6
The Evolution of Episodic Memory
TIMOTHY A. ALLEN* AND NORBERT J. FORTIN*†
One prominent view holds that episodic memory emerged recently in humans and lacks a “(neo)Darwinian evolution.” Here, we review evidence supporting the alternative perspective that episodic memory has a long evolutionary history. We show that fundamental features of episodic memory capacity are present in mammals and birds and that the major brain regions responsible for episodic memory in humans have anatomical and functional homologs in other species. We propose that episodic memory capacity depends on a fundamental neural circuit that is similar across mammalian and avian species, suggesting that protoepisodic memory systems exist across amniotes and, possibly, all vertebrates. The implication is that episodic memory in diverse species may primarily be due to a shared underlying neural ancestry, rather than the result of evolutionary convergence. We also discuss potential advantages that episodic memory may offer, as well as species-specific divergences that have developed on top of the fundamental episodic memory architecture. We conclude by identifying possible time points for the emergence of episodic memory in evolution, to help guide further research in this area.
_____________
*Center for the Neurobiology of Learning and Memory, Department of Neurobiology and Behavior, University of California, Irvine, CA 92697. †To whom correspondence should be addressed. E-mail: norbert.fortin@uci.edu.
In humans, episodic memory has been defined as the capacity to recall specific experiences, as if one were to “mentally time travel” to reexperience individual events (Tulving, 1972, 2002). Although a prominent view holds that episodic memory is unique to humans (Tulving and Markowitsch, 1998; Tulving, 2002), accumulating evidence indicates that birds and rodents can demonstrate a memory capacity that satisfies behavioral criteria for episodic memory (Clayton et al., 2003a; Eichenbaum et al., 2005; Crystal, 2010). Does this evidence imply that episodic memory capacity is fundamentally conserved across avian and mammalian species? Or does it suggest “episodic-like” memory capacity evolved separately in a few species and thus is the result of convergent evolution? Notably, these important questions cannot be answered by focusing on behavior alone because it is difficult, perhaps impossible, to distinguish between homologous and analogous memory capacities. Here, our objective is to shed light on the potential evolution of episodic memory. We go beyond previous efforts by integrating the behavioral evidence across species with a comparative analysis of the neurobiology and neural mechanisms underlying episodic memory capacity. We also discuss the potential functions of episodic memory in an evolutionary context, as well as species-specific divergences.
EPISODIC MEMORY CAPACITY ACROSS SPECIES
Episodic memory refers to the memory for specific personal experiences. Although accurate, this definition does not capture the considerable challenge associated with distinguishing episodic memory from other memory capacities. A common mistake is to assume that one-trial learning is a sufficient criterion for episodic memory capacity. This is clearly not the case, as nonepisodic memories can be formed after a single exposure [e.g., conditioned taste aversion or familiarity (Morris, 2001; Clayton et al., 2003a; Eichenbaum et al., 2007)]. In this section, we consider the main approaches used to define and demonstrate episodic memory capacity across species.
Subjective Measures of Episodic Recall
Because the concept of episodic memory was first studied in cognitive psychology, one approach is to define it in terms of the subjective experience associated with episodic recall. Specifically, Tulving (2002) proposed that episodic recall involves the ability to “mentally time travel” to reexperience specific events, a capacity that requires a sense of self, subjective time, and autonoetic awareness (conscious awareness that the experience occurred in the past). Although this definition may capture the phenom-
enological aspects associated with episodic memory in humans, it relies entirely on verbal reports of subjective mental experiences. Because this definition of episodic memory precludes its investigation in animals, the hypothesis that this capacity is unique to humans lacks falsifiability. The absence of objective measures for episodic memory is also not conducive to rigorous scientific investigation in human studies. A more productive approach to defining episodic memory is to identify fundamental features that can be measured experimentally.
Receiver Operating Characteristics
The main objective of the receiver operating characteristics (ROC) approach is to use signal detection analyses to characterize recognition memory performance. More specifically, this method can be used to objectively quantify the relative contributions of episodic recollection versus familiarity in a recognition memory task. Although this approach was originally developed for human studies, it was successfully adapted to rodents and provided strong evidence that rodents have recollective and familiarity processes similar to those of humans (Fortin et al., 2004). However, considerable effort is required to adjust the experimental parameters (e.g., response biases) for each species. Therefore, although this approach has distinct advantages [for a comprehensive review, see Eichenbaum et al. (2007, 2010)], it is unlikely to become widely used across species.
Memory for “Events in Context”
The events-in-context approach capitalizes on the fact that, in the episodic memory system, information about specific events is tied to the spatial, temporal, and other situational contexts in which they occurred (Tulving, 1972; Mishkin et al., 1997; Clayton and Dickinson, 1998). Based on this operational definition, demonstrations that animals can remember events in context (Clayton and Dickinson, 1998; Fortin et al., 2002; Babb and Crystal, 2006) provided compelling evidence that core properties of episodic memory are present in nonhumans. This capacity is often termed episodic-like to emphasize that, whereas it does not address the phenomenological aspects associated with episodic memory in humans, it satisfies three key behavioral criteria (Clayton et al., 2003a):
(i) Content: The individual remembers information about the event (“what”) and its context of occurrence (e.g., “where” or “when” it happened).
