Page 413
15—
Electrolytes
Sodium, potassium, and their attendant anions are important components of all body fluids. Sodium is the major cation of extracellular fluid, and potassium, of intracellular fluid. Complex mechanisms regulate electrolyte concentrations in the body fluids and the volume of both the extracellular and the intracellular fluid compartments. The processes that maintain gradient concentrations of these cations across cell membranes require energy; at least three transport systems seem to be involved. Regulation of fluid volumes and concentrations involves the cardiovascular and endocrine systems, the central nervous system, and the autonomic nervous system; all act chiefly by regulating the rate at which water and electrolytes are excreted by the kidneys.
Archaeological and anthropological studies show that the diets of hunter-gatherers during the Paleolithic period and the diets of present-day traditional societies outside the dominant culture have, with few exceptions, had high levels of potassium and very low levels of sodium (Denton, 1982). Salt has historically been scarce in most regions, and it was highly prized by early humans and by many ancient cultures in Asia, Africa, and Europe. It was used in rituals and for the preservation of food in many primitive cultures. In areas with a scarcity of salt, a mechanism to preserve extracellular fluid volume in the face of dehydration, trauma, hemorrhage, pregnancy, and lactation would be biologically useful. Thus, the physiology of mammals evolved to foster the conservation of salt by the kidneys, gastrointestinal tract, and sweat glands and to develop a taste for sodium chloride in the tongue and the salt appetite centers in the brain (Denton, 1982).
There is evidence for the existence of salt appetite centers in the central nervous system of some mammals, and a mechanism to taste salt is found in humans and many mammals. Craving for salt develops during acute salt depletion and hypovolemia. Some recent evidence suggests that two ranges of salt appetite exist: the physiological range of salt intake necessary to preserve body fluid volume and maintain sufficient arterial pressure to perfuse tissues adequately; and the higher range of salt appetite, which is determined by a learned desire to ingest salt in excess of physiological need (Beauchamp et al., 1985). Deprivation of these higher levels of salt for several months results in a preference for less salt.
Neolithic agricultural societies developed various methods for preserving and storing foods. For meat and dairy products, they usually used salt. Most modem processing methods, including the processing of grains and refined flours, increase sodium content and reduce potassium, whether needed for preservation or not. More sodium is added during the preparation of foods for the table and at the table itself.
Page 414
Evidence Associating Electrolytes with Chronic Diseases
Physiological regulating mechanisms normally maintain close control over the concentrations of sodium, potassium, and chloride as well as the total body content of several electrolytes. Failure of regulation profoundly affects body fluid volumes, blood pressure, cardiovascular function, and acid-base balance. Most perturbations in regulation and balance result from disease processes rather than from variations in dietary intake.
Sodium
Physiologic Requirements
In foods, sodium is present mainly in salt (sodium chloride). Sodium bicarbonate, sodium citrate, sodium glutamate, and other sodium salts are also consumed in small amounts. The quantity of dietary sodium is often expressed as milligrams of sodium or sodium chloride. In chemical terms, it may be expressed as mEq of ionized sodium or as mmol of sodium chloride. In this chapter, it is also expressed as milligrams of salt. Since potassium exists in the body in association with various chemicals and the chloride salt of potassium is not predominant, potassium is expressed in grams of elemental potassium and in chemical terms, as mEq.
The basal sodium requirement for growing and adult humans is no greater than 8 to 10 mmol/day (500 mg of sodium chloride) (AAP, 1974; Dahl, 1958) and may be lower. Dahl (1958) reported a mean sodium chloride intake of 10,340 mg (range, 4,000 to 24,200 mg) for 71 working men in New York based on a single 24-hour measurement of urinary excretion. Sanchez-Castillo et al. (1987a,b) performed metabolic balance studies using a lithium chloride marker to trace use of salt in cooking and at the table. Daily sodium chloride excretion over a 12-day period by men was 10,600 ± 0.550 mg and by women was 7,400 ± 2,900 mg. Ten percent of the salt came from the natural salt content of foods, 75% from processing and manufacturing, and 15% from discretionary addition of salt (both in cooking and at the table). Sweat and fecal excretion of sodium in these studies were found to contribute only 2 to 5% of total excretion; the remainder was excreted in the urine (Sanchez-Castillo et al., 1987a). Salt excretion was higher by men than by women and was directly proportional to body weight.
James et al. (1987) reported that the total salt excretion is similar in northern Europe and the United States; the lowest known intake is that of the Yanomami Indians of Brazil, who excrete an average of 1.5 mEq/day (87 mg of sodium chloride) in their urine (Oliver et al., 1975) and presumably lose some additional salt through sweating and lactation. Because the physiologic mechanisms for conserving salt are very efficient, chronic deficiencies do not ordinarily occur, even in populations such as the Yanomami and others on very low-sodium diets (Page, 1976, 1979). Nevertheless, it is likely that people subsisting on very-low-salt diets are slightly more vulnerable to acute losses resulting from diarrhea, trauma, or blood loss (Gothberg et al., 1983).
Acute depletion of salt resulting in heat exhaustion and cardiovascular collapse may follow heavy sweating in people who have a high-salt intake and in whom adrenal mechanisms for conserving sodium have not been sufficiently activated (Conn, 1949). Healthy Caucasian men, after adaptation to heat and a moderately low-salt intake for 1 week, maintain sodium balance on an intake of 90 mEq/day (5,200 mg of sodium chloride), even while sweating 9 liters/day (Conn, 1949). Lower salt intakes have not been tested under similar laboratory conditions. Normal men with salt intakes varying from 10 to 1,500 mEq/day (580 to 87,000 mg of sodium chloride) maintained sodium balance without acute ill effects for 3 days (Luft et al., 1979, 1982). Despite these apparently wide tolerances, variations in salt intake among populations are associated with substantial variations in blood pressure and, therefore, occurrence of hypertension.
Hypertension
Primary hypertension, or high blood pressure, is a major risk factor for cardiovascular disease and death in the United States (Pooling Project Research Group, 1978). Definitions of high blood pressure are arbitrary and vary substantially. Average levels of both systolic and diastolic blood pressure in populations vary with age and sex (see Chapter 20). The prevalence of hypertension increases with age and is present in approximately 15% of the U.S. population age 30 and over (WHO, 1978). Criteria for adults, recommended by an expert committee of the World Health Organization (WHO, 1978), are as follows:
· Normotensives: systolic £ 140 mm Hg
diastolic £ 90 mm Hg
Page 415
· Borderline: systolic 141-159 mm Hg
diastolic 91-94 mm Hg
· Hypertensives: systolic ³ 160 mm Hg
diastolic ³ 95 mm Hg
Additional definitions of hypertension are discussed in Chapter 5.
No single factor causes hypertension, which involves the renal, cardiovascular, endocrine, and central and autonomic nervous systems. It is generally agreed that different factors play greater or lesser roles in different people and that there are variations in the rapidity of onset and degree of severity. Hypertension is usually asymptomatic but increases the risk for stroke, heart attack, and other cardiovascular events.
There is a genetic predisposition to hypertension, but there are no known reliable specific genetic markers. The inherited susceptibility appears to be expressed as a result of interaction with other factors: obesity, alcohol, and a variety of nutrients, including sodium and potassium (Denton, 1982; Folkow, 1982). Intake of polyunsaturated and saturated fats and calcium and exposure to psychological stress may also have roles in the expression of hypertension (Kaplan et al., 1985; see also Chapter 13).
Although studies of the relationship of sodium to hypertension have been conducted since the beginning of the twentieth century (Folkow, 1982; Porter, 1983), there is still some controversy about the importance of salt in regulation of blood pressure and about the mechanisms by which salt influences blood pressure. By far the greatest difference of opinion, and the most strongly held opinions, relate to the desirability of recommending to the general public that dietary sodium intake should be restricted. Numerous research studies, reviews, symposia, and books on these topics provide an abundance of data and a wide range of interpretations. There is little likelihood that these controversies will be entirely resolved in the foreseeable future.
Methods for estimating salt intake have varied, and timed urine collections and dietary intake recalls frequently do not match. Liu et al. (1979) found wide day-to-day variations in repeated measurements of sodium excretion by urban workers. Such intraindividual variability will tend to obscure real associations. Multiple urine collections are necessary to obtain consistent results, but even these collections and meticulous attention to these methods may fail to give reproducible results (Cooper et al., 1982). In addition, other variables known to affect blood pressure, such as genetically determined salt sensitivity, obesity, alcohol intake, and emotional stress, can obscure the relationship of salt to blood pressure (Jacobs et al., 1979; Liu et al., 1979).
Studies of blood pressure and sodium intake or excretion in individuals within populations have often failed to show a statistically significant relationship (Dawber et al., 1967; Holden et al., 1983; Karvonen and Punsar, 1977; Ljungman et al., 1981; Schlierf et al., 1980), but most studies based on a single 24-hour urine collection have not had sufficient statistical power to detect a slope of 0.1 mm Hg/mmol sodium (Watt et al., 1983). Significant intrapopulation correlations have been observed by some investigators (Cooper et al., 1980; Kesteloot et al., 1980; Khaw and Barrett-Connor, 1987, 1988; Page et al., 1981; Tao et al., 1984; Voors et al., 1983).
Secondary analyses of results from cross-sectional surveys of various populations have generally shown a positive correlation between intake of sodium and level of systolic and diastolic blood pressures (Froment et al., 1979; Gleibermann, 1973; Simpson, 1985). However, interpretation of these results has been complicated because the data came from separate, individual studies with unstandardized measurements of sodium and blood pressure and because results were usually not adjusted for confounding factors other than age and sex.
The Intersalt Cooperative Research Group (1988) studied both intra- and interpopulation associations between 24-hour urinary excretion of electrolytes and blood pressure in 10,079 men and women, 20 to 59 years of age, who were sampled in groups of about 200 at each of 52 centers in 32 countries around the world. The investigators used a highly standardized protocol with central training of observers, a central laboratory, and extensive quality control procedures. Median systolic blood pressure varied from 95 to 132 mm Hg, median diastolic blood pressure from 61 to 82 mm Hg, and prevalence of hypertension (defined as systolic blood pressure ³ 140 mm Hg, or diastolic blood pressure ³ 90 mm Hg, or use of antihypertensive agents) from 0 to 33%. Median daily excretion of sodium chloride varied from 0.2 mmol (11.6 mg) in the Yanomami Indians of Brazil to 242.1 mmol (14,043 mg) in Tianjin, northern China, but the distribution within this range was not even. Four geographically isolated centers had mean values below 57 mmol/day, no center was between 57 and 103, 4 were between 104 and 138,
Page 416
and 44 were higher. Thus, data are limited on the association between sodium excretion and blood pressure for populations with mean sodium excretion less than 138 mmol/day (8 g/day).
