3
Identifying Clues and Testing Hypotheses
The challenge in elucidating the causes underlying the initial steep decline and continuing gradual decline in the western population of Steller sea lions (Eumetopias jubatus) lies in unraveling the complex multispecies relationships that characterize marine ecosystems. Direct and indirect trophic linkages among the members of these ecosystems present the potential for a population change in one species to profoundly influence the abundance of other species at other trophic levels. There have been several approaches to this problem, including analysis of food webs with few species (Elton, 1927), interaction webs containing a manageable number of multispecies interactions, and ecosystem models. The latter, while incorporating elements of the physical environment, often of necessity aggregate species into functional groupings, thus disguising the ecological roles of individual species (Paine, 1980).
Some blend of the above provides a useful conceptual framework for evaluating various hypotheses concerning the population collapse of Steller sea lions in western Alaska. Both population and ecosystem models are valuable as tools for identifying what types of factors could explain the population decline and for evaluating the available data for consistency with these potential causes. It is important to note, though, that models cannot compensate for the lack of fundamental data for most of the period of decline. Although models can be used to explore potential mechanisms, it will not be possible to establish the causal basis of the sea lion population decline based on these modeling exercises.
POPULATION MODELS
Population models have been used in conservation biology to estimate population viability, to determine the relative impact of different life history processes on population growth rates, and to predict the effects of management strategies on growth rates. Population viability analysis usually focuses on estimating the probability of extinction over a given time period and is conducted in the early listing stage for endangered species (Groom and Pascual, 1998; Gerber and VanBlaricom, 2001). This approach is limited, however, when there is a need to know which processes (fecundity, growth, or survival) in the life history of an organism are most likely to constrain recovery. To answer this question, researchers often conduct elasticity analyses (the proportional change in population growth rate due to a proportional change in a vital rate such as survival) or stochastic simulations (testing the effect of random variations in population vital rates across their statistical distribution on variations in population growth rate) to determine how variations in particular processes influence expected population growth rate (Heppell et al., 2000). In addition, these analyses can be used to compare the potential explanatory power of alternative hypotheses for population declines.
Population models vary in their structure from simple, deterministic, age-based matrix models to stochastic, individual-based models. Age-based models assume that all individuals of the same age experience similar dynamics. Individual-based models allow variation among individuals of the same age to influence population dynamics. But data are so limiting for most threatened species that the models are often relatively simple, leaving out features that may be important, such as stochasticity or well-defined geographic variation. Still these models have proven to be a useful way to integrate what we know, to point out unequivocally what we need to know, and to guide managers toward those solutions most likely to contribute to population recovery.
There have been a number of population models developed for Steller sea lions (Pascual and Adkison, 1994; York, 1994; York et al., 1996; Gerber and VanBlaricom, 2001). Each addresses different issues regarding population viability, population trends in the context of local populations, and regional metapopulations. Elasticity analyses of 44 population models based on 50 mammal life tables place Steller sea lions in a larger context. Populations of mammals with life histories like Steller sea lions are about twice as sensitive to changes in adult survival as to changes in juvenile survival (Heppell et al., 2000). Adult survival elasticity is 10-fold higher than elasticity related to fertility. So on first principles, factors that cause adult mortality might be expected to be much more important than those that cause juvenile mortality or that reduce fertility. York’s (1994) model
suggests that the observed 50% decline in adult sea lions from the mid-1970s to the mid-1980s and the concurrent increase in average age resulted from a 10 to 20% decrease in juvenile (ages 0 to 3) survival coupled with a very small decline in adult survival. Hence, the shift in age structure suggests that juvenile sea lions were disproportionately affected by whatever factor (or factors) was responsible for the steep population decline.
Pascual and Adkison (1994) used York’s model to examine a series of alternative hypotheses to explain the decline in sea lions. They argued that deterministic transient population behavior (time-delayed population response to an earlier event), historical pup harvesting, and short-term environmental stochasticity are unlikely to explain the decline. They concluded that from 1975 to 1990 sea lion declines were likely caused by a long-term or catastrophic change in environmental conditions that produced a 30 to 60% reduction in juvenile survival or a 70 to 100% reduction in fecundity because of food limitation. Both Pascual and Adkison and York et al. (1996) suggest that these processes vary spatially, leading to different local population trends. Pascual and Adkison acknowledge that anthropogenic causes (competition with fisheries, shooting, incidental takes) or long-term climatic fluctuations could explain the decline through a combination of bottom-up and top-down mechanisms.
