Supportive Care Along the Survivorship Trajectory
Most research on ovarian cancer focuses on improving diagnosis and therapy rather than on managing the side effects of treatment or on the psychological and social (psychosocial) issues that women with ovarian cancer face throughout the trajectory of care and survivorship. Addressing the general problem among all cancers, in 2006 the Institute of Medicine (IOM) concluded that “survivorship research is funded at relatively modest levels within both public and private sectors, especially as contrasted to levels of support for treatment-related research” (IOM, 2006, p. 13). While research on therapies that may provide life-saving benefits to patients is crucial, complementary research to support the quality of their survival—their survivorship—is also a critical concern for women and families affected by ovarian cancer.
This chapter reflects on the research base for the issues of survivorship and management (also known as supportive care) that are particularly relevant across the continuum of care for women diagnosed with ovarian cancer and their families. Because much of the research on survivorship is applicable to individuals with many types of cancer (or other serious diseases), a review of all survivorship literature is beyond the scope of this report. Instead, this chapter highlights the overarching issues in survivorship research and supportive care and discusses the evidence base for specific issues for women diagnosed with ovarian cancer.
Women are considered ovarian cancer survivors from the time of diagnosis to the end of life. In 2006, the IOM described cancer survivorship as follows:
Cancer survivorship, as defined in this report, is a distinct phase of the cancer trajectory, but has been relatively neglected in advocacy, education, clinical practice, and research. Raising awareness of the medical and psychosocial needs that may follow cancer treatment will help both survivors and their health care providers to ensure that appropriate assessments are completed and available interventions employed. The constellation of cancer’s long-term and late effects varies by cancer type, treatment modality, and individual characteristics, but there are common patterns of symptoms and conditions that must be recognized so that health and well-being can be improved. (IOM, 2006, p. 150)
The IOM study From Cancer Patient to Cancer Survivor stated that “although some cancer survivors recover with a renewed sense of life and purpose, what has often not been recognized is the toll taken by both cancer and its treatment—on health, functioning, sense of security, and well-being. Long-lasting effects of treatment may be apparent shortly after its completion or arise years later. Personal relationships change and adaptations to routines and work may be needed. Importantly, the survivor’s health care is forever altered” (IOM, 2006, p. 1). That study identified four essential components of survivorship care:
- Prevention of recurrent and new cancers (and other late effects);
- Surveillance for spread, recurrence, or second cancers and assessment of medical and psychosocial late effects;
- Intervention for consequences of cancer treatment (e.g., sexual dysfunction, pain, fatigue, psychological distress, and employment issues); and
- Coordination between specialists and primary care providers to ensure comprehensive care.
For most women with ovarian cancer, survivorship issues do not follow the cancer care continuum (see Figure 1-10 in Chapter 1) in a linear fashion, but rather they are evolving and overlapping from diagnosis to long-term survival (with or without active cancer) or to end-of-life care. The majority of women with ovarian cancer do not fit neatly into the traditional definition of survivorship. They experience survivorship as part of the long-term management of active disease because of the typical pattern of recurrence and multiple cycles of treatment. Consequently, this committee uses the
definition of survivorship proposed by the Society of Gynecologic Oncology (SGO): “the maintenance of physical, social, spiritual, sexual, and economic well-being by addressing short-term and long-term effects of cancer and its treatment” (SGO, 2011, p. 53).
OVERARCHING CHALLENGES IN SURVIVORSHIP RESEARCH FOR OVARIAN CANCER
Much of the research on survivorship that is relevant to women with ovarian cancer is done on individuals with any type of cancer, or else narrowed to women with any type of gynecologic cancer. Most studies that focus on ovarian cancer survivorship do not distinguish between younger and older survivors, who may have markedly different concerns. There also is little information available on the particular supportive care needs that different racial and ethnic groups have, which may contribute to the disparities in care and survivorship that have been observed. Furthermore, studies generally aggregate survivors at different phases of the disease trajectory and usually do not look at survivors by tumor subtype.
In part, these limitations are due to the relatively small numbers of ovarian cancer cases, which limits the power of the research. However, the ovarian cancer subtypes have different outcomes, and studies of women with ovarian cancer in general may not lead to the identification of the unique needs of women living with specific types of ovarian cancer. Finally, most studies on survivorship are retrospective and, therefore, may be biased because only the needs of those who lived long enough to be studied may be reflected in the analyses. Women who had worse prognoses and shorter life expectancies may have had very different concerns that could have been overlooked in a retrospective design.
An essential part of the comprehensive care of cancer patients is palliative care that begins at diagnosis. The World Health Organization describes palliative care as an approach that “improves the quality of life of patients and families who face life-threatening illness, by providing pain and symptom relief, spiritual and psychosocial support from diagnosis to the end of life and bereavement” (WHO, 2002). The IOM identifies palliative care as a component of high-quality cancer care that should span the continuum from diagnosis through end-of-life care (IOM, 2013). The SGO noted that “palliative care significantly improves the quality of life for patients and their families when faced with serious life-threatening illnesses, including advanced gynecologic cancer, and can also substantially reduce the cost of caring for such patients” (Rimel et al., 2015, p. 282). The American
Society of Clinical Oncology (ASCO) recommends that palliative care be included as a routine part of comprehensive cancer care by the year 2020 (Ferris et al., 2009).
Palliative care is not just end-of-life care; the inclusion of palliative care is appropriate at every point in the disease course, including concurrently with cancer therapies, even when the goal of therapy is cure (see Figure 5-1). Women with ovarian cancer can live for years, even with recurrent disease, and can enjoy substantial benefits from supportive care in terms of both symptom relief and life prolongation.
The Value of Integrating Palliative Care
Multiple national and international agencies have recommended that palliative care principles and practice be integrated into the overall care of women with ovarian cancer (Ferris et al., 2009; Levy et al., 2009; Rimel et al., 2015; WHO, 2002). Basic palliative care clinical services delivered by non–palliative care specialists are referred to as primary palliative care (PPC), whereas care provided by palliative care specialists is called specialty palliative care (SPC). PPC assessments and interventions for women with ovarian cancer need to engage both women and their health care
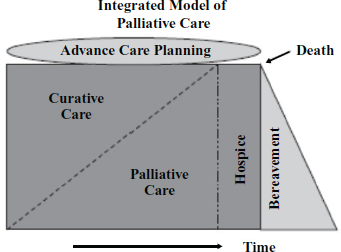
FIGURE 5-1 Integrated model of concurrent palliative and oncology care.
SOURCE: Reprinted with permission. Radwany, S. M., and V. E. Von Gruenigen. 2012. Palliative and end-of-life care for patients with ovarian cancer. Clinical Obstetrics and Gynecology 55(1):173-184.
providers. (Later in this chapter, see more on symptom assessment and self-management.)
The general oncology literature indicates that integrating SPC with usual care improves the control of symptoms and a patient’s quality of life and understanding of the prognosis and also maintains or improves survival (Bakitas et al., 2009; Brumley et al., 2007; Gade et al., 2008; Meyers et al., 2011; Pantilat et al., 2010; Rabow et al., 2004; Temel et al., 2010; Zimmermann et al., 2014). In addition, palliative care integrated with usual care often costs less overall than usual care alone (Brumley et al., 2007; Morrison et al., 2008, 2011). The few studies that have been conducted on the integration of SPC into the care of women with ovarian cancer show similar findings (Lefkowits et al., 2015; Lowery et al., 2013; Nevadunsky et al., 2014; Rugno et al., 2014). For example, one study of women with platinum-resistant ovarian cancer found that the addition of an early referral to SPC was associated with a cost savings of $1,285 per patient (Lowery et al., 2013).
Barriers to Integration of Palliative Care
Barriers to integrating palliative care into oncology care include limited workforce capacity, policy limitations, and misconceptions among both patients and providers (Hui et al., 2010; Lopez-Acevedo et al., 2013b; Von Roenn et al., 2013). For example, small for-profit and public hospitals are less likely to have inpatient SPC teams, and outpatient SPC is not uniformly available among all types of hospitals and cancer centers (Goldsmith et al., 2008; Hui et al., 2010). Providing SPC consultation remotely via telemedicine may help increase capacity (Hennemann-Krause et al., 2014). Furthermore, current health care policies and reimbursement mechanisms are not structured to support the early integration of palliative care (Lopez-Acevedo et al., 2013b). The limitations include a lack of institutional financial support, poor reimbursement, and a lack of legislation promoting and supporting the integration of palliative care into routine oncology care (Hui et al., 2010; Lopez-Acevedo et al., 2013b; Partridge et al., 2014).
Public knowledge about palliative care is limited, but people react more favorably when terminology other than “palliative care” is used, and they are willing to seek such care if it is recommended by their oncologist (McInturff and Harrington, 2011; Schenker et al., 2014b). Unfortunately, many providers are uninformed about locally available SPC services and mistakenly consider palliative care to be synonymous with end-of-life care and thus incompatible with anticancer therapy (McInturff and Harrington, 2011; Schenker et al., 2014a). Providers also respond more favorably to the use of the term “supportive care” rather than “palliative care” (Dalal et al., 2011; Fadul et al., 2009). The IOM report Dying in America con-
cluded that “one of the greatest remaining challenges is the need for better understanding of the role of palliative care among both the public and professionals across the continuum of care so that hospice and palliative care can achieve their full potential for patients and their families” (IOM, 2015).
INFORMATION NEEDS AND SHARED DECISION MAKING
In 2013, an IOM report stated, “A high-quality cancer care delivery system depends upon clinical research that gathers evidence of the benefits and harms of various treatment options so that patients, in consultation with their clinicians, can make treatment decisions that are consistent with their needs, values, and preferences” (IOM, 2013, p. 207). The report also said that “if the goal of clinical research is to improve the quality of cancer care, it is important to produce some of the types of evidence that would be most useful to patients and clinicians when making treatment decisions. For example, patients often want information about the estimated impact of a treatment regimen on their quality of life, functional status, symptoms, and overall experience with the disease, as well as information about other contextual factors” (IOM, 2013, p. 222).
Because ovarian cancer is typically at an advanced stage when it is diagnosed, its treatment requires women to make immediate decisions. As the SGO noted,
The diagnosis of cancer requires that people become almost instantaneously knowledgeable about their disease, their treatment options, possible toxicities, and likely outcomes. This disease, and the treatment it requires, will have a major impact on their home life, their caretakers, their economic situation and their overall [quality of life], and within this context, decisions need to be made. (SGO, 2011)
Survivorship in ovarian cancer is negatively affected by a lack of available information on the basics of ovarian cancer, especially as compared to other cancers (Ferrell et al., 2003c; Lockwood-Rayermann, 2006; McCorkle et al., 2003; Trivers et al., 2013). Survivors also often lack specific information on such things as fertility preservation options, complementary and alternative treatment options, and the disease’s impact on sexual function (Ferrell et al., 2003d; Sun et al., 2007). A longitudinal study of women following a diagnosis of ovarian cancer found consistent information needs including the likelihood of cure, the stage or current status of disease, and information about treatment options (Browall et al., 2004). Medical information is often prioritized over practical physical, social, psychological, or spiritual information, even though younger women and those with lower levels of education may place a higher value on information relating to social and psychological issues (Papadakos et al., 2012). However, physical and psychological symptoms are the most common and severe symptoms
reported by women with ovarian cancer (see sections on specific psychosocial and physical issues later in this chapter). Research shows that providing knowledge about the disease process and teaching coping skills can reduce stress and improve the emotional well-being of women with ovarian cancer (Parker et al., 2006; Roland et al., 2013; von Gruenigen et al., 2010).
Shared Decision Making
In the case of women with ovarian cancer, shared decision making can be complicated because so many of these women present at a late stage of the disease, making it necessary to intervene as quickly as possible. For example, younger survivors report that a rush to treatment often overshadowed their ability to explore fertility preservation options (Sun et al., 2007). While women with ovarian cancer say that they want to come to joint decisions about treatment with their physicians, many still place the highest value on their physicians’ recommendations, or even view the decisions as being made solely by their physicians (Elit et al., 2003; Fitch et al., 2003; Jolicoeur et al., 2009; Kitamura, 2010; Luketina et al., 2012; Stewart et al., 2000). In one study of women with ovarian cancer, only 36 percent reported having been very involved in the decision making about surgery, and only 40 percent reported being very involved in decision making about treatment; 20 percent of the women reported no involvement in decision making at all (Andersen et al., 2012). The study further found that women who were involved in decision making about their surgeries, lifestyle, and follow-up care had better emotional health.
In a review of issues for ovarian cancer survivors, Trivers and colleagues commented that “effective survivor–provider communication, including feeling satisfied with the information received and having their disease experiences validated, benefits overall well-being and [quality of life]” (Trivers et al., 2013, p. 2894). However, the quality of the relationship and of the communication between women and their providers can affect how information is exchanged and how care decisions are made. Women who received a positive diagnosis of ovarian cancer long after reporting symptoms to their providers can feel frustrated and angry by the perception of a delayed diagnosis, leading to lower confidence in those providers (Ferrell et al., 2003a). On the other hand, cancer patients undergoing active treatment, including those with ovarian cancer, can be reluctant to complain about their symptoms because of concerns that talking about them might distract their providers away from what they consider to be the more important treatment issues (Gunnarsdottir et al., 2002; Passik et al., 2002; Sun et al., 2007). In a study of women with ovarian cancer on active treatment, women reported experiencing 12 concurrent symptoms on average, but only 61 percent had discussed their most bothersome symptoms with their health care provider in the past month (Donovan et al.,
2005). Equally as concerning, only half of the women who discussed their symptoms with their providers received recommendations for symptom management. Women in this study had low perceptions of control over their symptoms, but those who had received recommendations for symptom management reported significantly higher perceptions of control than those who had not. (See more on symptom assessment and self-management later in this chapter.)
Optimizing the exchange of information and the medical management of treatment and its side effects requires an iterative approach that relies on in-depth conversations between the patient and her interdisciplinary team of providers. Given the current structure of the health care system and the time pressures to move patients through clinics, these types of interactions are difficult to achieve. Furthermore, as discussed in Chapter 4, patient-centered communication and shared decision making in oncology in general are “suboptimal,” with clinicians asking for patient preferences in medical decisions only about half the of the time. In part, this is due to “clinicians’ lack of training in communication and insensitivity to patients’ informational, cultural, and emotional needs” (IOM, 2013, p. 102). As such, it may be useful to involve other health care professionals (e.g., nurses and social workers) to expand on the communication and information provided by physicians or else to use technology (e.g., mobile health technologies) in innovative interdisciplinary models of care so that patient–provider communication between women and their care teams is enhanced.
Efforts to Facilitate Shared Decision Making in Ovarian Cancer
Many efforts are under way to facilitate the dissemination of necessary information so that women with ovarian cancer can better engage in shared decision making. These efforts are aimed both at helping better inform women about their choices and at ensuring that women’s individual preferences are included in the decision-making process. In many cases, decision aids are being developed to help women make informed choices. However, while some aids focus on helping women make specific treatment-related decisions (Anderson et al., 2011; Oostendorp et al., 2011), most aids for women with ovarian cancer focus on helping women at high risk for a genetic mutation make decisions about genetic counseling, risk management, and genetic testing (Juan et al., 2008; Tiller et al., 2003, 2006; Wakefield et al., 2008a,b).
