6 The Effectiveness of Bleach as a Disinfectant of Injection Drug Equipment
The needles and syringes used by injection drug users are produced for the delivery of legal drugs or biologics that require intravenous, intramuscular, intradermal, or subcutaneous delivery. They are manufactured according to exacting standards to ensure sterility and are intended for use by a single individual for a single injection on only a single occasion. As detailed in Chapter 5, the limited availability of adequate numbers of sterile needles and syringes results in their frequent reuse and sharing by injection drug users. The repeated use of needles and syringes necessarily compromises their sterility and safety. The sharing of injection equipment by
Note: a significant portion of the discussion in this chapter is based on Alice Gleghorn's (1994) paper commissioned by the panel, Practical Issues of Needle and Syringe Disinfection Programs. It reviews the history of bleach distribution programs and includes descriptions of these programs in San Francisco, Chicago, New York, and the various locations involved in the National AIDS Demonstration Research Project. Method variation in bleach use by injection drug users, encompassing substances and techniques used, comparisons of injection drug users who disinfect and those who do not, and problems with bleach disinfection are also discussed, as well as provisional federal regulations and the extension of bleach distribution programs. Information was also obtained from another commissioned paper, Inactivation of HIV: Application to Injecting Drug Use Equipment, by Linda Martin, Walter Bond, T. Stephen Jones, and Martin Favero (1994). This paper reviews the processes and biological aspects of disinfection and inactivation of the human immunodeficiency virus and discusses the applications of these studies to the cleaning and disinfection of syringes and needles reused by injection drug users.
injection drug users is particularly hazardous because it provides an efficient means for transmission of a number of blood-borne infectious diseases, including the HIV, hepatitis B (HBV), and hepatitis C viruses and the human T-cell leukemia viruses (HTLV-I and HTLV-II).
With the advent of the AIDS epidemic, the recognition of the rapid dissemination of HIV infection within injection drug user communities, and legal constraints associated with access to injection equipment (see Chapter 5), the use of bleach to clean and disinfect previously used needles and syringes has been encouraged by outreach workers in an attempt to improve the safety of the inherently unsafe practice of sharing needles and syringes among injection drug users. This chapter reviews the scientific literature on the efficacy of bleach as a disinfectant and the effectiveness of bleach distribution programs on reducing the potential harmful effects associated with injection drug use—risk behaviors and HIV transmission.
TRANSMISSION OF HIV AMONG INJECTION DRUG USERS
HIV is clearly a blood-borne pathogen. However, it is not yet known what proportion of HIV transmission between individuals results from the transfer of free infectious virus present in the plasma fraction of the blood of an infected individual, in the HIV-infected peripheral blood cells, or both. The sharing of needles and syringes by injection drug users can result in the transmission of pathogens spread as free virus (such as HBV) or via the transfer of infected cells (such as HTLV-I and HTLV-II). Resolution of the vehicle for HIV transmission may prove to be important, since HIV present within infected cells or as free virus may be differentially susceptible to the various available disinfection methods.
The actual amount (infectious dose) of HIV necessary to transfer the infection from one person to another is not known. In the absence of this information, attempts to model the effectiveness of any virus disinfection strategy are difficult because the level of inactivation needed to prevent transfer of the infection is unclear. Ideally, a disinfectant strategy will completely eliminate the infectivity of all HIV contaminating a surface. However, to be effective at limiting the transmission of HIV between individuals, such a strategy need only reduce the level of infectivity below the minimal infectious dose.
At present, the process of evaluating the effectiveness of various disinfection strategies relies on tissue culture models, in which virus inactivation is measured by the ability to isolate infectious virus following disinfectant treatment. Unfortunately, it is not known whether the available tissue culture-based HIV isolation methods are sensitive enough to detect the minimum infectious dose of virus responsible for transferring HIV infection between individuals. If virus isolation methods are not sensitive enough, a
preparation of HIV that was completely inactivated in the laboratory might still be able to transmit infection in vivo.
The amount of both free HIV and virus-infected cells present in the blood of infected individuals varies with the stage of the infection (see Chapter 1). During the acute infection stage, newly infected persons have extremely high levels of HIV present in circulating peripheral blood cells and in the plasma. When a host antiviral immune response is generated, typically within 1 to 3 months of initial infection, the levels of infected cells and plasma virus fall dramatically. HIV replication continues throughout the asymptomatic phase of the disease, primarily within the lymphoid organs of infected persons. During this period, recirculating HIV-infected lymphocytes and cell-free virus can be detected in the peripheral bloodstream. As a result, HIV-infected injection drug users are likely to be able to transmit the virus to others by sharing injection equipment throughout the entire course of their infection. Increasing levels of HIV and HIV-infected cells appear in the blood of infected persons as immune containment of virus replication fails and clinical disease becomes manifest.
Although epidemiologic evidence obtained from studies of heterosexual and perinatal transmission of HIV infection suggests that individuals who are in the acute or advanced stages of HIV infection may be more infectious than those in the early stages of the infection, no information concerning this issue has been reported in injection drug users (see Chapter 1). It seems reasonable to expect, however, that not all HIV-positive injection drug users are equally infectious. From the perspective of disinfection strategies for needles and syringes, effective methods need to be able to inactivate the viral burden present in contaminating blood derived from even the most highly infectious individuals.
Although it is commonly assumed that HIV transmission among injection drug users results from sharing contaminated needles and syringes, there are a number of additional factors that may account for virus spread. For example, HIV contamination of other materials used by injection drug users, including the paraphernalia used to mix and prepare drugs for injection ("cookers") and the water used to dissolve the drugs or rinse the needles and syringes between their use by different individuals, can potentially serve to transmit HIV. The relative roles of these different materials and processes in spreading HIV among injection drug users are not known.
BLEACH AS A DISINFECTANT
Definition of Terms
Although the terms sterilization and disinfection are widely employed, they are often misused (Block, 1983). Sterilization is the use of physical or
chemical procedures to destroy all forms of microbial life, including highly resistant bacterial endospores. Sterilization is frequently accomplished using high-pressure steam autoclaving, ethylene oxide gas, or prolonged exposure to dry heat. Disinfection is the elimination of all vegetative microorganisms and pathogenic viruses, but not bacterial or fungal spores, from an inanimate object. As such, disinfection processes lack the margin of safety achieved through sterilization procedures. Furthermore, the effectiveness of a disinfection procedure is influenced significantly by a number of factors, including the nature and number of contaminating microorganisms, the type and condition of the materials to be disinfected, and the amount of organic matter (such as blood) present (Klein and Deforest, 1965a; Rhame, 1986; Rutala and Weber, 1987; Rutala, 1987; Van Houton and Hayre, 1991).
A germicide is an agent that destroys microorganisms, especially pathogens, on either living or inanimate objects. Disinfectants can be categorized into high-, medium-, and low-level agents based on their potency of germicidal action. Disinfectants that are widely used at present include alcohols, hypochlorites (such as bleach) and other chlorine-containing compounds, formaldehyde, glutaraldehyde, hydrogen peroxide, iodophors, phenolics, and quaternary ammonium compounds. The level of contamination with organic matter of objects to be disinfected is referred to as the bioburden, and it is of particular importance in the disinfection of surfaces contaminated with blood that harbors infectious pathogens. Contact time is defined as the length of time that a contaminated object or surface is exposed to a disinfectant. Cleaning is the removal of foreign (especially organic) material. Decontamination is the removal of pathogenic microorganisms from objects so they are safe to handle.
The Role of Cleaning in Disinfection
Thorough cleaning must always precede chemical disinfection of any equipment (Klein and Deforest, 1965a; Rhame, 1986; Rutala and Weber, 1987; Rutala, 1987; Van Houton and Hayre, 1991). The mechanical action of cleaning can itself remove a large proportion of the microorganisms present. Cleaning also removes organic material that can inactivate or diminish the potency of the germicide used. The presence of blood on or in equipment being processed for reuse, including needles and syringes, can contribute to the failure of a given disinfection or sterilization procedure in three ways. First, organic material may contain large and/or diverse microbial populations. Second, it may trap microorganisms and prevent effective penetration of chemical germicides. Third, it may directly and rapidly inactivate certain germicidal chemicals, including bleach and other chlorine-containing solutions, iodine-based disinfectants, and quaternary ammonium-based
compounds. Physical cleaning is often the most important step in a disinfection process, and even a rigorous disinfection procedure may not inactivate contaminating bacteria or viruses if these are protected by organic material, such as blood.
Bleach is a solution of sodium hypochlorite, and it exerts rapid and broad-spectrum disinfecting action (Hugo and Russell, 1982; Ingraham, 1992; Klein and Deforest, 1965a, 1965b; Rutala and Cole, 1984; Rutala, 1990; Van Houton and Hayre, 1991). Sodium hypochlorite solutions in concentrations of 0.05 to 0.5 percent (500-5,000 milligrams per milliliter) free and available chlorine are generally considered to be intermediate-level disinfectants and are among the most effective, most convenient, and least expensive germicides. The precise mechanism through which bleach exerts its germicidal action is not known, but it is believed to be mediated by the ability of free chlorine to denature proteins, inactivate sulfhydryl-containing enzymes, and damage nucleic acids (RNA and DNA).