One of the most useful applications of population models has been to calculate the unexplained mortality that must have occurred to explain observed census results, assuming that reproductive rate has not decreased over the years. Unexplained mortality refers to the additional number of deaths required to make up the difference between the known mortality and the observed decrease in the population. Figure 3.1 shows one such “reconstruction” of past mortality rates, obtained with a spreadsheet age structure model using age-specific birth and survival rates supplied to the committee by Anne York (National Marine Fisheries Service, Alaska Fisheries Science Center, National Marine Mammal Laboratory, Seattle, personal communication, 2001). The spreadsheet simulated forward from 1950 to the present assuming constant (density-independent) age-specific rate schedules that would result in a steady population in 1950, with additional age-independent mortality rates for the periods shown in the figure. Excel’s nonlinear search procedure (Solver) was used to adjust the by-period additional mortality rates so as to match the population trend data as closely as possible.
In agreement with National Marine Fisheries Service calculations, the committee found that unexplained mortalities must have peaked at nearly 30% of the population, or around 15,000 to 20,000 animals per year, during the period of most rapid decline beginning in the mid-1980s. Current (late 1990s) unexplained mortality is estimated to be about 1,000 animals per year, a mortality rate 5% higher than expected for a steady population
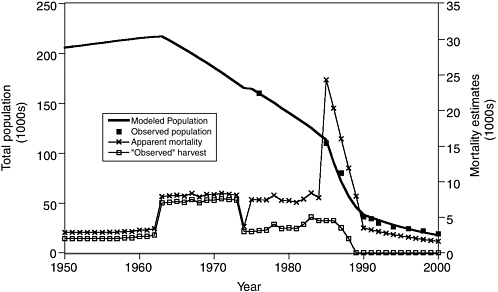
FIGURE 3.1 Fit of an age-structured population model to the western stock abundance data for Steller sea lions, using time-varying estimates of harvest compiled by Trites and Larkin (1992) and fitted by varying time-dependent “unexplained” mortality rates. Age-specific birth and survival rates were assumed to be constant over the simulation period. There was a brief period of very high, unexplained losses during the late 1980s but the unexplained loss from the mid-1970s to the mid-1980s is very close to the estimated commercial harvest from the 1960s to 1970s period of legal harvesting.
SOURCE: Figure based on data from Trites and Larkin (1992), supplied in spreadsheet format by T.R. Loughlin and A.E. York, National Marine Fisheries Service, Alaska Fisheries Science Center, Seattle, 2002.
size and 1,500 animals per year by comparison with the increasing eastern stock (see Box 3.1). These calculations of annual unexplained mortalities are not sensitive to alternative assumptions about the age distribution of the mortalities (e.g., concentrated on pups, yearlings, juveniles, or widely distributed over all ages), except that the losses during the 1980s were too large to have involved only pups and/or yearlings. The estimates are also not sensitive to assumptions about possible compensatory improvements in fecundity or early juvenile survival that might have occurred during the decline. If such improvements did occur, the unexplained mortalities would have to be higher.
During the steepest decline in the Steller sea lion population from 1985 to 1990, estimates of groundfishing-related takes were 1,350 to 1,600
|
BOX 3.1 The Missing 1,500 There are now approximately 28,000 to 33,000 nonpup Steller sea lions in the western population compared to about 150,000 in 1977. Most estimates of mortality rates assume a stable (neither increasing nor decreasing) population, but if there were no unnatural mortality, the expectation is that the depressed western stock would increase at a given rate, nominally at least the rate seen in southeastern Alaska. Demographic analyses (Loughlin and York, 2000) generated a projection of Steller sea lion mortality for 2001 that assumed the western stock would continue to decline at the rate observed from 1991 to 2000. If the population were stable (birth rate equal to death rate), there would be 4,710 mortalities. At the current rate of decline (5.2%), another 1,715 mortalities are projected, bringing the total mortality to 6,425 animals. Over this same time period, the Steller sea lion population in southeastern Alaska has been increasing at an average annual rate of 1.7%. Assuming this rate of increase is normal for the population when conditions are favorable, the western population would be projected to increase by 563 individuals. Hence, compared to the south-eastern Alaska population, the western stock would be short by 2,280 individuals (projected population of recovering stock, 33,680; projected population of declining stock, 31,400). Loughlin and York made a rough estimate of 779 mortalities from human takes and increases in predation by killer whales and sharks due to human activities. This leaves about 1,500 missing animals (unexplained mortalities projected for 2001) relative to a recovering population. |
per year (Perez and Loughlin, 1991; Alverson, 1992). Current takes are estimated to be low, about 30 per year (Ferrero et al., 2000). Subsistence takes have been estimated at 350 animals per year in the 1990s (Wolfe and Mishler, 1997; Wolfe and Hutchinson-Scarbrough, 1999). Shooting of Steller sea lions by fishermen was legal prior to 1972 and until 1990 was allowed in order to prevent gear damage. Trites and Larkin (1996) estimate there were 1,200 annual takes in 1985; it may now be as low as 50 to 100 per year. Predation by killer whales (Orcinus orca) is unknown but has been estimated at 1,100 to 1,200 per year based on a rough estimate of killer whale abundance (Barrett-Lennard et al., 1995; Loughlin and York, 2000). Shark predation could account for an additional 130 deaths (Loughlin and York, 2000). The total of these various estimated sources of mortality at their maximum values for the period 1985-1989 comes to fewer than 4,500 annual mortalities. This mortality is three- to fourfold lower than estimated from the model as unexplained mortality. The cause (or causes) of this large excess mortality has largely been attributed to nutritional stress, but it should be noted that estimates of killings by
humans and natural predators are based on limited data that may greatly underestimate the actual numbers.
