Prevention and Early Detection
Improving the prevention and early detection of ovarian carcinomas will be a critical component of reducing morbidity and mortality from ovarian cancer. This chapter discusses the genetic and nongenetic risk factors of the disease along with potential prevention strategies and methods for early detection and screening of ovarian cancer. In particular, this chapter identifies a number of gaps in knowledge related to identifying those women who are at highest risk for developing ovarian carcinomas, and it describes several challenges to developing screening tests for high-risk women, their families, and the general population. The chapter also explains how gaps in knowledge about the basic biology of ovarian carcinomas (as discussed in Chapter 2) hinder the development of better methods to prevent ovarian carcinomas or detect them at the earliest stage of disease progression.
RISK ASSESSMENT FOR OVARIAN CANCER
Although scientists’ understanding about the early carcinogenic events of ovarian cancer is incomplete (see Chapter 2), researchers have identified several factors associated with either an increased or a decreased risk of developing ovarian cancer (see Table 3-1). While some of these risks factors cannot be modified (e.g., age and ancestry), a number of others (e.g., hormone use and diet) can be altered through lifestyle changes, pharmacological interventions, or surgery. A critical drawback, however, is that nearly all of the identified risk factors are associated predominantly with the less common and less lethal ovarian cancer subtypes and not with the most common and lethal type—high-grade serous carcinoma (HGSC). Ovarian
TABLE 3-1
Risk Factors for Ovarian Cancer
| Increased Risk | Decreased Risk | |
| Age | Oral contraceptive (OC) use | |
| Family history of cancer | Oophorectomy | |
| Hereditary cancer syndromes | Hysterectomy | |
| Obesity | Tubal ligation | |
| Nulliparity | Lactation | |
| Hormone replacement therapy | Salpingectomy | |
| Increased numbers of lifetime ovulatory cycles | Bilateral salpingo-oophorectomy (BSO) | |
SOURCE: Adapted from Permuth-Wey et al., 2014.
cancer risk factors can also vary by histologic subtype, and thus a given risk factor may increase risk for one subtype while decreasing risk for another.
Age, Race, and Ethnicity
In general, cancer risk increases with age, in part because of the natural accumulation of genetic alterations and long-term exposure to environmental factors. As noted in Chapter 1, ovarian cancer incidence also increases with age, and the age-adjusted incidence rate for women ages 65 and older is more than five times the incidence rate for women younger than age 65 (Howlader et al., 2015). Race and ethnicity have variable associations with ovarian cancer (see Chapter 1 for more on the role of race and ethnicity in ovarian cancer demographics).
Inherited Genetic Risk
A family history of ovarian cancer has a strong association with risk for ovarian cancer, and having a large number of first-degree biological relatives with an ovarian carcinoma increases a woman’s risk (Jervis et al., 2014; Soegaard et al., 2009; Stratton et al., 1998; Werness and Eltabbakh, 2001). As a result, specific germline (inherited) genetic mutations are among the most well-established risk factors associated with ovarian cancer (Shulman and Dungan, 2010). Many women with a family history of cancer have a hereditary cancer syndrome (Garber and Offit, 2005). Table 3-2 describes some common genetic mutations and hereditary cancer syndromes that are found among women with ovarian cancer. Inherited genetic mutations are associated with approximately 5 to 15 percent of all ovarian carcino-
TABLE 3-2
Examples of High-Risk Ovarian Cancer Susceptibility Genes and Cancer Syndromes
| Mutated Gene(s) | Cancer Syndrome | |
| BRCA1, BRCA2 | Hereditary breast and ovarian cancer (HBOC) syndrome | |
| MLH1, MSH2, MSH6, PMS2, EPCAM | Lynch syndrome | |
| TP53 | Li–Fraumeni syndrome | |
| STK11/LKB1 | Peutz–Jeghers syndrome, sex cord and mucinous tumors | |
NOTE: Lynch syndrome is also known as hereditary nonpolyposis colorectal cancer.
SOURCES: Hampel et al., 2015; Hendriks et al., 2006; Kempers et al., 2011; Lu and Daniels, 2013; Shulman, 2010.
mas, though their distribution varies by subtype (Lynch et al., 2009). In some studies, inherited mutations have been found in up to 25 percent of women with HGSC, but up to half of these women did not have a family history of breast or ovarian cancer (Schrader et al., 2012; Walsh et al., 2011).
High- and Moderate-Risk Gene Mutations
BRCA1 and BRCA2 are the most recognizable of the genes that increase the risk of ovarian cancer. These genes were identified in the mid-1990s as breast and ovarian cancer–risk genes, and in recent years the effect that the BRCA1 and BRCA2 mutations have on the risk of breast and ovarian cancers has been established (Miki et al., 1994; Rebbeck et al., 2015; Wooster et al., 1994). Germline mutations in these genes are present in approximately 10 to 15 percent of all women diagnosed with invasive ovarian carcinomas and in approximately 15 to 23 percent of women diagnosed with HGSC (Alsop et al., 2012; Pal et al., 2005; Risch et al., 2006; Zhang et al., 2011). Recently discovered single-nucleotide polymorphisms (SNPs) in other genes appear to have a role in modulating the risk of ovarian cancer in those women who have an inherited BRCA1 or BRCA2 mutation (Ramus et al., 2011, 2012).
Lynch syndrome is caused by deleterious mutations in DNA mismatch repair genes (see Table 3-2). The lifetime risk of ovarian cancer associated with Lynch syndrome mutations is around 8 percent, with the greatest risks associated with MLH1 or MSH2 (Lu and Daniels, 2013). Many of the genes in Table 3-2 are typically included in the multiplex gene panels currently offered by commercial laboratories, and National Comprehensive Cancer Network (NCCN) guidelines suggest various interventions when a woman has one or more of these genes. For example, NCCN recommends
risk-reducing surgeries (e.g., bilateral salpingo-oophorectomy) for women with mutations in BRCA1, BRCA2, and genes associated with Lynch syndrome (NCCN, 2015). Risk-reducing surgeries are discussed later in this chapter. Other moderate-risk cancer susceptibility genes often included in the panels are CHEK2, PALB2, RAD51 family, BRIP1, and BARD1 (Walsh et al., 2011). RAD51C and RAD51D mutations appear to confer a lifetime risk of 10 to 15 percent for ovarian cancer (Lancaster et al., 2015).
Advances in DNA-sequencing technologies have dramatically reduced the cost of genetic testing, and in 2013 the U.S. Supreme Court ruled that, with few exceptions, patents on naturally isolated DNA sequences are invalid.1 The confluence of these events led to the rapid development and release of genetic tests that sequence multiple genes simultaneously—the so-called multiplex gene panels—and to the possibility of performing whole-genome sequencing (Rahman, 2014; Robson et al., 2015). With multiplex gene panels it may be possible to identify more individuals with hereditary cancer gene mutations than with testing for BRCA1 and BRCA2 alone (Desmond et al., 2015). However, the ability to find more mutations does not necessarily lead to a better understanding of disease risk, as little is known about the clinical impacts of a large fraction of these variants or mutations. In these cases where the clinical implications are unknown, the mutations are referred to as “variants of unknown significance” (Domchek and Nathanson, 2014; Eccles et al., 2015).
Recently, the American Society of Clinical Oncology (ASCO) recognized that for women with a personal or family history of cancer, multigene testing may be an efficient way to evaluate multiple high-penetrance genes of established clinical utility (Robson et al., 2015). ASCO further stated that providers with expertise in cancer risk assessment need to be involved in ordering and interpreting multigene panels when genes of uncertain clinical utility are included and that patients must give informed consent and be provided with pretest counseling prior to any genetic testing. ASCO also commented on the specific components that should be included in multigene panel testing.
Technological and legislative advances have combined to make genetic testing for BRCA1 and BRCA2 mutations much more accessible. The Patient Protection and Affordable Care Act2 requires health plans beginning on or after September 23, 2010, to cover genetic counseling and testing for BRCA1 and BRCA2 mutations in women whose family history indicates a high risk for cancer. The law does not specifically cover testing using multiplex gene panels or genome-wide sequencing, and testing is not required to be covered for women whose family history does not indicate an increased
________________
1Association for Molecular Pathology v. Myriad Genetics, Inc., 569 U.S. ___ (2013).
2 Patient Protection and Affordable Care Act, Public Law 148, 111th Cong., 2nd sess. (March 23, 2010).
risk (Nelson et al., 2005, 2014). Two testing issues that the act leaves unaddressed are that BRCA1 and BRCA2 mutations alone do not fully account for genetic risk and that many women with inherited cancer-predisposing mutations do not have a strong family history of breast or ovarian cancer (Jervis et al., 2014; Schrader et al., 2012; Walsh et al., 2011).
Women who test positive for germline genetic mutations associated with greatly increased risk for developing ovarian cancer may benefit from enhanced screening, risk-reducing prophylactic surgery, or chemoprevention. For instance, women with a known BRCA1 or BRCA2 mutation who have prophylactic bilateral salpingo-oophorectomy (BSO) can reduce their risk of ovarian cancer by more than 90 percent (and, for premenopausal women, can reduce their risk for breast cancer by 50 percent) (Domchek et al., 2010).
Low-Penetrance Alleles
Genome-wide association studies have identified some common low-penetrance alleles (genes associated with a low risk) that can also contribute to the familial risk associated with ovarian cancer (Song et al., 2014). Studies using consortia have identified as many as 17 low-penetrance SNP alleles associated with ovarian cancer risk (Bojesen et al., 2013; Bolton et al., 2010; Goode et al., 2010; Kuchenbaecker et al., 2015; Permuth-Wey et al., 2013; Pharoah et al., 2013; Song et al., 2009). In general, most of the risk alleles have been more strongly associated with serous tumors, but these associations may reflect the fact that there are relatively many cases of such tumors, making it easier to find risk alleles associated with them. Using consortia increases the number of cases available for study, which may lead to more associations being uncovered, especially for the less common subtypes. For example, one study of mucinous carcinomas using consortia identified several risk alleles that had not been identified in previous studies that included multiple histologic subtypes (Kelemen et al., 2015).
Genetic Testing Guidelines and Recommendations
Cancer genetic consultation services can provide an evaluation of personal and family history for possible features of hereditary cancer syndromes, a consideration of diagnoses, genetic testing if indicated and available, recommendations for the prevention and management of cancer, and information for at-risk relatives (Hampel et al., 2015). The American College of Surgeons (ACS) Commission on Cancer3 has issued standards for
________________
3 For more information, see http://www.facs.org/cancer/coc/cocprogramstandards2012.pdf (accessed September 25, 2015).
the provision of cancer risk assessment, genetic counseling, and genetic testing services (ACS, 2012). Several other clinical societies and organizations have also developed guidelines and recommendations for when referrals should be made to cancer genetic consultation (see Table 3-3). With the exception of the U.S. Preventive Services Task Force (USPSTF), all of the organizations recommend that all women diagnosed with ovarian carcinoma receive referral for genetic counseling and testing regardless of their family history. Guidelines from the Society of Gynecologic Oncology (SGO), the National Society of Genetic Counselors (NSGC), the American College of Medical Genetics and Genomics (ACMG), and the NCCN all recommend referral to a cancer genetics professional for women without a personal history of cancer but with a family history strongly suggestive of hereditary breast and ovarian cancer (HBOC) (Lancaster et al., 2015; NCCN, 2015).
In spite of the benefits of germline genetic testing for women diagnosed with ovarian cancer, the rate of referral to genetic counseling and testing in accordance with existing guidelines still remains low (Febbraro et al., 2015; Powell et al., 2013a). For example, one study of women with ovarian cancer found that only 14.5 percent of women who should have been counseled according to NCCN guidelines actually were referred to genetic counseling (Febbraro et al., 2015). Further, only 59.5 percent of the women who were referred actually did follow up with the genetic counseling, although of those who did receive counseling, approximately 95 percent ultimately underwent genetic testing.
One objective of Healthy People 20204 is that a greater percentage of women with a significant family history of breast or ovarian cancer receive genetic counseling (HHS, 2013). From 2006 to 2010 the proportion of women receiving genetic counseling rose from 34.6 to 52.9 percent (HHS, 2013). While this was a large increase, still nearly half of all eligible women are not receiving the recommended counseling. There are various patient-, provider-, and system-level barriers that make cancer genetics referrals less likely, including patients being unaware of a family history of cancer, the limited time that providers have to collect family history, and referral criteria that are complex and that vary by group (Hampel et al., 2015).
Relying on a patient’s family history alone to consider whether genetic testing should take place will inevitably overlook some women with inherited mutations that put them at risk for ovarian cancer. As mentioned previously, many women with inherited mutations do not have a significant family history for cancer. Furthermore, family history may not indicate a high risk for women with few close female relatives, women with female relatives who underwent risk-reducing surgery at early ages, women who may be adopted and thus do not know their biological family’s cancer his-
________________
4 For more information, see http://www.healthypeople.gov (accessed September 18, 2015).
TABLE 3-3
Current Recommendations for Cancer Genetic Consultation and Germline Genetic Testinga for Individuals with Personal or Family Historyb of Ovarian Cancer, 2013–2015
| Group | Criteria for Ovarian Cancer Patient | Genes | Criteria for Relatives of the Ovarian Cancer Patient | References | |
| Diagnosis | Family History | ||||
| NCCN | Invasive ovarian cancer (including fallopian tube and primary peritoneal cancer) | None required | Multiple genes |
|
NCCN, 2015 |
| USPSTF | Criteria unclear | Criteria unclear | BRCA1, BRCA2 |
|
CMS, 2015; Nelson et al., 2014 |
| SGO | High-grade epithelial ovarian, tubal, or peritoneal cancer | None required | Multiple genes |
|
Lancaster et al., 2015 |
| ACMG and NSGC | Ovarian cancer (including fallopian tube and primary peritoneal cancer) | None required | Multiple genes |
|
Hampel et al., 2015 |
| ASCO | Epithelial ovarian, fallopian tube, or primary peritoneal cancer (most commonly HGSC) | None required | BRCA1, BRCA2 |
|
Lu et al., 2014 |
aAll of the above organizations and the ACS Commission on Cancer recommend that the process of germline genetic testing include pre- and post-test counseling with a physician, genetic counselor, or other health care provider with expertise in cancer genetics.
bFamily history includes first- or second-degree relatives, and third-degree relatives in some recommendations.
NOTE: ACMG = American College of Medical Genetics and Genomics; ASCO = American Society of Clinical Oncology; NCCN = National Comprehensive Cancer Network; NSGC = National Society of Genetic Counselors; SGO = Society of Gynecologic Oncology; USPSTF = U.S. Preventive Services Task Force.
tory, or women who otherwise do not know the family health history of one or both parents (Lancaster et al., 2015).
Cascade Testing
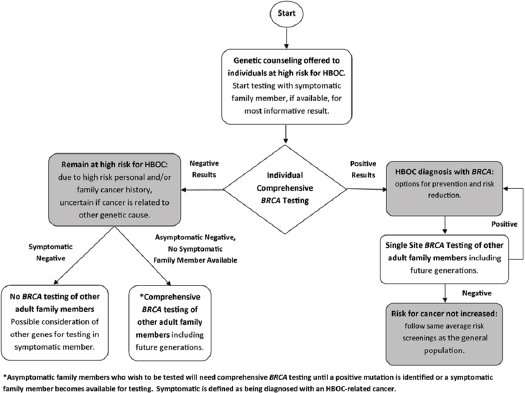
Cascade testing is a sequential process of identifying and testing close blood relatives of individuals who are at increased risk for genetic conditions; the process minimizes the number of tests and costs (George et al., 2015). If an individual has a known deleterious mutation, further genetic testing of close blood relatives requires testing only for the specific mutation rather than testing for the entire gene or doing multigene testing. Only single-site testing is needed for family members unless they are in a population with common and known founder mutations, such as Ashkenazi Jews. For relatives who are negative for the specific mutation, it is likely that their risk of cancer and their offspring’s risk of inheriting the mutation are no greater than the risk for the general population. For relatives who have the same mutation, the cascade testing continues with testing their close blood relatives, and so on. Asymptomatic women with a known mutation are often referred to as previvors (FORCE, 2008). Figure 3-1 illustrates how cascade genetic testing might be carried out for individuals at high risk for HBOC syndrome.
Several of the clinical societies and organizations in Table 3-3 recommend that biological relatives of an individual with a deleterious mutation for BRCA1, BRCA2, or the Lynch syndrome genes be referred for genetic consultation. The Centers for Disease Control and Prevention (CDC) also promotes cascade testing as an important public health intervention among family members at risk for HBOC syndrome and Lynch syndrome (Bowen et al., 2012).
Reproductive and Hormonal Risk Factors
A variety of reproductive and hormonal factors, including oral contraception, parity, infertility, endogenous hormones, and exogenous hormones, may increase or decrease a woman’s risk of developing an ovarian cancer. For some of these factors, there is not yet a robust evidence base for their association with ovarian cancer risk, particularly by tumor subtype.
Oral Contraception and Parity
Among the most well-established risk factors for ovarian cancer are the use of oral contraceptives (OCs) and parity. One large study found a 20 percent decrease in the risk of ovarian cancer for every 5 years of OC use; the association was similar for serous, endometrioid, and clear cell types,
although no association was observed for mucinous carcinomas (Beral et al., 2008). The protective effect of OCs waned with increasing time since last use and with later age at first use, suggesting that older women, who are at the highest risk, benefit less from prior OC use than younger women.
Parity is another clear protective factor for ovarian cancer. One analysis reported that the largest decrease in risk (approximately 30 to 40 percent) was associated with the first pregnancy, with about a 10 to 15 percent decrease in risk for each subsequent pregnancy (Whittemore et al., 1992). Age at first birth has not been consistently associated with risk, but later age at last birth may be weakly associated with decreased risk (Bevier et al., 2011). One meta-analysis suggests that breastfeeding among parous women reduces risk, with risk reduced by 8 percent for every 5 additional months of breastfeeding (Luan et al., 2013).
Most studies suggest that OCs and breastfeeding may reduce risk for all ovarian carcinoma types except mucinous carcinomas (Beral et al., 2008; Fortner et al., 2015; Gates et al., 2010; Merritt et al., 2013; Yang et al., 2012). Increasing parity only modestly reduces the risk for serous carcinomas or Type II tumors, but it significantly reduces the risk for endometrioid and clear cell carcinomas or Type I tumors (Setiawan et al., 2013).
