics to combat antibiotic-resistance. Washington, DC: Presidential Advisory Council on Combating Antibiotic-Resistant Bacteria.
HHS. 2019. Project bioshield overview. https://www.medicalcountermeasures.gov/barda/cbrn/project-bioshield-overview (accessed June 11, 2021).
HHS, FDA (Food and Drug Administration), CDER (Center for Drug Evaluation and Research), and CBER (Center for Biologics Evaluation and Research). 2016. Non-inferiority clinical trials to establish effectiveness guidance for industry. Washington, DC: Food and Drug Administration.
HHS, FDA, CDER, and CBER. 2019. Adaptive designs for clinical trials of drugs and biologics. Washington, DC: Center for Devices and Radiological Health and Center for Drug Evaluation and Research, Food and Drug Administration, Department of Health and Human Services.
Hillock, N. T., T. L. Merlin, J. Karnon, J. Turnidge, and J. Eliott. 2020. Feasibility of de-linking reimbursement of antimicrobials from sales: The Australian perspective as a qualitative case study. JAC Antimicrobial Resistance 2(2):dlaa023.
Hoelzer, K., L. Bielke, D. P. Blake, E. Cox, S. M. Cutting, B. Devriendt, E. Erlacher-Vindel, E. Goossens, K. Karaca, S. Lemiere, M. Metzner, M. Raicek, M. Collell Surinach, N. M. Wong, C. Gay, and F. Van Immerseel. 2018. Vaccines as alternatives to antibiotics for food producing animals. Part 1: Challenges and needs. Veterinary Research 49(1):64.
Hoseinifar, S. H., Y. Z. Sun, A. Wang, and Z. Zhou. 2018. Probiotics as means of diseases control in aquaculture, a review of current knowledge and future perspectives. Frontiers in Microbiology 9:2429.
Houchens, C., and J. Larsen. 2017. The role of the biomedical advanced research and development authority (BARDA) in promoting innovation in antibacterial product development. AMR Control August 2.
Humphries, R. M. 2018. Implementation of the 21st Century Cures Act for for breakpoints and interpretive categories. San Diego, CA: Clinical and Laboratory Standards Institute. https://clsi.org/media/2306/2018_june_ast_education_session_21st_century_cures.pdf (accessed June 4, 2021).
Humphries, R. M., and J. A. Hindler. 2016. Emerging resistance, new antimicrobial agents…. But no tests! The challenge of antimicrobial susceptibility testing in the current US regulatory landscape. Clinical Infectious Diseases 63(1):83-88.
Humphries, R. M., J. Hindler, M. Jane Ferraro, and A. Mathers. 2018a. Twenty-first century cures act and antimicrobial susceptibility testing: Clinical implications in the era of multidrug resistance. Clinical Infectious Diseases 67(7):1132-1138.
Humphries, R. M., J. A. Hindler, E. Epson, S. Horwich-Scholefield, L. G. Miller, J. Mendez, J. B. Martinez, J. Sinkowitz, D. Sinkowtiz, C. Hershey, P. Marquez, S. Bhaurla, M. Moran, L. Pandes, D. Terashita, and J. A. McKinnell. 2018b. Carbapenem-resistant enterobacteriaceae detection practices in California: What are we missing? Clinical Infectious Diseases 66(7):1061-1067.
Humphries, R. M., S. Kircher, A. Ferrell, K. M. Krause, R. Malherbe, A. Hsiung, and C. A. Burnham. 2018c. The continued value of disk diffusion for assessing antimicrobial susceptibility in clinical laboratories: Report from the Clinical and Laboratory Standards Institute methods development and standardization working group. Journal of Clinical Microbiology 56(8):e00437-18.
ICMRA (International Coalition of Medicines Regulatory Authorities). 2020. ICMRA statement on COVID-19. https://www.icmra.info/drupal/en/news/statement_on_COVID-19 (accessed September 22, 2021).
IDSA (Infectious Diseases Society of America). 2012. White paper: Recommendations on the conduct of superiority and organism-specific clinical trials of antibacterial agents for the treatment of infections caused by drug-resistant bacterial pathogens. Clinical Infectious Diseases 55(8):1031-1046.
IDSA. 2019. Disarm act provides framework needed to spur antibiotic R&D, protect existing drugs. https://www.idsociety.org/news--publications-new/articles/2019/disarm-act-provides-framework-needed-to-spur-antibiotic-rd-protect-existing-drugs (accessed June 3, 2021).
IDSA. 2021. FY 2022 budget recommendations for antimicrobial resistance programs. https://www.idsociety.org/contentassets/12ee9934b86742f8859f62945661acdc/fy2022-amrrecommendations-final.pdf (accessed June 29, 2021).
Jackson, S. A., J. L. Schoeni, C. Vegge, M. Pane, B. Stahl, M. Bradley, V. S. Goldman, P. Burguiere, J. B. Atwater, and M. E. Sanders. 2019. Improving end-user trust in the quality of commercial probiotic products. Frontiers in Microbiology 10(739):739.