ECOSYSTEM MODELS
There has been much interest in the possibility that ecosystem models might help to refute or support some of the more complex hypotheses for the Steller sea lion decline, by integrating information on past trophic interactions, fishery effects, and physical forcing factors. Models developed for the eastern Bering Sea system have ranged from relatively simple and static “snapshot” models of trophic mass balance using Ecopath (e.g., Trites et al., 1999; www.ecopath.org), to complex spatial models based on the Laevastu et al. (1976, 1982) Dynumes III approach (North Pacific Fishery Management Council, 1999). However, these models have generally not been compared to, fitted to, or “tested” against historical data with the same care as would be expected in single-species stock assessment modeling.
To determine whether trophic models can even come close to explaining historical changes, the committee developed an Ecopath/Ecosim model (Walters et al., 1997, 2000) for the historical period 1950-2000. They started with an Ecopath mass balance model for the 1950 abundances and trophic interactions proposed by Trites et al. (1999) for the eastern Bering Sea. Steps in the analysis are shown in the section below. The Ecopath/ Ecosim software system makes it easier to enter time series of historical “forcing” data (fishing mortality rates over time, physical regime indicators over time) and ecosystem “output” data (trends in relative abundances, catches, total mortality rates) than to run multiple simulations that compare model predictions to observed output patterns over time. A similar approach has been taken by the National Marine Fisheries Service (Kerim Aydin, National Marine Fisheries Service, personal communication, 2001).
Applying Ecopath/Ecosim
Four steps are involved in developing an Ecopath/Ecosim model and “testing” it against historical data: (1) establishing an initial system state, (2) defining a set of rules for dynamic change over time, (3) entering historical reference data, and (4) comparing/fitting predictions from the rules and historical “forcing” input patterns to available data.
The first step is to develop a static trophic flow “snapshot” of the ecosystem at one particular point in time, using Ecopath. This involves partitioning system biomass into a set of functional groups/species and providing basic information on these groups. Biomass estimates must be
provided for at least some groups. For all groups, estimates are generally provided of production/biomass (actually total mortality rate), food consumption/biomass (from bioenergetics studies), and diet composition. For groups lacking biomass estimates, estimates are provided of the proportion of total mortality attributable to modeled trophic/fishery flows (consumption by other species and fishery catch). This proportion along with calculated total consumption of the group allows back-calculation of biomass. Estimates can also be provided of the rate of biomass change at the reference time point or year. Ecopath then estimates total biomasses and total fluxes along each predator-prey linkage for a single reference year, so as to account for (balance) all components of biomass change over that year. These estimates provide an initial state for temporal simulations, and the flux components (total biomasses consumed of each prey by each predator in the food web) provide part of the information needed to predict changes in trophic flows over time. The committee used a 24-pool Ecopath model developed by Trites et al. (1999), with one extra pool added to represent the division of small pelagic fishes between nearshore/shallow habitat species like herring (Clupea pallasi) and sand lance (Ammodytes hexapterus) that are most available to Steller sea lions versus offshore/deepwater species (such as myctophids), which may be available mainly to the benthic piscivores like arrowtooth flounder (Atheresthes stomias) that have prospered in the eastern Bering Sea in the past two decades.
