Because of progress over the past few decades, newly diagnosed ovarian cancers are now more accurately and consistently staged than in the past, and, thanks in particularly to better characterization of tumor biology and a personalized medicine approach in therapeutic development, a wider variety of treatment options exist. While this progress is encouraging, there is still much to do, and there are a number of barriers that must be overcome to further improve outcomes for women with ovarian cancers. This chapter discusses the diagnosis and therapeutic management of women with newly diagnosed and recurrent ovarian cancer, discusses new therapies on the horizon, and gives an overview of the cancer clinical trials landscape. As noted in Chapter 1, these discussions focus primarily on the treatment of women diagnosed with high-grade serous carcinomas (HGSCs).
Because of the heterogeneity of ovarian cancers, it is essential that the initial diagnosis be accurate. Furthermore, because a variety of benign conditions can mimic ovarian cancer, it is crucial that women with suspected ovarian cancer be carefully examined to help determine the proper clinical management.
Signs and Symptoms
While there are no symptoms specific to ovarian cancer, most women diagnosed with ovarian cancer do experience such things as bloating, pelvic
or abdominal pain, difficulty eating, or urinary symptoms; the problem is that these symptoms are often overlooked until after a diagnosis has already been made. Studies show that 90 percent of women with early-stage ovarian cancer and 100 percent of women with the late-stage disease reported having at least one symptom (Goff et al., 2004; Lataifeh et al., 2005). There is only a marginal difference in the symptoms reported by women with the early-stage disease and those with the late-stage disease (Olsen et al., 2007), and patients and providers do not commonly associate these symptoms with gynecologic issues, which complicates early detection. One way to help raise awareness of the early signs and symptoms of ovarian cancer would be to increase the understanding of these symptoms among providers and then to improve communication between the providers and their patients.
Diagnosis
Several bodies have established guidelines for the initial assessment and treatment of women with suspected ovarian cancer. The Society for Gynecologic Oncologists (SGO), the American College of Obstetricians and Gynecologists (ACOG), and the National Comprehensive Cancer Network (NCCN) have all published guidelines for referral of patients with suspected ovarian cancer (ACOG, 2002; NCCN, 2015) (see Box 4-1). In addition to the difficulties created by the vagueness of the symptoms described above, it can be challenging to make an accurate diagnosis of ovarian cancer in women with widespread disease because gastrointestinal tumors can mimic an ovarian cancer at initial presentation (Munoz et al., 2012). In the clinic, a woman most often presents with carcinomatosis-associated ascites, and a diagnosis is often made by paracentesis with cytologic review of ascites, fine needle aspiration, or laparoscopic biopsy.
Pathological and Surgical Reporting
Complete and detailed pathological and surgical reporting is necessary for both the characterization of the disease and the determination of the treatment course. In general, the first step is to perform a tumor biopsy or primary debulking surgery (PDS). (See next section in the chapter for more on PDS.) Important information to collect in surgical operative reports includes an accurate description of the biopsy sites and an objective description and documentation of the extent of residual disease (Robboy et al., 2002). The collected tissues are used for pathological evaluation. The College of American Pathologists Protocol for the Examination of Specimens from Patients with Carcinoma of the Ovary lists the following crucial components for all pathology reports:
BOX 4-1
Ovarian Cancer Assessment Guidelines Summary
Clinical presentation
- Detection of pelvic mass on exam
- Symptoms: e.g., bloating, pelvic or abdominal pain, early satiety, urinary symptoms
- Concern raised on screening assays (e.g., CA-125)
- Incidental findings on previous surgery or tissue biopsy
Workup
- Family history of breast or ovarian cancer
- Abdominal/pelvic exam
- Imaging: chest, abdominal, and pelvic CT/MRI as indicated
- Blood tests: CBC, chemistry profile, liver function tests; CA-125 and other serum biomarkers (e.g., inhibin, AFP, beta-HCG) as indicated
- Needle aspiration should be avoided for diagnosis in patients with presumed early-stage disease to prevent tumor rupture and spread
NOTE: AFP = alpha-fetoprotein; CA-125 = cancer antigen 125; CBC = complete blood count; CT = computed tomography; HCG = human chorionic gonadotropin; MRI = magnetic resonance imaging.
SOURCES: ACOG, 2002; NCCN, 2015.
- Specimen integrity;
- Histology;
- Tumor grade (particularly for serous tumors); and
- Tumor stage (extent of involvement of other tissues/organs in a systematic manner) (Scully et al., 1996).
The accuracy of these records is crucial in ensuring effective patient care because many treatment decisions are based in large part on these reports. However, traditional operative and pathology reports frequently do not capture these important data (Donahoe et al., 2012; Gogoi et al., 2012). In one study, up to 25 percent of all operative reports lacked documentation of residual disease for ovarian cancer patients (Gogoi et al., 2012). Another evaluation of nearly 500 pathology reports for advanced ovarian, fallopian tube, and primary peritoneal cancers found that although most specimens were microscopically described (92.3 percent of reports), tumor size or weight was missing in 40.1 percent of reports, and, a description of the tumor origin was missing in 20.5 percent of surgical reports for PDS and 23.4 percent of surgical reports for interval debulking procedures (Verleye et al., 2011).
Once a diagnosis of ovarian cancer has been confirmed by pathological evaluation, patients with newly diagnosed ovarian cancer are generally managed with a combination of surgery and chemotherapy.
Surgical Management
The key goals for surgical intervention in women with suspected ovarian cancer include establishing the initial diagnosis, determining the extent of disease spread (staging), and deciding on the best course of therapy. Accurate staging is particularly relevant in those cases where the disease appears to be confined to the ovary, because the subsequent therapeutic course is largely prescribed according to tumor stage (Winter-Roach et al., 2012) (see Box 4-2 for staging criteria).
For women with early-stage (Stage I or II) ovarian cancer, complete surgical staging is also an important prognostic indicator (NIH Consensus Development Panel on Ovarian Cancer, 1995). For example, in a study of women with Stage I ovarian carcinoma who had received a lymphadenectomy (the surgical removal of one or more groups of lymph nodes), nearly 7 percent
BOX 4-2
International Federation of Gynecology and Obstetrics (FIGO)
Staging Criteria for Ovarian Cancer
Stage I: Tumor confined to one or both ovaries or fallopian tubes
IA. Tumor limited to one ovary (capsule intact) or fallopian tube
IB. Tumor limited to both ovaries or fallopian tubes
IC. Tumor limited to both ovaries or fallopian tubes with any of the following:
IC1. Surgical spill intraoperatively
IC2. Capsule ruptured before surgery or tumor on ovarian or fallopian tube surface
IC3. Malignant cells present in the ascites or peritoneal washings
Stage II: Tumor involves one or both ovaries or fallopian tubes with pelvic extension (below pelvic brim) or peritoneal cancer
IIA. Extension and/or implants on any one or more of the following: uterus, fallopian tube(s), ovaries
IIB. Extension to other pelvic intraperitoneal (IP) tissues
Stage III: Tumor involves one or both ovaries or fallopian tubes, or primary peritoneal cancer, with cytologically and histologically confirmed spread to the peritoneum outside the pelvis and/or metastasis to the retroperitoneal lymph nodes
of the women had greater disease-specific survival (Chan et al., 2007a). In the absence of complete staging, the full extent of residual disease may be unknown, which could affect decisions about the treatment course. Given the necessity for a sophisticated diagnostic and initial surgical approach, evidence shows that the surgical treatment for ovarian cancers is best performed by those with expertise in managing this disease specifically (e.g., gynecologic oncologists) (Giede et al., 2005; Hacker, 2011; Vernooij et al., 2007).
For women newly diagnosed with ovarian cancer, the amount of tumor remaining after PDS is among the most important prognostic factors for both progression-free survival (PFS) and overall survival (Bristow et al., 2002; du Bois et al., 2009; Eisenkop et al., 1998; Elattar et al., 2011; Griffiths, 1975; Hoskins et al., 1994). The goal of PDS is always to achieve a complete resection of the tumor. Several factors affect whether a patient experiences complete resection (no residual tumor), optimal cytoreduction (residual tumor ≤ 10 mm in diameter), or suboptimal cytoreduction (residual tumor > 10 mm in diameter) (Chi et al., 2012; du Bois et al., 2009; Hacker, 2013). These factors include the size of the tumor at diagnosis (as indicated by FIGO stage) and the aggressiveness of the surgical practice.
IIIA. Metastasis to the retroperitoneal lymph nodes with or without microscopic peritoneal involvement beyond the pelvis
IIIA1. Positive retroperitoneal lymph nodes only (cytologically or histologically proven)
IIIA1(i). Metastasis ≤ 10 mm in greatest dimension (note that this is a tumor dimension and not a lymph node dimension)
IIIA1(ii). Metastasis ≤ 10 mm in greatest dimension
IIIA2. Microscopic extrapelvic (above the pelvic brim) peritoneal involvement with or without positive retroperitoneal lymph nodes
IIIB. Macroscopic peritoneal metastases beyond the pelvic brim ≤ 2 cm in greatest dimension, with or without metastasis to the retroperitoneal lymph nodes
IIIC. Macroscopic peritoneal metastases beyond the pelvic brim > 2 cm in greatest dimension, with or without metastases to the retroperitoneal nodes
Stage IV: Distant metastasis excluding peritoneal metastases
IVA. Pleural effusion with positive cytology
IVB. Metastases to extra-abdominal organs
SOURCE: Mutch and Prat, 2014.
A study of nearly 1,300 women with ovarian carcinoma enrolled in three Phase III randomized trials testing different chemotherapy regimens found that both PFS and overall survival differed significantly for women who had complete resection (i.e., no residual tumor) versus those who had either optimal cytoreduction or suboptimal cytoreduction (du Bois et al., 2009). Overall survival differences were more pronounced for patients with Stage II (a 46-month improvement) or Stage III (a 60-month improvement) disease but were still significant for patients with Stage IV disease (average 30-month improvement).
In addition to women who have had suboptimal cytoreduction, there are women who are unable to undergo PDS at all because of the presence of comorbidities that preclude initial surgery. For these women, neoadjuvant chemotherapy (NACT) is used to reduce the tumor burden (the amount of cancer or the size of the tumor) and facilitate subsequent surgical resection (Morrison et al., 2012; Vergote et al., 2013). Increasingly, advanced-stage ovarian cancer patients with extensive metastasis (noted on imaging studies or at the time of diagnostic laparoscopy) are being managed with three to four cycles of NACT prior to an interval cytoreduction. Results from two separate trials suggested that patients who have undergone NACT have similar PFS and overall survival rates to patients who have undergone PDS (Kehoe et al., 2015; Vergote et al., 2010). However, these findings were met with skepticism from several groups in the gynecologic oncology community who criticized various elements of the trial design (Dai-yuan et al., 2013; du Bois et al., 2012; Gasparri et al., 2015; Hacker, 2013; Kehoe and Nankivell, 2015; Scaletta et al., 2015; Vergote et al., 2013). As such, the question of which women should receive initial PDS or NACT remains unresolved. The prognostic role of other endpoints—such as pathologic complete response, which is commonly utilized in NACT studies in breast cancer—may provide additional details in NACT studies in ovarian cancer. Nonetheless, NCCN guidelines do allow for the consideration (based on category 1 evidence) of NACT for patients with bulky Stage III to IV disease if maximum cytoreduction cannot be achieved (NCCN, 2015). Evidence suggests that a number of disease characteristics (e.g., tumor volume and stage) and patient characteristics (e.g., age at diagnosis) are significantly associated with outcome after PDS or NACT and need to be considered when evaluating patients (Horowitz et al., 2015).
Several prognostic and predictive biomarkers are currently used to determine whether other types of cancer patients will benefit from particular targeted therapies. These include HER2/Neu protein amplification for Herceptin® use against breast tumors, and epidermal growth factor receptor (EGFR) protein amplification for anti-EGFR therapeutics, which are used against a wide variety of tumors, including ovarian tumors (Baselga, 2006; Grunwald and Hidalgo, 2003; Yeon and Pegram, 2005). Although these
targeted therapies may not work in all ovarian cancers, HER2-positive mucinous carcinomas may be a viable target for anti-HER2 therapies (Anglesio et al., 2013; Teplinsky and Muggia, 2015). As discussed in Chapter 3, the protein biomarker cancer antigen 125 (CA-125) has been studied as a predictive biomarker for ovarian cancer risk, though its primary use is to monitor tumor progression. It has also been investigated for other uses, including predicting tumor response to chemotherapy and the probability of PFS and overall survival. It has been suggested that biomarkers such as CA-125 and human epididymis protein 4 (HE-4) can play a role in aiding clinical decision making for initial PDS or NACT (Nolen and Lokshin, 2013).
Chemotherapy Management
The earliest use of chemotherapy for the treatment of advanced ovarian cancer involved the intravenous (IV) delivery of doxorubicin (an anthracycline) and cyclophosphamide (a DNA cross-linking agent). These chemicals are cytotoxic, meaning that they kill cells, especially cancer cells. In the 1980s, the platinum-based agent cisplatin was shown to significantly improve response rates and survival when added to the protocol (Neijt et al., 1984; Omura et al., 1986). Later trials found little difference in clinical efficacy between the two-drug combination of cisplatin and cyclophosphamide and multidrug combinations including anthracyclines, so the combination of cisplatin and cyclophosphamide became standard chemotherapy for the first-line treatment of advanced ovarian cancer (Bertelsen et al., 1987; Omura et al., 1989).
In 1994, the first National Institutes of Health (NIH) Consensus Conference on Ovarian Cancer released recommendations for the chemotherapeutic management of newly diagnosed or post-operative ovarian cancer (NIH Consensus Development Panel on Ovarian Cancer, 1995). Based on the results of chemotherapy trials that had been conducted in the previous decade, the conference recommended that all women diagnosed with Stage IC–IV ovarian cancer receive adjuvant chemotherapy following PDS. (Women with Stage IA or IB ovarian cancer were only recommended to have surgery.) Since then, subsequent trials (see Table 4-1) using different treatment protocols for first-line and maintenance therapy have led to further improvements in PFS and in overall survival (Burger et al., 2011; du Bois et al., 2003; Katsumata et al., 2009; McGuire et al., 1996; Ozols et al., 2003; Perren et al., 2011; Piccart et al., 2000).
The current standard of care is a platinum-based chemotherapeutic agent (e.g., cisplatin or carboplatin), which may be given in combination with taxane agents (e.g., paclitaxel) (NCCN, 2015). Carboplatin administered every 3 weeks in combination with paclitaxel administered every 3 weeks or every week (dose dense) is now the standard treatment for ovar-
TABLE 4-1
Clinical Trials of Intravenous Chemotherapy Protocols for Ovarian Cancer
| Therapy | Trial(s) | Treatment Changes | References | |
| Front line | GOG-111, OV10 | Replacement of IV cyclophosphamide with IV paclitaxel in combination with IV cisplatin | McGuire et al., 1996; Piccart et al., 2000 | |
| AGO OVAR-3, GOG-158 | Replacement of IV cisplatin with IV carboplatin in combination with paclitaxel | du Bois et al., 2003; Ozols et al., 2003 | ||
| NCT00226915 | Dose-dense IV paclitaxel (80 mg/m2 weekly) in combination with standard IV carboplatin | Katsumata et al., 2009 | ||
| GOG-217, ICON 7 | Addition of bevacizumab to IV paclitaxel and IV carboplatin | Burger et al., 2011; Perren et al., 2011 | ||
| Maintenance | GOG-178 | Additional 12 months of paclitaxel | Markman et al., 2003 | |
ian cancer patients who have suboptimal cytoreduction or who are treated with NACT. For patients with advanced-stage ovarian cancer who have undergone optimal cytoreduction, intraperitoneal (IP) chemotherapy has proven to be one of the more effective treatment strategies.
Intraperitoneal Chemotherapy
For both primary and metastatic ovarian cancers, the peritoneal cavity is the principal site of disease upon diagnosis (Lengyel, 2010). Several routes exist for delivering chemotherapy that will reach that site: through an IV line, orally (by mouth), and through an implanted IP catheter.