Jacobs, A. 2019. Crisis looms in antibiotics as drug makers go bankrupt. https://www.nytimes.com/2019/12/25/health/antibiotics-new-resistance.html (accessed July 13, 2021).
Jit, M., D. H. L. Ng, N. Luangasanatip, F. Sandmann, K. E. Atkins, J. V. Robotham, and K. B. Pouwels. 2020. Quantifying the economic cost of antibiotic resistance and the impact of related interventions: Rapid methodological review, conceptual framework and recommendations for future studies. BMC Medicine 18(1):38.
JPIAMR (Joint Programming Initiative on Antimicrobial Resistance). n.d. About. https://www.jpiamr.eu/about (accessed June 10, 2021).
JPIAMR. 2021. JPIAMR activity report 2020. Stockholm, Sweden: Joint Programming Initiative on Antimicrobial Resistance.
Kadlec, R. 2019. Committee on Energy and Commerce Subcommittee on Oversight and Investigations—hearing on flu season: U.S. public health preparedness and response. https://docs.house.gov/meetings/IF/IF02/20191204/110278/HHRG-116-IF02-Wstate-KadlecMD-MTMHMSR-20191204-SD002.pdf (accessed December 4, 2020).
Kalelkar, P. P., M. Riddick, and A. J. García. 2021. Biomaterial-based antimicrobial therapies for the treatment of bacterial infections. Nature Reviews Materials. https://www.nature.com/articles/s41578-021-00362-4 (accessed September 30, 2021).
Kaufhold, S., R. Yaesoubi, and V. E. Pitzer. 2019. Predicting the impact of typhoid conjugate vaccines on antimicrobial resistance. Clinical Infectious Diseases 68(Suppl 2):S96-S104.
Keane, A. 2018. Plazomicin. Antimicrobial Drugs Advisory Committee Meeting, May 2, 2018, Washington, DC. https://www.fda.gov/media/113289/download (accessed June 10, 2021).
Kirienko, N. V., L. Rahme, and Y. H. Cho. 2019. Editorial: Beyond antimicrobials: Nontraditional approaches to combating multidrug-resistant bacteria. Frontiers in Cellular and Infection Microbiology 9:343.
Knisely, J. 2020. NIAID support to meet the carb challenge. Committee on the Long-Term Health and Economic Effects of Antimicrobial Resistance. Webinar. September 22-25, 2020. https://www.nationalacademies.org/event/09-22-2020/examining-the-long-term-health-and-economic-effects-of-antimicrobial-resistance-in-the-united-states-meeting-1 (accessed September 23, 2020).
Knox, J. 2020. A lifeline for antibiotic development. https://wellcome.org/news/lifeline-antibiotic-development (accessed June 10, 2021).
Krause, K. 2021a. The post-approval challenges of antimicrobial development. Committee on the Long-Term Medical And Economic Effects Of Antimicrobial Resistance: Meeting 3. Webinar. January 5-8, 2021. https://www.nationalacademies.org/event/01-05-2021/examining-the-long-term-health-and-economic-effects-of-antimicrobial-resistance-in-the-united-states-meeting-3 (accessed January 5, 2021).
Krause, K. 2021b. The relationship between antimicrobial susceptibility testing devices and antibiotic markets. Committee on the Long-Term Medical And Economic Effects Of Antimicrobial Resistance: Meeting 3. Webinar. January 5-8, 2021. https://www.nationalacademies.org/event/01-05-2021/examining-the-long-term-health-and-economic-effects-of-antimicrobial-resistance-in-the-united-states-meeting-3 (accessed January 6, 2021).
Lanini, S., J. P. A. Ioannidis, F. Vairo, M. Pletschette, G. Portella, V. Di Bari, A. Mammone, R. Pisapia, S. Merler, B. Nguhuni, M. Langer, A. Di Caro, S. J. L. Edwards, N. Petrosillo, A. Zumla, and G. Ippolito. 2019. Non-inferiority versus superiority trial design for new antibiotics in an era of high antimicrobial resistance: The case for post-marketing, adaptive randomised controlled trials. Lancet Infectious Diseases 19(12):e444-e451.
Larsen, J., and G. Disbrow. 2017. Project bioshield and the biomedical advanced research development authority: A 10-year progress report on meeting us preparedness objectives for threat agents. Clinical Infectious Diseases 64(10):1430–1434. https://doi.org/1410.1093/cid/cix1097.
Laxminarayan, R., T. Van Boeckel, and A. Teillant. 2015. The economic costs of withdrawing antimicrobial growth promoters from the livestock sector. OECD Food, Agriculture and Fisheries Papers, No. 78, Paris, France: OECD Publishing. https://doi.org/10.1787/5js64kst5wvl-en.
Ledley, F. D., S. S. McCoy, G. Vaughan, and E. G. Cleary. 2020. Profitability of large pharmaceutical companies compared with other large public companies. JAMA 323(9):834-843.
Lepore, C., and W. Kim. 2021. Opportunities in 2021 to fix the broken antibiotics market. https://globalforum.diaglobal.org/issue/january-2021/opportunities-in-2021-to-fix-the-broken-antibiotics-market (accessed June 11, 2021).