Among individuals within centers, 24-hour sodium excretion was positively and significantly associated with level of systolic blood pressure, and this association persisted after adjustment for age, sex, body mass index, alcohol consumption, and urinary excretion of potassium. A similar association was observed for diastolic blood pressure after adjustment for age and sex, but the regression coefficient was nearly zero and not statistically significant after adjustment also for body mass index, alcohol consumption, and urinary potassium. Sodium to potassium ratio was positively and significantly associated with both systolic and diastolic blood pressure after adjustment for age, sex, body mass index, and alcohol consumption.
In cross-center analyses involving all 52 centers, median sodium excretion was positively and significantly associated with median systolic blood pressure after adjustment for age, sex, body mass index, and alcohol consumption. As customarily happens when the range of a regressor is truncated, the regression coefficient was reduced in magnitude (and was no longer statistically significant) when the four centers with lowest median values for sodium excretion were excluded from the analysis. Associations with diastolic blood pressure were smaller and not statistically significant.
Median sodium excretion was positively and significantly associated with the age-related rise in systolic and diastolic blood pressure in all 52 centers and also in the 48 centers with the highest median values for sodium excretion. These associations persisted after adjustment for sex, body mass index, and alcohol consumption. The adjusted results for all 52 centers indicated that an increment of 100 mmol/day of sodium was associated with an increment of 10 mm Hg in mean systolic blood pressure and 6 mm Hg in mean diastolic blood pressure between the ages of 25 and 55.
Median sodium excretion was positively and significantly associated with prevalence of hypertension in cross-center analyses involving all 52 centers, but the association was not significant when the four centers with lowest median values for sodium excretion were excluded. Similarly, the ratio of sodium to potassium was positively associated with the slope of blood pressure with age and with the prevalence of hypertension, but these associations were statistically significant only in the analyses including all 52 centers.
These results indicate that in unacculturated populations where habitual salt intake is less than 4,500 mg/day, blood pressure does not rise with age, average adult blood pressure is low, and hypertension is rare or absent (Page, 1979). In the four Intersalt populations with median sodium excretion under 60 mmol/day (3,480 mg/day), age had little or no association with blood pressure, while in the populations with median sodium excretion of 100 mmol/day (5,800 mg/day) or higher, age was positively associated with blood pressure, and the rate of rise with age was proportional to the median sodium excretion. An absence of hypertension despite high intake of sodium has been reported in India (Malhotra, 1970) and among Buddhist farmers in Thailand (Henry and Cassel, 1969), but further examination of both cases indicated that the data were inadequate to support any conclusions about sodium intake and hypertension (Denton, 1982; Prineas and Blackburn, 1985).
Since blood pressure must rise with age for hypertension to become prevalent in a population, these results indicate that the average consumption of sodium is an important factor in determining the prevalence of hypertension in populations. The relatively weaker associations observed in the Intersalt populations between median sodium excretion and median levels of blood pressure may have been due to factors such as climate, physical activity, or other dietary components, which, while varying among centers, were similar within centers and could have biased comparisons of medians across centers without biasing the slope of blood pressure with age or other within-center associations. The committee recognizes that the results of Intersalt and other epidemiologic investigations of this topic are subject to various interpretations, but concludes that the weight of evidence supports the contention that intake of sodium is an important factor in the occurrence of hypertension.
A significant intrapopulation correlation has been found within other populations, at least for systolic blood pressures in Iran (Page et al., 1981), China (Tao et al., 1984), and Korea (Intersalt Cooperative Research Group, 1988; Kesteloot et al., 1980; Khaw and Barrett-Connor, 1987, 1988). Cooper et al. (1980) reported a slope of 0.06 mm Hg/mmol sodium in children, using seven consecutive 24-hour urine collections.
A positive correlation between blood pressure and salt intake among individuals might occur only in people with a genetic susceptibility to hypertension. American blacks account for an unusually
Page 417
high proportion of those genetically susceptible, and Voors et al. (1983) found a positive correlation between salt intake and blood pressure in black teenagers.
Several investigators have studied responses to changes in sodium intake and sodium depletion in an attempt to identify people who are sensitive or resistant to the effects of salt on blood pressure. Salt loading acutely lowers forearm vascular resistance in healthy, normal people, but it increases resistance in those with borderline hypertension (Mark et al., 1975). Luft et al. (1982) fed normotensive men six successively higher levels of salt, ranging from 10 to 1,500 mEq (580 to 87,000 mg of sodium chloride per day), each for a period of 3 days. The subjects maintained sodium balance, but nearly all subjects developed some increase in blood pressure as levels of salt increased. Hofman et al. (1983) compared two groups of newborn infants fed formulas with two different salt levels. One formula contained 6.3 mEq (370 mg) of sodium chloride per liter, approximately equal to that in human milk. The other contained 19.2 mEq (1,100 mg) of sodium chloride per liter, approximately equal to the level in cow's milk. At the end of the study, systolic pressure was 2.1 mmHg higher in those on the higher sodium formula, but no residual difference in blood pressure was found after 1 year. Weight and growth rate were the same in the two groups. Studies such as these indicate that short-term changes in sodium intake influence blood pressure. Long-term prospective controlled trials of variations in sodium intake have not been conducted.
Arterial blood pressure at any time is the result of a complex interplay of dynamic variables. Any satisfactory theory must consider short-term and long-term effects as well as the influence of heredity, body fluid volumes, cardiac and vascular factors, regulatory mechanisms mediated through the central and autonomic nervous systems, the endocrine system, and the kidneys. It must also take into account the other factors known to influence blood pressure, including race, body mass, psychosocial stresses, and nutrients other than sodium. Only selected facets of this large subject are presented herein, specifically those that seem most relevant to the long-term effects of habitual sodium intake on blood pressure in human populations.
Tobian and Binion (1952, 1954) reported increased intracellular sodium in the arteries of hypertensive patients. Recently, increased permeability of cell membranes to sodium has been found in several animal models of hypertension (Tobian et al., 1986) and in many patients with primary hypertension (Page, 1976). Abnormalities in membrane lipids and in calcium binding have also been found both in hypertensive humans and in animals (Tobian et al., 1985a). The possible role of calcium in hypertension is discussed in Chapter 13.
For 50 million years, almost all mammals have lived in a world where foodstuffs tend to have quite a low sodium content. The saltiest foods available to most mammals are other animal tissues; however, even completely carnivorous animals still have a salt intake substantially lower than that of most modern humans.
Studies suggest that a high-salt diet increases blood pressure in some animals, but not in others. When a high-salt diet raises blood pressure in animals, there is usually some defect in the ability of the kidneys to excrete salt rapidly. The combination of a high-salt intake and defective salt excretion tends to increase the salt content of the body, which appears to trigger a rise in blood pressure (Tobian, 1983; Tobian et al., 1977). In turn, the increased pressure entering the renal artery encourages a more rapid natriuresis, which tends to normalize the defect in salt excretion. Evidence suggests that two processes are involved. One process appears to be the interaction of a high-salt intake with a diminished renal capacity to excrete salt rapidly, which tends to cause sodium retention. The other process triggers a rise in blood pressure in response to increased body sodium, but the mechanism for this is not entirely clear. However, there is recent evidence that this sodium chloride signal is perceived in brain tissue around the third brain ventricle (Tobian, 1988).
Salt-induced high blood pressure can be observed in studies comparing Dahl S and Dahl R rats—now designated DS and DR (Dahl et al., 1962). When these two strains consume a low-salt diet, both have blood pressure well within the normal range, although the DS rat has a slightly higher blood pressure and some evidence of defective renal salt excretion. When dietary salt was increased, the blood pressure of the DR rat did not rise, whereas the blood pressure of the DS rat rose markedly over a 2-month period (Dahl et al., 1962). When salt excretion was facilitated by a thiazide diuretic, DS rats had little rise in blood pressure (Tobian et al., 1979).
Page 418
Kyoto spontaneously hypertensive rats (SHRs) are hypertensive on a fairly low salt diet. SHRs fed very low amounts of salt in the diet do not develop high blood pressure unless salt intake is too low to allow normal growth of the animal (Toal and Leenen, 1983; Wilczynski and Leenen, 1987).
A continuous excess of aldosterone or deoxycorticosterone intake causes rats and pigs to retain sodium and develop hypertension (Kaplan and Lieberman, 1986; Tobian, 1986), but hypertension does not occur when the same amount of deoxycorticosterone or aldosterone is combined with a very low salt diet. Continued high blood pressure tends to cause kidney damage, which further reduces sodium excretion by the kidney. Hypertension is aggravated in the rat by removal of one kidney.
A sodium-dependent type of hypertension can also be induced in rats by subtotal nephrectomy (Chanutin and Ferris, 1932). Hypertension supervenes when reduction in renal mass is accompanied by a high salt intake, but a low salt intake induces little or no hypertension. A similar type of hypertension was induced in dogs by Coleman and Guyton (1969). If the high-salt diet is discontinued, blood pressure returns to normal levels in the dog. A high-salt diet also increases blood pressure in some strains of monkeys (Srinivasan et al., 1984) and chickens (Sapirstein et al., 1950).
Muirhead et al. (1975) fed rats a high-salt diet and injected them with angiotensin, which stimulates the adrenal gland to secrete aldosterone and also stimulates renal proximal tubule sodium reabsorption. When angiotensin is combined with a high-salt diet, these rats develop ''angiotensin-salt" hypertension. Hypertension does not occur if the angiotensin injections are combined with a low-salt diet.
In DS rats made hypertensive by high salt intake, postsalt hypertension occurs after resumption of normal salt intake (Tobian et al., 1975). This is related to changes in the kidney, including destruction of nephrons and damage to glomeruli and tubules in the surviving nephrons. Tobian et al. (1986) reported that high-salt diets consumed for 20 days caused little rise of blood pressure but reduced intrinsic filtration capacity of the kidney to 50% below normal.
Destruction of certain parts of the central nervous system (CNS) can prevent salt-induced hypertension, suggesting that the CNS is involved in salt-induced hypertension (Tobian et al., 1982).
In summary, the combination of a high-salt diet and a diminished capacity for sodium excretion increases blood pressure in many animals. Studies suggest that this combination may also lead to hypertension in humans (Grim et al., 1979).
