4
Similarity in Form and Function of the Hippocampus in Rodents, Monkeys, and Humans
ROBERT E. CLARK*† ∥ AND LARRY R. SQUIRE*†‡§
We begin by describing an historical scientific debate in which the fundamental idea that species are related by evolutionary descent was challenged. The challenge was based on supposed neuroanatomical differences between humans and other primates with respect to a structure known then as the hippocampus minor. The debate took place in the early 1860s, just after the publication of Darwin’s famous book. We then recount the difficult road that was traveled to develop an animal model of human memory impairment, a matter that also turned on questions about similarities and differences between humans and other primates. We then describe how the insight that there are multiple memory systems helped to secure the animal model and how the animal model was ultimately used to identify the neuroanatomy of long-term declarative memory (sometimes termed explicit memory). Finally, we describe a challenge to the animal model and to cross-species comparisons by considering the case of the concurrent discrimination task, drawing on findings from humans and monkeys. We suggest that analysis of such cases, based on the understanding that there are multiple memory systems with different properties, has served to emphasize the similarities in memory function across mammalian species.
_____________
*Veterans Affairs Medical Center, San Diego, CA 92161; and Departments of †Psychiatry,‡ Neurosciences, and §Psychology, University of California at San Diego, La Jolla, CA 92093. ∥ To whom correspondence should be addressed. E-mail: reclark@ucsd.edu.
Here, we describe the successful development during the past few decades of a model of human memory and human memory impairment in monkeys and rodents. This work, which focused on the hippocampus and anatomically related brain regions, provides a robust illustration of similar neuroanatomy and function across species. The expectation that the study of experimental animals should yield insights into the organization of human memory is certainly reasonable when considered in the light of evolution and evolutionary theory. Indeed, the central idea behind the study of nonhuman mammals is the notion that what can be learned about mammalian behavior and mammalian brains can improve understanding of the human brain and the human condition. However, this perspective was not so easily attained. We begin by describing an historical scientific debate in which this fundamental idea (continuity between nonhumans and humans) was challenged on the basis of supposed neuroanatomical differences between humans and other primates. The debate took place in the early 1860s, just after the 1859 publication of Darwin’s (1859) On the Origin of Species. We then describe the difficult road that was traveled to develop an animal model of human memory impairment.
HIPPOCAMPUS MINOR AND BRAIN ANATOMY
In 1859, Sir Richard Owen (1804–1892), the renowned British comparative anatomist and paleontologist, and superintendent of the Natural History Department of the British Museum, member of the Royal Society, and author of more than 600 scientific publications, published a provocative and controversial paper (Owen, 1859). He argued that humans should be reclassified as distinct and separate from other primates. Up to this point, Linnaeus classification had placed humans, apes, and monkeys into the single order Primates. Owen’s new classification created not only a new order for humans (Bimana “two-handed ones”) but a new subclass for humans (Archencephala “ruling brain”), which placed humans well apart from other primates. On the final page of his essay, Owen provided the following summary: “Man is the sole species of his genus, the sole representative of his order and subclass. Thus I trust has been furnished the confutation of the notion of a transformation of the ape into man, which appears from a favourite old author to have been entertained by some in his day” (Owen, 1859).
It has been argued (Gross, 1993a) that this reclassification must have been due, at least in part, to Owen’s knowledge of the forthcoming publication of Charles Darwin’s (1859) On the Origin of Species. It was well known even before publication that Darwin’s book would emphasize a clear physiological and anatomical relationship among species and argue
that all species descended from common stock (in addition to proposing the theory of “natural selection” as the mechanism for driving evolution).
Owen based his new classification on three ostensible neuroanatomical differences in brain structure between humans and other animals, including nonhuman primates. He suggested that these three anatomical features were fundamental because these structures were not just different in humans and other primates but were found uniquely in the human brain (Owen, 1859). According to Owen, the first fundamental difference between humans and animals was that only in humans was there a posterior lobe. Specifically, only the human cerebrum extended over and beyond the extent of the cerebellum. The second difference was that only humans have a posterior horn or cornu of their lateral ventricles (a portion of the lateral ventricle that extends posteriorly and laterally). The third and most important difference was that only humans have a hippocampus minor.