Cancer advocacy organizations and policy organizations uniformly encourage self-advocacy, the act of speaking up for your own rights and preferences, as a means of addressing barriers to high-quality, patient-centered cancer care (ASCO, 2015; IOM, 2001; Shapiro et al., 2009). The term originated in the HIV/AIDS, disabilities, and mental health research and advocacy communities and has only recently been adopted into the
cancer lexicon. In these other populations, self-advocacy has been shown to be associated with better symptom management, higher quality of life, and more effective use of health care resources (Brashers et al., 2004; Pickett et al., 2010; Test et al., 2005). One emerging line of research that originated in the ovarian cancer community is aimed at defining the concept of self-advocacy, the skills required for effective self-advocacy, predictors of poor self-advocacy, and the short- and longer-term consequences of self-advocacy in order to help guide the development of interventions (Hagan and Donovan, 2013a,b).
Many different stakeholders act as sources of reliable information and actively work to support women with ovarian cancer in making decisions about their own care. For example, Facing Our Risk of Cancer Empowered, or FORCE, strives to raise awareness and provide resources and information related to genetic counseling and testing for women at high risk for breast and ovarian cancer (FORCE, 2015). The National Ovarian Cancer Coalition’s Take Early Action & Live (TEAL) Initiative focuses on creating awareness of the symptoms of undiagnosed ovarian cancer, and its TEAL Totes provide educational materials to women newly diagnosed with ovarian cancer (NOCC, 2015). Other programs focus on increasing awareness among health care providers.
Patient-Centered Outcomes Research Institute
The Patient-Centered Outcomes Research Institute (PCORI), authorized by the Patient Protection and Affordable Care Act,1 has a mission to help “people make informed healthcare decisions, and improve healthcare delivery and outcomes, by producing and promoting high-integrity, evidence-based information that comes from research guided by patients, caregivers, and the broader healthcare community” (PCORI, 2015a). Two ovarian cancer–specific projects are funded by PCORI. The American BRCA Outcomes and Utilization of Testing Patient-Powered Research Network, also known as the ABOUT Network, is designed to increase community engagement in research, promote hereditary breast and ovarian cancer (HBOC) research opportunities, and optimize enrollment into HBOC-specific studies as well as to report new research findings to the community (PCORI, 2015b). The other project is developing an ovarian cancer patient-centered decision aid (PCORI, 2015c). The aid will allow patients to assimilate information and identify trade-offs about the impact of combining intraperitoneal and intravenous therapy (versus intravenous therapy alone) on their quality of life and survival, based on their own preferences and personal clinical characteristics.
________________
1 Patient Protection and Affordable Care Act, Public Law 148, 111th Cong., 2nd sess. (March 23, 2010).
PHYSICAL AND PSYCHOSOCIAL EFFECTS OF OVARIAN CANCER DIAGNOSIS AND TREATMENT
Because women are often diagnosed with ovarian cancer at an advanced stage, the urgency of treating the primary disease or a lack of available expertise may lead to neglect of the management of the physical and psychosocial effects of such a diagnosis and the subsequent treatment. And while many of the effects that women with ovarian cancer experience may be similar to those of individuals undergoing treatment for other types of cancer, women with ovarian cancer may also have specific concerns or needs because of their typically long and recurring periods of active treatment. Unfortunately, as mentioned earlier in this chapter, there is little existing literature on the physical and psychosocial effects of ovarian cancer specifically, and, generally, such literature as does exist seldom takes into account the different ovarian cancer subtypes, different racial and ethnic groups, or different age groups, and it is mostly based on small studies that are insufficient to fully inform practice at this time. The following sections give an overview of the particular physical and psychosocial concerns for women with ovarian cancer. Later in this chapter there is a discussion of interventions aimed at addressing these concerns, and, in recognition of the fact that the accurate assessment of symptoms depends on patient-reported outcomes (PROs), there is also a discussion of the role of PROs and self-management.
Physical Effects of Diagnosis and Treatment
Women with ovarian cancer usually undergo prolonged and intensive courses of active treatment, often for many years (see Chapter 4), and these courses of treatment have numerous side effects that require long-term active management (Chase and Wenzel, 2011). Most commonly, these side effects are similar to those experienced by other cancer patients undergoing chemotherapy and include pain, fatigue, peripheral neuropathy, abdominal symptoms, nausea, hair loss, weight loss, and loss of appetite (Arriba et al., 2010; Badr et al., 2006; Ferrell et al., 2003a; Lockwood-Rayermann, 2006; Otis-Green et al., 2008; Sun et al., 2007). Younger survivors may experience different side effects than older survivors, and the effects may also vary by ovarian cancer subtype (Matei et al., 2009).
Neuropathy is a particularly concerning side effect for women undergoing treatment for ovarian cancer and is especially prevalent because of the typical course of multiple cycles of chemotherapy over many years (Ezendam et al., 2014; Josephs-Cowan, 2006; Nurgalieva et al., 2010; Pignata et al., 2006; Postma et al., 1999; Verstappen et al., 2003; Visovsky and Daly, 2004; Wenzel et al., 2007). Fatigue is also of high concern for
women with ovarian cancer (Anderson and Hacker, 2008; Clevenger et al., 2012, 2013; Liavaag et al., 2007; Payne, 2002; Schrepf et al., 2013; Shinde et al., 2015; Sun et al., 2005). Pain is another particular concern, especially toward the end of life (Portenoy et al., 1994; Rolnick et al., 2007). Some of these side effects can last for years, even after treatment has ceased (Shinde et al., 2015).
The morbidity and mortality of ovarian cancer depends in part on a woman’s physical health. For example, in a study of 30-day morbidity in onco-geriatric surgical patients (including women with ovarian cancer), baseline physical function prior to surgery was an accurate predictor of major complications (Huisman et al., 2014). Among women with Stage III ovarian cancer, baseline physical well-being has been associated with overall survival (von Gruenigen et al., 2012). Overall survival was lower for women in the lowest quartile of baseline physical well-being compared with those in the highest quartile. Cancer prehabilitation (i.e., the assessment of baseline functional level in order to identify impairments and provide targeted interventions to improve health and prevent future impairments) has shown promise in patients with gastric cancer (Silver and Baima, 2013; West et al., 2015) and might be applicable to women with ovarian cancer.
Psychosocial Effects of Diagnosis and Treatment
The diagnosis and treatment of cancer in general leads to a wide range of psychosocial effects with a significant impairment in quality of life. The 2008 IOM report Cancer Care for the Whole Patient reported:
[I]ndividuals diagnosed with cancer often report that their care providers do not understand their psychosocial needs; do not consider psychosocial support an integral part of their care; are unaware of psychosocial health care resources; and fail to recognize, adequately treat, or offer referral for depression or other sequelae of stress due to illness in patients and their families. (IOM, 2008, p. 5)
Quality of Life and Personal Growth
In a review of the social and psychological needs of ovarian cancer survivors, Roland and colleagues noted that “despite the many challenges of living with [ovarian cancer], survivors often experience personal growth through their spiritual lives, personal relationships, and renewed perspectives on life” (Roland et al., 2013). Ovarian cancer survivors report a good quality of life overall, and an improved quality of life is most likely to be reported when a patient has supportive relationships, a reprieve from treatment, and a lack of physical side effects (Bodurka-Bevers et al., 2000; Champion et al., 2007; Ferrell et al., 2005; Fox and Lyon, 2007; Kornblith
et al., 2010; Otis-Green et al., 2008; Roland et al., 2013; Stewart et al., 2001; Von Gruenigen et al., 2009, 2010; Wenzel et al., 2002). The stage at which ovarian cancer is diagnosed also shows mixed associations with quality of life, and younger survivors report lower quality of life than older survivors (Bodurka-Bevers et al., 2000; Ferrell et al., 2005; Mirabeau-Beale et al., 2009; Roland et al., 2013; von Gruenigen et al., 2010).
Women often cope in survivorship by being active, spending time with family, supporting other survivors, participating in advocacy, and “living for the moment” (Ferrell et al., 2003b; Roland et al., 2013). Findings on spirituality and ovarian cancer survivorship are mixed; some women who have experienced ovarian cancer report feeling increased meaning and purpose of life, with faith providing strength and hope, while others report a loss in spiritual faith (Champion et al., 2007; Ferrell et al., 2003d; Kornblith et al., 2010; Matulonis et al., 2008; Monahan et al., 2008; Seibaek et al., 2012; Swenson et al., 2003; Wenzel et al., 2002).
Social Supports and Relationships
Social supports and relationships have a positive impact on self-esteem, depression, anxiety, and the overall health of cancer survivors (Champion et al., 2007; Nausheen et al., 2009; Norton et al., 2004, 2005; Pinquart and Duberstein, 2010a; Roland et al., 2013). Given the relative rarity of ovarian cancer, many women with the disease report feeling isolated and seek out other survivors, but many lack access to support groups or other resources (Ferrell et al., 2003b,c,d; Jackson et al., 2007; Roland et al., 2013; Swenson et al., 2003; Wenzel et al., 2002). Advocacy organizations have been particularly important in providing a community for women with ovarian cancer to come together, both at the national level and at the local level (see Chapter 1). For example, the Ovarian Cancer National Alliance provides an online community for women with ovarian cancer in general, including a specific group for women diagnosed with granulosa cell tumor (OCNA, 2015). Given the rarity of ovarian cancer, such online resources can provide a community for women who might not otherwise have access to speaking with other women with similar diagnoses. Social supports from family and friends are also important, such as the caregiving often provided to women with ovarian cancer. (See Chapter 4 for more on family caregiving.)
These sorts of supports can also influence the course of the disease itself. Women with ovarian cancer who report low levels of social support have been shown to have alterations in markers of inflammation, angiogenesis, invasion, innate immunity, and gene expression (Costanzo et al., 2005; Lutgendorf et al., 2002, 2005, 2008, 2009, 2011). One of the first studies to link social supports to ovarian cancer disease outcomes, a prospective study of 168 women newly diagnosed with ovarian carcinoma,
found a lower likelihood of death among those women with higher social attachment, defined as “an individual’s experience of emotional connection to others that provides a sense of well-being, intimacy, or security” (Lutgendorf et al., 2012). By contrast, instrumental support, defined as “availability of help, information, and advice from other people,” was not associated with survival. It is worth noting that controlling for depression did not change the relationship between social attachment and survival, that the distribution of marital relationships was similar among women with high and low levels of social attachment, and that even moderate reductions in social attachment placed women in the high-risk category. These findings have important implications for screening women for deficits in social attachment and identifying interventions to strengthen or supplement women’s existing support networks.
Psychological Distress
Ovarian cancer survivors have high levels of depression and anxiety as compared with the general population and non-gynecologic-cancer survivors (Bodurka-Bevers et al., 2000; Ferrell et al., 2005; Norton et al., 2004; Roland et al., 2013), and younger survivors tend to have higher levels of distress and depression than older survivors (Bodurka-Bevers et al., 2000; Norton et al., 2004; Ponto et al., 2010). The point of diagnosis can be particularly stressful because of the sudden change in health status and feelings of losing control (Ferrell et al., 2003b,d, 2005; Norton et al., 2004; Wenzel et al., 2002). Associations between psychological distress and the stage of disease at diagnosis have been variable (Bodurka-Bevers et al., 2000; Kornblith et al., 2010; Norton et al., 2004). Roland and colleagues noted that “[ovarian cancer] survivors experiencing greater physical symptoms have higher levels of distress, depression, and anxiety, possibly because of symptoms being perceived as disease progression” (Roland et al., 2013).
Ovarian cancer survivors, especially younger survivors, commonly report distress related to fears about recurrence and death (Kornblith et al., 2010; Matulonis et al., 2008; Otis-Green et al., 2008; Shinn et al., 2009; Wenzel et al., 2002). These fears may persist for years after diagnosis and affect quality of life even when the woman is living without evidence of disease (Ferrell et al., 2003b,d; Wenzel et al., 2002). Women may also be distressed by waiting for test results, such as with surveillance for recurrent disease by monitoring CA-125 levels (see Chapter 3) (Parker et al., 2006).
Survivors may experience guilt or fear related to their genetic predisposition for ovarian cancer. In families with a history of ovarian cancer, survivors report feeling sympathy for family members as they recalled their own pain in supporting other relatives with ovarian cancer, and they also feel guilty about passing genetic mutations to their daughters (Ferrell et al.,
2003b,c,d; Sun et al., 2007; Trivers et al., 2013). Women with germline mutations expressed further fear and concern for the lack of effective screening for their family members (Ferrell et al., 2003d). (See Chapter 3 for more on genetic counseling and testing for family members.)
Reproductive and Sexual Health
Studies of the reproductive and sexual concerns of women with cancer often include women with all types of gynecologic cancer or, even more broadly, women with any type of cancer. Thus, women with ovarian cancer may be underrepresented in these studies (Abbott-Anderson and Kwekkeboom, 2012; Deshpande et al., 2015). Survivors of ovarian cancer at all ages have many concerns about their sexual health, including treatment-induced menopause, pain or discomfort during sex, poor body image, decrease in sexual desire and satisfaction, and difficulty in communication about sexuality with a partner (Buković et al., 2008; Carmack Taylor et al., 2004; Ferrell et al., 2003c; Gershenson et al., 2007; Kornblith et al., 2010; Liavaag et al., 2008; Matulonis et al., 2008; Mirabeau-Beale et al., 2009; Roland et al., 2013; von Gruenigen et al., 2009; Wenzel et al., 2002; Wilmoth et al., 2011). Younger survivors report particular concerns about decreases in sexual desire, activity, and pleasure (Champion et al., 2007; Gershenson et al., 2007; Monahan et al., 2008; Swenson et al., 2003).
Infertility or reproductive concerns are pronounced among younger ovarian cancer survivors (Trivers et al., 2013). Young survivors who desire children report that infertility is more distressing than the initial diagnosis itself, and many experience anger or regret about not receiving information or exploring options for fertility preservation prior to treatment (Sun et al., 2007). Fertility preservation may be feasible through conservative surgery or the cryopreservation of oocytes, embryos, or ovarian tissue (Alvarez et al., 2014; Dittrich et al., 2015; Henes et al., 2014; Lambertini et al., 2015; Letourneau et al., 2015; Morice et al., 2011; Prasath et al., 2014). ASCO guidelines state that all cancer patients of childbearing age should receive information about fertility preservation options, yet in practice this does not always happen (Lee et al., 2006; Quinn et al., 2008; Schover et al., 1999; Tomao et al., 2015). Unfortunately, little is known about the extent to which young women with ovarian cancer receive fertility preservation counseling or the impact of counseling on important psychological outcomes during survivorship. One review found that women with ovarian cancer who received fertility preservation counseling had a reduction in long-term regret, regardless of age or parity (Deshpande et al., 2015). As most women with ovarian carcinomas are diagnosed at later ages (see Chapter 1), issues of fertility preservation may be most relevant for women with less common types of ovarian cancer, most notably germ cell tumors,
which tend to occur in teenage girls or young women. One review of 145 women with malignant ovarian germ cell tumors found no difference in menstruation, pregnancy, or offspring after fertility-preserving treatment (Zhang et al., 2012).
Finances and Employment
Ovarian cancer treatment often affects women’s ability to work (Trivers et al., 2013). Some survivors report needing to take time off from work to receive care, and other survivors retire after diagnosis (Ferrell et al., 2003c; Matulonis et al., 2008; von Gruenigen et al., 2009). However, research shows that survivors often remain in the workforce or report a need to return to work for economic reasons (e.g., to recover lost wages and maintain insurance coverage), but also as a means of returning to a “normal” life (Ferrell et al., 2003c). Employment and higher income are associated with a higher quality of life for women with ovarian cancer (Ferrell et al., 2005; von Gruenigen et al., 2009). Younger survivors particularly report needing to change jobs because their ability to work was affected but also report fearing that a change of employment might result in the loss of health insurance coverage (Matei et al., 2009).