(ii) Structure: The information about the event and its context is integrated in a single representation.
(iii) Flexibility: The memory can be expressed to support adaptive behavior in novel situations.
These criteria have provided a solid theoretical framework for behavioral tests of episodic memory. It is important to note that the criteria are usually satisfied using converging evidence from multiple studies, as it is impractical to address them all in every experiment. Here, we examine the three main approaches used to study the memory for events in context: (i) “what-where-when,” (ii) “what-where,” and (iii) “what-when.” The distinct content requirements of these models provide an opportunity to investigate different aspects of episodic memory capacity.
Memory for What-Where-When
An influential animal model of episodic memory took advantage of the natural caching behavior of scrub jays. In an ingenious paradigm, Clayton and Dickinson (1998) demonstrated that scrub jays could remember what food they stored (worms or peanuts), as well as where (the location in the cage) and when (4 hours or 124 hours ago) it was cached, thus fully satisfying the content criterion. Similar evidence of what-where-when memory has also been reported in other bird species, including other corvids [magpies (Zinkivskay et al., 2009)] and noncorvids [black-capped chickadees (Feeney et al., 2009)]. This approach has also been adapted to many mammalian species, including rats (Ergorul and Eichenbaum, 2004; Eacott et al., 2005; Babb and Crystal, 2006; Kart-Teke et al., 2006), mice (Dere et al., 2005), meadow voles (Ferkin et al., 2008), pigs (Kouwenberg et al., 2009), nonhuman primates (Hoffman et al., 2009; Martin-Ordas et al., 2010), and humans (Hayne and Imuta, 2011; Holland and Smulders, 2011). It is important to note that the structure and flexibility criteria have been much less investigated than the content criterion, so it remains to be determined whether all these species will meet all three behavioral criteria. As of now, there is evidence for what-where-when integration (structure criterion) in birds (Clayton et al., 2001), rodents (Ergorul and Eichenbaum, 2004), and primates (Hoffman et al., 2009). Evidence for the flexibility criterion comes from the demonstration that the what-where-when memory can be updated with new information [birds (Clayton and Dickinson, 1999; Clayton et al., 2003b); rodents (Babb and Crystal, 2006)], and that it can be expressed spontaneously [i.e., without training or in response to an unexpected test; birds (Singer and Zentall, 2007); rodents (Dere et al., 2005; Kart-Teke et al., 2006)]. Although this approach has been momentous, leading to the development of a number of animal models of episodic memory, it also has limitations. In particular, the content criterion is very stringent, requiring memory for what, where, and when. On one hand,
this is a positive aspect of the model as it established a very high threshold for the first convincing behavioral demonstration of episodic memory in animals. On the other hand, this criterion may be overly restrictive. In fact, there is no clear evidence that all episodic memories contain all three types of information. Therefore, other forms of memory for events in context should also be considered episodic, such as memories involving a subset of the two (e.g., what-where), other types of contextual information [e.g., internal context (Kennedy and Shapiro, 2004)], or possibly where-when associations [no “what” component (Mankin et al., 2012)].
Memory for What-Where
This approach focuses on the memory for the spatial context of episodic memory, the ability to remember where specific events occurred. It is important to note that this capacity does not simply correspond to spatial memory (memory for “where”), as it requires animals to remember specific what-where associations (i.e., specific items in specific places). In these paradigms, the “what” component refers to the presentation of a specific item (e.g., odor, object). The “where” component varies depending on the species, typically referring to a specific place in an environment in rodent studies, or to a specific location on a screen (or complex visual scene) in primate studies. Tasks involving item-place associations have been used extensively in rats (e.g., Gilbert and Kesner, 2002, 2003; Day et al., 2003; Rajji et al., 2006) and nonhuman primates [e.g., item-scene associations (Gaffan, 1994)], particularly to study the neural basis of episodic memory. Paradigms relying on spontaneous preference, which require no training, have also been developed [e.g., Dix and Aggleton (1999)]. A more detailed review of what-where approaches, including their use as preclinical tests for assessment of cognitive function in animal models of aging and Alzheimer’s disease, is available elsewhere (Snigdha et al., 2013).
Memory for What-When
This approach requires subjects to remember the temporal context in which specific events occurred, a defining feature of episodic memory (Tulving, 1972, 2002). There are different forms of memory for when events occurred, including memory for the order of events in a sequence, for how long ago events happened, and for the time of day at which they took place (Friedman, 1993; Roberts, 2002; Eichenbaum and Fortin, 2003; Crystal, 2010; Eacott and Easton, 2010; Jacobs et al., 2013). The vast majority of studies have focused on memory for the order of events, which reflects the capacity of episodic memory to preserve the “flow of events”
as they occurred in experience (Tulving, 1972, 2002). The typical paradigm involves the presentation of a sequence of items (e.g., odors, objects), followed by a choice between two of the presented items. Memory for order is expressed by selecting [e.g., Fortin et al. (2002) and Kesner et al. (2002)], or preferentially exploring [e.g., Hannesson et al. (2004)], the item that appeared earlier in the sequence. Importantly, information about the spatial context is irrelevant to performance. This basic approach has been used in rodents (Fortin et al., 2002; Kesner et al., 2002), and similar approaches have been developed in nonhuman primates (Petrides, 1995; Naya and Suzuki, 2011; Templer and Hampton, 2013) and humans (Kumaran and Maguire, 2006; Lehn et al., 2009; Ross et al., 2009). Notably, the NIH Toolbox Cognition Battery proposes a what-when paradigm, which requires memory for sequences of pictured events, as the new standard measure for episodic memory capacity in humans [for a review, see Snigdha et al. (2013) and Weintraub et al. (2013)].