Lewnard, J. A., N. C. Lo, N. Arinaminpathy, I. Frost, and R. Laxminarayan. 2020. Childhood vaccines and antibiotic use in low- and middle-income countries. Nature 581(7806):94-99.
Li Bassi, G., J. Suen, A. G. Barnett, A. Corley, J. Millar, J. Fanning, I. Lye, S. Colombo, K. Wildi, S. Livingstone, G. Abbate, S. Hinton, B. Liquet, S. Shrapnel, H. Dalton, J. F. Fraser, and COVID-19 Critical Care Consortium Investigators. 2020. Design and rationale of the COVID-19 Critical Care Consortium international, multicentre, observational study. BMJ Open 10(12):e041417.
Lipsitch, M., and G. R. Siber. 2016. How can vaccines contribute to solving the antimicrobial resistance problem? mBio 7(3):e00428-00416.
Mahase, E. 2020. UK launches subscription style model for antibiotics to encourage new development. BMJ 369:m2468.
Martinez Cruz, P., A. L. Ibanez, O. A. Monroy Hermosillo, and H. C. Ramirez Saad. 2012. Use of probiotics in aquaculture. ISRN Microbiology 2012:916845.
McAteer, J., G. Derado, M. Hughes, A. Bhatnagar, F. Medalla, K. Chatham-Stevens, G. D. Appiah, and E. Mintz. 2020. Typhoid fever in the US pediatric population, 1999-2015: Opportunities for improvement. Clinical Infectious Diseases 73(11):e4581–e4589.
McKenna, M. 2020. The antibiotic paradox: Why companies can’t afford to create life-saving drugs. Nature 584(7821):338-341.
MedicalCountermeasures.gov. 2019a. Broad spectrum antimicrobials. https://www.medicalcountermeasures.gov/barda/cbrn/broad-spectrum-antimicrobials (accessed June 11, 2021).
MedicalCountermeasures.gov. 2019b. Project bioshield overview. https://www.medicalcountermeasures.gov/barda/cbrn/project-bioshield-overview (accessed June 11, 2021).
Mereish, K. 2018. Threats to pharmaceutical supply chains. Washington, DC: Department of Homeland Security.
Mikulic, M. 2021. World pharmaceutical market distribution by submarket 2014-2020. https://www.statista.com/statistics/266547/total-value-of-world-pharmaceutical-market-by-submarket-since-2006 (accessed June 11, 2021).
MMV (Medicines for Malaria Venture). 2021. Working with regulators to advance access to quality medicines. https://www.mmv.org/research-development/working-regulators-advance-access-quality-medicines (accessed July 13, 2021).
Moeller, G., S. Buchsbaum, P. Wilson, A. Shepherd, P. Kaspar, M. Giovanni, L. Penny, M. Urdea, P. Vickerman, C. Dye, J. Cunningham, S. Reed, C. Hanson, P. Small, M. Perkins, R. Allan, A. Magill, T. Taylor, R. Guerrant, J. Agosti, L. von Seidlein, J. Hughes, M. Miller, C. Mason, P. Tarr, C. Wilfert, K. Holmes, P. Mwaba, N. Hellmann, R. Peeling, et al. 2007. Estimating the global health impact of improved diagnostic tools for the developing world. Santa Monica, CA: RAND Corporation.
Moore, M. R., R. Link-Gelles, W. Schaffner, R. Lynfield, C. Lexau, N. M. Bennett, S. Petit, S. M. Zansky, L. H. Harrison, A. Reingold, L. Miller, K. Scherzinger, A. Thomas, M. M. Farley, E. R. Zell, T. H. Taylor, T. Pondo, L. Rodgers, L. McGee, B. Beall, J. H. Jorgensen, and C. G. Whitney. 2015. Effect of use of 13-valent pneumococcal conjugate vaccine in children on invasive pneumococcal disease in children and adults in the USA: Analysis of multisite, population-based surveillance. The Lancet Infectious Diseases 15(3):301-309.
Mossialos, E., C. M. Morel, S. Edwards, J. Berenson, M. Gemmill-Toyama, D. Brogan, and World Health Organization. 2010. Policies and incentives for promoting innovation in antibiotic research. Geneva, Switzerland: World Health Organization. Regional Office for Europe.
Mullard, A. 2020. UK outlines its antibiotic pull incentive plan. Nature Reviews Drug Discovery 19(5):298.
Murray, D. D., A. G. Babiker, J. V. Baker, C. E. Barkauskas, S. M. Brown, C. C. Chang, V. J. Davey, A. C. Gelijns, A. A. Ginde, B. Grund, E. Higgs, F. Hudson, V. L. Kan, H. C. Lane, T. A. Murray, R. Paredes, M. K. B. Parmar, S. Pett, A. N. Phillips, M. N. Polizzotto, C. Reilly, U. Sandkovsky, S. Sharma, M. Teitelbaum, B. T. Thompson, B. E. Young, J. D. Neaton, and J. D. Lundgren. 2021. Design and implementation of an international, multi-arm, multi-stage platform master protocol for trials of novel SARS-CoV-2 antiviral agents: Therapeutics for inpatients with COVID-19 (tico/activ-3). medRxiv. https://www.ncbi.nlm.nih.gov/pubmed/33215168 (accessed June 28, 2021).