Within the floor of the lateral ventricles are two prominent structures: a large structure referred to at that time as the hippocampus major (now known simply as the hippocampus) and a smaller structure known then as the hippocampus minor. The hippocampus minor is a ridge in the floor of the posterior horn of the lateral ventricle. The original term for the hippocampus minor, and the term in current use, is the calcar avis. The calcar avis is a Latin term meaning “cock’s spur” due to its resemblance to the spurs found on rooster legs.
Owen’s reclassification was met with a vigorous opposition, led principally by Thomas Henry Huxley (1825–1895). Huxley was a British biologist, paleontologist, and anatomist. In his lifetime, he served as president of the Royal Society, president of the Geological Society, and president of the British Association for the Advancement of Science, and he was one of Darwin’s most vocal scientific supporters. The debate between Owen and Huxley was anticlimactic in one sense because Huxley was easily able to demonstrate that Owen’s three criteria, including the hippocampus minor, were not only identifiable in all primate species available for study but that the relative size of the hippocampus minor was larger in some primate species than in humans (Huxley, 1861a,b, 1863). During the course of his investigations, Huxley also discovered that the hippocampal fissure in primates actually consisted of two separate fissures. The more anterior fissure retained the name hippocampal fissure, whereas the more posterior fissure was renamed by Huxley as the calcarine sulcus because the calcar avis, or hippocampus minor, is, in fact, formed by the deep inward penetration of the calcarine sulcus (Huxley, 1861a). The hippocampus minor debate can be said to have concluded when Sir Charles Lyell, Great Britain’s leading geologist and one of its most eminent scientists, reviewed in detail the hippocampus minor debate (Lyell, 1863). Lyell authoritatively concluded,
without reservation, that the hippocampus minor debate was settled in Huxley’s favor. Lyell’s conclusion was all the more compelling because Lyell had not fully embraced Darwin’s theory of evolution.
Despite its brief lifetime, the debate over the hippocampus minor was important. It has been suggested by some historians (Desmond, 1984; Gross, 1993b) that Huxley intended to show not only that Owen was incorrect but that he must have been intentionally obfuscating the facts. Huxley admitted as much in a letter he sent to Sir Charles Lyell on August 17, 1862 [the letter is reproduced in Huxley (1900)]. In this letter, Huxley references a line from a well-known poem by the noted paleontologist, Sir Philip Egerton. Huxley wrote: “I do not think you will find room to complain of any want of distinctness in my definition of Owen’s position touching the Hippocampus question. I mean to give the whole history of the business in a note, so that the paraphrase of Sir Ph. Egerton’s line ‘To which Huxley replies that Owen he lies,’ shall be unmistakable.” Huxley’s effort damaged the scientific reputation of perhaps the most prominent antievolutionist of the time and helped pave the way for the broad scientific acceptance of evolution by scientists during the following decades [excellent reviews of the hippocampus minor debate are provided by Gross (1993a,b)].
In the end, the hippocampus minor debate provided another example of the similarities in neuroanatomical organization among all primate species, including humans. The same would later prove to be true for hippocampal function and for the organization of long-term memory across species ranging from rodents to primates. However, as described in the sections that follow, the continuity of memory function across species was in serious doubt for nearly two decades.
HIPPOCAMPUS AND THE MODERN ERA OF MEMORY RESEARCH
The modern era of memory research, and the first insights about the hippocampus and the organization of memory, began with the description of patient H.M. by William Beecher Scoville and Brenda Milner. Patient H.M. had a history of minor and major seizures that were unresponsive to antiepileptic medication. Eventually, with the consent of the family, the surgeon Scoville attempted to relieve the seizures with an experimental surgical intervention that involved resecting the medial aspect of the temporal lobe bilaterally. Subsequently, MRI scans showed that the lesion was bilaterally symmetrical and included the medial temporal polar cortex, virtually all the entorhinal cortex and amygdaloid complex, and the anterior half of the intraventricular aspect of the hippocampal formation [i.e., dentate gyrus (DG), hippocampus, subicular complex]. The perirhi-
nal cortex was substantially damaged as well, with some sparing of its ventrocaudal aspect (Corkin et al., 1997). The surgery succeeded in that it reduced the frequency and severity of the seizures, but it left H.M. with profound amnesia.
The subsequent systematic evaluation of H.M. and other patients with similar damage established three fundamental principles (Squire, 2009). First, memory is a distinct cerebral ability that is separate from other cognitive functions, such as perception, intelligence, personality, and motivation. Second, short-term memory and long-term memory are distinct functions, because H.M. had severely impaired long-term memory. However, he could maintain and use information for a short time in immediate memory (and working memory) so long as the material could be effectively rehearsed. With distraction, the information was lost. Third, medial temporal lobe structures are not the ultimate repository of long-term memory because H.M.’s memory for remote events remained largely intact.