Familial aggregation of blood pressure has been demonstrated repeatedly (Feinleib et al., 1975; Hamilton et al., 1954; Zinner et al., 1976), but the mode of inheritance has not been determined. A bimodal blood pressure distribution, originally proposed by Platt (1947), has received some recent support in an analysis by McManus (1983). Most studies support a polygenic mode of inheritance (Hamilton et al., 1954; Miller and Grim, 1983). The term hypertension is used in these studies to designate the upper end of the biologic distribution curve for blood pressure in a normal population.
Many kinds of evidence support a hereditary, polygenic predisposition to hypertension (Folkow, 1982; Miall and Oldham, 1963; Pickering, 1968), although the prediction of hypertension based on the blood pressure of first-degree relatives is imperfect and qualitative. For example, Feinleib et al. (1975) showed closer correlations in blood pressure in monozygotic than in dizygotic twins. Consequently, many efforts have been made to identify genetic markers in individuals likely to develop hypertension in response to excess sodium intake or other environmental variables, but these efforts have not been successful. In recent years, several ion transport systems have been described, and there have been attempts to identify abnormalities in such systems that might represent markers for genetic susceptibility to hypertension (Weissberg et al., 1983).
A polygenic predisposition to primary hypertension would have important implications. For example, it would indicate that several or many components of the cardiovascular control mechanisms are involved and that a variety of constellations in genetically controlled responses to environmental influences are likely to be present in human populations. It would also help to explain the apparent heterogeneity of mechanisms in the genesis of hypertension and make it unlikely that one hypothesis will apply to all cases. Furthermore, it complicates efforts to identify genetic markers that could reliably predict who is or is not at risk for development of hypertension.
Mechanisms for Development of Hypertension
The maintenance of high-concentration gradients for sodium and potassium across cell mem-
Page 419
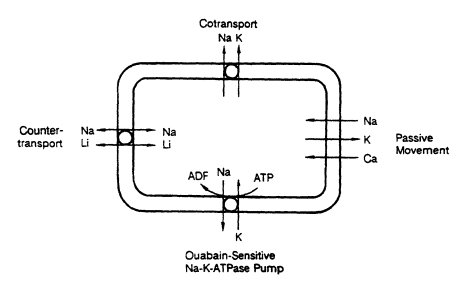
FIGURE 15-1
Four sodium transport mechanisms that have been demonstrated
in red blood cells. From Kaplan and Lieberman (1986).
branes requires expenditure of energy. Several separate systems for maintaining high concentrations of sodium outside cells and high concentrations of potassium inside cells have been described (Hilton, 1986). Many studies of ion transport systems have tested the hypothesis that they reflect the action of one or more genes that determine hereditary susceptibility to hypertension and, thus, may be used to identify people at risk prior to onset of the disease. Figure 15-1 is a schematic diagram of the most important ion transport systems.
In the sodium-potassium adenosine triphosphatase (ATP-ase) pump, a membrane-bound enzyme (ATP-ase) releases energy from ATP to transport sodium and potassium in opposite directions across the cell membrane. The system is inhibited by ouabain and other digitalis glycosides. The activity of this sodium pump is decreased in hypertensives and some of their relatives (Swales et al., 1982). Plasma from hypertensives inhibits this mechanism in the white cells of normotensives (Poston et al., 1981). Abnormalities in this system have been observed in spontaneous and induced hypertension in animals (Overbeck et al., 1981). Variations in activity of this system occur in different racial groups (Beutler et al., 1983) and can be induced by changes in weight and diet (Bradlaugh et al., 1984; De Luise et al., 1980). Thus, the sodium-potassium ATP-ase pump does not appear to be a reliable specific genetic marker for hypertension.
The sodium-potassium cotransport system brings about simultaneous inward or outward coupled transport of sodium and potassium. This system is inhibited by furosemide and was reported to be decreased in patients with hypertension and in their first-degree relatives (Garay and Meyer, 1979). Subsequent results have been inconsistent (Weissberg et al., 1983). Thus, this system has proved to be a disappointing candidate as a genetic marker.
An increased rate of sodium-lithium countertransport has been observed in hypertensives (Canessa et al., 1980) and in many normotensive relatives of white hypertensives, but no relationship to blood pressure has been found in black hypertensives (Weissberg et al., 1983). Countertransport cannot alter the net concentration of sodium within cells. Nevertheless, numerous studies of the sodium-lithium countertransport system have shown consistently increased activity in variable proportions of Caucasian hypertensives studied in different laboratories (Motulsky et al., 1987). Familial aggregation and higher concordance of sodium-lithium countertransport in monozygotic, but not in dizygotic, twins has been demonstrated in several studies (Dadone et al., 1984; Woods et al., 1982). The system is unaffected by sodium restriction and antihypertensive medications (Burke et al., 1984; Cooper et al., 1984). Motulsky et al. (1987) tested several different genetic models for their control of increased countertransport and found that results were most consistent with a major gene determining increased sodium-lithium countertransport activity superimposed on a background of polygenic control of blood pressure. The findings thus support the thesis that hypertension has a heterogeneous genetic basis. Further investigation on genetic linkage relationships for this putative gene may clarify the role of at least one possible genetic marker for hypertension.
Page 420
In several studies, acute or short-term sodium loading and sodium depletion have been used to classify types of genetic susceptibility to salt-induced hypertension. Their duration has ranged from hours to days, or at most, to a few weeks. Most of the studies have shown considerable variation in responsiveness of hypertensive patients to salt, without a clear separation between salt-sensitive and salt-resistant subjects (Bittle et al., 1985; Fujita et al., 1980; Kawasaki et al., 1978). A variety of procedures and protocols have been used by different investigators to identify sodium sensitivity in hypertensives (Logan, 1986). The proportion of hypertensives identified by these investigators as sodium sensitive has varied from 35 to 73%.
Weinberger et al. (1986) showed that acute responses to variations in sodium intake occurred in both hypertensive and normotensive subjects. Responses were somewhat greater in the hypertensives, but there was considerable overlap. Greater increases in blood pressure and lesser natriuretic responses after sodium loading have been reported in blacks under 40 years of age and in blacks and whites older than 40 years (Luft et al., 1979).
The studies cited above fail to show clear-cut differences in sodium sensitivity between hypertensive and normotensive subjects, although quantitative differences were noted in response to the procedures used. All studies were designed to show responses to acute changes in sodium; none were analogous to the changes observed during primary hypertension, where blood pressure rises slowly over many years. These studies do not convincingly demonstrate the presence of sharply defined hereditary sodium sensitivity in the genesis of chronic hypertension.
At present, knowledge of blood pressure in first-degree relatives is the most superior method for predicting the development of hypertension. Although epidemiologic studies provide strong evidence that sensitivity to excess sodium chloride is an important factor in initiating hypertension, there is no certain method to predict individual responses or even for estimating the percentage of a susceptible group of individuals that become hypertensive as a result of excess sodium intake. Other environmental influences, such as psychosocial stresses, obesity, and habitual alcohol intake, may elevate blood pressure singly or in combination with sodium intake, depending on the genetic profile of the individual. Body mass and obesity, and heavy use of ethanol, are powerful determinants of blood pressure in population studies (see Chapter 20). Nevertheless, among the nutrients, indirect evidence is strongest and most extensive for the role of sodium chloride.
Although treatment of established hypertension by sodium chloride restriction is relevant to dietary recommendations, neither a response to such treatment nor the absence of response is adequate to prove or disprove the role of sodium in the etiology of hypertension. Diets with very low levels of sodium chloride, such as a rice diet, were shown to be effective in treating moderate and severe hypertension before modem drug treatment was available (Kempner, 1948). Most studies of moderate sodium restrictions in the treatment of mild to moderate hypertension have been small, brief, and flawed by lack of blinding or absence of appropriate controls. Larger trials over a longer period are needed to clarify the importance of salt restriction in the treatment of hypertension.
Patients whose hypertension had been maintained in the normotensive range by medication over a 5-year period in a large intervention trial (HDFP, 1979a,b) subsequently participated in a second trial (Langford et al., 1985). Eligible subjects were randomized into control and discontinued-medication groups, with and without dietary intervention. Interventions were weight loss or sodium reduction to 40 mEq/day. Both dietary interventions, continued for 56 weeks, increased the likelihood of remaining normotensive without medication. The highest success rate was obtained in nonoverweight, mild hypertensives on sodium restriction. That study demonstrates the value of salt restriction in preventing recurrence of hypertension in previously hypertensive patients. Studies such as that by Langford et al. (1985) also demonstrate the feasibility and safety of prolonged moderate sodium restriction. Clinical trials of hypertension prevention using different levels of dietary salt in normotensive subjects over an extended period would provide additional evidence on the etiologic role of sodium chloride in hypertension.
Gastric Cancer
Correa et al. (1985) reported a high urinary excretion of sodium in Colombian populations with elevated gastric cancer mortality rates and
Page 421
with documented high prevalence of atrophic gastritis. Correa (1982) postulated that salt is important in the development of precursors of stomach cancer, particularly atrophic gastritis. Nevertheless, cross-sectional and time-trend correlation analyses do not show consistent associations between per-capita salt consumption and gastric cancer mortality rates in Japan (Howson et al., 1986; Kono et al., 1983). Difficulties in obtaining valid estimates of salt intake have limited similar analyses in other countries. Some investigators have reported a positive correlation between gastric cancer mortality and stroke incidence and mortality, which they took as a surrogate measure of salt intake (Joossens, 1980; Joossens and Geboers, 1981; Tuomilehto et al., 1984), but others have reported no association (Harrington, 1981; Walker, 1982; Whelton and Goldblatt, 1982).
The association of salt with gastric cancer becomes stronger and more uniform when salt intake from salted, smoked, and pickled foods is considered (Joossens and Geboers, 1984; NRC, 1982; West, 1984). Consumption of salted, smoked, and pickled fish products and other salted products, such as salt-cured ham, sausage, and salami, has been associated with increased gastric cancer risk (Bjelke, 1974; Correa et al., 1983, 1985; Haenszel et al., 1972; Hara et al., 1985; Hirayama, 1979; Ishiwata et al., 1975; Kolonel et al., 1980; Sato et al., 1959, 1961). Similar foods may account for the elevated gastric cancer rates observed in Iceland, Chile, Portugal, and the Adriatic coast of Yugoslavia (Alderson, 1981; Howson et al., 1986).