In the 1950s, IP chemotherapy was used for patients with malignant pleural, peritoneal, or pericardial effusions from several types of primary cancer (Weisberger et al., 1955). In delivering pharmaceutical agents for the treatment of ovarian carcinoma, the method that consistently led to the highest concentrations of the drug in ascites was the IP route (Ward et al., 1987). Subsequent studies have confirmed that IP delivery results in an increased drug concentration at the tumor site and have indicated that IP delivery also results in fewer adverse systemic side effects than IV or oral routes (Jaaback et al., 2011). However, IV and oral chemotherapy are the most common modes of treatment for ovarian cancer in general, while fewer than half of patients with ovarian carcinoma receive IP chemotherapy in addition to IV chemotherapy (Wright et al., 2015).
In the past two decades, several clinical trials have tested different combinations and doses of IP chemotherapy agents, including three trials that had not yet released results as this report was being written (Alberts et al., 1996; Armstrong et al., 2006; Mackay et al., 2011; Markman et al., 2001). While these trials have had variable results, they all have shown that combined IV and IP chemotherapy administration improves survival in women with Stage III, optimally resected disease compared with standard IV administration alone (Hess et al., 2007; Jaaback et al., 2011). The seminal results from the 2006 GOG-172 clinical trial showed that treatment with IP chemotherapy resulted in a 5-month improvement in median PFS and a 16-month improvement in median overall survival over IV chemotherapy (Armstrong et al., 2006). A subsequent long-term follow-up found a nearly 10-month increase in median survival and a 23 percent decreased risk of death for patients treated with the IP protocols compared with IV protocols, suggesting a sustained benefit from IP chemotherapy (Tewari et al., 2015).
However, tumor histology may play a role in how well a patient responds to IP chemotherapy, so that not all women with ovarian cancer benefit equally from IP chemotherapy. Among patients with aberrant BRCA1 gene expression, those treated with IP chemotherapy had a significantly better median overall survival (84 months) than those treated using only IV chemotherapy (47 months), but no significant difference was seen among patients with normal BRCA1 expression (Lesnock et al., 2013). Among the various subtypes of ovarian cancer, the advantage of IP over IV chemotherapy is most pronounced for serous tumors, but only if all six cycles of therapy are delivered.
The adoption of IP chemotherapy protocols is not yet widespread among clinicians because of concerns about some potential serious adverse effects of IP administration, including drug-related toxicities, complications from catheter use, delivery of chemotherapy beyond the peritoneum, and poor tolerance of the treatment (Wright et al., 2015). Another concern is that the delivery of IP chemotherapy is sometimes inconsistent (e.g., the full six cycles of intended therapy are not always completed). Furthermore, among hospitals that have adopted IP chemotherapy into the standard of care, there appears to be great variation in the number and types of patients who receive it (Wright et al., 2015). The delivery of dose-dense paclitaxel, a newer IV dosing regimen, has demonstrated similar efficacy to the IP protocols without many of the accompanying toxicities (Katsumata et al., 2009). However, questions remain concerning direct comparisons between IP chemotherapy regimens and the equivalent doses and schedules that are administered via the IV route. Three large-scale trials are currently examining carboplatin-based IP chemotherapy:
- iPocc Trial (GOTIC-001/JGOG-3019);
- GOG-252; and
- OV-21/CGIG.
Although these trials have different patient populations and different treatment regimens, they will help answer important questions regarding the administration and comparative effectiveness of IP versus IV chemotherapy.
Maintenance Therapy
Maintenance therapies may be beneficial for women with advanced ovarian cancers, especially given the high rates of recurrence with standard therapies. Several trials have tested a variety of agents for use in maintenance therapy, including hormones, radiation, chemotherapy, immunotherapy, biological therapy, and complementary medicines. At its interim analysis, GOG-178, a Phase III trial, found that 12 additional months of paclitaxel provided a 7-month improvement in PFS, and the study was closed to further accrual (Markman et al., 2003). GOG-212 is a Phase III confirmatory trial that has completed accrual and will provide further guidance regarding the role of cytotoxic chemotherapy agents as a maintenance strategy in ovarian cancer.
Within the past decade, several randomized studies have investigated the various roles of anti-angiogenic agents (e.g., bevacizumab, pazopanib, and cediranib) and poly ADP ribose polymerase (PARP) inhibitors (e.g., olaparib) as a maintenance strategy in patients with advanced or relapsed ovarian cancers. Trials have demonstrated an improvement in PFS with the use of bevacizumab as a maintenance regimen (after primary chemotherapy). Trials investigating PARP inhibitors alone and in combination with chemotherapy have likewise demonstrated improvement in PFS and have led to U.S. Food and Drug Administration (FDA) approval of olaparib as a monotherapy for ovarian cancer patients with a BRCA mutation who have received three prior chemotherapy regimens (Kaye et al., 2012; Kim et al., 2015; Ledermann et al., 2012). Additional trials are now in progress to investigate several other anti-angiogenic agents and PARP inhibitors as maintenance options. For instance, SOLO2 is investigating maintenance treatment with olaparib, with results expected in 2016. The FDA did not support an accelerated approval of olaparib and stated that its decision on whether to approve olaparib for maintenance therapy needed to be delayed until the final results were available (FDA, 2014c). Some of the issues that the FDA was concerned about were the validity of the effects, the trial’s statistical analysis, and potential risks associated with the drug. In addition, questions remain regarding which endpoints may be necessary to accurately capture the effects of maintenance therapy in ovarian cancer (Ledermann
and El-Khouly, 2015). It may be necessary to explore additional clinical trial endpoints in order to adequately assess maintenance therapy and ensure that patients do indeed benefit from this treatment.
Despite the high response rate to aggressive initial treatments, most women diagnosed with advanced ovarian cancer will eventually have a recurrence (Coleman et al., 2013). The rate of relapse in ovarian cancers is highly dependent on the initial stage at diagnosis, the histologic type, and the presence of residual disease at the time of primary or interval debulking. Women with Type II ovarian cancers (e.g., HGSC) are also likely to have higher rates of recurrence.
Several organizations have established guidelines for monitoring women for recurrence (Salani et al., 2011). Although many women with recurrent ovarian cancer will be symptomatic or have detectable signs of recurrence upon physical examination, an increasing number of women are being (or could be) diagnosed with recurrent ovarian cancer through the detection of incremental rises in CA-125 and through the use of more sophisticated imaging technology (Bhosale et al., 2010; Prat et al., 2009). Diagnosing recurrent ovarian cancer earlier may improve physicians’ ability to achieve maximum secondary cytoreduction. However, there are limited data to suggest that an earlier diagnosis of recurrence improves overall survival, and, indeed, it may instead lead to interventions that negatively affect the patient’s quality of life (Clarke et al., 2014; Fleming et al., 2011; Rustin et al., 2010). Some argue that following up with CA-125 testing after primary therapy may be unneccesary and that its use should be discouraged (Rustin, 2011). Additional studies may be needed to examine the various follow-up methods and assess whether early intervention improves survival or quality of life, especially as new treatments for relapsed ovarian cancer become available. Additional considerations such as the distress and other psychosocial effects caused by continued monitoring for recurrence need to be considered when evaluating the effectiveness of surveillance (Parker et al., 2006). (For more on supportive care for the physicial and psychosocial effects of diagnosis and treatment, see Chapter 5.)
Patients with recurrent ovarian cancers have traditionally been categorized as either platinum sensitive (if recurrence is diagnosed more than 6 months after prior therapy) or platinum resistant (if recurrence is diagnosed less than 6 months after prior therapy). However, it may be time to consider developing a new classification paradigm for recurrent ovarian cancers, particularly given the ability to diagnose these recurrences at earlier time points, the increased understanding of the impact that BRCA mutation status has on a patient’s response to therapy for recurrence, and the hetero-
geneous responses noted in patients who are classified as platinum resistant (Davis et al., 2014; Guth et al., 2010).
Surgical Management
Like newly diagnosed women, women with recurrent ovarian cancer have improved outcomes when there is a complete resection of the cancer (Al Rawahi et al., 2013; Harter et al., 2006). There are several ongoing trials evaluating secondary cytoreduction in patients with recurrent ovarian cancer, two of which have overall survival as the primary endpoint (van de Laar et al., 2014). Secondary (and further) cytoreduction is generally reserved for patients with isolated, resectable, platinum-sensitive disease. Validated scoring systems can be used to predict which women with recurrent ovarian cancers undergoing secondary cytoreduction are most likely to achieve complete resection of all visible disease (Harter et al., 2011, 2014; Nick et al., 2015; Tian et al., 2012; Wimberger et al., 2007). Limited studies of tertiary (and beyond) cytoreduction in patients with recurrent ovarian cancer that suggest complete resection is feasible, and this remains the most important factor in predicting a patient’s survival (Fotopoulou et al., 2011, 2013; Liu et al., 2014b).
Chemotherapy Management
Once ovarian cancer recurs, the appropriate chemotherapy strategy will depend on the length of time to relapse (Friedlander et al., 2011; Stuart et al., 2011). Several trials have found better outcomes with carboplatin-based combination cytotoxic chemotherapies than for single-agent carboplatin for patients with platinum-sensitive recurrent ovarian cancers (Parmar et al., 2003; Pfisterer et al., 2006; Raja et al., 2013; Wagner et al., 2012). Patients with cancers that are not responsive to front-line therapy are typically treated with other agents (e.g., liposomal doxorubicin, gemcitabine, topotecan, or etoposide), but the overall response to these agents remains poor (less than 20 percent) and the median PFS is only 3 months on average (Agarwal and Kaye, 2003; Luvero et al., 2014). A better understanding of the genetic differences between the initial ovarian cancer cells and the drug-resistant ovarian cancer cells could help direct efforts to design targeted therapies.
Two new agents have recently been approved by the FDA for patients with recurrent ovarian cancer. Bevacizumab, a humanized antibody that blocks vascular endothelial growth factor (VEGF), in combination with one of three chemotherapy regimens (i.e., dose-dense paclitaxel, liposomal doxorubicin, and topotecan), is now approved for the treatment of patients with platinum-resistant ovarian cancers. Olaparib, a PARP inhibitor, has
been approved as a monotherapy for BRCA mutation–positive, recurrent ovarian cancer patients who have had three prior chemotherapy treatments.
Evaluating Chemosensitivity and Chemoresistance
Understanding which tumors are likely to be resistant to chemotherapeutic agents after recurrence is a key step in improving overall survival. Several assays have been developed (or are in development) for determining the likelihood that primary and relapsed tumors will respond to various chemotherapeutic agents (Rutherford et al., 2013). There is some evidence to suggest that sensitivity to platinum agents differs by tumor histologic subtype, with Type I tumors showing greater sensitivity than Type II tumors (Berns and Bowtell, 2012; Gershenson, 2012; Kobel et al., 2008; Storey et al., 2008; Vang et al., 2009). This heterogeneity complicates assay development efforts in which tumor samples must be divided into small subsets in order to more accurately determine therapy efficacy.
No systematic review supports the role of chemosensitivity assay–directed care for patients with recurrent ovarian cancer. Similarly, molecular profiling for the identification of potential molecular targeted therapies for patients with recurrent ovarian cancer, while feasible, has not been demonstrated to be associated with improved outcomes (Kohn, 2014; Plamadeala et al., 2015; Rutherford et al., 2013). New clinical trials, such as the National Cancer Institute-Molecular Analysis for Therapy Choice (NCI-MATCH), seek to analyze patient tumors for genetic abnormalities in order to determine if a targeted drug exists for use in treatment (NCI, 2015). Some of the targeted therapies in this trial may be effective in treating ovarian cancer.
THE DELIVERY OF OVARIAN CANCER CARE
The Institute of Medicine (IOM) reports Ensuring Quality Cancer Care and Delivering High-Quality Cancer Care looked at the delivery of cancer care overall (IOM and NRC, 1999; IOM, 2013) and offered a variety of findings and conclusions. The 2013 report found that “cancer care is often not as patient-centered, accessible, coordinated, or evidence-based as it could be” (IOM, 2013, p. 2). The growing demand for care (mostly due to the aging population), a shrinking workforce, and rising costs are issues that affect the care of patients with many types of cancer (and other serious diseases). Furthermore, “in the current cancer care system, many patients lack access to affordable, high-quality cancer care” (IOM, 2013, p. 309). Clinical trials in cancer care tend to have lower levels of enrollment among older adults and racial and ethnic minorities (Lewis et al., 2003; Murthy et al., 2004; Stewart et al., 2007; Talarico et al., 2004). There are major
disparities in cancer outcomes that negatively affect individuals who are of lower socioeconomic status, are racial or ethnic minorities, or who are underinsured or lack health insurance coverage (IOM, 2013). Furthermore, a focus on the delivery of treatment for the disease often overshadows consideration for the patient’s preferences and their supportive care needs, including the treatment and management of the physical and psychosocial effects of diagnosis and treatment (see Chapter 5). A review of the literature on the delivery of cancer care (or the challenges in the health care system in general that affect cancer care) is beyond the scope of this report, but the following sections highlight the overarching findings in the research of cancer care delivery and discuss the specific evidence related to the delivery of care for women with ovarian cancer.
Standard-of-Care Guidelines
According to the 2013 IOM report, “clinical research leads to improvements in the quality of care only if these research results are translated into clinical practice” (IOM, 2013, p. 293). Professional societies and organizations help to develop standard-of-care guidelines based on currently available data. Women with ovarian cancer who receive care in accordance with NCCN clinical practice guidelines have considerably better clinical outcomes (e.g., improved survival and fewer surgical complications) (Bristow et al., 2013b; Chan et al., 2007b; Eisenkop, et al., 1992; Goff, 2015). However, 56 percent of women with ovarian cancer nationwide do not receive care that adheres to these guidelines (Cliby et al., 2015). Furthermore, as was discussed in Chapter 3, the percentage of women who are referred to genetic counseling and testing in accordance with existing guidelines is low, and nearly half of all eligible women are not receiving recommended counseling (Febbraro et al., 2015; HHS, 2013; Powell et al., 2013). (See Table 3-3 for current recommendations for cancer genetic consultation and germline genetic testing for women with a personal or family history of ovarian cancer.)
The most significant predictors of whether women will receive the standard of care are whether they are treated by a gynecologic oncologist (as compared to a general gynecologist or general surgeon) and whether they are treated in a high-volume (often urban) or a low-volume (often rural) hospital or cancer center; treatment by a gynecologic oncologist and in a high-volume setting are associated with better outcomes (Bristow et al., 2013a, 2014). Significant predictors of nonadherence to the standard of care include advanced age at diagnosis, the presence of one or more treatment-limiting comorbidities, non-white race, and lower socioeconomic status (Bristow et al., 2013a,b; Chase et al., 2012; Du et al., 2008; Erickson et al., 2014; Goff et al., 2007; Harlan et al., 2003; Howell et al., 2013;
Jordan et al., 2013; Joslin et al., 2014; Thrall, 2011). Some studies have found a geographic variation in the patterns of cancer care, which may be due to socioeconomic or other factors (Fairfield et al., 2010; Polsky et al., 2006; Ulanday et al., 2014).
Individual Patient Considerations
A physician developing a treatment plan for a woman with ovarian cancer needs to consider not only the disease diagnosis itself, but also other factors related to demographics and patient preferences.
Caring for Older Adults
The 2013 IOM report noted that “the population of those 65 years and older comprises the majority of patients who are diagnosed with cancer and who die from cancer, as well as the majority of cancer survivors” (IOM, 2013, p. 2). Similarly, most ovarian cancers are diagnosed in older women (see Chapter 1). The care for older adults with cancer in general can be affected by alterations in their physiology and by their functional impairments, comorbidities, and treatment goals (IOM, 2013). Some of the areas that have been identified as in need of further research in general for the cancer care of older adults include the efficacy and toxicity of commonly used therapeies in older patients, clinical trials to better understand specific issues for older patients (including consideration of comorbidities), and the outcome measures that are most important for older adults (Hurria et al., 2012; Moy et al., 2014). NRG Oncology and the NCI are collaborating on an open observational trial to study “comprehensive patient questionnaires in predicting complications in older patients with gynecologic cancer undergoing surgery. Comprehensive patient questionnaires completed before surgery may help identify complications, such as the need for assistance in taking medications, decreased mobility, decreased social activity, and falls, and may improve outcomes for older patients with gynecologic cancer” (NRG Oncology, 2015).