Section Summary
The evidence reviewed strongly suggests that core properties of episodic memory are present across mammals, as well as in a number of bird species. Although the ROC method has distinct advantages, the memory for the events-in-context approach is more practical and widely used. Therefore, the latter is more appropriate to examine episodic memory capacity across species and shed light on its evolution. What-where-when paradigms have the strictest behavioral criteria and thus are better suited for determining whether a given species has the capacity for episodic memory. In contrast, paradigms that focus on isolating a specific form of contextual information (e.g., what-where, what-when) are promising for investigating the types of contextual information fundamental to episodic memories, as well as elucidating its critical neurobiological substrate (see below). Although no single definition or approach is likely to capture all features of episodic memory, converging evidence from these operational approaches has greatly furthered our understanding of episodic memory across phylogeny.
BRAIN STRUCTURES IMPORTANT FOR EPISODIC MEMORY
Studies of neurological patients and functional neuroimaging in humans have shown that episodic memory critically depends on the integrity of the hippocampus (Vargha-Khadem et al., 1997; Tulving and Markowitsch, 1998; Eichenbaum and Fortin, 2005) but also involves a large network of cortical areas that includes the adjacent parahippocampal region and the prefrontal cortex (Cabeza and St. Jacques, 2007; Schacter
et al., 2007). In this section, we review basic anatomical and functional evidence to determine the extent to which these structures are conserved in mammals and birds.
Hippocampus
The hippocampus has been identified in many species, including a large breadth of mammals (Insausti, 1993; Manns and Eichenbaum, 2006), birds (Székely, 1999; Atoji and Wild, 2006), reptiles [medial cortex (Rodríguez et al., 2002)], and teleost fish [dorsolateral telencephalon (Rodríguez et al., 2002; Broglio et al., 2005)]. The neurobiological and functional evidence strongly suggests that the hippocampus is a homologous structure across species.
In mammals, the hippocampus is remarkably conserved across species, including humans, nonhuman primates, pigs, rodents, and bats (Insausti, 1993; Manns and Eichenbaum, 2006). The cytoarchitecture can be easily identified by the dense layers of folded cell bodies that make up hippocampal subregions, including the subiculum, dentate gyrus, and cornu ammonis (CA) fields (Amaral and Witter, 1989; Manns and Eichenbaum, 2006; van Strien et al., 2009) (Fig. 6.1). Major inputs to the hippocampus originate from the entorhinal cortex and synapse on all subfields. Within the hippocampus, the dentate gyrus projects to CA3 through mossy fiber connections. CA3 projects to itself, through recurrent connections, as well as to CA1, through the Schaffer collaterals. The major outputs of the hippocampus originate from CA1 and the subiculum, and terminate in the entorhinal cortex [for a comprehensive account of the hippocampal circuitry, see van Strien et al. (2009)]. Additionally, a major anatomical characteristic of the mammalian hippocampus is a connection with the septum, which is conserved across all mammals. The function of the hippocampus is also well conserved across mammalian species. In fact, the hippocampus is critical for spatial memory in rats [reviewed in O’Keefe and Nadel (1978)], nonhuman primates (Banta Lavenex and Lavenex, 2009), and humans (Burgess et al., 2002). Moreover, neurophysiological studies have identified hippocampal neurons that encode specific places in an environment (place cells) in rodents (O’Keefe and Dostrovsky, 1971; Wilson and McNaughton, 1993; Knierim et al., 2006), nonhuman primates (Nishijo et al., 1997; Matsumura et al., 1999), and humans (Ekstrom et al., 2003), as well as in bats (Yartsev et al., 2011).
Birds also have a hippocampus, which arises from the same developmental origin as in mammals (Székely, 1999; Atoji and Wild, 2006; Rattenborg and Martinez-Gonzalez, 2011). As in mammals, a hippocampal–septal pathway is a major feature of the avian hippocampus (Atoji and Wild, 2004; Rattenborg and Martinez-Gonzalez, 2011). The avian hippo-
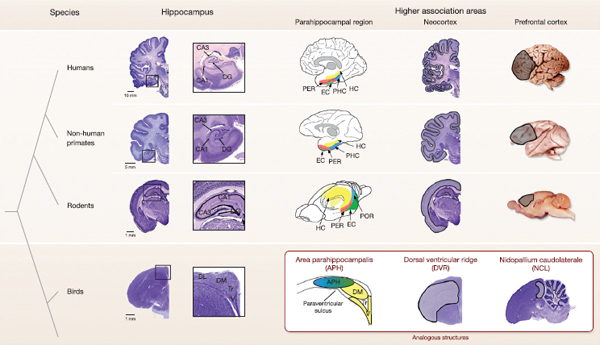
FIGURE 6.1 Brain regions important for episodic memory. Anatomical comparison of the hippocampus (avian hippocampus), parahippocampal region (avian area parahippocampalis), associational neocortex (avian dorsal ventricular ridge), and prefrontal cortex (avian nidopallium caudolaterale). The mammalian hippocampus shows distinct subregions, which are less evident in the avian hippocampus. The mammalian parahippocampal region is shown in diagrams [adapted with permission from Furtak et al. (2007), copyright Wiley-Liss, Inc.] to highlight the conserved relative spatial locations among species, with similar adjacent locations of area parahippocampalis and hippocampus in birds. Neocortical areas in mammals and associational areas of the dorsal ventricular ridge are outlined. The prefrontal cortex is shown in whole brains in mammals (medial surface in rat) and in a sagittal section in the bird. Human, nonhuman primate (Macaca mulatta), and rodent (Rattus norvegicus) sections were adapted with permission from http://www.brains.rad.msu.edu, and www.brainmuseum.org supported by the U.S. National Science Foundation, and bird (Taeniopygia guttata) sections from http://zebrafinch.brainarchitecture.org. DG, dentate gyrus; DL, dorsolateral region; DM, dorsomedial region; EC, entorhinal cortex, HC, hippocampus; PER, perirhinal cortex; PHC, parahippocampal cortex; POR, postrhinal cortex; Tr, triangular region; V, V-shaped layer.