Infertility
Most studies of infertility suggest that female-factor infertility is associated with a modestly increased risk for ovarian cancer (Tworoger et al., 2007). Many fertility drugs can increase the level of gonadotropins and ovulation, both of which are hypothesized to influence the risk for ovarian cancer. While the results have been somewhat inconsistent, several larger studies with long follow-up have found little or no association of clomiphene citrate (an ovulatory stimulant) or exogenous gonadotropins with ovarian cancer risk, although one study did find suggestive evidence of an increased risk for nulliparous women using clomiphene citrate (Jensen et al., 2009; Trabert et al., 2013).
Endometriosis, which is defined by the presence of endometrial tissue outside the uterine cavity, is a common cause of infertility and has been positively associated with ovarian cancer risk. One analysis of women with and without endometriosis found a two- to threefold increased risk for ovarian cancer, specifically for endometrioid and clear cell carcinomas, among women with endometriosis (Pearce et al., 2012). Other ovarian cancer subtypes, including HGSC and mucinous carcinoma, were not associated with endometriosis (though there was a suggestive association for low-grade serous carcinoma) (Guo, 2015; Pearce et al., 2012). These results support the hypothesis that endometriotic tissue may be a precursor lesion for certain ovarian cancer subtypes (see Chapter 2).
Polycystic ovarian syndrome (PCOS) has been hypothesized to be associated with ovarian cancer because of the increased levels of androgens that it produces and its association with infertility (Daniilidis and Dinas, 2009). However, several studies have found PCOS to have no associations with ovarian cancer risk, although there was a suggestive association among older women in one study (Barry et al., 2014; Gottschau et al., 2015; Shen et al., 2015).
Other Reproductive Factors
Other reproductive factors, such as age at menarche and age at menopause, have been weakly or inconsistently associated with ovarian cancer risk (Permuth-Wey and Sellers, 2009). However, the estimated number of ovulatory years (e.g., age at menopause minus age at menarche minus years of OC use and pregnancy) appears to be strongly positively associated with risk for all types of ovarian carcinoma (Fortner et al., 2015; Gates et al., 2010). Overall, these results suggest that ovulation and the associated hormonal and inflammatory process (e.g., ovarian surface epithelium repair after ovulation) are likely to be important in the etiology of ovarian carcinomas. The strong protective effect that pregnancy has for endometrioid and clear cell carcinomas suggests a potential role for progesterone exposure in these tumor types, although this has not been directly assessed.
Hormones
Steroid hormones are also thought to play a role in ovarian cancer risk (Lukanova and Kaaks, 2005). Androgens have been hypothesized to increase risk, for example, but most prospective studies of circulating androgens (e.g., testosterone, DHEA) have not observed an association, although one study did report an increased risk for Type I tumors—and a decreased risk for Type II tumors—with increasing androstenedione levels (Ose et al., 2015a; Risch, 1998; Tworoger et al., 2008b). One small study of endogenous hormones that included estrogen and progesterone found that neither of these hormones had a significant association with ovarian cancer risk, although a more recent study found that high estradiol levels during pregnancy were associated with an increased risk of endometrioid tumors (Helzlsouer et al., 1995; Schock et al., 2014b).
Experiments in cell and animal models indicate that the hormones insulin-like growth factor I (IGF-1) and placental growth hormone (GH) are expressed in the ovary and are involved in ovarian function or the progression of ovarian cancer (Beauchamp et al., 2010; Bruchim and Werner, 2013; Hull and Harvey, 2001). A study of IGF-1 and GH serum concentration from early pregnancy found that GH was not associated with risk for
ovarian carcinomas and that IGF-1 showed borderline significant associations, with a decreased risk for all ovarian carcinoma subtypes together as well as invasive and endometrioid tumors individually (with slightly stronger associations in women diagnosed under the age of 55) (Schock et al., 2015). Other hormones, such as prolactin and anti-Müllerian hormone, may be associated with ovarian cancer risk, but larger studies are needed (Clendenen et al., 2013; Schock et al., 2014a).
Further support for the role of hormones in ovarian cancer etiology includes the positive association observed between postmenopausal hormone replacements and increased ovarian cancer risk. In the largest prospective study to date, the use of any hormone therapy at any time was associated with a 20 percent increase in risk for ovarian cancer (Collaborative Group on Epidemiological Studies of Ovarian Cancer, 2015). The observed increase in risk was slightly stronger for the use of estrogen only (34 percent higher risk) than for the use of estrogen plus progestin (14 percent increased risk), and durations of hormone use of greater than 5 years were associated with the highest risk. Multiple studies have reported that hormone therapy is more strongly associated with risk for serous and endometrioid carcinomas than for other types, although one study found no difference in risk for Type I versus Type II cancers (Beral et al., 2015; Fortner et al., 2015; Gates et al., 2010; Yang et al., 2012).
Behavioral and Inflammatory Risk Factors
In general, lifestyle factors such as diet, physical activity, adiposity, and smoking have not been strongly associated with ovarian cancer risk. However, a number of recent smaller studies have found some evidence for the role of lifestyle factors in risk for ovarian cancer.
Diet
Studies of overall diet quality and dietary patterns have generally not shown an association with ovarian cancer risk; however, data from the low-fat dietary intervention in the Women’s Health Initiative5 indicated that there was a 40 percent lower risk for ovarian cancer in women who followed the low-fat dietary intervention for more than 4 years (Chandran et al., 2011; Chang et al., 2007; Edefonti et al., 2008, 2009; Kolahdooz et al., 2009; Prentice et al., 2007; Romaguera et al., 2012; Thomson et al., 2014; Xie et al., 2014). Several studies have reported that both total fat intake and polyunsaturated fat intake have modest positive associations with
________________
5 For more information on the Women’s Health Initiative, see https://www.nhlbi.nih.gov/whi (accessed September 2, 2015).
ovarian cancer risk, although a pooled analysis of 12 prospective studies did not observe an association (Blank et al., 2012; Genkinger et al., 2006b; Merritt et al., 2014b). Studies on the risk associated with meat consumption have been inconsistent (Kolahdooz et al., 2010; Wallin et al., 2011).
Several studies have found no risk associated with overall lactose intake, but a single study did suggest that the intake of lactose and dairy foods is inversely associated with risk for endometrioid carcinomas (Cramer, 1989; Genkinger et al., 2006a; Liu et al., 2015; Merritt et al., 2014a). Increased consumption of tea and its components (e.g., flavonoids and caffeine) has been suggested to lower risk (Cassidy et al., 2014; Gates et al., 2009; Gosvig et al., 2015; Lueth et al., 2008; Rossi et al., 2008, 2010; Song et al., 2008; Tworoger et al., 2008a; Wang et al., 2009; Zhang et al., 2014). Studies of other dietary factors, including alcohol, fruits, vegetables, and vitamin intake, have not demonstrated any associations with ovarian cancer risk (Genkinger et al., 2006c; Koushik et al., 2005, 2006).
Adiposity and Body Size
Adiposity and body size are important lifestyle factors associated with a number of cancers (Renehan et al., 2015). While height is consistently associated with an increased risk of ovarian cancer, associations of ovarian cancer risk with various measures of adiposity have been inconsistent, possibly because of differences due to menopausal status (Aune et al., 2015). In one large study, only premenopausal ovarian cancer was positively associated with body mass index (BMI) (Schouten et al., 2008). One meta-analysis reported that higher adult BMIs were modestly associated with an increased ovarian cancer risk and that the risk was greater for higher BMIs in early adulthood (Aune et al., 2015). The risks associated with other measures of adiposity (e.g., waist and hip circumference) are not as well studied.
Physical Activity
Physical activity and sedentary behavior have been shown to be associated with risk of breast, colon, and other cancers (Phillips et al., 2015). However, physical activity has not been clearly or consistently associated with ovarian cancer risk; it is possible that the association is different for premenopausal versus postmenopausal activity (Huang et al., 2015a; Zhong et al., 2014). Several studies have reported conflicting results as to whether measures of a sedentary lifestyle (e.g., total sitting or the amount of time spent watching television) are associated with an increased risk of ovarian cancer (Hildebrand et al., 2015; Patel et al., 2006; Xiao et al., 2013; Zhang et al., 2004).
Smoking
The association of smoking with risk for ovarian cancer varies by subtype. A recent meta-analysis found a 7 percent increased risk of ovarian cancer for current smokers versus women who had never smoked, but the association varied significantly by histologic subtype (Beral et al., 2012). Smoking was associated with an approximately 20 percent lower risk for endometrioid and clear cell carcinomas and an approximately 80 percent increased risk for mucinous carcinomas among current smokers versus never-smokers; serous carcinomas were not associated with smoking.
Inflammation
Studies of the inflammatory marker C-reactive protein suggest a possible association between inflammation and an increased risk of ovarian cancer (Ose et al., 2015b; Poole et al., 2013). Other specific inflammatory factors have also been associated with ovarian cancer. A meta-analysis reported that exposure to asbestos was associated with a 77 percent increased risk of ovarian cancer mortality (Camargo et al., 2011), and the International Agency for Research on Cancer determined that there was sufficient evidence to support a causal relationship between asbestos exposure and ovarian cancer (Straif et al., 2009). This has led to studies of talc use, which is chemically similar to asbestos and can cause an inflammatory response. The use of perineal talcum powder has been associated with a 20 to 30 percent increased risk of ovarian cancer, although it also has been show to vary by histologic subtype (Cramer et al., 2015; Terry et al., 2013). One analysis reported a 9 percent lower ovarian cancer risk with regular aspirin use, with stronger results among daily users (Trabert et al., 2014). However, most cohort studies have not observed a similar reduction in risk (Brasky et al., 2014; Lacey et al., 2004; Murphy et al., 2012; Ni et al., 2013; Pinheiro et al., 2009; Prizment et al., 2010; Setiawan et al., 2012).
As mentioned previously, endometriosis is associated with an increased risk of ovarian cancer, and tubal ligation and hysterectomy (which may limit the ability of endometrial tissues to access the fallopian tubes, ovaries, and pelvic region by retrograde menstruation) act to decrease this risk. Hysterectomy is associated with an approximately 30 percent decreased risk of ovarian cancer, and tubal ligation has been associated with a 26 percent decreased risk of ovarian cancer overall and a 55 percent lower risk of endometrioid cancer, the ovarian cancer type most strongly associated with endometriosis (Rice et al., 2012). The exact causes for the increased risk of ovarian cancer from endometriosis are unknown. However, endometriosis is associated with an inflammatory environment characterized by elevated levels of cytokines and growth factors (Arici, 2002; Malutan
et al., 2015a,b). This pro-inflammatory environment may produce an environment favorable for tumor development and growth (Hanahan and Weinberg, 2011). As previously discussed, endometriosis is associated with infertility and nulliparity, which may further increase the risk of ovarian cancer.
Other conditions that increase local peritoneal inflammation may also be associated with ovarian cancer risk. Studies of the association of chronic pelvic inflammatory disease (PID), which develops from sexually transmitted infections, and ovarian cancer risk have had mixed results (Lin et al., 2011; Ness et al., 2000; Parazzini et al., 1996; Rasmussen et al., 2013; Risch and Howe, 1995; Rowlands et al., 2011; Shu et al., 1989). However, PID was not uniformly defined in these studies, which may have led to misclassifications. In some studies, serologic evidence of chlamydia infection, a common cause of PID, has been associated with an increased ovarian cancer risk, particularly for cancer arising in the fallopian tube (Idahl et al., 2011; Ness et al., 2003, 2008).
Psychological, Social, and Environmental Risk Factors
Experimental evidence suggests that psychosocial stress can influence processes that may be relevant to ovarian cancer development, such as inflammation and wound healing (Kiecolt-Glaser et al., 2010; Powell et al., 2013b; Walburn et al., 2009) For example, in a study of mice with a Trp53 gene mutation, chronic stress led to increased tumor incidence and decreased latency (i.e., time from exposure to tumor growth) (Feng et al., 2012). Although ovarian tumors were not studied explicitly, there is a clear link between TP53 alterations and ovarian cancer (see Chapter 2). Furthermore, mice subjected to physical restraint or social isolation and injected with ovarian cancer cells were shown to have more than twice the tumor weight and nodule count than found in controls that had been injected with cancer cells but not stressed; half of all the stressed mice had metastases versus none in the controls (Thaker et al., 2006). Specifically, norepinephrine, a stress hormone, has been shown to alter cell adhesion, migration, invasion, and angiogenesis in stressed mice (Armaiz-Pena et al., 2013; Thaker et al., 2006). Norepinephrine also increases the invasive potential of the cells and the expression of vascular endothelial growth factor (Nilsson et al., 2007; Sood et al., 2006).
The role that chronic stress and its biologic response play in ovarian carcinogenesis is understudied in human populations. One meta-analysis found that reports of psychosocial stress were modestly, but significantly, associated with a 6 to 20 percent increase in overall cancer risk; the associations were stronger in studies with large sample sizes and long follow-up (Chida et al., 2008). A recent prospective study found that women with
depressive symptoms had a significant 30 percent higher risk of ovarian cancer (Huang et al., 2015b). Very little research has been conducted on the association between socioeconomic status and ovarian cancer risk. One study in Denmark suggested a positive association between disposable income and ovarian cancer risk, but no other socioeconomic indicators were associated with risk (Jensen et al., 2008).
RISK FACTORS AND TUMOR SUBTYPES
While some factors affect risk for all ovarian cancer subtypes, many factors appear to affect risk for different ovarian cancer subtypes in different ways, including increasing risk for one or more subtypes and decreasing risk for others (Yang et al., 2012). Generally, studies have used histology with or without tumor grade as a surrogate for tumor type. The inability to demonstrate clear associations for certain risk factors may be due in part to the limited power of individual studies to assess associations by tumor type.
The complex associations that these factors have with ovarian cancer risk are driven in part by the differing etiologies for the diverse ovarian cancer subtypes. Many of the studies described earlier in this chapter used the Type I and Type II classification dichotomy. This schema has greater power to detect associations (by increasing the number of cases in each category), but it ignores the unique biology of each subtype and thereby likely masks particular associations.
Risk prediction models for women at average genetic risk are important tools for informing women of their 5- and 10-year risk for ovarian cancer, for improving clinical decision making, and for identifying women for enrollment in prevention studies (Freedman et al., 2005). Table 3-4 outlines four studies that have developed risk prediction models for ovarian cancer among women at average genetic risk. Overall, these studies do not demonstrate a strong predictive ability that would be meaningful in clinical decision making, most likely because of the relative rarity of ovarian cancer and the somewhat modest effect sizes of the known risk factors. Improving the discriminatory ability of these models will likely require the consideration of differential associations by tumor subtype and the identification of new risk factors.
Most medical strategies designed to prevent the occurrence of ovarian cancer are structured around modulating female hormone cycles and the
TABLE 3-4
Risk Prediction Models for Ovarian Cancer in Average Risk Populations
| References | Study Used to Develop Model | Validation of Model | Risk Factors Included in Risk Prediction Model | AUCa |
| Hartge et al., 1994 | Seven case-control studies | No | Parity, OC use, family history of breast or ovarian cancer | None provided |
| Rosner et al., 2005 | Nurses’ Health Study I and II | Yes | Estimated ovulatory years (incorporates parity and OC), duration of menopause, tubal ligation | 0.60 |
| Pfeiffer et al., 2013 | PLCO screening trial, NIH-AARP Diet and Health Study | Yes | Parity, OC use, family history of breast or ovarian cancer, menopausal hormone use | 0.59 |
| Li et al., 2015 | European Prospective Investigation into Cancer and Nutrition | Yes | Parity, OC use, menopausal status, age at menopause, menopausal hormone use, unilateral oophorectomy, BMI | 0.64 |
NOTE: AUC = area under the curve; BMI = body mass index; NIH = National Institutes of Health; OC = oral contraceptive; PLCO = prostate, lung, colorectal, and ovarian.
aAn AUC of 0.5 predicts outcomes no better than chance (generally an AUC greater than 0.7 may be considered for clinical use).
surgical removal or modification of gynecological tract components, including the fallopian tubes (salpingectomy), ovaries (oophorectomy), and uterus (hysterectomy).
Bilateral Salpingo-Oophorectomy (BSO)
BSO, also known as risk-reducing salpingo-oophorectomy (RRSO), is the surgical removal of the fallopian tubes and ovaries, which dramatically reduces the risk of ovarian cancer in women at average risk and high risk due to inherited genetic susceptibility (see Table 3-5). The USPSTF and SGO advocate RRSO for women at high genetic risk, but it may also be effective for women at average or unknown genetic risk (Evans et al., 2009; Nelson et al., 2014; Walker et al., 2015). The GOG-0199 study, also known as the National Ovarian Cancer Prevention and Early Detection Study, a number of factors linked to a higher risk of ovarian cancer were found during RRSO, including being postmenopausal or having mutated BRCA1 or BRCA2 genes, abnormal CA-125 test results, and abnormal transvaginal ultrasound (TVU) results (Sherman et al., 2014). However, the
TABLE 3-5
Risk Reduction After Bilateral Salpingo-Oophorectomy
| Genetic Risk | Study | Number of Subjects | Relative Risk of Ovarian Cancer With vs. Without RRSO (95% confidence interval) | Risk Reduction (%) |
| High (BRCA1 or 2 positive) | Rutter et al., 2003 | 249 | 0.29 (0.12–0.73) | 71 |
| Finch et al., 2006 | 1,828 | 0.20 (0.07–0.58) | 80 | |
| Finkelman et al., 2012 | 3,787 | 0.08 (0.04–0.16) | 92 | |
| Average or low | Rutter et al., 2003 | 598 | 0.05 (0.01–0.22) | 95 |
| Parker et al., 2009a | 29,390 | 0.04 (0.01–0.09) | 96 | |
| Jacoby et al., 2011a | 25,448 | 0.07 (0.02–0.22) | 93 | |
| Unknown | Evans et al., 2009 | 803 | 0.08 (0.01–0.57) | 92 |
aOnly among women with a hysterectomy.
risk reduction achieved with RRSO is not 100 percent, and the procedure is not without inherent risks and side effects, including early menopause, osteoporosis, cardiovascular disease, and increased overall mortality (Finch et al., 2014). As such, no formal body has recommended RRSO for the primary prevention of ovarian cancer for the general population.