INTERVENTIONS FOR SUPPORTIVE CARE AND IMPROVING OUTCOMES
Researchers need to better understand how to manage the side effects of disease and treatment, as well as how to develop interventions that can improve disease and treatment outcomes for women with ovarian cancer. Many of the physical and psychosocial effects of ovarian cancer diagnosis and treatment may be better managed by combining pharmacologic and non-pharmacologic therapies, but studies provide little insight into best practices. The following sections describe strategies for managing the physical and psychosocial side effects of treatment, including specific activities that might alleviate symptoms, and how modifying behaviors may affect outcomes.
Pharmaceutical Interventions
The management of the physical side effects of treatment for ovarian cancer often follows the same course as for patients with any other type of gynecologic cancer or even any type of cancer in general, because the most common side effects arise from treatments that are common to all the different types of cancer (e.g., chemotherapy). Studies have looked specifically at the question of how to help women with ovarian cancer deal with the nausea and vomiting that often accompany treatment (Choi et al.,
2014; Timmins et al., 2008; Walker and Lane, 2007; You et al., 2009), pain (Rolnick et al., 2007), and malnutrition (Gadducci et al., 2001). While much can be learned from the literature that is not specific to ovarian cancer, more research is needed on how to improve the pharmaceutical management of some of the side effects of current therapies. Furthermore, there should be more effort placed on developing new disease therapies and approaches that have lower levels of side effects rather than on developing new therapies to manage the side effects of current therapy options (Chase and Wenzel, 2011; Chase et al., 2012, 2015; Teefey et al., 2013). (See Chapter 4 for more on clinical trials and the development of newer therapies with lower levels of toxicity.)
Non-Pharmaceutical Interventions
In addition to taking advantage of decision-making aids (see earlier in this chapter), ovarian cancer survivors can also participate in various activities that not only can help manage the effects of their diagnosis and treatment, but also may improve outcomes. Such efforts are generally not well developed, but there are a number of areas, described below, that warrant further study. (See Chapter 3 for more on the potential role that some of these areas can play in modifying the risk for developing ovarian cancer.)
Complementary and Alternative Medicine
Like other cancer patients, women with ovarian cancer often explore a number of nonstandard treatments (e.g., acupuncture, yoga, vitamins, herbs) to manage their symptoms or even in an attempt to cure the disease (Arriba et al., 2010; Chan et al., 2011; Ferrell et al., 2005; Helpman et al., 2011; Helpman Bek et al., 2009; Lu et al., 2009; Matulonis et al., 2008; von Gruenigen et al., 2006; You et al., 2009). These approaches need to be studied for their potential benefits in managing the side effects of treatment or even improving outcomes, but they may also need to be evaluated for any potential negative interactions with prescribed medications (Andersen et al., 2013).
Exercise
Exercise and yoga have been associated with improvements in physical functioning and qualify of life as well as decreases in pain, fatigue, anxiety, and depression among women with ovarian cancer (Danhauer et al., 2008; Lowe et al., 2012; Sohl et al., 2010, 2012; Stevinson et al., 2009). On the other hand, a review of the literature finds mixed evidence for associations—either a positive association or no association, depending on the
study design—between recreational physical activity and ovarian cancer risk and survival, but the authors emphasized “the greater body of scientific evidence which has demonstrated that [recreational physical activity] results in a plethora of health benefits that can be achieved in all populations, including those with cancer” (Cannioto and Moysich, 2015). (See below for more on sedentary lifestyle.)
Nutrition
Few studies have looked at the role of nutritional assessment in ovarian cancer as part of the treatment plan or at the impact of treatment on nutritional status (Billson et al., 2013; Geisler et al., 2007; Glaser et al., 2012). However, poor nutrition, sedentary lifestyle, and obesity have been associated with a poorer quality of life and survival among women with ovarian cancer (Nagle et al., 2015; Pavelka et al., 2006; Smits et al., 2015; Torres et al., 2013). (See Chapter 4 for more on obesity as a factor in caring for women with ovarian cancer.)
Sleep
Treatment-related sleep disturbances can lead to fatigue, depression, and decreased quality of life for women with ovarian cancer (Clevenger et al., 2013; Mizrahi et al., 2013; Sandadi et al., 2011). Interventions to improve sleeping patterns may help improve quality of life and even treatment outcomes.
Biobehavioral Pathways and Outcomes
Psychosocial and biobehavioral factors can influence the tumor microenvironment and tumor progression (Lutgendorf and Andersen, 2015). In particular, given the evidence of their association with cancer progression and cancer death, three important psychosocial and biobehavioral factors—stress, depressive symptoms, and social support or attachment—are promising targets for intervention (Chida et al., 2008; Pinquart and Duberstein, 2010a,b). These factors are known to modulate tumor characteristics such as angiogenesis, metastasis, and immune response. For example, ovarian tumors in chronically stressed animals have been shown to be larger, more highly vascularized, and having an enhanced expression of proteins known to promote aggressive tumor behavior (Thaker et al., 2006). Similarly, ovarian cancer patients reporting depression and low social support had higher levels of proteins associated with angiogenesis, tumor growth, and immune response (Costanzo et al., 2005; Lutgendorf et al., 2002, 2008). Epidemiologic studies show that improved social support is associated with longer
survival in many cancers, including ovarian cancer (Lutgendorf et al., 2012; Pinquart and Duberstein, 2010a).
Psychosocial interventions have been shown to improve the survival of breast cancer patients, but the results of the studies are inconsistent (Andersen et al., 2008). Unfortunately, to date, few intervention studies in ovarian cancer have been adequately designed to test the same mechanisms. More research is needed on the relationship between biobehavioral factors and signaling pathways in ovarian cancer if researchers are to identify therapies that can block the interactions that promote tumor growth. In addition, research to identify how best to monitor and assess patterns of behavior (e.g., using smart phones and sensors) may provide insight into the relationship between biobehavioral and lifestyle factors. Work investigating how exercise and diet may influence not only patient survival but also tumor response to therapies is needed in order to identify potential novel nonpharmacologic approaches to complement pharmacologic approaches. One current example of such research is a Phase III clinical trial that is studying the impact of changes in diet and physical activity on progression-free survival for patients with previously treated ovarian, fallopian tube, or primary peritoneal cancer (GOG, 2015).
SYMPTOM ASSESSMENT AND SELF-MANAGEMENT
The systematic assessment of symptoms and quality of life in ovarian cancer patients has so far not been a major focus in clinical practice or research. Providers need to ask women about their symptoms, and survivors need to report any new information about their symptoms to their providers in order to inform the process of developing a treatment plan or supportive care interventions. Furthermore, women need to make their personal preferences for care known to their providers. For example, some women may be willing to forego aggressive treatment in order to enjoy, in their opinion, a better quality of life. The following sections discuss the role of the patient in the assessment and management of symptoms (including some of the strategies discussed in the previous sections of this chapter) and in discussing personal preferences for care plans.
Patient-Reported Outcomes
The U.S. Food and Drug Administration defines PROs as the “measurement of any aspect of a patient’s health status that comes directly from the patient without the interpretation of the patient responses by a physician or anyone else” (FDA, 2009). Paying attention to PROs will be an important way to inform the development of newer therapies with side-effect profiles that are more acceptable to patients. In 2010 the IOM noted that for cancer
clinical trials in general, “publicly funded clinical trials play a vital role by addressing questions that are important to patients but are less likely to be top priorities of industry,” including trials focusing on quality of life (IOM, 2010, p. 10). In 2013, the IOM called on the National Cancer Institute (NCI), PCORI, and others to “develop a common set of data elements that captures [PROs], relevant patient characteristics, and health behaviors that researchers should collect from randomized clinical trials and observational studies” (IOM, 2013, p. 12). Collecting PROs is also important for the management of the symptoms of disease and treatment that providers may be unaware of.
PROs facilitate the systematic assessment of symptoms and impairments in quality of life in clinical and research settings (Cleeland and Sloan, 2010; FDA, 2009; Friedlander and King, 2013; King et al., 2014; Williams et al., 2013). Providers tend to rely on asking open-ended questions, which can lead to significant underreporting of symptoms (Homsi et al., 2006). In the research setting, the use of adverse event reporting using the Common Terminology Criteria for Adverse Events (CTCAE) is commonly used as a substitute for the assessment of patients’ symptoms and quality-of-life experiences. However, the use of CTCAE can be unreliable, clinicians may underestimate CTCAE symptoms compared with what is indicated in patient self-reporting, and patients can be better than physicians at detecting serious impairments (Atkinson et al., 2012; Basch, 2010).
The systematic assessment of PROs is essential to ensure safety, to identify and intervene on life-affecting symptoms, to inform shared decision making, to accurately identify and report treatment-related toxicities, and to inform the design and conclusions of clinical trials (see Chapter 4). The NCI sponsored the development and validation of a patient-reported version of CTCAE for use in clinical trials (Dueck et al., 2015), and the Patient-Reported Outcomes Measurement Information System is a national initiative to develop tools for assessing key PROs (Cella et al., 2010; Wagner et al., 2015). Unfortunately, little is known about how best to support clinicians and patients in integrating systematic assessment of PROs into clinical settings or about the optimal timing and frequency of assessments.
PROs in Ovarian Cancer
The NCI’s Symptom Management and Health-Related Quality of Life Steering Committee convened a group of experts to identify a core set of symptoms to be assessed routinely in cancer clinical trials for three cancers with a high symptom burden: ovarian, head and neck, and prostate (Reeve et al., 2014). The review of symptoms in ovarian cancer largely focused on women with recurrent disease (Donovan et al., 2014b). The panel recommended prioritizing three areas of symptoms to be monitored with
PROs: symptoms similar to those experienced in other cancers, abdominal symptoms, and symptoms particularly prevalent or important to women with ovarian cancer. Again, many of the most important symptoms for women with ovarian cancer were the same as those for patients with all types of cancer (Reeve et al., 2014). The committee’s list of PROs that are particularly important to assess for women with ovarian cancer included abdominal pain, bloating, cramping, fear of recurrence/disease progression, indigestion, sexual dysfunction, vomiting, weight gain, and weight loss (Donovan et al., 2014b).
The inclusion of PROs like these into clinical trials is not new, but the importance of including them has become more widely appreciated and accepted (Friedlander and King, 2013). However, assessment is not enough. Assessment without adequate strategies for managing unmet symptom and quality-of-life needs will lead to frustration for patients and clinicians alike, which only underscores the need to identify better approaches to symptom management for women with ovarian cancer. Furthermore, as more novel agents are used to treat ovarian cancer, new and distinct side effects may arise. The assessment of PROs specific to ovarian cancer will complement the typical primary outcome of concern—survival—and will provide useful data on the benefits and risks of new treatments (FDA, 2009). Therefore, PROs need to evolve along with the nature of symptoms (Donovan et al., 2014b; Han et al., 2009).
Individual Preferences
Ovarian cancer affects women of all ages, races, and social statuses. As such, preferences regarding treatment types and the importance of quality of life versus quantity of life can vary from woman to woman. For example, younger women with ovarian cancer tend to focus on fertility, the impact to family life, and employment (Trivers et al., 2013). One study found that religious beliefs can affect treatment decisions, particularly care near death (Phelps et al., 2009). Because many ovarian cancers tend to recur, women’s preferences can change over the course of treatment, depending on the stage of the cancer and the number of recurrences. Still, survivors tend not to transition to palliative care even in the face of an unfavorable prognosis (Sun et al., 2007). Therefore, regular discussion needs to occur between the woman and her care team to ensure that her needs are being addressed, and treatment options need to be tailored to the prognosis, needs, and desires of each woman.
Self-Management
Self-management is broadly defined as an individual’s engagement in the management of the symptoms and consequences of a health problem, including treatment and the wide range of psychological, social, physical, and lifestyle and role changes (Barlow et al., 2002). Necessary skills for self-management include forming effective partnerships with health care providers, adhering to medication and treatment recommendations, problem solving and decision making, and taking action (Lorig and Holman, 2003). A key aspect of self-management is that it works best in conjunction with family members and providers encouraging the individual patients to become informed about their conditions, engage in a lifelong process of self-monitoring and self-evaluation, and shift from a perspective of illness to one of wellness (Davis et al., 2000; Dunbar et al., 2008; Grey et al., 2006; Lorig and Holman, 2003; Wiecha and Pollard, 2004). (See Chapter 4 for more on the role of family caregivers in providing needed supports.) In 2003, the IOM report Priority Areas for National Action: Transforming Health Care Quality called for providing greater support for self-management (IOM, 2003).
A great deal of research supports the value of providing self-management training for various chronic diseases, such as diabetes and depression (Barlow et al., 2002; Bodenheimer et al., 2002; Davis et al., 2000; Ferguson, 2011; Fredericks et al., 2012; Houle et al., 2013; Lorig and Holman, 2003; Norris et al., 2001, 2002; Schulman-Green et al., 2012; Steed et al., 2003; Wiecha and Pollard, 2004). As cancer survival rates have increased over the past 20 years, researchers, clinicians, and survivors have come to see the value of adopting the chronic care model of self-management into supportive care services for patients with cancer, and self-management interventions to improve coping with cancer-related symptoms have shown promising results (Cimprich et al., 2005; Cockle-Hearne and Faithfull, 2010; Foster and Fenlon, 2011; Hoffman et al., 2013; Koller et al., 2012; Lee et al., 2013; van den Berg et al., 2012).
A recent review of 32 self-management intervention studies characterized self-management interventions in three areas: skills needed during treatment, skills needed during the transition from primary treatment to survivorship, and skills for persons with advanced cancer approaching the end of life (McCorkle et al., 2011). Only one of these studies focused on women with gynecologic cancers (McCorkle et al., 2009). In this particular study, advanced practice nurses provided home visits and telephone calls for 6 months following surgery for gynecological cancers in order to teach self-management skills for dealing with the short- and long-term physical and
psychosocial symptoms associated with treatment. Uncertainty2 decreased in the intervention group, and for those with high baseline distress, the intervention resulted in improvements in symptom distress and in mental and physical distress over time. More recently, a pilot study of the Written Representational Intervention To Ease Symptoms (WRITE Symptoms) program showed that providing Web-based symptom management support for women with ovarian cancer resulted in lower symptom-related distress (Donovan et al., 2014a). In this study, women with recurrent ovarian cancer were connected to nurses via private Internet message boards for 8 weeks to learn how to monitor and manage their symptoms. The Gynecologic Oncology Group recently compared a nurse-delivered WRITE Symptoms intervention with a self-directed, computer-mediated WRITE Symptoms intervention to see how effective each was in decreasing symptom severity, distress, consequences, and depression and improving quality of life among women with recurrent ovarian, fallopian tube, or primary peritoneal cancer (NIH, 2013). Primary findings from this study are pending.
While small improvements are possible with self-management skills training alone, making substantial improvements requires ongoing interactions between patients and clinicians as well as care delivery modifications designed to support patients and families in their self-management efforts (McCorkle et al., 2011). Future research could identify ways to disseminate and implement promising theory-guided interventions in clinical practice. One challenge to this approach is that the amount of work required for self-management during a time when the disease and the treatment are physically, cognitively, and psychologically overwhelming can be burdensome (Granger et al., 2009; Maeng et al., 2012; Russell et al., 2005). Thus, there is a pressing need for research on interventions that focuses not only on enabling self-management but also on elucidating the mechanisms by which interventions can support self-management behaviors (Hammer et al., 2015). Finally, many interventions that promote the self-management of chronic diseases are effective in the short term, but the self-management behaviors typically diminish over time (Ory et al., 2010; Tang et al., 2012), and the strategies and theoretical mechanisms for sustaining self-management behaviors are not well understood. In 2014, the National Institutes of Nursing Research convened a workshop to discuss the state of the science in self-management; the participants identified a number of areas of research needed to advance self-management science, including
________________
2 The Mishel Uncertainty in Illness Scale, based on a theory of uncertainty in illness, helps to measure “uncertainty in symptomatology, diagnosis, treatment, relationship with caregivers, and planning for the future” (Mishel, 1981, p. 258).