A concurrence of declining gastric cancer mortality and decreasing consumption of salted, dried fish and salted vegetables in Japan has been noted in time-trend analyses (Japan Ministry of Health and Welfare, 1980). One clinical study indicated that consumption of gherkins pickled in high-salt soy sauce led to abnormal changes in gastric mucosa (MacDonald et al., 1967).
Salt alone has not been carcinogenic in the gastrointestinal tract in a variety of animal bioassays or in short-term studies in rodents (Ohgaki et al., 1984; Takahashi et al., 1983; Tatematsu et al., 1975). In rodents, however, a high salt intake increased absorption of polycyclic aromatic hydrocarbons (PAHs), which are known to be gastric carcinogens (Capoferro and Torgersen, 1974). High-salt diets in the presence of other gastric carcinogens have also been reported to facilitate both gastric cancer initiation (Kodama et al., 1984; Takahashi et al., 1983) and promotion (Hanawa et al., 1980; Ohgaki et al., 1984; Takahashi, 1986), although the evidence for the latter is not consistent (Takahashi et al., 1983).
In addition to salt, salted, smoked, and dried fish contain PAHs (Stewart, 1967) as well as nitrosated products (Kodama et al., 1982; Yano, 1981), which include at least one as yet unidentified substance that can induce glandular stomach tumors in rats (Weisburger, 1981; Weisburger et al., 1980). In one study, mice fed a diet of dried cod containing 7% sodium chloride developed lesions characteristic of acute and chronic gastritis (Sato et al., 1959). In humans, chronic gastritis is suspected of being a precursor to gastric cancer (Correa, 1982).
Despite the lack of direct evidence that dietary salt is a gastric carcinogen per se, experimental evidence suggests that a high salt intake may enhance tumor initiation by irritating and damaging the gastric mucosal barrier, thereby facilitating mutation of the target cell by a suitable carcinogen (Howson et al., 1986). If such enhancement occurs, it may be related in part to a salt-induced reduction in the viscosity of hyaluronic acid, one of the mucopolysaccharides that protect the gastric mucosa (Takahashi et al., 1983).
In summary, stomach cancer is associated with diets containing large amounts of foods preserved with salt and possibly also containing precursors of nitrosamines.
Potassium
Evidence that potassium is important in human nutrition has grown steadily in the past four decades. Prehistoric humans and hominids ate only the food that they could obtain by hunting or collecting vegetation such as roots, fruits, tubers, nuts, grains, and seeds. Such foods contain small amounts of sodium and large amounts of potassium (Denton, 1982; Eaton and Konner, 1985). Studies indicate that present-day hunter-gatherers consume between 200 and 285 mEq (7.8 and 11.0 g of elemental potassium per day) (Denton, 1982; Eaton and Konner, 1985). Today, most people consume less than one-quarter of the potassium eaten by prehistoric hunter-gatherers. Urban whites in the United States eat about 2.5 g of elemental potassium per day (Khaw and Barrett-Connor, 1987). Lower intakes have been observed in blacks, whose average excretion rate has been
Page 422
reported to be 25 to 30 mEq/day (0.98 to 1.17 g/day ) (Grim et al., 1980; Langford, 1985). There is a wide diversity in habitual daily potassium intake among various populations of the world.
Hypertension, Cardiovascular Diseases, and Stroke
A substantial body of evidence in humans and animals indicates that dietary potassium exerts a beneficial effect on hypertension. This is partly because of its effect on lowering blood pressure and partly because of its separate, protective effect against vascular damage and stroke. Several studies have shown that supplemental potassium decreases the blood pressure of hypertensive patients (Addison, 1928; Iimura et al., 1981; Kaplan et al., 1985; MacGregor et al., 1982; Morino et al., 1978; Priddle, 1931; Svetkey et al., 1986).
Populations with habitually low-potassium diets appear to have an increased incidence of various cardiovascular morbid events. In the United States, for example, blacks consume less potassium than do whites (Cushman and Langford, 1983; Dai et al., 1984; Frate and Langford, 1985; Grim et al., 1980; Voors et al., 1983; Walker et al., 1979; Watson et al., 1980; Zinner et al., 1976). These differences in potassium intake may be related to the higher frequency of hypertension and related complications in blacks. Blacks in the southeastern part of the United States who eat a very low-potassium diet appear to have a higher stroke rate than any other geographic or ethnic group in the United States (Langford, 1985). Blacks also have a very high incidence of end-stage renal disease caused by hypertension. The incidence of hypertensive renal damage is 18 times higher in blacks than in whites (Rostand et al., 1982) and is correlated with differences in potassium intake.
Diets in Scotland are relatively low in potassium, averaging about 46 mEq of elemental potassium per day (1.80 g). There is a considerably greater incidence of cardiovascular diseases in Scotland than in France, Italy, or southern England, where higher levels of potassium are consumed (Tobian, 1986). Sasaki et al. (1959) described two adjoining prefectures in northern Japan with contrasting diets. The Aamori diet provides a greater potassium and a lower sodium intake, and the Aamori population has a much lower stroke rate than do people in Akita.
Langford (1985) studied 101 black women about 20 years of age and found no significant correlation between blood pressure and sodium excretion. However, he found a modest positive correlation between the urinary sodium-potassium ratio and diastolic blood pressure. In a study by the Intersalt Cooperative Research Group (1988), potassium excretion was negatively correlated with blood pressure after adjustment for confounding variables.
Walker et al. (1979) found no meaningful correlation between blood pressure and sodium intake in a large cohort in Baltimore, Maryland. However, a negative correlation was found between potassium excretion and recumbent diastolic blood pressure.
Page et al. (1981) found a correlation between blood pressure and sodium intake in men among pastoral nomads in Iran. Women of the same tribe did not show a correlation between blood pressure and sodium excretion, but their blood pressure was positively correlated with the urinary sodium-potassium ratio. Langford (1985) observed a very high correlation between mean urinary sodium-potassium ratio and mean diastolic blood pressure in Norwegian populations from three different areas (Langford, 1985).
In several recent studies (Khaw and Barrett-Connor, 1984; Khaw and Rose, 1982; Reed et al., 1985), investigators found a positive correlation of blood pressure with the dietary sodium-potassium ratio and a negative correlation with the urinary potassium-creatinine ratio and with potassium in the diet. Similarly, potassium intake was inversely correlated with blood pressure (Kromhout et al., 1985). In the Intersalt study, the sodium-potassium ratio followed a pattern similar to that of sodium (Intersalt Cooperative Research Group, 1988).
Khaw and Barrett-Connor (1987) showed that over a period of 12 years the incidence of stroke-related deaths in people over 50 years of age in a retirement community was negatively correlated with the daily intake of potassium. Results were unchanged when the correlation of potassium with death from stroke was adjusted for age, caloric intake, systolic or diastolic blood pressure, and fiber, magnesium, and calcium in the diet. No similar relationship of potassium to coronary artery events was found in this study. The corrected data suggest that an increase of 10 mEq (400 mg) of elemental potassium per day would lead to an approximately 40% decrease in the incidence of stroke-related deaths over the 12 years of the study; this amounts to only one or two extra servings of fruits, fruit juices, vegetables, or potatoes per day.
Page 423
In this study, the protective effect of added potassium in the diet appeared to be independent of any change in blood pressure.
In summary, evidence from epidemiologic studies suggests that a diet with high levels of potassium and low levels of sodium may be beneficial in lowering blood pressure and that a high potassium intake may be independently protective against death from stroke. An intake of ³3.5 g/day of elemental potassium is associated with a beneficial effect, and no threshold for this effect is known.
DS rats fed a diet with 4% sodium chloride and 0.75% potassium developed hypertension, a gradual, progressive destruction of the kidney tubules, and an increase in the wall thickness of the renal arterioles. With supplemental potassium (2.11%), control DS rats had fewer lesions in the renal cortex, outer medulla, and papillae, and the average thickness of their renal arterioles was not greater than that of normotensive DR rats, although there was no lowering of blood pressure in the salt-fed rats (Tobian et al., 1984). Added potassium salts attenuated the expected thickening of the arteriolar walls and resulted in a lowering of renal lesions (Tobian et al., 1984).
In SHRSP rats (a stroke-prone substrain of spontaneously hypertensive rats), a Japanese rat chow diet markedly increased the incidence of stroke compared to that of a U.S. rat chow diet, mainly because the Japanese diet had a 40% lower potassium content than the U.S. diet and a lower concentration of glycine and sulfur-containing amino acids (Tobian et al., 1985a,b). SHRSP rats surviving on 0.75% potassium intake (normal level) had much more histopathological evidence of brain infarcts than did SHRSP animals given a 2.11% high-potassium diet.
Moderately severe hypertension can cause rents, irregularities, and severe stretching in a tense arterial endothelial layer, which could increase the permeability of the endothelial lining (Goldby and Beilin, 1972). If potassium reduces such endothelial irregularities in cerebral arteries, it could also reduce the incidence of strokes.
A protective effect of high-potassium diets against endothelial cell damage could explain both intimal and medial protection, since it is associated with diminished release of endothelial, macrophage, and platelet-derived growth factors. This in turn may result in reduced medial hypertrophy and intimal hypercellularity. Ratios of heart weight to body weight were reduced in hypertensive rats on high-potassium diets, even though blood pressures were the same in animals without potassium supplements.
Potassium added to the diet of the SHRSP preserves normal function in the arterial endothelial cells, including those with chronically high intraarterial pressure. It also prevents thickening of the intimal layer (Tobian, 1986). Moreover, high-potassium diets preserve the full degree of endothelium-dependent relaxation of the arterial wall, whereas the normal potassium diet allows two-thirds of this relaxation to disappear in the SHRSP rats. Those studies suggest that a high-potassium diet protects the arterial endothelial cells against hypertensive damage.
Meneely and Ball (1958) reported that the addition of potassium substantially prolonged the survival rate in Sprague-Dawley rats on high-salt diets, although blood pressure was not reduced below control levels. Gordon and Drury (1956) produced renal hypertension in rabbits by narrowing a renal artery. Supplements of potassium did not reduce blood pressure in these rabbits, but greatly reduced the frequency of mesenteric hemorrhages.
Although the mechanism by which additional dietary potassium reduces deaths is not known, the evidence suggests that it exerts a protective effect partly through reducing blood pressure and partly through an effect on the vascular system independent of blood pressure (Khaw and Barrett-Conner, 1987; Tobian et al., 1985a).