Bilateral Salpingectomy with Ovarian Retention (BSOR)
To avoid the long-term complications associated with removing the ovaries, BSOR, a surgical procedure that removes the fallopian tubes but leaves the ovaries intact, may prove to be a valuable option for women at risk for developing ovarian cancer (Daly et al., 2015). There is a growing evidence base suggesting that the various ovarian carcinoma subtypes have different sites of origins (see Chapter 2). However, the proportion of ovarian cancers that originate from sites outside the ovaries is unknown, and therefore the effectiveness of BSOR in preventing ovarian cancer is uncertain and may differ by subtype. For example, BSOR may be most effective in preventing the ovarian carcinoma subtypes postulated to arise in the fallopian tubes. Furthermore, BSOR can prevent retrograde menstrua-
tion of endometrial tissue, which is thought to be the origin and cause of endometriosis and possibly to be associated with endometrioid and clear cell carcinomas, as discussed in Chapter 2 (Leblanc et al., 2011; Salamanca and Beltran, 1995; Sampson, 1927; Sanfilippo et al., 1986).
BSOR might allow high-risk women to delay removal of ovaries until the procedure is desired or warranted. For average-risk women, BSOR at the time of a planned hysterectomy may be a prevention opportunity. Until recently, salpingectomy was typically not performed as part of a standard hysterectomy unless the ovaries were also being removed. Data suggest that a salpingectomy at the time of hysterectomy is feasible, safe, and does not affect short-term ovarian function (McAlpine et al., 2014). However, establishing the safety and efficacy of this procedure will require additional data from more women with a longer follow-up.
Recent population-based studies suggest that salpingectomy may reduce the incidence of ovarian cancer in the general population (Falconer et al., 2015; Guldberg et al., 2013). In a study from Sweden, salpingectomy (indicated for medical reasons) was associated with a 35 percent reduction in ovarian cancer risk (Falconer et al., 2015). A second study reported no significant reduction in risk for unilateral salpingectomy, but a 42 percent reduction in risk for BSOR (Madsen et al., 2015). However, these results were based on a small number of cases, and neither study had sufficient cases to assess the association by histologic subtype. In light of the evidence of the distal fallopian tube epithelium as the site of origin of at least some HGSCs, the SGO and American Congress of Obstetricians and Gynecologists have issued statements recommending consideration of opportunistic BSOR to reduce ovarian cancer mortality in the general population (ACOG Committee on Gynecologic Practice, 2015; SGO, 2013).
Tubal Ligation
Tubal ligation, a surgical procedure in which the fallopian tubes are tied or blocked in such a way that eggs released from the ovary cannot reach the uterus, reduces the risk for ovarian cancer in both high-genetic-risk and average-genetic-risk populations (Rice et al., 2012). One meta-analysis found that the risk for ovarian cancer for women who underwent tubal ligation dropped by 33 percent compared with women who did not undergo surgery (Cibula et al., 2011). The reduction in risk is associated primarily with the endometrioid and clear cell histologic subtypes and appears to last for up to 14 years post-surgery (Madsen et al., 2015; Rice et al., 2013; Sieh et al., 2013). Similar reductions in risk have been reported among BRCA1 mutation carriers in whom HGSCs are the most common subtype of ovarian cancer (Antoniou et al., 2009; Narod et al., 2001).
Hysterectomy
Women who have undergone a hysterectomy, the surgical removal of the uterus, have a lower risk of ovarian cancer (Rice et al., 2012; Vitonis et al., 2011). As noted earlier, endometriosis is associated with increased peritoneal inflammation and is commonly associated with endometrioid carcinoma and clear cell carcinoma (Arici, 2002; Leblanc et al., 2011; Malutan et al., 2015a,b). A hysterectomy may prevent ovarian cancer by limiting the ability of endometriotic tissue to access the fallopian tubes and the ovaries through retrograde menstruation, thereby stopping the associated inflammation and protumorigenic environment. Thus, a hysterectomy may prevent the development of certain types of ovarian cancer, but no formal body has recommended hysterectomy as a prevention strategy.
Prescription Medications
One of the primary alternatives to surgical intervention for the prevention of ovarian cancer is use of hormone-modulating prescription drugs such as OCs. The amount or type of hormones (i.e., estrogen and progestin) in OCs can affect ovarian cancer risk. However, research has produced conflicting results on the effects of the various types of hormones present in OCs (Greer et al., 2005; Hankinson et al., 1992; Schildkraut et al., 2002). As was noted earlier in this chapter, OCs have consistently been associated with a lower risk of ovarian cancer, including a reduction in the risk for nearly all histologic subtypes. One analysis estimated that two ovarian cancer cases and one ovarian cancer–related death are prevented for every 500 woman-years of OC use (Beral et al., 2008). The analysis further estimated that in developed countries, where ovarian cancer incidence is high and OC use is very common, approximately 13 percent of the ovarian cancer that would have occurred in women younger than age 75 is prevented by OC use. But there are also some risks associated with OC use, including a slightly increased breast cancer risk and a two- to threefold higher risk of venous thromboembolism and ischemic stroke (Bassuk and Manson, 2015; Havrilesky et al., 2013). These associations, however, are primarily related to the current use of OCs, and the diseases whose risks are increased by OC use are relatively rare in young women (Bassuk and Manson, 2015).
The SGO has stated that OCs “reduce the risk of ovarian cancer for average-risk women and BRCA1 and BRCA2 mutation carriers. Appropriate counseling about side effects and contraindications will allow each patient to weigh the risks and the benefits” (Walker et al., 2015, p. 2116). However, the CDC has concluded that insufficient evidence is available to address the use of OCs in ovarian cancer prevention (Havrilesky et al., 2013).
As discussed in Chapter 1, one of the chief causes of the significant morbidity and mortality from ovarian cancer is the late stage at which most women are diagnosed (see Figure 1-5), which is partly due to the lack of clear and unique ovarian cancer–specific symptoms. Because of this absence of specific symptoms, researchers have investigated other strategies for early ovarian cancer detection, such as assaying for one or more biomarkers, often in combination with imaging technologies. While the use of these strategies in large screening trials has resulted in more women being diagnosed with ovarian cancer at earlier stages, to date these strategies have not reduced overall mortality. Furthermore, because some ovarian carcinoma subtypes originate away from the ovaries, it is difficult to know where to look to detect the earliest lesions associated with ovarian cancer. Because this is an issue specific to ovarian cancer, it is difficult to draw on early detection methods from other heterogeneous cancers. Thus developing effective and reliable early detection strategies for ovarian cancer will require ongoing research aimed at better understanding the early molecular and genetic events associated with the carcinogenesis of each subtype of ovarian cancer, along with better assessment of disease-specific symptoms.
Biomarkers
A biomarker is “a biological molecule found in blood, other body fluids, or tissues that is a sign of a normal or abnormal process, or of a condition or disease. A biomarker may be used to see how well the body responds to a treatment for a disease or condition” (NCI, 2015). Although the most commonly used biomarkers in clinical care are proteins, the definition includes a broad spectrum of biochemical substances, including nucleic acids (e.g., DNA and various types of RNA), lipids, small metabolites, and even whole cells (IOM, 2007). Biomarkers are used throughout the cancer care continuum. Predictive biomarkers are used in risk assessment and to measure the biological response to an intervention, and prognostic biomarkers are used to describe outcomes such as progression-free or overall survival (Huang et al., 2010; IOM, 2007). In research on ovarian cancer, the most extensively studied and frequently used biomarkers are two proteins, cancer antigen 125 (CA-125) and human epididymis protein 4 (HE-4). The following sections discuss how these and other biomarkers have affected our understanding of ovarian cancer risk and describe these biomarkers’ utility in screening and early detection.
CA-125
CA-125 gained prominence following a study that identified an antibody against CA-125 that reacted predominantly with malignant ovarian tissue (Bast et al., 1981). Nearly 80 percent of women with an advanced (Stage III or IV) ovarian carcinoma have elevated CA-125 serum levels at diagnosis (Niloff et al., 1984). Research that followed up on the original study found that serum levels of CA-125 correlate both with disease stage and with the response to chemotherapy, suggesting that CA-125 could be useful as a marker of disease progression as well as a prognostic biomarker (Bast et al., 1983; Hawkins et al., 1989; Hising et al., 1991; Kobayashi et al., 2012). CA-125’s potential use as a tool in early detection was extrapolated from these studies and also case reports noting its rise in asymptomatic women in advance of being diagnosed with ovarian cancer (Bast et al., 1985).
The biology behind CA-125’s apparent association with ovarian cancer risk and prognosis is currently unclear. Laboratory research suggests that CA-125 may play roles in metastasis to the peritoneal cavity and in promoting chemoresistance to several drugs that are used in standard ovarian cancer chemotherapy protocols, but these findings have not been replicated clinically (Boivin et al., 2009; Felder et al., 2014; Gubbels et al., 2006).
For early detection, CA-125 is a predictive tool that becomes increasingly powerful with proximity to diagnosis and may signal the presence of precursor lesions (Jacobs et al., 1999). Using trends in CA-125 levels to select women for imaging may improve its screening performance (Karlan et al., 2014). This strategy is currently being tested in the United Kingdom Collaborative Trial of Ovarian Cancer Screening (UKCTOCS). Several algorithms developed over the past decade can help predict the presence of ovarian cancer in women with a pelvic mass so that they may be triaged to appropriate specialists (i.e., gynecologic oncologists). The majority of these algorithms have incorporated CA-125 along with other biomarkers or diagnostic indicators (e.g., OVA-1, the Risk of Ovarian Malignancy Algorithm [ROMA], and the Risk of Malignancy Index) (Bast et al., 2012; Cohen et al., 2014; Jacobs et al., 1990; Moore et al., 2009; Skates et al., 1995).
Challenges with the use of CA-125 for early detection of or screening for ovarian cancer include its lack of specificity and sensitivity. For example, CA-125 may be markedly elevated in patients who have a variety of benign or nonovarian malignant conditions, and in approximately 20 percent of women with ovarian cancer it is expressed not at all or only in trace amounts (Cohen et al., 2014; Miralles et al., 2003; Moore et al., 2008). Finally, serum levels of CA-125 are significantly elevated above baseline in only half of women diagnosed with early stage (Stage I or II) ovarian cancer (Woolas et al., 1993).
HE-4
A 1999 study looking for genes that are significantly overexpressed in ovarian tumors when compared with normal ovarian tissue singled out WFDC2, which encodes the HE-4 protein, as a potential diagnostic marker for ovarian cancer (Schummer et al., 1999). Subsequent research confirmed this expression pattern in ovarian carcinomas, and additional work in cell and animal models suggested a role for HE-4 in mediating resistance to chemotherapy and promoting tumor growth (Moore et al., 2014; Welsh et al., 2001). Compared with CA-125, HE-4 has a similar sensitivity for detecting late-stage ovarian cancer but a greater specificity in differentiating between malignant and benign tumors (Bast et al., 2005). In 2011, the U.S. Food and Drug Administration (FDA) approved the use of ROMA, which combines measurements of HE-4 and CA-125 with menopausal status to determine whether a woman presenting with a pelvic mass is at a high or low risk of malignancy (Moore et al., 2009, 2011). However, as is the case with CA-125, elevated serum levels of HE-4 are not unique to women with ovarian tumors and are found in individuals with tumors of gynecologic and pulmonary origin (Drapkin et al., 2005; Karlsen et al., 2014). Nonetheless, the dramatic increases in HE-4 serum concentration seen in women with ovarian carcinomas (and in the serous and endometrioid subtypes in particular) support its usefulness as a biomarker with high specificity for the early detection of ovarian cancer (Escudero et al., 2011; Hertlein et al., 2012; Karlsen et al., 2014).
Other Ovarian Cancer Biomarkers
As described in Chapter 2, microRNAs (miR) appear to play a role in several biological processes related to ovarian cancer. A number of microRNAs are expressed at either a higher or lower level in ovarian cancer tissue than in normal ovarian surface epithelium, and they also differ in their levels of expression among the various ovarian cancer subtypes (Lee et al., 2009; Zhang et al., 2015). The altered levels of several of these microRNAs can be detected in a woman’s peripheral blood and may serve as early detection biomarkers alone or in tandem with other commonly used biomarkers (Taylor and Gercel-Taylor, 2008; Zhang et al., 2015).
Advances in protein and nucleic acid analysis technologies such as microfluidic chips, nuclear magnetic resonance, and other high-throughput platforms make possible the analysis of small amounts of patient-derived samples for numerous potential biomarkers. The ovarian cancer sample sources that are currently investigated include tumor cells in ascites (i.e., fluid that accumulates in the peritoneal cavity after metastasis), circulating tumor cells in the blood, and exosomes (i.e., small membrane-bound vesicles secreted by cancer cells into bodily fluids) (Castro et al., 2015).
Biomarker Tests
FDA-approved protein tumor markers include ROMA (HE-4 and CA-125), OVA-1 (measures levels of apolipoprotein A1, beta 2 microglobulin, CA-125, prealbumin, and transferrin), HE-4, and CA-125 (Fung, 2010; Moore et al., 2010; Moss et al., 2005; Muller, 2010; Wu et al., 2012). Although a great deal of research is being carried out on identifying and developing new biomarkers for ovarian cancer, scientists often find it difficult to navigate the analytical, diagnostic, and regulatory requirements for a clinical assay (Fuzery et al., 2013). Currently, none of the FDA-approved protein tumor markers are approved as screening tests for ovarian cancer.
Imaging Technologies for Early Detection
Imaging technologies help measure the size of tumors and the extent to which they have spread after the masses have been detected during a clinical examination, and these same technologies may have a role to play in earlier detection of ovarian cancer. Most of these technologies are noninvasive or minimally invasive and may be performed in the outpatient setting using no anesthesia or only local anesthesia. The most common imaging technologies used for ovarian cancer are ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI). Modifications to these technologies that incorporate radiologic markers or sound waves have improved image quality and resolution, and some newer techniques may enable up-close visualization of tumor growth in the fallopian tubes and on the ovary surfaces.
Transvaginal Ultrasound
TVU (also known as transvaginal sonography) is the most widely used imaging technique for the examination of pelvic organs (Manegold-Brauer et al., 2014). TVU is used primarily to evaluate gynecologic symptoms and pathologies, including pain or pressure in the pelvic region, irregular bleeding, fibroids, and adnexal masses (e.g., ovarian cysts, ectopic pregnancies, and tumors near the uterus). While TVU can identify most adnexal masses, the majority of these masses are benign, and TVU is limited in its ability to differentiate between malignant tumors and benign tumors. Overall, TVU has not yet shown value as a primary screening tool for ovarian cancer, but it may be useful with specific populations (e.g., women at high risk) or in conjunction with biomarkers for ovarian cancer screening (van Nagell et al., 2007).
Doppler and contrast-enhanced ultrasound techniques have been added to routine TVU to provide information on tissue vascularity and
angiogenesis in an effort to improve the ability to differentiate between benign and malignant masses. However, studies using Doppler imaging with TVU for ovarian cancer screening have revealed a wide range of specificities and a lower sensitivity than TVU alone (Kinkel et al., 2000). The use of non-targeted and targeted microbubbles to distinguish benign from malignant ovarian lesions is still in the investigational stage (Lutz et al., 2011).
Magnetic Resonance Imaging
MRI is commonly used for a number of diseases and disorders, and it may be useful for the diagnosis and staging of adnexal masses (Manegold-Brauer et al., 2014). One meta-analysis found a 91.9 percent sensitivity and 88.4 percent specificity for classifying adnexal masses as malignant (Dodge et al., 2012). These values are similar to the sensitivity (96.0 percent) and specificity (90.0 percent) of TVU for classifying adnexal masses as malignant (Timmerman et al., 1999). As a result, TVU is often the first choice to identify masses, although MRI is useful to determine malignant potential of these masses when TVU may be unreliable (Dodge et al., 2012).
Computed Tomography
While CT scanning is commonly used in the management of ovarian cancer, it is primarily used as a staging tool. However, the early stages of ovarian cancer development may be readily missed by CT alone as its ability to distinguish benign from malignant masses is lower that that of MRI or TVU (Alt et al., 2011; Bharwani et al., 2011).
18F-fluoro-deoxyglucose Positron Emission Tomography (18F-FDG-PET)
An improved understanding of the role of glucose metabolism in tumor development has led to the use of glucose-based positron emission tomography (18F-FDG-PET) for tumor imaging. While basic functional imaging techniques such as PET can detect actively growing masses, distinguishing between benign and malignant lesions is better done with 18F-FDG-PET combined with CT scanning (18F-FDG-PET/CT) (Manegold-Brauer et al., 2014; Yamamoto et al., 2008). Only a few studies have examined the usefulness of this technique for ovarian cancer screening, and early results indicate there is a high likelihood of missing borderline and low-grade tumors when using 18F-FDG-PET/CT (Castellucci et al., 2007; Risum et al., 2007; Yamamoto et al., 2008).
CHALLENGES TO EARLY DETECTION OF OVARIAN CANCER
Because of the marked heterogeneity of ovarian carcinomas, it is likely that no single tumor biomarker will be sufficient to aid in the early detection of all the histologic subtypes. Research shows, for instance, that the distinct carcinoma subtypes express different sets of post-translationally modified proteins and microRNAs (Hua et al., 2013; Lee et al., 2009). There are also questions concerning the timing and type of patient samples that will need to be collected in clinical studies, although one screening trial suggests that serial biomarker measurements have better predictive power than single-time-point sampling (see detailed discussion in next section) (Cohen et al., 2014; Menon et al., 2015). It is becoming clearer that a more individualized approach to measuring CA-125 may be needed rather than having a single threshold for all women (Skates et al., 2011). This individualized approach could include longitudinal biomarker, genetics, and epidemiologic results in order to more accurately assess the risk for ovarian cancer. Another outstanding challenge is determining which marker or combination of markers meets the sensitivity and specificity requirements for early detection of a rare and heterogeneous disease. The difficulties of performing such validation studies are exacerbated by the low incidence of ovarian cancer, especially when separating out the different subtypes. However, recent screening trials (e.g., GOG-0199) actively collected DNA, serum, plasma, and tissue samples from high-risk women, which resulted in a valuable repository of samples for future studies (Greene et al., 2008).