- Technology that can support self-monitoring and self-management;
- Brain-behavior links and potential environment moderators of successful self-management;
- Interventions to support the self-management of symptoms across chronic conditions;
- Strategies to increase the sustainability of self-management interventions across the lifespan;
- Factors that facilitate the translation of interventions into clinical practice;
- Modifiable epigenetic factors that are influenced by self-management interventions and how they may shape biological and behavioral outcomes; and
- Community interventions to reduce disparities and support self-management in rural and underserved areas (NINR, 2015).
The IOM report Dying in America is largely generalizable to women with ovarian cancer in the late stages of their disease, in that the target group for that report is patients with “a serious illness or medical condition who may be approaching death” (IOM, 2015, p. 1). The report recommends that all stakeholders (including patients and their families, policy makers, clinicians, leaders in health care delivery and financing, researchers, funders, religious and community leaders, advocates, journalists, and members of the public) learn what constitutes good care for people nearing the end of life. Other recommendations that are generalizable to women with ovarian cancer include (1) to assess the woman’s physical, emotional, social, and spiritual well-being frequently; (2) to pay attention to physical symptoms, emotional distress, family support, social needs, and spiritual and religious needs; (3) to offer referral to expert-level palliative care; and (4) to offer referral to hospice if the patient has a prognosis of 6 months or less. The following sections describe some of the literature that is specific to end-of-life care for women with gynecologic cancers.
Hospice Care
Hospice care is a specialized end-of-life service, most often covered by the Medicare Hospice Benefit, which is available to patients with a prognosis of 6 months or less who are no longer pursuing anticancer therapy. Retrospective data in non-gynecologic cancers suggest that hospice care is associated with no decrease in the length of survival and an improved length of survival in some cancers and that family members of hospice patients are more likely to report better qualify of life, lower rates of physical
and psychosocial distress, and better quality of death for their loved ones than patients treated in the hospital at the end of life (Connor et al., 2007; Teno et al., 2004; Wright et al., 2010).
In one study of women with recurrent gynecologic cancer, the median overall survival for patients using hospice care was 17 months, compared with 9 months for the group not using hospice (Keyser et al., 2010). Various studies suggest that between 20 and 60 percent of patients who die of ovarian cancer use hospice (Fairfield et al., 2012; Lewin et al., 2005; Lopez-Acevedo et al., 2013a; von Gruenigen et al., 2008; Wright et al., 2014). Other studies show that between 3.5 and 12.1 percent of women with ovarian cancer enroll in hospice in the last 3 days of life (an indicator of poor-quality end-of-life care) (Brown et al., 2014; Fairfield et al., 2012; Lopez-Acevedo et al., 2013a; von Gruenigen et al., 2008). Gynecologic oncology patients are more likely to use hospice services and reap the benefits for themselves and their families when hospice is recommended by their oncology team (Brown et al., 2014).
Barriers to timely hospice use include physicians overestimating how long a patient will likely live and both physicians and patients and their families not being aware of or misunderstanding the benefits of hospice care (Christakis and Iwashyna, 2000; Friedman et al., 2002; Gazelle, 2007; Glare et al., 2003). The requirement that patients forego anticancer therapy in order to enroll in hospice also is a barrier to hospice use, particularly with the recent growth of targeted anticancer therapies with fewer side effects (Lopez-Acevedo et al., 2013b).
Advance Care Planning
According to the IOM report Dying in America, “[A]dvance care planning refers to the whole process of discussion of end-of-life care, clarification of related values and goals, and embodiment of preferences through written documents and medical orders” (IOM, 2015, p. 122). Such conversations between providers and women with ovarian cancer are best not left until the point where it is being considered or recommended that the anticancer therapy be discontinued. Oncologists and other providers can contribute to the quality of end-of-life care by having conversations about patients’ goals and preferences earlier and often throughout the disease course (Lopez-Acevedo et al., 2013a; Wright et al., 2014). Such conversations have been associated with improved clinical outcomes, including improved patient quality of life, less aggressive medical care near death, earlier hospice referrals, and a reduction in both surrogate distress and costs, and they have not been associated with increased anxiety or depression or loss of hope (Bernacki et al., 2014; Fried et al., 2003; Wright et al., 2008).
Studies of conversations about advanced care planning or the goals of care that include women with ovarian cancer tend to focus on conversations specific to end-of-life care. For example, in a study of admissions of gynecologic oncology patients that resulted in the patient being discharged to a hospice, patients who had had an outpatient discussion about hospice care prior to hospital admission had shorter lengths of stay and were more likely to receive palliative care consultation (Doll et al., 2013). Another study of women who eventually died of ovarian cancer found that 80 percent had a documented conversation about end-of-life care before their death, but those conversations occurred a median of 29 days prior to death (Lopez-Acevedo et al., 2013a). The researchers found that women who had a conversation about end-of-life care at least 30 days before death had less aggressive interventions at the end of life (e.g., less chemotherapy within 14 days of death, lower rates of hospitalization during the last 30 days of life, lower rates of intensive care unit admission during last 30 days of life, and lower rates of admission to hospice within 3 days of death).
Provider Training in Palliative and End-of-Life Care
Gynecologic oncologists are not being well trained in palliative care (Ramondetta et al., 2004). A survey of gynecologic oncology fellows found that while 89 percent felt that palliative and end-of-life care were integral to their training, only 11 percent reported having such training (Lesnock et al., 2013). They also reported that the quantity and quality of training in palliative care was lower than training in other common procedural and oncologic issues. In a separate survey of both gynecologic oncology fellows and candidate members of the SGO, only about 8 percent reported having received formal training (Eskander et al., 2014). A survey of gynecologic oncology fellowship directors found that all reported that their programs had covered at least one palliative care topic in didactic sessions in the previous year and that 48 percent offered a required or elective palliative care rotation, but that only 14 percent had a written palliative care curriculum (Lefkowits et al., 2014). A recent SGO white paper on palliative care suggested that “the SGO should be at the forefront of developing both [palliative care] curricula for our trainees as well as continuing education for current gynecologic oncologists” (Landrum et al., 2015).
The committee offers the following findings and conclusions:
- Most ovarian cancer research focuses on treatment, not supportive care issues.
- Much of the research on survivorship aggregates individuals with different types of cancer, including both gynecologic and non-gynecologic cancers.
- Research specific to survivors of ovarian cancer rarely distinguishes between the needs of older and younger women, of women from different racial and ethnic groups, or of women who have been diagnosed at different stages of disease.
- Retrospective studies may neglect the experiences of women who did not have long-term survival.
- Prospective studies are needed on women starting from initial diagnosis in order to determine issues that are particularly relevant to ovarian cancer and to how survivorship changes over time.
- More research is needed on risk factors for specific physical or psychosocial effects.
- More research is needed on how both health care providers and women themselves can better manage the physical and psychosocial effects of treatment, as well as on how health care providers and others can intervene to help women better self-manage their symptoms.
- Providers need to assess physical and psychological symptoms throughout the care continuum.
- Women with ovarian cancer often undergo active treatment until the end of their lives; both women and their providers need a better understanding of quality-of-life issues in order to determine the benefits of continued treatment versus transitioning to end-of-life care.
- As is the case with other cancers, ovarian cancer care is seldom integrated with palliative care, and gynecologic oncologists may need more training in palliative and end-of-life care.
Abbott-Anderson, K., and K. L. Kwekkeboom. 2012. A systematic review of sexual concerns reported by gynecological cancer survivors. Gynecologic Oncology 124(3):477-489.
Alvarez, M., M. Solé, M. Devesa, R. Fábregas, M. Boada, R. Tur, B. Coroleu, A. Veiga, and P. N. Barri. 2014. Live birth using vitrified-warmed oocytes in invasive ovarian cancer: Case report and literature review. Reproductive BioMedicine Online 28(6):663-668.
Andersen, B. L., H. C. Yang, W. B. Farrar, D. M. Golden-Kreutz, C. F. Emery, L. M. Thornton, D. C. Young, and W. E. Carson, 3rd. 2008. Psychologic intervention improves survival for breast cancer patients: A randomized clinical trial. Cancer 113(12):3450-3458.
Andersen, M. R., E. Sweet, K. A. Lowe, L. J. Standish, C. W. Drescher, and B. A. Goff. 2012. Involvement in decision-making about treatment and ovarian cancer survivor quality of life. Gynecologic Oncology 124(3):465-470.
Andersen, M. R., E. Sweet, K. A. Lowe, L. J. Standish, C. W. Drescher, and B. A. Goff. 2013. Dangerous combinations: Ingestible cam supplement use during chemotherapy in patients with ovarian cancer. Journal of Alternative & Complementary Medicine 19(8):714-720.
Anderson, C., J. Carter, K. Nattress, P. Beale, S. Philp, J. Harrison, and I. Juraskova. 2011. “The booklet helped me not to panic”: A pilot of a decision aid for asymptomatic women with ovarian cancer and with rising CA-125 levels. International Journal of Gynecological Cancer 21(4):737-743.
Anderson, N. J., and E. D. Hacker. 2008. Fatigue in women receiving intraperitoneal chemotherapy for ovarian cancer: A review of contributing factors. Clinical Journal of Oncology Nursing 12(3):445-454.
Arriba, L. N., A. N. Fader, H. E. Frasure, and V. E. Von Gruenigen. 2010. A review of issues surrounding quality of life among women with ovarian cancer. Gynecologic Oncology 119(2):390-396.
ASCO (American Society of Clinical Oncology). 2015. Taking charge of your care. http://www.cancer.net/all-about-cancer/cancernet-feature-articles/cancer-basics/self-advocacy-participating-your-cancer-care (accessed October 22, 2015).
Atkinson, T. M., Y. Li, C. W. Coffey, L. Sit, M. Shaw, D. Lavene, A. V. Bennett, M. Fruscione, L. Rogak, J. Hay, M. Gonen, D. Schrag, and E. Basch. 2012. Reliability of adverse symptom event reporting by clinicians. Quality of Life Research 21(7):1159-1164.
Badr, H., K. Basen-Engquist, C. L. C. Taylor, and C. De Moor. 2006. Mood states associated with transitory physical symptoms among breast and ovarian cancer survivors. Journal of Behavioral Medicine 29(5):461-475.
Bakitas, M., K. D. Lyons, M. T. Hegel, S. Balan, F. C. Brokaw, J. Seville, J. G. Hull, Z. Li, T. D. Tosteson, I. R. Byock, and T. A. Ahles. 2009. Effects of a palliative care intervention on clinical outcomes in patients with advanced cancer: The Project Enable II randomized controlled trial. Journal of the American Medical Association 302(7):741-749.
Barlow, J., C. Wright, J. Sheasby, A. Turner, and J. Hainsworth. 2002. Self-management approaches for people with chronic conditions: A review. Patient Education and Counseling 48(2):177-187.
Basch, E. 2010. The missing voice of patients in drug-safety reporting. New England Journal of Medicine 362(10):865-869.
Bernacki, R. E., S. D. Block, and American College of Physicians High Value Care Task Force. 2014. Communication about serious illness care goals: A review and synthesis of best practices. JAMA Internal Medicine 174(12):1994-2003.
Billson, H. A., C. Holland, J. Curwell, V. L. Davey, L. Kinsey, L. J. Lawton, A. J. Whitworth, and S. Burden. 2013. Perioperative nutrition interventions for women with ovarian cancer. Cochrane Database of Systematic Reviews (9).
Bodenheimer, T., K. Lorig, H. Holman, and K. Grumbach. 2002. Patient self-management of chronic disease in primary care. Journal of the American Medical Association 288(19): 2469-2475.
Bodurka-Bevers, D., K. Basen-Engquist, C. L. Carmack, M. A. Fitzgerald, J. K. Wolf, C. De Moor, and D. M. Gershenson. 2000. Depression, anxiety, and quality of life in patients with epithelial ovarian cancer. Gynecologic Oncology 78(3 I):302-308.
Brashers, D. E., J. L. Neidig, and D. J. Goldsmith. 2004. Social support and the management of uncertainty for people living with HIV or AIDS. Health Communication 16(3):305-331.
Browall, M., M. Carlsson, and G. Horvath. 2004. Information needs of women with recently diagnosed ovarian cancer—A longitudinal study. European Journal of Oncology Nursing 8(3):200-207.
Brown, A. J., C. C. Sun, L. S. Prescott, J. S. Taylor, L. M. Ramondetta, and D. C. Bodurka. 2014. Missed opportunities: Patterns of medical care and hospice utilization among ovarian cancer patients. Gynecologic Oncology 135(2):244-248.
Brumley, R., S. Enguidanos, P. Jamison, R. Seitz, N. Morgenstern, S. Saito, J. McIlwane, K. Hillary, and J. Gonzalez. 2007. Increased satisfaction with care and lower costs: Results of a randomized trial of in-home palliative care. Journal of the American Geriatrics Society 55(7):993-1000.
Buković, D., H. Silovski, T. Silovski, I. Hojsak, K. Šakić, and Z. Hrgović. 2008. Sexual functioning and body image of patients treated for ovarian cancer. Sexuality and Disability 26(2):63-73.
Cannioto, R. A., and K. B. Moysich. 2015. Epithelial ovarian cancer and recreational physical activity: A review of the epidemiological literature and implications for exercise prescription. Gynecologic Oncology 137(3):559-573.
Carmack Taylor, C. L., K. Basen-Engquist, E. H. Shinn, and D. C. Bodurka. 2004. Predictors of sexual functioning in ovarian cancer patients. Journal of Clinical Oncology 22(5):881-889.
Cella, D., W. Riley, A. Stone, N. Rothrock, B. Reeve, et al. 2010. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005–2008. Journal of Clinical Epidemiology 63(11):1179-1194.
Champion, V., S. D. Williams, A. Miller, K. M. Reuille, K. Wagler-Ziner, P. O. Monahan, Q. Zhao, D. Gershenson, and D. Cella. 2007. Quality of life in long-term survivors of ovarian germ cell tumors: A Gynecologic Oncology Group study. Gynecologic Oncology 105(3):687-694.
Chan, K. K., T. J. Yao, B. Jones, J. F. Zhao, F. K. Ma, C. Y. Leung, S. K. Lau, M. W. Yip, and H. Y. Ngan. 2011. The use of Chinese herbal medicine to improve quality of life in women undergoing chemotherapy for ovarian cancer: A double-blind placebo-controlled randomized trial with immunological monitoring. Annals of Oncology 22(10):2241-2249.
Chase, D. M., and L. Wenzel. 2011. Health-related quality of life in ovarian cancer patients and its impact on clinical management. Expert Review of Pharmacoeconomics & Outcomes Research 11(4):421-431.
Chase, D. M., L. B. Wenzel, and B. J. Monk. 2012. Quality-of-life results used to endorse changes in standard of care for recurrent platinum-sensitive ovarian cancer. Expert Review of Pharmacoeconomics & Outcomes Research 12(3):279-281.
Chase, D. M., H. Huang, C. D. Foss, L. B. Wenzel, B. J. Monk, and R. A. Burger. 2015. Neurotoxicity in ovarian cancer patients on Gynecologic Oncology Group (GOG) protocol 218: Characteristics associated with toxicity and the effect of substitution with docetaxel: An NRG Oncology/Gynecologic Oncology Group study. Gynecologic Oncology 136(2):323-327.