Household bleach manufactured in the United States contains approximately 5 percent sodium hypochlorite (50,000 milligrams per milliliter chlorine [Cl2]). Bleach solutions exhibit sporicidal activity, are tuberculocidal, inactivate vegetative bacteria, and are fungicidal and virucidal. Klein and Deforest (1963) reported that 25 viruses tested, including picornaviruses, were inactivated within 10 minutes by as little as 0.02 percent (200 milligrams per milliliter) available chlorine.
The efficacy of a bleach solution to act as a disinfectant is determined by the concentration of free and available chlorine present in the solution (Klein and Deforest, 1965a, 1965b; Rutala and Cole, 1984; Rutala, 1990; Van Houton and Hayre, 1991). Chlorine demand is a term used to describe the amount of chlorine that is expended in the course of reaction with inorganic and organic materials. After this demand is met, any remaining chlorine is referred to as available chlorine . The level of available chlorine present to effect disinfection is influenced by a number of variables, including the amount of organic material in the infectious material and the temperature, pH, and hardness of the water used to dilute the bleach solution. Serum proteins and other organic material in blood will reduce the chlorine in bleach available for microbial inactivation (Favero and Bond, 1991; Klein and Deforest, 1965a; Rutala and Cole, 1984; Rutala, 1987). In practice in the health care setting, it is thus recommended that blood-contaminated surfaces first be cleaned to remove as much visible blood as possible prior to treatment with bleach.
The disinfectant potential of a bleach solution is not constant; rather, it decreases with time because of the relative instability of the active chlorine component. Bleach solutions lose potency at an accelerated rate when exposed to sunlight, oxygen, and heat. Bleach solutions prepared with tap
water at pH 8 or greater are stable for about one month when stored at room temperature in a closed, opaque container.
Susceptibility of HIV to Disinfection
Microorganisms vary widely in their level of resistance to chemical germicides (Favero and Bond, 1991; Hugo and Russell, 1982; Rutala and Weber, 1987; Klein and Deforest, 1965a). The types of microorganisms present can have a significant effect on the contact time and the concentration of germicide necessary to ensure the sterilization or disinfection of a contaminated object. Among pathogenic microorganisms, bacterial spores are the most resistant to sterilization and disinfection. In general, the presence of lipid in a virus is associated with a high degree of susceptibility to all germicides. The blood-borne pathogens HIV and HBV both are surrounded by lipid-containing membranes and thus are expected to be among the microorganisms least resistant to disinfection and sterilization. In principle, and in practice in the laboratory and the health care environment, there is no reason to believe that HIV is resistant to disinfection (Conte, 1986; Centers for Disease Control [CDC], 1987).
The identification of HIV as the cause of AIDS and the availability of tissue culture models to study virus infectivity permitted the laboratory evaluation of methods to disinfect HIV present on surfaces. The primary goal of the initial studies of disinfection methods was to determine the optimal approaches to disinfect reusable medical devices and to safely clean surfaces contaminated with blood from HIV-infected patients. Although varying in their precise protocols, all early studies were performed using HIV obtained from the supernatants of virus-infected tissue culture cell lines (Spire et al., 1984; Resnick et al., 1986; Martin et al., 1985). The virus present in such supernatants is not associated with infected cells, and this is referred to as cell-free virus. The infectious titer of cell-free HIV found in tissue culture supernatants ranges from 104 to 106 tissue culture infectious doses (TCID) per milliliter volume; these titers are several orders of magnitude higher than those typically present in the blood of HIV-infected individuals (Pantaleo et al., 1993). In addition, the amounts of proteins and other organic materials present in tissue culture fluids are usually less than those found in blood.
It is important to emphasize that preparations of cell-free HIV may demonstrate significantly different levels of susceptibility to disinfecting agents than samples of virus-infected cells, particularly as they may be found in blood specimens that are rich in cellular and extracellular (plasma) protein content. Cell-free HIV derived from supernatants of tissue culture cells is most susceptible to disinfection, whereas infected cells (and possibly cell-free virus present in the plasma) contained within blood specimens
are more difficult to inactivate (Flynn et al., 1994; Shapshak et al., 1994). The role that the HIV contained in clotted blood present within needles and syringes may play in virus transmission is not known. Should HIV trapped in clotted blood remain infectious, it may be more difficult to inactivate by disinfectant treatment because of the substantial increase in contact time that may be needed (see special issue of the Journal of Acquired Immune Deficiency Syndromes 7:7, 1994).
To date, no satisfactory approaches to measure the infectivity of HIV present in clotted blood or model the efficiency of inactivation of the virus contained in clotted blood specimens have been devised. For these reasons, the experimental methods that have been used to monitor HIV inactivation by disinfectants have (to various degrees) approximated, but not reproduced, the circumstances through which HIV transmission occurs between injection drug users sharing contaminated injection equipment.
Laboratory Studies
Laboratory-based HIV inactivation studies commonly have been performed by mixing high-titer preparations of cell-free HIV with various dilutions of disinfectants for varying periods of time. Following designated time(s) of exposure, serial dilutions of each virus-disinfectant mixture are then added to CD4+ T-cells (that are susceptible to HIV infection) in tissue culture. The cultures are then monitored for evidence of active replication of HIV, as evidenced by increasing levels of virus-specific antigens (the p24 core antigen) or enzymatic activities (reverse transcriptase). Increasing levels of HIV p24 or reverse transcriptase activity in the tissue culture supernatants from cells exposed to disinfectant-treated virus preparations is indicative of the failure of disinfection. The absence of HIV p24 antigen or reverse transcriptase in test culture provides evidence that the concentration and exposure time of disinfectant used successfully inactivated the amount of HIV present in the original inoculum. Because of the inherent toxicity of disinfectants, it is essential that appropriate controls be performed to determine if the test disinfectant killed the indicator CD4+ T-cells used to propagate the virus; such toxicity can undermine the sensitivity of the virus isolation procedure and lead to overestimating the effectiveness of the disinfectant at inactivating the virus itself.
Studies performed to date have usually employed laboratory-adapted strains of HIV and immortalized T-lymphocyte cell lines. Although there is no known reason to expect differences between the laboratory strains of HIV and primary virus isolates with respect to their intrinsic susceptibility to disinfection, this issue has not been addressed by researchers in the field. Similarly, most experimental evaluations of HIV disinfection methods have used immortalized CD4+ T-cell lines as indicator cells rather than primary
cultures of T lymphocytes or macrophages for HIV isolation. The impact of the choice of indicator cells on the sensitivity of virus detection following disinfection is not known.
Spire and colleagues (1984), using an assay for the presence of HIV reverse transcriptase activity (and not the residual infectivity of the virus preparation itself), examined the inactivation of cell-free HIV by commonly used disinfectants. Their results suggested that 25 percent ethanol or 1 percent glutaraldehyde should be sufficient to disinfect medical instruments, and that 0.2 percent sodium hypochlorite should be sufficient to disinfect contaminated environmental surfaces. They also reported that HIV was inactivated by a 1:400 dilution of beta-propiolactone or 30 millimolar sodium hydroxide, but that formalin 0.1 percent required 48 hours to render reverse transcriptase activity undetectable.
Resnick and colleagues (1986) pointed out that assays for reverse transcriptase activity are not reliable alternatives to tests for infectious virus. These authors, using concentrated preparations of cell-free HIV, found that infectious virus could be recovered from dried material for 3 to 7 days. In an aqueous environment, cell-free HIV retained its infectivity for more than 2 weeks at room temperature. However, HIV infectivity was completely eliminated following 1 minute of exposure to sodium hypochlorite 0.5 percent (a 10 percent solution of household bleach), alcohol 70 percent, or a 0.5 percent solution of the detergent NP-40.
Martin and colleagues (1985), using antigen detection methods to monitor virus replication in tissue culture, reported that cell-free HIV could be inactivated by a variety of disinfectants at concentrations well below those commonly employed. They reported that HIV infectivity was efficiently inactivated by 0.3 percent hydrogen peroxide, 50 percent ethanol, 0.5 percent paraformaldehyde, 0.5 percent Lysol (a proprietary mixture of phenolics and surfactants), and a 1/1,000 dilution of household bleach (approximately 50 milligram per milliliter chlorine). Since that time, many laboratories have performed HIV inactivation studies with various compounds. The results of many of these studies have been reviewed by Sattar and Springthorpe (1991) and confirm the predicted susceptibility of HIV to inactivation by a wide variety of chemical disinfectants.
CDC Recommendations
Based on the results of these early studies, recommendations were formulated by the Centers for Disease Control for the sterilization, disinfection, and housekeeping procedures in health care settings (Centers for Disease Control, 1987). These guidelines continue to serve effectively and have not been modified. An example of an effective disinfection procedure used in the health care environment relevant to the consideration of inactivation
of HIV present on blood-contaminated surfaces is provided by the example of the disinfection of hemodialysis machines. Patients infected with HIV who are undergoing hemodialysis do not require isolation from other dialysis patients. Although the dialyzer itself is disposable, the recommendations for disinfecting the dialysis fluid pathways of the hemodialysis machine (exposure to 500 to 750 milligrams per liter of sodium hypochlorite for 30 to 40 minutes)—originally intended for controlling bacterial contamination—are also effective in inactivating HIV (Centers for Disease Control, 1986; Conte, 1986).