Several challenges stand in the way of developing reliable and accurate early detection technologies for ovarian carcinomas. Imaging technologies have improved markedly over the years—including becoming less invasive and providing finer resolution of images—but a major challenge remains in the incomplete understanding of early carcinogenesis. While other types of cancer also have multiple subtypes, ovarian cancer is distinct in that the different subtypes likely develop from different tissues of origin. Once researchers have a better understanding of the cell of origin for each of the ovarian carcinoma subtypes, they may then have more success with the use of imaging technologies to find these ovarian carcinomas at earlier stages.
Screening—checking for disease when there are no symptoms (NCI, 2015)—is a key tool in the early detection of disease. In general, the effectiveness of a screening test is evaluated in terms of the ability of the test to identify those individuals who truly have the disease in question and to rule out those individuals who do not have the disease. Furthermore, a standard goal is that no more than 10 exploratory diagnostic operations are
performed for every 1 actual case of ovarian cancer (Bast, 2003). Establishing these minimum targets is important primarily to avoid an unacceptable level of unnecessary and potentially harmful surgical or chemotherapeutic interventions (Etzioni et al., 2013; Jacobs and Menon, 2004).
No reliable screening method exists to detect ovarian cancer at earlier stages, and, as a result, no professional organization recommends screening in the general population. Current methods for detecting ovarian cancer include physical examination, assessment of symptoms, imaging methods (e.g., TVU), and the use of serum levels of CA-125 (as described previously) (Bast et al., 1998; Fishman et al., 2005; Higgins et al., 1990; Kinkel et al., 2000).
Ovarian Cancer Screening Trials
Early detection screening trials evaluate the effectiveness of screening strategies in reducing the morbidity and mortality from a disease. General screening strategies have proved to be effective in detecting early-stage disease for several forms of cancer (e.g., breast and colon cancer) in both general and high-risk populations (Nelson et al., 2009; Schoen et al., 2012). However, there is limited evidence to suggest that these strategies significantly decrease the short- or long-term mortality from these cancers (Harding et al., 2015). Several large trials have been conducted to determine whether screening for ovarian cancer within either the high-risk or the general population helps reduce mortality from the disease (see Table 3-6).
Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial
The Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial, which used CA-125 screening and TVU, showed no reduction in disease-specific mortality in women at average risk for ovarian cancer (Buys et al., 2011). However, an increase in invasive medical procedures and associated harms was observed. One limitation of this study was its use of a single fixed cutoff for CA-125 serum levels. Follow-up modeling studies of the PLCO study’s results suggest that nearly 20 percent of ovarian cancer could have been detected at an earlier point if a CA-125 level trajectory had been taken into account that used the risk ROCA employed in later trials, although using the trajectory would have not affected mortality (Buys et al., 2011; Drescher et al., 2013; Pinsky et al., 2013). ROCA is based on annual measurements of CA-125 evaluated in a serial fashion so that each woman serves as her own baseline (Menon et al., 2005).
To study the predictive value of biomarkers beyond CA-125, the PLCO trial distributed sets of Phase III patient serum samples that were collected prior to the clinical diagnosis of ovarian cancer (Cramer et al., 2011). Al-
TABLE 3-6
Ovarian Cancer Prospective Screening Trials Results Summary
| Study | Study Design | Screening Cohort Size | Screening Strategy | Interpretation of CA-125 | Sensitivity (%) | Mortality/ Surrogate Results | References |
| PLCO | RCT, general population | 30,630 | CA-125 and TVU | Fixed cutoff, 35 U/mL | OC/FT: 69.5 IOC/FT: 68.2 | No mortality benefit | Buys et al., 2011 |
| UKCTOCS | RCT, general population, two arms | 101,247 | 1. CA-125 followed by MMS 2. TVU only | ROCA (longitudinal sampling) | OC/FT: MMS (89.4)/ TVU (84.9) IOC/FT: MMS (84.9)/ TVU (75.0) IOC/FT: MMS (88.6)/TVU (65.8) | Relative mortality reduction in MMS (14%) and USS (11%) groups over no screening, but reductions were not significant in the primary analysis | Jacobs et al., 2015; Menon et al., 2015 |
| Japanese Shizuoka Cohort | RCT, low-risk postmenopausal | 41,688 | Physical exam, CA-125, and TVU | Fixed cutoff, 35 U/mL | OC/FT: 77.1 | Stage shift; Stage I OC in screened (63%) versus control (38%) | Kobayashi et al., 2008 |
| Kentucky Screening Study | Single-arm prospective, general population | 25,327 | TVU | None | OC/FT: 81 IOC/FT: 76.3 | 5-year survival in screened (74.8%) versus unscreened (53.7%) | van Nagell et al., 2007 |
NOTE: CA-125 = cancer antigen 125; IOC/FT = invasive cancer or fallopian tube; MMS = multimodal screening; OC/FT = ovarian cancer or fallopian tube cancer; PLCO = prostate, lung, colorectal, and ovarian cancer screening; RCT = randomized controlled trial; ROCA = risk of ovarian cancer algorithm; TVU = transvaginal ultrasound; UKCTOCS = United Kingdom Collaborative Trial of Ovarian Cancer Screening; USS = ultrasound scanning.
SOURCE: Adapted from Menon et al., 2014.
though researchers analyzed 35 different biomarkers that had been identified in PLCO trial specimens for use in ovarian cancer screening, CA-125 remained the single best biomarker among the samples tested (Cramer et al., 2011).
United Kingdom Collaborative Trial of Ovarian Cancer Screening
The UKCTOCS is the largest randomized controlled ovarian cancer screening trial to date, with 200,000 women enrolled and each assigned to one of three arms in order to compare two screening strategies (Sharma et al., 2012). The first strategy used ROCA to evaluate the trajectory of CA-125 serum concentration over time, starting from a baseline concentration (measured prior to diagnosis) and using concentrations measured annually throughout the 10-year screening period (Skates et al., 1995, 2001). This longitudinal sampling contrasts with the single fixed cutoff used in previous screening trials. In the first screening arm, the CA-125 trajectory was evaluated, and TVU was used as a second line test for women who had significantly elevated CA-125 levels. The second screening arm used only an annual TVU imaging, while the third, control arm used no intervention at all. In addition to evaluating patient survival, the UKCTOCS collected data on the cost of screening, acceptance of screening modalities by patients, and a variety of physiological and psychosocial comorbidities linked to screening with TVU and measuring CA-125 levels.
The UKCTOCS found that using ROCA and TVU resulted in a notable improvement in the early detection of ovarian cancer at earlier stages (Menon et al., 2015) (see Table 3-6). Compared to using different fixed cutoffs for CA-125 concentration, screening with ROCA doubled the number of ovarian or tubal carcinomas detected in the trial. However, as most of the cancer detected at earlier stages was not of the deadly HGSC subtype, this improvement in early diagnosis may not lead to a similarly significant improvement in mortality. Recently published mortality data indicated that there was a 15 percent mortality reduction with CA-125 plus TVU and an 11 percent mortality reduction in TVU alone compared with no screening over 14 years (Jacobs et al., 2015). When the analysis was restricted to years 7 through 14, when most of the mortality occurred, the benefits of the CA-125 and TVU screening were more obvious, as it reduced mortality by 23 percent in this time frame. Although these results are promising, the current methods still need further refining, and it is likely that distinct multimodal approaches will be needed in order to detect each of the various subtypes at their earliest stages.
Japanese Shizuoka Cohort
The Shizuoka Cohort Study of Ovarian Cancer Screening prospective randomized controlled trial involved more than 80,000 asymptomatic postmenopausal women who were enrolled between 1985 and 1999 (Kobayashi et al., 2008). Nearly half of the women were assigned to the intervention arm, which consisted of annual screening with pelvic ultrasound and CA-125 serum measurement. The control arm included no screening. The ultrasound techniques used for the prevalence or first incidence scans were TVU (in the majority of cases screened after 1990) or transabdominal ultrasound. Women with abnormal ultrasound findings or elevated CA-125 levels (or both) were referred for further medical evaluation or investigative surgery by gynecologic oncologists. The trial closed in December 2002 with a mean follow-up period of 9.2 years. A higher proportion of women with Stage I ovarian cancer were identified in the screened group than in the control group, but it was not statistically significant (Kobayashi et al., 2008).
Kentucky Screening Study
Between 1987 and 2005, more than 25,000 women participated in a screening study at the University of Kentucky (van Nagell et al., 2007). In this single-arm study, women received annual TVU screening. The 5-year survival rate in women whose screens were positive and who were diagnosed with invasive ovarian carcinoma was significantly higher than among women treated at the University of Kentucky during the same time period who did not undergo screening (75 versus 54 percent). As improved therapies continue to be developed, even a modest improvement in detection time may improve survival rates.
The committee offers the following findings and conclusions:
- Given the relative rarity of ovarian cancer and the heterogeneity of the disease, consortia are likely to be necessary to achieve the statistical confidence (power) to evaluate risk factor associations by tumor histologic subtype.
- While several risk factors for ovarian cancer have been identified, their associations with specific histologic subtypes require further clarification.
- Not all women who carry germline genetic mutations in cancer-predisposition genes have an apparent family history.
- The contribution that known mutations in the BRCA1 and BRCA2 genes, as well as others linked to hereditary cancer syndromes (e.g., Lynch syndome), make to inherited genetic risk is well characterized, yet it does not account for all of the inherited risk for ovarian cancer.
- New mutations identified in recent sequencing studies need further risk quantification before they can be integrated into clinical practice.
- The current understanding of risk factors has limited utility in accurately predicting risk at the individual level; thus, there is a clear need for improved and validated risk prediction models that can be used to screen the general population of women.
- BSO has been shown to drastically reduce a woman’s risk of ovarian cancer and is recommended for women known to be at high genetic risk for developing ovarian cancer. Adverse side effects (e.g., surgical complications, loss of fertility, and premature menopause) and the low incidence of ovarian cancer in the general population preclude performance of these surgeries as part of a risk-reducing strategy in women at low or average genetic risk for ovarian cancer.
- The potential impact of opportunistic salpingectomy on reducing risk for women at high risk and average risk of developing ovarian cancer needs to be studied.
- Current imaging technologies for ovarian cancer screening are effective at detecting pelvic masses but are limited in their sensitivity to detect small, early lesions. Efforts to improve early detection through technology are hampered by an incomplete understanding of tumorigenesis, including knowledge about the cell or tissue of origin.
- Current screening methods have not had a substantial impact on overall mortality for general or high-risk populations irrespective of the biomarkers, imaging strategy, or risk prediction algorithms that have been used. In addition, the best currently available protocol for early detection of ovarian cancer (a combination of screening for elevated CA-125 and use of TVU) does not meet the risk–benefit ratio criteria for support by the USPSTF.
- The largest and most recent screening trial, UKCTOCS, has demonstrated a significant improvement in early detection, although the improvement in mortality was modest.
- No reliable biomarker or panel test exists for the detection of early-stage ovarian cancer.
ACOG (American College of Obstetricians and Gynecologists) Committee on Gynecologic Practice. 2015. Committee opinion no. 620: Salpingectomy for ovarian cancer prevention. Obstetrics and Gynecology 125(1):279-281.
ACS (American College of Surgeons). 2012. Cancer program standards 2012: Ensuring patient-centered care. https://www.facs.org/~/media/files/quality%20programs/cancer/coc/programstandards2012.ashx (accessed September 15, 2015).
Alsop, K., S. Fereday, C. Meldrum, A. DeFazio, C. Emmanuel, J. George, A. Dobrovic, M. J. Birrer, P. M. Webb, C. Stewart, M. Friedlander, S. Fox, D. Bowtell, and G. Mitchell. 2012. BRCA mutation frequency and patterns of treatment response in BRCA mutation-positive women with ovarian cancer: A report from the Australian Ovarian Cancer Study Group. Journal of Clinical Oncology 30(21):2654-2663.
Alt, C. D., K. A. Brocker, M. Eichbaum, C. Sohn, F. U. Arnegger, H. U. Kauczor, and P. Hallscheidt. 2011. Imaging of female pelvic malignancies regarding MRI, CT, and PET/CT: Part 2. Strahlentherapie und Onkologie 187(11):705-714.
Antoniou, A. C., M. Rookus, N. Andrieu, R. Brohet, J. Chang-Claude, et al. 2009. Reproductive and hormonal factors, and ovarian cancer risk for BRCA1 and BRCA2 mutation carriers: Results from the International BRCA1/2 Carrier Cohort Study. Cancer Epidemiology Biomarkers and Prevention 18(2):601-610.
Arici, A. 2002. Local cytokines in endometrial tissue: The role of interleukin-8 in the pathogenesis of endometriosis. Annals of the New York Academy of Sciences 955:101-109; discussion 118, 396-406.
Armaiz-Pena, G. N., S. W. Cole, S. K. Lutgendorf, and A. K. Sood. 2013. Neuroendocrine influences on cancer progression. Brain, Behavior, and Immunity 30(Suppl):S19-S25.
Aune, D., D. A. Navarro Rosenblatt, D. S. Chan, L. Abar, S. Vingeliene, A. R. Vieira, D. C. Greenwood, and T. Norat. 2015. Anthropometric factors and ovarian cancer risk: A systematic review and nonlinear dose-response meta-analysis of prospective studies. International Journal of Cancer 136(8):1888-1898.
Barry, J. A., M. M. Azizia, and P. J. Hardiman. 2014. Risk of endometrial, ovarian and breast cancer in women with polycystic ovary syndrome: A systematic review and meta-analysis. Human Reproduction Update 20(5):748-758.
Bassuk, S. S., and J. E. Manson. 2015. Oral contraceptives and menopausal hormone therapy: Relative and attributable risks of cardiovascular disease, cancer, and other health outcomes. Annals of Epidemiology 25(3):193-200.
Bast, R. C., Jr. 2003. Status of tumor markers in ovarian cancer screening. Journal of Clinical Oncology 21(10 Suppl):200s-205s.
Bast, R. C., Jr., M. Feeney, H. Lazarus, L. M. Nadler, R. B. Colvin, and R. C. Knapp. 1981. Reactivity of a monoclonal antibody with human ovarian carcinoma. Journal of Clinical Investigation 68(5):1331-1337.
Bast, R. C., Jr., T. L. Klug, E. St John, E. Jenison, J. M. Niloff, H. Lazarus, R. S. Berkowitz, T. Leavitt, C. T. Griffiths, L. Parker, V. R. Zurawski, Jr., and R. C. Knapp. 1983. A radioimmunoassay using a monoclonal antibody to monitor the course of epithelial ovarian cancer. New England Journal of Medicine 309(15):883-887.
Bast, R. C., Jr., F. P. Siegal, C. Runowicz, T. L. Klug, V. R. Zurawski, Jr., D. Schonholz, C. J. Cohen, and R. C. Knapp. 1985. Elevation of serum CA 125 prior to diagnosis of an epithelial ovarian carcinoma. Gynecologic Oncology 22(1):115-120.
Bast, R. C., Jr., F. J. Xu, Y. H. Yu, S. Barnhill, Z. Zhang, and G. B. Mills. 1998. CA 125: The past and the future. International Journal of Biological Markers 13(4):179-187.
Bast, R. C., Jr., D. Badgwell, Z. Lu, R. Marquez, D. Rosen, J. Liu, K. A. Baggerly, E. N. Atkinson, S. Skates, Z. Zhang, A. Lokshin, U. Menon, I. Jacobs, and K. Lu. 2005. New tumor markers: CA125 and beyond. International Journal of Gynecological Cancer 15(Suppl 3):274-281.
Bast, R. C., Jr., S. Skates, A. Lokshin, and R. G. Moore. 2012. Differential diagnosis of a pelvic mass: Improved algorithms and novel biomarkers. International Journal of Gynecological Cancer 22(Suppl 1):S5-S8.
Beauchamp, M. C., A. Yasmeen, A. Knafo, and W. H. Gotlieb. 2010. Targeting insulin and insulin-like growth factor pathways in epithelial ovarian cancer. Journal of Oncology Print 2010:257058.
Beral, V., R. Doll, C. Hermon, R. Peto, G. Reeves, et al. 2008. Ovarian cancer and oral contraceptives: Collaborative reanalysis of data from 45 epidemiological studies including 23,257 women with ovarian cancer and 87,303 controls. Lancet 371(9609):303-314.
Beral, V., K. Gaitskell, C. Hermon, K. Moser, G. Reeves, and R. Peto. 2012. Ovarian cancer and smoking: Individual participant meta-analysis including 28,114 women with ovarian cancer from 51 epidemiological studies. Lancet Oncology 13(9):946-956.
Beral, V., K. Gaitskell, C. Hermon, K. Moser, G. Reeves, and R. Peto. 2015. Menopausal hormone use and ovarian cancer risk: Individual participant meta-analysis of 52 epidemiological studies. Lancet 385(9980):1835-1842.
Bevier, M., J. Sundquist, and K. Hemminki. 2011. Does the time interval between first and last birth influence the risk of endometrial and ovarian cancer? European Journal of Cancer 47(4):586-591.
Bharwani, N., R. H. Reznek, and A. G. Rockall. 2011. Ovarian cancer management: The role of imaging and diagnostic challenges. European Journal of Radiology 78(1):41-51.
Blank, M. M., N. Wentzensen, M. A. Murphy, A. Hollenbeck, and Y. Park. 2012. Dietary fat intake and risk of ovarian cancer in the NIH-AARP Diet and Health Study. British Journal of Cancer 106(3):596-602.
Boivin, M., D. Lane, A. Piche, and C. Rancourt. 2009. CA125 (MUC16) tumor antigen selectively modulates the sensitivity of ovarian cancer cells to genotoxic drug-induced apoptosis. Gynecologic Oncology 115(3):407-413.
Bojesen, S. E., K. A. Pooley, S. E. Johnatty, J. Beesley, K. Michailidou, et al. 2013. Multiple independent variants at the TERT locus are associated with telomere length and risks of breast and ovarian cancer. Nature Genetics 45(4):371-384.
Bolton, K. L., J. Tyrer, H. Song, S. J. Ramus, M. Notaridou, et al. 2010. Common variants at 19p13 are associated with susceptibility to ovarian cancer. Nature Genetics 42(10):880-884.