Chida, Y., M. Hamer, J. Wardle, and A. Steptoe. 2008. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nature Clinical Practice Oncology 5(8):466-475.
Choi, C. H., M. K. Kim, J. Y. Park, A. Yoon, H. J. Kim, Y. Y. Lee, T. J. Kim, J. W. Lee, B. G. Kim, and D. S. Bae. 2014. Safety and efficacy of aprepitant, ramosetron, and dexamethasone for chemotherapy-induced nausea and vomiting in patients with ovarian cancer treated with paclitaxel/carboplatin. Supportive Care in Cancer 22(5):1181-1187.
Christakis, N. A., and T. J. Iwashyna. 2000. Impact of individual and market factors on the timing of initiation of hospice terminal care. Medical Care 38(5):528-541.
Cimprich, B., N. K. Janz, L. Northouse, P. A. Wren, B. Given, and C. W. Given. 2005. Taking charge: A self-management program for women following breast cancer treatment. Psychooncology 14(9):704-717.
Cleeland, C. S., and J. A. Sloan. 2010. Assessing the symptoms of cancer using patient-reported outcomes (ASCPRO): Searching for standards. Journal of Pain and Symptom Management 39(6):1077-1085.
Clevenger, L., A. Schrepf, D. Christensen, K. DeGeest, D. Bender, A. Ahmed, M. J. Goodheart, F. Penedo, D. M. Lubaroff, A. K. Sood, and S. K. Lutgendorf. 2012. Sleep disturbance, cytokines, and fatigue in women with ovarian cancer. Brain, Behavior, and Immunity 26(7):1037-1044.
Clevenger, L., A. Schrepf, K. DeGeest, D. Bender, M. Goodheart, A. Ahmed, L. Dahmoush, F. Penedo, J. Lucci III, P. H. Thaker, L. Mendez, A. K. Sood, G. M. Slavich, and S. K. Lutgendorf. 2013. Sleep disturbance, distress, and quality of life in ovarian cancer patients during the first year after diagnosis. Cancer 119(17):3234-3241.
Cockle-Hearne, J., and S. Faithfull. 2010. Self-management for men surviving prostate cancer: A review of behavioural and psychosocial interventions to understand what strategies can work, for whom and in what circumstances. Psychooncology 19(9):909-922.
Connor, S. R., B. Pyenson, K. Fitch, C. Spence, and K. Iwasaki. 2007. Comparing hospice and nonhospice patient survival among patients who die within a three-year window. Journal of Pain and Symptom Management 33(3):238-246.
Costanzo, E. S., S. K. Lutgendorf, A. K. Sood, B. Andersen, J. Sorosky, and D. M. Lubaroff. 2005. Psychosocial factors and interleukin-6 among women with advanced ovarian cancer. Cancer 104(2):305-313.
Dalal, S., S. Palla, D. Hui, L. Nguyen, R. Chacko, Z. Li, N. Fadul, C. Scott, V. Thornton, B. Coldman, Y. Amin, and E. Bruera. 2011. Association between a name change from palliative to supportive care and the timing of patient referrals at a comprehensive cancer center. Oncologist 16(1):105-111.
Danhauer, S. C., J. A. Tooze, D. F. Farmer, C. R. Campbell, R. P. McQuellon, R. Barrett, and B. E. Miller. 2008. Restorative yoga for women with ovarian or breast cancer: Findings from a pilot study. Journal of the Society for Integrative Oncology 6(2):47-58.
Davis, R. M., E. G. Wagner, and T. Groves. 2000. Advances in managing chronic disease. Research, performance measurement, and quality improvement are key. BMJ 320(7234): 525-526.
Deshpande, N. A., I. M. Braun, and F. L. Meyer. 2015. Impact of fertility preservation counseling and treatment on psychological outcomes among women with cancer: A systematic review. Cancer (Epub ahead of print).
Dittrich, R., J. Hackl, L. Lotz, I. Hoffmann, and M. W. Beckmann. 2015. Pregnancies and live births after 20 transplantations of cryopreserved ovarian tissue in a single center. Fertility and Sterility 103(2):462-468.
Doll, K. M., J. E. Stine, L. Van Le, D. T. Moore, V. Bae-Jump, W. R. Brewster, J. T. Soper, J. F. Boggess, P. A. Gehrig, and K. H. Kim. 2013. Outpatient end of life discussions shorten hospital admissions in gynecologic oncology patients. Gynecologic Oncology 130(1):152-155.
Donovan, H. S., E. M. Hartenbach, and M. W. Method. 2005. Patient–provider communication and perceived control for women experiencing multiple symptoms associated with ovarian cancer. Gynecologic Oncology 99(2):404-411.
Donovan, H. S., S. E. Ward, S. M. Sereika, J. E. Knapp, P. R. Sherwood, C. M. Bender, R. P. Edwards, M. Fields, and R. Ingel. 2014a. Web-based symptom management for women with recurrent ovarian cancer: A pilot randomized controlled trial of the WRITE Symptoms intervention. Journal of Pain and Symptom Management 47(2):218-230.
Donovan, K. A., H. S. Donovan, D. Cella, M. E. Gaines, R. T. Penson, S. C. Plaxe, V. E. von Gruenigen, D. W. Bruner, B. B. Reeve, and L. Wenzel. 2014b. Recommended patient-reported core set of symptoms and quality-of-life domains to measure in ovarian cancer treatment trials. Journal of the National Cancer Institute 106(7):dju128.
Dueck, A. C., T. R. Mendoza, S. A. Mitchell, B. B. Reeve, K. M. Castro, et al. 2015. Validity and reliability of the U.S. National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). JAMA Oncology (Epub ahead of print).
Dunbar, S. B., P. C. Clark, C. Quinn, R. A. Gary, and N. J. Kaslow. 2008. Family influences on heart failure self-care and outcomes. Journal of Cardiovascular Nursing 23(3):258-265.
Elit, L., C. Charles, I. Gold, A. Gafni, S. Farrell, S. Tedford, D. Dal Bello, and T. Whelan. 2003. Women’s perceptions about treatment decision making for ovarian cancer. Gynecologic Oncology 88(2):89-95.
Eskander, R. N., K. Osann, E. Dickson, L. L. Holman, J. A. Rauh-Hain, L. Spoozak, E. Wu, L. Krill, A. N. Fader, and K. S. Tewari. 2014. Assessment of palliative care training in gynecologic oncology: A gynecologic oncology fellow research network study. Gynecologic Oncology 134(2):379-384.
Ezendam, N. P., B. Pijlman, C. Bhugwandass, J. F. Pruijt, F. Mols, M. C. Vos, J. M. Pijnenborg, and L. V. van de Poll-Franse. 2014. Chemotherapy-induced peripheral neuropathy and its impact on health-related quality of life among ovarian cancer survivors: Results from the population-based profiles registry. Gynecologic Oncology 135(3):510-517.
Fadul, N., A. Elsayem, J. L. Palmer, E. Del Fabbro, K. Swint, Z. Li, V. Poulter, and E. Bruera. 2009. Supportive versus palliative care: What’s in a name?: A survey of medical oncologists and midlevel providers at a comprehensive cancer center. Cancer 115(9):2013-2021.
Fairfield, K. M., K. M. Murray, H. R. Wierman, P. K. Han, S. Hallen, S. Miesfeldt, E. L. Trimble, J. L. Warren, and C. C. Earle. 2012. Disparities in hospice care among older women dying with ovarian cancer. Gynecologic Oncology 125(1):14-18.
FDA (U.S. Food and Drug Administration). 2009. Guidance for industry patient-reported outcome measures: Use in medical product development to support labeling claims. http://www.fda.gov/downloads/drugs/guidances/ucm193282 (accessed October 21, 2015).
Ferguson, M. J. 2011. The decisions study: Synopsis of evidence for shared decision-making and quality patient–provider communication. Translational Behavioral Medicine 1(2): 205-206.
Ferrell, B., S. Smith, C. Cullinane, and C. Melancon. 2003a. Symptom concerns of women with ovarian cancer. Journal of Pain and Symptom Management 25(6):528-538.
Ferrell, B., S. L. Smith, C. A. Cullinane, and C. Melancon. 2003b. Psychological well-being and quality of life in ovarian cancer survivors. Cancer 98(5):1061-1071.
Ferrell, B. R., S. L. Smith, K. S. Ervin, J. Itano, and C. Melancon. 2003c. A qualitative analysis of social concerns of women with ovarian cancer. Psychooncology 12(7):647-663.
Ferrell, B. R., S. L. Smith, G. Juarez, and C. Melancon. 2003d. Meaning of illness and spirituality in ovarian cancer survivors. Oncology Nursing Forum 30(2):249-257.
Ferrell, B., C. A. Cullinane, K. Ervin, C. Melancon, G. C. Uman, and G. Juarez. 2005. Perspectives on the impact of ovarian cancer: Women’s views of quality of life. Oncology Nursing Forum 32(6):1143-1149.
Ferris, F. D., E. Bruera, N. Cherny, C. Cummings, D. Currow, D. Dudgeon, N. Janjan, F. Strasser, C. F. von Gunten, and J. H. Von Roenn. 2009. Palliative cancer care a decade later: Accomplishments, the need, next steps—from the American Society of Clinical Oncology. Journal of Clinical Oncology 27(18):3052-3058.
Fitch, M. I., K. Deane, and D. Howell. 2003. Living with ovarian cancer: Women’s perspectives on treatment and treatment decision-making. Canadian Oncology Nursing Journal 13(1):8-20.
FORCE (Facing Our Risk of Cancer Empowered). 2015. Our mission. http://www.facingour-risk.org/our-role-and-impact/our-impact/our-mission.php (accessed October 23, 2015).
Foster, C., and D. Fenlon. 2011. Recovery and self-management support following primary cancer treatment. British Journal of Cancer 105(Suppl 1):S21-S28.
Fox, S. W., and D. Lyon. 2007. Symptom clusters and quality of life in survivors of ovarian cancer. Cancer Nursing 30(5):354-361.
Fredericks, S., J. Lapum, and J. Lo. 2012. Anxiety, depression, and self-management: A systematic review. Clinical Nursing Research 21(4):411-430.
Fried, T. R., E. H. Bradley, and J. O—Leary. 2003. Prognosis communication in serious illness: Perceptions of older patients, caregivers, and clinicians. Journal of the American Geriatrics Society 51(10):1398-1403.
Friedlander, M. L., and M. T. King. 2013. Patient-reported outcomes in ovarian cancer clinical trials. Annals of Oncology 24(Suppl 10):x64-x68.
Friedman, B. T., M. K. Harwood, and M. Shields. 2002. Barriers and enablers to hospice referrals: An expert overview. Journal of Palliative Medicine 5(1):73-84.
Gadducci, A., S. Cosio, A. Fanucchi, and A. R. Genazzani. 2001. Malnutrition and cachexia in ovarian cancer patients: Pathophysiology and management. Anticancer Research 21(4 B):2941-2947.
Gade, G., I. Venohr, D. Conner, K. McGrady, J. Beane, R. H. Richardson, M. P. Williams, M. Liberson, M. Blum, and R. Della Penna. 2008. Impact of an inpatient palliative care team: A randomized control trial. Journal of Palliative Medicine 11(2):180-190.
Gazelle, G. 2007. Understanding hospice—An underutilized option for life’s final chapter. New England Journal of Medicine 357(4):321-324.
Geisler, J. P., G. C. Linnemeier, A. J. Thomas, and K. J. Manahan. 2007. Nutritional assessment using prealbumin as an objective criterion to determine whom should not undergo primary radical cytoreductive surgery for ovarian cancer. Gynecologic Oncology 106(1):128-131.
Gershenson, D. M., A. M. Miller, V. L. Champion, P. O. Monahan, Q. Zhao, D. Cella, and S. D. Williams. 2007. Reproductive and sexual function after platinum-based chemotherapy in long-term ovarian germ cell tumor survivors: A Gynecologic Oncology Group study. Journal of Clinical Oncology 25(19):2792-2797.
Glare, P., K. Virik, M. Jones, M. Hudson, S. Eychmuller, J. Simes, and N. Christakis. 2003. A systematic review of physicians’ survival predictions in terminally ill cancer patients. BMJ 327(7408):195-198.
Glaser, G., L. Hartman, W. Cliby, M. Torres, Z. Tabbaa, K. Kalli, A. Weaver, A. Jatoi, and A. Mariani. 2012. Impact of neoadjuvant chemotherapy (NACT) on nutritional status and treatment-related morbidity in medically unfit women with advanced ovarian cancer (AOC). Gynecologic Oncology 125(2):S192-S193.
Goldsmith, B., J. Dietrich, Q. Du, and R. S. Morrison. 2008. Variability in access to hospital palliative care in the United States. Journal of Palliative Medicine 11(8):1094-1102.
GOG (Gynecologic Oncology Group). 2015. Diet and physical activity change or usual care in improving progression-free survival in patients with previously treated stage II, III, or IV ovarian, fallopian tube, or primary peritoneal cancer. Bethesda, MD: National Library of Medicine. https://clinicaltrials.gov/ct2/show/NCT00719303 (accessed December 13, 2015).
Granger, B. B., M. Sandelowski, H. Tahshjain, K. Swedberg, and I. Ekman. 2009. A qualitative descriptive study of the work of adherence to a chronic heart failure regimen: Patient and physician perspectives. Journal of Cardiovascular Nursing 24(4):308-315.
Grey, M., K. Knafl, and R. McCorkle. 2006. A framework for the study of self- and family management of chronic conditions. Nursing Outlook 54(5):278-286.
Gunnarsdottir, S., H. S. Donovan, R. C. Serlin, C. Voge, and S. Ward. 2002. Patient-related barriers to pain management: The Barriers Questionnaire II (BQ-II). Pain 99(3):385-396.
Hagan, T. L., and H. S. Donovan. 2013a. Ovarian cancer survivors’ experiences of self-advocacy: A focus group study. Oncology Nursing Forum 40(2):140-147.
Hagan, T. L., and H. S. Donovan. 2013b. Self-advocacy and cancer: A concept analysis. Journal of Advanced Nursing 69(10):2348-2359.
Hammer, M. J., E. A. Ercolano, F. Wright, V. V. Dickson, D. Chyun, and G. D. E. Melkus. 2015. Self-management for adult patients with cancer: An integrative review. Cancer Nursing 38(2):E10-E26.
Han, E. S., P. Lin, and M. Wakabayashi. 2009. Current status on biologic therapies in the treatment of epithelial ovarian cancer. Current Treatment Options in Oncology 10(1-2):54-66.
Helpman, L., S. E. Ferguson, M. MacKean, A. Rana, L. Le, M. A. Atkinson, A. Rogerson, and H. MacKay. 2011. Complementary and alternative medicine use among women receiving chemotherapy for ovarian cancer in 2 patient populations. International Journal of Gynecological Cancer 21(3):587-593.
Helpman Bek, L., S. E. Ferguson, M. Mackean, L. Le, A. Rogerson, A. Dewan, and H. Mackay. 2009. Use of complementary medicine (CAM) among women receiving chemotherapy for ovarian cancer: A comparison of attitudes between two patient populations. Journal of Clinical Oncology 1:e20545.
Henes, M., F. Neis, B. Krämer, C. Walter, S. Brucker, M. Von Wolff, R. Rothmund, B. Lawrenz, and K. Rall. 2014. Possibilities of fertility preservation in young patients with ovarian cancer. Anticancer Research 34(7):3851-3854.