In sum, when used appropriately, there is no doubt that bleach can effectively disinfect surfaces contaminated by HIV-infected blood.
BLEACH DISTRIBUTION PROGRAMS
In the mid-1980s, the recognition of an impending epidemic in the spread of HIV infection in injection drug users led to discussion among health care professionals and providers concerning potential intervention strategies to limit virus transmission. Although HIV infection had entered the injection drug user population in San Francisco by 1985, it had not yet assumed the high level of prevalence that was seen among injection drug users in the New York metropolitan area. A San Francisco group of community health outreach workers that came to be known as the Mid-City Consortium to Combat AIDS proposed that the types of education and behavior change efforts that had been successfully conducted among homosexual and bisexual men in the area (Becker and Joseph, 1988) might also attenuate the transmission of HIV among injection drug users (Newmeyer, 1988). However, the opportunities for public health interventions were limited.
California's paraphernalia and prescription laws made the prospects for legally authorized needle exchange remote. The most effective risk reduction behavior, cessation of drug use, was not a realistic option for many injection drug users because of the nature of the addictive process and limited treatment resources (Murphy, 1987). Social norms for sharing injection equipment among injection drug users, as well as legal and economic constraints, all placed restrictions on an individual's ability and motivation to keep a needle and syringe set for personal use only (Des Jarlais et al., 1986; Hopkins, 1988; Feldman and Biernacki, 1988). Therefore, the option of disinfecting needles and syringes seemed to hold the greatest promise for realistic interventions to limit HIV transmission among injection drug users.
Ethnographic studies were conducted in San Francisco to determine characteristics that would make a disinfection strategy acceptable to injection drug users (Newmeyer, 1988). Newmeyer listed five essential features
of a disinfection technique as follows: ''(1) it should be quick, preferably taking less than 60 seconds; (2) it should be inexpensive; (3) it should use materials conveniently available; (4) it should be safe to the user and his/her injection equipment; and (5) it should be effective at neutralizing viruses" (Newmeyer, 1988:160). In determining an appropriate disinfectant, published studies available at the time—particularly those of Martin et al. (1985) and Resnick and colleagues (1986)—were the primary sources of information regarding the efficacy of various disinfectants for inactivation of HIV under laboratory conditions.
Based on these studies, several disinfectants were considered, including bleach, hydrogen peroxide, and isopropyl alcohol. According to Newmeyer (1988), hydrogen peroxide was rejected due to its limited shelf life and sensitivity to light exposure; the potential for isopropyl alcohol to be confused with other readily available sources of alcohol in beer, wine, and hard liquors eliminated it from consideration. Boiling syringes in water for 15 minutes was another strategy considered; however, this strategy was deemed impractical due to the length of time required, the facilities needed, and the fact that boiling melted the syringe (Murphy, 1987). As a result of these considerations, household bleach (approximately 5 percent sodium hypochlorite) was determined to be the most feasible. Bleach met the criteria for cost and availability, and reviews of the available literature (Becker et al., 1974; Froner et al., 1987) suggested that accidental injection of bleach would not produce great harm to injection drug users. Aside from fading the numbers inscribed on the syringe barrel (Newmeyer, 1988) and corrosion of the rubber stopper of the plunger of the syringe, bleach did not seem to have excessive deleterious effects on the injection equipment.
Following the selection of bleach as the disinfectant of choice, community health outreach workers in San Francisco began a campaign of bleach distribution and provided simple, graphic instructions for its use in disinfecting needles and syringes (Watters, 1987). The initial bleach distribution efforts were part of a broader education program to decrease the risk of HIV infection among injection drug users that included condom distribution and voluntary HIV counseling and testing. The educational message delivered was: "The best protection is to stop using drugs; if you can't stop using, don't inject; if you can't stop injecting, at least keep your own outfit and don't share it with anyone; if you do share, use bleach to reduce your risk." The recommended protocol for bleach disinfection was to fill needles and syringes twice with bleach, followed by two water rinses (Newmeyer, 1988; Froner et al., 1987; Watters, 1987). These initial disinfection guidelines recommended the use of full-strength bleach, which was believed by the San Francisco community health outreach workers to provide a more potent and convenient disinfectant than the diluted bleach preparations used in the early laboratory studies.
Program Evaluation
Prior to 1993, the instructions for bleach use disseminated across the country were relatively consistent with the methods developed by the Mid-City Consortium in San Francisco (Gleghorn, 1993). With few exceptions (Flynn et al., 1994), bleach has been exclusively promoted as the disinfectant of choice for injection equipment. As a result, awareness of bleach disinfection strategies among injection drug users is high (Meyers et al., 1990). Therefore, a review of the studies that have evaluated cleaning techniques used by injection drug users provides indications concerning user compliance with the recommended bleach techniques.
The bleach distribution program in San Francisco was well accepted by injection drug users. Among those interviewed in neighborhoods with high drug use, Watters (1987, 1994) reported that prior to initiation of the outreach program, only 3 percent of the sample used bleach to clean their equipment. In contrast, 9 months after beginning the community health outreach worker (CHOW) intervention, 76 percent of injection drug users interviewed reported bleach use. In addition, prior to program initiation, regular cleaning of needles and syringes was reported by only 21 percent of those who reported any cleaning attempts. In the follow-up interview, injection drug users who used bleach reported doing so 90 percent of the time. Although other reports (Chaisson et al., 1987) from San Francisco reported less dramatic (approximately fivefold) increases in bleach use, they noted important additional effects of outreach efforts that included bleach distribution on other HIV-related risk behaviors, including decreases in needle sharing and increases in condom use (Chaisson et al., 1987; Sorensen et al., 1989).
Moreover, in these early studies of the adoption of bleach disinfection by injection drug users, a significant positive relationship was observed between access to community health outreach workers and reported use of bleach disinfection (Watters, 1987). Another important variable in the success of bleach education programs appears to be the direct provision of bleach to injection drug users. For example, the AIDS Outreach Program (AOP) provided street-level education on bleach use but did not actually distribute bleach (Neaigus et al., 1990). Evaluation of the impact of this program found that only low levels of regular bleach use were noted both at baseline and during follow-up interviews (16 and 18 percent, respectively). In interpreting these results, Neaigus and colleagues (1990) suggested that outreach efforts aimed at increasing bleach use should include distribution of bleach.
Following the lead of San Francisco, bleach distribution programs proliferated rapidly in cities across the United States. Using techniques adopted from medical epidemiology and community ethnography, workers in Chicago
(Wiebel, 1990, 1993a) initiated an active program of education, intervention, and research, employing a field staff with extensive knowledge of and familiarity with the local community of injection drug users. In an evaluation of this approach (Wiebel et al., 1990), active injecting drug users were queried about their use of bleach disinfection at baseline and at 6 months following the initiation of the educational program. Bleach use as a disinfection strategy was adopted by many injection drug users. Two-thirds of the study participants who initially used bleach "less than always" reported an increase in bleach use, with one-third of the follow-up sample reporting "always" using bleach.
Similarly, researchers in New York (Friedman et al., 1990) helped form an organization of injection drug users in an effort to decrease HIV risk behaviors. Injection drug users in recovery provided HIV risk-reduction education for active users. These groups encouraged the adoption of safe sexual and drug-related behaviors and offered AIDS education, individual counseling, and HIV testing. In an evaluation of this program, follow-up comparisons were made between injection drug users who participated in the education and organizing effort and those who did not. Although use of new needles and condoms increased in both groups, only the organized injection drug users showed an increase in consistent bleach use.
Additional studies sponsored by the National Institute on Drug Abuse (NIDA) evaluated AIDS risk behaviors and trends in HIV infection in injection drug users in methadone treatment programs (Battjes and Pickens, 1992, 1993). Reported trends over the five-year period included increased use of bleach and alcohol by injection drug users to clean injection equipment in four of the seven cities studied: New York; Asbury Park, N.J.; Trenton, N.J.; Baltimore, Md.; Chicago, Ill.; San Antonio, Tex.; and Los Angeles. Additional studies conducted in Indianapolis and Los Angeles County have reported increased rates of bleach use (McKee et al., 1992; Longshore et al., 1993). These changes appear to reflect the increasing awareness and acceptance of bleach as an HIV prevention strategy.
In a survey of nearly 3,000 injection drug users in Baltimore conducted in 1988 and 1989, Latkin and colleagues (1992) reported that a variety of substances, including water, bleach, and alcohol, were used by injection drug users to clean contaminated needles and syringes. In addition to the use of various agents, the methods reportedly used by injection drug users for syringe disinfection were often imprecise and variable. Gleghorn and colleagues (Gleghorn et al., 1994; Gleghorn, 1994) studied cleaning practices used by injection drug users via videotaping mock needle and syringe cleaning sessions. They found that more than 80 percent of the 161 subjects studied used bleach for less than 30 seconds, although they reported cleaning for longer periods of time.