As noted above, older women with ovarian cancer are less likely to be treated by a gynecologic oncologist and are less likely to receive care in accordance with standard-of-care guidelines. For example, one study of women with advanced ovarian carcinoma showed that only 39.1 percent of women older than age 65 had surgery and at least six cycles of chemotherapy (Thrall et al., 2011). Given that ovarian cancer is largely a disease of older women, more research is needed on the issues that are particularly relevant for the care of older women with ovarian cancer, including differing preferences, treatment goals, and supportive care needs along the survivorship trajectory (see Chapter 5).
Obesity
Obesity is a growing challenge in the treatment of cancer in general. Obesity complicates treatment by creating challenges in determining the correct dosage for chemotherapy, in performing surgery (e.g., airway complications), and in administering radiation therapy (Calle et al., 2003; Griggs et al., 2012; Sparreboom et al., 2007; Washam, 2012; Welsh et al., 2011). Obesity creates similar challenges in the treatment of gynecologic cancers (Blikkendaal et al., 2015; Modesitt and van Nagell, 2005). Poor nutrition, sedentary lifestyle, and obesity have been associated with a lower quality of life and survival rates among women with ovarian cancer (Nagle et al., 2015; Pavelka et al., 2006; Smits et al., 2015a; Torres et al., 2013). (See Chapter 5 for more on survivorship issues related to diet and exercise.) However, one study did find that survival rates were similar between obese and non-obese women when optimal debulking surgery was performed in both sets of patients (Matthews et al., 2009), and another showed that obesity was associated with wound complications and prolonged hospital stay, but not with cytoreduction status or 30-day mortality (Smits et al., 2015b). Therefore, more research is needed both on how to adjust treatment approaches for obese women and also on how to incorporate weight and nutrition counseling into the treatment plan.
Patient Engagement
The 2013 IOM report stated, “A high-quality cancer care delivery system depends upon clinical research that gathers evidence of the benefits and harms of various treatment options so that patients, in consultation with their clinicians, can make treatment decisions that are consistent with their needs, values, and preferences” (IOM, 2013, p. 207). However, the report found that patient-centered communication and shared decision making in oncology is “suboptimal,” with clinicians asking for patient preferences in medical decisions only about half the of the time (IOM, 2013). The patient’s voice is often lost when clinicians are developing treatment plans for women with ovarian cancer, and more needs to be done to ensure better communication between women and their care providers when developing such plans. (See Chapter 5 for more on patient preferences, shared decision making, and patient–provider communication.)
The possibilities for patient engagement go beyond simply tailoring treatment plans to take patient preferences into account. In practice, all types of patients play a role in their own care teams, and they can contribute to their own care in a variety of ways. One example is the use of patient-reported outcomes (PROs) to help inform the care team. PROs can facilitate the systematic assessment of disease- and treatment-related
symptoms and impairments in quality of life in both clinical and research settings, but these data are often underreported. (See Chapter 5 for more on patient-reported outcomes.) Researchers should consider using endpoints in clinical trials that better reflect the outcomes of greatest importance to women with ovarian cancer (e.g., quality of life versus overall survival). Furthermore, more research is needed into the issue of how better to include the collection and validation of PROs in the health care delivery system. For example, various technologies, such as wearable sensors, and real-time data may be useful in improving both patient self-management and provider assessment and monitoring (Appelboom et al., 2014; Cereda et al., 2007; Dobkin and Dorsch, 2011).
Access to Care
The previous IOM reports on cancer care identified several factors that affect access to cancer care, including
- Health insurance coverage;
- Affordability of care (e.g., cost of care, reduction in work productivity, and transportation);
- Health care delivery system attributes (e.g., geographic distribution of facilities);
- Patient attributes (e.g., health literacy, language, or cultural factors); and
- Clinician attributes (e.g., communication style and cultural competency) (IOM and NRC, 1999; IOM, 2013).
For women with ovarian cancer, receiving care in cancer centers that have experience in treating ovarian cancer (i.e., high-volume centers) or from gynecologic oncologists is associated with an increased likelihood of receiving the standard of care and with better outcomes, but getting access to such care can be a challenge (Bristow et al., 2009, 2014; Cliby et al., 2015). A recent study found that two-thirds of all centers providing initial management of ovarian cancer treat one to seven cases annually (Cliby et al., 2015). The limited research that has been done with women with ovarian cancer has found disparities in access to high-volume cancer centers, with women from racial and ethnic minority groups, women with low socioeconomic status, and women on Medicaid being less likely to have access to these centers (Aranda et al., 2008; Bristow et al., 2014). One study showed that women treated in public hospitals in New York City were less likely to have surgery by a gynecologic oncologist and less likely to have a surgeon who treated a high volume of ovarian cancer cases (Boyd et al., 2011). Tele-oncology may be one way to provide improved access to care to
women in rural areas or at centers without gynecologic oncologic specialists (Shalowitz et al., 2015). Other models of care will need to be developed and assessed for their ability to improve access to the standard of care.
Women with ovarian cancer also experience challenges in accessing recommended genetic counseling and testing (see Chapter 3). There are various patient-, provider-, and system-level barriers that hinder cancer genetics referrals, including patients being unaware that they have a family history of cancer, the limited time that providers have to collect family histories, and complex and non-standardized referral criteria (Hampel et al., 2015). Again, it will be necessary to develop and assess new models of care and new methods for delivering necessary information (e.g., the use of video) to patients.
Finally, women with ovarian cancer may not receive the appropriate standard of care when being treated for ovarian cancer because of insurance coverage policies that preclude them from going to particular providers (e.g., gynecologic oncologists and high-volume cancer centers) or that preclude payment for certain genetic tests.
Health Care Workforce
The 2013 IOM report Delivering High-Quality Cancer Care noted that “the cancer care team includes those with specialized training in oncology, such as oncologists and oncology nurses, other specialists and primary care clinicians, as well as family caregivers and direct care workers” (IOM, 2013, p. 153). The report pointed to the need for adequate numbers of health care clinicians with training in oncology and for new models of interprofessional, team-based care. The delivery of care in accordance with the standard of care in part depends on the number of qualified and available members of the cancer care team, the capacity to adequately train a qualified (and larger) cancer care workforce, and the ability to pay for specialty care (e.g., genetic counseling) (IOM, 2009, 2013).
Research has shown that women with ovarian cancer who are provided treatment (e.g., assessment and cytoreductive surgery) by a gynecologic oncologist have longer survival times than women treated by a general gynecologist or general surgeon (Bristow et al., 2009, 2013b; Chan et al., 2007b; Eisenkop et al., 1992). Furthermore, NCCN guidelines recommend that surgery on an ovarian carcinoma be carried out by a gynecologic oncologist (NCCN, 2015). One study has also shown that while the use of gynecologic oncologists did not vary across racial and ethnic groups, women older than 70 were less likely than younger women to receive care from a gynecologic oncologist (Austin et al., 2013). Little evidence exists on the level and types of training that health care professionals other than gynecologic oncologists receive in caring for women with ovarian cancer.
Given that most cancers are diagnosed in older adults, enhancing the geriatrics competence of cancer care teams may be important (IOM, 2008, 2013). Consultation with clinicians with expertise in geriatrics may also be warranted (Girre et al, 2008; Schiphorst et al., 2015). The 2013 IOM report recommended that “members of the cancer care team should coordinate with each other and with primary/geriatrics and specialist care teams to implement patients’ care plans and deliver comprehensive, efficient, and patient-centered care” (IOM, 2013, p. 10). Furthermore, as noted above, patient–provider communication about patient preferences is often lacking. In part, this is due to “clinicians’ lack of training in communication and insensitivity to patients’ informational, cultural, and emotional needs” (IOM, 2013, p. 102). Given this situation, more consideration may be needed for the use of other health care professionals (e.g., nurses and social workers) or the use of technology (e.g., mobile health technologies) in innovative interdisciplinary models of care so that patient–provider communication between women and their care teams is enhanced and the delivery of important information is improved.
While family caregivers provide much needed care and support in general, they are often neglected as an important part of the health care workforce. For older adults, family and friends often play such roles as helping make treatment decisions, accompanying patients on health care visits, providing assistance with daily actitivies, and even providing an increasingly complex level of medical care within the home (IOM, 2008). In 2013, the IOM noted that family caregivers are “particularly important in cancer care because of the debilitating effect of the disease; the side effects associated with many of the common cancer treatments; the complexity of the medical decisions; and the ongoing need for medical treatment, home care, and surveillance” (IOM, 2013, p. 183). Little research has been done specifically on family caregivers for women with ovarian cancer, but one study showed that these caregivers express concerns about a lack of support from the health care community in addressing disease-specific needs and also express a need to connect with others in similar situations (Ferrell et al., 2002).
As noted above, patients themselves also play a sizeable role on their own care team. As the 2008 IOM report noted, “Patients play a major part in determining treatment plans, navigating the delivery system to obtain services, and ensuring overall adherence to the selected course of treatment. For older adults, these care plans often involve multiple providers and settings, and they can find themselves functioning as coordinators of the entire process” (IOM, 2008, p. 241). Chapter 5 provides a more detailed discussion on the role for women with ovarian cancer in helping to determine and manage their treatment plan and course, particularly through the use of shared decision making, self-management, and PROs.
Cancer Quality Metrics
Cancer quality metrics “serve a number of roles in assessing quality of care by providing a standardized and objective means of measurement” (IOM, 2013, p. 272). Several organizations (e.g., the National Quality Forum, the Agency for Healthcare Research and Quality, the American Society for Clinical Oncology, and the American College of Surgeons’ Commission on Cancer Care have developed or endorsed quality measures for cancer care (or measures that can be applied to cancer care) (IOM, 2013). Such measures can be used for performance improvement initiatives and to drive continuous quality improvement in the delivery of the standard of care. The 2013 IOM report reviewed several challenges to the development of cancer care quality metrics, including the failure to address the entire trajectory of cancer care and the underrepresentation of older adults in quality measurement. In particular, the report found that “there are few or no measures for other rare cancers, such as brain and ovarian cancers” (IOM, 2013, p. 281).
The SGO has developed a list of quality indicators for ovarian cancer based on current evidence (see Box 4-3). One study of 123 women with ovarian cancer foud compliance with such quality indicators to be variable (Liang, 2015).
A better understanding of histologic subtypes of ovarian carcinomas and their molecular features (see Chapter 2) has led to a more targeted approach in the development and use of new therapeutic treatments. In light of the growing number of new therapeutics, innovative early-phase clinical trials that incorporate molecular biomarkers of efficacy are needed to help identify which ovarian cancer histologic and molecular subtypes are likely to be resistant or responsive to specific therapies. This section covers research on the newly introduced classes of therapeutics along with novel therapy development strategies and technologies.
Targeted Therapies: Current Approaches
During the past 20 years improvements in the overall survival rates for ovarian cancer have lagged behind those for a number of other solid malignancies (Vaughan et al., 2011). The incremental gain in overall survival for ovarian cancer is largely attributable to changes in chemotherapy schedules and routes of administration (Armstrong et al., 2006). A recent study found that the addition of a third cytotoxic agent (on top of the platinum-taxane standard) provided no benefit in PFS or overall survival (Bookman et al., 2009).
BOX 4-3
SGO Quality Indicators for Ovarian Cancer
- Operative report with documentation of residual disease within 48 hours of cytoreduction for women with invasive ovarian, fallopian tube, or peritoneal cancer
- Complete staging for women with invasive Stage I–IIIB ovarian, fallopian tube, or peritoneal cancer who have undergone cytoreduction
- Intraperitoneal chemotherapy offered within 42 days of optimal cytoreduction to women with invasive Stage III ovarian, fallopian tube, or peritoneal cancer
- Intraperitoneal chemotherapy administered within 42 days of optimal cytoreduction to women with invasive Stage III ovarian, fallopian tube, or peritoneal cancer
- Platin or taxane administered within 42 days following cytoreduction to women with invasive Stage I (grade 3), IC–IV ovarian, fallopian tube, or peritoneal cancer
- Venous thromboembolism prophylaxis administered within 24 hours of cytoreduction to women with invasive ovarian, fallopian tube, or peritoneal cancer
- Order for prophylactic parenteral antibiotic administration within 1–2 hours before cytoreduction for women with invasive ovarian, fallopian tube, or peritoneal cancer
- Order for prophylactic parenteral antibiotic discontinuation within 24 hours after cytoreduction for women with invasive ovarian, fallopian tube, or peritoneal cancer
SOURCE: SGO, 2015.
Thus, to maintain progress, researchers have turned to molecularly targeted agents, a number of which have been developed and used to manage various malignancies. Unlike most traditional cytotoxic anticancer drugs, these drugs target tumor cells, tumor stroma, tumor vasculature, and aberrant signaling mechanisms in the tumor. For example, the Cancer Therapy Evaluation Program (CTEP)1 of the NCI, which sponsors clinical trials for new anticancer agents, focuses on agents that target pathways involved in apoptosis, survival and proliferation, migration and invasion, angiogenesis, mitosis, protein turnover, immunomodulation, DNA repair and epigenetics, and stem cell signaling (see Appendix C for a list of current ovarian cancer clinical trials compiled from www.ClinicalTrials.gov).
________________
1 For more information, see http://www.ctep.cancer.gov (accessed September 22, 2015).
Anti-Angiogenic Therapy
Angiogenesis (blood vessel development) in tumors increases the perfusion of oxygen and nutrients into tumor cells and thereby promotes tumor growth and facilitates tumor metastasis (Spannuth et al., 2008). Several proteins, such as VEGF and its receptor (VEGFR), play important roles in angiogenesis. VEGF stimulates cellular pathways that lead to the formation and branching of new tumor blood vessels, tumor growth, and increased metastatic potential (Hicklin and Ellis, 2005). It is also hypothesized that anti-angiogenic therapies help to balance angiogenic signaling and normalize the vasculature, thus improving the delivery of anticancer drugs to the tumor (Goel et al., 2012). Anti-angiogenic therapies have also been shown to reverse immunosuppression, which suggests it might be effective to use anti-angiogenic therapies in combination with immunotherapies (Mauge et al., 2014). Accordingly, therapies have been designed to inhibit the VEGF–VEGFR pathway with the goal of producing a rapid and sustained anti-angiogenic and antitumor response, and research has found such an approach to be effective. In particular, anti-angiogenic therapies (e.g., bevacizumab) have been shown to be effective in treating recurrent ovarian cancer both when used as a single agent and in combination with chemotherapy or other targeted agents (Liu et al., 2014a). Although no anti-angiogenic therapies have been approved by the FDA for the front-line treatment of ovarian cancer, the FDA has approved the use of bevacizumab in combination with chemotherapy for the treatment of women with platinum-resistant, recurrent ovarian cancer (Eskander and Tewari, 2014). In Europe, the European Medicines Agency has approved a number of anti-angiogeneic therapies (including bevacizumab) for the treatment of advanced primary, recurrent, and metastatic ovarian cancer (Eskander and Tewari, 2014).
The anti-angiogenic signaling process is complex—it is characterized by multiple pathways that not only overlap but have compensatory mechanisms. Besides the VEGF–VEGFR pathway, a number of other pathways also regulate tumor growth and metastasis and provide compensatory mechanisms when VEGF signaling is blocked. Therefore, recent anti-angiogenic therapies have been designed to simultaneously block both VEGF and VEGFR signaling and other pathways that are critical to angiogenesis and tumor growth. The use of additional anti-angiogenic agents that target VEGFR and other kinases has had variable results (Biagi et al., 2011; Campos et al., 2013; Coleman et al., 2011; Davidson and Secord, 2014; du Bois et al., 2014; Friedlander et al., 2010; Gotlieb et al., 2012; Hirte et al., 2015; Ledermann et al., 2011). This variability in response is most likely a reflection of the highly heterogeneous nature of the disease, the high rate of drug resistance, and the presence of compensatory angiogenic mechanisms. Therefore, dis-
rupting multiple signaling pathways with a combinatorial approach may provide an improved response.