Second, Ecosim uses the initial or base state rate estimates from Ecopath and adds additional parameters to predict changes in the trophic interaction (predation) rates for other system states (biomasses) besides the original Ecopath state. That is, Ecosim postulates functional relationships based on the recent behavioral ecology theory of foraging arenas (see Walters and Kitchell, 2001) to predict effects of changes in prey and predator abundances on the predation rate components. The functional models for rate change can include effects of predator satiation, changes in foraging times by prey and predator, and changes in physical factors that affect production rates (in case of primary producers) or prey availability/predator effective search rates. Additionally, Ecosim allows replacement of simple biomass dynamics rate equations with more complex delay difference accounting for population age-size structure for species that have strong habitat/trophic ontogeny.
Third, a simple Excel file is used to define a historical reference period (1950-2000) and to specify annual time forcing historical “inputs” and measured model-testing abundance and mortality “output” patterns over the reference period. The committee used results from single-species assessments to provide fishing mortality rate histories for herring, walleye pollock, cod, flatfish (rock sole, arrowtooth flounders), whales, and
Steller sea lions (culling rates). As outputs, relative abundance series were provided, ranging from the Steller sea lion western stock total abundance estimates to total pollock, cod, and flatfish abundance estimates from single-species stock assessments. Time series estimates of total jellyfish, plankton, and forage fish abundance also were included from Brodeur et al. (1999).
Fourth, Ecosim simulations were used to determine how well the model performs at “replaying” past system changes in two testing improvement modes: exploratory game playing and formal fitting procedures. The exploratory/game playing mode of model fitting tends to lead in one or a few promising directions of explanation but certainly does not rule out others. In this mode, simulated and observed abundance trajectories are compared and obvious model data discrepancies are corrected by changing model parameters that may have caused the discrepancies. For example, in this mode it was quickly found that Ecosim would not predict as much variation as seen in the historical eastern Bering Sea data unless parameters representing vulnerability to predation were increased considerably (for all trophic linkages) from their Ecosim default settings. It was also found that inverse patterns of abundance change for benthic piscivores (increase) versus Steller sea lions (decrease) could not be produced when strong feeding preference for small pelagics was assumed, unless it was admitted that different components of the pelagic biomass might be available to these predator types.
The second mode is a more formal “fitting” of the model by nonlinear search procedures, allowing predation vulnerabilities, initial abundances, and annual primary production “anomalies” to vary. The fitting criterion used was a likelihood function for the observed relative abundance time series, assuming independent and log-normal observation errors for these relative abundances. The formal fitting procedures can demonstrate whether or not the data are consistent to at least some degree with a hypothesis of long-term variation in primary productivity, but this consistency is in no way proof that such variations actually took place. This mode was used only to estimate patterns of apparent change in primary production; patterns that the estimation procedure will “see” in this case are those that simultaneously improve fit to multiple relative abundance time series (i.e., that represent apparent ecosystem-scale productivity anomalies).
As a word of warning, it must be emphasized that step four cannot in principle be used to demonstrate that there is some unique “best” model to describe the historical data. We are dealing with fragmentary, incomplete data, and a natural dynamics that historically involved a multitude of dynamic parameters, most of which have not been measured directly. In such situations there is certain to be a wide variety of model structures
and rate parameter combinations that will fit the data equally well (i.e., that are equally good hypotheses to explain the data).
Ecosim Modeling Applied to the Eastern Bering Sea
The available data are insufficient to develop models that will provide unequivocal conclusions about cause and effect with regard to the Steller sea lion decline. Instead, modeling is used here to identify more plausible scenarios for the decline of the Steller sea lion population.
It is quite easy to obtain reasonably good fits to at least qualitative trends in the abundance of major species groups, especially when Ecosim is allowed to estimate apparent temporal anomalies in annual primary production rates (see Figures 3.2 and 3.3). The apparent anomalies that give best fits (and explain about 30% of the variation in the data series shown in Figure 3.2) appear to involve the sort of decadal regime shifts that have been proposed for the region based on climatic data and anomaly patterns in recruitment rates for single-species stock assessment models. Interestingly, little of the variation is explained by historical changes in fishing mortality rates alone. In terms of trophic interactions, much of the good fit is due to effects following from reduction in sperm and baleen whale abundances, represented as a temporal cascade of events.
The model predicts (hindcasts) that reduction in sperm whales, in particular, likely led to substantial increases in their main prey, squids, and this resulted in considerable increase in squid predation on small pelagics. Reduction in small pelagics was then partly responsible for an initial simulated decline in Steller sea lion population and to the beginnings of an increase in jellyfish (more zooplankton available following reduction in pelagic fish abundance). Pelagic fisheries (herring fishery) then hastened the reduction in availability of prey to Steller sea lions, and permitted increase in other pelagics that in turn fueled an increase in benthic piscivores like arrowtooth flounder. Obviously, each of the steps cannot be tested in this complex causal “hypothesis” with the fragmentary data available on the history of the eastern Bering Sea.