McCoy and colleagues (1992, 1994) studied injection drug users in Miami
who had been taught a bleach disinfection procedure consisting of two complete fillings of the syringe with bleach, followed by two complete fillings with rinse water, and discarding of the used cleaning solutions. Study participants were then evaluated for proper disinfection technique 6 to 12 months following initial training. At follow-up, a substantial proportion of the injection drug users did not perform all of the previously taught steps, and less than half completely filled the syringe with bleach or rinsed the syringe twice with bleach. These investigators also reported that compliance with disinfection methods decreased as the number of steps required increased.
Several behavioral characteristics of injection drug users have been found to be associated with use or nonuse of needle and syringe disinfection (Brown and Beschner, 1993; Latkin et al., 1992; Gleghorn et al., 1994; Edlin et al., 1993). Injection drug users who use shooting galleries, or who share needles and syringes with more than one partner, may be more likely to use bleach or alcohol for disinfection (Latkin et al., 1992). Neither injection frequency nor cocaine injection has been found to be associated with disinfectant use (Latkin et al., 1992). Injection drug users who also smoke crack cocaine report a lower rate of cleaning needles and syringes prior to injecting than nonsmokers, even though they inject less frequently (Edlin et al., 1993). Crack cocaine-smoking injection drug users are also more likely to use unsafe needles and to share rinse water, cookers, and cotton (Edlin et al., 1993).
Injection drug users who disinfect their injection equipment with bleach or alcohol are also more likely to be acquainted with other HIV risk reduction behaviors (Celentano et al., 1991). Knowledge of bleach effectiveness, condom effectiveness, and several other HIV transmission risks is higher among injection drug users with a history of bleach or alcohol disinfection. Further studies of beliefs and attitudes that differentiate bleach users from nonusers or that predict bleach use are needed to increase the adoption of effective cleaning strategies.
The National AIDS Demonstration Research (NADR) program represents a large-scale multisite prevention effort supported by the National Institute on Drug Abuse (National Institute on Drug Abuse, 1994). A total of 28 sites participated in this aspect of the NIDA initiative. This undertaking has produced what is probably the largest single database on out-of-treatment injection drug users with evaluative data on outreach program effectiveness. Brown (1994) describes the NADR "standard" intervention as typically involving at least: (1) the provision of information about and distribution of condoms and bleach by outreach and counseling staffs; (2) one session of individual educational counseling regarding AIDS transmission and strategies for AIDS risk reduction; (3) an offer to make available HIV testing; (4) posttest counseling for those accepting HIV testing;
and (5) prior to counseling, the administration of a structured interview schedule. In addition, 18 sites developed "enhanced" interventions involving didactic training sessions and other additional initiatives, including: (1) individual and group counseling on behavior change strategies; (2) couples counseling; (3) cognitive skills training; (4) peer networks; and (5) social skills training. As described in Chapter 3, it is apparent that these outreach programs are multicomponent AIDS prevention programs that do not rely exclusively on distributing bleach bottles in their attempt to reduce HIV risk behaviors and the transmission of HIV infection.
The NADR report (National Institute on Drug Abuse, 1994) indicates that these 28 sites assessed 33,407 injection drug users at intake. However, only those participants whose initial and follow-up data could be linked (13,475) formed the basis of the program evaluation component of this project. As such, these data represent a fraction of the total number of injection drug users who were served (or at least assessed) by the NADR projects (a 40 percent follow-up rate). For sex partners, 6,216 were assessed and 1,637 (26 percent) were followed.1 As reflected by the substantial variation in follow-up rates2 across sites, the quality of data provided by individual sites varied considerably. Given the considerable potential problems associated with attempting to integrate these data, the following section summarizes the evaluation results of one specific NADR site that has provided detailed information on program delivery and comprehensive longitudinal data on meaningful outcome measures (risk behaviors and HIV seroconversion).
The Chicago Experience
Chicago's NADR program (which is now supported by local funding) collected longitudinal data over a 4-year period in an attempt to assess program impact on risk behaviors and HIV seroconversion (Wiebel et al., 1993b, 1994). This program was similar to the other NADR sites in its focus on out-of-treatment injectors and use of street outreach workers as HIV prevention educators; however, it had unique elements as well. The project's prevention activities has six key features:
-
The intervention is carried out by street outreach workers who are former injectors.
-
Outreach staff identify and target social networks of injectors, rather than isolated individuals.
-
Outreach staff educate network members about HIV and AIDS and work with them to realistically assess their own risks for HIV infection. Complete confidentiality is stressed throughout.
-
Staff then offer multiple options for coping with identified risks.
-
Options may include referral to treatment to stop injecting altogether, social strategies to avoid sharing injection equipment, and, for those who still share, instruction on how to clean injection equipment with bleach and water to reduce the risk of HIV transmission. Outreach workers carry supplies of bleach, water, cotton, condoms, and information pamphlets at all times and distribute materials as needed in all encounters.
-
In repeated encounters with injection drug users, outreach workers support and reinforce behavioral change.
-
The intervention's impact is further extended by promoting prevention advocacy within injection drug user social networks for HIV risk reduction.
Chicago's program represents the more intensive end of the range of NADR outreach programs in the frequency and duration of contacts that take place between street outreach workers and networks of injection drug users. As stated above, this program cannot be considered simply a bleach distribution program, although it uses bleach and other injection supplies as tools to facilitate interaction with street addicts and to reduce injection harm. It focuses on total risk awareness, education, and reduction, rather than solely on distributing bleach and providing instruction in bleach use.
To monitor trends in risk behavior and HIV incidence, a panel of injection drug users was recruited for study and followed over a 4-year period. The results of seven waves (baseline and six follow-ups) of interview and serologic data collection from 1988 through 1992 were analyzed.
The sample was recruited from the three Chicago neighborhoods in which the outreach was conducted: the mostly African American South Side, the ethnically mixed North Side, and the largely Puerto Rican Northwest Side. These neighborhoods were selected for their high concentration of injection drug activity, the variety of injectable drugs represented in the areas, and the broad demographic mix of user characteristics known to exist in Chicago. Recruitment targeted injectors in the three outreach areas in community settings such as street corners, copping (drug sales) areas, and shooting galleries. Injectors were identified on the basis of street outreach workers' knowledge of them and injectors' identification of others in their injection networks (targeted sampling).
Between March and August 1988, baseline interviews and blood specimens were collected from a cohort of 850 injection drug users. Of this initial cohort, 209 (24.6 percent) were HIV seropositive at their first serologic evaluation. Those injection drug users initially HIV seronegative and thus at risk for infection (n = 641) were the focus of the evaluation.
Subsequent to the 1988 baseline assessment, six waves of follow-up interviews and blood specimen collections were performed through fall 1992. The questionnaires used at the baseline and follow-up waves assessed the
demographic characteristics, medical and drug treatment history, and recent drug-use and sexual behavior of respondents. Serologic screening for HIV-1 antibodies used venous specimens. Specimens repeatedly reactive in whole-virus lysate enzyme-linked immunosorbent assays (ELISA) were confirmed by Western blot.
The sample of 641 seronegative injection drug users was primarily male (76 percent); about two-thirds were age 40 or younger; somewhat more than half were African American, 26 percent white, and 18 percent Hispanic; and almost half reported injecting twice a day or more at recruitment. As of September 1992—4 years after the program began—the status of the cohort was 83 (13 percent) observed to have seroconverted, 42 (7 percent) had died and were seronegative at last serologic evaluation, 208 (32 percent) were seronegative at last evaluation and were lost to follow-up, and 308 (48 percent) were seronegative at the end of the study period. The cohort had second measures on 88 percent (513 of 641) of the baseline sample, and over-time follow-up response rates were 85, 84, 82, 78, and 68 percent, respectively, of the base sample in each of the successive waves.
Figure 6.1 shows the rate of HIV seroconversions per 100 person years (py). A decline in seroincidence was observed, from 8.4 per hundred py at the first follow-up wave to approximately 2.0 per hundred py after 1990.
Table 6.1 presents the percentages of the cohort reporting that they had engaged in HIV-related risk behavior since their last interview. The denominator for each wave is the number observed at each wave at risk of seroconverting (i.e., prior seroconverters are eliminated). The gender-specific prevalence of sexual risk behavior is nearly identical at each wave.
Survival analysis (i.e., Cox proportional hazard survival model) of HIV seroconversion incorporated the two behavioral risk variables, injecting risk and sexual risk, that varied over interviewing waves, as well as gender, age, race, and site/neighborhood. The only variable found to be associated with HIV seroconversion was the time-dependent behavioral risk variable "injecting risk" (RR = 9.8, p < .001). The time-dependent sexual risk variable and all demographic variables were not found to be associated with seroconverting.