Various challenges remain in finding effective biomarkers or molecular signatures that will predict sensitivity to anti-angiogenic therapies or predict increased risk of toxicity (Aghajanian et al., 2012; Collinson et al., 2013; Eskander and Tewari, 2014; Monk et al., 2014; Pujade-Lauraine et al., 2014). Currently, bevacizumab is licensed as a molecularly unselected agent, meaning that patients are not molecularly stratified into groups to determine if one group responds better than another group. However, certain molecular features may actually predict a tumor’s response to treatment (Collinson et al., 2013; Gourley et al., 2014; Symeonides and Gourley, 2015; Wehland et al., 2013). These biomarkers will need further verification, but hopefully, by stratifying patients according to the histologic and molecular subtypes of each ovarian cancer subtype, it will be possible to achieve better outcomes for women with ovarian cancer.
PARP Inhibitors
Another cellular process that is key for the long-term survival of tumors is the cell’s DNA repair in response to damage caused by chemotherapeutic agents and genomic instability. The main DNA repair process is high-fidelity (nearly error-free) homologous recombination (HR), which is mediated by several protein products, including those of the BRCA1 or BRCA2 genes (Gudmundsdottir and Ashworth, 2006). If the cell does not have a functional HR pathway, alternative damage repair processes are activated, such as base excision repair (BER) and error-prone nonhomologous end joining (NHEJ). The rate-limiting step of BER is the activation and activity of the PARP enzyme (Amé et al., 2004; Dantzer et al., 2000). If both HR and BER are disabled and only NHEJ remains functional, then the already genetically instable tumor cells rapidly acquire enough genetic mutations to cause cell death (Ashworth, 2008; Bryant et al., 2005; Farmer et al., 2005). Thus, researchers expected that drugs of the newly developed class of PARP inhibitors would be effective against tumors with faulty HR repair processes (Ashworth, 2008; Farmer et al., 2005; Feng et al., 2015).
Studies have indeed shown that PARP inhibitors, alone or in combination with chemotherapy, are effective in women with recurrent ovarian cancer, particularly those with a BRCA1 or BRCA2 mutation. In particular, olaparib improves PFS when used as a maintenance therapy in the context of BRCA1 or BRCA2 mutations, platinum sensitivity, and recurrent disease (Audeh et al., 2010; Fong et al., 2009; Ledermann et al., 2012; Menear et al., 2008). In December 2014, the FDA approved olaparib as a therapy for women with recurrent ovarian cancer who have a BRCA1 or BRCA2 mutation and who have had three prior chemotherapy treatments
(FDA, 2014b). Two months before that, the European Medicines Agency had approved olaparib as first-line maintenance therapy for women with platinum-sensitive, recurrent HGSC who are in complete or partial response to platinum-based chemotherapy (EMA, 2014). Additional evidence suggests that the addition of olaparib to chemotherapy improves PFS in platinum-sensitive recurrent ovarian cancer (Oza et al., 2015). Current trials are exploring the use of PARP inhibitors, including in combination with other molecularly targeted agents (see Appendix C). Preliminary evidence suggests that the combination of the anti-angiogenic cedirinab and the PARP inhibitor olaparib may be as effective as cytotoxic chemotherapy regimens (Liu et al., 2014b).
Targeted Therapies on the Horizon
Advancements in technology can catalyze drug discovery. For example, next-generation sequencing (NGS) technologies and data from such initiatives as The Cancer Genome Atlas (TCGA) have allowed researchers to improve their understanding of aberrations and the pathways that are perturbed in ovarian cancer (Cancer Genome Atlas Research Network, 2011). In addition, functional genomics approaches make possible large-scale screening methods based on the loss of gene expression (Cong et al., 2013; Mali et al., 2013; Wood et al., 2011). Immunoscreening has also been used to help identify ovarian-specific immune targets (Sahin et al., 1995). The integration of these approaches with genomic platforms may help researchers identify additional targets.
Precision Medicine
Precision medicine involves the development of molecularly targeted therapies that take into account the variability in genes among individuals. The genomic information derived from modern sequencing technologies aids in the development of precision medicine and may lead to a new paradigm for clinical investigation and for crafting treatment decisions for individual patients. The use of NGS has led to the development of targeted therapies for various solid tumors (Chapman et al., 2011; Garnett et al., 2012; Kwak et al., 2010), and these data may also lead to new indications for already approved cancer drugs if identical mutations exist in other cancer types, including ovarian cancer. The NCI-MATCH trial represents a new method for evaluating targeted therapies, which is needed for precision medicine initiatives. However, it should be noted that some ovarian carcinoma subtypes, such as HGSC, may have limited actionable mutations. While targeting specific genetic changes in defined patient subsets has been successful, there could be a wide range of responses to appropriately
selected therapies. Furthermore, a large number of cancer drugs have not been linked to specific genomic alterations that could be used as biomarkers to specify their potential therapeutic efficacy.
The molecular analysis of HGSC by TCGA Research Network revealed the presence of “TP53 mutations in almost all tumors (96 percent) [and] low prevalence but statistically recurrent somatic mutations in nine further genes” (Cancer Genome Atlas Research Network, 2011, p. 609). Because p53 is the nexus of various tumor suppressive pathways, it would probably be of benefit to reactivate or restore p53 function in order to revert or rescue cells from resistance to standard chemotherapeutic treatments (Carrillo et al., 2015; Mohell et al., 2015). A clinical trial is currently under way to investigate the use of a p53-reactivating compound APR-246 in ovarian cancer (Aprea, 2014).
As newer mutations are discovered, novel therapeutics may be developed to block the pathways that promote tumor growth and survival. For example, low-grade serous ovarian carcinomas frequently harbor mutations in KRAS or BRAF and express active mitogen-activated protein kinase (MAPK) (Coward et al., 2015). Patients with these carcinomas may benefit from agents that inactivate the ERK1/2 pathway. PI3K is found to be mutated in 33 percent of patients with clear cell ovarian cancer, while a loss of PTEN gene expression is noted in 40 percent of these patients. Studies of various inhibitors of the PI3K/AKT pathway are ongoing. TCGA data are guiding the development of other molecularly targeted therapies as well. Notable targets include PI3K, BRAF, MEK, ERK, and MAPK.
Immunotherapy
Cancer immunoediting is a process whereby host immunity functions not only as an extrinsic tumor suppressor but also to shape tumor immunogenicity (Dunn et al., 2004). A central theme of the cancer immunoediting process is that tumor cells express antigens that distinguish them from their nontransformed counterparts, thus permitting their recognition by T cells and their eventual destruction by immunological mechanisms. In ovarian cancer, the presence of intraepithelial CD8+ T cell infiltrates has been shown to correlate with improved survival rates (Hwang et al., 2012; Sato et al., 2005; Shankaran et al., 2001; Zhang et al., 2003). These findings suggest that the endogenous T cell compartment of ovarian cancer patients is able to recognize antigens on the surface of the malignant cells. Therefore, researchers are looking at approaches to generating antitumor immune responses that lead to tumor eradication, including antibody-based therapies, cancer vaccines, immune modulation, and adoptive cellular therapies.
One challenge in evaluating immunotherapeutic approaches to treating ovarian cancer lies in deciding where in the disease course the agent should
be evaluated. A large number of immunotherapeutic strategies are under investigation for women with ovarian cancer (see Appendix C). Most of these trials are pilot studies or small Phase I or Phase II trials with the goal of assessing safety and immunogenicity. Some have correlated improved outcome with a surrogate such as antibody or T cell response, and most current trials aim to produce cellular responses. However, few of these trials are adequately powered, and none has yet shown definitive efficacy.
Another important consideration in the development of immunotherapy is the need for reliable markers of clinical efficacy. Clinical outcomes to immunotherapy are often not accurately assessed by the conventional imaging measures for solid tumors. Indeed, the infiltration of a tumor by active immune cells following therapy may be misleadingly diagnosed as tumor growth and disease progression. These considerations led some researchers to propose a set of immune-related response criteria in an attempt to capture the additional response patterns observed with immune therapy (Wolchok et al., 2009).
Antibody-Based Therapies
The anti-CA-125 antibody oregovomab binds with high affinity to circulating CA-125 and generates both a cellular and a humoral immune response. Its use in ovarian cancer has had mixed results (Baum et al., 1993; Berek et al., 2003, 2004; Ehlen et al., 2005; Mobus et al., 2003; Noujaim et al., 2001). Another anti-CA-125 antibody, abagovomab, failed to demonstrate significant prolongation of PFS or overall survival in ovarian cancer patients in first remission (Sabbatini et al., 2013). Although neither approach showed a survival improvement, interest remains in considering CA-125 as a target for studies with other immunotherapy approaches because CA-125 is overexpressed on most ovarian carcinoma cells and expressed at low levels in other tissue sites, and because preclinical data support its role in the modulation of ovarian tumor growth and invasiveness (Gubbels et al., 2006).
Cancer Vaccines in Ovarian Cancer
The development of ways to analyze humoral and cellular immune reactivity to cancer has led to the molecular characterization of tumor antigens that are recognized by autologous CD8+ T cells or antibodies (Alpen et al., 2002; Boon and van der Bruggen, 1996; Chen et al., 1997; Sahin et al., 1995; Scanlan et al., 2000; Van den Eynde and Boon, 1997; van der Bruggen et al., 1991). When the NCI developed a priority-ranked list of cancer vaccine target antigens (Cheever et al., 2009), the top 10 antigens included WT1, MAGE-A3, and NY-ESO-1, all of which are tumor
antigens that are well known to be expressed in a portion of ovarian cancer patients (Daudi et al., 2014; Hylander et al., 2006; Odunsi et al., 2003). Among these antigens, NY-ESO-1 has been investigated most extensively for patients with ovarian cancer (Odunsi et al., 2007, 2012, 2014; Sabbatini et al., 2012). While studies with these antigens have found that using vaccines to generate integrated humoral and T cell immune responses appears to have some clinical benefit, it is not yet possible to come to definitive conclusions on efficacy because of the small study sizes. In addition, early preclinical and clinical data suggest promise for vaccination with neoantigens, but more study is needed (Castle et al., 2012; Duan et al., 2014; Gubin et al., 2014; Linnemann et al., 2015; Rizvi et al., 2015; Wick et al., 2014). Remaining questions include: Are monovalent or multiantigen vaccine approaches likely to provide better results? At what disease state is vaccination appropriate? What is the optimal frequency and duration of vaccination? How should antigen-specific immune responses be monitored? How should the induced immune response be sustained?
Immune Modulation
Immune modulation acts to reinstate an existing anticancer immune response or to elicit novel responses as a result of antigen spreading. Studies of immune modulation have been carried out in a variety of solid tumors, but there has been more limited research with ovarian cancers. One approach to immune modulation, checkpoint blockade (the inhibition of immunosuppressive receptors expressed by activated T lymphocytes), has been shown to induce robust and durable responses in studies on a variety of solid tumors (Brahmer et al., 2012; Robert et al., 2014). A number of checkpoint-blocking antibodies are approved by international regulatory agencies for use in humans (Hodi et al., 2010; Tumeh et al., 2014). The use of checkpoint-blocking antibodies in treating cancer is expanding, but these agents have not been tested extensively nor have they been approved for use in treating ovarian cancer (Brahmer et al., 2012; Disis et al., 2015; Varga et al., 2015). One challenge in treating ovarian cancer is that because of the heterogeneity of the disease, it is important to identify the full spectrum of immune resistance mechanisms. For example, emerging evidence suggests that the clinical efficacy of immunomodulatory antibodies (especially checkpoint blockers) may be profoundly influenced by the mutational burden and neoantigens specific to the particular cancer subtype (Snyder et al., 2014). A higher neoantigen load can lead to recruitment of a diverse repertoire of neoantigen-specific T cells, and checkpoint blockade may help to restore a favorable ratio of antigen-specific effector cells to regulatory T cells. Researchers do not yet know which patients are most likely to respond to
immune modulation and checkpoint blockade in the treatment of ovarian cancer, so studies on this issue are warranted.
Adoptive Cellular Therapies
Adoptive cell transfer (ACT) involves the live collection, modification, expansion, and activation of circulating or tumor-infiltrating lymphocytes that are then reinfused into patients (Rosenberg et al., 2008). Initial studies demonstrated the potential of T cell immunotherapy to eradicate solid tumors (Dudley et al., 2002, 2005). In order to improve the therapeutic potential of transferred cells, peripheral blood lymphocytes with unique antigen specificity can be genetically modified (Kalos et al., 2011; Robbins et al., 2011; Rosenberg et al., 2008). Although remarkable responses have been observed in patients with other solid tumors (Dudley et al., 2005; Robbins et al., 2011), the FDA has not approved any ACT protocol for use in ovarian cancer patients. An ongoing clinical trial in patients with ovarian cancer may shed some light on the potential role of ACT in this disease (Adaptimmune, 2012). The goals of future studies of ACT in patients with ovarian cancer should include identifying biomarkers of response or nonresponse and determining whether this approach should be combined with other immunotherapy strategies.
Immunostimulatory Cytokines
Immunostimulatory cytokines are generally used as adjuvants for other anticancer immunotherapeutics. Although many attempts have been made to use these cytokines in patients with ovarian cancer, no randomized consolidation study has demonstrated a significant improvement in overall survival (Alberts et al., 2006; Hall et al., 2004).
CLINICAL TRIALS FOR OVARIAN CANCER
Clinical researchers are regularly developing new trial designs in order to accommodate new approaches to the development of anticancer agents, to make the trial designs more efficient, and to study types of therapies that have not been evaluated before. Many of these new approaches are due to the shift away from a primarily empiric development of cytotoxic therapies toward the development of molecularly targeted agents. Limited resources for conducting clinical trials and shrinking patient populations for which precision targeted agents are appropriate have made it more difficult than ever to come up with efficient clinical trial designs. Design complexity and novelty do not guarantee design quality or utility. Thorough evaluation and transparency are needed if one is to choose an optimal clinical trial design
that most efficiently and convincingly answers a specific clinical question. The following sections provide a brief overview of clinical trial design and how study designs are being adapted to increase efficiency and to include biomarker analyses relevant to modern molecularly targeted therapies in ovarian cancer.
Preclinical Studies
Preclinical studies involve the testing of anticancer agents in biological systems such as tumor cell line cultures or animals (see Chapter 2). Although preclinical models do not always accurately represent the human manifestation of ovarian cancer, researchers may still glean much useful evidence regarding anticancer agent activity against tumor cells or on a particular molecular target within those cells. Preclinical studies can also provide greater insights into the mechanisms of action of an agent and into an agent’s potential toxicities.
A better understanding of the biological mechanisms underpinning a drug’s activity make it easier to develop optimal designs of clinical trials aimed at identifying patients who are most likely to benefit from a new therapy and least likely to experience toxicities. Few patient-derived xenograft models have been developed from HGSCs (Scott et al., 2013). Greater incentives and resources for the creation of such models could benefit ovarian cancer drug development programs and, if the models are successful, could increase the number of promising drugs for human clinical trials.
Clinical Studies
If they meet toxicity and efficacy benchmarks in preclinical studies, novel agents are moved into human clinical studies. These studies are conducted in a series of phases (see Box 4-4) in which new agents are given to various patient populations and examined for the mechanism of action, dose-related toxicities and overall safety, and effectiveness (Rubinstein, 2000).
Phase 0 Studies and Window-of-Opportunity Studies
Phase 0 studies are exploratory first-in-human studies that are intended to bridge evidence from preclinical animal studies; these Phase 0 studies are not conducted with therapeutic intent, and they are intended primarily to examine biological target modulation rather than antitumor activity (Kummar et al., 2007). Phase 0 trials are sometimes embedded as window-of-opportunity (WOO) studies in Phase II or Phase III trials to evaluate the clinical benefit of a new therapy, but WOO trials typically use clinical
BOX 4-4
Clinical Study Phases
Phase 0
- These are exploratory “first time in human” studies to test an agent’s mechanisms of action, bioavailability, pharmacodynamics, and pharmacokinetics (no therapeutic intent).
- Small numbers of patients are given subtherapeutic doses for short durations to compare drug metrics with preclinical results.
Phase I
- The primary intent is to assess an agent’s toxicity profile, determine the maximum tolerated dose, determine related dose-limiting toxicities, and establish a suitable dose for further human testing.
- Phase I studies usually enroll patients with advanced cancer who have exhausted standard treatment options.