At the time that H.M. was first described, the anatomy of the medial temporal lobe was poorly understood and it was not known what specific damage (within the large region included in his surgery) was responsible for his memory impairment. Accordingly, efforts were begun to address this question in experimental animals. In fact, these efforts began almost immediately when Scoville himself went to Montreal and performed the same surgery in monkeys that he had performed with H.M (Correll and Scoville, 1965). However, these monkeys and others with medial temporal lobe lesions were able to learn tasks that seemed similar to tasks that H.M. could not learn.
The difficulty was that it was not yet appreciated that humans and experimental animals can approach ostensibly similar tasks using different strategies (Multiple Memory Systems). For example, monkeys learn visual discrimination tasks gradually over many trials in a fashion that is now referred to as habit learning. In the monkey, this kind of learning depends on the basal ganglia rather than the medial temporal lobe (Mishkin et al., 1984; Teng et al., 2000). By contrast, humans learn the same task by directly memorizing the stimulus material. In the decade that followed H.M.’s initial description, extensive work in rats with hippocampal lesions also failed to reproduce H.M.’s impairment. For example, rats with hippocampal lesions, like monkeys, performed normally on simple visual discrimination problems (Kimble, 1963). Lesioned rats were also unimpaired at learning to press a bar for food and at various shock avoidance tasks [reviewed by Douglas (1967)]. In short, it was clear from the work carried out during the 1960s that the behavioral impairments observed in rats with hippocampus lesions did not provide an adequate description of the memory impairment seen in humans with hippocampal damage.
Accordingly, investigators studying rats tended not to relate their work to studies of memory-impaired patients and were more likely to interpret their findings within the framework of response inhibition as first outlined by Pavlov (1927). In fact, in a review of the literature on the hippocampus and behavior published a decade after the description of H.M., Robert Douglas (1967) wrote:
Hippocampal lesions obviously do not impair learning in general, even when the learning involves retention for long periods of time. Thus, the animal and human data would appear to be in contradiction. This contradiction could be “resolved” by postulating that the hippocampus has a different basic function in man and beast. Such a solution to this problem is generally unacceptable to physiological psychologists, however. Another possible resolution of this paradox is that the recent memory loss in man is a secondary effect of a different type of primary disorder. The author has chosen the latter course, and suggests that the recent memory loss in man is a genuine phenomenon, but that it is a byproduct of interference during storage and not due to a lack of ability to store, per se.
These comments made it clear that a large animal literature was substantially out of register with the human work and that experimentalists were beginning to doubt the basic idea that medial temporal lobe damage produced an impairment in memory. Furthermore, if one did suppose that medial temporal lobe damage in humans impaired memory, then to understand the data from animals, one must consider the possibility, however unsatisfactory, that memory is organized differently in humans and other animals.
MULTIPLE MEMORY SYSTEMS
During the 1960s and 1970s, it was not yet understood that different tasks of learning and memory could be supported by different brain systems. Many of the tasks given to animals with hippocampal lesions were, in fact, skill-based tasks that amnesic patients would have been able to acquire (Milner, 1962), or they were tasks that animals could learn as a skill even if humans tended to learn the task by memorizing the material. Establishing an animal model of human memory impairment would require developing tasks for animals that assessed the same kind of memory that is impaired in humans after medial temporal lobe damage.
The kind of memory that is impaired is now typically referred to as declarative memory (Cohen and Squire, 1980). Declarative memory is flexibly expressed and provides conscious access to facts and events. It is impaired when structures of the medial temporal lobe are damaged. In contrast, the term nondeclarative memory refers to various acquired
skills and abilities that are not accessible to conscious knowledge but are expressed through performance and depend on different brain systems (Squire, 2004). Nondeclarative memory is independent of the medial temporal lobe.