However, the committee was unable to find Ecosim parameter combinations that would predict a short period of rapid decline during the late 1970s or early 1980s for Steller sea lions, related only to changes in trophic interactions caused by measured fishery changes and fitted primary production changes. This is not because the Ecosim functional structure precludes such predictions; see Figure 3.4 for an example where it did predict rapid populaton decline for Hawaiian monk seals (Monachus schauinslandi) under combined effects of fishing on lobsters and a climatic regime shift. Note the decline predicted in Figure 3.2 is much more
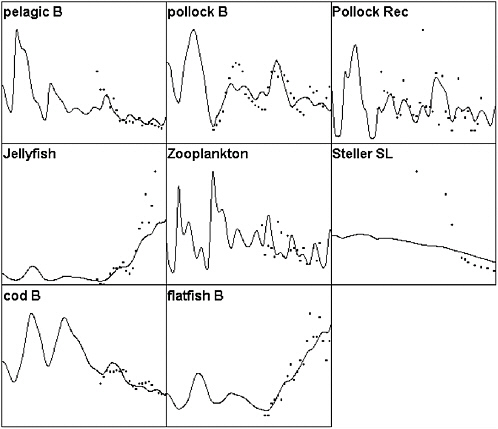
FIGURE 3.2 Ecopath/Ecosim simulation of the eastern Bering Sea ecosystem for the period 1950-2000 compared to selected time series data. Solid lines represent simulated biomasses over time (abscissa is time, 1950-2000; ordinate is relative biomass) and dots represent survey or stock assessment estimates rescaled to the same mean as the simulated biomass series. Simulated biomasses were initialized with the 25 pool Ecopath model based on the mass balance estimates of Trites et al. (1999). Simulated results include effects of time-varying fishing mortality rates, along with fitted estimates of relative primary production rates (fitted relative production rate series shown in Figure 3.3). The base model does not predict as severe or rapid a decline in Steller sea lions as was observed; no parameter combination was found that predicted a rapid decline during the mid-1980s involving only trophic interactions and known impacts of fisheries.
SOURCES: Figure created based on Ecosim simulation of Ecopath model by Trites et al. (1999); times series data for cod, pollock, flatfish came from Blackburn et al. (2001); data on small pelagics, zooplankton, and jellyfish came from Anderson and Piatt (1999).
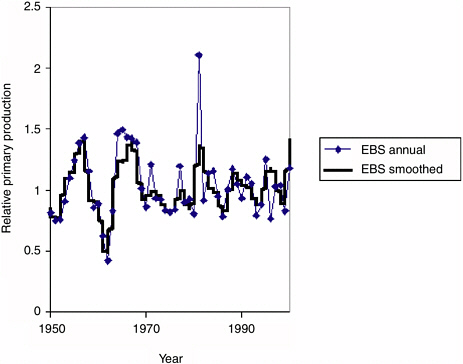
FIGURE 3.3 Fitted time series of relative primary production rates for the eastern Bering Sea (EBS) used to obtain the fits to Ecosim shown in Figure 3.2. A nonlinear search procedure in Ecosim was used to vary annual (or spline function with 2-year nodes) primary production “anomalies” so as to improve the fit of the model to the time series shown in Figure 3.2. These anomaly sequences explain about 30% of the variability in the time series data not explained by historical fishing and trophic interaction effects.
gradual and prolonged than indicted by the census data and that there are no measured rapid changes in prey abundance just prior to or during the rapid Steller sea lion decline. Of course, a more rapid ecosystem-scale regime shift could be forced on the model dynamics in order to produce a more rapid decline in Steller sea lions, but such scenarios result in poor fits to trend data for the fish species.