Bowen, M. S., K. Kolor, W. D. Dotson, R. M. Ned, and M. J. Khoury. 2012. Public health action in genomics is now needed beyond newborn screening. Public Health Genomics 15(6):327-334.
Brasky, T. M., J. Liu, E. White, U. Peters, J. D. Potter, R. B. Walter, C. S. Baik, D. S. Lane, J. E. Manson, M. Z. Vitolins, M. A. Allison, J. Y. Tang, and J. Wactawski-Wende. 2014. Non-steroidal anti-inflammatory drugs and cancer risk in women: Results from the Women’s Health Initiative. International Journal of Cancer 135(8):1869-1883.
Bruchim, I., and H. Werner. 2013. Targeting IGF-1 signaling pathways in gynecologic malignancies. Expert Opinion on Therapeutic Targets 17(3):307-320.
Buys, S. S., E. Partridge, A. Black, C. C. Johnson, L. Lamerato, et al. 2011. Effect of screening on ovarian cancer mortality: The Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Randomized Controlled Trial. Journal of the American Medical Association 305(22):2295-2303.
Camargo, M. C., L. T. Stayner, K. Straif, M. Reina, U. Al-Alem, P. A. Demers, and P. J. Landrigan. 2011. Occupational exposure to asbestos and ovarian cancer: A meta-analysis. Environmental Health Perspectives 119(9):1211-1217.
Cassidy, A., T. Huang, M. S. Rice, E. B. Rimm, and S. S. Tworoger. 2014. Intake of dietary flavonoids and risk of epithelial ovarian cancer. American Journal of Clinical Nutrition 100(5):1344-1351.
Castellucci, P., A. M. Perrone, M. Picchio, T. Ghi, M. Farsad, C. Nanni, C. Messa, M. C. Meriggiola, G. Pelusi, A. Al-Nahhas, D. Rubello, F. Fazio, and S. Fanti. 2007. Diagnostic accuracy of 18F-FDG PET/CT in characterizing ovarian lesions and staging ovarian cancer: Correlation with transvaginal ultrasonography, computed tomography, and histology. Nuclear Medicine Communications 28(8):589-595.
Castro, C. M., H. Im, C. Le, H. Lee, R. Weissleder, and M. J. Birrer. 2015. Exploring alternative ovarian cancer biomarkers using innovative nanotechnology strategies. Cancer and Metastasis Reviews 34(1):75-82.
Chandran, U., E. V. Bandera, M. G. Williams-King, L. E. Paddock, L. Rodriguez-Rodriguez, S. E. Lu, S. Faulkner, K. Pulick, and S. H. Olson. 2011. Healthy eating index and ovarian cancer risk. Cancer Causes and Control 22(4):563-571.
Chang, E. T., V. S. Lee, A. J. Canchola, C. A. Clarke, D. M. Purdie, P. Reynolds, H. Anton-Culver, L. Bernstein, D. Deapen, D. Peel, R. Pinder, R. K. Ross, D. O. Stram, D. W. West, W. Wright, A. Ziogas, and P. L. Horn-Ross. 2007. Diet and risk of ovarian cancer in the California Teachers Study cohort. American Journal of Epidemiology 165(7):802-813.
Chida, Y., M. Hamer, J. Wardle, and A. Steptoe. 2008. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nature Clinical Practice: Oncology 5(8):466-475.
Cibula, D., M. Widschwendter, O. Majek, and L. Dusek. 2011. Tubal ligation and the risk of ovarian cancer: Review and meta-analysis. Human Reproduction Update 17(1):55-67.
Clendenen, T. V., A. A. Arslan, A. E. Lokshin, M. Liu, E. Lundin, K. L. Koenig, F. Berrino, G. Hallmans, A. Idahl, V. Krogh, A. Lukanova, A. Marrangoni, P. Muti, B. M. Nolen, N. Ohlson, R. E. Shore, S. Sieri, and A. Zeleniuch-Jacquotte. 2013. Circulating prolactin levels and risk of epithelial ovarian cancer. Cancer Causes and Control 24(4):741-748.
CMS (Centers for Medicare & Medicaid Services). 2015. FAQs about Affordable Care Act implementation (part XXVI). https://www.cms.gov/CCIIO/Resources/Fact-Sheets-and-FAQs/Downloads/aca_implementation_faqs26.pdf (accessed October 2, 2015).
Cohen, J. G., M. White, A. Cruz, and R. Farias-Eisner. 2014. In 2014, can we do better than CA125 in the early detection of ovarian cancer? World Journal of Biological Chemistry 5(3):286-300.
Collaborative Group on Epidemiological Studies of Ovarian Cancer. 2015. Menopausal hormone use and ovarian cancer risk: Individual participant meta-analysis of 52 epidemiological studies. Lancet 385(9980):1835-1842.
Cramer, D. W. 1989. Lactase persistence and milk consumption as determinants of ovarian cancer risk. American Journal of Epidemiology 130(5):904-910.
Cramer, D. W., R. C. Bast, Jr., C. D. Berg, E. P. Diamandis, A. K. Godwin, et al. 2011. Ovarian cancer biomarker performance in Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial specimens. Cancer Prevention Research 4(3):365-374.
Cramer, D. W., A. F. Vitonis, K. L. Terry, W. R. Welch, L.J. Titus. 2015. The association between talc use and ovarian cancer: A retrospective case-control study in two U.S. states. Epidemiology (Epub ahead of print).
Daly, M. B., C. W. Dresher, M. S. Yates, J. M. Jeter, B. Y. Karlan, D. S. Alberts, and K. H. Lu. 2015. Salpingectomy as a means to reduce ovarian cancer risk. Cancer Prevention Research (Philadelphia, PA) 8(5):342-348.
Daniilidis, A., and K. Dinas. 2009. Long term health consequences of polycystic ovarian syndrome: A review analysis. Hippokratia 13(2):90-92.
Desmond, A., A. W. Kurian, M. Gabree, M. A. Mills, M. J. Anderson, Y. Kobayashi, N. Horick, S. Yang, K. M. Shannon, N. Tung, J. M. Ford, S. E. Lincoln, and L. W. Ellisen. 2015. Clinical actionability of multigene panel testing for hereditary breast and ovarian cancer risk assessment. JAMA Oncology 1(7):943-951.
Dodge, J. E., A. L. Covens, C. Lacchetti, L. M. Elit, T. Le, M. Devries-Aboud, M. Fung-Kee-Fung, and Gynecology Cancer Disease Site Group. 2012. Management of a suspicious adnexal mass: A clinical practice guideline. Current Oncology (Toronto, Ontario) 19(4):e244-e257.
Domchek, S. M., and K. L. Nathanson. 2014. Panel testing for inherited susceptibility to breast, ovarian, and colorectal cancer. Genetics in Medicine 16(11):827-829.
Domchek, S. M., T. M. Friebel, C. F. Singer, D. G. Evans, H. T. Lynch, et al. 2010. Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. Journal of the American Medical Association 304(9):967-975.
Drapkin, R., H. H. von Horsten, Y. Lin, S. C. Mok, C. P. Crum, W. R. Welch, and J. L. Hecht. 2005. Human epididymis protein 4 (HE4) is a secreted glycoprotein that is overexpressed by serous and endometrioid ovarian carcinomas. Cancer Research 65(6):2162-2169.
Drescher, C. W., C. Shah, J. Thorpe, K. O—Briant, G. L. Anderson, C. D. Berg, N. Urban, and M. W. McIntosh. 2013. Longitudinal screening algorithm that incorporates change over time in CA125 levels identifies ovarian cancer earlier than a single-threshold rule. Journal of Clinical Oncology 31(3):387-392.
Eccles, D., G. Mitchell, A. N. Monteiro, R. Schmutzler, F. J. Couch, A. B. Spurdle, E. B. Gomez-Garcia, and E. C. W. Group. 2015. BRCA1 and BRCA2 genetic testing—Pitfalls and recommendations for managing variants of uncertain clinical significance. Annals of Oncology 26(10):2057-2065.
Edefonti, V., A. Decarli, C. La Vecchia, C. Bosetti, G. Randi, S. Franceschi, L. Dal Maso, and M. Ferraroni. 2008. Nutrient dietary patterns and the risk of breast and ovarian cancers. International Journal of Cancer 122(3):609-613.
Edefonti, V., G. Randi, A. Decarli, C. La Vecchia, C. Bosetti, S. Franceschi, L. Dal Maso, and M. Ferraroni. 2009. Clustering dietary habits and the risk of breast and ovarian cancers. Annals of Oncology 20(3):581-590.
Escudero, J. M., J. M. Auge, X. Filella, A. Torne, J. Pahisa, and R. Molina. 2011. Comparison of serum human epididymis protein 4 with cancer antigen 125 as a tumor marker in patients with malignant and nonmalignant diseases. Clinical Chemistry 57(11):1534-1544.
Etzioni, R., R. Gulati, L. Mallinger, and J. Mandelblatt. 2013. Influence of study features and methods on overdiagnosis estimates in breast and prostate cancer screening. Annals of Internal Medicine 158(11):831-838.
Evans, D. G., R. Clayton, P. Donnai, A. Shenton, and F. Lalloo. 2009. Risk-reducing surgery for ovarian cancer: Outcomes in 300 surgeries suggest a low peritoneal primary risk. European Journal of Human Genetics 17(11):1381-1385.
Falconer, H., L. Yin, H. Gronberg, and D. Altman. 2015. Ovarian cancer risk after salpingectomy: A nationwide population-based study. Journal of the National Cancer Institute 107(2).
Febbraro, T., K. Robison, J. S. Wilbur, J. Laprise, A. Bregar, V. Lopes, R. Legare, and A. Stuckey. 2015. Adherence patterns to National Comprehensive Cancer Network (NCCN) guidelines for referral to cancer genetic professionals. Gynecologic Oncology 138(1):109-114.
Felder, M., A. Kapur, J. Gonzalez-Bosquet, S. Horibata, J. Heintz, R. Albrecht, L. Fass, J. Kaur, K. Hu, H. Shojaei, R. J. Whelan, and M. S. Patankar. 2014. MUC16 (CA125): Tumor biomarker to cancer therapy, a work in progress. Molecular Cancer 13:129.
Feng, Z., L. Liu, C. Zhang, T. Zheng, J. Wang, M. Lin, Y. Zhao, X. Wang, A. J. Levine, and W. Hu. 2012. Chronic restraint stress attenuates p53 function and promotes tumorigenesis. Proceedings of the National Academy of Sciences of the United States of America 109(18):7013-7018.
Finch, A., M. Beiner, J. Lubinski, H. T. Lynch, P. Moller, et al. 2006. Salpingo-oophorectomy and the risk of ovarian, fallopian tube, and peritoneal cancers in women with a BRCA1 or BRCA2 mutation. Journal of the American Medical Association 296(2):185-192.
Finch, A. P. M., J. Lubinski, P. Møller, C. F. Singer, B. Karlan, et al. 2014. Impact of oophorectomy on cancer incidence and mortality in women with a BRCA1 or BRCA2 mutation. Journal of Clinical Oncology 32(15):1547-1553.
Finkelman, B. S., W. S. Rubinstein, S. Friedman, T. M. Friebel, S. Dubitsky, et al. 2012. Breast and ovarian cancer risk and risk reduction in Jewish BRCA1/2 mutation carriers. Journal of Clinical Oncology 30(12):1321-1328.
Fishman, D. A., L. Cohen, S. V. Blank, L. Shulman, D. Singh, K. Bozorgi, R. Tamura, I. Timor-Tritsch, and P. E. Schwartz. 2005. The role of ultrasound evaluation in the detection of early-stage epithelial ovarian cancer. American Journal of Obstetrics and Gynecology 192(4):1214-1221; discussion 1221-1222.
FORCE (Facing Our Risk of Cancer Empowered). 2008. Previvor: Past, present, & future. http://www.facingourrisk.org/get-involved/HBOC-community/BRCA-HBOC-blogs/FORCE/general/previvor-past-present-future (accessed November 23, 2015).
Fortner, R. T., J. Ose, M. A. Merritt, H. Schock, A. Tjønneland, et al. 2015. Reproductive and hormone-related risk factors for epithelial ovarian cancer by histologic pathways, invasiveness and histologic subtypes: Results from the EPIC cohort. International Journal of Cancer 137(5):1196-1208.
Freedman, A. N., D. Seminara, M. H. Gail, P. Hartge, G. A. Colditz, R. Ballard-Barbash, and R. M. Pfeiffer. 2005. Cancer risk prediction models: A workshop on development, evaluation, and application. Journal of the National Cancer Institute 97(10):715-723.
Fung, E. T. 2010. A recipe for proteomics diagnostic test development: The OVA1 test, from biomarker discovery to FDA clearance. Clinical Chemistry 56(2):327-329.
Fuzery, A. K., J. Levin, M. M. Chan, and D. W. Chan. 2013. Translation of proteomic biomarkers into FDA approved cancer diagnostics: Issues and challenges. Clinical Proteomics 10(1):13.
Garber, J. E., and K. Offit. 2005. Hereditary cancer predisposition syndromes. Journal of Clinical Oncology 23(2):276-292.
Gates, M. A., A. F. Vitonis, S. S. Tworoger, B. Rosner, L. Titus-Ernstoff, S. E. Hankinson, and D. W. Cramer. 2009. Flavonoid intake and ovarian cancer risk in a population-based case-control study. International Journal of Cancer 124(8):1918-1925.
Gates, M. A., B. A. Rosner, J. L. Hecht, and S. S. Tworoger. 2010. Risk factors for epithelial ovarian cancer by histologic subtype. American Journal of Epidemiology 171(1):45-53.
Genkinger, J. M., D. J. Hunter, D. Spiegelman, K. E. Anderson, A. Arslan, et al. 2006a. Dairy products and ovarian cancer: A pooled analysis of 12 cohort studies. Cancer Epidemiology Biomarkers and Prevention 15(2):364-372.
Genkinger, J. M., D. J. Hunter, D. Spiegelman, K. E. Anderson, W. L. Beeson, et al. 2006b. A pooled analysis of 12 cohort studies of dietary fat, cholesterol and egg intake and ovarian cancer. Cancer Causes and Control 17(3):273-285.
Genkinger, J. M., D. J. Hunter, D. Spiegelman, K. E. Anderson, J. E. Buring, et al. 2006c. Alcohol intake and ovarian cancer risk: A pooled analysis of 10 cohort studies. British Journal of Cancer 94(5):757-762.
George, R., K. Kovak, and S. L. Cox. 2015. Aligning policy to promote cascade genetic screening for prevention and early diagnosis of heritable diseases. Journal of Genetic Counseling 24(3):388-399.
Goode, E. L., G. Chenevix-Trench, H. Song, S. J. Ramus, M. Notaridou, et al. 2010. A genome-wide association study identifies susceptibility loci for ovarian cancer at 2q31 and 8q24. Nature Genetics 42(10):874-879.
Gosvig, C. F., S. K. Kjaer, J. Blaakaer, E. Hogdall, C. Hogdall, and A. Jensen. 2015. Coffee, tea, and caffeine consumption and risk of epithelial ovarian cancer and borderline ovarian tumors: Results from a Danish case-control study. Acta Oncologica 1-8.
Gottschau, M., S. K. Kjaer, A. Jensen, C. Munk, and L. Mellemkjaer. 2015. Risk of cancer among women with polycystic ovary syndrome: A Danish cohort study. Gynecologic Oncology 136(1):99-103.
Greene, M. H., M. Piedmonte, D. Alberts, M. Gail, M. Hensley, Z. Miner, P. L. Mai, J. Loud, G. Rodriguez, J. Basil, J. Boggess, P. E. Schwartz, J. L. Kelley, K. E. Wakeley, L. Minasian, and S. Skates. 2008. A prospective study of risk-reducing salpingo-oophorectomy and longitudinal CA-125 screening among women at increased genetic risk of ovarian cancer: Design and baseline characteristics: A gynecologic oncology group study. Cancer Epidemiology, Biomarkers and Prevention 17(3):594-604.
Greer, J. B., F. Modugno, G. O. Allen, and R. B. Ness. 2005. Androgenic progestins in oral contraceptives and the risk of epithelial ovarian cancer. Obstetrics and Gynecology 105(4):731-740.
Gubbels, J. A., J. Belisle, M. Onda, C. Rancourt, M. Migneault, M. Ho, T. K. Bera, J. Connor, B. K. Sathyanarayana, B. Lee, I. Pastan, and M. S. Patankar. 2006. Mesothelin-MUC16 binding is a high affinity, N-glycan dependent interaction that facilitates peritoneal metastasis of ovarian tumors. Molecular Cancer 5(1):50.
Guldberg, R., S. Wehberg, C. W. Skovlund, O. Mogensen, and O. Lidegaard. 2013. Salpingectomy as standard at hysterectomy? A Danish cohort study, 1977–2010. BMJ Open 3(6).
Guo, S. W. 2015. Endometriosis and ovarian cancer: Potential benefits and harms of screening and risk-reducing surgery. Fertility and Sterility 104(4):813-830.
Hampel, H., R. L. Bennett, A. Buchanan, R. Pearlman, G. L. Wiesner, and the Guideline Development Group of the American College of Medical Genetics and Genomics Professional Practice and Guidelines Committee and the National Society of Genetic Counselors Practice Guidelines Committee. 2015. A practice guideline from the American College of Medical Genetics and Genomics and the National Society of Genetic Counselors: Referral indications for cancer predisposition assessment. Genetics in Medicine 17(1):70-87.
Hanahan, D., and R. A. Weinberg. 2011. Hallmarks of cancer: The next generation. Cell 144(5):646-674.
Hankinson, S. E., G. A. Colditz, D. J. Hunter, T. L. Spencer, B. Rosner, and M. J. Stampfer. 1992. A quantitative assessment of oral contraceptive use and risk of ovarian cancer. Obstetrics and Gynecology 80(4):708-714.
Harding, C., F. Pompei, D. Burmistrov, H. G. Welch, R. Abebe, and R. Wilson. 2015. Breast cancer screening, incidence, and mortality across U.S. counties. JAMA Internal Medicine 175(9):1483-1489.
Hartge, P., A. S. Whittemore, J. Itnyre, L. McGowan, and D. Cramer. 1994. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstetrics and Gynecology 84(5):760-764.