A key advance in establishing a model of human medial temporal lobe amnesia was the development of one-trial memory tests for the monkey. In 1974, Gaffan suggested that many tests of memory in animals with hippocampal damage might not be similar to the tests that reveal memory impairment in patients. Accordingly, if one intends to relate work in animals to work in humans, it is not adequate to use any convenient task in which an animal must utilize memory. Rather, one must use “specifically designed animal analogs of those tests that do reveal impairment in human amnesiacs” (Gaffan, 1974). One of the tests that Gaffan used was a one-trial test of visual recognition memory, which had been refined for use in monkeys by Benjamin Weinstein (1941). In this test (initially referred to as the “matching-from-sample delayed reaction procedure”), monkeys were presented with a single object that they displaced for a food reward (the sample phase). Memory for the sample object was then tested after a delay by presenting the sample object together with a new object (choice phase). The monkey was trained to select (i.e., match) the object that had been presented previously during the sample phase. Unique objects were used on each trial so that judgment of familiarity was sufficient to identify the correct object. The demand on memory could be increased by increasing the delay between the sample and choice phases. This task became known as the “delayed matching to sample” (DMS) task. Monkeys with lesions of the fornix, thought to mimic the effects of hippocampal damage, impaired performance in a delay-dependent fashion. Performance was intact at a delay of 10 seconds but impaired at delays of 70 seconds and 130 seconds (Gaffan, 1974).
An alternative version of the DMS task was also developed and referred to as the delayed nonmatching to sample (DNMS) task (Mishkin and Delacour, 1975). In this case, monkeys were trained to select the new object during the choice phase (i.e., nonmatch the sample object). In a critical experiment (Mishkin, 1978), monkeys were given lesions designed to mimic the damage sustained by H.M. The key group received hippocampus plus amygdala lesions, together with the cortex underlying these two structures. Other monkeys received smaller lesions of the hippocampus (and underlying cortex) or lesions of the amygdala (and underlying cortex). Note that the underlying cortical areas damaged by surgery were not specifically targeted but were damaged during the surgical approach to the hippocampus and amygdala. Postoperatively, the animals reacquired the nonmatching rule, and the delay between the sample and choice phases was then increased progressively from 10 seconds to 30 seconds, 60
seconds, and 120 seconds. The result was that hippocampal or amygdala lesions (combined with the underlying cortical areas that were damaged in this study) yielded only a mild impairment but that the combined lesion (hippocampus plus amygdala and underlying cortex) produced a marked, delay-dependent deficit that was especially severe at the longer delays.
The demonstration of delay-dependent impairments in performance was critical for identifying this impairment as a memory impairment. When performance is spared at short delays and selectively impaired at longer delays (when the demand on memory is greatest), one can rule out a variety of alternative explanations for the impairment (e.g., the ability to identify objects; motivational changes; as well as secondary effects of the lesion, such as hyperactivity, increased distractibility, motor impairments, and other nonspecific effects). This study, and subsequent studies, that relied especially on the DNMS task (Zola-Morgan et al., 1982), documented the successful establishment of an animal model of human medial temporal lobe amnesia.
The DNMS task with trial-unique stimuli was subsequently adapted for use with rats (Mumby et al., 1990). This new task used trial-unique objects that could be displaced to obtain food rewards just as in the version developed for monkeys (Mumby et al., 1990). Additionally, the test apparatus was designed so that the delay between the sample and choice phases could be as short as 4 seconds. Work with this task demonstrated that lesions of the hippocampus, or lesions of the cortical regions adjacent to the hippocampus, produced a delay-dependent memory impairment similar to what had been reported in the monkey (Mumby and Pinel, 1994). Thus, this new task successfully extended the animal model of medial temporal lobe memory impairment to the rat. Further work with this task demonstrated that the impairment in rats with hippocampal lesions was unambiguously a delay-dependent memory impairment (Clark et al., 2001) and that delay dependence was not a simple consequence of the large amount of training given at the short delay during learning of the nonmatching rule (Ringo, 1993).
Since the successful establishment of the DNMS task, another memory task has become widely used. This task allows one-trial learning to be studied in humans and experimental animals in a straightforward and simple way (Clark and Squire, 2010). The visual paired-comparison (VPC) task was initially developed to study the development of vision in human infants (Fagan, 1970). In this task, two identical stimuli are presented side-by-side for a period (e.g., 1–2 min). Immediately afterward, the infant is presented with the recently viewed stimulus along with a novel stimulus. The finding is that infants preferentially look at the novel stimulus, presumably because they remember the already presented and now less interesting stimulus. This work established the VPC task as a reliable test
of visual recognition memory. The task takes advantage of a spontaneous preference for novelty (which is preserved across mammalian species), and it has the advantage of not requiring any verbal instruction or rule learning. This feature makes the task an excellent tool for studying preverbal human infants and experimental animals.