NCATS GARD (National Center for Advancing Translational Sciences Genetic and Rare Diseases Information Center). 2021. FAQs about rare diseases. https://rarediseases.info.nih.gov/diseases/pages/31/faqs-about-rare-diseases (accessed June 10, 2021).
Nielsen, T. B., E. P. Brass, D. N. Gilbert, J. G. Bartlett, and B. Spellberg. 2019. Sustainable discovery and development of antibiotics—is a nonprofit approach the future? New England Journal of Medicine 381(6):503.
NIH (National Institutes of Health). 2021. Radx programs. https://www.nih.gov/researchtraining/medical-research-initiatives/radx/radx-programs (accessed September 24, 2021).
NLM (National Library of Medicine). National Database of Antibiotic Resistant Organisms (NDARO). 2021. https://www.ncbi.nlm.nih.gov/pathogens/antimicrobial-resistance (accessed June 10, 2021).
Novo Nordisk Foundation. 2018. Repair impact fund. https://www.repair-impact-fund.com/about (accessed June 11, 2021).
Nwokoro, E., R. Leach, C. Ardal, E. Baraldi, K. Ryan, and J. Plahte. 2016. An assessment of the future impact of alternative technologies on antibiotics markets. Journal of Pharmaceutical Policy and Practice 9:34.
O’Brien, M. K., and P. Chu. 2020. A market failure for antimicrobial resistant medicines. https://www.appliedclinicaltrialsonline.com/view/market-failure-antimicrobial-resistant-medicines (accessed October 12, 2021).
OECD (Organisation for Economic Co-operation and Development), WHO (World Health Organization), FAO (Food and Agriculture Organization of the United Nations), and OIE (World Organisation for Animal Health). 2017. Tackling antimicrobial resistance: Ensuring sustainable R&D. Organisation for Economic Co-operation and Development, World Health Organization, Food and Agriculture Organization of the United Nations, and World Organisation for Animal Health. https://www.oecd.org/g20/summits/hamburg/Tackling-Antimicrobial-Resistance-Ensuring-Sustainable-RD.pdf (accessed June 11, 2021).
Oehler, R. L., and S. G. Gompf. 2020. Shortcomings in the US pharmaceutical supply chain: Potential risks associated with international manufacturing and trade-related tariffs. JAMA 324(2):143-144.
OIE (World Organisation for Animal Health). 2015. Report of the meeting of the OIE ad hoc group on prioritisation of diseases for which vaccines could reduce antimicrobial use in animals, 21-23 April 2015, Paris, France. https://www.oie.int/fileadmin/SST/adhocreports/Diseases%20for%20which%20Vaccines%20could%20reduce%20Antimicrobial%20Use/AN/AHG_AMUR_Vaccines_Apr2015.pdf (accessed May 24, 2021).
OIE. 2018. Report of the meeting of the OIE ad hoc group on prioritisation of diseases for which vaccines could reduce antimicrobial use in animals, 7-9 May 2018, Paris, France. https://www.oie.int/fileadmin/SST/adhocreports/Diseases%20for%20which%20Vaccines%20could%20reduce%20Antimicrobial%20Use/AN/AHG_AMUR_Vaccines_ruminants_May2018.pdf (accessed May 24, 2021).
Okeke, I. N. 2009. Cholera vaccine will reduce antibiotic use. Science 325(5941):674.
Okeke, I. N., R. W. Peeling, H. Goossens, R. Auckenthaler, S. S. Olmsted, J. F. de Lavison, B. L. Zimmer, M. D. Perkins, and K. Nordqvist. 2011. Diagnostics as essential tools for containing antibacterial resistance. Drug Resistance Updates 14(2):95-106.
Okhravi, C., S. Callegari, S. McKeever, C. Kronlid, E. Baraldi, O. Lindahl, and F. Ciabuschi. 2018. Simulating market entry rewards for antibiotics development. Journal of Law, Medicine and Ethics 46(1 Suppl):32-42.
O’Neill, J. 2016. Tackling drug-resistant infections globally: Final report and recommendations. 2016. London, UK: HM Government and Wellcome Trust. https://wellcomecollection.org/works/thvwsuba (accessed April 8, 2021).
Outterson, K. 2019. A shot in the arm for new antibiotics. Nature Biotechnology 37(10):1110-1112.
Outterson, K., U. Gopinathan, C. Clift, A. D. So, C. M. Morel, and J. A. Rottingen. 2016. Delinking investment in antibiotic research and development from sales revenues: The challenges of transforming a promising idea into reality. PLoS Medicine 13(6):e1002043.