Havrilesky, L. J., J. M. Gierisch, P. G. Moorman, R. R. Coeytaux, R. P. Urrutia, W. J. Lowery, M. Dinan, A. J. McBroom, L. Wing, M. D. Musty, K. R. Lallinger, V. Hasselblad, G. D. Sanders, and E. R. Myers. 2013. Oral contraceptive use for the primary prevention of ovarian cancer: Evidence reports/technology assessments, no. 212. Rockville, MD: Agency for Healthcare Research and Quality.
Hawkins, R. E., K. Roberts, E. Wiltshaw, J. Mundy, and V. R. McCready. 1989. The clinical correlates of serum CA125 in 169 patients with epithelial ovarian carcinoma. British Journal of Cancer 60(4):634-637.
Helzlsouer, K. J., A. J. Alberg, G. B. Gordon, C. Longcope, T. L. Bush, S. C. Hoffman, and G. W. Comstock. 1995. Serum gonadotropins and steroid hormones and the development of ovarian cancer. Journal of the American Medical Association 274(24):1926-1930.
Hendriks, Y. M., A. E. de Jong, H. Morreau, C. M. Tops, H. F. Vasen, J. T. Wijnen, M. H. Breuning, and A. H. Brocker-Vriends. 2006. Diagnostic approach and management of Lynch syndrome (hereditary nonpolyposis colorectal carcinoma): A guide for clinicians. CA: A Cancer Journal for Clinicians 56(4):213-225.
Hertlein, L., P. Stieber, A. Kirschenhofer, K. Krocker, D. Nagel, M. Lenhard, and A. Burges. 2012. Human epididymis protein 4 (HE4) in benign and malignant diseases. Clinical Chemistry and Laboratory Medicine 50(12):2181-2188.
HHS (U.S. Department of Health and Human Services). 2013. Increase the proportion of women with a family history of breast and/or ovarian cancer who receive genetic counseling. http://www.healthypeople.gov/2020/topics-objectives/topic/genomics/objectives (accessed September 15, 2015).
Higgins, R. V., J. R. van Nagell, Jr., C. H. Woods, E. A. Thompson, and R. J. Kryscio. 1990. Interobserver variation in ovarian measurements using transvaginal sonography. Gynecologic Oncology 39(1):69-71.
Hildebrand, J. S., S. M. Gapstur, M. M. Gaudet, P. T. Campbell, and A. V. Patel. 2015. Moderate-to-vigorous physical activity and leisure-time sitting in relation to ovarian cancer risk in a large prospective U.S. cohort. Cancer Causes and Control 26(11):1691-1697.
Hising, C., I. M. Anjegard, and N. Einhorn. 1991. Clinical relevance of the CA 125 assay in monitoring of ovarian cancer patients. American Journal of Clinical Oncology 14(2):111-114.
Howlader, N., A. M. Noone, M. Krapcho, J. Garshell, D. Miller, S. F. Altekruse, C. L. Kosary, M. Yu, J. Ruhl, Z. Tatalovich, A. Mariotto, D. R. Lewis, H. S. Chen, E. J. Feuer, and K. A. Cronin. 2015. SEER cancer statistics review, 1975–2011. Bethesda, MD: National Cancer Institute.
Hua, S., C. C. Williams, L. M. Dimapasoc, G. S. Ro, S. Ozcan, S. Miyamoto, C. B. Lebrilla, H. J. An, and G. S. Leiserowitz. 2013. Isomer-specific chromatographic profiling yields highly sensitive and specific potential N-glycan biomarkers for epithelial ovarian cancer. Journal of Chromatography A 1279:58-67.
Huang, J., W. Hu, and A. K. Sood. 2010. Prognostic biomarkers in ovarian cancer. Cancer Biomarkers 8(4-5):231-251.
Huang, T., A. H. Eliassen, S. E. Hankinson, O. I. Okereke, L. D. Kubzansky, M. Wang, E. M. Poole, J. E. Chavarro, and S. S. Tworoger. 2015a. A prospective study of leisure-time physical activity and risk of incident epithelial ovarian cancer: Impact by menopausal status. International Journal of Cancer (Epub ahead of print).
Huang, T., E. M. Poole, O. I. Okereke, L. D. Kubzansky, A. H. Eliassen, A. K. Sood, M. Wang, and S. S. Tworoger. 2015b. Depression and risk of epithelial ovarian cancer: Results from two large prospective cohort studies. Gynecologic Oncology (Epub ahead of print).
Hull, K. L., and S. Harvey. 2001. Growth hormone: Roles in female reproduction. Journal of Endocrinology 168(1):1-23.
Idahl, A., E. Lundin, M. Jurstrand, U. Kumlin, F. Elgh, N. Ohlson, and U. Ottander. 2011. Chlamydia trachomatis and mycoplasma genitalium plasma antibodies in relation to epithelial ovarian tumors. Infectious Diseases in Obstetrics and Gynecology 2011:824627.
IOM (Institute of Medicine). 2007. Cancer biomarkers: The promises and challenges of improving detection and treatment. Washington, DC: The National Academies Press.
Jacobs, I. J., and U. Menon. 2004. Progress and challenges in screening for early detection of ovarian cancer. Molecular and Cellular Proteomics 3(4):355-366.
Jacobs, I., D. Oram, J. Fairbanks, J. Turner, C. Frost, and J. G. Grudzinskas. 1990. A risk of malignancy index incorporating CA 125, ultrasound and menopausal status for the accurate preoperative diagnosis of ovarian cancer. British Journal of Obstetrics and Gynaecology 97(10):922-929.
Jacobs, I. J., S. J. Skates, N. MacDonald, U. Menon, A. N. Rosenthal, A. P. Davies, R. Woolas, A. R. Jeyarajah, K. Sibley, D. G. Lowe, and D. H. Oram. 1999. Screening for ovarian cancer: A pilot randomised controlled trial. Lancet 353(9160):1207-1210.
Jacobs, I. J., U. Menon, A. Ryan, A. Gentry-Maharaj, M. Burnell, et al. 2016. Ovarian cancer screening and mortality in the U.K. collaborative trial of ovarian cancer screening (UKCTOCS): A randomised controlled trial. Lancet 387(10022):945-956.
Jacoby, V. L., D. Grady, J. Wactawski-Wende, J. E. Manson, M. A. Allison, M. Kuppermann, G. E. Sarto, J. Robbins, L. Phillips, L. W. Martin, M. J. O’Sullivan, R. Jackson, R. J. Rodabough, and M. L. Stefanick. 2011. Oophorectomy vs ovarian conservation with hysterectomy: Cardiovascular disease, hip fracture, and cancer in the Women’s Health Initiative observational study. Archives of Internal Medicine 171(8):760-768.
Jensen, A., H. Sharif, K. Frederiksen, and S. K. Kjaer. 2009. Use of fertility drugs and risk of ovarian cancer: Danish population-based cohort study. BMJ (Online) 338(7694):580-582.
Jensen, K. E., C. G. Hannibal, A. Nielsen, A. Jensen, B. Nohr, C. Munk, and S. K. Kjaer. 2008. Social inequality and incidence of and survival from cancer of the female genital organs in a population-based study in Denmark, 1994–2003. European Journal of Cancer 44(14):2003-2017.
Jervis, S., H. Song, A. Lee, E. Dicks, J. Tyrer, P. Harrington, D. F. Easton, I. J. Jacobs, P. P. Pharoah, and A. C. Antoniou. 2014. Ovarian cancer familial relative risks by tumour subtypes and by known ovarian cancer genetic susceptibility variants. Journal of Medical Genetics 51(2):108-113.
Karlan, B. Y., J. Thorpe, K. Watabayashi, C. W. Drescher, M. Palomares, M. B. Daly, P. Paley, P. Hillard, M. R. Andersen, G. Anderson, R. Drapkin, and N. Urban. 2014. Use of CA125 and HE4 serum markers to predict ovarian cancer in elevated-risk women. Cancer Epidemiology, Biomarkers and Prevention 23(7):1383-1393.
Karlsen, N. S., M. A. Karlsen, C. K. Hogdall, and E. V. Hogdall. 2014. HE4 tissue expression and serum HE4 levels in healthy individuals and patients with benign or malignant tumors: A systematic review. Cancer Epidemiology, Biomarkers and Prevention 23(11):2285-2295.
Kelemen, L. E., K. Lawrenson, J. Tyrer, Q. Li, J. M. Lee, et al. 2015. Genome-wide significant risk associations for mucinous ovarian carcinoma. Nature Genetics 47(8):888-897.
Kempers, M. J., R. P. Kuiper, C. W. Ockeloen, P. O. Chappuis, P. Hutter, et al. 2011. Risk of colorectal and endometrial cancers in EPCAM deletion-positive Lynch syndrome: A cohort study. Lancet Oncology 12(1):49-55.
Kiecolt-Glaser, J. K., L. Christian, H. Preston, C. R. Houts, W. B. Malarkey, C. F. Emery, and R. Glaser. 2010. Stress, inflammation, and yoga practice. Psychosomatic Medicine 72(2):113-121.
Kinkel, K., H. Hricak, Y. Lu, K. Tsuda, and R. A. Filly. 2000. U.S. characterization of ovarian masses: A meta-analysis. Radiology 217(3):803-811.
Kobayashi, E., Y. Ueda, S. Matsuzaki, T. Yokoyama, T. Kimura, K. Yoshino, M. Fujita, T. Kimura, and T. Enomoto. 2012. Biomarkers for screening, diagnosis, and monitoring of ovarian cancer. Cancer Epidemiology, Biomarkers and Prevention 21(11):1902-1912.
Kobayashi, H., Y. Yamada, T. Sado, M. Sakata, S. Yoshida, R. Kawaguchi, S. Kanayama, H. Shigetomi, S. Haruta, Y. Tsuji, S. Ueda, and T. Kitanaka. 2008. A randomized study of screening for ovarian cancer: A multicenter study in Japan. International Journal of Gynecological Cancer 18(3):414-420.
Kolahdooz, F., T. I. Ibiebele, J. C. van der Pols, and P. M. Webb. 2009. Dietary patterns and ovarian cancer risk. American Journal of Clinical Nutrition 89(1):297-304.
Kolahdooz, F., J. C. van der Pols, C. J. Bain, G. C. Marks, M. C. Hughes, D. C. Whiteman, and P. M. Webb for the Australian Cancer Study (Ovarian Cancer) and the Australian Ovarian Cancer Study Group. 2010. Meat, fish, and ovarian cancer risk: Results from 2 Australian case-control studies, a systematic review, and meta-analysis. American Journal of Clinical Nutrition 91(6):1752-1763.
Koushik, A., D. J. Hunter, D. Spiegelman, K. E. Anderson, A. A. Arslan, et al. 2005. Fruits and vegetables and ovarian cancer risk in a pooled analysis of 12 cohort studies. Cancer Epidemiology, Biomarkers and Prevention 14(9):2160-2167.
Koushik, A., D. J. Hunter, D. Spiegelman, K. E. Anderson, J. E. Buring, et al. 2006. Intake of the major carotenoids and the risk of epithelial ovarian cancer in a pooled analysis of 10 cohort studies. International Journal of Cancer 119(9):2148-2154.
Kuchenbaecker, K. B., S. J. Ramus, J. Tyrer, A. Lee, H. C. Shen, et al. 2015. Identification of six new susceptibility loci for invasive epithelial ovarian cancer. Nature Genetics 47(2):164-171.
Lacey, J. V., Jr., M. E. Sherman, P. Hartge, A. Schatzkin, and C. Schairer. 2004. Medication use and risk of ovarian carcinoma: A prospective study. International Journal of Cancer 108(2):281-286.
Lancaster, J. M., C. B. Powell, L. M. Chen, D. L. Richardson, and the Society of Gynecologic Oncology Clinical Practice Committee. 2015. Society of Gynecologic Oncology statement on risk assessment for inherited gynecologic cancer predispositions. Gynecologic Oncology 136(1):3-7.
Leblanc, E., F. Narducci, I. Farre, J. P. Peyrat, S. Taieb, C. Adenis, and P. Vennin. 2011. Radical fimbriectomy: A reasonable temporary risk-reducing surgery for selected women with a germ line mutation of BRCA 1 or 2 genes? Rationale and preliminary development. Gynecologic Oncology 121(3):472-476.
Lee, C. H., S. Subramanian, A. H. Beck, I. Espinosa, J. Senz, S. X. Zhu, D. Huntsman, M. van de Rijn, and C. B. Gilks. 2009. MicroRNA profiling of BRCA1/2 mutation-carrying and non-mutation-carrying high-grade serous carcinomas of ovary. PLoS ONE 4(10):e7314.
Li, K., A. Hüsing, R. T. Fortner, A. Tjønneland, L. Hansen, et al. 2015. An epidemiologic risk prediction model for ovarian cancer in Europe: The EPIC study. British Journal of Cancer 112(7):1257-1265.
Lin, H. W., Y. Y. Tu, S. Y. Lin, W. J. Su, W. L. Lin, W. Z. Lin, S. C. Wu, and Y. L. Lai. 2011. Risk of ovarian cancer in women with pelvic inflammatory disease: A population-based study. Lancet Oncology 12(9):900-904.
Liu, J., W. Tang, L. Sang, X. Dai, D. Wei, Y. Luo, and J. Zhang. 2015. Milk, yogurt, and lactose intake and ovarian cancer risk: A meta-analysis. Nutrition and Cancer 67(1):68-72.
Lu, K. H., and M. Daniels. 2013. Endometrial and ovarian cancer in women with Lynch syndrome: Update in screening and prevention. Familial Cancer 12(2):273-277.
Lu, K. H., M. E. Wood, M. Daniels, C. Burke, J. Ford, N. D. Kauff, W. Kohlmann, N. M. Lindor, T. M. Mulvey, L. Robinson, W. S. Rubinstein, E. M. Stoffel, C. Snyder, S. Syngal, J. K. Merrill, D. S. Wollins, and K. S. Hughes. 2014. American Society of Clinical Oncology expert statement: Collection and use of a cancer family history for oncology providers. Journal of Clinical Oncology 32(8):833-840.
Luan, N. N., Q. J. Wu, T. T. Gong, E. Vogtmann, Y. L. Wang, and B. Lin. 2013. Breastfeeding and ovarian cancer risk: A meta-analysis of epidemiologic studies. American Journal of Clinical Nutrition 98(4):1020-1031.
Lueth, N. A., K. E. Anderson, L. J. Harnack, J. A. Fulkerson, and K. Robien. 2008. Coffee and caffeine intake and the risk of ovarian cancer: The Iowa Women’s Health Study. Cancer Causes and Control 19(10):1365-1372.
Lukanova, A., and R. Kaaks. 2005. Endogenous hormones and ovarian cancer: Epidemiology and current hypotheses. Cancer Epidemiology, Biomarkers and Prevention 14(1):98-107.
Lutz, A. M., J. K. Willmann, C. W. Drescher, P. Ray, F. V. Cochran, N. Urban, and S. S. Gambhir. 2011. Early diagnosis of ovarian carcinoma: Is a solution in sight? Radiology 259(2):329-345.
Lynch, H. T., M. J. Casey, C. L. Snyder, C. Bewtra, J. F. Lynch, M. Butts, and A. K. Godwin. 2009. Hereditary ovarian carcinoma: Heterogeneity, molecular genetics, pathology, and management. Molecular Oncology 3(2):97-137.
Madsen, C., L. Baandrup, C. Dehlendorff, and S. K. Kjaer. 2015. Tubal ligation and salpingectomy and the risk of epithelial ovarian cancer and borderline ovarian tumors: A nationwide case-control study. Acta Obstetricia et Gynecologica Scandinavica 94(1):86-94.
Malutan, A. M., T. Drugan, R. Ciortea, R. F. Mocan-Hognogi, C. Bucuri, M. P. Rada, and D. Mihu. 2015a. Serum anti-inflammatory cytokines for the evaluation of inflammatory status in endometriosis. Journal of Research in Medical Sciences 20(7):668-674.
Malutan, A. M., T. Drugan, N. Costin, R. Ciortea, C. Bucuri, M. P. Rada, and D. Mihu. 2015b. Pro-inflammatory cytokines for evaluation of inflammatory status in endometriosis. Central European Journal of Immunology 40(1):96-102.
Manegold-Brauer, G., A. K. Bellin, S. Tercanli, O. Lapaire, and V. Heinzelmann-Schwarz. 2014. The special role of ultrasound for screening, staging and surveillance of malignant ovarian tumors: Distinction from other methods of diagnostic imaging. Archives of Gynecology and Obstetrics 289(3):491-498.
McAlpine, J. N., G. E. Hanley, M. M. Woo, A. A. Tone, N. Rozenberg, K. D. Swenerton, C. B. Gilks, S. J. Finlayson, D. G. Huntsman, D. M. Miller, and Ovarian Cancer Research Program of British Columbia. 2014. Opportunistic salpingectomy: Uptake, risks, and complications of a regional initiative for ovarian cancer prevention. American Journal of Obstetrics and Gynecology 210(5):471.e1-e11.
Menon, U., S. J. Skates, S. Lewis, A. N. Rosenthal, B. Rufford, K. Sibley, N. Macdonald, A. Dawnay, A. Jeyarajah, R. C. Bast, Jr., D. Oram, and I. J. Jacobs. 2005. Prospective study using the risk of ovarian cancer algorithm to screen for ovarian cancer. Journal of Clinical Oncology 23(31):7919-7926.
Menon, U., M. Griffin, and A. Gentry-Maharaj. 2014. Ovarian cancer screening—Current status, future directions. Gynecologic Oncology 132(2):490-495.
Menon, U., A. Ryan, J. Kalsi, A. Gentry-Maharaj, A. Dawnay, et al. 2015. Risk algorithm using serial biomarker measurements doubles the number of screen-detected cancers compared with a single-threshold rule in the United Kingdom Collaborative Trial of Ovarian Cancer Screening. Journal of Clinical Oncology 33(18):2062-2071.
Merritt, M. A., M. De Pari, A. F. Vitonis, L. J. Titus, D. W. Cramer, and K. L. Terry. 2013. Reproductive characteristics in relation to ovarian cancer risk by histologic pathways. Human Reproduction 28(5):1406-1417.