The VPC test was subsequently adapted for the monkey (Bachevalier et al., 1993), the rat (Ennaceur and Delacour, 1988), and the mouse (Tang et al., 1999). When used with primates, this task is generally referred to as the VPC task, because it is a test of visual recognition memory. In the rat and mouse, the animal physically explores the objects so that visual, olfactory, and tactile information can be used to guide performance during the test phase. Accordingly, in the rodent, this task is typically referred to with other descriptors, such as “novel object recognition” (NOR), “novel object preference,” “spontaneous object preference,” or “spontaneous object recognition.” Here, we refer to it as the NOR task.
Recent findings with the VPC and NOR tasks are consistent with findings using the DNMS task. For example, memory-impaired patients with damage that included the hippocampus exhibited memory impairment on the VPC test (McKee and Squire, 1993). Furthermore, monkeys (Zola et al., 2000), rats (Clark et al., 2000), and mice (Hammond et al., 2004) with selective hippocampal damage, or transient disruption of hippocampal function, exhibited delay-dependent memory impairments when tested with the VPC/NOR task [a summary of the work in animals with hippocampal lesions is provided in Clark and Martin (2005) and Squire et al. (2007)]. The NOR task has now largely replaced the DNMS task for the assessment of recognition memory in the experimental animal (Clark and Martin, 2005; Winters et al., 2008).
ORGANIZATION OF LONG-TERM DECLARATIVE MEMORY
Work in the experimental animal during the past three decades used these tasks to identify a system of anatomically connected structures in the medial temporal lobe that, when damaged, produce memory impairment like the impairment first revealed by the study of H.M. [reviewed in Clark and Squire (2010)]. The system of structures important for memory includes the hippocampus and the adjacent entorhinal, perirhinal, and parahippocampal cortices (Squire and Zola-Morgan, 1991). The amygdala is not part of this system. Note also that in the rat, the parahippocampal cortex is referred to as the postrhinal cortex; however, anatomical organization is preserved across the three species (Fig. 4.1).
The hippocampus, defined here as the DG, cornu ammonis (CA)3, and CA1, is anatomically situated to receive highly processed information from widespread neocortical regions through three cortical areas, the ento-
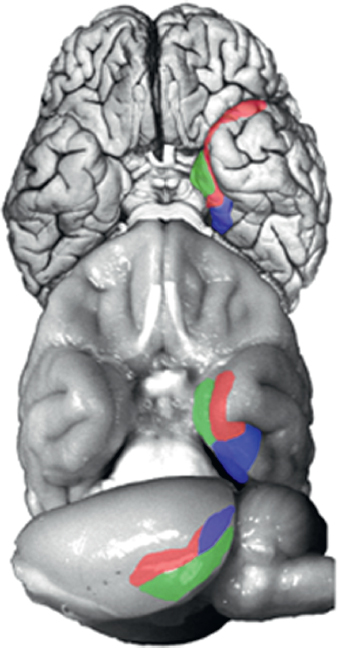
FIGURE 4.1 Ventral view of a human brain (Top), ventral view of a monkey brain (Middle), and lateral view of a rat brain (Bottom). The major cortical components of the medial temporal lobe are highlighted [perirhinal cortex (red), parahippocampal/postrhinal (blue), and entorhinal cortex (green)]. The organization and connections of these structures are highly conserved across these species. Brains are not drawn to scale. [NOTE: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu/catalog.php?record_id18573.]