Phase II
- The primary intent is to assess the efficacy of new agents (potentially in combination with each other or standard treatments), gather more data on safety, and determine whether an agent looks sufficiently promising to be further developed in Phase III trials.
- The agent dose is estimated from Phase I studies.
- Phase II studies usually enroll patients who were treated with one or more standard therapies without eradication of their cancer or who have a cancer for which no standard effective therapy exists.
Phase III
- Phase III studies are comparative trials intended to assess the effectiveness of a new agent or agent combination compared to existing standard of care.
- Randomized controlled trials are considered to be the gold standard design for Phase III studies, but in some situations historical control or concurrent control designs may be considered acceptable.
Phase IV
- The main goal is to acquire more real-world experience for a new agent following marketing authorization in order to gain information in a broader patient population that encompasses a variety of disease severities and comorbid conditions, with the goal of better understanding the agent’s effectiveness and to identify rare or late-appearing adverse effects.
- Phase IV studies may include studies that collect extended follow-up data on patients who have completed Phase III trials, noninterventional observational studies, large simple trials, and post-marketing surveillance studies.
tumor response or PFS endpoints to establish evidence of antitumor activity for a new agent (Glimelius and Lahn, 2011). In a WOO trial, a patient agrees to delay conventional therapy for a few weeks in order to observe the antitumor effect of a new agent.
Figure 4-1 shows one example of a WOO study embedded in an adjuvant therapy trial. At the investigators’ institution, the standard clinical evaluation plan (i.e., the Anderson algorithm) specifies that, prior to any treatment, women who are presumed to have advanced-stage ovarian cancer undergo laparoscopic evaluation to determine the likelihood that the tumor can be completely resected. If the tumor is unlikely to be completely resected (a score of 8 or more), the recommended course of action includes immediate NACT with a novel agent in combination with a standard chemotherapy regimen, followed by debulking surgery. Women who have a low score are offered participation in a WOO study that involves the administration of a novel single agent prior to debulking surgery and the option to enroll post-operatively in a randomized adjuvant therapy study comparing novel therapy to standard therapy. In both cases, tissue samples are collected both at the time of the laparoscopy and at the time of surgery, which allows researchers to examine the in vivo effects of the novel agents and may provide valuable insights into the mechanism of action or resistance for those agents.
Phase I Clinical Trials
Phase I studies attempt to find a dose that provides an acceptable balance between killing tumor cells and patient toxicity by delivering increasing doses of the experimental agent to successive patients while they are
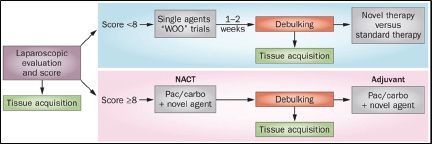
FIGURE 4-1 Novel clinical trial design using the Anderson algorithm.
SOURCE: Reprinted by permission from Macmillan Publishers Ltd: Nature Reviews: Clinical Oncology. Nick, A. M., R. L. Coleman, P. T. Ramirez, and A. K. Sood. A framework for a personalized surgical approach to ovarian cancer. 12(4):239-245. Copyright 2015.
carefully monitored for signs of toxicity. The dose escalation ceases when the maximally tolerated dose is identified. The various Phase I designs differ in the criteria they use for progressing through a sequence of doses. Three of the most commonly used Phase I study designs are the 3+3 cohort expansion, accelerated titration, and continual reassessment designs (Ivy et al., 2010). The conventional 3+3 cohort expansion design increases doses in successive cohorts of three patients. Accelerated titration designs are characterized by a more rapid initial dose escalation and the potential for doses to be increased within patients in addition to across patients (Simon et al., 1997). The continual reassessment method relies on a continually updated mathematical model that expresses the relationship between dose and the probability of toxicity (O—Quigley et al., 1990). If rapid dose escalation were more widely adopted when there are scant preliminary data to suggest the starting dose level, Phase I studies could be completed more quickly with fewer patients undertreated and with minimal increase in risk of serious toxicities (Horstmann et al., 2005; Kurzrock and Benjamin, 2005; Simon et al., 1997). Phase I clinical trials have increasingly enrolled additional patients in expansion cohorts, but they often lack statistical justification for the cohort size (Dahlberg et al., 2014). The recent PARP inhibitor studies are an example of the successful use of expansion cohorts for ovarian cancer clinical trials (Fong et al., 2010). Clearer guidance is needed so that the expectations for the goals, study designs, and reporting of Phase I trials are better understood and so that the available resources for clinical research are most appropriately utilized.
Phase II Clinical Trials
Randomized Phase II trials allow for a preliminary assessment of the clinical benefits of an experimental therapy relative to other therapies, but they also require larger sample sizes. Single-arm trials with a tumor-response endpoint have been a mainstay of oncology Phase II trials, but newer anticancer agents often do not fit into the traditional paradigm of cytotoxic chemotherapies. Randomized Phase II trials may be needed in situations where it is difficult to specify historical benchmarks for the clinical endpoint of interest or when a new agent will be evaluated in combination with a standard therapy that is known to be effective (Rubinstein et al., 2009). When the new agent is evaluated in combination with a known effective therapy, a single-arm trial cannot determine the relative contribution of the new agent (Rubinstein et al., 2005).
Biomarkers are increasingly being used in Phase II cancer clinical trials, largely because of the rise in the number of molecularly targeted agents under development (McShane and Hunsberger, 2015; McShane et al., 2009). Key considerations in such trials include whether the trial eligibility should
be restricted to only those patients whose tumors possess the presumed target of the new agent and whether trials of targeted agents should be agnostic to type of tumor so that the focus is the target molecular characteristics, regardless of tumor histology. In ovarian cancer clinical trials, biomarkers are occasionally incorporated into the definition of the trial endpoint. Most notably, the biomarker CA-125 has sometimes been used as part of a composite endpoint to indicate the progression of the disease. For example, in the clinical trial GOG-218, the incorporation of CA-125 into the definition of progression resulted in an increased estimate of the treatment’s effect (Burger et al., 2011). However, it remains debatable whether incorporating CA-125 in this way strengthens the quality of evidence and whether the composite endpoint more reliably predicts overall survival (Herzog et al., 2014).
Phase III Clinical Trials
Phase III clinical trials are intended to provide definitive evidence about whether an experimental therapy has clinical efficacy when compared to a standard treatment or no treatment. These trials are usually randomized to guard against biases resulting from patient selection. Exceptions to randomization include where there are no known effective therapies or when patients are expected to have extremely good outcomes on standard therapy and a randomized trial would have to be prohibitively large to allow a reliable comparison of the treatments. In the case of ovarian cancer, overall survival and PFS are most commonly used as the primary endpoint for late-phase clinical trials, although the merits of PFS are increasingly being debated (Korn and Crowley, 2013; Korn et al., 2011). (See the discussion of clinical trial endpoint selection later in this chapter.)
An increasing number of Phase III oncology clinical trial designs incorporate biomarkers (producing what is referred to as “biomarker-driven trial designs”) under the hypothesis that the biomarkers will help identify a subset of patients who are more likely to benefit from the experimental therapy. However, biomarkers are also used as stratification factors or eligibility criteria to control the variability in expected patient outcomes due to differing baseline prognoses associated with the biomarker values. (See the discussion of prognostic and predictive [treatment-selection] biomarker-based tests later in this chapter.)
The three basic types of biomarker-driven Phase III trial designs are the enrichment design, the stratified design, and the strategy design. The enrichment design randomizes only patients whose tumors are positive for the biomarker, while the stratified design uses a randomization procedure stratified on biomarker values. The strategy design randomizes patients into a biomarker-testing arm or a no-biomarker arm, and patients in the arm
with biomarker testing are assigned to treatment based on a predefined algorithm, depending on the biomarker. The IOM and others have discussed the advantages and disadvantages of the different biomarker-driven designs (Freidlin et al., 2010; IOM, 2012; Sargent et al., 2005). The best choice of design in a particular situation depends on the strength of the evidence supporting the biomarker, the biomarker’s relationship to treatment benefit or outcome, and the feasibility of its use (Freidlin and Korn, 2014).
Multiarm Phase II and III Clinical Trial Designs
Extending the conventional single- and two-arm clinical trial designs to three or more arms is relatively straightforward and can offer substantial advantages. In a multiarm trial, a single clinical trial infrastructure can be used, and a greater percentage of patients are assigned to an experimental therapy arm. Multiarm randomized trials are also statistically efficient in that multiple research questions can be addressed with fewer total patients (because data from any treatment arm can be reused) (Freidlin et al., 2008). For example, in GOG-218, women with newly diagnosed Stage III or Stage IV ovarian carcinoma who were unable to have a complete resection of their tumors were randomized to one of three treatment arms (Burger et al., 2011). All three arms included IV paclitaxel plus carboplatin for cycles 1 through 6, plus a study treatment for cycles 2 through 22. The control arm included a placebo for cycles 2 through 22, the second arm added bevacizumab in cycles 2 through 6 and then a placebo in cycles 7 through 22, and the third arm added bevacizumab in cycles 2 through 22. The trial found a prolongation of median PFS by 4 months with use of bevacizumab during and up to 10 months.
A more specialized type of multiarm trial design is a factorial design. In a 2x2 factorial design, four possible combinations of two treatments (e.g., A and B) can be considered: neither treatment A nor B (if appropriate), A alone, B alone, and the combination of A and B. Data from the arm with both therapies can be combined with data from either arm A or arm B to evaluate the effects of the individual therapies as long as the combination of A and B can reasonably be assumed to have an additive effect (Green, 2005). The use of factorial designs is likely to become increasingly important as more effective monotherapies become available and interest turns to the evaluation of combination therapies.
The I-SPY2 Phase II breast cancer trial is the first oncology clinical trial in the United States to use a platform trial structure in which multiple agents can be evaluated seamlessly over time (Barker et al., 2009). This study required the development of protocols that allowed the rapid addition of new investigational agents. In I-SPY2, each arm corresponds to a particular drug under investigation in a randomized comparison against a standard
therapy. Multiple pharmaceutical companies provide their drugs for evaluation, but drugs from competing companies are not directly compared to one another. A platform trial design of this sort is adaptable to any cancer type as long as there are sufficient numbers of promising experimental therapies for inclusion in the trial.
Molecular profiling trials can evaluate large numbers of experimental agents along with the biomarkers intended to guide their use. The biomarkers may be measured using a combination of different assay types. High-throughput assays (e.g., NGS platforms), which are capable of searching tumor DNA and RNA sequences for thousands of potential mutations, are being increasingly used. In molecular profiling trials, patients whose tumors have specific subsets of mutations are assigned to treatment arms that include an experimental therapy designed to target tumors with those specific mutations. There are various types of molecular profiling trial designs, each of which offers different advantages and disadvantages (Kummar et al., 2015).
Phase IV Clinical Trials (Post-Marketing Clinical Studies)
After a drug has demonstrated clinical efficacy and has received regulatory approval to be marketed, additional studies are usually conducted to gain experience with the drug’s performance in real-world settings (Suvarna, 2010). These studies may be mandated by regulatory authorities as a condition of drug approval, or the company may choose to acquire more experience with the drug in a broader population under the conditions in which the drugs are likely to be prescribed and used. Post-marketing studies are generally needed because the previous clinical trials typically would involve no more than a few thousand patients, and those patients would have been followed for a limited amount of time. Furthermore, the earlier studies may have excluded sicker patients or patients with a variety of comorbid conditions, providing limited opportunity to observe extremely rare toxicities or toxicities that might appear years after treatment or in the context of certain comorbid conditions. Post-marketing studies are rare, but they may be particularly valuable for use with ovarian cancer treatments due to the cancer’s rarity, the relatively small size of ovarian cancer trials, their typically long clinical trajectory, and the substantial potential for late adverse effects associated with many commonly used treatments for ovarian cancer. One post-marketing study that is currently being set up will evaluate the safety and efficacy of bevacizumad in routine clinical practice in patients with advanced ovarian carcinoma, fallopian tube cancer, or primary peritoneal cancer (Hoffmann-La Roche, 2015). The goal of this study is to measure the incidence of adverse events for approximately 18 months after patients begin taking bevacizumab.
Clinical Trial Endpoint Selection
Ovarian cancers frequently recur, and women may experience multiple episodes of disease progression followed by additional therapies. This pattern presents challenges for the selection of interpretable and clinically meaningful endpoints for clinical trials. It may be difficult to assess an experimental agent’s effect on overall survival because of the dilution effect of a woman having received multiple therapies. PFS may be a good alternative endpoint in these settings, but in order to be considered clinically meaningful, increases in PFS must be substantial and preferably accompanied by some increase in overall survival or increased quality of life, lower toxicity, or other benefits. Furthermore, if an experimental agent is not expected to shrink tumors, then PFS or overall survival is a more informative endpoint than tumor response. A white paper from the SGO discusses the advantages and disadvantages of a variety of endpoints used in ovarian cancer clinical trials. Table 4-2 summarizes the comparisons made in the white paper; the table does not offer an exhaustive list of all potential endpoints, but rather covers those endpoints that are particularly useful in ovarian cancer studies. For example, disease control rate is increasingly being used in trials with targeted agents. However, its utility has been questioned, and because it may not be beneficial to use in ovarian cancer clinical trials, it was not included in the table (Sznol, 2010). PROs are becoming more appreciated as an endpoint because quality of life is a major factor in the treatments that patients select, but which symptoms are most important to patients and how PROs relate to clinical outcome still need further study. The SGO stressed the importance of considering the meaningfulness of various endpoints and patients’ preferences for therapeutics with more dramatic improvements in outcomes relative to existing therapies (Herzog et al., 2014). The importance of limiting toxicities was another clear message, but women surveyed by the SGO indicated that they have a greater tolerance for toxicity related to curative therapy then for toxicity from palliative therapy.
Comparative Effectiveness Studies
In a comparative effectiveness study, the therapies under study have already been shown to have efficacy in the carefully controlled settings of clinical trials, and the goal is to determine which of two or more therapies provides an overall greater benefit (balanced against any risks) in real-world settings (CBO, 2007; Lyman, 2009). These studies can be particularly helpful for diseases like ovarian cancer where there are several available therapies and some significant drug toxicities that need to be weighed against benefits in an objective way. While a randomized trial is considered by many to be the gold standard for a comparative effectiveness study, some
TABLE 4-2
Comparison of Potential Endpoints for Ovarian Cancer Trials
| Endpoint | Definition | Advantages | Disadvantages |
| Response rate | Assessed by the RECIST criteria on the basis of imaging studies, GCIG has defined changes in CA-125 as a response criterion | Objective Quantifiable Results quickly available Tumor shrinkage Appealing to patient and physician | Difficult to measure accurately and reproducibly in ovarian cancer Not considered sufficient as a primary endpoint for Phase III trials Not necessarily a clinically relevant benefit for the patient |
| Patient-recorded outcomes | Symptom-based parameters | Direct clinical benefit as perceived and quantified by patient | Need randomized blinded studies Subjective and dependent on limited validation instruments |
| Progression-free survival | Time from entry into trial to progression of disease, death, or lost to follow-up | Provides answer sooner Avoids the impact of post-progression therapy Preferred to time to progression by regulatory agencies | Does not include deaths |
| Time to progression | Time from entry into trial to progression of disease | Similar to progression-free survival | Does not include deaths |
| Overall survival | Time from entry into trial to death or lost to follow-up | Clear-cut endpoint of death Indisputably indicative of clinical benefit | Longer time to answer Impacted by post-progression therapy |
| Time to tumor growth | Uses prescribed longitudinal tumor models | Novel metric In some models is best predictor of OS Reduces time and cost | Not validated in ovarian cancer Subjectivity in assessment |
NOTE: GCIG = Gynecologic Cancer Intergroup; OS = overall survival; RECIST = Response Evaluation Critera in Solid Tumors.
SOURCE: Herzog et al., 2014.
argue that large observational studies with access to high-quality data—such as might be obtainable from electronic medical records, registries, and adverse event reporting systems—can also be valuable and may be more reflective of real-world experiences. Such studies require careful modeling of the relationships between outcomes, therapies received, and other clinical
and demographic variables and also extreme vigilance for the possibility of confounding effects that could distort the estimated treatment effects (Korn and Freidlin, 2012; Lyman, 2009).