campal subregions are not as visually obvious (Fig. 6.1) but nonetheless show homologies to those in mammals. Based on anatomical connectivity, Atoji and Wild (2006) noted that the dorsomedial area of the hippocampus is similar to the mammalian subiculum and CA regions, whereas the V-shaped layer in the ventromedial portion is similar to the mammalian dentate gyrus. However, a consensus on the exact homologies of hippocampal subregions is lacking (Székely, 1999; Atoji and Wild, 2006;
Rattenborg and Martinez-Gonzalez, 2011). Functionally, the avian hippocampus is similar to the mammalian hippocampus. Neurons in the avian hippocampus also show distinct place fields [reviewed in Bingman and Sharp (2006)], and lesions to the avian hippocampus specifically disrupt spatial memories (Hampton and Shettleworth, 1996; Colombo et al., 1997; Gagliardo et al., 1999). Notably, hippocampal lesions similarly impair spatial memories in turtles and goldfish (Rodríguez et al., 2002), further evidence that these functional similarities result from a long neurobiological ancestry.
Parahippocampal Region
In mammals, the hallmark of cortical–hippocampal connectivity is the existence of associative cortical structures that serve as an interface between the hippocampus and the rest of the neocortex. These associative regions include the entorhinal cortex, perirhinal cortex, and parahippocampal cortex [postrhinal cortex in rodents (Furtak et al., 2007)], which are collectively referred to as the parahippocampal region (Fig. 6.1). There are two main information processing pathways within the parahippocampal region (Fig. 6.2A). The “what” pathway, composed of the perirhinal and lateral entorhinal cortex, is important for processing and representing features of specific objects or items. In rodents and primates, this system receives information from all sensory modalities (Lavenex and Amaral, 2000; Suzuki and Amaral, 2004; Furtak et al., 2007), is critical for object memory (Brown and Aggleton, 2001; Squire et al., 2004; Feinberg et al., 2012), and contains neurons that respond to specific objects (Fried et al., 2002; Allen et al., 2007; Naya and Suzuki, 2011; Deshmukh et al., 2012). The second pathway processes “where” information and is composed of the parahippocampal/postrhinal cortex and medial entorhinal cortex. This system primarily receives visuospatial information (Suzuki and Amaral, 2004; Furtak et al., 2007). Consistent with a role in processing “where” information, neurons in a subregion of the medial entorhinal cortex fire in a triangular grid pattern as animals explore an environment [grid cells (Fyhn et al., 2004)]. Evidence for grid cells has been reported in rodents (Fyhn et al., 2004), nonhuman primates (Killian et al., 2012), and humans (Doeller et al., 2010), as well as in bats (Yartsev et al., 2011). Although species differences exist in the information processed by these pathways, the distinct informational segregation is conserved across rats, nonhuman primates, and humans (Burwell, 2000; Suzuki and Amaral, 2004; Eichenbaum et al., 2007).