Merritt, M. A., E. M. Poole, S. E. Hankinson, W. C. Willett, and S. S. Tworoger. 2014a. Dairy food and nutrient intake in different life periods in relation to risk of ovarian cancer. Cancer Causes and Control 25(7):795-808.
Merritt, M. A., E. Riboli, E. Weiderpass, K. K. Tsilidis, K. Overvad, et al. 2014b. Dietary fat intake and risk of epithelial ovarian cancer in the European Prospective Investigation into Cancer and Nutrition. Cancer Epidemiology 38(5):528-537.
Miki, Y., J. Swensen, D. Shattuck-Eidens, P. A. Futreal, K. Harshman, et al. 1994. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 266(5182): 66-71.
Miralles, C., M. Orea, P. Espana, M. Provencio, A. Sanchez, B. Cantos, R. Cubedo, E. Carcereny, F. Bonilla, and T. Gea. 2003. Cancer antigen 125 associated with multiple benign and malignant pathologies. Annals of Surgical Oncology 10(2):150-154.
Moore, R. G., A. K. Brown, M. C. Miller, S. Skates, W. J. Allard, T. Verch, M. Steinhoff, G. Messerlian, P. DiSilvestro, C. O. Granai, and R. C. Bast, Jr. 2008. The use of multiple novel tumor biomarkers for the detection of ovarian carcinoma in patients with a pelvic mass. Gynecologic Oncology 108(2):402-408.
Moore, R. G., D. S. McMeekin, A. K. Brown, P. DiSilvestro, M. C. Miller, W. J. Allard, W. Gajewski, R. Kurman, R. C. Bast, Jr., and S. J. Skates. 2009. A novel multiple marker bioassay utilizing HE4 and CA125 for the prediction of ovarian cancer in patients with a pelvic mass. Gynecologic Oncology 112(1):40-46.
Moore, R. G., M. Jabre-Raughley, A. K. Brown, K. M. Robison, M. C. Miller, W. J. Allard, R. J. Kurman, R. C. Bast, and S. J. Skates. 2010. Comparison of a novel multiple marker assay vs the risk of malignancy index for the prediction of epithelial ovarian cancer in patients with a pelvic mass. American Journal of Obstetrics and Gynecology 203(3):228.e1-e6.
Moore, R. G., M. C. Miller, P. Disilvestro, L. M. Landrum, W. Gajewski, J. J. Ball, and S. J. Skates. 2011. Evaluation of the diagnostic accuracy of the risk of ovarian malignancy algorithm in women with a pelvic mass. Obstetrics and Gynecology 118(2 Pt 1):280-288.
Moore, R. G., E. K. Hill, T. Horan, N. Yano, K. Kim, S. MacLaughlan, G. Lambert-Messerlian, Y. D. Tseng, J. F. Padbury, M. C. Miller, T. S. Lange, and R. K. Singh. 2014. HE4 (WFDC2) gene overexpression promotes ovarian tumor growth. Scientific Reports 4:3574.
Moss, E. L., J. Hollingworth, and T. M. Reynolds. 2005. The role of CA125 in clinical practice. Journal of Clinical Pathology 58(3):308-312.
Muller, C. Y. 2010. Doctor, should I get this new ovarian cancer test-OVA1? Obstetrics and Gynecology 116(2 Pt 1):246-247.
Murphy, M. A., B. Trabert, H. P. Yang, Y. Park, L. A. Brinton, P. Hartge, M. E. Sherman, A. Hollenbeck, and N. Wentzensen. 2012. Non-steroidal anti-inflammatory drug use and ovarian cancer risk: Findings from the NIH-AARP Diet and Health Study and systematic review. Cancer Causes and Control 23(11):1839-1852.
Narod, S. A., P. Sun, P. Ghadirian, H. Lynch, C. Isaacs, J. Garber, B. Weber, B. Karlan, D. Fishman, B. Rosen, N. Tung, and S. L. Neuhausen. 2001. Tubal ligation and risk of ovarian cancer in carriers of BRCA1 or BRCA2 mutations: A case-control study. Lancet 357(9267):1467-1470.
NCCN (National Comprehensive Cancer Network). 2015. Genetic/familial high-risk assessment: Breast and ovarian. Fort Washington, PA: NCCN.
NCI (National Cancer Institute). 2015. NCI dictionary of cancer terms. http://www.cancer.gov/publications/dictionaries/cancer-terms (accessed September 16, 2015).
Nelson, H. D., L. H. Huffman, R. Fu, E. L. Harris, and U.S. Preventive Services Task Force. 2005. Genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility: Systematic evidence review for the U.S. Preventive Services Task Force. Annals of Internal Medicine 143(5):362-379.
Nelson, H. D., K. Tyne, A. Naik, C. Bougatsos, B. K. Chan, L. Humphrey, and U.S. Preventive Services Task Force. 2009. Screening for breast cancer: An update for the U.S. Preventive Services Task Force. Annals of Internal Medicine 151(10):727-737, W237-W242.
Nelson, H. D., M. Pappas, B. Zakher, J. P. Mitchell, L. Okinaka-Hu, and R. Fu. 2014. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer in women: A systematic review to update the U.S. Preventive Services Task Force recommendation. Annals of Internal Medicine 160(4):255-266.
Ness, R. B., J. A. Grisso, C. Cottreau, J. Klapper, R. Vergona, J. E. Wheeler, M. Morgan, and J. J. Schlesselman. 2000. Factors related to inflammation of the ovarian epithelium and risk of ovarian cancer. Epidemiology 11(2):111-117.
Ness, R. B., M. T. Goodman, C. Shen, and R. C. Brunham. 2003. Serologic evidence of past infection with chlamydia trachomatis, in relation to ovarian cancer. Journal of Infectious Diseases 187(7):1147-1152.
Ness, R. B., C. Shen, D. Bass, C. Jackson, K. Moysich, R. Edwards, and R. C. Brunham. 2008. Chlamydia trachomatis serology in women with and without ovarian cancer. Infectious Diseases in Obstetrics and Gynecology 2008:219672.
Ni, X., J. Ma, Y. Zhao, Y. Wang, and S. Wang. 2013. Meta-analysis on the association between non-steroidal anti-inflammatory drug use and ovarian cancer. British Journal of Clinical Pharmacology 75(1):26-35.
Niloff, J. M., R. C. Knapp, E. Schaetzl, C. Reynolds, and R. C. Bast, Jr. 1984. CA125 antigen levels in obstetric and gynecologic patients. Obstetrics and Gynecology 64(5):703-707.
Nilsson, M. B., G. Armaiz-Pena, R. Takahashi, Y. G. Lin, J. Trevino, Y. Li, N. Jennings, J. Arevalo, S. K. Lutgendorf, G. E. Gallick, A. M. Sanguino, G. Lopez-Berestein, S. W. Cole, and A. K. Sood. 2007. Stress hormones regulate interleukin-6 expression by human ovarian carcinoma cells through a Src-dependent mechanism. Journal of Biological Chemistry 282(41):29919-29926.
Ose, J., R. T. Fortner, S. Rinaldi, H. Schock, K. Overvad, et al. 2015a. Endogenous androgens and risk of epithelial invasive ovarian cancer by tumor characteristics in the European Prospective Investigation into Cancer and Nutrition. International Journal of Cancer 136(2):399-410.
Ose, J., H. Schock, A. Tjønneland, L. Hansen, K. Overvad, et al. 2015b. Inflammatory markers and risk of epithelial ovarian cancer by tumor subtypes: The EPIC cohort. Cancer Epidemiology, Biomarkers and Prevention 24(6):951-961.
Pal, T., J. Permuth-Wey, J. A. Betts, J. P. Krischer, J. Fiorica, H. Arango, J. LaPolla, M. Hoffman, M. A. Martino, K. Wakeley, G. Wilbanks, S. Nicosia, A. Cantor, and R. Sutphen. 2005. BRCA1 and BRCA2 mutations account for a large proportion of ovarian carcinoma cases. Cancer 104(12):2807-2816.
Parazzini, F., C. La Vecchia, E. Negri, S. Moroni, D. dal Pino, and L. Fedele. 1996. Pelvic inflammatory disease and risk of ovarian cancer. Cancer Epidemiology, Biomarkers and Prevention 5(8):667-669.
Parker, W. H., M. S. Broder, E. Chang, D. Feskanich, C. Farquhar, Z. Liu, D. Shoupe, J. S. Berek, S. Hankinson, and J. E. Manson. 2009. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstetrics and Gynecology 113(5):1027-1037.
Patel, A. V., C. Rodriguez, A. L. Pavluck, M. J. Thun, and E. E. Calle. 2006. Recreational physical activity and sedentary behavior in relation to ovarian cancer risk in a large cohort of U.S. women. American Journal of Epidemiology 163(8):709-716.
Pearce, C. L., C. Templeman, M. A. Rossing, A. Lee, A. M. Near, et al. 2012. Association between endometriosis and risk of histological subtypes of ovarian cancer: A pooled analysis of case-control studies. The Lancet Oncology 13(4):385-394.
Permuth-Wey, J., and T. Sellers. 2009. Epidemiology of ovarian cancer. In Cancer epidemiology, Vol. 472, edited by M. Verma. New York: Humana Press. Pp. 413-437.
Permuth-Wey, J., K. Lawrenson, H. C. Shen, A. Velkova, J. P. Tyrer, et al. 2013. Identification and molecular characterization of a new ovarian cancer susceptibility locus at 17q21.31. Nature Communications 4:1627.
Permuth-Wey, J., A. Besharat, and T. Sellers. 2014. Epidemiology of ovarian cancer: An update. In Advances in diagnosis and management of ovarian cancer, edited by S. A. Farghaly. New York: Springer Science and Business Media. Pp. 1-21.
Pfeiffer, R. M., Y. Park, A. R. Kreimer, J. V. Lacey, Jr., D. Pee, R. T. Greenlee, S. S. Buys, A. Hollenbeck, B. Rosner, M. H. Gail, and P. Hartge. 2013. Risk prediction for breast, endometrial, and ovarian cancer in white women aged 50 y or older: Derivation and validation from population-based cohort studies. PLoS Medicine 10(7):e1001492.
Pharoah, P. D. P., Y. Y. Tsai, S. J. Ramus, C. M. Phelan, E. L. Goode, et al. 2013. GWAS meta-analysis and replication identifies three new susceptibility loci for ovarian cancer. Nature Genetics 45(4):362-370.
Phillips, S. M., K. W. Dodd, J. Steeves, J. McClain, C. M. Alfano, and E. McAuley. 2015. Physical activity and sedentary behavior in breast cancer survivors: New insight into activity patterns and potential intervention targets. Gynecologic Oncology 138(2):398-404.
Pinheiro, S. P., S. S. Tworoger, D. W. Cramer, B. A. Rosner, and S. E. Hankinson. 2009. Use of nonsteroidal antiinflammatory agents and incidence of ovarian cancer in 2 large prospective cohorts. American Journal of Epidemiology 169(11):1378-1387.
Pinsky, P. F., C. Zhu, S. J. Skates, A. Black, E. Partridge, S. S. Buys, and C. D. Berg. 2013. Potential effect of the risk of ovarian cancer algorithm (ROCA) on the mortality outcome of the Prostate, Lung, Colorectal and Ovarian (PLCO) trial. International Journal of Cancer 132(9):2127-2133.
Poole, E. M., I. M. Lee, P. M. Ridker, J. E. Buring, S. E. Hankinson, and S. S. Tworoger. 2013. A prospective study of circulating C-reactive protein, interleukin-6, and tumor necrosis factor α receptor 2 levels and risk of ovarian cancer. American Journal of Epidemiology 178(8):1256-1264.
Powell, C. B., R. Littell, E. Hoodfar, F. Sinclair, and A. Pressman. 2013a. Does the diagnosis of breast or ovarian cancer trigger referral to genetic counseling? International Journal of Gynecological Cancer 23(3):431-436.
Powell, N. D., A. J. Tarr, and J. F. Sheridan. 2013b. Psychosocial stress and inflammation in cancer. Brain, Behavior, and Immunity 30(Suppl):S41-S47.
Prentice, R. L., C. A. Thomson, B. Caan, F. A. Hubbell, G. L. Anderson, S. A. Beresford, M. Pettinger, D. S. Lane, L. Lessin, S. Yasmeen, B. Singh, J. Khandekar, J. M. Shikany, S. Satterfield, and R. T. Chlebowski. 2007. Low-fat dietary pattern and cancer incidence in the Women’s Health Initiative dietary modification randomized controlled trial. Journal of the National Cancer Institute 99(20):1534-1543.
Prizment, A. E., A. R. Folsom, and K. E. Anderson. 2010. Nonsteroidal anti-inflammatory drugs and risk for ovarian and endometrial cancers in the Iowa Women’s Health Study. Cancer Epidemiology, Biomarkers and Prevention 19(2):435-442.
Rahman, N. 2014. Realizing the promise of cancer predisposition genes. Nature 505(7483): 302-308.
Ramus, S. J., C. Kartsonaki, S. A. Gayther, P. D. P. Pharoah, O. M. Sinilnikova, et al. 2011. Genetic variation at 9p22.2 and ovarian cancer risk for BRCA1 and BRCA2 mutation carriers. Journal of the National Cancer Institute 103(2):105-116.
Ramus, S. J., A. C. Antoniou, K. B. Kuchenbaecker, P. Soucy, J. Beesley, X. Chen, L. McGuffog, O. M. Sinilnikova, S. Healey, D. Barrowdale, A. Lee, and M. Thomassen. 2012. Ovarian cancer susceptibility alleles and risk of ovarian cancer in BRCA1 and BRCA2 mutation carriers. Human Mutation 33(4):690-702.
Rasmussen, C. B., M. T. Faber, A. Jensen, E. Hogdall, C. Hogdall, J. Blaakaer, and S. K. Kjaer. 2013. Pelvic inflammatory disease and risk of invasive ovarian cancer and ovarian borderline tumors. Cancer Causes and Control 24(7):1459-1464.
Rebbeck, T. R., N. Mitra, F. Wan, O. M. Sinilnikova, S. Healey, et al. 2015. Association of type and location of BRCA1 and BRCA2 mutations with risk of breast and ovarian cancer. Journal of the American Medical Association 313(13):1347-1361.
Renehan, A. G., M. Zwahlen, and M. Egger. 2015. Adiposity and cancer risk: New mechanistic insights from epidemiology. Nature Reviews: Cancer 15(8):484-498.
Rice, M. S., M. A. Murphy, and S. S. Tworoger. 2012. Tubal ligation, hysterectomy and ovarian cancer: A meta-analysis. Journal of Ovarian Research 5:13.
Rice, M. S., M. A. Murphy, A. F. Vitonis, D. W. Cramer, L. J. Titus, S. S. Tworoger, and K. L. Terry. 2013. Tubal ligation, hysterectomy and epithelial ovarian cancer in the New England Case-Control Study. International Journal of Cancer 133(10):2415-2421.
Risch, H. A. 1998. Hormonal etiology of epithelial ovarian cancer, with a hypothesis concerning the role of androgens and progesterone. Journal of the National Cancer Institute 90(23):1774-1786.
Risch, H. A., and G. R. Howe. 1995. Pelvic inflammatory disease and the risk of epithelial ovarian cancer. Cancer Epidemiology, Biomarkers and Prevention 4(5):447-451.
Risch, H. A., J. R. McLaughlin, D. E. Cole, B. Rosen, L. Bradley, I. Fan, J. Tang, S. Li, S. Zhang, P. A. Shaw, and S. A. Narod. 2006. Population BRCA1 and BRCA2 mutation frequencies and cancer penetrances: A kin-cohort study in Ontario, Canada. Journal of the National Cancer Institute 98(23):1694-1706.
Risum, S., C. Hogdall, A. Loft, A. K. Berthelsen, E. Hogdall, L. Nedergaard, L. Lundvall, and S. A. Engelholm. 2007. The diagnostic value of PET/CT for primary ovarian cancer—A prospective study. Gynecologic Oncology 105(1):145-149.
Robson, M. E., A. R. Bradbury, B. Arun, S. M. Domchek, J. M. Ford, H. L. Hampel, S. M. Lipkin, S. Syngal, D. S. Wollins, and N. M. Lindor. 2015. American Society of Clinical Oncology policy statement update: Genetic and genomic testing for cancer susceptibility. Journal of Clinical Oncology (Epub ahead of print).
Romaguera, D., A. C. Vergnaud, P. H. Peeters, C. H. Van Gils, D. S. M. Chan, et al. 2012. Is concordance with World Cancer Research Fund/American Institute for Cancer Research guidelines for cancer prevention related to subsequent risk of cancer? Results from the EPIC study. American Journal of Clinical Nutrition 96(1):150-163.
Rosner, B. A., G. A. Colditz, P. M. Webb, and S. E. Hankinson. 2005. Mathematical models of ovarian cancer incidence. Epidemiology 16(4):508-515.
Rossi, M., E. Negri, P. Lagiou, R. Talamini, L. Dal Maso, M. Montella, S. Franceschi, and C. La Vecchia. 2008. Flavonoids and ovarian cancer risk: A case-control study in Italy. International Journal of Cancer 123(4):895-898.
Rossi, M., C. Bosetti, E. Negri, P. Lagiou, and C. La Vecchia. 2010. Flavonoids, proanthocyanidins, and cancer risk: A network of case-control studies from Italy. Nutrition and Cancer 62(7):871-877.
Rowlands, I. J., C. M. Nagle, A. B. Spurdle, P. M. Webb, Australian National Endometrial Cancer Study Group, and Australian Ovarian Cancer Study Group. 2011. Gynecological conditions and the risk of endometrial cancer. Gynecologic Oncology 123(3):537-541.
Rutter, J. L., S. Wacholder, A. Chetrit, F. Lubin, J. Menczer, S. Ebbers, M. A. Tucker, J. P. Struewing, and P. Hartge. 2003. Gynecologic surgeries and risk of ovarian cancer in women with BRCA1 and BRCA2 Ashkenazi founder mutations: An Israeli population-based case-control study. Journal of the National Cancer Institute 95(14):1072-1078.
Salamanca, A., and E. Beltran. 1995. Subendometrial contractility in menstrual phase visualized by transvaginal sonography in patients with endometriosis. Fertility and Sterility 64(1):193-195.