Adaptive Clinical Trial Designs
Adaptive clinical trial designs are one potential way to make clinical trials more efficient (e.g., fewer patients and shorter duration) or more informative and, ultimately, to deliver effective new therapies to patients more rapidly. There are many types of adaptive designs, but all share the use of outcome measures or other data to make adaptations in the trial quickly (relative to the pace of accrual) (Coffey and Kairalla, 2008; FDA, 2010). Adaptations may include restricting eligibility to a subset of patients who appear to be benefitting most from the experimental therapy, increasing the sample size to more accurately estimate treatment effect, changing the primary endpoint, adjusting or adding drug dose levels, adjusting randomization ratios to favor the treatment arm that appears more promising, or stopping a trial early because sufficient evidence has been obtained to confidently answer the treatment question. Interim monitoring rules, which are applied in nearly all Phase III and many Phase II oncology clinical trials, are one example of an adaptive design feature (Redman, 2012).
Bayesian trial designs are a type of adaptive design that combines information obtained from outside the trial with data accumulating within the trial (Berry, 2006). Traditional designs use only data observed within the trial for decision making, although outside data such as anticipated treatment effects and event rates (e.g., relapses or deaths) may be used informally. Bayesian methods summarize prior beliefs about the values of key parameters such as the treatment effect using formal mathematical models called “prior distributions.” As new data are accumulated in the trial, the models are mathematically updated to “posterior distributions” that are used for decision making during the trials and after completion. This mathematical modeling framework offers the flexibility to adapt many aspects of the trial design.
Phase II/III clinical trials, another example of adaptive design, allow for transiting directly from a Phase II to a Phase III trial when an experimental agent demonstrates a positive effect on an intermediate endpoint (Hunsberger et al., 2009; Korn et al., 2012). The intermediate endpoint is the Phase II trial endpoint, most often PFS or tumor response. Key considerations in the design of a Phase II/III trial are the choice of the intermediate endpoint, the target effect size, and the timing of the interim analysis. A biomarker may serve as the intermediate endpoint if there is sufficient evidence that a certain magnitude of effect on the biomarker is likely to translate to a meaningful effect on the definitive Phase III clinical endpoint. A Phase II/
III design is efficient because there is minimal administrative startup time for the Phase III trial and patients accrued during Phase II are combined with the additional Phase III patients for the final analysis.
The flexibility offered by adaptive designs presents some challenges in evaluating their performance. Withouth careful design, adaptive clinical trials are particularly prone to the potential for biased estimates of treatment effects and an increased likelihood of drawing false conclusions (e.g., declaring that a treatment has clinical benefit when it does not) because they are intentionally engineered to repeatedly assess accumulating data in order to alter aspects of the trial. Adaptive designs need thorough and transparent evaluation to ensure that their properties are well understood. These evaluations may require extensive computer simulations conducted under a variety of conditions—both conditions that are consistent with the assumptions made in developing the design and alternative conditions that are still plausible but may differ from the design assumptions (FDA, 2010).
Some adaptive trial design methods are controversial. For example, outcome adaptive randomization is the process of dynamically adjusting randomization ratios in favor of treatment arms that appear more promising as data accumulate during the course of a trial. Some have questioned whether these designs are ethical because the randomization may become substantially imbalanced by the end of the trial (Hey and Kimmelman, 2015). While a larger proportion of patients will ultimately be assigned to the better treatment arm (Berry, 2015), a larger total number of patients might be accrued, which would lead to a larger absolute number of patients being assigned to the inferior arm (Berry, 2011; Korn and Freidlin, 2011a,b; Yuan and Yin, 2011). Other risks include biased treatment effect estimates if the characteristics of patients accrued over time drift toward those associated with more favorable prognosis (Rosenberger and Lachin, 1993). The impact of outcome adaptive randomization on analyses of secondary endpoints and associations between patient covariates or biomarkers and outcome is also uncertain. This situation emphasizes the complexities in assessment of adaptive clinical trial designs and the need to evaluate them carefully in order to understand their performance under a range of possible conditions.
Prognostic and Predictive (Treatment-Selection) Biomarker-Based Tests
Prognostic and predictive biomarker tests may aid in making decisions about therapy (IOM, 2010). These tests are also referred to as treatment-selection, treatment-guiding, or treatment-modifier tests and advanced diagnostic laboratory tests. They can be based on a single biomarker or may evaluate a multi-biomarker signature (e.g., omics-based tests, which typically combine multiple biomarker values from omics assays according to
some algorithm or mathematical model) (IOM, 2012; McShane and Polley, 2013). Prognostic tests aim to predict the clinical course of a disease either in the absence of additional therapy (natural course) or in the context of a standard therapy that all patients are likely to receive. Predictive tests indicate a likely benefit or lack of benefit (or potential harm) from a particular therapy relative to other available therapies (Polley et al., 2013). In some situations (e.g., early-phase clinical trials) the term “predictive” is also used to refer to a test designed to indicate whether the patient is likely to respond to a specific therapeutic agent. That response may or may not translate to a clinical benefit such as improved survival. Prognostic and predictive tests must be carefully evaluated to determine whether their use leads to better treatment decisions and to benefits for patients.
There are a few biomarker tests that have been used or are currently under study for their potential to guide treatment decisions for patients with ovarian cancer. Tests for alterations in BRCA1 and BRCA2 genes are currently of interest as possible predictive tests that could identify those patients most like to benefit from PARP inhibitors (Michels et al., 2014). The OVA1 test is approved by the FDA for use in guiding referral to a gynecologic oncologist.2 It is “a qualitative serum test that combines the results of five immunoassays into a single numerical score for women with an ovarian adnexal mass for which surgery is planned as an aid to further assess the likelihood that malignancy is present when the physician’s independent clinical and radiological evaluation does not indicate malignancy” (FDA, 2011, p. 1). Several chemosensitivity assays have been developed to predict which chemotherapies are likely to elicit a tumor response; however, a review of evidence for the utility of several different chemosensitivity assays concluded that there was insufficient evidence to support a claim that their use would lead to clinical benefits for patients (Kohn, 2014). Few of the studies evaluating these chemosensitivity assays have been randomized clinical trials. An exception is the Tumor Chemosensitivity Assay Ovarian Cancer Study, a randomized biomarker-strategy trial that evaluated whether the use of a chemosensitivity assay that evaluated the impact of 12 chemotherapy agents on cancer cells taken from a patient’s biopsy and was then used to direct chemotherapy choices resulted in better outcome for patients with recurrent platinum-resistant ovarian cancer when compared with a physician’s selection of chemotherapy (Cree et al., 2007). The trial did not find that the use of the assay resulted in significantly improved PFS or overall survival. Other nonrandomized studies have been conducted in an attempt to assess the value of chemosensitivity tests, but it has gener-
________________
2 For more information, see http://vermillion.com/providers/ova-1/ova1 (accessed October 7, 2015).
ally been difficult to interpret their results (Grendys et al., 2014; Korn and Freidlin, 2015).
Compared to the strict regulatory requirements for the evaluation of drugs in clinical trials prior to marketing, there is little regulatory oversight in the United States for laboratory tests. In the past several years, a number of unvalidated laboratory tests were prematurely marketed or used in clinical trials, leading to calls for more rigorous development and validation of laboratory tests and for greater regulatory oversight (ASCO, 2015; FDA, 2014a; Hayes et al., 2013; IOM, 2012; Sawyers and van ’t Veer, 2014).
Advancing Clinical Trials for Ovarian Cancer
The clinical trial designs described are broadly applicable to a variety of cancer types. How readily they can be applied to ovarian cancer depends on a variety of factors that may evolve over time, including the development of new biological insights and new treatments, particularly molecularly targeted therapies. Certain features of ovarian cancer make some of the designs more readily applicable than others at present. For example, in order to reap the efficiency benefits of platform and molecular profiling trials (e.g., NCI-MATCH), it will be necessary to have a broad selection of novel therapies ready for clinical evaluation, and some promising molecular targets of therapies will need to be identified. Platform trials and molecular profiling trials shift the emphasis away from a tumor’s site of origin to its molecular profile, which may be beneficial for relatively rare cancers that may not have a large patient population or resources to warrant its own clinical trial, although advances in the development of novel anticancer agents and therapeutic targets for ovarian cancer have been slow relative to some other cancers, thereby limiting opportunities for the efficient evaluation of new therapies in platform and profiling trials.
There are some features of ovarian cancer that may facilitate novel trial designs, such as the relative accessibility of tumor tissue for molecular analysis for molecularly driven trials and the possibility of carrying out perform WOO trials to directly observe the molecular changes induced by treatment. Such evaluations could help inform the design of trials aimed at preventing or overcoming therapeutic resistance, perhaps by use of multiple therapeutic agents, either in combination or in sequence. A major challenge that is likely to persist is the long clinical trajectory for many women with ovarian cancer. This long trajectory necessitates an extended follow-up in order to observe survival endpoints and makes the interpretation of clinical endpoints particularly complex in those settings where multiple treatments may have been received over a period of several years.
Finally, the improved stratification of women with ovarian cancer may help account for the varying clinical trial outcomes and may ultimately lead
to more clinically meaningful data. For instance, differences in outcomes based on the presence or absence of residual disease after surgery are well documented. Stratification based on whether or not maximum cytoreduction was achieved may be beneficial in accurately assessing clinical trial outcomes. Also, trial designs should account for the various subtypes of ovarian cancer. When looking at individual subtypes, the rarity of cases leads to individual epidemiologic and treatment studies having limited power to draw accurate conclusions. Therefore, the use of consortia and the leveraging of existing data in pooled studies will be important for all studies of ovarian cancer. Such consortia will require infrastructure to support cross-cutting activities such as data harmonization and the development of new statistical methods. New quantitative analysis approaches, including novel statistical, bioinformatic, and computational methods applied to biological, epidemiologic, and clinical data, will be essential for gaining insights into disease etiology, developing biologically driven treatments, and designing efficient clinical trials. Rapidly evolving molecular assay technologies will make it necessary to develop new analysis methods to process and integrate molecular data representing DNA, RNA, and protein profiles of tumors as well as other characterizations, such as tumor epigenetic profiles or host germline genomic profiles. These analyses may provide new insights to help researchers better understand tumor development, growth, metastatsis, and response to therapy. Clinical trials that leverage these new biological insights to optimize treatment selection or adapt to data as they accumulate during the course of the trial may also require novel statistical methods for their design and evaluation.
The committee offers the following findings and conclusions:
- PDS is an effective first-line treatment method, yet maximum cytoreduction may be unachievable in women with Stage III or IV ovarian cancer unless NACT is employed. An evidence-based decision-making strategy or tool to determine which of these options is most appropriate for a patient is needed.
- The majority of women with ovarian cancer respond well to initial treatment with platinum-based (e.g., carboplatin) chemotherapy, although a large fraction of tumors become platinum resistant within 6 months of completing treatment. Given the heterogeneity in both platinum-sensitive and platinum-resistant patient populations at initial diagnosis and recurrence, there is a need for better tools to predict near- and long-term response to treatments for both newly diagnosed and recurrent disease.
- The definition of disease recurrence is generally accepted to be the detection of macroscopic disease within 6 months of complete cytoreduction from initial treatment. There is a debate regarding how the timing of secondary cytoreductive surgery relates to its efficacy in improving patient outcome. As secondary cytoreductive surgery is normally performed at the time of the diagnosis of recurrence, the definition of ovarian recurrence needs to be further refined.
- Given the high rate of drug resistance after the initial response to front-line therapy, additional treatment options are necessary in both the up-front and recurrent settings in order to improve patient outcomes.
- A better understanding is needed of the mechanisms of the development of recurrent disease.
- More well-validated biomarker tests with demonstrated ability to aid in treatment decisions that lead to better outcomes for women with ovarian cancer are needed.
- There is considerable variability in the quality of care provided to women with ovarian cancer nationwide. This variability is influenced by several factors, including the type of hospitals where patients are treated, whether they are treated by a gynecologic oncologist, and whether their care adheres to the proper guidelines. The delivery of more consistent and higher-quality care could have a significant impact on improving outcomes for ovarian cancer patients.
- Significant predictors of nonadherence to the standard of care include advanced age at diagnosis, the presence of one or more treatment-limiting comorbidities, non-white race, and lower socioeconomic status.
- Most ovarian cancers occur in older women, yet little research focuses on how to improve care for older women, especially those with comorbidities, and clinical trials tend to enroll younger patients.
- Given that the effectiveness of novel therapeutics may vary within and among subtypes, it will be necessary to evaluate a variety of approaches, including new combinations of existing drugs, new drug formulations, targeted biologics, protein inhibitors, TP53-directed therapies, anti-angiogenics, immunotherapies, and nonpharmacologic interventions.
- Stratification based on the presence or absence of residual disease after surgery and by tumor subtype may be used to control for the different outcomes in clinical trials and may lead to more clinically meaningful data.
- High-quality preclinical and translational research, improved clinical trial infrastructure, and incentives to facilitate more rapid
accrual of patients to clinical trials are all essential to the rapid delivery of effective new treatments for women with ovarian cancer.
ACOG (American College of Obstetricians and Gynecologists). 2002. The role of the generalist obstetrician-gynecologist in the early detection of ovarian cancer. Gynecologic Oncology 87(3):237-239.
Adaptimmune. 2012. CT antigen TCR-redirected T cells for ovarian cancer. Bethesda, MD: National Library of Medicine. https://clinicaltrials.gov/ct2/show/NCT01567891 (accessed October 21, 2015).
Agarwal, R., and S. B. Kaye. 2003. Ovarian cancer: Strategies for overcoming resistance to chemotherapy. Nature Reviews: Cancer 3(7):502-516.
Aghajanian, C., S. V. Blank, B. A. Goff, P. L. Judson, M. G. Teneriello, A. Husain, M. A. Sovak, J. Yi, and L. R. Nycum. 2012. Oceans: A randomized, double-blind, placebo-controlled phase III trial of chemotherapy with or without bevacizumab in patients with platinum-sensitive recurrent epithelial ovarian, primary peritoneal, or fallopian tube cancer. Journal of Clinical Oncology 30(17):2039-2045.
Al Rawahi, T., A. D. Lopes, R. E. Bristow, A. Bryant, A. Elattar, S. Chattopadhyay, and K. Galaal. 2013. Surgical cytoreduction for recurrent epithelial ovarian cancer. Cochrane Database of Systematic Reviews 2:CD008765.
Alberts, D. S., P. Y. Liu, E. V. Hannigan, R. O—Toole, S. D. Williams, J. A. Young, E. W. Franklin, D. L. Clarke-Pearson, V. K. Malviya, and B. DuBeshter. 1996. Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. New England Journal of Medicine 335(26):1950-1955.
Alberts, D. S., E. V. Hannigan, P. Y. Liu, C. Jiang, S. Wilczynski, L. Copeland, and M. Markman. 2006. Randomized trial of adjuvant intraperitoneal alpha-interferon in stage III ovarian cancer patients who have no evidence of disease after primary surgery and chemotherapy: An intergroup study. Gynecologic Oncology 100(1):133-138.
Alpen, B., A. O. Gure, M. J. Scanlan, L. J. Old, and Y. T. Chen. 2002. A new member of the NY-ESO-1 gene family is ubiquitously expressed in somatic tissues and evolutionarily conserved. Gene 297(1-2):141-149.
Amé, J. C., C. Spenlehauer, and G. de Murcia. 2004. The PARP superfamily. Bioessays 26(8):882-893.
Anglesio, M. S., S. Kommoss, M. C. Tolcher, B. Clarke, L. Galletta, et al. 2013. Molecular characterization of mucinous ovarian tumours supports a stratified treatment approach with HER2 targeting in 19% of carcinomas. Journal of Pathology 229(1):111-120.
Appelboom, G., E. Camacho, M. E. Abraham, S. S. Bruce, E. L. P. Dumont, B. E. Zacharia, R. D—Amico, J. Slomian, J. Y. Reginster, O. Bruyere, and E. S. Connolly, Jr. 2014. Smart wearable body sensors for patient self-assessment and monitoring. Archives of Public Health 72(1):28.