In birds, the primary inputs and outputs of the hippocampus originate in the area parahippocampalis (Atoji and Wild, 2006) (Figs. 6.1 and 6.2B). Afferents to area parahippocampalis arise from several locations, includ-
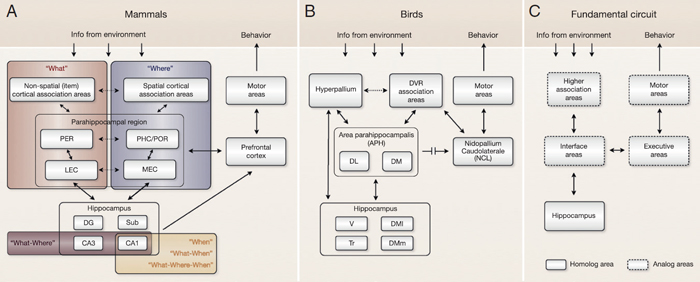
FIGURE 6.2 Neural circuits underlying episodic memory capacity in mammalian and avian species. (A) Schematic diagram of neural mechanisms supporting episodic memory encoding and expression in mammals. After information from the environment reaches the neocortex, the processing of “what” and “where” information is divided in parallel streams of cortical association areas. This functional segregation is maintained in the parahippocampal region, where the information is further processed before it reaches the hippocampus. Episodic memories are formed when the hippocampus integrates information about a specific event (what happened) with the context in which it occurred (e.g., where and/or when it happened). Although what-where coding has been shown in regions CA3 and CA1, lesion studies suggest that this type of integration depends specifically on region CA3. Recent evidence suggests that region CA1 provides an internal representation of elapsed time (when), which could support the formation of what-when and what-where-when associations. Episodic recall is thought to occur when the integrated event-in-context representation is reactivated in the hippocampal network, which leads to the reactivation of the associated representations in parahippocampal and neocortical association areas. The process by which the retrieved memories can guide behavior depends on the prefrontal cortex. (B) Comparable circuit in the avian brain. Note that connectivity between the area parahippocampalis and the nidopallium caudolaterale is indirect, as depicted with a broken arrow. (C) Fundamental circuit hypothesized to support episodic memory across species. Anatomical, behavioral, and physiological evidence demonstrates that this system involves homologous and analogous structures. DG, dentate gyrus; DL, dorsolateral region; DM, dorsomedial region (lateral and medial); DVR, dorsal ventricular ridge; LEC, lateral entorhinal cortex; MEC, medial entorhinal cortex; PER, perirhinal cortex; PHC, parahippocampal cortex; POR, postrhinal cortex; Sub, subiculum; Tr, triangular region; V, V-shaped layer.
ing the dorsal ventricular ridge and hyperpallium. Its efferents project back to the same structures and to the V-shaped layer and triangular region of the avian hippocampus. Therefore, the avian hippocampus has access to information from all modalities through the area parahip-
pocampalis (Atoji and Wild, 2006), much like the mammalian system. However, it is unknown whether the dorsolateral and dorsomedial subregions of area parahippocampalis are involved in segregated informational streams. As in the medial entorhinal cortex in mammals, grid-like cells have been observed near the avian hippocampus, although their exact location remains unclear (Bingman and Sharp, 2006).
To summarize, the extent to which the mammalian parahippocampal region and the avian area parahippocampalis are homologous remains to be determined. However, it is clear that there are similarities in the circuit organization and functions of these regions across mammals and birds, and especially within mammals.
Prefrontal Cortex
The size of the prefrontal cortex varies greatly across mammals, especially between primates and rodents (Fig. 6.1), but there is strong evidence of anatomical and functional correspondence across species (Kesner, 1998; Brown and Bowman, 2002; Uylings et al., 2003). The prefrontal cortex receives information from most cortical association areas and strongly projects to cortical and subcortical motor regions, suggesting that it plays a key role in the representation and execution of actions (Goldman-Rakic, 1996; Fuster, 2001) (Fig. 6.2A). The prefrontal cortex is also connected to the hippocampus by a direct pathway from CA1 (Verwer et al., 1997) and indirect connections through the parahippocampal region (Lavenex and Amaral, 2000; Furtak et al., 2007). Importantly, individual prefrontal neurons exhibit delay-related activity in nonhuman primates [reviewed in Goldman-Rakic (1996) and Fuster (2001)] and rodents (Jung et al., 1998), activity that may contribute to working memory, inferential reasoning, and decision-making abilities. These findings are consistent with the view that the prefrontal cortex is the primary executive region of the brain, a structure particularly important for bridging perception, memory, and action (Goldman-Rakic, 1996; Fuster, 2001).
Birds also have an executive region thought to be similar to the mammalian prefrontal cortex, called the nidopallium caudolaterale (Güntürkün, 2005; Herold et al., 2011). Importantly, the nidopallium caudolaterale directly projects to motor regions and has indirect access to the hippocampus and the area parahippocampalis [Güntürkün (2005); see also Allen and Fortin (2013)] (Fig. 6.2B). Delay-related neuronal activity has also been observed in individual nidopallium caudolaterale neurons (Rose and Colombo, 2005). However, it is important to note that, despite these similarities to the mammalian prefrontal cortex, the nidopallium caudolaterale is not homologous to its mammalian counterpart [i.e., the similarities are due to convergent evolution (Rose and Colombo, 2005)].
Section Summary
In sum, the hippocampus, parahippocampal region, and prefrontal cortex form a neural system that is thought to underlie episodic memory capacities in humans, but this basic neurobiology is not unique to humans. Considerable evidence shows that this circuit is present across mammals and that a comparable circuit exists in the avian brain. Interestingly, regions that are homologous to the hippocampus also exist in reptiles and bony (teleost) fish. Considering the long evolutionary history and structure–function similarities, it seems reasonable to hypothesize that the human episodic memory circuit shares an ancestral protoepisodic memory system with other mammals and possibly birds.
NEURAL MECHANISMS UNDERLYING EPISODIC MEMORY
Episodic memory in mammals depends on the hippocampus, the parahippocampal region, and the prefrontal cortex. However, until recently, it was unclear how this network of structures could give rise to episodic memory. In fact, considerable progress has been made in recent years toward understanding the specific contribution of each structure, as well as the nature of their functional relationships. Here, we describe a model, derived primarily from rodent and primate studies, summarizing the neural mechanisms thought to support the encoding and expression of episodic memories in mammals (Fig. 6.2A).