Chloride
Hypertension
It appears from several studies that both sodium and chloride are necessary to produce hypertension, but the matter remains unresolved. In DS rats, nonchloride-containing sodium salt did not raise blood pressure as much as equimolar amounts of sodium chloride (Whitescarver et al., 1984). Similar observations have been made in studies of other animal models with deoxycorticosterone hypertension (Kurtz and Morris, 1985). Conversely, severe hypertension has been reported in Dahl S rats given supplements of sodium citrate (Tobian et al., 1984). Kurtz et al. (1987) studied five hypertensive men who were selected because their blood pressure was normalized after 7 days of sodium chloride restriction to 10 mmol/day (600 mg). Dietary supplements of sodium chloride at 240 mmol/day (14,000 mg), given in capsules, increased blood pressure by 16/8 mm Hg; equimo-
Page 424
lar sodium citrate induced no rise in blood pressure. More studies on the effects of different sodium salts are needed.
Salt Substitutes
Many salt substitutes are now commercially available. They include mixtures of sodium chloride and potassium chloride and mixtures of nonsodium salts, 5'-nucleotides, and herbs. The moderate use of these substances for flavoring food does not harm healthy people. Because of the rapid absorption of the potassium, they may be dangerous if used in large amounts by people with renal insufficiency or certain diseases of the endocrine system. The regular use of these condiments warrants medical supervision.
Summary
Epidemiologic data strongly indicate that habitual sodium intake is an important determinant of blood pressure in humans, but that there is wide variation in genetic susceptibility to salt-induced hypertension. Reliable genetic markers for susceptibility to the effect of sodium have not been identified. Blood pressure rises slowly with age in susceptible people. Once induced by a high sodium intake, blood pressure is not necessarily corrected by resumption of a moderately low intake. Evidence points to the kidney as responsible for this elevation of blood pressure.
Studies of hypertension in a variety of animal species and under various experimental conditions confirm the importance of both sodium and potassium intake as determinants of both normal blood pressure and hypertension. Animal models have contributed in several important ways to present understanding of hypertension. Hereditary susceptibility to the effects of sodium has been demonstrated in rodents, pigs, and primates. In the salt-sensitive DS rats, heredity alone does not produce hypertension, but blood pressure increases progressively during and following sodium loading. In SHRs, hypertension occurs as a result of genetic susceptibility alone and without sodium loading, but the hypertension is greatly aggravated by increased sodium intakes.
Hypertension develops to a greater extent in rats or pigs given deoxycorticosterone or aldosterone than in those given high-salt diets. The mechanisms by which salt causes hypertension in animals have not been fully elucidated and appear to vary somewhat from one animal model to another. A common feature is a reduced ability to excrete sodium, which may result from alterations in regulatory mechanisms or from damage to the kidneys. An important role for the central and autonomic nervous systems in modifying the response to salt loading has been demonstrated in some animal models.
Experimental data indicate that a high salt intake can damage stomach mucosa and induce severe gastritis. These data combined with the consistent epidemiologic findings linking salted, dried, and pickled foods to increased gastric cancer risk suggest that salt can act as a cocarcinogen, perhaps by compromising the gastric mucosal barrier and facilitating initiation by a gastric carcinogen.
Diets with a high sodium content tend to be low in potassium, whereas those with high potassium levels have a low sodium content. An important role for potassium in regulation of blood pressure and in modification of the sequelae of hypertension has been documented in humans and animals. Data strongly indicate that a high potassium intake is protective against fatal stroke in humans and in SHRSP and DS rats. It also protects against arterial hypertrophy and injury in animals. This effect is exerted partly through modulation or reduction of blood pressure, but high potassium intake reduces cardiovascular mortality and morbidity due to hypertension, even apart from its effects on blood pressure. In animal models, potassium also prevents strokes as well as hypertrophy and intimal damage of blood vessels without reducing blood pressure.
Epidemiologic studies suggest that dietary potassium protects against death from strokes in humans. Some studies have shown that potassium supplements lower blood pressure, but others—in human populations and in animal models—suggest that potassium exerts a protective effect against stroke that is unrelated to blood pressure.
Anions appear to modify the effects of cations on blood pressure in some studies, but no recommendation concerning anion nutrient content of the diet is warranted at present.
Directions for Research
Recommendations for research and the rationale for them are presented below;
· Epidemiologic, clinical, and animal studies indicate a wide genetic variability in blood pressure and in the susceptibility of individuals to the effects of electrolyte intake and other environmen-
Page 425
tal variables. Except for such crude indices as race and family history, however, there are no reliable genetic markers for susceptibility to hypertension. More specific markers are needed for genetic susceptibility to sodium chloride and the modifying effect of potassium. The availability of such markers may make possible the targeting of specific dietary recommendations to susceptible people rather than only to the general public.
· Long-term prospective studies of dietary intervention to prevent hypertension in normotensives would be of great value. Such studies should include variables such as sodium restriction, potassium supplementation, weight control, and possibly other variables such as calcium supplementation or variation in protein and polyunsaturated fat. These studies should be conducted in children as well as in adults.
· Research on the fundamental cellular and physiological mechanisms leading to hypertension and vascular injury from hypertension is currently widely pursued and should be encouraged. Such research should include studies of the interactions of sodium, potassium, and their attendant anions and of the relationships between these dietary electrolytes and minerals such as calcium.
· Further research is needed on the interrelationships between electrolytes, other nutrients, and obesity in the fundamental pathogenesis of hypertension. Special attention should be given to the interactions of nutrients and other environmental factors in different age, sex, and racial groups.
· Anionic composition of sodium salts in human hypertension deserves further study. Sodium and chloride are highly correlated in the urine of humans, and it would be difficult to devise a diet for humans in which the major sodium salt would not be sodium chloride. The principal relevance of studies on anions at present is the possibility that the results could be used as a probe for mechanisms of blood pressure regulation.
References
AAP (American Academy of Pediatrics). 1974. Salt intake and eating patterns of infants and children. Pediatrics 53: 115-121.
Addison, W.LT. 1928. Use of sodium chloride, potassium chloride, sodium bromide, and potassium bromide in cases of arterial hypertension which are amenable to potassium chloride. Can. Med. Assoc. J. 18:281-285.
Alderson, M.R. 1981. International Mortality Statistics. Facts on File, New York. 524 pp.
Beauchamp, G.K., M. Bertino, and K. Engelman. 1985. Sensory basis for human salt consumption. Pp. 113-124 in M.J. Horan, M. Blaustein, J.B. Dunbar, W. Kachadorian, N.M. Kaplan, and A.P. Simopoulos, eds. NIH Workshop on Nutrition and Hypertension: Proceedings from a Symposium. Biomedical Information Corp., New York.
Beutler, E., W. Kuhl, and P. Sacks. 1983. Sodium-potassium-ATPase activity is influenced by ethnic origin and not by obesity. N. Engl. J. Med. 309:756-760.
Bittle, C.C., Jr., D.J. Molina, and F.C. Batter. 1985. Salt sensitivity in essential hypertension as determined by the Cosinor Method. Hypertension 7:989-994.
Bjelke, E. 1974. Epidemiologic studies of cancer of the stomach, colon, and rectum; with special emphasis on the role of diet. Scand. J. Gastroenterol. 31:1-235.
Bradlaugh, R., A.M. Heagerty, R.F. Bing, J.D. Swales, and H. Thurston. 1984. Rat thymocyte sodium transport. Effects of changes in sodium balance and experimental hypertension. Hypertension 6:454-459.
Burke, W., S. Hornung, B.R. Copeland, C.E. Furlong, and A.G. Motulsky. 1984. Red cell sodium-lithium countertransport in hypertensives. Pp. 88-99 in H. Villarreal and M.P. Sambhi, eds. Topics in Pathophysiology of Hypertension. Martinus Nijhoff, Boston.
Canessa, M., N. Adragna, H.S. Solomon, T.M. Connolly, and D.C. Tosteson. 1980. Increased sodium-lithium countertransport in red cells of patients with essential hypertension. N. Engl. J. Med. 302:772-776.
Capoferro, R., and O. Torgersen. 1974. The effect of hypertonic saline on the uptake of tritiated 7,12-dimethylbenz[a]anthracene by the gastric mucosa. Scand J. Gastroenterol. 9:343-349.
Chanutin, A., and E.B. Ferris, Jr. 1932. Experimental renal insufficiency produced by partial nephrectomy; control diet. Arch. Intern. Med. 49:767-787.
Coleman, T.G., and A.C. Guyton. 1969. Hypertension caused by salt loading in the dog. III. Onset transients of cardiac output and other circulatory variables. Circ. Res. 25: 153-160.
Conn, J.W. 1949. The mechanisms of acclimatization to heat. Adv. Intern. Med. 3:373-393.
Cooper, R., I. Soltero, K. Liu, D. Berkson, S. Levinson, and J. Stamler. 1980. The association between urinary sodium excretion and blood pressure in children. Circulation 62:97-104.
Cooper, R., K. Liu, M. Trevisan, W. Miller, and J. Stamler. 1982. Urinary sodium excretion and blood pressure in children: absence of a reproducible association. Hypertension 5:135-139.
Cooper, R., M. Trevisan, L Van Horn, E. Larbi, K. Liu, S. Nanas, H. Ueshima, C. Sempos, D. Ostrow, and J. Stamler. 1984. Effect of dietary sodium reduction on red blood cell sodium concentration and sodium-lithium countertransport. Hypertension 6:731-735.
Correa, P. 1982. Precursors of gastric and esophageal cancer. Cancer 50 Suppl. 11:2554-2565.
Correa, P., C. Cuello, L.F. Fajardo, W. Haenszel, O Bolaños, and B. de Ramirez. 1983. Diet and gastric cancer: nutrition survey in a high-risk area. J. Natl. Cancer Inst. 70:673-678.
Correa, P., G. Montes, C. Cuello, W. Haenszel, G. Liuzza, G. Zarama, E. de Marin, and D. Zavala. 1985. Urinary sodium-to-creatinine ratio as an indicator of gastric cancer risk. Natl. Cancer Inst. Monogr. 69:121-123.
Cushman, W.C., and H.G. Langford. 1983. Urinary electrolyte differences in black and white hypertensives: for Veter-
Page 426
ans Administration Cooperative Study Group on Antihypertensive Agents. Clin. Res. 31:843A.
Dadone, M.M., S.J. Hasstedt, S.C. Hunt, J.B. Smith, O.K. Ash, and R.R. Williams. 1984. Genetic analysis of sodium-lithium countertransport in 10 hypertension-prone kindreds. Am. J. Med. Genet. 17:565-577.
Dahl, L.K. 1958. Salt intake and salt need. N. Engl. J. Med. 258:1152-1157.