Hennemann-Krause, L., A. J. Lopes, J. A. Araujo, E. M. Petersen, and R. A. Nunes. 2014. The assessment of telemedicine to support outpatient palliative care in advanced cancer. Palliative and Supportive Care 1-6.
Hoffman, A. J., R. A. Brintnall, J. K. Brown, A. V. Eye, L. W. Jones, G. Alderink, D. Ritz-Holland, M. Enter, L. H. Patzelt, and G. M. Vanotteren. 2013. Too sick not to exercise: Using a 6-week, home-based exercise intervention for cancer-related fatigue self-management for postsurgical non-small cell lung cancer patients. Cancer Nursing 36(3):175-188.
Homsi, J., D. Walsh, N. Rivera, L. A. Rybicki, K. A. Nelson, S. B. LeGrand, M. Davis, M. Naughton, D. Gvozdjan, and H. Pham. 2006. Symptom evaluation in palliative medicine: Patient report vs systematic assessment. Supportive Care in Cancer 14(5):444-453.
Houle, J., M. Gascon-Depatie, G. Bélanger-Dumontier, and C. Cardinal. 2013. Depression self-management support: A systematic review. Patient Education and Counseling 91(3): 271-279.
Hui, D., A. Elsayem, M. De la Cruz, A. Berger, D. S. Zhukovsky, S. Palla, A. Evans, N. Fadul, J. L. Palmer, and E. Bruera. 2010. Availability and integration of palliative care at U.S. cancer centers. Journal of the American Medical Association 303(11):1054-1061.
Huisman, M. G., B. L. Van Leeuwen, G. Ugolini, I. Montroni, J. Spiliotis, C. Stabilini, N. D. Carino, E. Farinella, G. H. De Bock, and R. A. Audisio. 2014. “Timed up & go”: A screening tool for predicting 30-day morbidity in onco-geriatric surgical patients? A multicenter cohort study. PLoS ONE 9(1):e86863.
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
IOM. 2003. Priority areas for national action: Transforming health care quality. Washington, DC: The National Academies Press.
IOM. 2006. From cancer patient to cancer survivor: Lost in transition. Washington, DC: The National Academies Press.
IOM. 2008. Cancer care for the whole patient: Meeting psychosocial health needs. Washington, DC: The National Academies Press.
IOM. 2010. A national cancer clinical trials system for the 21st century: Reinvigorating the NCI Cooperative Group Program. Washington, DC: The National Academies Press.
IOM. 2013. Delivering high-quality cancer care: Charting a new course for a system in crisis. Washington, DC: The National Academies Press.
IOM. 2015. Dying in America: Improving quality and honoring individual preferences near the end of life. Washington, DC: The National Academies Press.
Jackson, J. M., S. J. Rolnick, S. S. Coughlin, C. Neslund-Dudas, M. C. Hornbrook, J. Darbinian, D. J. Bachman, and L. J. Herrinton. 2007. Social support among women who died of ovarian cancer. Supportive Care in Cancer 15(5):547-556.
Jolicoeur, L. J., A. M. O—Connor, L. Hopkins, and I. D. Graham. 2009. Women’s decision-making needs related to treatment for recurrent ovarian cancer: A pilot study. Canadian Oncology Nursing Journal 19(3):117-121.
Josephs-Cowan, C. 2006. Peripheral neuropathy in the ovarian cancer patient. Journal of Gynecologic Oncology Nursing 16(1):6-11.
Juan, A. S., C. Wakefield, N. A. Kasparian, J. Kirk, J. Tyler, and K. Tucker. 2008. Development and pilot testing of a decision aid for men considering genetic testing for breast and/or ovarian cancer-related mutations (BRCA1/2). Genetic Testing 12(4):523-532.
Keyser, E. A., B. G. Reed, W. J. Lowery, M. J. Sundborg, W. E. Winter, 3rd, J. A. Ward, and C. A. Leath, 3rd. 2010. Hospice enrollment for terminally ill patients with gynecologic malignancies: Impact on outcomes and interventions. Gynecologic Oncology 118(3):274-277.
King, M. T., M. R. Stockler, P. Butow, R. O—Connell, M. Voysey, A. M. Oza, K. Gillies, H. S. Donovan, R. Mercieca-Bebber, J. Martyn, K. Sjoquist, and M. L. Friedlander. 2014. Development of the measure of ovarian symptoms and treatment concerns: Aiming for optimal measurement of patient-reported symptom benefit with chemotherapy for symptomatic ovarian cancer. International Journal of Gynecological Cancer 24(5):865-873.
Kitamura, Y. 2010. Decision-making process of patients with gynecological cancer regarding their cancer treatment choices using the analytic hierarchy process. Japan Journal of Nursing Science 7(2):148-157.
Koller, A., C. Miaskowski, S. De Geest, O. Opitz, and E. Spichiger. 2012. A systematic evaluation of content, structure, and efficacy of interventions to improve patients’ self-management of cancer pain. Journal of Pain and Symptom Management 44(2):264-284.
Kornblith, A. B., K. Mirabeau-Beale, H. Lee, A. K. Goodman, R. T. Penson, L. Pereira, and U. A. Matulonis. 2010. Long-term adjustment of survivors of ovarian cancer treated for advanced-stage disease. Journal of Psychosocial Oncology 28(5):451-469.
Lambertini, M., E. S. Ginsburg, and A. H. Partridge. 2015. Update on fertility preservation in young women undergoing breast cancer and ovarian cancer therapy. Current Opinion in Obstetrics and Gynecology 27(1):98-107.
Landrum, L. M., S. Blank, L. M. Chen, L. Duska, V. Bae-Jump, P. S. Lee, L. Levine, C. McCourt, K. N. Moore, and R. R. Urban. 2015. Comprehensive care in gynecologic oncology: The importance of palliative care. Gynecologic Oncology 137(2):193-202.
Lee, M. K., H. A. Park, Y. H. Yun, and Y. J. Chang. 2013. Development and formative evaluation of a Web-based self-management exercise and diet intervention program with tailored motivation and action planning for cancer survivors. Journal of Medical Internet Research 15(2).
Lee, S. J., L. R. Schover, A. H. Partridge, P. Patrizio, W. H. Wallace, K. Hagerty, L. N. Beck, L. V. Brennan, and K. Oktay. 2006. American Society of Clinical Oncology recommendations on fertility preservation in cancer patients. Journal of Clinical Oncology 24(18):2917-2931.
Lefkowits, C., P. Sukumvanich, R. Claxton, M. Courtney-Brooks, J. L. Kelley, M. A. McNeil, and A. Goodman. 2014. Needs assessment of palliative care education in gynecologic oncology fellowship: We—re not teaching what we think is most important. Gynecologic Oncology 135(2):255-260.
Lefkowits, C., W. Teuteberg, M. Courtney-Brooks, P. Sukumvanich, R. Ruskin, and J. L. Kelley. 2015. Improvement in symptom burden within one day after palliative care consultation in a cohort of gynecologic oncology inpatients. Gynecologic Oncology 136(3):424-428.
Lesnock, J. L., R. M. Arnold, L. A. Meyn, M. K. Buss, M. Quimper, T. C. Krivak, R. P. Edwards, and J. C. Chang. 2013. Palliative care education in Gynecologic Oncology: A survey of the fellows. Gynecologic Oncology 130(3):431-435.
Letourneau, J., J. Chan, W. Salem, S. W. Chan, M. Shah, E. Ebbel, C. McCulloch, L. M. Chen, M. Cedars, and M. Rosen. 2015. Fertility sparing surgery for localized ovarian cancers maintains an ability to conceive, but is associated with diminished reproductive potential. Journal of Surgical Oncology 112(1):26-30.
Levy, M. H., A. Back, C. Benedetti, J. A. Billings, S. Block, B. Boston, E. Bruera, S. Dy, C. Eberle, K. M. Foley, S. B. Karver, S. J. Knight, S. Misra, C. S. Ritchie, D. Spiegel, L. Sutton, S. Urba, J. H. Von Roenn, and S. M. Weinstein. 2009. NCCN clinical practice guidelines in oncology: Palliative care. Journal of the National Comprehensive Cancer Network 7(4):436-473.
Lewin, S. N., B. M. Buttin, M. A. Powell, R. K. Gibb, J. S. Rader, D. G. Mutch, and T. J. Herzog. 2005. Resource utilization for ovarian cancer patients at the end of life: How much is too much? Gynecologic Oncology 99(2):261-266.
Liavaag, A. H., A. Dørum, S. D. Fosså, C. Tropé, and A. A. Dahl. 2007. Controlled study of fatigue, quality of life, and somatic and mental morbidity in epithelial ovarian cancer survivors: How lucky are the lucky ones? Journal of Clinical Oncology 25(15):2049-2056.
Liavaag, A. H., A. Dørum, T. Bjøro, H. Oksefjell, S. D. Fosså, C. Tropé, and A. A. Dahl. 2008. A controlled study of sexual activity and functioning in epithelial ovarian cancer survivors: A therapeutic approach. Gynecologic Oncology 108(2):348-354.
Lockwood-Rayermann, S. 2006. Survivorship issues in ovarian cancer: A review. Oncology Nursing Forum 33(3):553-562.
Lopez-Acevedo, M., L. J. Havrilesky, G. Broadwater, A. H. Kamal, A. P. Abernethy, A. Berchuck, A. Alvarez Secord, J. A. Tulsky, F. Valea, and P. S. Lee. 2013a. Timing of end-of-life care discussion with performance on end-of-life quality indicators in ovarian cancer. Gynecologic Oncology 130(1):156-161.
Lopez-Acevedo, M., W. J. Lowery, A. W. Lowery, P. S. Lee, and L. J. Havrilesky. 2013b. Palliative and hospice care in gynecologic cancer: A review. Gynecologic Oncology 131(1):215-221.
Lorig, K., and H. Holman. 2003. Self-management education: History, definition, outcomes, and mechanisms. Annals of Behavioral Medicine 26(1):1-7.
Lowe, K. A., M. R. Andersen, E. Sweet, L. Standish, C. W. Drescher, and B. A. Goff. 2012. The effect of regular exercise and yoga on health-related quality of life among ovarian cancer survivors. Journal of Evidence-Based Complementary and Alternative Medicine 17(3):155-160.
Lowery, W. J., A. W. Lowery, J. C. Barnett, M. Lopez-Acevedo, P. S. Lee, A. A. Secord, and L. Havrilesky. 2013. Cost-effectiveness of early palliative care intervention in recurrent platinum-resistant ovarian cancer. Gynecologic Oncology 130(3):426-430.
Lu, W., U. A. Matulonis, A. Doherty-Gilman, H. Lee, E. Dean-Clower, A. Rosulek, C. Gibson, A. Goodman, R. B. Davis, J. E. Buring, P. M. Wayne, D. S. Rosenthal, and R. T. Penson. 2009. Acupuncture for chemotherapy-induced neutropenia in patients with gynecologic malignancies: A pilot randomized, sham-controlled clinical trial. Journal of Alternative and Complementary Medicine 15(7):745-753.
Luketina, H., C. Fotopoulou, R. R. Luketina, A. Pilger, and J. Sehouli. 2012. Treatment decision-making processes in the systemic treatment of ovarian cancer: Review of the scientific evidence. Anticancer Research 32(9):4085-4090.
Lutgendorf, S. K., and B. L. Andersen. 2015. Biobehavioral approaches to cancer progression and survival: Mechanisms and interventions. American Psychologist 70(2):186-197.
Lutgendorf, S. K., E. L. Johnsen, B. Cooper, B. Anderson, J. I. Sorosky, R. E. Buller, and A. K. Sood. 2002. Vascular endothelial growth factor and social support in patients with ovarian carcinoma. Cancer 95(4):808-815.
Lutgendorf, S. K., A. K. Sood, B. Anderson, S. McGinn, H. Maiseri, M. Dao, J. I. Sorosky, K. De Geest, J. Ritchie, and D. M. Lubaroff. 2005. Social support, psychological distress, and natural killer cell activity in ovarian cancer. Journal of Clinical Oncology 23(28):7105-7113.
Lutgendorf, S. K., D. M. Lamkin, N. B. Jennings, J. M. G. Arevalo, F. Penedo, K. DeGeest, R. R. Langley, J. A. Lucci III, S. W. Cole, D. M. Lubaroff, and A. K. Sood. 2008. Biobehavioral influences on matrix metalloproteinase expression in ovarian carcinoma. Clinical Cancer Research 14(21):6839-6846.
Lutgendorf, S. K., K. DeGeest, C. Y. Sung, J. M. Arevalo, F. Penedo, J. Lucci, 3rd, M. Goodheart, D. Lubaroff, D. M. Farley, A. K. Sood, and S. W. Cole. 2009. Depression, social support, and beta-adrenergic transcription control in human ovarian cancer. Brain, Behavior, and Immunity 23(2):176-183.
Lutgendorf, S. K., K. DeGeest, L. Dahmoush, D. Farley, F. Penedo, D. Bender, M. Goodheart, T. E. Buekers, L. Mendez, G. Krueger, L. Clevenger, D. M. Lubaroff, A. K. Sood, and S. W. Cole. 2011. Social isolation is associated with elevated tumor norepinephrine in ovarian carcinoma patients. Brain, Behavior, and Immunity 25(2):250-255.
Lutgendorf, S. K., K. De Geest, D. Bender, A. Ahmed, M. J. Goodheart, L. Dahmoush, M. B. Zimmerman, F. J. Penedo, J. A. Lucci III, P. Ganjei-Azar, P. H. Thaker, L. Mendez, D. M. Lubaroff, G. M. Slavich, S. W. Cole, and A. K. Sood. 2012. Social influences on clinical outcomes of patients with ovarian cancer. Journal of Clinical Oncology 30(23):2885-2890.
Maeng, D. D., G. R. Martsolf, D. P. Scanlon, and J. B. Christianson. 2012. Care coordination for the chronically ill: Understanding the patient’s perspective. Health Services Research 47(5):1960-1979.
Matei, D., A. M. Miller, P. Monahan, D. Gershenson, Q. Zhao, D. Cella, V. L. Champion, and S. D. Williams. 2009. Chronic physical effects and health care utilization in long-term ovarian germ cell tumor survivors: A Gynecologic Oncology Group study. Journal of Clinical Oncology 27(25):4142-4149.
Matulonis, U. A., A. Kornblith, H. Lee, J. Bryan, C. Gibson, C. Wells, J. Lee, L. Sullivan, and R. Penson. 2008. Long-term adjustment of early-stage ovarian cancer survivors. International Journal of Gynecological Cancer 18(6):1183-1193.
McCorkle, R., J. Pasacreta, and S. T. Tang. 2003. The silent killer: Psychological issues in ovarian cancer. Holistic Nursing Practice 17(6):300-308.
McCorkle, R., M. Dowd, E. Ercolano, D. Schulman-Green, A.-L. Williams, M. L. Siefert, J. Steiner, and P. Schwartz. 2009. Effects of a nursing intervention on quality of life outcomes in post-surgical women with gynecological cancers. Psychooncology 18(1):62-70.
McCorkle, R., E. Ercolano, M. Lazenby, D. Schulman-Green, L. S. Schilling, K. Lorig, and E. H. Wagner. 2011. Self-management: Enabling and empowering patients living with cancer as a chronic illness. CA: A Cancer Journal for Clinicians 61(1):50-62.
McInturff, B., and E. Harrington. 2011. 2011 public opinion research on palliative care: A report based on research by public opinion strategies. https://media.capc.org/filer_public/18/ab/18ab708c-f835-4380-921d-fbf729702e36/2011-public-opinion-research-on-palliative-care.pdf (accessed October 16, 2015).