Aprea, A. B. 2014. P53 suppressor activation in recurrent high grade serous ovarian cancer, a phase Ib/II study of systemic carboplatin combination chemotherapy with or without APR-246. Bethesda, MD: National Library of Medicine. https://clinicaltrials.gov/ct2/show/NCT02098343 (accessed October 21, 2015).
Armstrong, D. K., B. Bundy, L. Wenzel, H. Q. Huang, R. Baergen, S. Lele, L. J. Copeland, J. L. Walker, and R. A. Burger, for the Gynecologic Oncology Group. 2006. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. New England Journal of Medicine 354(1):34-43.
ASCO (American Society of Clinical Oncology). 2015. ASCO supports FDA’s proposed regulation of laboratory developed tests. http://www.asco.org/advocacy/asco-supports-fda%E2%80%99s-proposed-regulation-laboratory-developed-tests (accessed October 1, 2015).
Ashworth, A. 2008. A synthetic lethal therapeutic approach: Poly (ADP) ribose polymerase inhibitors for the treatment of cancers deficient in DNA double-strand break repair. Journal of Clinical Oncology 26(22):3785-3790.
Audeh, M. W., J. Carmichael, R. T. Penson, M. Friedlander, B. Powell, K. M. Bell-McGuinn, C. Scott, J. N. Weitzel, A. Oaknin, N. Loman, K. Lu, R. K. Schmutzler, U. Matulonis, M. Wickens, and A. Tutt. 2010. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: A proof-of-concept trial. Lancet 376(9737):245-251.
Austin, M., Y. Martin, Y. Kim, E. M. Funkhouser, E. E. Partridge, and M. Pisu. 2013. Disparities in the use of gynecologic oncologists for women with ovarian cancer in the United States. Health Services Research 48(3):1135-1153.
Barker, A. D., C. C. Sigman, G. J. Kelloff, N. M. Hylton, D. A. Berry, and L. J. Esserman. 2009. I-SPY 2: An adaptive breast cancer trial design in the setting of neoadjuvant chemotherapy. Clinical Pharmacology and Therapeutics 86(1):97-100.
Baselga, J. 2006. Targeting tyrosine kinases in cancer: The second wave. Science 312(5777): 1175-1178.
Baum, R. P., A. A. Noujaim, A. Nanci, V. Moebus, A. Hertel, A. Niesen, B. Donnerstag, T. Sykes, G. Boniface, and G. Hor. 1993. Clinical course of ovarian cancer patients under repeated stimulation of HAMA using MAb OC125 and B43.13. Hybridoma 12(5):583-589.
Berek, J. S., B. C. Schultes, and C. F. Nicodemus. 2003. Biologic and immunologic therapies for ovarian cancer. Journal of Clinical Oncology 21(10 Suppl):168-174.
Berek, J. S., P. T. Taylor, A. Gordon, M. J. Cunningham, N. Finkler, J. Orr, Jr., S. Rivkin, B. C. Schultes, T. L. Whiteside, and C. F. Nicodemus. 2004. Randomized, placebo-controlled study of oregovomab for consolidation of clinical remission in patients with advanced ovarian cancer. Journal of Clinical Oncology 22(17):3507-3516.
Berns, E. M., and D. D. Bowtell. 2012. The changing view of high-grade serous ovarian cancer. Cancer Research 72(11):2701-2704.
Berry, D. A. 2006. Bayesian clinical trials. Nature Reviews: Drug Discovery 5(1):27-36.
Berry, D. 2011. Adaptive clinical trials: The promise and the caution. Journal of Clinical Oncology 29(6):606-609.
Berry, D. 2015. Commentary on Hey and Kimmelman. Clinical Trials (London, England) 12(2):107-109.
Bertelsen, K., A. Jakobsen, J. E. Andersen, S. Ahrons, P. H. Pedersen, H. Kiaer, E. Arffmann, P. Bichel, E. Boestofte, I. Stroyer, et al. 1987. A randomized study of cyclophosphamide and cis-platinum with or without doxorubicin in advanced ovarian carcinoma. Gynecologic Oncology 28(2):161-169.
Bhosale, P., S. Peungjesada, W. Wei, C. F. Levenback, K. Schmeler, E. Rohren, H. A. Macapinlac, and R. B. Iyer. 2010. Clinical utility of positron emission tomography/computed tomography in the evaluation of suspected recurrent ovarian cancer in the setting of normal CA-125 levels. International Journal of Gynecologic Cancer 20(6):936-944.
Biagi, J. J., A. M. Oza, H. I. Chalchal, R. Grimshaw, S. L. Ellard, U. Lee, H. Hirte, J. Sederias, S. P. Ivy, and E. A. Eisenhauer. 2011. A phase II study of sunitinib in patients with recurrent epithelial ovarian and primary peritoneal carcinoma: An NCIC clinical trials group study. Annals of Oncology 22(2):335-340.
Blikkendaal, M. D., E. M. Schepers, E. W. van Zwet, A. R. H. Twijnstra, and F. W. Jansen. 2015. Hysterectomy in very obese and morbidly obese patients: A systematic review with cumulative analysis of comparative studies. Archives of Gynecology and Obstetrics 292(4):723-738.
Bookman, M. A., M. F. Brady, W. P. McGuire, P. G. Harper, D. S. Alberts, M. Friedlander, N. Colombo, J. M. Fowler, P. A. Argenta, K. De Geest, D. G. Mutch, R. A. Burger, A. M. Swart, E. L. Trimble, C. Accario-Winslow, and L. M. Roth. 2009. Evaluation of new platinum-based treatment regimens in advanced-stage ovarian cancer: A phase III trial of the gynecologic cancer intergroup. Journal of Clinical Oncology 27(9):1419-1425.
Boon, T., and P. van der Bruggen. 1996. Human tumor antigens recognized by T lymphocytes. Journal of Experimental Medicine 183(3):725-729.
Boyd, L. R., A. P. Novetsky, and J. P. Curtin. 2011. Ovarian cancer care for the underserved: Are surgical patterns of care different in a public hospital setting? Cancer 117(4): 777-783.
Brahmer, J. R., S. S. Tykodi, L. Q. Chow, W. J. Hwu, S. L. Topalian, et al. 2012. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. New England Journal of Medicine 366(26):2455-2465.
Bristow, R. E., R. S. Tomacruz, D. K. Armstrong, E. L. Trimble, and F. J. Montz. 2002. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: A meta-analysis. Journal of Clinical Oncology 20(5):1248-1259.
Bristow, R. E., J. Chang, A. Ziogas, and H. Anton-Culver. 2013a. Adherence to treatment guidelines for ovarian cancer as a measure of quality care. Obstetrics and Gynecology 121(6):1226-1234.
Bristow, R. E., M. A. Powell, N. Al-Hammadi, L. Chen, J. P. Miller, P. Y. Roland, D. G. Mutch, and W. A. Cliby. 2013b. Disparities in ovarian cancer care quality and survival according to race and socioeconomic status. Journal of the National Cancer Institute 105(11):823-832.
Bristow, R. E., J. Chang, A. Ziogas, L. M. Randall, and H. Anton-Culver. 2014. High-volume ovarian cancer care: Survival impact and disparities in access for advanced-stage disease. Gynecologic Oncology 132(2):403-410.
Bryant, H. E., N. Schultz, H. D. Thomas, K. M. Parker, D. Flower, E. Lopez, S. Kyle, M. Meuth, N. J. Curtin, and T. Helleday. 2005. Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 434(7035):913-917.
Burger, R. A., M. F. Brady, M. A. Bookman, G. F. Fleming, B. J. Monk, H. Huang, R. S. Mannel, H. D. Homesley, J. Fowler, B. E. Greer, M. Boente, M. J. Birrer, and S. X. Liang, for the Gynecologic Oncology Group. 2011. Incorporation of bevacizumab in the primary treatment of ovarian cancer. New England Journal of Medicine 365(26):2473-2483.
Calle, E. E., C. Rodriguez, K. Walker-Thurmond, and M. J. Thun. 2003. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. New England Journal of Medicine 348(17):1625-1638.
Campos, S. M., R. T. Penson, U. Matulonis, N. S. Horowitz, C. Whalen, L. Pereira, K. Tyburski, M. Roche, J. Szymonifka, and S. Berlin. 2013. A phase II trial of sunitinib malate in recurrent and refractory ovarian, fallopian tube and peritoneal carcinoma. Gynecologic Oncology 128(2):215-220.
Cancer Genome Atlas Research Network. 2011. Integrated genomic analyses of ovarian carcinoma. Nature 474(7353):609-615.
Carrillo, A. M., M. Hicks, D. Khabele, and C. M. Eischen. 2015. Pharmacologically increasing Mdm2 inhibits DNA repair and cooperates with genotoxic agents to kill p53-inactivated ovarian cancer cells. Molecular Cancer Research 13(8):1197-1205.
Castle, J. C., S. Kreiter, J. Diekmann, M. Lower, N. van de Roemer, J. de Graaf, A. Selmi, M. Diken, S. Boegel, C. Paret, M. Koslowski, A. N. Kuhn, C. M. Britten, C. Huber, O. Tureci, and U. Sahin. 2012. Exploiting the mutanome for tumor vaccination. Cancer Research 72(5):1081-1091.
CBO (Congressional Budget Office). 2007. Research on the comparative effectiveness of medical treatments. https://www.cbo.gov/sites/default/files/110th-congress-2007-2008/reports/12-18-comparativeeffectiveness.pdf (accessed October 1, 2015).
Cereda, E., M. Turrini, D. Ciapanna, L. Marbello, A. Pietrobelli, and E. Corradi. 2007. Assessing energy expenditure in cancer patients: A pilot validation of a new wearable device. Journal of Parenteral and Enteral Nutrition 31(6):502-507.
Chan, J. K., E. G. Munro, M. K. Cheung, A. Husain, N. N. Teng, J. S. Berek, and K. Osann. 2007a. Association of lymphadenectomy and survival in stage I ovarian cancer patients. Obstetrics and Gynecology 109(1):12-19.
Chan, J. K., D. S. Kapp, J. Y. Shin, A. Husain, N. N. Teng, J. S. Berek, K. Osann, G. S. Leiserowitz, R. D. Cress, and C. O—Malley. 2007b. Influence of the gynecologic oncologist on the survival of ovarian cancer patients. Obstetrics and Gynecology 109(6):1342-1350.
Chapman, P. B., A. Hauschild, C. Robert, J. B. Haanen, P. Ascierto, et al. 2011. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. New England Journal of Medicine 364(26):2507-2516.
Chase, D. M., S. Fedewa, T. S. Chou, A. Chen, E. Ward, and W. R. Brewster. 2012. Disparities in the allocation of treatment in advanced ovarian cancer: Are there certain patient characteristics associated with nonstandard therapy? Obstetrics and Gynecology 119(1):68-77.
Cheever, M. A., J. P. Allison, A. S. Ferris, O. J. Finn, B. M. Hastings, T. T. Hecht, I. Mellman, S. A. Prindiville, J. L. Viner, L. M. Weiner, and L. M. Matrisian. 2009. The prioritization of cancer antigens: A National Cancer Institute pilot project for the acceleration of translational research. Clinical Cancer Research 15(17):5323-5337.
Chen, Y. T., M. J. Scanlan, U. Sahin, O. Tureci, A. O. Gure, S. Tsang, B. Williamson, E. Stockert, M. Pfreundschuh, and L. J. Old. 1997. A testicular antigen aberrantly expressed in human cancers detected by autologous antibody screening. Proceedings of the National Academy Sciences of the United States of America 94(5):1914-1918.
Chi, D. S., F. Musa, F. Dao, O. Zivanovic, Y. Sonoda, M. M. Leitao, D. A. Levine, G. J. Gardner, N. R. Abu-Rustum, and R. R. Barakat. 2012. An analysis of patients with bulky advanced stage ovarian, tubal, and peritoneal carcinoma treated with primary debulking surgery (PDS) during an identical time period as the randomized EORTC-NCIC trial of PDS vs neoadjuvant chemotherapy (NACT). Gynecologic Oncology 124(1):10-14.
Clarke, T., K. Galaal, A. Bryant, and R. Naik. 2014. Evaluation of follow-up strategies for patients with epithelial ovarian cancer following completion of primary treatment. Cochrane Database System Review 9:CD006119.
Cliby, W. A., M. A. Powell, N. Al-Hammadi, L. Chen, J. Philip Miller, P. Y. Roland, D. G. Mutch, and R. E. Bristow. 2015. Ovarian cancer in the United States: Contemporary patterns of care associated with improved survival. Gynecologic Oncology 136(1): 11-17.
Coffey, C. S., and J. A. Kairalla. 2008. Adaptive clinical trials: Progress and challenges. Drugs in Research and Development 9(4):229-242.
Coleman, R. L., L. R. Duska, P. T. Ramirez, J. V. Heymach, A. A. Kamat, S. C. Modesitt, K. M. Schmeler, R. B. Iyer, M. E. Garcia, D. L. Miller, E. F. Jackson, C. S. Ng, V. Kundra, R. Jaffe, and A. K. Sood. 2011. Phase 1-2 study of docetaxel plus aflibercept in patients with recurrent ovarian, primary peritoneal, or fallopian tube cancer. Lancet Oncology 12(12):1109-1117.
Coleman, R. L., B. J. Monk, A. K. Sood, and T. J. Herzog. 2013. Latest research and treatment of advanced-stage epithelial ovarian cancer. Nature Reviews: Clinical Oncology 10(4):211-224.
Collinson, F., M. Hutchinson, R. A. Craven, D. A. Cairns, A. Zougman, T. C. Wind, N. Gahir, M. P. Messenger, S. Jackson, D. Thompson, C. Adusei, J. A. Ledermann, G. Hall, G. C. Jayson, P. J. Selby, and R. E. Banks. 2013. Predicting response to bevacizumab in ovarian cancer: A panel of potential biomarkers informing treatment selection. Clinical Cancer Research 19(18):5227-5239.
Cong, L., F. A. Ran, D. Cox, S. Lin, R. Barretto, N. Habib, P. D. Hsu, X. Wu, W. Jiang, L. A. Marraffini, and F. Zhang. 2013. Multiplex genome engineering using CRISPR/Cas systems. Science 339(6121):819-823.
Coward, J. I., K. Middleton, and F. Murphy. 2015. New perspectives on targeted therapy in ovarian cancer. International Journal of Women’s Health 7:189-203.
Cree, I. A., C. M. Kurbacher, A. Lamont, A. C. Hindley, S. Love, and TCA Ovarian Cancer Trial Group. 2007. A prospective randomized controlled trial of tumour chemosensitivity assay directed chemotherapy versus physician’s choice in patients with recurrent platinum-resistant ovarian cancer. Anti-Cancer Drugs 18(9):1093-1101.
Dahlberg, S. E., G. I. Shapiro, J. W. Clark, and B. E. Johnson. 2014. Evaluation of statistical designs in phase I expansion cohorts: The Dana-Farber/Harvard Cancer Center experience. Journal of the National Cancer Institute 106(7).
Dai-yuan, M., T. Bang-xian, L. Xian-fu, Z. Ye-qin, and C. Hong-Wei. 2013. A meta-analysis: Neoadjuvant chemotherapy versus primary surgery in ovarian carcinoma FIGO stage III and IV. World Journal of Surgical Oncology 11(267).
Dantzer, F., G. de La Rubia, J. Menissier-De Murcia, Z. Hostomsky, G. de Murcia, and V. Schreiber. 2000. Base excision repair is impaired in mammalian cells lacking poly(ADP-ribose) polymerase-1. Biochemistry 39(25):7559-7569.
Daudi, S., K. H. Eng, P. Mhawech-Fauceglia, C. Morrison, A. Miliotto, A. Beck, J. Matsuzaki, T. Tsuji, A. Groman, S. Gnjatic, G. Spagnoli, S. Lele, and K. Odunsi. 2014. Expression and immune responses to MAGE antigens predict survival in epithelial ovarian cancer. PLoS ONE 9(8):e104099.
Davidson, B. A., and A. A. Secord. 2014. Profile of pazopanib and its potential in the treatment of epithelial ovarian cancer. International Journal of Women’s Health 6:289-300.
Davis, A., A. V. Tinker, and M. Friedlander. 2014. “Platinum resistant” ovarian cancer: What is it, who to treat and how to measure benefit? Gynecologic Oncology 133(3):624-631.