Dahl, L.K., M. Heine, and L. Tassinari. 1962. Effects of chronic excess salt ingestion: evidence that genetic factors play an important role in susceptibility to experimental hypertension. J. Exp. Med. 115:1173-1190.
Dai, W.S., L.H. Kuller, and G. Miller. 1984. Arterial blood pressure and urinary electrolytes. J. Chronic Dis. 37:75-84.
Dawber, T.R., W.B. Kannel, A. Kagan, R.K. Donabedian, P.M. McNamara, and G. Pearson. 1967. Environmental factors in hypertension. Pp. 255-288 in J. Stamler, R. Stamler, and T.N. Pullman, eds. The Epidemiology of Hypertension: Proceedings of an International Symposium Sponsored by the Chicago Heart Association and the American Heart Association, Chicago, Illinois, February 3-7, 1964. Grune and Stratton, New York.
De Luise, M., G.L. Blackburn, and J.S. Flier. 1980. Reduced activity of red-cell sodium-potassium pump in human obesity. N. Engl. J. Med. 303:1017-1022.
Denton, D. 1982. The Hunger for Salt: An Anthropological, Physiological and Medical Analysis. Springer-Verlag, Berlin. 650 pp.
Eaton, S.B., and M. Konner. 1985. Paleolithic nutrition: a consideration of its nature and current implications. N. Engl. J. Med. 312:283-289.
Feinleib, M., R. Garrison, N. Borhani, R. Rosenman, and J. Christian. 1975. Studies of hypertension in twins. Pp. 3-20 in O. Paul, ed. Epidemiology and Control of Hypertension. Stratton Intercontinental Medical Book Co., New York.
Folkow, B. 1982. Physiological aspects of primary hypertension. Physiol. Rev. 62:347-504.
Frate, D.A., and H.G. Langford. 1985. Potassium and Hypertension. Pp. 147-153 in M.J. Horan, M. Blaustein, J.B. Dunbar, W. Kachadorian, N.M. Kaplan, and A.P. Simopoulos, eds. NIH Workshop on Nutrition and Hypertension: Proceedings from a Symposium. Biomedical Information Corp., New York.
Froment, A., H. Milon, and C. Gravier. 1979. Relation entre consommation sodée et hypertension artérielle. Contribution de l'épidémiologie géographique. Rev. Epidemiol. Sante Publique 27:437-454.
Fujita, T., W.L. Henry, F.C. Bartter, C.R. Lake, and CS. Delea. 1980. Factors influencing blood pressure in salt-sensitive patients with hypertension. Am. J. Med. 69:334-344.
Garay, R.P., and P. Meyer. 1979. A new test showing abnormal net Na+ and K+ fluxes in erythrocytes of essential hypertensive patients. Lancet 1:349-353.
Gleibermann, L. 1973. Blood pressure and dietary salt in human populations. Ecol. Food Nutr. 2:143-156.
Goldby, F.S., and L.J. Beilin. 1972. Relationship between arterial pressure and the permeability of arterioles to carbon particles in acute hypertension in the rat. Cardiovasc. Res. 6:384-390.
Gordon, D.B., and D.R. Drury. 1956. The effect of potassium on the occurrence of petechial hemorrhages in renal hypertensive rabbits. Circ. Res. 4:167-172.
Gothberg, G., S. Lundin, M. Aurell, and B. Folkow. 1983. Responses to slow, graded bleeding in salt-depleted rats. J. Hypertension Suppl. 2:24-26.
Grim, C.E., F.C. Luft, J.Z. Miller, P.L. Brown, M.A. Gannon, and M.H. Weinberger. 1979. Effects of sodium loading and depletion in normotensive first-degree relatives of essential hypertensives. J. Lab. Clin. Med. 94:764-771.
Grim, C.E., F.C. Luft, J.Z. Miller, G.R. Meneely, H.D. Battarbee, C.G. Hames, and L.K. Dahl. 1980. Racial differences in blood pressure in Evans County, Georgia: relationship to sodium and potassium intake and plasma renin activity. J. Chronic Dis. 33:87-94.
Haenszel, W., M. Kurihara, M. Segi, and R.K.C. Lee. 1972. Stomach cancer among Japanese in Hawaii. J. Natl. Cancer Inst. 49:969-988.
Hamilton, M., G.W. Pickering, J.A.F. Roberts, and G.S.C. Sowry. 1954. Aetiology of essential hypertension: role of inheritance. Clin. Sci. 13:273-304.
Hanawa, K., S. Yamada, H. Suzuki, et al. 1980. Effects of sodium chloride on gastric cancer induction by N-methyl-N'-nitro-N-nitrosoguanidine (MNNG) in rats. P. 49 in Proceedings of the Thirty-ninth Annual Meeting of the Japanese Cancer Association. Japanese Cancer Association, Tokyo.
Hara, N., K. Sakata, M. Nagai, Y. Fujita, T. Hashimoto, and H. Yanagawa. 1985. Geographical difference of mortality of digestive cancers and food consumption. Gan No Rinsho 30:1665-1674.
Harrington, J.S. 1981. Advances in cancer epidemiology in South Africa. S. Afr. Cancer Bull. 25:9-18.
HDFP (Hypertension Detection and Follow-up Program Cooperative Group). 1979a. Five-year findings of the hypertension detection and follow-up program. I. Reduction in mortality of persons with high blood pressure, including mild hypertension. J. Am. Med. Assoc. 242:2562-2571.
HDFP (Hypertension Detection and Follow-up Program Cooperative Group). 1979b. Five-year findings of the hypertension detection and follow-up program. II. Mortality by race-sex and age. J. Am. Med. Assoc. 242:2572-2577.
Henry, J.P., and J.C. Cassel. 1969. Psychosocial factors in essential hypertension. Recent epidemiologic and animal experimental evidence. Am. J. Epidemiol. 90:171-200.
Hilton, P.J. 1986. Cellular sodium transport in essential hypertension. N. Engl. J. Med. 314:222-229.
Hirayama, T. 1979. Diet and cancer. Nutr. Cancer 1:67-81.
Hofman, A., A. Hazebroek, and H.A. Valkenburg. 1983. A randomized trial of sodium intake and blood pressure in newborn infants. J. Am. Med. Assoc. 250:370-373.
Holden, R.A., A.M. Ostfeld, D.H. Freeman, Jr., K.G. Hellenbrand, and D.A. D'Atri. 1983. Dietary salt intake and blood pressure. J. Am. Med. Assoc. 250:365-369.
Howson, C.P., T. Hiyama, and EL. Wynder. 1986. The decline in gastric cancer: epidemiology of an unplanned triumph. Epidemiol. Rev. 8:1-27.
Iimura, O., T. Kijima, K. Kikuchi, A. Miyama, T. Ando, T. Nakao, and Y. Takigami. 1981. Studies on the hypotensive effect of high potassium intake in patients with essential hypertension. Clin. Sci. 61:77s-80s.
Intersalt Cooperative Research Group. 1988. Intersalt: an international study of electrolyte excretion and blood pressure. Results for a 24 hour urinary sodium and potassium excretion. Br. Med. J. 297:319-328.
Ishiwata, H., P. Boriboon, Y. Nakamura, M. Harada, A. Tanimura, and M. Ishidate. 1975. Studies on in vivo formation of nitroso compounds. II. Changes of nitrite and nitrate
Page 427
concentrations in human saliva after ingestion of vegetables or sodium nitrate. J. Food Hyg. Soc. 16:19-24.
Jacobs, D.R., Jr., J.T. Anderson, and H. Blackburn. 1979. Diet and serum cholesterol: do zero correlations negate the relationship? Am. J. Epidemiol. 110:77-87.
James, W.P., A. Ralph, and C.P. Sanchez-Castillo. 1987. The dominance of salt in manufactured food in the sodium intake of affluent societies. Lancet 1:426-429.
Japan Ministry of Health and Welfare. 1980. Kosei-no-shihyo "Cancer." Health and Welfare Statistics Association, Japan Ministry of Health and Welfare, Tokyo.
Joossens, J.V. 1980. Stroke, stomach cancer and salt: a possible clue to the prevention of hypertension. Pp. 489-508 in H. Kesteloot and J.V. Joossens, eds. Epidemiology of Arterial Blood Pressure. Martinus Nijhoff, The Hague.
Joossens, J.V., and J. Geboers.' 1981. Nutrition and gastric cancer. Proc. Nutr. Soc. 40:37-46.
Joossens, J.V., and J. Geboers. 1984. Diet and environment in the etiology of gastric cancer. Pp. 167-183 in B. Levin and R.H. Riddell, eds. Frontiers in Gastrointestinal Cancer. Elsevier, New York.
Kaplan, N.M., and E. Lieberman. 1986. Clinical Hypertension, 4th ed. Williams & Wilkins, Baltimore. 492 pp.
Kaplan, N.M., A. Carnegie, P. Raskin, J.A. Heller, and M. Simmons. 1985. Potassium supplementation in hypertensive patients with diuretic-induced hypokalemia. N. Engl. J. Med. 312:746-749.
Karvonen, M.J., and S. Punsar. 1977. Sodium excretion and blood pressure of West and East Finns. Acta Med. Scand. 202:501-507.
Kawasaki, T., C.S. Delea, F.C. Bartter, and H. Smith. 1978. The effect of high-sodium and low-sodium intakes on blood pressure and other related variables in human subjects with idiopathic hypertension. Am. J. Med. 64:193-198.
Kempner, W. 1948. Treatment of hypertensive vascular disease with rice diet. Am. J. Med. 4:545-577.
Kesteloot, H., B.C. Park, C.S. Lee, E. Brems-Heyns, J. Claessens, and J.V. Joossens. 1980. A comparative study of blood pressure and sodium intake in Belgium and in Korea. Eur. J. Cardiol. 11:169-182.
Khaw, K.T., and E. Barrett-Connor. 1984. Dietary potassium and blood pressure in a population. Am. J. Clin. Nutr. 39: 963-968.
Khaw, K.T., and E. Barrett-Connor. 1987. Dietary potassium and stroke-associated mortality. A 12-year prospective population study. N. Engl. J. Med. 316:235-240.
Khaw, K.T., and E. Barrett-Connor. 1988. The association between blood pressure, age, and dietary sodium and potassium: a population study. Circulation 77:53-61.
Khaw, K.T., and G. Rose. 1982. Population study of blood pressure and associated factors in St. Lucia, West Indies. Int. J. Epidemiol. 11:372-377.