Outterson, K., E. S. F. Orubu, J. Rex, C. Ardal, and M. H. Zaman. 2021. Patient access in fourteen high-income countries to new antibacterials approved by the FDA, EMA, PMDA, or Health Canada, 2010-2020. Clinical Infectious Diseases. Epub ahead of print. https://doi.org/10.1093/cid/ciab612.
PACCARB (Presidential Advisory Council on Combating Antibiotic-Resistant Bacteria). 2019. Priorities for the national action plan on combating antibiotic-resistant bacteria: 2020–2025. Department of Health and Human Services. https://www.hhs.gov/sites/default/files/PACCARB%20NAP%20Report%20FINAL%20Approved%20by%20Council.pdf (accessed June 11, 2021).
Palzkill, T. 2013. Metallo-beta-lactamase structure and function. Annals of the New York Academy of Sciences 1277:91-104.
Paul, M., S. Harbarth, A. Huttner, G. E. Thwaites, U. Theuretzbacher, M. J. M. Bonten, and L. Leibovici. 2021. Investigator-initiated randomized controlled trials in infectious diseases: Better value for money for registration trials of new antimicrobials. Clinical Infectious Diseases 72(7):1259-1264.
PCAST (President’s Council of Advisors on Science and Technology). 2015. National action plan for combating antibiotic-resistant bacteria. Washington, DC: The White House.
Peeling, R. W., and D. Mabey. 2010. Point-of-care tests for diagnosing infections in the developing world. Clinical Microbiology and Infection 16(8):1062-1069.
Peplow, M. 2020. Developing countries face diagnostic challenges as the COVID-19 pandemic surges. Chemical and Engineering News. https://cen.acs.org/analytical-chemistry/diagnostics/Developing-countries-face-diagnostic-challenges/98/i27 (accessed June 29, 2021).
Perkins, M., and D. Glover. 2020. How the “NHS model” to tackle antimicrobial resistance (AMR) can set a global standard. National Health Service. https://www.england.nhs.uk/blog/how-the-nhs-model-to-tackle-antimicrobial-resistance-amr-can-set-a-global-standard (accessed June 3, 2021).
Pew. 2014. Antibiotics currently in global clinical development. https://www.pewtrusts.org/en/research-and-analysis/data-visualizations/2014/antibiotics-currently-in-clinicaldevelopment (accessed June 11, 2021).
Pew. 2021. The shared platform for antibiotic research and knowledge (SPARK). https://www.pewtrusts.org/en/research-and-analysis/articles/2018/09/21/the-shared-platform-for-antibiotic-research-and-knowledge (accessed June 10, 2021).
Plackett, B. 2020. Why big pharma has abandoned antibiotics. Nature 586(7830):S50-S52.
Renwick, M., and E. Mossialos. 2020. Fostering R&D of novel antibiotics and other technologies to prevent and treat infection. European Journal of Public Health 30(Suppl 5):ckaa165.
Renwick, M. J., D. M. Brogan, and E. Mossialos. 2016. A systematic review and critical assessment of incentive strategies for discovery and development of novel antibiotics. Journal of Antibiotics 69(2):73-88.
Rex, J., and K. Outterson. 2020. UK antibiotic subscription pilot implies pull incentive of up to $4b across the G20. https://amr.solutions/2020/03/29/uk-antibiotic-subscription-pilotimplies-pull-incentive-of-up-to-4b-across-the-g20 (accessed June 3, 2021).
Rex, J. H., M. Goldberger, B. I. Eisenstein, and C. Harney. 2014. The evolution of the regulatory framework for antibacterial agents. Annals of the New York Academy of Sciences 1323:11-21.
Rex, J. H., G. H. Talbot, M. J. Goldberger, B. I. Eisenstein, R. M. Echols, J. F. Tomayko, M. N. Dudley, and A. Dane. 2017. Progress in the fight against multidrug-resistant bacteria 2005-2016: Modern noninferiority trial designs enable antibiotic development in advance of epidemic bacterial resistance. Clinical Infectious Diseases 65(1):141-146.
Roope, L. S. J., R. D. Smith, K. B. Pouwels, J. Buchanan, L. Abel, P. Eibich, C. C. Butler, P. S. Tan, A. S. Walker, J. V. Robotham, and S. Wordsworth. 2019. The challenge of antimicrobial resistance: What economics can contribute. Science 364(6435).
Rost, L. M., M. H. Nguyen, C. J. Clancy, R. K. Shields, and E. S. Wright. 2021. Discordance among antibiotic prescription guidelines reflects a lack of clear best practices. Open Forum Infectious Diseases 8(1):ofaa571.
Rubin, D. 2016. Evaluating antibacterial drugs in unmet need settings. https://www.fda.gov/files/drugs/published/Evaluating-antibacterial-drugs-in-unmet-need-settings.pdf (accessed July 18, 2021).
Russell, P. 2007. Project bioshield: What it is, why it is needed, and its accomplishments so far. Clinical Infectious Diseases 45(Suppl 1):S68–S72. https://doi.org/10.1086/518151.