Risky injection behavior was reported by 100 percent of the study cohort at baseline (1988) and only 14 percent at wave six (1992). (Neither risk behavior nor HIV status was an initial recruitment factor.) The observed decline in seroincidence in this cohort appears to have resulted from changes in drug-use risk behaviors attributable, at least in part, to the intervention.
This study design obviously cannot completely rule out alternative explanations for the observed decline in seroconversion in the studied cohort. One such alternative explanation could be that the findings were the result of other prevention efforts, especially given the increased amount of media attention directed at HIV risk reduction and other community efforts occurring
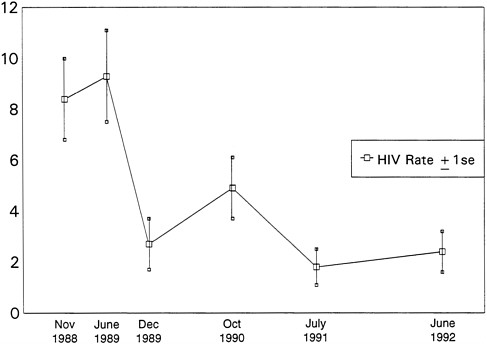
FIGURE 6.1 HIV seroconversion (per 100 person years) among out-of-treatment injection drug users in Chicago 1988-1992. SOURCE: Prevention of New HIV Infections Among Out-of-Treatment Injection Drug Users: A Four Year Prospective Study (Wiebel et al., 1994).
during that period. However, two independent data sets do provide support for the potential causal program effect on the observed findings.
During the sixth reinterview wave, the researchers recruited from nearby neighborhoods 248 previously unsurveyed street injection drug users. The prevalence of drug-use risk behavior among those who were seronegative was 50 percent for those with no reported outreach program contact. In addition, NIDA's surveys of drug treatment entrants in Chicago over a number of years include questions about needle sharing. The prevalence of self-reported needle sharing among 500 injection drug user treatment entrants surveyed in 1992 was 67 percent. The higher rates of risky injections found in these independent samples of injection drug users in the same communities lend some credence to the potential causal effect of the intervention on the observed decline in injection drug-use risk behaviors and HIV seroconversion. Nonetheless, it should be noted that these two separate data sets reflect substantially lower levels of drug-use risk behaviors than were observed at baseline among members of the intervention group (i.e., 100 percent). This implies that one cannot completely rule out some
TABLE 6.1 Percentage of Those at Risk of Converting (HIV Negative) Engaging in Sex Risk and Injecting Risk Behaviors
|
Midpoint |
May 1988 |
November 1988 |
June 1989 |
December 1989 |
October 1990 |
July 1991 |
June 1992 |
|
Sexual risk |
71 |
66 |
60 |
61 |
— |
55 |
45 |
|
Injecting risk |
100 |
54 |
46 |
37 |
27 |
19 |
14 |
|
SOURCE: Prevention of New HIV Infections Among Out-of-Treatment Injection Drug Users: A Four Year Prospective Study (Wiebel et al., 1994). |
|||||||
partial secular trend effect on the intervention group's observed drug-use risk behavior change.
There are a number of other possible uncontrolled factors that could account for the observed reduction in the injection drug-use risk behaviors and HIV seroconversion. Although participants were not "selected" for services on the basis of their measured level of risk, it is possible that they agreed to become a participant when their needle risk was at an abnormally high level. Due to maturation or instability in their levels of use, we would expect that on subsequent assessment, these clients would reveal a lower level of risk behavior (i.e., they would regress toward the population mean). Without a control group, it is not possible to assess the extent to which trait instability or maturation is at work.
Saturation of the at-risk pool of injectors (which happens when most of the susceptible injectors have already been infected) is yet another possible alternative explanation for the result depicted in Figure 6.1. Although the Chicago researchers contend that saturation could not account for the reported decline in seroincidence, because HIV seroprevalence remained constant in their cohort at 32 percent throughout their prospective study, it is still possible that saturation did impact on their findings. That is, saturation among those who remained susceptible (susceptibility being defined on the basis of risk of exposure through drug-use risk behavior) may have occurred, given that a substantial proportion of the cohort removed their risk of exposure (i.e., their drug-use risk behavior) and were no longer in the pool of susceptibles (assuming no risk of exposure through sexual-risk behavior).
Nonetheless, the data tend to suggest that there was a decrease in risky drug-use behavior over time, with an attendant decrease in new HIV infection in the cohort as originally assembled (irrespective of other risk behaviors).3 It also highlights that bleach distribution occurs in the context of street outreach, making it difficult to disentangle bleach effect per se from the outreach worker effect. Moreover, the corresponding decline in self-report drug-use risk behavior and HIV incidence does also provide supporting evidence for the construct validity of self-report data.
The null results of epidemiologic studies reviewed earlier in conjunction with the program evaluation results reviewed above reveal the potential critical role of outreach activities in achieving meaningful behavioral change. In fact, these investigators (Wiebel et al., 1994) reported that injection drug users in the study group most frequently reduced risk not by always using bleach to clean injection equipment, but, rather, by eliminating the sharing of syringes and other injection equipment altogether. These changes in injection practices occurred over a time when there was no syringe exchange in Chicago, nor were there local laws permitting the possession of
nonprescription needles, which suggests a high level of motivation on the part of cohort members to avoid HIV infection.
Therefore, in light of these findings, it would be misleading to consider the outreach efforts undertaken solely as bleach distribution programs.
Limitations of Bleach Disinfection
Although bleach, when properly used, provides an effective disinfectant in the health care setting, a number of variables may limit its effectiveness when used by injection drug users to disinfect contaminated needles and syringes. The physical and biological parameters that govern the effectiveness of any disinfectant (discussed earlier in this chapter) are critically important factors in determining whether the cleaning and disinfectant method used by a given injection drug user will result in effective disinfection of their injection equipment. Needles and syringes are not designed for reuse and are difficult to completely clean and disinfect. The presence of organic matter in the residual blood, either liquid or dried, remaining in the contaminated injection equipment decreases the potency of bleach as a disinfectant. The presence of blood in a previously used syringe necessitates thorough rinsing with water prior to bleach treatment. Residual, potentially infectious HIV present within clotted blood may be even more resistant to elimination by cleaning and disinfection methods. Furthermore, the infectivity of HIV-infected cells found within the blood remaining in used needles and syringes may be more resistant to disinfection than the cell-free or cell-associated HIV preparations studied in laboratory settings.
Reports by Shapshak and colleagues (Shapshak et al., 1993, 1994; McCoy et al., 1994) indicate that complete inactivation of preparations of cell-free HIV that had been concentrated (''pelleted") by centrifugation required prolonged (greater than 30 second) exposure to undiluted bleach. Although the laboratory model used in this study does not directly approximate the real-life circumstances of bleach disinfection of contaminated injection equipment, these reports resulted in significant concern and confusion about the validity of previous recommendations concerning bleach disinfection methods.
Another factor that may limit the effectiveness of disinfection efforts is that needles and syringes may become recontaminated if other drug injection paraphernalia, such as the cooker or the cotton used, are not cleaned or replaced (Koester et al., 1990). However, two studies (Marmor et al., 1987; Samuels et al., 1991) have found no association between shared cookers and HIV seroprevalence. Sharing water for mixing drugs or for rinsing the syringe after bleach use is another source of contamination (Koester et al., 1990; Inciardi and Page, 1991). Various methods of dividing drugs for sharing purposes also hold the potential for contamination (see Chapter 1).
These practices include frontloading (removing the needle from one syringe and filling that syringe by inserting the needle from another drug-filled syringe directly into the barrel), and backloading (removing the plunger from the back of the syringe and filling that syringe through the back with drugs from a second syringe). Studies of these factors have to date yielded conflicting results, with a report from New York finding a positive association between frontloading and HIV seroprevalence (Jose et al., 1992), and a study in Baltimore (Samuels et al., 1991) reporting similar seroprevalence rates for injection drug users who did and did not practice frontloading. Samuels and colleagues noted, however, that these results do not mean "that the behaviors are without risk of HIV infection among [injection drug users] … (but) that the associated risks are probably low relative to other (HIV risk) behaviors."
Impact of Bleach Disinfection on HIV Transmission
If disinfection of injection equipment is an effective means of preventing HIV infection, then HIV seroconversion rates would be expected to be lower among injection drug users who consistently disinfect their injection equipment. Prior to the initiation of the original CHOW program in San Francisco, Chaisson and colleagues (1987) found no protective effect for those injection drug users who reported cleaning their needles and syringes with boiling water or alcohol. Later, Moss and Chaisson (1988) reported that there was no relationship found between behavior change, including reported bleach use, and seroprevalence. In contrast, Watters (1994) reported that, although the seroprevalence rate among heterosexual injection drug users in San Francisco nearly doubled between 1986 and 1987, there was a stabilization of seroprevalence following the introduction of the bleach distribution and other HIV prevention programs. However, seroprevalent infections for which the date of onset of infection is unknown are not the optimal endpoint from which to measure effectiveness of an intervention, since other risk reduction factors, such as decreased needle sharing, were operating simultaneously. In such circumstances, any change in seroprevalence cannot be attributed solely to the bleach distribution programs.