Good fits to the Steller sea lion data, including the rapid decline, could be obtained by forcing the Ecosim model with various “hidden variable” hypotheses for historical change not directly supported (or refuted) by available field data (see Figure 3.5). One parsimonious hypothesis that gives a good fit (see Figure 3.6) is to simply assume there was much more unreported culling of Steller sea lions by trawl and crab fishers as these fisheries developed and peaked in the early 1980s. Another is
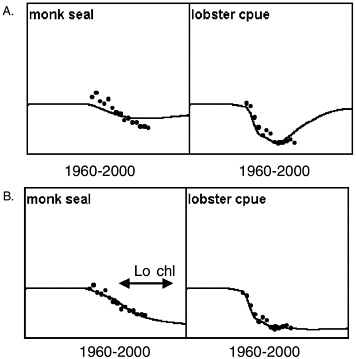
FIGURE 3.4 Prediction of a strong decline in a marine mammal population using Ecosim. Results are for a model of reef ecosystems in the northwestern Hawaiian Islands (French Frigate Shoals), testing whether rock lobster fishery development alone could explain monk seal decline. Data points are for the years 1983-1998. Initial model runs (A) showed that fishing and trophic interactions alone did not explain the monk seal decline and predicted recovery of the lobster population. Polovina’s insight (B) was that satellite chlorophyll data indicate a persistent 40-50% decline in primary production beginning around 1990. This example illustrates that Ecosim functional structure is capable of modeling a rapid population decline of marine mammals.
SOURCE: Jeffery Polovina, National Marine Fisheries Service, Honolulu, personal communication, January 1999.
to assume that there were widespread, relatively sudden declines in small pelagics, particularly herring, due to disease outbreaks like those that apparently caused mass mortality in Prince William Sound (Meyers et al., 1994; Stokesbury et al., 2002). Still another is to assume that physical regime changes in 1976-1977 led to reduced vulnerability of various fish prey to Steller sea lions, perhaps by affecting prey depth distributions and/or prey use of coastal areas near rookeries.
There is always the possibility that future, more detailed, modeling
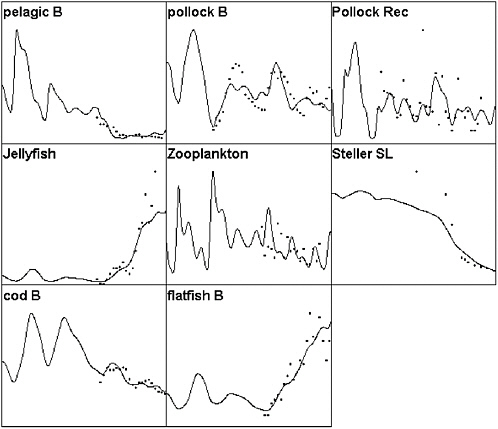
FIGURE 3.5 Comparison of Ecosim-predicted biomass patterns to observed data for the same model as in Figure 3.2 but including a few years of “unexplained” high mortality rate for small pelagic fishes (herring, sand lance) that are available to the Steller sea lion. Note that this scenario gives a somewhat better fit to both the sea lion and pelagic abundance data.
SOURCES: Figure created based on Ecosim simulation of Ecopath model by Trites et al. (1999); times series data for cod, pollock, and flatfish came from Blackburn et al. (2001); data on small pelagics, zooplankton, and jellyfish came from Anderson and Piatt (1999).
exercises, comparison of models for the eastern Bering Sea and Gulf of Alaska regions, and further comparisons to historical data may reveal a “complete” explanation for the Steller sea lion decline in terms of trophic changes. But the committee seriously doubts that this is going to happen, especially in view of gaps in the historical forcing data that would have to be supplied to such models. Hence, models will probably not be sufficient to definitively identify the cause of the rapid decline in the sea lion population.
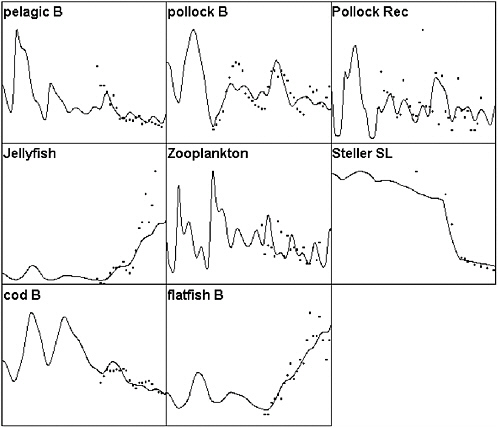
FIGURE 3.6 A further comparison of Ecosim to eastern Bering Sea time series data, but with the addition of direct mortality of Steller sea lions during the mid-1980s due to a hypothesized direct fishery impact (unmonitored culling by crab and pollock fishers). Note that this hypothesis fails to explain the early decline in the pelagic fish abundance index and also fails to explain relatively high rates of decline observed since 1990 in the sea lion population.