Processing Information About Events and Elements of Context
After being processed by sensory receptors and thalamic nuclei, information from the external world reaches primary sensory areas of the neocortex. A hierarchy of association cortical areas then processes this information at increasing levels of complexity and abstraction, culminating in multimodal representations. This information is funneled into the parahippocampal region, which mediates communications between the neocortex and the hippocampus (McClelland and Goddard, 1996).
The processing of “what” (e.g., stimuli, items) and “where” information is generally segregated into parallel streams. This functional segregation is maintained in the parahippocampal region (Burwell, 2000; Lavenex and Amaral, 2000; Eichenbaum et al., 2007): the perirhinal and lateral entorhinal areas play a critical role in item memory (what) whereas the postrhinal and medial entorhinal areas are important for the memory of contextual information (where). In contrast, the neural basis of the memory for “when” is much less understood. Although the hippocampus may play a critical role in processing “when” information under specific conditions (Meck et al., 1984; Jacobs et al., 2013), this capacity is gener-
ally thought to depend on other cortical and subcortical structures [e.g., striatum (Buhusi and Meck, 2005)].
Integrating Event and Context Information
Before reaching the hippocampus, information about the “what,” “where,” or “when” of individual events is not yet integrated into a single representation and thus does not satisfy the structure criterion for episodic memory. Episodic memory requires the integration of the representation of a single event with its distinctive contextual information, and it is this process that critically depends on the hippocampus [for potential mechanisms, see Buzsáki and Moser (2013)].
What-Where Integration
Studies in rodents (Gilbert and Kesner, 2002, 2003; Day et al., 2003; Rajji et al., 2006) and primates (Gaffan, 1994) show that the hippocampus plays a critical role in forming specific item–place associations. It is important to note that the spatial layout is already well learned in these paradigms, so deficits after hippocampus lesions cannot be solely attributed to an impairment in processing “where” information. Similarly, the deficits cannot be attributed to a deficiency in processing “what” information, as this capacity is normal in animals with hippocampal damage (Fortin et al., 2002; Gilbert and Kesner, 2002; Feinberg et al., 2012). The integration of what-where information can also be demonstrated in the coding properties of individual hippocampal neurons. A study by Wood et al. (1999) showed that different subsets of neurons selectively coded for “what” (e.g., a specific odor) and “where” (e.g., a specific location) information, whereas others coded for specific what-where conjunctions (a specific odor in a particular place). More recent studies have shown that the emergence of what-where coding parallels the learning of item-place associations (Komorowski et al., 2009; Kim et al., 2011). Although lesion studies suggest what-where integration depends on subregion CA3 but not CA1 (Gilbert and Kesner, 2003), what-where neural coding has been reported in both subregions with no significant differences reported (Wood et al., 1999; Komorowski et al., 2009).
What-When Integration
Accumulating evidence suggests that the hippocampus also plays a critical role in forming what-when associations, including memory for the order in which specific events occurred. For instance, in sequence memory paradigms, rats with hippocampal damage were shown to have normal
memory for the individual items presented (what) but consistently failed to remember the temporal relationships among events [what-when (Fortin et al., 2002; Kesner et al., 2002)]. Functional neuroimaging studies have shown that the hippocampus is strongly engaged during performance of similar tasks in humans as well (Kumaran and Maguire, 2006; Lehn et al., 2009; Ross et al., 2009). Furthermore, recent electrophysiological evidence suggests that a fundamental role of the hippocampus is to provide an internal representation of elapsed time, which could support the formation of what-when memories (MacDonald et al., 2011; Naya and Suzuki, 2011; Shapiro, 2011). In fact, recent studies have shown that individual hippocampal neurons exhibit robust timing signals during stimulus-free intervals [“time cells” (Pastalkova et al., 2008; MacDonald et al., 2011)] and during the presentation of sequences of events (Naya and Suzuki, 2011). In addition, the pattern of activity in hippocampal ensembles has been shown to gradually change over time, a form of population coding that could serve as a timing signal (Manns et al., 2007; Mankin et al., 2012). The above lesion and electrophysiological studies provide converging evidence that this capacity primarily depends on subregion CA1 of the hippocampus.
Episodic Recall and Response Selection
Episodic recall is thought to occur when the integrated event-in-context representation is reactivated, involving a pattern completion process that can be initiated by cueing the hippocampal network with elements of the event or context. This hippocampal reactivation leads to the reactivation of the corresponding representations in the parahippocampal region and other cortical association areas (McClelland and Goddard, 1996; Eichenbaum et al., 2007). The process by which the retrieved information can guide behavior is thought to critically depend on the prefrontal cortex (Goldman-Rakic, 1996; Fuster, 2001; Ninokura et al., 2003; Eichenbaum and Fortin, 2009). First, the episode-specific patterns of activity retrieved in the hippocampus are thought to reach the prefrontal cortex, either directly or through the parahippocampal region. The prefrontal cortex then evaluates the retrieved information and plans the appropriate course of action, which is then conveyed to motor regions (Goldman-Rakic, 1996; Fuster, 2001; Ninokura et al., 2003; Eichenbaum and Fortin, 2009).