More recently, Vlahov and colleagues have studied the relationship between the reported injection equipment disinfection practices of injection drug users and rates of HIV seroconversion. Their initial report (Vlahov et al., 1991) described a small nested case-control study of HIV seroconverters and HIV-seronegative injection drug users by their self-report frequency of using disinfectants all the time, less than all the time, and no use of disinfectants. Although no statistically significant protective effect was detected, the results of this study suggested a possible modest protective effect of bleach or alcohol disinfection among injection drug users who reported
always using disinfectants. A follow-up study by these investigators reexamined this result (Vlahov et al., 1994), using a larger sample size and examining the influence of potentially confounding variables on the study outcome, including the effect of drug-use variables, the possibility of sexual acquisition of HIV infection (Solomon et al., 1993), and the potential for study participants to provide socially desirable responses to the investigator's questions concerning disinfection practices (Latkin et al., 1993). Nevertheless, similar null results were obtained, with injection drug users who reported using disinfectants all the time compared with those who reported no use of disinfectants (Vlahov et al., 1994). Thus, bleach use, as practiced in Baltimore in the early 1990s, did not eliminate or substantially reduce the risk of HIV seroconversion among injection drug users who reported using it all the time.
Similar findings were subsequently found in New York (Titus et al., 1994), in a case-control study. After adjusting for possible confounders, no evidence that bleach use protected against incident HIV infection was reported. Another study of HIV seroconversion among injection drug users in San Francisco showed no protective effect with bleach disinfection (Moss et al., 1994). It remains to be established whether these disappointing results derive from exaggerated reporting of injection drug users about their actual disinfection practices, the use of inadequate disinfection methods, contamination of water or ancillary injection paraphernalia with HIV, or the inability of bleach or alcohol to effectively disinfect contaminated needles and syringes within the context of their actual use by injection drug users.
Responding to concerns arising from laboratory studies about prevailing methods for bleach inactivation of HIV and epidemiologic studies that suggest little, if any, protective effect of needle and syringe disinfection efforts, the Centers for Disease Control, the National Institute on Drug Abuse, and the Center for Substance Abuse Treatment (CSAT) sponsored a meeting in February 1993 at which the current status of bleach outreach and research was reviewed. Discussions from this meeting provided the basis for the publication of provisional guidelines on bleach disinfection (Curran et al., 1993). These published guidelines included the following recommendations:
-
Cleaning should be done twice—once immediately after use and again just before reuse of needles and syringes.
-
Before using bleach, wash out the needle and syringe by filling them several times with clean water. (This will reduce the amount of blood and other debris in the syringe. Blood reduces the effectiveness of bleach.)
-
Then, use full-strength liquid household bleach (not diluted bleach).
-
Completely fill the needle and syringe with bleach several times. (Some suggest filling the syringe at least three times.)
-
The longer the syringe is completely full of bleach, the more likely HIV will be inactivated. (Some suggest the syringe should be full of bleach for at least 30 seconds.)
-
After using bleach, rinse the syringe and needle by filling several times with clean water. Don't reuse water used for initial prebleach washing; it may be contaminated.
-
For every filling of the needle and syringe with prebleach wash water, bleach, and rinse water, fill the syringe completely ("to the top").
-
Shaking and tapping the syringe are recommended when the syringe is filled with prebleach wash water, bleach, and rinse water. Shaking the syringe should improve the effectiveness of all steps.
-
Taking the syringe apart (removing the plunger) may improve the cleaning/disinfection of parts (e.g., behind the plunger) that might not be reached by solutions in the syringe.
Although the intent of these guidelines was to provide clear recommendations to be used by outreach workers in risk reduction programs in the injection drug user community and to attempt to maximize the effectiveness of bleach disinfection methods, responses to this revision have been mixed. Some, including researchers as well as injection drug users and outreach workers, have interpreted the revised guidelines to mean that bleach is ineffective and have called for using bleach only as a last resort (Donoghoe and Power, 1993). Other outreach workers have expressed concern that changing the guidelines undermines their credibility with injection drug users (Haverkos and Jones, 1994). Whereas implementing these recommendations is likely to increase the effectiveness of bleach disinfection of needles and syringes, the lack of adequate laboratory models of injection equipment disinfection makes this difficult to document. Given the importance of limiting HIV transmission among injection drug users, the margin of safety in disinfection methods is desirable. However, should recommended disinfection strategies prove too complex or cumbersome, they may be less likely to be followed.
At least one report indicates that injection drug users accept the majority of the guidelines (Corby, 1993). In the 5 months following publication of the guidelines, 17 focus groups were held in eight cities across the country to gauge injection drug users' acceptance of the new guidelines. Two of the eight recommended practices were judged as unlikely to be adopted: cleaning both before and after injection and disassembling the syringe. Many injection drug users felt that the first was unnecessary and the second would damage the equipment or would be too difficult to do.
SUMMARY
The reuse of needles and syringes that are intended for use by a single individual on a single occasion is an inherently unsafe practice. The repeated use of needles and syringes commonly violates their sterility and predisposes the user to life-threatening infections from a wide variety of bacterial and viral pathogens. The sharing of injection equipment by injection drug users provides an efficient means of transmitting HIV, hepatitis B and C viruses, and the human T-cell leukemia viruses (HTLV-I and HTLV-II). The only reliable ways to limit the transmission of HIV and other viral infections among injection drug users are either to refrain from injecting drugs or not to share injection equipment with other users. If an injection drug user is unable or unwilling to refrain from using injection drugs, the use of a sterile, disposable needle and syringe for each injection represents the next best alternative. However, in circumstances in which supplies of sterile needles and syringes are inadequate or unavailable, disinfection strategies provide another means to decrease the risk of transmission of HIV and other infectious agents between injection drug users who are compelled to share injection equipment.
Bleach disinfection of contaminated needles and syringes is the most commonly used and familiar strategy employed by injection drug users, and, if used according to current recommendations made by the Centers for Disease Control and Prevention, the National Institute on Drug Abuse, and the Center for Substance Abuse Treatment, is likely to be effective in disinfecting contaminated injection equipment. Definition of the precise efficacy of bleach as a disinfectant of HIV-contaminated needles and syringes has been hampered by the lack of a laboratory model that faithfully recapitulates the real-life circumstances of injection drug use. Unfortunately, too little attention and too few resources have been devoted to studies to define the simplest, most effective disinfection methods for injection equipment used by injection drug users.
The inability of initial epidemiologic studies to demonstrate a significant protective effect of disinfection practice against HIV infection of injection drug users is disconcerting. Unfortunately, evaluation and epidemiologic studies performed to date do not clearly identify the reasons for the similar rates of incident HIV infections reported between those who always clean their needles and syringes and those who never do. It is not yet known whether this results from inadequate disinfection techniques used by injection drug users who become infected, their overestimation of the frequency of their use of needle and syringe disinfection in interviews with researchers, or physical or biological factors that compromise the ability of bleach, a potent disinfectant in other circumstances, to effectively disinfect contaminated injection equipment. Certainly, a number of observational
studies indicate that injection drug users may use variable, and often clearly inadequate, disinfection strategies. Furthermore, adherence to recommended disinfection protocols after training may decrease with time. Increasingly complex disinfection strategies may be less likely to be retained or adopted by injection drug users.
Additional research is needed to identify approaches that most effectively transmit to injection drug users the importance of disinfection of contaminated injection equipment and that ensure maximal compliance with recommended procedures. In the interim, promotion of a clear, consistent method of disinfection procedures, such as those recommended in the recent CDC/NIDA/CSAT publication, is essential to help decrease the risk of HIV infection for injection drug users who continue to share needles and syringes.
Bleach can be an effective and potentially life-saving intervention for injection drug users who share needles and syringes. Bleach distribution programs have been a popular and effective component of community health outreach efforts to decrease HIV risk behaviors among injection drug users. Continued advocacy of bleach disinfection will be necessary even if sterile needles and syringes become more widely available through exchange programs or the relaxation of prescription laws.
Research efforts to better model the efficacy of disinfection strategies for blood-contaminated needles and syringes should be actively encouraged. Emphasis should be placed on models that are relevant to the typical practices and circumstances of injection drug use behaviors. Additional field research studies are needed to further evaluate the effectiveness of bleach disinfection in decreasing the risk of HIV infection among injection drug users. A better understanding of the reasons for the limited protective impact of needle and syringe disinfection practices documented to date is essential. Research to identify effective education strategies to maximize injection drug user familiarity with and practice of effective bleach disinfection methods is needed.
Conclusions
The panel concludes that:
-
Bleach, if used according to the recommendations of the Centers for Disease Control and Prevention, the National Institute on Drug Abuse, and the Center for Substance Abuse Treatment, is likely to be an effective HIV prevention strategy for injection drug users who share needles and syringes.
-
Concerted efforts are essential to increase the awareness of injection
-
drug users of the importance of disinfecting shared injection equipment and the importance of following the appropriate procedures.