SOURCES: Figure created based on Ecosim simulation of Ecopath model by Trites et al., (1999); times series data for cod, pollock, and flatfish came from Blackburn et al. (2001); data on small pelagics, zooplankton, and jellyfish came from Anderson and Piatt (1999).
EVALUATING HYPOTHESES FOR THE CAUSE OF THE STELLER SEA LION DECLINE
A variety of complex hypotheses involving changes in trophic interactions in the eastern Bering Sea and Gulf of Alaska have been suggested as possible explanations for the Steller sea lion decline. These hypotheses mainly involve factors that could have resulted in two types of immediate effects on the sea lions: (1) reductions in the availability of pelagic, high-
quality (fatty), prey like herring and sand lance and/or (2) reductions in alternative prey for predators such as killer whales, which might have led these predators to switch to Steller sea lions. Note that these immediate effects do not involve geographic patterning or directional changes in ecosystem productivity. On a global scale (eastern Bering Sea plus western Gulf of Alaska), the data from single-species stock assessments do not support the proposition that fisheries and/or climatic factors have led to a reduction in the total abundance of food available to Steller sea lions. Some widely distributed and very abundant fish species like pollock and cod have increased considerably over the period of the Steller sea lion decline, as have some bird species (Dragoo et al., 2000). One climatic index, mean wind speed over the eastern Bering Sea, has declined considerably. This could result in a decrease in annual primary production, due to decreased vertical mixing and supply of nutrients to the photic zone. However, the mean wind decrease is not well correlated with most indicators of productivity at higher trophic levels.
Pelagic prey availability could have changed for at least two reasons: changes in prey distributions related to physical changes (e.g., changes in depth distributions associated with warming) and changes in total prey abundance. There is little evidence of major changes in prey vertical or horizontal distributions, but there is considerable evidence of changes in pelagic prey abundance, especially herring. Hypotheses that could explain pelagic species abundance change include:
-
Herring reduction fisheries (ended in late 1960s) and later roe fish eries. Herring stock assessments do not indicate complete recovery in stock sizes following closure of the reduction fisheries as was observed farther south in British Columbia. BC assessments generally show dramatic increases in herring stocks during the early 1970s (Hourston and Haegele, 1980), while Wespestad’s (1991) assessment and more recent trends reported by Brodeur et al. (1999) show continuing low abundance in the eastern Bering Sea. However, under this hypothesis, the Steller sea lion decline should have occurred much sooner than currently estimated.
-
Food web impacts of whale fisheries. Whaling in the 1950s and 1960s led to considerable reduction in sperm whale populations (National Research Council, 1996). This may have permitted squid, their main prey, to increase. Squid feed on small pelagics and may now “capture” much more of the net productivity of the pelagics than was possible when whales were abundant. This hypothesis is “supported” by Ecopath/ Ecosim modeling results but again should have led to an earlier sea lion decline than was observed.
-
Competition with jellyfish. Brodeur et al. (1999) suggest that major increases in jellyfish abundance since at least 1980 may have resulted in
-
both higher direct predation on eggs and larvae and greater food competition with pelagic, plankton-feeding fishes. However, the rapid decline in Steller sea lions that began in the late 1970s cannot be explained by an increase in jellyfish during the 1980s.
-
Shift in fish community dominance from pelagic to “benthic” species. Herring and other small pelagics may compete with juveniles of pollock and other large fish species for food and may also prey on juveniles of these species. Thus, they are not simply prey for the larger fish. Such reciprocal mortality interactions, where the prey can cause increased juvenile mortality of its predators as well as predators causing higher prey mortality, can lead to so-called cultivation-depensation effects (Walters and Kitchell, 2001) and to multiple equilibria in community structure. In this case, one equilibrium might involve dominance by small pelagics, and another might involve dominance by larger fishes. The shift toward benthic dominance may have been triggered by herring fisheries, climatic regime shifts, or both (Anderson and Piatt, 1999). The benthic piscivore community has increased considerably in biomass despite the coincident development of the fishery (North Pacific Fishery Management Council, 2001a, 2001b). A fishery closure might result in even higher fish biomass absent effects of fishing on piscivore age-size structure, unless cannibalism were to increase considerably. The shifts in community dominance do not coincide precisely with the timing of the Steller sea lion decline; the shifts apparently began during the 1970s and were largely complete by the time of rapid decline in the late 1980s.