Meyers, F. J., M. Carducci, M. J. Loscalzo, J. Linder, T. Greasby, and L. A. Beckett. 2011. Effects of a problem-solving intervention (COPE) on quality of life for patients with advanced cancer on clinical trials and their caregivers: Simultaneous care educational intervention (SCEI): Linking palliation and clinical trials. Journal of Palliative Medicine 14(4):465-473.
Mirabeau-Beale, K. L., A. B. Kornblith, R. T. Penson, H. Lee, A. Goodman, S. M. Campos, L. Duska, L. Pereira, J. Bryan, and U. A. Matulonis. 2009. Comparison of the quality of life of early and advanced stage ovarian cancer survivors. Gynecologic Oncology 114(2):353-359.
Mishel, M. H. 1981. The measurement of uncertainty in illness. Nursing Research 30(5): 258-263.
Mizrahi, D., F. Naumann, C. Broderick, M. Ryan, and M. Friedlander. 2013. Physical activity and sleep disturbances for women with recurrent ovarian cancer undergoing chemotherapy. International Journal of Gynecological Cancer 1:949.
Monahan, P. O., V. L. Champion, Q. Zhao, A. M. Miller, D. Gershenson, S. D. Williams, and D. Cella. 2008. Case-control comparison of quality of life in long-term ovarian germ cell tumor survivors: A Gynecologic Oncology Group study. Journal of Psychosocial Oncology 26(3):19-42.
Morice, P., D. Denschlag, A. Rodolakis, N. Reed, A. Schneider, V. Kesic, and N. Colombo. 2011. Recommendations of the fertility task force of the European Society of Gynecologic Oncology about the conservative management of ovarian malignant tumors. International Journal of Gynecological Cancer 21(5):951-963.
Morrison, R. S., J. D. Penrod, J. B. Cassel, M. Caust-Ellenbogen, A. Litke, L. Spragens, D. E. Meier, and G. Palliative Care Leadership Centers’ Outcomes. 2008. Cost savings associated with U.S. hospital palliative care consultation programs. Archives of Internal Medicine 168(16):1783-1790.
Morrison, R. S., J. Dietrich, S. Ladwig, T. Quill, J. Sacco, J. Tangeman, and D. E. Meier. 2011. Palliative care consultation teams cut hospital costs for medicaid beneficiaries. Health Affairs 30(3):454-463.
Nagle, C. M., S. C. Dixon, A. Jensen, S. K. Kjaer, F. Modugno, et al. 2015. Obesity and survival among women with ovarian cancer: Results from the Ovarian Cancer Association Consortium. British Journal of Cancer 113(5):817-826.
Nausheen, B., Y. Gidron, R. Peveler, and R. Moss-Morris. 2009. Social support and cancer progression: A systematic review. Journal of Psychosomatic Research 67(5):403-415.
Nevadunsky, N. S., S. Gordon, L. Spoozak, A. Van Arsdale, Y. Hou, M. Klobocista, S. Eti, B. Rapkin, and G. L. Goldberg. 2014. The role and timing of palliative medicine consultation for women with gynecologic malignancies: Association with end of life interventions and direct hospital costs. Gynecologic Oncology 132(1):3-7.
NIH (National Institutes of Health). 2013. Symptom management in patients with recurrent or persistent ovarian cancer, fallopian tube cancer, or primary peritoneal cancer: Nct00958698. https://clinicaltrials.gov/ct2/show/NCT00958698 (accessed October 21, 2015).
NINR (National Institute for Nursing Research). 2015. Self-management: Improving quality of life for individuals with chronic illness. http://www.ninr.nih.gov/newsandinformation/iq/self-management-workshop#.Vie2Jn6rSHt (accessed October 21, 2015).
NOCC (National Ovarian Cancer Coalition). 2015. Programs. http://www.ovarian.org/programs.php (accessed October 22, 2015).
Norris, S. L., M. M. Engelgau, and K. M. V. Narayan. 2001. Effectiveness of self-management training in type 2 diabetes: A systematic review of randomized controlled trials. Diabetes Care 24(3):561-587.
Norris, S. L., P. J. Nichols, C. J. Caspersen, R. E. Glasgow, M. M. Engelgau, L. Jack, Jr., S. R. Snyder, V. G. Carande-Kulis, G. Isham, S. Garfield, P. Briss, and D. McCulloch. 2002. Increasing diabetes self-management education in community settings: A systematic review. American Journal of Preventive Medicine 22(4 Suppl 1):39-66.
Norton, T. R., S. L. Manne, S. Rubin, J. Carlson, E. Hernandez, M. I. Edelson, N. Rosenblum, D. Warshal, and C. Bergman. 2004. Prevalence and predictors of psychological distress among women with ovarian cancer. Journal of Clinical Oncology 22(5):919-926.
Norton, T. R., S. L. Manne, S. Rubin, E. Hernandez, J. Carlson, C. Bergman, and N. Rosenblum. 2005. Ovarian cancer patients’ psychological distress: The role of physical impairment, perceived unsupportive family and friend behaviors, perceived control, and self-esteem. Health Psychology 24(2):143-152.
Nurgalieva, Z., R. Xia, C. C. Liu, K. Burau, D. Hardy, and X. L. Du. 2010. Risk of chemotherapy-induced peripheral neuropathy in large population-based cohorts of elderly patients with breast, ovarian, and lung cancer. American Journal of Therapeutics 17(2):148-158.
OCNA (Ovarian Cancer National Alliance). 2015. Ovarian cancer community. http://www.ovariancancer.org/just-diagnosed/ovarian-cancer-community (accessed October 22, 2015).
Oostendorp, L. J. M., P. B. Ottevanger, W. T. A. Van Der Graaf, and P. F. M. Stalmeier. 2011. Assessing the information desire of patients with advanced cancer by providing information with a decision aid, which is evaluated in a randomized trial: A study protocol. BMC Medical Informatics and Decision Making 11(1).
Ory, M. G., M. L. Smith, N. Mier, and M. M. Wernicke. 2010. The science of sustaining health behavior change: The Health Maintenance Consortium. American Journal of Health Behavior 34(6):647-659.
Otis-Green, S., B. Ferrell, V. Sun, M. Spolum, R. Morgan, and D. MacDonald. 2008. Feasibility of an ovarian cancer quality-of-life psychoeducational intervention. Journal of Cancer Education 23(4):214-221.
Pantilat, S. Z., D. L. O—Riordan, S. L. Dibble, and C. S. Landefeld. 2010. Hospital-based palliative medicine consultation: A randomized controlled trial. Archives of Internal Medicine 170(22):2038-2040.
Papadakos, J., S. Bussiere-Cote, N. Abdelmutti, P. Catton, A. J. Friedman, C. Massey, S. Urowitz, and S. E. Ferguson. 2012. Informational needs of gynecologic cancer survivors. Gynecologic Oncology 124(3):452-457.
Parker, P. A., A. Kudelka, K. Basen-Engquist, J. Kavanagh, J. De Moor, and L. Cohen. 2006. The associations between knowledge, CA125 preoccupation, and distress in women with epithelial ovarian cancer. Gynecologic Oncology 100(3):495-500.
Partridge, A. H., D. S. Seah, T. King, N. B. Leighl, R. Hauke, D. S. Wollins, and J. H. Von Roenn. 2014. Developing a service model that integrates palliative care throughout cancer care: The time is now. Journal of Clinical Oncology 32(29):3330-3336.
Passik, S. D., K. L. Kirsh, K. Donaghy, E. Holtsclaw, D. Theobald, D. Cella, and W. Breitbart. 2002. Patient-related barriers to fatigue communication: Initial validation of the fatigue management barriers questionnaire. Journal of Pain and Symptom Management 24(5):481-493.
Pavelka, J. C., R. S. Brown, B. Y. Karlan, I. Cass, R. S. Leuchter, L. O. Lagasse, and A. J. Li. 2006. Effect of obesity on survival in epithelial ovarian cancer. Cancer 107(7):1520-1524.
Payne, J. K. 2002. The trajectory of fatigue in adult patients with breast and ovarian cancer receiving chemotherapy. Oncology Nursing Forum 29(9):1334-1340.
PCORI (Patient-Centered Outcomes Research Institute). 2015a. About us. http://www.pcori.org/about-us (accessed October 17, 2015).
PCORI. 2015b. American BRCA Outcomes and Utilization of Testing Patient-Powered Research Network (ABOUT Network). http://www.pcori.org/research-results/2013/american-brca-outcomes-and-utilization-testing-patient-powered-research (accessed October 16, 2015).
PCORI. 2015c. Ovarian cancer patient-centered decision aid. http://www.pcori.org/research-results/2013/ovarian-cancer-patient-centered-decision-aid (accessed October 16, 2015).
Phelps, A. C., P. K. Maciejewski, M. Nilsson, T. A. Balboni, A. A. Wright, M. E. Paulk, E. Trice, D. Schrag, J. R. Peteet, S. D. Block, and H. G. Prigerson. 2009. Religious coping and use of intensive life-prolonging care near death in patients with advanced cancer. Journal of the American Medical Association 301(11):1140-1147.
Pickett, S. A., S. Diehl, P. J. Steigman, J. D. Prater, A. Fox, and J. A. Cook. 2010. Early outcomes and lessons learned from a study of the Building Recovery of Individual Dreams and Goals through Education and Support (BRIDGES) program in Tennessee. Psychiatric Rehabilitation Journal 34(2):96-103.
Pignata, S., S. De Placido, R. Biamonte, G. Scambia, G. Di Vagno, et al. 2006. Residual neurotoxicity in ovarian cancer patients in clinical remission after first-line chemotherapy with carboplatin and paclitaxel: The Multicenter Italian Trial in Ovarian cancer (MITO-4) retrospective study. BMC Cancer 6:5.
Pinquart, M., and P. R. Duberstein. 2010a. Associations of social networks with cancer mortality: A meta-analysis. Critical Reviews in Oncology/Hematology 75(2):122-137.
Pinquart, M., and P. R. Duberstein. 2010b. Depression and cancer mortality: A meta-analysis. Psychological Medicine 40(11):1797-1810.
Ponto, J. A., L. Ellington, S. Mellon, and S. L. Beck. 2010. Predictors of adjustment and growth in women with recurrent ovarian cancer. Oncology Nursing Forum 37(3):357-364.
Portenoy, R. K., A. B. Kornblith, G. Wong, V. Vlamis, J. M. Lepore, D. B. Loseth, T. Hakes, K. M. Foley, and W. J. Hoskins. 1994. Pain in ovarian cancer patients: Prevalence, characteristics, and associated symptoms. Cancer 74(3 Suppl):907-915.
Postma, T. J., K. Hoekman, J. M. G. H. Van Riel, J. J. Heimans, and J. B. Vermorken. 1999. Peripheral neuropathy due to biweekly paclitaxel, epirubicin and cisplatin in patients with advanced ovarian cancer. Journal of Neuro-Oncology 45(3):241-246.
Prasath, E. B., M. L. H. Chan, W. H. W. Wong, C. J. W. Lim, M. D. Tharmalingam, M. Hendricks, S. F. Loh, and Y. N. Chia. 2014. First pregnancy and live birth resulting from cryopreserved embryos obtained from in vitro matured oocytes after oophorectomy in an ovarian cancer patient. Human Reproduction 29(2):276-278.
Quinn, G. P., S. T. Vadaparampil, B. A. Bell-Ellison, C. K. Gwede, and T. L. Albrecht. 2008. Patient–physician communication barriers regarding fertility preservation among newly diagnosed cancer patients. Social Science & Medicine 66(3):784-789.
Rabow, M. W., S. L. Dibble, S. Z. Pantilat, and S. J. McPhee. 2004. The comprehensive care team: A controlled trial of outpatient palliative medicine consultation. Archives of Internal Medicine 164(1):83-91.
Radwany, S. M., and V. E. Von Gruenigen. 2012. Palliative and end-of-life care for patients with ovarian cancer. Clinical Obstetrics and Gynecology 55(1):173-184.
Ramondetta, L. M., G. Tortolero-Luna, D. C. Bodurka, D. Sills, K. Basen-Engquist, J. Gano, and C. Levenback. 2004. Approaches for end-of-life care in the field of gynecologic oncology: An exploratory study. International Journal of Gynecological Cancer 14(4):580-588.
Reeve, B. B., S. A. Mitchell, A. C. Dueck, E. Basch, D. Cella, C. M. Reilly, L. M. Minasian, A. M. Denicoff, A. M. O—Mara, M. J. Fisch, C. Chauhan, N. K. Aaronson, C. Coens, and D. W. Bruner. 2014. Recommended patient-reported core set of symptoms to measure in adult cancer treatment trials. Journal of the National Cancer Institute 106(7).
Rimel, B. J., W. M. Burke, R. V. Higgins, P. S. Lee, C. V. Lutman, and L. Parker. 2015. Improving quality and decreasing cost in gynecologic oncology care. Society of Gynecologic Oncology recommendations for clinical practice. Gynecologic Oncology 137(2):280-284.
Roland, K. B., J. L. Rodriguez, J. R. Patterson, and K. F. Trivers. 2013. A literature review of the social and psychological needs of ovarian cancer survivors. Psychooncology 22(11):2408-2418.
Rolnick, S. J., J. Jackson, W. W. Nelson, A. Butani, L. J. Herrinton, M. Hornbrook, C. Neslund-Dudas, D. J. Bachman, and S. S. Coughlin. 2007. Pain management in the last six months of life among women who died of ovarian cancer. Journal of Pain & Symptom Management 33(1):24-31.
Rugno, F. C., B. S. Paiva, and C. E. Paiva. 2014. Early integration of palliative care facilitates the discontinuation of anticancer treatment in women with advanced breast or gynecologic cancers. Gynecologic Oncology 135(2):249-254.
Russell, L. B., D. C. Suh, and M. A. Safford. 2005. Time requirements for diabetes self-management: Too much for many? Journal of Family Practice 54(1):52-56.
Sandadi, S., H. E. Frasure, M. J. Broderick, S. E. Waggoner, J. A. Miller, and V. E. Von Gruenigen. 2011. The effect of sleep disturbance on quality of life in women with ovarian cancer. Gynecologic Oncology 123(2):351-355.
Schenker, Y., M. Crowley-Matoka, D. Dohan, M. W. Rabow, C. B. Smith, D. B. White, E. Chu, G. A. Tiver, S. Einhorn, and R. M. Arnold. 2014a. Oncologist factors that influence referrals to subspecialty palliative care clinics. Journal of Oncology Practice 10(2):e37-e44.
Schenker, Y., S. Y. Park, R. Maciasz, and R. M. Arnold. 2014b. Do patients with advanced cancer and unmet palliative care needs have an interest in receiving palliative care services? Journal of Palliative Medicine 17(6):667-672.
Schover, L. R., L. A. Rybicki, B. A. Martin, and K. A. Bringelsen. 1999. Having children after cancer. A pilot survey of survivors’ attitudes and experiences. Cancer 86(4):697-709.
Schrepf, A., L. Clevenger, D. Christensen, K. DeGeest, D. Bender, A. Ahmed, M. J. Goodheart, L. Dahmoush, F. Penedo, J. A. Lucci, 3rd, P. Ganjei-Azar, L. Mendez, K. Markon, D. M. Lubaroff, P. H. Thaker, G. M. Slavich, A. K. Sood, and S. K. Lutgendorf. 2013. Cortisol and inflammatory processes in ovarian cancer patients following primary treatment: Relationships with depression, fatigue, and disability. Brain, Behavior, & Immunity 30(Suppl):S126-S134.