Recommendations
The panel recommends that:
-
Health research funding agencies (e.g., the National Institutes of Health, the Centers for Disease Control and Prevention, and the Agency for Health Care Policy and Research) should support research directed toward identifying the simplest to use and most effective disinfection strategies, employing agents that are readily available to injection drug users.
-
Health research funding agencies (e.g., the National Institutes of Health, the Centers for Disease Control and Prevention) should support development of effective education strategies for familiarizing injection drug users with methods of effective bleach disinfection.
NOTES
|
1. |
The disparity between the number of follow-up and the number of individuals included in the analyses (in particular, the sex partners) is not discussed in the final report. |
|
2. |
The follow-up rates per site were not reported for all sites. Of those reported, the rates ranged from a low of 2 percent to a high of 74 percent. |
|
3. |
The Chicago study was completed before the needle exchange evaluation study reviewed in Appendix A. As the reader will note, this cohort of injection drug users (which represented a sizable proportion of the needle exchange sample) had experienced substantial reductions in drug-use risk behavior and HIV incidence prior to the needle exchange evaluation study. This provides an historical context for reviewing the needle exchange evaluation project presented in Appendix A. |
REFERENCES
Battjes, R., and R. Pickens 1992 Trends in HIV Infection and AIDS Risk Behaviors Among Intravenous Drug Users in Selected U.S. Cities. Abstract No. PoC 4247. International Conference on AIDS 8(2):C286. 1993 Trends in HIV Infection and AIDS Risk Behaviors Among Intravenous Drug Users in Selected U.S. Cities. Abstract No. PO-C15-2950. International Conference on AIDS 9(2):709.
Becker, M.H., and J.G. Joseph 1988 AIDS and behavioral change to reduce risk: A review. American Journal of Public Health 78(4):394-410.
Becker, G.L., S. Cohen, and R. Borer 1974 The sequelae of accidentally injecting sodium hypochlorite beyond the root apex. Oral Surgery 38(4):633-638.
Block, S.S., ed. 1983 Definition of terms. Pp. 877-881 in Disinfection, Sterilization and Preservation, 3rd edition. Philadelphia, PA: Lea & Febiger.
Brown, B.S. 1994 Review of the Effects of Bleach Distribution Programs. Paper commissioned by the Panel on Needle Exchange and Bleach Distribution Programs.
Brown, B.S., and G.M. Beschner, eds. 1993 Handbook on Risk of AIDS—Injection Drug Users and Sexual Partners . Westport, CT: Greenwood Press.
Celentano, D.D., D. Vlahov, A.S. Menon, and B.F. Polk 1991 HIV knowledge and attitudes among intravenous drug users: Comparison to the U.S. population and by drug use behaviors. Journal of Drug Issues 21(3):635-649.
Centers for Disease Control 1986 Recommendations for providing dialysis treatment to patients infected with human T-lymphotrophic virus type III/lymphadenopathy-associated virus. Morbidity and Mortality Weekly Report 35(23):376-378, 383. 1987 Recommendations for prevention of HIV transmission in health-care settings. Morbidity and Mortality Weekly Report 1S-18S.
Chaisson, R.E., A.R. Moss, R. Onishi, D. Osmond, and J.R. Carlson 1987 Human immunodeficiency virus infection in heterosexual intravenous drug users in San Francisco. American Journal of Public Health 77(2):169-172.
Conte, J.E., Jr. 1986 Infection with human immunodeficiency virus in the hospital. Epidemiology, infection control, and biosafety considerations. Annals of Internal Medicine 105(5):730-736.
Corby, N. 1993 Injection Drug Users Willingness to Adopt New Bleaching Recommendations. A paper presented at the 6th Annual AIDS Update in San Francisco, California.
Curran, J.W., L.W. Scheckel, and R.A. Millstein 1993 HIV/AIDS Prevention Bulletin, April 19. Washington, DC: Department of Health and Human Services, Public Health Service, and Centers for Disease Control and Prevention.
Des Jarlais, D.C., S.R. Friedman, and D. Strug 1986 AIDS and needle sharing within the IV-drug use subculture. Pp. 111-125 in The Social Dimensions of AIDS: Methods and Theory, D. Feldman and T. Johnson, eds. New York, NY: Preager.
Donoghoe, M.D., and R. Power 1993 Household bleach as disinfectant for use by injecting drug users. Lancet 341(8861):1658.
Edlin, B.R, K. Irwin, Y. Serrano, P. Evans, and C. McCoy 1993 The Impact of Cocaine Smoking on Injection Practices of Street-Recruited Drug Injectors. Abstract No. PO-C15-2952. International Conference on AIDS 9(2):709.
Favero, M.S., and W.W. Bond 1991 Disinfection, sterilization and preservation. In Chemical Disinfection of Medical and Surgical Materials. Philadelphia, PA: Lea & Febiger.
Feldman, H.W., and P. Biernacki 1988 The ethnography of needle sharing among IVDUs and implications for public policies and intervention strategies. In Needle Sharing Among IVDUs: National and International Perspectives, R.J. Battjes and R.W. Pickens, eds. NIDA Research Monograph No. 80:28-39. Washington, DC: National Institute on Drug Abuse.
Flynn, N., S. Jain, E.M. Keddie, J.R. Carlson, M.B. Jennings, H.W. Haverkos, N. Nassar, R. Anderson, S. Cohen, and D. Goldberg 1994 In vitro activity of readily available household materials against HIV-1: Is bleach enough? Journal of Acquired Immune Deficiency Syndromes 7(7):747-753.
Friedman, S.R., M. Sufian, A. Neaigus, B. Stepherson, et al. 1990 Organizing Drug Users Against AIDS: A Comparison With Outreach for Producing Risk Reduction. Abstract No. S.C. 733. International Conference on AIDS 6(3):272.
Froner, G.A., G.W. Rutherford, and M. Rokeach 1987 Injection of sodium hypochlorite by intravenous drug users [letter]. Journal of the American Medical Association 258(3):325.
Gleghorn, A.A. 1993 Limitations of Bleach Use. A paper presented at the 6th Annual AIDS Update, San Francisco, California. 1994 Use of bleach by injection drug users. Pp. 294-302 in Proceedings, Workshop on Needle Exchange and Bleach Distribution Programs. Washington, DC : National Academy Press.
Gleghorn, A.A., M.C. Doherty, D. Vlahov, D.D. Celentano, and T.S. Jones 1994 Inadequate bleach contact times during syringe cleaning among injection drug users. Journal of Acquired Immune Deficiency Syndromes 7(7):767-772.
Haverkos, H.W., and T.S. Jones 1994 HIV, drug-use paraphernalia, and bleach. Journal of Acquired Immune Deficiency Syndromes 7(7):741-742.
Hopkins, W. 1988 Needle sharing and street behavior in response to AIDS in New York City. In Needle Sharing Among IVDUs: National and International Perspectives, R.J. Battjes and R.W. Pickens, eds. NIDA Research Monograph No. 80:18-27. Washington, DC: National Institute on Drug Abuse.
Hugo, W.B., and A.D. Russell 1982 Types of antimicrobial agents. Pp. 8-106 in Principles and Practice Disinfection, Preservation and Sterilization, W.B. Hugo and G.A. Ayliffe, eds. Oxford, England: Blackwell.
Inciardi, J.A., and J.B. Page 1991 Drug sharing among intravenous drug users. AIDS 5(6):772-773.
Ingraham, A.S. 1992 The chemistry of disinfectants and sterilants. Contemporary Topics 31:18-23.
Jose, B., S.R. Friedman, A. Neaigus, R. Curtis, and D.C. Des Jarlais 1992 "Frontloading" Is Associated With HIV Infection Among Drug Injectors in New York City. Abstract No. ThC1551. International Conference on AIDS Th76.
Klein, M. and A. Deforest 1963 Antiviral action of germicides. Soap Chemistry Specialist 39:70-72, 95-97. 1965a Principles of viral inactivation. Pp. 422-434 in Disinfection, Sterilization, and Preservation, S.S. Block, ed. Philadelphia, PA: Lea & Febiger. 1965b The chemical inactivation of viruses. Federation Proceedings 24:319.
Koester, S., R. Booth, and W. Wiebel 1990 The risk of HIV transmission from sharing water, drug-mixing containers and cotton filters among intravenous drug users. International Journal of Drug Policy 1(6):28-30.
Latkin, C.A., D. Vlahov, J.C. Anthony, S. Cohn, and W. Mandell 1992 Needle-cleaning practices among intravenous drug users who share injection equipment in Baltimore, Maryland. International Journal of the Addictions 27(6):717-725.
Latkin, C.A., D. Vlahov, and J.C. Anthony 1993 Socially desirable responding and self-reported HIV infection risk behaviors among intravenous drug users. Addiction 88:517-526.
Longshore, D., M.D. Anglin, K. Annon, and S. Hsieh 1993 Trends in self-reported HIV risk behavior: Injection drug users in Los Angeles. Journal of Acquired Immune Deficiency Syndromes 6(1):82-90.
Marmor, M., D.C. Des Jarlais, H. Cohen, S.R. Friedman, S.T. Beatrice, N. Dubin, W. el-Sadr, D. Mildvan, S. Yancovitz, U. Mathor, et al. 1987 Risk factors for HIV infection with human immunodeficiency virus among intravenous drug users in New York City. AIDS 1(1):39-44.