Sampson, J. A. 1927. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. American Journal of Obstetrics and Gynecology 14:422-469.
Sanfilippo, J. S., N. G. Wakim, K. N. Schikler, and M. A. Yussman. 1986. Endometriosis in association with uterine anomaly. American Journal of Obstetrics and Gynecology 154(1):39-43.
Schildkraut, J. M., B. Calingaert, P. A. Marchbanks, P. G. Moorman, and G. C. Rodriguez. 2002. Impact of progestin and estrogen potency in oral contraceptives on ovarian cancer risk. Journal of the National Cancer Institute 94(1):32-38.
Schock, H., E. Lundin, M. Vaarasmaki, K. Grankvist, A. Fry, J. F. Dorgan, E. Pukkala, M. Lehtinen, H. M. Surcel, and A. Lukanova. 2014a. Anti-Müllerian hormone and risk of invasive serous ovarian cancer. Cancer Causes and Control 25(5):583-589.
Schock, H., H. M. Surcel, A. Zeleniuch-Jacquotte, K. Grankvist, H. A. Lakso, R. T. Fortner, R. Kaaks, E. Pukkala, M. Lehtinen, P. Toniolo, and E. Lundin. 2014b. Early pregnancy sex steroids and maternal risk of epithelial ovarian cancer. Endocrine-Related Cancer 21(6):831-844.
Schock, H., R. T. Fortner, H. M. Surcel, K. Grankvist, E. Pukkala, M. Lehtinen, and E. Lundin. 2015. Early pregnancy IGF-I and placental GH and risk of epithelial ovarian cancer: A nested case-control study. International Journal of Cancer 137(2):439-447.
Schoen, R. E., P. F. Pinsky, J. L. Weissfeld, L. A. Yokochi, T. Church, et al. 2012. Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. New England Journal of Medicine 366(25):2345-2357.
Schouten, L. J., C. Rivera, D. J. Hunter, D. Spiegelman, H. O. Adami, et al. 2008. Height, body mass index, and ovarian cancer: A pooled analysis of 12 cohort studies. Cancer Epidemiology, Biomarkers and Prevention 17(4):902-912.
Schrader, K. A., J. Hurlburt, S. E. Kalloger, S. Hansford, S. Young, D. G. Huntsman, C. B. Gilks, and J. N. McAlpine. 2012. Germline BRCA1 and BRCA2 mutations in ovarian cancer: Utility of a histology-based referral strategy. Obstetrics and Gynecology 120(2 Pt 1):235-240.
Schummer, M., W. V. Ng, R. E. Bumgarner, P. S. Nelson, B. Schummer, D. W. Bednarski, L. Hassell, R. L. Baldwin, B. Y. Karlan, and L. Hood. 1999. Comparative hybridization of an array of 21,500 ovarian cDNAs for the discovery of genes overexpressed in ovarian carcinomas. Gene 238(2):375-385.
Setiawan, V. W., R. K. Matsuno, G. Lurie, L. R. Wilkens, M. E. Carney, B. E. Henderson, L. N. Kolonel, and M. T. Goodman. 2012. Use of nonsteroidal anti-inflammatory drugs and risk of ovarian and endometrial cancer: The multiethnic cohort. Cancer Epidemiology, Biomarkers and Prevention 21(9):1441-1449.
Setiawan, V. W., H. P. Yang, M. C. Pike, S. E. McCann, H. Yu, et al. 2013. Type I and II endometrial cancers: Have they different risk factors? Journal of Clinical Oncology 31(20):2607-2618.
SGO (Society of Gynecologic Oncology). 2013. SGO clinical practice statement: Salpingectomy for ovarian cancer prevention. https://www.sgo.org/clinical-practice/guidelines/sgo-clinical-practice-statement-salpingectomy-for-ovarian-cancer-prevention (accessed October 1, 2015).
Sharma, A., S. Apostolidou, M. Burnell, S. Campbell, M. Habib, et al. 2012. Risk of epithelial ovarian cancer in asymptomatic women with ultrasound-detected ovarian masses: A prospective cohort study within the U.K. collaborative trial of ovarian cancer screening (UKCTOCS). Ultrasound in Obstetrics and Gynecology 40(3):338-344.
Shen, C. C., A. C. Yang, J. H. Hung, L. Y. Hu, and S. J. Tsai. 2015. A nationwide population-based retrospective cohort study of the risk of uterine, ovarian and breast cancer in women with polycystic ovary syndrome. Oncologist 20(1):45-49.
Sherman, M. E., M. Piedmonte, P. L. Mai, O. B. Ioffe, B. M. Ronnett, et al. 2014. Pathologic findings at risk-reducing salpingo-oophorectomy: Primary results from gynecologic oncology group trial gog-0199. Journal of Clinical Oncology 32(29):3275-3283.
Shu, X. O., L. A. Brinton, Y. T. Gao, and J. M. Yuan. 1989. Population-based case-control study of ovarian cancer in Shanghai. Cancer Research 49(13):3670-3674.
Shulman, L. P. 2010. Hereditary breast and ovarian cancer (HBOC): Clinical features and counseling for BRCA1 and BRCA2, Lynch syndrome, Cowden syndrome, and Li-Fraumeni syndrome. Obstetrics and Gynecology Clinics of North America 37(1):109-133.
Shulman, L. P., and J. S. Dungan. 2010. Cancer genetics: Risks and mechanisms of cancer in women with inherited susceptibility to epithelial ovarian cancer. Cancer Treatment and Research 156:69-85.
Sieh, W., S. Salvador, V. McGuire, R. P. Weber, K. L. Terry, et al. 2013. Tubal ligation and risk of ovarian cancer subtypes: A pooled analysis of case-control studies. International Journal of Epidemiology 42(2):579-589.
Skates, S. J., F. J. Xu, Y. H. Yu, K. Sjovall, N. Einhorn, Y. Chang, R. C. Bast, Jr., and R. C. Knapp. 1995. Toward an optimal algorithm for ovarian cancer screening with longitudinal tumor markers. Cancer 76(10 Suppl):2004-2010.
Skates, S. J., D. K. Pauler, and I. J. Jacobs. 2001. Screening based on the risk of cancer calculation from Bayesian hierarchical changepoint and mixture models of longitudinal markers. Journal of the American Statistical Association 96(454):429-439.
Skates, S. J., P. Mai, N. K. Horick, M. Piedmonte, C. W. Drescher, et al. 2011. Large prospective study of ovarian cancer screening in high-risk women: CA125 cut-point defined by menopausal status. Cancer Prevention Research (Philadelphia, PA) 4(9):1401-1408.
Soegaard, M., K. Frederiksen, A. Jensen, E. Hogdall, C. Hogdall, J. Blaakaer, S. J. Ramus, S. A. Gayther, and S. K. Kjaer. 2009. Risk of ovarian cancer in women with first-degree relatives with cancer. Acta Obstetricia et Gynecologica Scandinavica 88(4):449-456.
Song, H., S. J. Ramus, J. Tyrer, K. L. Bolton, A. Gentry-Maharaj, et al. 2009. A genome-wide association study identifies a new ovarian cancer susceptibility locus on 9p22.2. Nature Genetics 41(9):996-1000.
Song, H., M. S. Cicek, E. Dicks, P. Harrington, S. J. Ramus, J. M. Cunningham, B. L. Fridley, J. P. Tyrer, J. Alsop, M. Jimenez-Linan, S. A. Gayther, E. L. Goode, and P. D. Pharoah. 2014. The contribution of deleterious germline mutations in BRCA1, BRCA2 and the mismatch repair genes to ovarian cancer in the population. Human Molecular Genetics 23(17):4703-4709.
Song, Y. J., A. R. Kristal, K. G. Wicklund, K. L. Cushing-Haugen, and M. A. Rossing. 2008. Coffee, tea, colas, and risk of epithelial ovarian cancer. Cancer Epidemiology, Biomarkers and Prevention 17(3):712-716.
Sood, A. K., R. Bhatty, A. A. Kamat, C. N. Landen, L. Han, P. H. Thaker, Y. Li, D. M. Gershenson, S. Lutgendorf, and S. W. Cole. 2006. Stress hormone-mediated invasion of ovarian cancer cells. Clinical Cancer Research 12(2):369-375.
Straif, K., L. Benbrahim-Tallaa, R. Baan, Y. Grosse, B. Secretan, F. E. Ghissassi, V. Bouvard, N. Guha, C. Freeman, L. Galichet, V. Cogliano, and WHO International Agency for Research on Cancer Monograph Working Group. 2009. A review of human carcinogens—part C: Metals, arsenic, dusts, and fibres. Lancet 10:453-454.
Stratton, J. F., P. Pharoah, S. K. Smith, D. Easton, and B. A. Ponder. 1998. A systematic review and meta-analysis of family history and risk of ovarian cancer. British Journal of Obstetrics and Gynaecology 105(5):493-499.
Taylor, D. D., and C. Gercel-Taylor. 2008. MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecologic Oncology 110(1):13-21.
Terry, K. L., S. Karageorgi, Y. B. Shvetsov, M. A. Merritt, G. Lurie, et al. 2013. Genital powder use and risk of ovarian cancer: A pooled analysis of 8,525 cases and 9,859 controls. Cancer Prevention Research (Philadelphia, PA) 6(8):811-821.
Thaker, P. H., L. Y. Han, A. A. Kamat, J. M. Arevalo, R. Takahashi, et al. 2006. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nature Medicine 12(8):939-944.
Thomson, C. A., L. Van Horn, B. J. Caan, A. K. Aragaki, R. T. Chlebowski, J. E. Manson, T. E. Rohan, L. F. Tinker, L. H. Kuller, L. Hou, D. S. Lane, K. C. Johnson, M. Z. Vitolins, and R. L. Prentice. 2014. Cancer incidence and mortality during the intervention and postintervention periods of the Women’s Health Initiative dietary modification trial. Cancer Epidemiology, Biomarkers and Prevention 23(12):2924-2935.
Timmerman, D., P. Schwarzler, W. P. Collins, F. Claerhout, M. Coenen, F. Amant, I. Vergote, and T. H. Bourne. 1999. Subjective assessment of adnexal masses with the use of ultrasonography: An analysis of interobserver variability and experience. Ultrasound in Obstetrics and Gynecology 13(1):11-16.
Trabert, B., E. J. Lamb, B. Scoccia, K. S. Moghissi, C. L. Westhoff, S. Niwa, and L. A. Brinton. 2013. Ovulation-inducing drugs and ovarian cancer risk: Results from an extended follow-up of a large United States infertility cohort. Fertility and Sterility 100(6):1660-1666.
Trabert, B., R. B. Ness, W. H. Lo-Ciganic, M. A. Murphy, E. L. Goode, et al. 2014. Aspirin, nonaspirin nonsteroidal anti-inflammatory drug, and acetaminophen use and risk of invasive epithelial ovarian cancer: A pooled analysis in the Ovarian Cancer Association Consortium. Journal of the National Cancer Institute 106(2).
Tworoger, S. S., K. M. Fairfield, G. A. Colditz, B. A. Rosner, and S. E. Hankinson. 2007. Association of oral contraceptive use, other contraceptive methods, and infertility with ovarian cancer risk. American Journal of Epidemiology 166(8):894-901.
Tworoger, S. S., D. M. Gertig, M. A. Gates, J. L. Hecht, and S. E. Hankinson. 2008a. Caffeine, alcohol, smoking, and the risk of incident epithelial ovarian cancer. Cancer 112(5):1169-1177.
Tworoger, S. S., I. M. Lee, J. E. Buring, and S. E. Hankinson. 2008b. Plasma androgen concentrations and risk of incident ovarian cancer. American Journal of Epidemiology 167(2):211-218.
van Nagell, J. R., Jr., P. D. DePriest, F. R. Ueland, C. P. DeSimone, A. L. Cooper, J. M. McDonald, E. J. Pavlik, and R. J. Kryscio. 2007. Ovarian cancer screening with annual transvaginal sonography: Findings of 25,000 women screened. Cancer 109(9):1887-1896.
Vitonis, A. F., L. Titus-Ernstoff, and D. W. Cramer. 2011. Assessing ovarian cancer risk when considering elective oophorectomy at the time of hysterectomy. Obstetrics and Gynecology 117(5):1042-1050.
Walburn, J., K. Vedhara, M. Hankins, L. Rixon, and J. Weinman. 2009. Psychological stress and wound healing in humans: A systematic review and meta-analysis. Journal of Psychosomatic Research 67(3):253-271.
Walker, J. L., C. B. Powell, L. M. Chen, J. Carter, V. L. Bae Jump, L. P. Parker, M. E. Borowsky, and R. K. Gibb. 2015. Society of Gynecologic Oncology recommendations for the prevention of ovarian cancer. Cancer (Epub ahead of print).
Wallin, A., N. Orsini, and A. Wolk. 2011. Red and processed meat consumption and risk of ovarian cancer: A dose-response meta-analysis of prospective studies. British Journal of Cancer 104(7):1196-1201.
Walsh, T., S. Casadei, M. K. Lee, C. C. Pennil, A. S. Nord, A. M. Thornton, W. Roeb, K. J. Agnew, S. M. Stray, A. Wickramanayake, B. Norquist, K. P. Pennington, R. L. Garcia, M. C. King, and E. M. Swisher. 2011. Mutations in 12 genes for inherited ovarian, fallopian tube, and peritoneal carcinoma identified by massively parallel sequencing. Proceedings of the National Academy of Sciences of the United States of America 108(44):18032-18037.
Wang, L., I. M. Lee, S. M. Zhang, J. B. Blumberg, J. E. Buring, and H. D. Sesso. 2009. Dietary intake of selected flavonols, flavones, and flavonoid-rich foods and risk of cancer in middle-aged and older women. American Journal of Clinical Nutrition 89(3):905-912.
Welsh, J. B., P. P. Zarrinkar, L. M. Sapinoso, S. G. Kern, C. A. Behling, B. J. Monk, D. J. Lockhart, R. A. Burger, and G. M. Hampton. 2001. Analysis of gene expression profiles in normal and neoplastic ovarian tissue samples identifies candidate molecular markers of epithelial ovarian cancer. Proceedings of the National Academy of Sciences of the United States of America 98(3):1176-1181.
Werness, B. A., and G. H. Eltabbakh. 2001. Familial ovarian cancer and early ovarian cancer: Biologic, pathologic, and clinical features. International Journal of Gynecological Pathology 20(1):48-63.
Whittemore, A. S., R. Harris, and J. Itnyre. 1992. Characteristics relating to ovarian cancer risk: Collaborative analysis of 12 U.S. case-control studies. II. Invasive epithelial ovarian cancers in white women. Collaborative Ovarian Cancer Group. American Journal of Epidemiology 136(10):1184-1203.
Woolas, R. P., F. J. Xu, I. J. Jacobs, Y. H. Yu, L. Daly, A. Berchuck, J. T. Soper, D. L. Clarke-Pearson, D. H. Oram, and R. C. Bast, Jr. 1993. Elevation of multiple serum markers in patients with stage I ovarian cancer. Journal of the National Cancer Institute 85(21):1748-1751.
Wooster, R., S. L. Neuhausen, J. Mangion, Y. Quirk, D. Ford, et al. 1994. Localization of a breast cancer susceptibility gene, BRCA2, to chromosome 13q12-13. Science 265(5181): 2088-2090.
Wu, L., Z. Y. Dai, Y. H. Qian, Y. Shi, F. J. Liu, and C. Yang. 2012. Diagnostic value of serum human epididymis protein 4 (HE4) in ovarian carcinoma: A systematic review and meta-analysis. International Journal of Gynecological Cancer 22(7):1106-1112.
Xiao, Q., H. P. Yang, N. Wentzensen, A. Hollenbeck, and C. E. Matthews. 2013. Physical activity in different periods of life, sedentary behavior, and the risk of ovarian cancer in the NIH-AARP Diet and Health Study. Cancer Epidemiology, Biomarkers and Prevention 22(11):2000-2008.
Xie, J., E. M. Poole, K. L. Terry, T. T. Fung, B. A. Rosner, W. C. Willett, and S. S. Tworoger. 2014. A prospective cohort study of dietary indices and incidence of epithelial ovarian cancer. Journal of Ovarian Research 7(1):112.
Yamamoto, Y., H. Oguri, R. Yamada, N. Maeda, S. Kohsaki, and T. Fukaya. 2008. Preoperative evaluation of pelvic masses with combined 18f-fluorodeoxyglucose positron emission tomography and computed tomography. International Journal of Gynaecology and Obstetrics 102(2):124-127.
Yang, H. P., B. Trabert, M. A. Murphy, M. E. Sherman, J. N. Sampson, L. A. Brinton, P. Hartge, A. Hollenbeck, Y. Park, and N. Wentzensen. 2012. Ovarian cancer risk factors by histologic subtypes in the NIH-AARP Diet and Health Study. International Journal of Cancer 131(4):938-948.
Zhang, M., X. Xie, A. H. Lee, and C. W. Binns. 2004. Sedentary behaviours and epithelial ovarian cancer risk. Cancer Causes and Control 15(1):83-89.
Zhang, S., R. Royer, S. Li, J. R. McLaughlin, B. Rosen, H. A. Risch, I. Fan, L. Bradley, P. A. Shaw, and S. A. Narod. 2011. Frequencies of BRCA1 and BRCA2 mutations among 1,342 unselected patients with invasive ovarian cancer. Gynecologic Oncology 121(2):353-357.
Zhang, S., Z. Lu, A. K. Unruh, C. Ivan, K. A. Baggerly, G. A. Calin, Z. Li, R. C. Bast, Jr., and X. F. Le. 2015. Clinically relevant microRNAs in ovarian cancer. Molecular Cancer Research 13(3):393-401.
Zhang, Y. F., Q. Xu, J. Lu, P. Wang, H. W. Zhang, L. Zhou, X. Q. Ma, and Y. H. Zhou. 2014. Tea consumption and the incidence of cancer: A systematic review and meta-analysis of prospective observational studies. European Journal of Cancer Prevention 24(4):353-362.
Zhong, S., L. Chen, M. Lv, T. Ma, X. Zhang, and J. Zhao. 2014. Nonoccupational physical activity and risk of ovarian cancer: A meta-analysis. Tumour Biology 35(11):11065-11073.