-
Changes in the seasonal timing of water temperature and plankton production. There are apparently at least two types of “growing seasons” in the Bering Sea. One involves late ice cover that results in a spring bloom when the water temperature is low enough to inhibit zooplankton productivity. The other involves less ice cover, resulting in a delayed spring bloom with warmer temperatures that favor the growth of zooplankton. The second of these regimes appears to favor recruitment of planktivorous fish and their predators, such as pollock. During a warm regime, strong year classes of pollock could suppress the populations of forage fish (also see hypothesis 4). However, in the Gulf of Alaska, where sea ice is not a factor in regulating the timing of the spring bloom, this mechanism would not apply.
EVALUATING MECHANISMS IN RELATION TO SEA LION POPULATION DYNAMICS
Considering the natural complexity of the eastern Bering Sea food web, further complicated by the effects of fisheries and oceanographic conditions on the recruitment of individual species, a variety of other
hypotheses could be proposed that invoke changes in food web structure. And doubtless, at least some support for these alternative hypotheses could be found in the biological and physical data that have been collected. But all of these stories would share two key features: they require many contingencies to fit the observations, and they do not predict the particular timing of the Steller sea lion decline.
There is good evidence (from fine-mesh trawl surveys) of a rapid shift from pelagic to benthic community dominance in the western Gulf of Alaska at just about the right time to explain rapid sea lion declines in the region from Prince William Sound to the eastern Aleutians (Anderson and Piatt, 1999). Anderson and Piatt argue that this shift may have involved a change in seasonal timing of plankton production (hypothesis 5 above), from a late-bloom pattern that may favor small pelagics, shrimp, and crabs to an earlier bloom pattern that may favor pollock and some flatfishes. They note that increases in benthic predator recruitment would then exaggerate the decline in the smaller, late-spawning species, through top-down predation effects.
It is quite possible that the Gulf of Alaska “community reorganization” involved strong trophic interaction (cultivation/depensation) effects along with the physical regime effects described by Anderson and Piatt. If so, fishery reductions and closures aimed at the groundfish stocks may help to reinforce benthic community dominance and prevent recovery of a community structure that favors Steller sea lions.
Shifts in food web organization due to both fishing and environmental change might have altered feeding patterns of killer whales and other predators, resulting in higher predation rates than might be expected based on a straightforward change in relative prey abundance. Such “predator switching” effects constitute a distinct hypothesis for the Steller sea lion decline, discussed in more detail in Chapter 6. Here we simply note that any of the mechanisms above that may have led to a change in the “prey field” available to the Steller sea lion also may have affected killer whales and other potential predators. These predators may then have compensated by switching prey and exaggerating the effects of the other mechanisms. Ecosim/Ecopath models fit this hypothesis if there is at least a doubling of killer whale predation on Steller sea lions coincident with the Steller sea lion decline and another mechanism (like disease or a cultivation/depensation shift) caused a change in pelagic prey availability to both Steller sea lions and killer whales during the early 1980s.
SUMMARY
Both the population and ecosystem models demonstrate the necessity of quantitative historical data. Lack of this information constrains the
interpretation of modeling exercises, and without input of archival or unanalyzed data, these models will not be able to definitively test any of the current hypotheses about the cause or causes of the historical decline of Steller sea lions. However, these models clearly identify what types of data should be collected to monitor current population trends and sources of mortality.
The models encourage serious examination of many of the previously underemphasized top-down hypotheses. The population models suggest that increased adult mortality more readily accounts for the rapid decline in the 1980s than either reduced fertility or increased juvenile mortality due to food limitation. Therefore, the question shifts away from “Is it food?” to “Were they food?” (e.g., sources of sea lion mortality from predation by humans or other top carnivores). However, these conclusions are based on old demographic data of limited sample size, and until more current data become available, this interpretation rests on a shaky foundation. Nonetheless, population models examined in a global spatial context suggest that multiple factors have contributed to the decline. It is not sufficient to consider regime shifts or extreme fluctuations in availability of important prey populations in the absence of other indirect effects arising from the multiple linkages in food webs. The ecosystem models develop a varied menu of hypotheses to explain the historical decline. Thus, based on available trajectories for the population trends of many species, Ecosim is unable to reproduce the rapid decline in sea lion abundance. The model could not reproduce the steep population decline involving only trophic interactions and known impacts of fisheries. Adding other, often undocumented, mortality factors substantially improves the fit. The value of inventing and using undocumented levels of historical forcing, again acknowledging the constraints imposed by insufficient information, is that they identify a tantalizing variety of possible causes of the decline. These approaches, despite their implicit and identified limitations, provide a framework for evaluating the various hypothesized causes of the continuing decline in the sea lion population and identifying the most fruitful avenues for future research.


