Section Summary
Significant progress has been made in our understanding of the neural circuits underlying episodic memory capacity in mammals. In its essence, the circuit requires higher association areas to process the sensory
information (neocortex), interface areas to communicate with the hippocampus (parahippocampal region), the hippocampus to integrate and retrieve information about the episode, and executive areas to produce the appropriate behavior (prefrontal cortex). Although little is known about the neural mechanisms underlying episodic memory in birds, it is important to note that they have a similar circuit that could perform the same fundamental operations. The corresponding system in birds involves a combination of homologous (the hippocampus and, to some degree, the area parahippocampalis) and analogous (dorsal ventricular ridge, nidopallium caudolaterale) structures (Fig. 6.2B). Therefore, we hypothesize that a fundamental circuit may be shared between species that demonstrate episodic memory abilities (Fig. 6.2C).
FUNCTIONS OF EPISODIC MEMORY ACROSS SPECIES
As we examine the evolution of episodic memory, it is important to consider its potential functions across species. What are its potential contributions to the fitness of an individual? What advantage could it provide? Episodic memory is not necessary for animals to find food, shelter, mates, or to avoid dangerous situations. However, given the dynamic nature of the environment, the ability to remember unique experiences could certainly help animals be more successful. This advantage may be especially beneficial under conditions of limited resources, when incremental gains in the likelihood of success can amount to large effects on long-term survival. As mentioned earlier, our central argument is that fundamental properties of episodic memory, as well as their underlying neural circuits, are shared across mammals and birds. Therefore, some basic functions of episodic memory should be common across species.
Memory-Based Predictions
The purpose of memory is not to reminisce about the past, but to allow us to think, reason, and plan for the future (McGaugh, Chapter 9, this volume). Along these lines, we propose that the main function of episodic memory is to provide memory-based predictions to support adaptive behavior in the present or immediate future (Eichenbaum and Fortin, 2009). There are two ways in which episodic memory could contribute to this capacity. First, episodic memory is the only memory system to provide spatially and temporally specific information about single experiences. For instance, when faced with a specific need (e.g., a tool), an individual could use episodic memory to make predictions as to how to satisfy this need (e.g., look where the tool was last seen). This unparalleled specificity allows animals to take into account unique events in
guiding their behavior and to quickly adapt to changing circumstances. Second, episodic memory could contribute to memory-based predictions by supporting the capacity to make novel inferences. In fact, it has been proposed that a fundamental role of the hippocampus is to integrate episodic and semantic memories into a relational (declarative) memory network (Eichenbaum et al., 1999; Eichenbaum and Fortin, 2009). Because many of our memories overlap in information content, the network is thought to represent relationships among memories by linking them using their common elements. This network structure could support the flexible expression of inferred relationships between elements that were never experienced together, such as deducing a novel trajectory between two locations or the social hierarchy among a group of individuals. It should be noted that nondeclarative memory abilities also extract regularities from the environment to support the ability to generalize to other situations, but the process requires multiple exposures and lacks flexibility of expression (i.e., is tied to specific cues).
Planning for the Distant Future
Future planning involves making predictions about the distant future (many hours ahead) to anticipate future needs, an extension of the capacity for memory-based predictions described above. In humans, future planning involves “episodic future thought,” the ability to simulate plausible future events or scenarios [e.g., imagining future activities to determine what to pack for an upcoming trip (Addis et al., 2007; Hassabis et al., 2007; Szpunar et al., 2007)]. Interestingly, there is considerable overlap between the neural circuits involved in retrieving episodic memories and those involved in simulating future events, suggesting that the two capacities are intrinsically linked (Addis et al., 2007; Hassabis et al., 2007; Szpunar et al., 2007). Does this capacity for future planning extend beyond humans? Any attempt to examine future planning in animals must address the Bischof-Köhler hypothesis, which states that only humans can dissociate themselves from their current motivational state and take action for future needs (Suddendorf and Corballis, 2007). The criteria for demonstrating future planning in animals are as follows: (i) the behavior involved should be a novel action or combination of actions, (ii) the action should be appropriate for the future motivational state, and (iii) the anticipatory action should not have been extensively reinforced (Clayton et al., 2003a; Raby et al., 2007; Shettleworth, 2007; Suddendorf and Corballis, 2007). The first study satisfying all criteria has been conducted in scrub jays. In this study, the birds demonstrated the ability to make provisions for a future need, thus showing that they could dissociate themselves from their current motivational state and spontaneously plan for the next
day (Raby et al., 2007). Accumulating evidence suggests that apes are also capable of future planning, as they can save tools for future use (Mulcahy and Call, 2006) and can override immediate drives in favor of future needs (Naqshbandi and Roberts, 2006; Osvath and Osvath, 2008). Although it is clear that the behavior of other animals can be future oriented or based on future consequences [e.g., selecting an item to receive a reward (Roberts, 2002)], it remains to be determined whether animals other than humans, apes, and scrub jays are capable of future planning.
Building Social Relationships and Networks
Episodic memory could be particularly useful for processing and using social information. Although some aspects of social information are static (e.g., who is related to whom?), others can change over time (e.g., who has been cooperative? who has been aggressive?) and thus could depend on the capacity to remember specific experiences. Interestingly, the species in which episodic memory capacity has been most convincingly demonstrated (primates, rodents, and scrub jays) are highly social (Emery and Clayton, 2004; Brennan and Kendrick, 2006). Recent evidence suggests that humans with episodic memory impairments have social circles that are limited compared with controls, suggesting that episodic memory may be crucial for establishing and/or maintaining social bonds (Davidson et al., 2012). Thus, there may be a relationship between episodic memory capacity and social interactions.