Kodama, M., H. Saito, and Z. Yamaizumi. 1982. Formation of alkylureas in the environment. Pp. 131-136 in H. Bartsch, I.K. O'Neill, M. Castegnaro, M. Okada, and W. Davis, eds. N-Nitroso Compounds: Occurrence and Biological Effects—Proceedings of the VIIth International Symposium on N-Nitroso Compounds Held in Tokyo, 28 September-1 October 1981. IARC Scientific Publ. No. 41. International Agency for Research on Cancer, Lyon, France.
Kodama, M., T. Kodama, H. Suzuki, and K. Kondo. 1984. Effect of rice and salty rice diets on the structure of mouse stomach. Nutr. Cancer 6:135-147.
Kolonel, L.N., M.W. Hinds, and .H. Hankin. 1980. Cancer patterns among migrant and native-born Japanese in Hawaii in relation to smoking, drinking, and dietary habits. Pp. 327-340 in H.B. Gelboin, B. MacMahon, T. Matsushima, T. Sugimura, S. Takayama, and H. Takebe, eds. Genetic and Environmental Factors in Experimental and Human Cancer: Proceedings of the 10th International Symposium of the Princess Takamatsu Cancer Research Fund, Tokyo, 1979. Japan Scientific Societies Press, Tokyo.
Kono, S., M. Ikeda, and M. Ogata. 1983. Salt and geographical mortality of gastric cancer and stroke in Japan. J. Epidemiol. Community Health 37:43-46.
Kromhout, D., E.B. Bosschieter, and C.D. Coulander. 1985. Potassium, calcium, alcohol intake and blood pressure: the Zutphen Study. Am. J. Clin. Nutr. 41:1299-1304.
Kurtz, T.W., and P.C. Morris. 1985. Dietary chloride as a determinant of "sodium dependent" hypertension and hypercalemia. Pp. 137-146 in M.J. Horan, M. Blaustein, J.B. Dunbar, W. Kachadorian, N.M. Kaplan, and A.P. Simopoulos, eds. NIH Workshop on Nutrition and Hypertension: Proceedings from a Symposium. Biomedical Information Corp., New York.
Kurtz, T.W., H.H. Al-Bander, and R.C. Morris, Jr. 1987. "Salt-sensitive" essential hypertension in men. Is the sodium ion alone important? N. Engl. J. Med. 317:1043-1048.
Langford, H.G. 1985. Dietary potassium and hypertension. Pp. 147-153 in M.J. Horan, M. Blaustein, J.B. Dunbar, W. Kachadorian, N.M. Kaplan, and A.P. Simopoulos, eds. NIH Workshop on Nutrition and Hypertension: Proceedings from a Symposium. Biomedical Information Corp., New York.
Langford, H.G., M.D. Blaufox, A. Oberman, C.M. Hawkins, J.D. Curb, G.R. Cutter, S. Wassertheil-Smoller, S. Pressel, C. Babcock, J.D. Abernethy, J. Hotchkiss, and M. Tyler. 1985. Dietary therapy slows the return of hypertension after stopping prolonged medication. J. Am. Med. Assoc. 253: 657-664.
Liu, K., R. Cooper, J. McKeever, P. McKeever, R. Byington, I. Soltero, R. Stamler, F. Gosch, E. Stevens, and J. Stamler. 1979. Assessment of the association between habitual salt intake and high blood pressure: methodological problems. Am. J. Epidemiol. 110:219-226.
Ljungman, S., M. Aurell, M. Hartford, J. Wikstrand, L. Wilhelmsen, and G. Berglund. 1981. Sodium excretion and blood pressure. Hypertension 3:318-326.
Logan, A.G. 1986. Sodium manipulation in the management of hypertension. The view against its general use. Can. J. Physiol. Pharmacol. 64:793-802.
Luft, F.C., C.E. Grim, N. Fineberg, and M.C. Weinberger. 1979. Effects of volume expansion and contraction in normotensive whites, blacks, and subjects of different ages. Circulation 59:643-650.
Luft, F.C., M.C. Weinberger, and C.E. Grimm. 1982. Sodium sensitivity and resistance in normotensive humans. Am. J. Med. 72:726-736.
MacDonald, W.C., F.H. Anderson, and S. Hashimoto. 1967. Histological effect of certain pickles on the human gastric mucosa: a preliminary report. Can. Med. Assoc. J. 96:1521-1525.
MacGregor, G.A., S.J. Smith, N.D. Markandu, R.A. Banks, and G.A. Sagnella. 1982. Moderate potassium supplementation in essential hypertension. Lancet 2:567-570.
Malhotra, S.L. 1970. Dietary factors causing hypertension in India. Am. J. Clin. Nutr. 23:1353-1363.
Mark, A.L., W.J. Lawton, F.M. Abboud, A.E. Fitz, W.E.
Page 428
Connor, and D.D. Heistad. 1975. Effects of high and low sodium intake on arterial pressure and forearm vascular resistance in borderline hypertension. A preliminary report. Circ. Res. 36 Suppl. 1:1-194 to 1-198.
McManus, I.C. 1983. Bimodality of blood pressure levels. Star. Med. 2:253-258.
Meneely, G.R., and C.O.T. Ball. 1958. Experimental epidemiology of chronic sodium chloride toxicity and the protective effect of potassium chloride. Am. J. Med. 25: 713-725.
Miall, W.E., and P.D. Oldham. 1963. The heredity factor in arterial blood pressure. Br. Med. J. 1:75-80.
Miller, J.Z., and C.E. Grim. 1983. Heritability of blood pressure. Pp. 79-90 in T.A. Kotchen and J.M. Kotchen, eds. Clinical Approaches to High Blood Pressure in the Young. John Wright, PSG Inc., Boston.
Morino, T., R. McCaa, and H.G. Langford. 1978. Effect of potassium loading on blood pressure, sodium excretion and plasma renin activity in hypertensive patients. Clin. Res. 26:805A.
Motulsky, A.G., W. Burke, P.R. Billings, and R.H. Ward. 1987. Hypertension and the genetics of red cell membrane abnormalities. Pp. 150-166 in G. Bock and G.M. Collins, eds. Molecular Approaches to Human Polygenic Disease: Ciba Foundation Symposium 130. Wiley, Chicester, United Kingdom.
Muirhead, E.E., B.E. Leach, J.O. Davis, F.B. Armstrong, Jr., J.A. Pitcock, and W.L. Brosius. 1975. Pathophysiology of angiotensin-salt hypertension. J. Lab. Clin. Med. 85:734-745.
NRC (National Research Council). 1982. Diet, Nutrition, and Cancer. Report of the Committee on Diet, Nutrition, and Cancer, Assembly of Life Sciences. National Academy Press, Washington, D.C. 478 pp.
Ohgaki, H., T. Kato, K. Morino, N. Matsukura, S. Sato, S. Takayama, and T. Sugimura. 1984. Study of the promoting effect of sodium chloride on gastric carcinogenesis by N-methyl-N'-nitro-N-nitrosoguanidine in inbred Wistar rats. Gann 75:1053-1057.
Oliver, W.J., E.L Cohen, and J.V. Neel. 1975. Blood pressure, sodium intake and sodium related hormones in the Yanomamo Indians, a ''no-salt" culture. Circulation 52: 146-151.
Overbeck, H.W., D.D. Ku, and J.P. Rapp. 1981. Sodium pump activity in arteries of Dahl salt-sensitive rats. Hypertension 3:306-312.
Page, L.B. 1976. Epidemiologic evidence on the etiology of human hypertension and its possible prevention. Am. Heart J. 91:527-534.
Page, L.B. 1979. Hypertension and atherosclerosis in primitive and acculturating societies. Pp. 1-12 in J.C. Hunt, ed. Hypertension Update, Vol. 1. Health Learning Systems, Lyndhurst, N.J.
Page, L.B., D.E. Vandevert, K. Nader, N.K. Lubin, and J.R. Page. 1981. Blood pressure of Qash'qai pastoral nomads in Iran in relation to culture, diet, and body form. Am. J. Clin. Nutr. 34:527-538.
Pickering, G. 1968. High Blood Pressure, 2nd ed. Grune & Stratton, New York. 717 pp.
Platt, R. 1947. Heredity in hypertension. Q. J. Med. 16:111-133.
Pooling Project Research Group. 1978. Relationship of blood pressure, serum cholesterol, smoking habit, relative weight and ECG abnormalities to incidence of major coronary events: final report of the Pooling Project. J. Chronic Dis. 31:201-306.
Porter, G.A. 1983. Chronology of the sodium hypothesis and hypertension. Ann. Intern. Med. 98:720-723.
Poston, L., R.B. Sewell, S.P. Wilkinson, P.J. Richardson, R. Williams, E.M. Clarkson, G.A. MacGregor, and H.E. De Wardener. 1981. Evidence for a circulating sodium transport inhibitor in essential hypertension. Br. Med. J. 282: 847-849.
Priddle, W.W. 1931. Observations on the management of hypertension. Can. Med. Assoc. J. 25:5-8.
Prineas, R.J., and H. Blackburn. 1985. Clinical and epidemiologic relationships between electrolytes and hypertension. Pp. 63-85 in M.J. Horan, M. Blaustein, J.B. Dunbar, W. Kachadorian, N.M. Kaplan, and A.P. Simopoulos, eds. NIH Workshop on Nutrition and Hypertension: Proceedings from a Symposium. Biomedical Information Corp., New York.
Reed, D., D. McGee, K. Yano, and J. Hankin. 1985. Diet, blood pressure, and multicollinearity. Hypertension 7:405-410.
Rostand, S.G., K.A. Kirk, E.A. Rutsky, and B.A. Pate. 1982. Racial differences in the incidence of treatment for end-stage renal disease. N. Engl. J. Med. 306:1276-1279.
Sanchez-Castillo, C.P., S. Warrender, T.P. Whitehead, and W.P. James. 1987a. An assessment of the sources of dietary salt in a British population. Clin. Sci. 72:95-102.
Sanchez-Castillo, C.P., W.J. Branch, and W.P. James. 1987b. A test of the validity of the lithium-marker technique for monitoring dietary sources of salt in men. Clin. Sci. 72:87-94.
Sapirstein, L.A., W.L. Brandt, and D.R. Drury. 1950. Production of hypertension in the rat by substituting hypertonic sodium chloride solutions for drinking water. Proc. Soc. Exp. Biol. Med. 73:82-85.
Sasaki, N., T. Mitsuhashi, and S. Fukushi. 1959. Effects of the ingestion of large amounts of apples on blood pressure of farmers in Akita prefecture. Igaku To Seibutsugaku 51:103-105.