rhinal, perirhinal, and parahippocampal cortices, as well as through other direct projections to the entorhinal cortex from outside the temporal lobe (Fig. 4.2). The main pathway for the transmission of sensory information to the hippocampus is the entorhinal cortex. Layer II of the entorhinal cortex provides its major input. This unidirectional projection, forming part
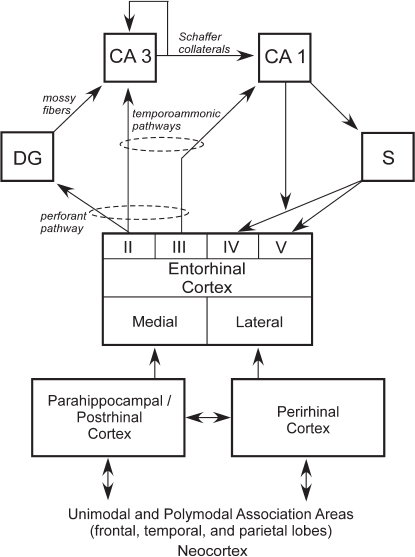
FIGURE 4.2 Schematic view of the connections within the medial temporal lobe memory system. The hippocampus, defined here as the DG, CA3, CA1, and subiculum (S), is anatomically situated to receive highly processed information from widespread neocortical regions through three temporal cortical areas, the entorhinal, perirhinal, and parahippocampal cortices (in the rat, the term postrhinal cortex replaces the term parahippocampal cortex), as well as through other direct projections to the entorhinal cortex from areas outside the temporal lobe. The figure shows a simplified view of the way in which information enters the hippocampus from the superficial layers (II and III) of the entorhinal cortex and then flows in a largely unidirectional feed forward direction to return (predominantly) ultimately to the deep layers of entorhinal cortex (IV and V). The output and input layers refer to the entire entorhinal cortex and not to its medial or lateral subdivisions.
of the perforant pathway, provides a substantial input to the DG, which, in turn, provides the major input to CA3 via the mossy fiber projection. There is also a smaller unidirectional projection to CA3 from layer II of the entorhinal cortex. CA3 provides the major input to CA1 via the Schaffer collateral/commissural pathway, but there is also a substantial recurrent associational projection within the CA3 field. CA1 also receives a direct projection from layer III of the entorhinal cortex, as does the subiculum (the temporoammonic pathway). Both the Schaffer collateral and temporoammonic projections to CA1 are unidirectional. CA1 primarily projects to the subiculum but also sends a projection to layer V of the entorhinal cortex. The subiculum sends projections primarily to layers IV and V of the entorhinal cortex (Witter and Amaral, 2004).
The hippocampus can be viewed as residing at the end of the processing hierarchy of the medial temporal lobe, receiving input from both the perirhinal and parahippocampal cortices, as well as the entorhinal cortex (Fig. 4.2). Guided by the anatomy, it seems plausible that the hippocampus extends and combines functions performed by the structures that project to it (Squire et al., 2007). Note also that anatomical connections from different regions of neocortex enter the medial temporal lobe at different points. Thus, the higher visual areas TE and TEO project preferentially to the perirhinal cortex. Conversely, spatial information that comes to the medial temporal lobe from parietal cortex arrives exclusively at the parahippocampal cortex. Consistent with these anatomical facts, damage to parahippocampal cortex was found to impair spatial memory more than did damage to perirhinal cortex (Parkinson et al., 1988; Malkova and Mishkin, 2003), and damage to perirhinal cortex impaired performance on the visual DNMS task more than did damage to parahippocampal cortex (Zola-Morgan et al., 1994). Finally, the relative segregation of visual and spatial information appears to be maintained within the entorhinal cortex, such that the lateral entorhinal cortex processes more visual information and the medial entorhinal cortex processes more spatial information.
CHALLENGE TO THE ANIMAL MODEL AND TO CROSS-SPECIES COMPARISONS
As discussed earlier, during the 1960s and early 1970s, the development of an animal model of human memory and human memory impairment was challenged by the fact that animals could use nondeclarative memory to solve some memory tasks that humans typically approached using declarative memory. It therefore became important to understand under what conditions this occurs and to identify what kind of memory is used in each case.
This issue can be illustrated by considering concurrent discrimination learning, a standard task that has been used to study mammalian memory for more than 50 years. In a common version of the task, eight pairs of objects are presented side-by-side five times each day, one pair at a time in a mixed order, for a total of 40 trials. One object in each pair is always correct, and a choice of the correct object yields a reward. The left/right position of the correct object varies randomly across trials. Humans readily learn this task, performing at a level better than 80 percent correct after only 1 or 2 days of training (Squire et al., 1988). That performance ordinarily depends on declarative memory, and on memorizing which object is correct in each pair, is demonstrated by the fact that task performance was correlated highly with the ability to describe the objects and by the fact that memory-impaired patients performed poorly during the period that their controls mastered the task. In contrast, this same task was acquired at a normal rate by monkeys with hippocampal lesions (Teng et al., 2000). Importantly, monkeys with hippocampal damage that also included damage to the caudate nucleus (Teng et al., 2000) or monkeys with selective damage to the caudate nucleus (Fernandez-Ruiz et al., 2001) were severely impaired at learning the task. These findings suggested that monkeys learn the concurrent discrimination task via a trial-and-error stimulus/ response strategy that relies on nondeclarative memory. If so, the question naturally arises as to whether humans also have a similar capacity for nondeclarative memory that could support learning of this same task.