Scheftel, J., M. T. Jay-Russell, T. W. Lehenbauer, D. R. Smith, M. Papich, L. Schnabel, T. M. Parker, C. Brookshire, R. Singer, H. Cervantes, J. E. Whaley, P. S. Gaunt, A. Canon, and P. Davies. 2020. Antimicrobial resistant pathogens affecting animal health in the United States. American Veterinary Medical Association. https://www.avma.org/resources-tools/one-health/antimicrobial-use-and-antimicrobial-resistance/antimicrobial-resistantpathogens-affecting-animal-health (accessed July 13, 2021).
Schmidt, V., M. Gomez-Chiarri, C. Roy, K. Smith, and L. Amaral-Zettler. 2017. Subtle microbiome manipulation using probiotics reduces antibiotic-associated mortality in fish. mSystems 2(6):e00133-00117.
Schneider, M. 2020. New rules expand application of the NTAP program for innovative antibiotics. https://www.contagionlive.com/view/new-rules-expand-application-of-the-ntap-program-for-innovative-antibiotics (accessed April 7, 2021).
Schneider, M., G. W. Daniel, N. R. Harrison, and M. B. McClellan. 2020a. Delinking us antibiotic payments through a subscription model in Medicare. Duke-Margolis Center for Health Policy. https://healthpolicy.duke.edu/sites/default/files/2020-02/margolis_subscription_model_14jan2020.pdf (accessed June 3, 2021).
Schneider, M., N. R. Harrison, and M. B. McClellan. 2020b. Recommended post-market incentive strategies to support the development of innovative antibiotics. Duke-Margolis Center for Health Policy. https://healthpolicy.duke.edu/sites/default/files/2020-09/Recommendations%20Report%20Sept%202020.pdf (accessed June 3 2021).
Schulz, L. T., S. Y. Kim, A. Hartsell, and W. E. Rose. 2019. Antimicrobial stewardship during a time of rapid antimicrobial development: Potential impact on industry for future investment. Diagnostic Microbiology and Infectious Disease 95(3):114857.
Sciarretta, K., A. Opalska, A. Van-Hengel, and J. Larsen. Economic incentives for antimicrobial therapy development: Summary from the transatlantic task force on antimicrobial resistance. https://www.cdc.gov/drugresistance/pdf/economic-incentives-for-antimicrobialdevelopment-tatfar.pdf (accessed June 3, 2021).
ScienceDirect. 2021a. Disk diffusion. https://www.sciencedirect.com/topics/immunologyand-microbiology/disk-diffusion (accessed June 4, 2021).
ScienceDirect. 2021b. High throughput screening. https://www.sciencedirect.com/topics/medicine-and-dentistry/high-throughput-screening (accessed June 10, 2021).
Segerman, A. 2019. The DISARM Act can help fix the broken antibiotics market. In BIOtech-NOW. Biotechnology Innovation Organization. https://www.bio.org/blogs/disarm-act-can-help-fix-broken-antibiotics-market (accessed June 3, 2021).
Shawar, R. 2016. Overview of AST device clearance process: CDRH perspective. Public Workshop—Coordinated Development of Antimicrobial Drugs and Antimicrobial Susceptibility Test Devices, Silver Spring, MD. https://www.fda.gov/drugs/news-events-human-drugs/coordinated-development-antimicrobial-drugs-and-antimicrobialsusceptibility-test-devices (accessed June 4, 2021).
Simpkin, V. L., M. J. Renwick, R. Kelly, and E. Mossialos. 2017. Incentivising innovation in antibiotic drug discovery and development: Progress, challenges and next steps. Journal of Antibiotics 70(12):1087-1096.
Singer, A. C., C. Kirchhelle, and A. P. Roberts. 2020. (inter)nationalising the antibiotic research and development pipeline. Lancet Infectious Diseases 20(2):e54-e62.
Spellberg, B. 2021. A new sustainable model of antibiotic R&D. Committee on the Long-Term Health and Economic Effects on Antimicrobial Resistance in the United States: Meeting 3. Webinar. January 5, 2021. https://www.nationalacademies.org/event/01-05-2021/examining-the-long-term-health-and-economic-effects-of-antimicrobial-resistance-in-the-united-states-meeting-3 (accessed January 5, 2021).
Spellberg, B., J. G. Bartlett, and D. N. Gilbert. 2013. The future of antibiotics and resistance. New England Journal of Medicine 368(4):299-302.
Stafford, K. A., M. Boutin, S. R. Evans, and A. D. Harris. 2014. Difficulties in demonstrating superiority of an antibiotic for multidrug-resistant bacteria in nonrandomized studies. Clinical Infectious Diseases 59(8):1142-1147.
Steedman, M., and K. Taylor. 2019. Ten years on measuring the return from pharamceutical innovation. London, UK: Deloitte LLP.
Strich, J. R., S. Warner, Y. L. Lai, C. Y. Demirkale, J. H. Powers, 3rd, R. L. Danner, and S. S. Kadri. 2020. Needs assessment for novel gram-negative antibiotics in US hospitals: A retrospective cohort study. Lancet Infectious Diseases 20(10):1172-1181.