Schulman-Green, D., S. Jaser, F. Martin, A. Alonzo, M. Grey, R. McCorkle, N. S. Redeker, N. Reynolds, and R. Whittemore. 2012. Processes of self-management in chronic illness. Journal of Nursing Scholarship 44(2):136-144.
Seibaek, L., L. K. Petersen, J. Blaakaer, and L. Hounsgaard. 2012. Hoping for the best, preparing for the worst: The lived experiences of women undergoing ovarian cancer surgery. European Journal of Cancer Care 21(3):360-371.
SGO (Society of Gynecologic Oncology). 2011. Pathways to progress in women’s cancer: A research agenda proposed by the Society of Gynecologic Oncology. https://www.sgo.org/wp-content/uploads/2012/10/Pathways-to-Progress.pdf (accessed September 21, 2015).
Shapiro, C. L., M. S. McCabe, K. L. Syrjala, D. Friedman, L. A. Jacobs, P. A. Ganz, L. Diller, M. Campell, K. Orcena, and A. C. Marcus. 2009. The LIVESTRONG Survivorship Center of Excellence Network. Journal of Cancer Survivorship 3(1):4-11.
Shinde, S., T. Wanger, P. Novotny, M. Grudem, and A. Jatoi. 2015. Disease-free ovarian cancer patients report severe pain and fatigue over time: Prospective quality of life assessment in a consecutive series. European Journal of Gynaecological Oncology 36(2):155-160.
Shinn, E. H., C. L. Taylor, K. Kilgore, A. Valentine, D. C. Bodurka, J. Kavanagh, A. Sood, Y. Li, and K. Basen-Engquist. 2009. Associations with worry about dying and hopelessness in ambulatory ovarian cancer patients. Palliative & Supportive Care 7(3):299-306.
Silver, J. K., and J. Baima. 2013. Cancer prehabilitation: An opportunity to decrease treatment-related morbidity, increase cancer treatment options, and improve physical and psychological health outcomes. American Journal of Physical Medicine and Rehabilitation 92(8):715-727.
Smits, A., E. Smits, A. Lopes, N. Das, G. Hughes, A. Talaat, A. Pollard, F. Bouwman, L. Massuger, R. Bekkers, and K. Galaal. 2015. Body mass index, physical activity and quality of life in ovarian cancer survivors: Time to get moving? Gynecologic Oncology 139(1):148-154
Sohl, S., J. Schnur, L. Daly, K. Suslov, and G. Montgomery. 2010. Development of a brief yoga intervention implemented while patients are undergoing chemotherapy for recurrent ovarian cancer: The journey. Journal of the Society for Integrative Oncology 8(4):170-171.
Sohl, S. J., S. C. Danhauer, J. B. Schnur, L. Daly, K. Suslov, and G. H. Montgomery. 2012. Feasibility of a brief yoga intervention during chemotherapy for persistent or recurrent ovarian cancer. Explore: The Journal of Science & Healing 8(3):197-198.
Steed, L., D. Cooke, and S. Newman. 2003. A systematic review of psychosocial outcomes following education, self-management and psychological interventions in diabetes mellitus. Patient Education and Counseling 51(1):5-15.
Stevinson, C., H. Steed, W. Faught, K. Tonkin, J. K. Vallance, A. B. Ladha, A. Schepansky, V. Capstick, and K. S. Courneya. 2009. Physical activity in ovarian cancer survivors: Associations with fatigue, sleep, and psychosocial functioning. International Journal of Gynecological Cancer 19(1):73-78.
Stewart, D. E., F. Wong, A. M. Cheung, J. Dancey, M. Meana, J. I. Cameron, M. P. McAndrews, T. Bunston, J. Murphy, and B. Rosen. 2000. Information needs and decisional preferences among women with ovarian cancer. Gynecologic Oncology 77(3):357-361.
Stewart, D. E., F. Wong, S. Duff, C. H. Melancon, and A. M. Cheung. 2001. “What doesn—t kill you makes you stronger”: An ovarian cancer survivor survey. Gynecologic Oncology 83(3):537-542.
Sun, C. C., D. C. Bodurka, C. B. Weaver, R. Rasu, J. K. Wolf, M. W. Bevers, J. A. Smith, J. T. Wharton, and E. B. Rubenstein. 2005. Rankings and symptom assessments of side effects from chemotherapy: Insights from experienced patients with ovarian cancer. Supportive Care in Cancer 13(4):219-227.
Sun, C. C., P. T. Ramirez, and D. C. Bodurka. 2007. Quality of life for patients with epithelial ovarian cancer. Nature Clinical Practice Oncology 4(1):18-29.
Swenson, M. M., J. S. MacLeod, S. D. Williams, A. M. Miller, and V. L. Champion. 2003. Quality of life after among ovarian germ cell cancer survivors: A narrative analysis. Oncology Nursing Forum 30(3):380.
Tang, T. S., M. M. Funnell, S. Noorulla, M. Oh, and M. B. Brown. 2012. Sustaining short-term improvements over the long term: Results from a 2-year diabetes self-management support (DSMS) intervention. Diabetes Research and Clinical Practice 95(1):85-92.
Teefey, P., N. B. Zgheib, S. M. Apte, J. Gonzalez-Bosquet, P. L. Judson, W. S. Roberts, J. M. Lancaster, and R. M. Wenham. 2013. Factors associated with improved toxicity and tolerability of intraperitoneal chemotherapy in advanced-stage epithelial ovarian cancers. American Journal of Obstetrics and Gynecology 208(6):501.e501-e507.
Temel, J. S., J. A. Greer, A. Muzikansky, E. R. Gallagher, S. Admane, V. A. Jackson, C. M. Dahlin, C. D. Blinderman, J. Jacobsen, W. F. Pirl, J. A. Billings, and T. J. Lynch. 2010. Early palliative care for patients with metastatic non-small-cell lung cancer. New England Journal of Medicine 363(8):733-742.
Teno, J. M., B. R. Clarridge, V. Casey, L. C. Welch, T. Wetle, R. Shield, and V. Mor. 2004. Family perspectives on end-of-life care at the last place of care. Journal of the American Medical Association 291(1):88-93.
Test, D. W., C. H. Fowler, D. M. Brewer, and W. M. Wood. 2005. A content and methodological review of self-advocacy intervention studies. Exceptional Children 72(1):101-125.
Thaker, P. H., L. Y. Han, A. A. Kamat, J. M. Arevalo, R. Takahashi, et al. 2006. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nature Medicine 12(8):939-944.
Tiller, K., B. Meiser, E. Reeson, M. Tucker, L. Andrews, C. Gaff, J. Kirk, K. A. Phillips, and M. Friedlander. 2003. A decision aid for women at increased risk for ovarian cancer. International Journal of Gynecological Cancer 13(1):15-22.
Tiller, K., B. Meiser, C. Gaff, J. Kirk, T. Dudding, K. A. Phillips, M. Friedlander, and K. Tucker. 2006. A randomized controlled trial of a decision aid for women at increased risk of ovarian cancer. Medical Decision Making 26(4):360-372.
Timmins III, P., D. Kredentser, K. Hancock, M. Messing, K. A. Boehm, S. Mull, and L. Asmar. 2008. Results of an open-label study to evaluate the efficacy and tolerability of aprepitant for the prevention of chemotherapy-induced nausea and vomiting associated with carboplatin-containing chemotherapy in patients with ovarian cancer, primary peritoneal, or fallopian tube carcinoma (stage I–IV) or papillary serous cancer of the uterus. Clinical Ovarian Cancer 1(1):60-65.
Tomao, F., F. Peccatori, L. D. Pup, D. Franchi, V. Zanagnolo, P. B. Panici, and N. Colombo. 2015. Special issues in fertility preservation for gynecologic malignancies. Critical Reviews in Oncology/Hematology (Epub ahead of print).
Torres, M. L., L. C. Hartmann, W. A. Cliby, K. R. Kalli, P. M. Young, A. L. Weaver, C. L. Langstraat, A. Jatoi, S. Kumar, A. Mariani. 2013. 129(3): 548-553.
Trivers, K. F., J. R. Patterson, K. B. Roland, and J. L. Rodriguez. 2013. Issues of ovarian cancer survivors in the USA: A literature review. Supportive Care in Cancer 21(10):2889-2898.
van den Berg, S. W., M. F. M. Gielissen, P. B. Ottevanger, and J. B. Prins. 2012. Rationale of the BREAst cancer e-healTH [BREATH] multicentre randomised controlled trial: An internet-based self-management intervention to foster adjustment after curative breast cancer by decreasing distress and increasing empowerment. BMC Cancer 12.
Verstappen, C. C. P., T. J. Postma, K. Hoekman, and J. J. Heimans. 2003. Peripheral neuropathy due to therapy with paclitaxel, gemcitabine and cisplatin in patients with advanced ovarian cancer. Journal of Neuro-Oncology 63(2):201-205.
Visovsky, C., and B. J. Daly. 2004. Clinical evaluation and patterns of chemotherapy-induced peripheral neuropathy. Journal of the American Academy of Nurse Practitioners 16(8):353-359.
von Gruenigen, V. E., H. E. Frasure, E. L. Jenison, M. P. Hopkins, and K. M. Gil. 2006. Longitudinal assessment of quality of life and lifestyle in newly diagnosed ovarian cancer patients: The roles of surgery and chemotherapy. Gynecologic Oncology 103(1):120-126.
von Gruenigen, V., B. Daly, H. Gibbons, J. Hutchins, and A. Green. 2008. Indicators of survival duration in ovarian cancer and implications for aggressiveness of care. Cancer 112(10):2221-2227.
von Gruenigen, V. E., H. Q. Huang, K. M. Gil, H. E. Gibbons, B. J. Monk, P. G. Rose, D. K. Armstrong, D. Cella, and L. Wenzel. 2009. Assessment of factors that contribute to decreased quality of life in Gynecologic Oncology Group ovarian cancer trials. Cancer 115(20):4857-4864.
von Gruenigen, V. E., H. Q. Huang, K. M. Gil, H. E. Gibbons, B. J. Monk, P. G. Rose, D. K. Armstrong, D. Cella, and L. Wenzel. 2010. A comparison of quality-of-life domains and clinical factors in ovarian cancer patients: A Gynecologic Oncology Group study. Journal of Pain and Symptom Management 39(5):839-846.
von Gruenigen, V. E., H. Q. Huang, K. M. Gil, H. E. Frasure, D. K. Armstrong, and L. B. Wenzel. 2012. The association between quality of life domains and overall survival in ovarian cancer patients during adjuvant chemotherapy: A Gynecologic Oncology Group study. Gynecologic Oncology 124(3):379-382.
Von Roenn, J. H., R. Voltz, and A. Serrie. 2013. Barriers and approaches to the successful integration of palliative care and oncology practice. Journal of the National Comprehensive Cancer Network 11(Suppl 1):S11-S16.
Wagner, L. I., J. Schink, M. Bass, S. Patel, M. V. Diaz, N. Rothrock, T. Pearman, R. Gershon, F. J. Penedo, S. Rosen, and D. Cella. 2015. Bringing promise to practice: Brief and precise symptom screening in ambulatory cancer care. Cancer 121(6):927-934.
Wakefield, C. E., B. Meiser, J. Homewood, M. Peate, A. Taylor, et al. 2008a. A randomized controlled trial of a decision aid for women considering genetic testing for breast and ovarian cancer risk. Breast Cancer Research and Treatment 107(2):289-301.
Wakefield, C. E., B. Meiser, J. Homewood, A. Taylor, M. Gleeson, et al. 2008b. A randomized trial of a breast/ovarian cancer genetic testing decision aid used as a communication aid during genetic counseling. Psychooncology 17(8):844-854.
Walker, J., and P. Lane. 2007. Challenges and choices: An audit of the management of nausea, vomiting and bowel obstruction in metastatic ovarian cancer. Contemporary Nurse 27(1):39-46.
Wenzel, L. B., J. P. Donnelly, J. M. Fowler, R. Habbal, T. H. Taylor, N. Aziz, and D. Cella. 2002. Resilience, reflection, and residual stress in ovarian cancer survivorship: A Gynecologic Oncology Group study. Psychooncology 11(2):142-153.
Wenzel, L. B., H. Q. Huang, D. K. Armstrong, J. L. Walker, and D. Cella. 2007. Health-related quality of life during and after intraperitoneal versus intravenous chemotherapy for optimally debulked ovarian cancer: A Gynecologic Oncology Group study. Journal of Clinical Oncology 25(4):437-443.
West, M. A., L. Loughney, D. Lythgoe, C. P. Barben, R. Sripadam, G. J. Kemp, M. P. W. Grocott, and S. Jack. 2015. Effect of prehabilitation on objectively measured physical fitness after neoadjuvant treatment in preoperative rectal cancer patients: A blinded interventional pilot study. British Journal of Anaesthesia 114(2):244-251.
WHO (World Health Organization). 2002. Palliative care is an essential part of cancer control. http://www.who.int/cancer/palliative/en (accessed October 23, 2015).
Wiecha, J., and T. Pollard. 2004. The interdisciplinary eHealth team: Chronic care for the future. Journal of Medical Internet Research 6(3):e22.
Williams, L. A., S. Agarwal, D. C. Bodurka, A. K. Saleeba, C. C. Sun, and C. S. Cleeland. 2013. Capturing the patient’s experience: Using qualitative methods to develop a measure of patient-reported symptom burden: An example from ovarian cancer. Journal of Pain and Symptom Management 46(6):837-845.
Wilmoth, M. C., E. Hatmaker-Flanigan, V. LaLoggia, and T. Nixon. 2011. Ovarian cancer survivors: Qualitative analysis of the symptom of sexuality. Oncology Nursing Forum 38(6):699-708.
Wright, A. A., B. Zhang, A. Ray, J. W. Mack, E. Trice, T. Balboni, S. L. Mitchell, V. A. Jackson, S. D. Block, P. K. Maciejewski, and H. G. Prigerson. 2008. Associations between end-of-life discussions, patient mental health, medical care near death, and caregiver bereavement adjustment. Journal of the American Medical Association 300(14):1665-1673.
Wright, A. A., N. L. Keating, T. A. Balboni, U. A. Matulonis, S. D. Block, and H. G. Prigerson. 2010. Place of death: Correlations with quality of life of patients with cancer and predictors of bereaved caregivers’ mental health. Journal of Clinical Oncology 28(29): 4457-4464.
Wright, A. A., L. A. Hatfield, C. C. Earle, and N. L. Keating. 2014. End-of-life care for older patients with ovarian cancer is intensive despite high rates of hospice use. Journal of Clinical Oncology 32(31):3534-3539.
You, Q., H. Yu, D. Wu, Y. Zhang, J. Zheng, and C. Peng. 2009. Vitamin B6 points PC6 injection during acupuncture can relieve nausea and vomiting in patients with ovarian cancer. International Journal of Gynecological Cancer 19(4):567-571.
Zhang, R., Y.C. Sun, G.Y. Zhang, L.Y. Wu, and J. Zuo. 2012. Treatment of malignant ovarian germ cell tumors and preservation of fertility. European Journal of Gynaecological Oncology 33(5):489-492.
Zimmermann, C., N. Swami, M. Krzyzanowska, B. Hannon, N. Leighl, A. Oza, M. Moore, A. Rydall, G. Rodin, I. Tannock, A. Donner, and C. Lo. 2014. Early palliative care for patients with advanced cancer: A cluster-randomised controlled trial. Lancet 383(9930): 1721-1730.










