To address this question, patients with large lesions of the medial temporal lobe, and essentially no residual declarative memory, were given extended training on the concurrent discrimination task over a period of several weeks (Bayley et al., 2005). Fig. 4.3A shows the performance of four control participants on the task. The controls learned the task easily within three sessions. One key feature of declarative memory is that it can be expressed flexibly. To demonstrate this feature, the controls were given a sorting task 3–6 days after learning. The 16 objects used in this task were placed together on a table, and participants were told that some of the objects had been consistently designated as correct. They were then asked to sort the objects, placing the correct objects to one side of the table and the other objects to the other side. Fig. 4.3A shows that controls had no difficulty with this modification of the task.
By contrast, patient E.P. gradually learned the object pairs across 36 test sessions (Fig. 4.3B) but then failed the sorting task altogether 5 days later. Nonetheless, immediately afterward, E.P. performed well in the standard task format (Fig. 4.3B). Seventeen days later, E.P. again failed the sorting task but performed well in the original task format. The results for patient G.P. were essentially the same as for E.P. (Fig. 4.3C).
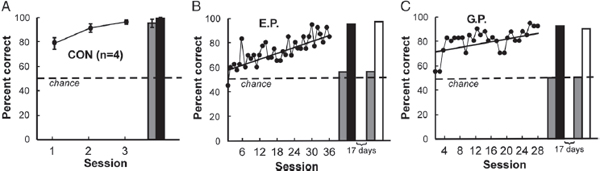
FIGURE 4.3 Performance on the concurrent discrimination task. (A) Controls (n = 4) learned the task easily within three sessions and performed well on the sorting task 3–6 days later (gray bar). The black bar shows performance immediately afterward, when participants were asked to verbalize their choices rather than reach for objects. Results are means ± SEM. (B) Patient E.P. gradually learned the object pairs across 18 weeks. Five days later, he failed the sorting task (gray bar) but then, immediately afterward, performed well in the standard task format while verbalizing his responses (black bar). Seventeen days later, E.P. again failed the sorting task (gray bar) but performed perfectly when 40 trials were given exactly as in original training (white bar). (C) Patient G.P. learned the object pairs gradually during 14 weeks. Like E.P., he failed the sorting task on two different occasions, 5 days after training and again 17 days later. In both instances, he performed well immediately afterward when the original task format was reinstated [verbalizing (black bar), standard task (white bar)]. The dashed line indicates chance performance (50% correct). Reproduced from Bayley et al. (2005).
Some informal observations are informative. At the beginning of each testing day, the patients were asked about the objects and about what had occurred on previous visits. However, they were never able to remember anything about earlier test sessions and could not describe how the objects had been used. Nonetheless, as training proceded from day to day, both patients (without explicit instructions about what to do on each trial) readily picked up one of the objects in each pair and turned it over to receive feedback (the word “correct” or “incorrect” was printed under the base of each object).
Thus, both patients gradually acquired the standard eight-pair object discrimination task over many weeks; however, at the start of each session, they could not describe the task, the instructions, or the objects. The acquired knowledge was rigidly organized, and performance collapsed when the task format was altered. The findings indicate that humans possess a robust capacity for gradual trial-and-error learning that can operate outside awareness for what is learned and independent of the medial temporal lobe. What is acquired is inflexible and altogether diffe-
rent from declarative memory. These findings show that humans possess a nondeclarative learning system like other primates and suggest what the characteristics of this system are likely to be (e.g., inflexible, inaccessible to awareness).
In summary, the development of a model of human memory and human memory impairment in the monkey and rat is a success story, and the work has provided important insight into the anatomy and organization of mammalian memory. Still, challenges can arise because these species have evolved under different selection pressures that can bias them to approach tasks with different strategies than the strategies used by humans. Analysis of such cases, based on our understanding that there are multiple memory systems with different properties, has, in the end, served to emphasize the similarities in memory function across mammalian species.
ACKNOWLEDGMENTS
This work was supported by the Medical Research Service of the Department of Veterans Affairs, National Science Foundation Grant 1120395, and National Institute of Mental Health Grant 24600.
