Suzuki, H. 2021. Building resilient global supply chains—the geopolitics of the Indo-Pacific Region. Washington, DC: Center for Strategic and International Studies. https://www.csis.org/analysis/building-resilient-global-supply-chains-geopolitics-indo-pacific-region (accessed June 25, 2021).
Talbot, G. H., A. Jezek, B. E. Murray, R. N. Jones, R. H. Ebright, G. J. Nau, K. A. Rodvold, J. G. Newland, H. W. Boucher, and Infectious Diseases Society of America. 2019. The Infectious Diseases Society of America’s 10 × 20 initiative (10 new systemic antibacterial agents U.S. Food and Drug Administration approved by 2020): Is 20 × 20 a possibility? Clinical Infectious Diseases 69(1):1-11.
Terpou, A., A. Papadaki, I. K. Lappa, V. Kachrimanidou, L. A. Bosnea, and N. Kopsahelis. 2019. Probiotics in food systems: Significance and emerging strategies towards improved viability and delivery of enhanced beneficial value. Nutrients 11(7):1591.
The Pew Charitable Trusts. 2020a. Correspondence re: CMS-1735-p: Medicare program; hospital inpatient prospective payment systems for acute care hospitals and the long term care hospital prospective payment system and proposed policy changes and fiscal year 2021 rates; quality reporting and medicare and medicaid promoting interoperability programs requirements for eligible hospitals and critical access hospitals proposed rule. Washington, DC: The Pew Charitable Trusts. https://www.pewtrusts.org/-/media/assets/2020/07/fy2021-ippspew-comments.pdf (accessed September 22, 2021).
The Pew Charitable Trusts. 2020b. Tracking the global pipeline of antibiotics in development, April 2020. Washington, DC: The Pew Charitable Trusts.
The Rockefeller Foundation. 2020. A landscape analysis of laboratory technologies for COVID-19. https://www.rockefellerfoundation.org/wp-content/uploads/2020/12/LMIC-Covid19-Landscape-Analysis_20-Nov-2020.pdf (accessed June 29, 2021).
Theuretzbacher, U., and L. J. V. Piddock. 2019. Non-traditional antibacterial therapeutic options and challenges. Cell Host & Microbe 26(1):61-72.
Theuretzbacher, U., K. Outterson, A. Engel, and A. Karlen. 2020. The global preclinical antibacterial pipeline. Nature Reviews: Microbiology 18(5):275-285.
Thomas, J., M. Navre, A. Rubio, and A. Coukell. 2018. Shared platform for antibiotic research and knowledge: A collaborative tool to spark antibiotic discovery. ACS Infectious Diseases 4(11):1536-1539.
Towse, A., C. K. Hoyle, J. Goodall, M. Hirsch, J. Mestre-Ferrandiz, and J. H. Rex. 2017. Time for a change in how new antibiotics are reimbursed: Development of an insurance framework for funding new antibiotics based on a policy of risk mitigation. Health Policy 121(10):1025-1030.
Trudil, D. 2015. Phage lytic enzymes: A history. Virologica Sinica 30(1):26-32.
Trusheim, M. R., A. A. Shrier, Z. Antonijevic, R. A. Beckman, R. K. Campbell, C. Chen, K. T. Flaherty, J. Loewy, D. Lacombe, S. Madhavan, H. P. Selker, and L. J. Esserman. 2016. Pipelines: Creating comparable clinical knowledge efficiently by linking trial platforms. Clinical Pharmacology and Therapeutics 100(6):713-729.
Tsai, J. M., N. V. Tolan, A. K. Petrides, S. Kanjilal, M. Brigl, N. I. Lindeman, Y. D. Li, M. J. Tanasijevic, S. S. Basu, and S. E. F. Melanson. 2021. How SARS-CoV-2 transformed the clinical laboratory: Challenges and lessons learned. Journal of Applied Laboratory Medicine 6(5):1338-1354.
USAMRIID (United States Army Medical Research Institute of Infectious Diseases). 2021. USAMRIID: Biodefense solutions to protect our nation. https://www.usamriid.army.mil (accessed September 21, 2021).
USDA (U.S. Department of Agriculture). 2014. Antimicrobial resistance action plan. U.S. Department of Agriculture. https://www.usda.gov/sites/default/files/documents/usda-antimicrobial-resistance-action-plan.pdf (accessed July 13, 2021).
USDT (U.S. Department of the Treasury). 2021. G7 finance ministers & central bank governors communiqué. https://home.treasury.gov/news/press-releases/jy0215 (accessed September 22, 2021).