Martin, L.S., J.S. McDougal, and S.L. Lokoski 1985 Disinfection and inactivation of the human T-lymphotrophic virus type-III/lymphadenopathy-associated virus. Journal of Infectious Diseases 152(2):400-403.
McCoy, C.B., H.V. McCoy, P. Shapshak, N. Weatherby, et al. 1992 HIV Intervention Strategies Based on Field and Laboratory Studies. Abstract No. PoC 4292. International Conference on AIDS 8(2):C293.
McCoy, C.B., J.E. Rivers, H.V. McCoy, P. Shapshak, N.L. Weatherby, D.D. Chitwood, J.B. Page, J.A. Inciardi, and D.C. McBride 1994a Compliance to bleach disinfection protocols among injecting drug users in Miami. Journal of Acquired Immune Deficiency Syndromes 7(7):773-776.
McCoy, C.B., P. Shapshak, S.M. Shah, H.V. McCoy, J.E. Rivers, J.B. Page, D.D. Chitwood, N.L. Weatherby, J.A. Inciardi, D.C. McBride, D.C. Mash, and J.K. Watters 1994b HIV-1 prevention: Interdisciplinary studies and reviews on efficacy of bleach and compliance to bleach prevention protocols. Pp. 255-283 in Proceedings, Workshop on Needle Exchange and Bleach Distribution Programs. Washington, DC: National Academy Press.
McKee, M.M., V.A. Caine, and L.M. Bryson 1992 Promoting Safe Injection Technique and Condom Usage Among Injection Drug Users in Indianapolis, IN. Abstract No. PoD 5085. International Conference on AIDS 8(2):D401.
Meyers, M., R. Nemeth-Coslett, F. Snyder, and P. Young 1990 Needle Cleaning Methods Reported by Intravenous Drug Users Who Are Not in Drug Treatment. Abstract No. S.C. 743. International Conference on AIDS 6(3):274.
Moss, A.R., and R.E. Chaisson 1988 AIDS and intravenous drug use in San Francisco. AIDS and Public Policy Journal 3:37-41.
Moss, A.R., K. Vranizan, R. Gorter, P. Bacchetti, J. Watters, and D. Osmond 1994 HIV seroconversion in intravenous drug users in San Francisco, 1985-1990. AIDS 8:223-231.
Murphy, S. 1987 Intravenous drug use and AIDS: Notes on the social economy of needle sharing. Contemporary Drug Problems Fall:373-395.
National Institute on Drug Abuse 1994 Outreach/Risk Reduction Strategies for Changing HIV-Related Risk Behaviors Among Injection Drug Users: The National AIDS Demonstration Research (NADR) Project. NIH Publication No. 94-3726. Bethesda, MD: National Institutes of Health.
Neaigus, A., M. Sufian, S.R. Friedman, D.S. Goldsmith, B. Stepherson, P. Mota, J. Pascal, and D.C. Des Jarlais 1990 Effects of outreach intervention on risk reduction among intravenous drug users. AIDS Education and Prevention 2(4):253-271.
Newmeyer, J.A. 1988 Why bleach? Fighting AIDS contagion among intravenous drug users: The San Francisco experience. Journal of Psychoactive Drugs 20(2):159-163.
Pantaleo, G., C. Graziosi, and A.S. Fauci 1993 New concepts in the immunopathogenesis of human immunodeficiency virus infection. New England Journal of Medicine 328(5):327-335.
Resnick, L., K. Veren, Z. Salahuddin, S. Tondreau, and P.D. Markham 1986 Stability and inactivation of HTLV-III/LAV under clinical and laboratory environments. Journal of the American Medical Association 255(14):1887-1891.
Rhame, F.S. 1986 The inanimate environment. Pp. 233-249 in Hospital Infections , J.V. Bennett and P.S. Brachmann, eds. Boston, MA: Little, Brown and Co.
Rutala, W.A. 1987 Disinfection, sterilization, and waste disposal. Pp. 257-282 in Prevention and Control of Nosocomial Infections, R.P. Wenzel, ed. Baltimore, MD: Williams and Wilkins. 1990 APIC guideline for selection and use of disinfectants. American Journal of Infection Control 18(2):99-117.
Rutala, W.A., and E.C. Cole 1984 Antiseptics and disinfectants—Safe and effective? Infection Control 5(5):215-218.
Rutala, W.A., and D.J. Weber 1987 Environmental issues and nosocomial infections. Pp. 131-171 in Clinics and Critical Care Medicine: Infection Control in Intensive Care. New York, NY: Churchill Livingston.
Samuels, J.F., D. Vlahov, J.C. Anthony, L. Solomon, and D.D. Celentano 1991 The practice of ''frontloading" among intravenous drug users: Association with HIV-antibody [letter]. AIDS 5(3):343.
Sattar, S.A., and V.S. Springthorpe 1991 Survival and disinfectant inactivation of the human immunodeficiency virus: A critical review. Reviews of Infectious Diseases 13(3):430-447.
Shapshak, P., C.B. McCoy, J.E. Rivers, D.D. Chitwood, D.C. Mash, N.L. Weatherby, J.A. Inciardi, S.M. Shah, and B.S. Brown 1993 Inactivation of human immunodeficiency virus-1 at short time intervals using undiluted bleach [letter]. Journal of Acquired Immune Deficiency Syndromes 6(2):218-219.
Shapshak, P., C.B. McCoy, S.M. Shah, J. B. Page, J.E. Rivers, N.L. Weatherby, D.D. Chitwood, and D.C. Mash 1994 Preliminary laboratory studies of inactivation of HIV-1 in needles and syringes containing infected blood using undiluted household bleach. Journal of Acquired Immune Deficiency Syndromes 7(7):754-759.
Solomon, L., J. Astemborski, D. Warren, A. Muñoz, S. Cohn, D. Vlahov, and K.E. Nelson 1993 Difference in risk factors for human immunodeficiency virus type 1 seroconversion among male and female intravenous drug users. American Journal of Epidemiology 137:892-898.
Sorensen, J.L., J. Guydish, M. Costantini, and S.L. Batki 1989 Changes in needle sharing and syringe cleaning among San Francisco drug abusers. New England Journal of Medicine 320(12):807.
Spire, B., F. Barré-Sinoussi, L. Montagnier, and J.C. Chermann 1984 Inactivation of lymphadenopathy associated virus by chemical disinfectants. Lancet 2(8408):899-901.
Titus, S., M. Marmor, D. Des Jarlais, M. Kim, H. Wolfe, and S. Beatrice 1994 Bleach use and HIV seroconversion among New York City injection drug users. Journal of Acquired Immune Deficiency Syndromes 7(7):700-704.
Van Houton, J., and M.D. Hayre 1991 Disinfectants and sterilants: Their chemistry, use and evaluation. AALAS Bulletin 30:24-27.
Vlahov, D., A. Muñoz, D.D. Celentano, S. Cohn, J.C. Anthony, H. Chilcoat, and K.E. Nelson 1991 HIV seroconversion and disinfection of injection equipment among intravenous drug users, Baltimore, Maryland. Epidemiology 2(6):444-446.
Vlahov, D., J. Astemborski, L. Solomon, and K.E. Nelson 1994 Field effectiveness of needle disinfection among injection drug users. Journal of Acquired Immune Deficiency Syndromes 7(7):760-766.
Watters, J. 1987 A street-based outreach model of AIDS prevention for intravenous drug users: Preliminary evaluation. Contemporary Drug Problems Fall:411-423. 1994 Historical perspective on the use of bleach in HIV/AIDS prevention. Journal of Acquired Immune Deficiency Syndromes 7(7):743-746.
Wiebel, W.W. 1990 Identifying and gaining access to hidden populations. In The Collection and Interpretation of Data from Hidden Populations, E.Y. Lambert, ed. NIDA Research Monograph No. 98:4-11. Washington, DC: National Institute on Drug Abuse. 1993 The Indigenous Leader Outreach Model, Intervention Manual. National Institute on Drug Abuse, Department of Health and Human Services Publication no. 93-3581.
Wiebel, W.W., D. Chene, and W. Johnson 1990 Adoption of Bleach Use in a Cohort of Street Intravenous Drug Users in Chicago. Abstract No. S.C. 742. International Conference on AIDS 6(3):274.
Weibel, W.A., Jimenez, N. Johnson, L. Ouellet, L. Lampinen, J. Murray, B. Jovanovic, and M.U. O'Brien 1993 Positive Effect on HIV Seroconversion of Street Outreach Intervention with IDU in Chicago: 1988-1992. Presented at the Ninth International Conference on AIDS, June 6-11, Berlin.
Wiebel, W.W., W. Johnson, B. Jovanovic, and J. Murray 1994 Prevention of New HIV Infections Among Out-of-Treatment Injection Drug Users: A Four Year Prospective Study. Draft paper.