Sato, T., T. Fukuyama, T. Suzuki, J. Takayanagri, T. Murakami, N. Shiotsuki, R. Tanaka, and R. Tsuji. 1959. Studies of the causation of gastric cancer. 2. The relation between gastric cancer mortality rate and salted food intake in several places in Japan. Koshu Eisei In Kenkyu Hokoku 8:187-198.
Sato, T., T. Fukuyama, T. Suzuki, J. Takayangai, and Y. Sakai. 1961. Studies of the causation of gastric cancer: intake of highly brined foods in several places with high mortality rate in Europe. Koshu Eisei In Kenkyu Hokoku 10: 9-17.
Schlierf, G., L. Arab, B. Schellenberg, P. Oster, R. Mordasini, H. Schmidt-Gayk, and G. Vogel. 1980. Salt and hypertension: data from the "Heidelberg Study." Am. J. Clin. Nutr. 33:872-875.
Simpson, F.O. 1985. Blood pressure and sodium intake. Pp. 175-190 in C.J Bulpitt, ed. Handbook of Hypertension, Vol. 6. The Epidemiology of Hypertension. Elsevier Science Publ., New York.
Srinivasan, S.R., E.R. Dalferes,.Jr., R.H. Wolf, B. Radhakrishnamurthy, T.A. Foster, and G.S. Berenson. 1984. Variability in blood pressure response to dietary sodium intake among African green monkeys (Cercopithecus aethiops). Am. J. Clin. Nutr. 39:792-796.
Page 429
Stewart, H.L. 1967. Experimental alimentary tract cancer. Natl. Cancer Inst. Monogr. 25:199-217.
Svetkey, L.P., W.E. Yarger, J.R. Feussner, E. DeLong, and P.E. Klotman. 1986. Placebo-controlled trial of oral potassium in the treatment of mild hypertension. Clin. Res. 34: 487A.
Swales, J.D., R.F. Bing, A. Heagerty, J.E. Pohl, G.I. Russell, and H. Thurston. 1982. Treatment of refractory hypertension. Lancet 1:894-896.
Takahashi, M. 1986. Enhancing effect of a high salt diet on gastrointestinal carcinogenesis. Gan No Rinsho 32:667-673.
Takahashi, M., T. Kokubo, F. Furukawa, Y. Kurokawa, M. Tatematsu, and Y. Hayashi. 1983. Effect of high salt diet on rat gastric carcinogenesis induced by N-methyl-N'-nitro-N-nitrosoguanidine. Gann 74:28-34.
Tatematsu, M., M. Takahashi, S. Fukushima, M. Hananouchi, and T. Shirai. 1975. Effects in rats of sodium chloride on experimental gastric cancers induced by N-methyl-N'nitro-N-nitrosoguanidine or 4-nitroquinoline-l-oxide. J. Natl. Cancer Inst. 55:101-106.
Tao, S.C., R.S. Tsai, Z.G. Hong, and J. Stamler. 1984. Timed overnight sodium and potassium excretion and blood pressure in steel workers in Peoples' Republic of China, (PRC)-North. CVD Epidemiol. Newsletter (AHA) 35:64.
Toal, C.B., and F.H. Leenen. 1983. Dietary sodium restriction and development of hypertension in spontaneously hypertensive rats. Am. J. Physiol. 245:H1081-H1084.
Tobian, L. 1983. Human essential hypertension: implications of animal studies. Ann. Intern. Med. 98:729-734.
Tobian, L. 1986. High potassium diets markedly protect against stroke deaths and kidney disease in hypertensive rats, a possible legacy from prehistoric times. Can. J. Physiol. Pharmocol. 64:840-848.
Tobian, L. 1988. The Volhard lecture: potassium and sodium in hypertension. J. Hypertens. Suppl. 4:S12-S24.
Tobian, L, Jr., and J.T. Binion. 1952. Tissue cations and water in arterial hypertension. Circulation 5:754-758.
Tobian, L., Jr., and J. Binion. 1954. Artery wall electrolytes in renal and DCA hypertension. J. Clin. Invest. 33:1407-1414.
Tobian, L., M.A. Johnson, J. Lange, and S. Magraw. 1975. Effect of varying perfusion pressures on the output of sodium and renin and the vascular resistance in kidneys of rats with "post-salt" hypertension and Kyoto spontaneous hypertension. Circ. Res. 36 Suppl. 1:1-162 to 1-170.
Tobian, L, J. Lange, S. Azar, J. Iwai, D. Koop, and K. Coffee. 1977. Reduction of intrinsic natriuretic capacity in kidneys of Dahl hypertension-prone rats. Trans. Assoc. Am. Physicians 90:401-406.
Tobian, L, J. Lange, J. Iwai, K. Hiller, M.A. Johnson, and P. Goossens. 1979. Prevention with thiazide of NaCl-induced hypertension in Dahl "S" rats: evidence for a Na-retaining humoral agent in "S" rats. Hypertension 1:316-323.
Tobian, L., M. Ganguli, A. Goto, T. Ikeda, M.A. Johnson, and J. Iwai. 1982. The influence of renal prostaglandins, central nervous system and NaCl on hypertension in Dahl S rats. Clin. Exp. Pharmacol. Physiol. 9:341-353.
Tobian, L., D. MacNeill, M.A. Johnson, M.C Ganguli, and J. Iwai. 1984. Potassium protection against lesions of the renal tubules, arteries, and glomeruli and nephron loss in salt-loaded hypertensive Dahl S rats. Hypertension 6:I-170 to I-176.
Tobian, L., J. Lange, K. Ulm, L. Wold, and J. Iwai. 1985a. Potassium reduces cerebral hemorrhage and death rate in hypertensive rats, even when blood pressure is not lowered. Hypertension 7 Suppl 1:I-110 to I-114.
Tobian, L., Y. Uehara, and J. Iwai. 1985b. Prostaglandin alterations in barely hypertensive Dahl S rats. Trans. Assoc. Am. Physicians XCVIII:378-383.
Tobian, L., J. Lange, K. Ulm, L. Wold, and J. Iwai. 1986. NaCl feeding in Dahl S rats markedly reduces GFR and RBF in isolated kidneys perfused at normotensive inflow pressures. J. Hypertension 4 Suppl. 6:S370-S372.
Tuomilehto, J., J. Geboers, J.V. Joossens, J.T. Salonen, and A. Tanskanen. 1984. Trends in stomach cancer and stroke in Finland. Comparison to Northwest Europe and USA. Stroke 15:823-828.
Voors, A.W., E.R. Dalferes, Jr., G.C. Frank, G.G. Aristimuno, and G.S. Berenson. 1983. Relation between ingested potassium and sodium balance in young blacks and whites. Am. J. Clin. Nutr. 37:583-594.
Walker, A.R. 1982. Changing disease patterns in South Africa. S. Afr. Med. J. 61:126-129.
Walker, W.G., P.K. Whelton, H. Saito, R.P. Russell, and J. Hermann. 1979. Relation between blood pressure and renin, renin substrate, angiotensin II, aldosterone and urinary sodium and potassium in 574 ambulatory subjects. Hypertension 1:287-301.
Watson, R.L., H.G. Langford, J. Abernethy, T.Y. Barnes, and M.J. Watson. 1980. Urinary electrolytes, body weight, and blood pressure: pooled cross-sectional results among four groups of adolescent females. Hypertension 2:93-98.
Watt, G.C., C.J. Foy, and J.T. Hart. 1983. Comparison of blood pressure, sodium intake, and other variables in offspring with and without a family history of high blood pressure. Lancet 1:1245-1248.
Weinberger, M.H., J.Z. Miller, F.C. Luft, C.E. Grimm, and N.S. Fineberg. 1986. Definitions and characteristics of sodium sensitivity and blood pressure resistance. Hypertension 8 Suppl. II:II-127 to II-134.
Weisburger, J.H. 1981. N-Nitroso compounds: diet and cancer trends. An approach to the prevention of gastric cancer. Pp. 305-317 in R.A. Scanlan and S.R. Tannenbaum, eds. N-Nitroso Compounds. Based on a Symposium Cosponsored by the Divisions of Agricultural and Food Chemistry and Pesticide Chemistry of the 181st Meeting of the American Chemical Society, Atlanta, Georgia, March 31-April 1, 1981. ACS Symposium Series 174. American Chemical Society, Washington, D.C.
Weisburger, J.H., H. Marquardt, N. Hirota, H. Mori, and G.M. Williams. 1980. Induction of cancer of the glandular stomach in rats by an extract of nitrite-treated fish. J. Natl. Cancer Inst. 64:163-167.
Weissberg, P.L., M.R. Wilkins, and L.J. Kricka. 1983. Test for circulating Na+-K+ ATPase inhibitors. Lancet 1:1219.
West, R.C., ed. 1984. European Collaborative Study on the Role of Diet and Other Factors in the Aetiology of Atrophic Gastritis: A Precancerous Lesion of Gastric Cancer. Euro-Nut, The Netherlands.
Whelton, P.K., and P. Goldblatt. 1982. An investigation of the relationship between stomach cancer and cerebrovascular disease: evidence for and against the salt hypothesis. Am. J. Epidemiol. 115:418-427.
Whitescarver, S.A., C.E. Ott, B.A. Jackson, G.P. Guthrie, Jr., and T.A. Kotchen. 1984. Salt-sensitive hypertension:
Page 430
contribution of chloride. Science 223:1430-1432.
WHO (World Health Organization). 1978. Arterial Hypertension. Report of a WHO Expert Committee. Technical Report Series 628. World Health Organization, Geneva. 58 pp.
Wilczynski, E.A., and F.H. Leenen. 1987. Dietary sodium intake and age in spontaneously hypertensive rats: effects on blood pressure and sympathetic activity. Life Sci. 41:707-715.
Woods, J.W., R.J. Falk, A.W. Pittman, P.J. Klemmer, B.S. Watson, and K. Namboodiri. 1982. Increased red-cell sodium-lithium countertransport in normotensive sons of hypertensive parents. N. Engl. J. Med. 306:593-595.
Yano, K. 1981. Alkylating activity of processed fish products treated with sodium nitrite in simulated gastric juice. Gann 72:451-454.
Zinner, S.H., H.S. Margolius, B. Rosner, H.R. Keiser, and E.H. Kass. 1976. Familial aggregation of urinary kallikrein concentration in childhood: relation to blood pressure, race and urinary electrolytes. Am. J. Epidemiol. 104:124-132.


