Vallabhaneni, S., J. Y. Huang, J. E. Grass, A. Bhatnagar, S. Sabour, J. D. Lutgring, D. Campbell, M. Karlsson, A. J. Kallen, E. Nazarian, E. A. Snavely, S. Morris, C. Wang, R. Lee, M. Koag, R. Lewis, B. Garcia, EIP Work Group, A. C. Brown, and M. S. Walters. 2021. Antimicrobial susceptibility profiles to predict the presence of carbapenemase genes among carbapenem-resistant pseudomonas aeruginosa isolates. Journal of Clinical Microbiology 59(6):e02874-02820.
van Belkum, A., T. T. Bachmann, G. Ludke, J. G. Lisby, G. Kahlmeter, A. Mohess, K. Becker, J. P. Hays, N. Woodford, K. Mitsakakis, J. Moran-Gilad, J. Vila, H. Peter, J. H. Rex, W. M. Dunne, Jr., and the JPIAMR AMR-RDT Working Group on Antimicrobial Resistance and Rapid Diagnostic Testing. 2019. Developmental roadmap for antimicrobial susceptibility testing systems. Nature Reviews: Microbiology 17(1):51-62.
van Belkum, A., C. D. Burnham, J. W. A. Rossen, F. Mallard, O. Rochas, and W. M. Dunne, Jr. 2020. Innovative and rapid antimicrobial susceptibility testing systems. Nature Reviews: Microbiology 18(5):299-311.
van de Sande-Bruinsma, N., H. Grundmann, D. Verloo, E. Tiemersma, J. Monen, H. Goossens, M. Ferech, European Antimicrobial Resistance Surveillance System Group, and European Surveillance of Antimicrobial Consumption Project Group. 2008. Antimicrobial drug use and resistance in Europe. Emerging Infectious Diseases 14(11):1722-1730.
Vekemans, J., M. Hasso-Agopsowicz, G. Kang, W. P. Hausdorff, A. Fiore, E. Tayler, E. J. Klemm, R. Laxminarayan, P. Srikantiah, M. Friede, and M. Lipsitch. 2021. Leveraging vaccines to reduce antibiotic use and prevent antimicrobial resistance: A World Health Organization action framework. Clinical Infectious Diseases 73(4):e1011-e1017.
Ventola, C. L. 2015. The antibiotic resistance crisis: Part 1: Causes and threats. P&T 40(4):277-283.
Warrington, J. S., J. W. Crothers, A. Goodwin, L. Coulombe, T. Hong, L. Bryan, C. Wojewoda, M. Fung, G. Warrington, V. Clark, L. Risley, and M. Lewis. 2021. All hands-on deck and all decks on hand: Surmounting supply chain limitations during the COVID-19 pandemic. Academic Pathology 8:23742895211011928.
WEF (World Economic Forum). 2018. Antimicrobial resistance tackling the gap in R&D resources with pull incentives. Wellcome Trust. Geneva, Switzerland: World Economic Forum. https://www.weforum.org/reports/antimicrobial-resistance-tackling-the-gap-in-r-d-resources-with-pull-incentives-in-collaboration-with-wellcome-trust (accessed June 3, 2021).
WHO (World Health Organization). 2015. Vaccinating salmon: How Norway avoids antibiotics in fish farming. https://www.euro.who.int/en/health-topics/disease-prevention/antimicrobial-resistance/news/news/2015/10/vaccinating-salmon-how-norwayavoids-antibiotics-in-fish-farming (accessed August 22, 2021).
WHO. 2019a. Antibacterial agents in clinical development: An analysis of the antibacterial clinical development pipeline. Geneva, Switzerland: World Health Organization.
WHO. 2019b. Antibacterial agents in preclinical development: An open access database. Geneva, Switzerland: World Health Organization.
WHO. 2020. Global partnership to make available 120 million affordable, quality COVID-19 rapid tests for low- and middle-income countries. https://www.who.int/news/item/28-09-2020-global-partnership-to-make-available-120-million-affordable-quality-covid-19-rapid-tests-for-low--and-middle-income-countries (accessed June 29, 2021).
WHO. 2021. 2020 antibacterial agents in clinical and preclinical development: An overview and analysis. Geneva, Switzerland: World Health Organization.
Wouters, O. J., M. McKee, and J. Luyten. 2020. Estimated research and development investment needed to bring a new medicine to market, 2009-2018. JAMA 323(9):844-853.
Zimmer, B. 2021. Regulatory challenges in antimicrobial diagnostics. Committee on the Long-Term Health and Economic Effects of Antimicrobial Resistance: Meeting 3. Webinar. January 6, 2021. https://www.nationalacademies.org/event/01-05-2021/examining-the-long-term-health-and-economic-effects-of-antimicrobial-resistance-in-the-united-states-meeting-3 (accessed January 6, 2021).
Zumla, A., J. A. Al-Tawfiq, V. I. Enne, M. Kidd, C. Drosten, J. Breuer, M. A. Muller, D. Hui, M. Maeurer, M. Bates, P. Mwaba, R. Al-Hakeem, G. Gray, P. Gautret, A. A. Al-Rabeeah, Z. A. Memish, and V. Gant. 2014. Rapid point of care diagnostic tests for viral and bacterial respiratory tract infections—needs, advances, and future prospects. The Lancet Infectious Diseases 14(11):1123-113.














































