Species-Specific Uses of Episodic Memory
Although we have so far emphasized the commonalities in episodic memory capacity, there are also clear differences across species. These divergences include unique uses of episodic memory, as well as species-specific attributes. For instance, in humans, episodic memory is thought to be intrinsically tied to other mental capacities such as language, a sense of self, empathy, and theory of mind (Tulving, 2002; Schacter et al., 2007; Duff and Brown-Schmidt, 2012). Although such characteristics were initially used as evidence that episodic memory is unique to humans, according to the present conceptual framework, they represent species-specific attributes (or modules) associated with the expansion of neocortical (particularly prefrontal) areas in humans. Other species-specific uses of episodic memory may include meadow voles predicting when and where sexually receptive females will be located (Ferkin et al., 2008) and hummingbirds keeping track of the location, quality, and renewal rate of different sources of nectar (González-Gómez et al., 2011). Episodic memory may be of particular importance in hummingbirds because of the enormous energy cost
in gathering nectar, which makes repeat visits or poor planning highly detrimental (González-Gómez et al., 2011).
Section Summary
Given that several species demonstrate episodic memory capacity, it is reasonable to assume that it offers significant advantages. Some of these benefits could be common across species; others may be species-specific. However, further research is needed before we can understand the specific nature of these advantages or establish that they are causally linked with an increase in fitness.
CONCLUSIONS
Episodic memory is the remarkable capacity to remember specific personal experiences. Although it was originally thought that this capacity was particular to humans, the ample evidence reviewed here indicates that core properties of episodic memory are present across mammals, as well as in birds. This cross-species approach to episodic memory research is made possible by the use of operational definitions that can be applied across species, a method we strongly suggest should be used in animal and human studies. The most common approach to investigate episodic memory capacity across species is to determine whether animals can remember events within the context in which they occurred (e.g., memory for what-where-when, what-where, or what-when). Using this conceptual framework, we showed that episodic memory in mammals depends on a functional relationship between the hippocampus, parahippocampal region, neocortical association areas, and prefrontal cortex. Importantly, we described a comparable neural circuit in birds, which includes homologous (hippocampus, and to some degree, parahippocampal region) and analogous (dorsal ventricular ridge association areas, nidopallium caudolaterale “prefrontal” area) structures. Finally, we submit that this fundamental circuit underlies episodic memory capacity across species but that species-specific divergences have also evolved around this central architecture.
When did episodic memory emerge? Unfortunately, the available evidence cannot support a definitive answer at this time. We speculate that it evolved at a stage when the hippocampus was already present because the hippocampus is known to be a critical substrate. However, we are not implying a one-to-one relationship between the hippocampus and episodic memory (e.g., if the hippocampus is present, the animal has episodic memory capacity). Because the hippocampus is essential for spatial memory across species, ranging from humans to teleost fish, it is
likely that its role was limited to the processing of “where” information when it first emerged (Fig. 6.3). We propose that episodic memory capacity emerged at a later time, when the hippocampus began supporting the integration of information about events in context (e.g., “what,” “where,” and/or “when” information). As the neural architecture of the hippocampus indicates, the content of its associations is determined by its inputs. Thus, the change to supporting episodic memory likely occurred when the hippocampus began receiving highly processed event and contextual information from higher association areas. In light of the cross-species behavioral and neurobiological similarities reviewed here, it is tempting to conclude that episodic memory capacity emerged before mammals and reptiles diverged (possibility 1 in Fig. 6.3). However, because of the limited data available in nonavian reptiles, the hypothesis that it resulted from convergent evolution (e.g., possibilities 2 and 3 in Fig. 6.3) cannot be rejected at this time. Addressing this important issue will require converg-
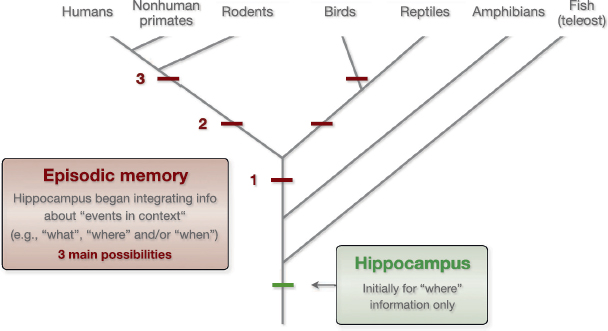
FIGURE 6.3 Possible time points for the emergence of episodic memory in evolution. Initially, the role of hippocampus was likely limited to the processing of spatial information (“where”). We hypothesize that episodic memory capacity emerged later on, when the hippocampus began supporting the integration of information about events in context (e.g., “what,” “where,” and/or “when” information). The striking behavioral and neurobiological similarities reviewed in this chapter suggest that episodic memory capacity emerged before mammals and reptiles diverged (possibility 1). However, additional evidence from birds and reptiles is needed before the alternative hypothesis that episodic memory is the result of convergent evolution (e.g., possibilities 2 and 3) can be safely rejected.




