Disis, M. L., M. R. Patel, S. Pant, J. R. Infante, A. C. Lockhart, K. Kelly, J. T. Beck, M. S. Gordon, G. J. Weiss, and S. Ejadi. 2015. Avelumab (MSB0010718C), an anti-PD-L1 antibody, in patients with previously treated, recurrent or refractory ovarian cancer: A phase Ib, open-label expansion trial. Journal of Clinical Oncology 33(Suppl):abstract 5509.
Dobkin, B. H. and A. Dorsch. 2011. The promise of mHealth: Daily activity monitoring and outcome assessments by wearable sensors. Neurorehabilitation and Neural Repair 25(9): 788-798.
Donahoe, L., S. Bennett, W. Temple, A. Hilchie-Pye, K. Dabbs, E. Macintosh, and G. Porter. 2012. Completeness of dictated operative reports in breast cancer—The case for synoptic reporting. Journal of Surgical Oncology 106(1):79-83.
Du, X. L., C. C. Sun, M. R. Milam, D. C. Bodurka, and S. Fang. 2008. Ethnic differences in socioeconomic status, diagnosis, treatment, and survival among older women with epithelial ovarian cancer. International Journal of Gynecological Cancer 18(4):660-669.
du Bois, A., H. J. Luck, W. Meier, H. P. Adams, V. Mobus, S. Costa, T. Bauknecht, B. Richter, M. Warm, W. Schroder, S. Olbricht, U. Nitz, C. Jackisch, G. Emons, U. Wagner, W. Kuhn, J. Pfisterer, and Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group. 2003. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. Journal of the National Cancer Institute 95(17):1320-1329.
du Bois, A., A. Reuss, E. Pujade-Lauraine, P. Harter, I. Ray-Coquard, and J. Pfisterer. 2009. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: A combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: By the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO-OVAR) and the Groupe d—Investigateurs Nationaux Pour les Etudes des Cancers de l—Ovaire (GINECO). Cancer 115(6):1234-1244.
du Bois, A., C. Marth, J. Pfisterer, P. Harter, F. Hilpert, A. G. Zeimet, and J. Sehouli. 2012. Neoadjuvant chemotherapy cannot be regarded as adequate routine therapy strategy of advanced ovarian cancer. International Journal of Gynecological Cancer 22(2):182-185.
du Bois, A., A. Floquet, J. W. Kim, J. Rau, J. M. del Campo, et al. 2014. Incorporation of pazopanib in maintenance therapy of ovarian cancer. Journal of Clinical Oncology 32(30):3374-3382.
Duan, F., J. Duitama, S. Al Seesi, C. M. Ayres, S. A. Corcelli, A. P. Pawashe, T. Blanchard, D. McMahon, J. Sidney, A. Sette, B. M. Baker, I. I. Mandoiu, and P. K. Srivastava. 2014. Genomic and bioinformatic profiling of mutational neoepitopes reveals new rules to predict anticancer immunogenicity. Journal of Experimental Medicine 211(11):2231-2248.
Dudley, M. E., J. R. Wunderlich, P. F. Robbins, J. C. Yang, P. Hwu, D. J. Schwartzentruber, S. L. Topalian, R. Sherry, N. P. Restifo, A. M. Hubicki, M. R. Robinson, M. Raffeld, P. Duray, C. A. Seipp, L. Rogers-Freezer, K. E. Morton, S. A. Mavroukakis, D. E. White, and S. A. Rosenberg. 2002. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298(5594):850-854.
Dudley, M. E., J. R. Wunderlich, J. C. Yang, R. M. Sherry, S. L. Topalian, et al. 2005. Adoptive cell transfer therapy following non-myeloablative but lymphodepleting chemotherapy for the treatment of patients with refractory metastatic melanoma. Journal of Clinical Oncology 23(10):2346-2357.
Dunn, G. P., L. J. Old, and R. D. Schreiber. 2004. The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21(2):137-148.
Ehlen, T. G., P. J. Hoskins, D. Miller, T. L. Whiteside, C. F. Nicodemus, B. C. Schultes, and K. D. Swenerton. 2005. A pilot phase 2 study of oregovomab murine monoclonal antibody to CA125 as an immunotherapeutic agent for recurrent ovarian cancer. International Journal of Gynecological Cancer 15(6):1023-1034.
Eisenkop, S. M., N. M. Spirtos, T. W. Montag, R. H. Nalick, and H. Wang. 1992. The impact of subspecialty training on the management of advanced ovarian cancer. Gynecologic Oncology 47(2):203-209.
Eisenkop, S. M., R. L. Friedman, and H. J. Wang. 1998. Complete cytoreductive surgery is feasible and maximizes survival in patients with advanced epithelial ovarian cancer: A prospective study. Gynecologic Oncology 69(2):103-108.
Elattar, A., A. Bryant, B. A. Winter-Roach, M. Hatem, and R. Naik. 2011. Optimal primary surgical treatment for advanced epithelial ovarian cancer. Cochrane Database of Systematic Reviews 8:CD007565.
EMA (European Medicines Agency). 2014. Lynparza. http://www.ema.europa.eu/docs/en_GB/document_library/Summary_of_opinion_-_Initial_authorisation/human/003726/WC500176336.pdf (accessed October 21, 2015).
Erickson, B. K., J. Y. Martin, M. M. Shah, J. M. Straughn, Jr., and C. A. Leath, 3rd. 2014. Reasons for failure to deliver National Comprehensive Cancer Network (NCCN)-adherent care in the treatment of epithelial ovarian cancer at an NCCN cancer center. Gynecologic Oncology 133(2):142-146.
Eskander, R. N., and K. S. Tewari. 2014. Incorporation of anti-angiogenesis therapy in the management of advanced ovarian carcinoma—Mechanistics, review of phase III randomized clinical trials, and regulatory implications. Gynecologic Oncology 132(2):496-505.
Fairfield, K. M., F. L. Lucas, C. C. Earle, I. Small, E. L. Trimble, and J. L. Warren. 2010. Regional variation in cancer-directed surgery and mortality among women with epithelial ovarian cancer in the Medicare population. Cancer 116(20):4840-4848.
Farmer, H., N. McCabe, C. J. Lord, A. N. Tutt, D. A. Johnson, T. B. Richardson, M. Santarosa, K. J. Dillon, I. Hickson, C. Knights, N. M. Martin, S. P. Jackson, G. C. Smith, and A. Ashworth. 2005. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434(7035):917-921.
FDA (U.S. Food and Drug Administration). 2010. Guidance for industry: Adaptive design clinical trials for drugs and biologics. http://www.fda.gov/downloads/Drugs/Guidances/ucm201790.pdf (accessed October 1, 2015).
FDA. 2011. 510(k) substantial equivalence determination decision summary. http://www.accessdata.fda.gov/cdrh_docs/reviews/K081754.pdf (accessed October 1, 2015).
FDA. 2014a. Framework for regulatory oversight of 8 laboratory developed tests (LDTs). http://www.fda.gov/downloads/medicaldevices/deviceregulationandguidance/guidance-documents/ucm416685.pdf (accessed October 1, 2015).
FDA. 2014b. Summary review for regulatory action. http://www.accessdata.fda.gov/drug-satfda_docs/nda/2014/206162Orig1s000SumR.pdf (accessed August 3, 2015).
FDA. 2014c. Meeting of the Onvologic Drugs Advisory Committee June 25, 2014 transcript. http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/OncologicDrugsAdvisoryCommittee/UCM410457.pdf (accessed December 21, 2015).
Febbraro, T., K. Robison, J. S. Wilbur, J. Laprise, A. Bregar, V. Lopes, R. Legare, and A. Stuckey. 2015. Adherence patterns to National Comprehensive Cancer Network (NCCN) guidelines for referral to cancer genetic professionals. Gynecologic Oncology 138(1):109-114.
Feng, F. Y., J. S. de Bono, M. A. Rubin, and K. E. Knudsen. 2015. Chromatin to clinic: The molecular rationale for PARP1 inhibitor function. Molecular Cell 58(6):925-934.
Fleming, N. D., I. Cass, C. S. Walsh, B. Y. Karlan, and A. J. Li. 2011. CA125 surveillance increases optimal resectability at secondary cytoreductive surgery for recurrent epithelial ovarian cancer. Gynecologic Oncology 121(2):249-252.
Fong, P. C., D. S. Boss, T. A. Yap, A. Tutt, P. Wu, M. Mergui-Roelvink, P. Mortimer, H. Swaisland, A. Lau, M. J. O—Connor, A. Ashworth, J. Carmichael, S. B. Kaye, J. H. Schellens, and J. S. de Bono. 2009. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. New England Journal of Medicine 361(2):123-134.
Fong, P. C., T. A. Yap, D. S. Boss, C. P. Carden, M. Mergui-Roelvink, C. Gourley, J. De Greve, J. Lubinski, S. Shanley, C. Messiou, R. A—Hern, A. Tutt, A. Ashworth, J. Stone, J. Carmichael, J. H. Schellens, J. S. de Bono, and S. B. Kaye. 2010. Poly(ADP)-ribose polymerase inhibition: Frequent durable responses in BRCA carrier ovarian cancer correlating with platinum-free interval. Journal of Clinical Oncology 28(15):2512-2519.
Fotopoulou, C., R. Richter, I. E. Braicu, S. C. Schmidt, P. Neuhaus, W. Lichtenegger, and J. Sehouli. 2011. Clinical outcome of tertiary surgical cytoreduction in patients with recurrent epithelial ovarian cancer. Annals of Surgical Oncology 18(1):49-57.
Fotopoulou, C., R. Zang, M. Gultekin, D. Cibula, A. Ayhan, D. Liu, R. Richter, I. Braicu, S. Mahner, P. Harter, F. Trillsch, S. Kumar, M. Peiretti, S. C. Dowdy, A. Maggioni, C. Trope, and J. Sehouli. 2013. Value of tertiary cytoreductive surgery in epithelial ovarian cancer: An international multicenter evaluation. Annals of Surgical Oncology 20(4):1348-1354.
Freidlin, B., and E. L. Korn. 2014. Biomarker enrichment strategies: Matching trial design to biomarker credentials. Nature Reviews: Clinical Oncology 11(2):81-90.
Freidlin, B., E. L. Korn, R. Gray, and A. Martin. 2008. Multi-arm clinical trials of new agents: Some design considerations. Clinical Cancer Research 14(14):4368-4371.
Freidlin, B., L. M. McShane, and E. L. Korn. 2010. Randomized clinical trials with biomarkers: Design issues. Journal of the National Cancer Institute 102(3):152-160.
Friedlander, M., K. C. Hancock, D. Rischin, M. J. Messing, C. A. Stringer, G. M. Matthys, B. Ma, J. P. Hodge, and J. J. Lager. 2010. A phase II, open-label study evaluating pazopanib in patients with recurrent ovarian cancer. Gynecologic Oncology 119(1):32-37.
Friedlander, M., E. Trimble, A. Tinker, D. Alberts, E. Avall-Lundqvist, et al. 2011. Clinical trials in recurrent ovarian cancer. International Journal of Gynecological Cancer 21(4):771-775.
Garnett, M. J., E. J. Edelman, S. J. Heidorn, C. D. Greenman, A. Dastur, et al. 2012. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature 483(7391):570-575.
Gasparri, M. L., P. B. Panici, and A. Papadia. 2015. Primary chemotherapy versus primary surgery for ovarian cancer. Lancet 386(10009):2142-2143.
Gershenson, D. M. 2012. Treatment of ovarian cancer in young women. Clinical Obstetrics and Gynecology 55(1):65-74.
Giede, K. C., K. Kieser, J. Dodge, and B. Rosen. 2005. Who should operate on patients with ovarian cancer? An evidence-based review. Gynecologic Oncology 99(2):447-461.
Girre, V., M. C. Falcou, M. Gisselbrecht, G. Gridel, V. Mosseri, C. Bouleuc, R. Poinsot, L. Vedrine, L. Ollivier, V. Garabige, J. Y. Pierga, V. Dieras, and L. Mignot. 2008. Does a geriatric oncology consultation modify the cancer treatment plan for elderly patients? The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 63(7):724-730.
Glimelius, B., and M. Lahn. 2011. Window-of-opportunity trials to evaluate clinical activity of new molecular entities in oncology. Annals of Oncology 22(8):1717-1725.
Goel, S., A. H. Wong, and R. K. Jain. 2012. Vascular normalization as a therapeutic strategy for malignant and nonmalignant disease. Cold Spring Harbor Perspectives in Medicine 2(3):a006486.
Goff, B. 2015. Measuring ovarian cancer care: Why are we still failing? Gynecologic Oncology 136(1):1-2.
Goff, B. A., L. S. Mandel, C. H. Melancon, and H. G. Muntz. 2004. Frequency of symptoms of ovarian cancer in women presenting to primary care clinics. Journal of the American Medical Association 291(22):2705-2712.
Goff, B. A., B. J. Matthews, E. H. Larson, C. H. Andrilla, M. Wynn, D. M. Lishner, and L. M. Baldwin. 2007. Predictors of comprehensive surgical treatment in patients with ovarian cancer. Cancer 109(10):2031-2042.
Gogoi, R. P., R. Urban, H. Sun, and B. Goff. 2012. Evaluation of Society of Gynecologic Oncologists (SGO) ovarian cancer quality surgical measures. Gynecologic Oncology 126(2):217-219.
Gotlieb, W. H., F. Amant, S. Advani, C. Goswami, H. Hirte, D. Provencher, N. Somani, S. D. Yamada, J. F. Tamby, and I. Vergote. 2012. Intravenous aflibercept for treatment of recurrent symptomatic malignant ascites in patients with advanced ovarian cancer: A phase 2, randomised, double-blind, placebo-controlled study. Lancet Oncology 13(2):154-162.
Gourley, C., A. McCavigan, T. Perren, J. Paul, C. O. Michie, M. Churchman, A. Williams, W. G. McCluggage, M. Parmar, R. S. Kaplan, L. A. Hill, I. A. Halfpenny, E. J. O—Brien, O. Raji, S. Deharo, T. Davison, P. Johnston, K. E. Keating, D. P. Harkin, R. D. Kennedy. 2015. Molecular subgroup of high-grade serous ovarian cancer (HGSOC) as a predictor of outcome following bevacizumab. Journal of Clinical Oncology 32(5s):abstr 5502.
Green, S. 2005. Factorial designs with time to event endpoints. In Handbook of statistics in clinical oncology, 2nd ed., edited by J. Crowley and D. Ankerst. Boca Raton, FL: Chapman and Hall/CRC. Pp. 181-189.
Grendys, E. C., Jr., J. V. Fiorica, J. W. Orr, Jr., R. Holloway, D. Wang, C. Tian, J. K. Chan, and T. J. Herzog. 2014. Overview of a chemoresponse assay in ovarian cancer. Clinical and Translational Oncology 16(9):761-769.
Griffiths, C. T. 1975. Surgical resection of tumor bulk in the primary treatment of ovarian carcinoma. National Cancer Institute Monographs 42:101-104.
Griggs, J. J., P. B. Mangu, H. Anderson, E. P. Balaban, J. J. Dignam, W. M. Hryniuk, V. A. Morrison, T. M. Pini, C. D. Runowicz, G. L. Rosner, M. Shayne, A. Sparreboom, L. E. Sucheston, and G. H. Lyman. 2012. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. Journal of Clinical Oncology 30(13):1553-1561.
Grunwald, V., and M. Hidalgo. 2003. Developing inhibitors of the epidermal growth factor receptor for cancer treatment. Journal of the National Cancer Institute 95(12):851-867.
Gubbels, J. A., J. Belisle, M. Onda, C. Rancourt, M. Migneault, M. Ho, T. K. Bera, J. Connor, B. K. Sathyanarayana, B. Lee, I. Pastan, and M. S. Patankar. 2006. Mesothelin-MUC16 binding is a high affinity, N-glycan dependent interaction that facilitates peritoneal metastasis of ovarian tumors. Molecular Cancer 5(1):50.
Gubin, M. M., X. Zhang, H. Schuster, E. Caron, J. P. Ward, et al. 2014. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature 515(7528): 577-581.
Gudmundsdottir, K., and A. Ashworth. 2006. The roles of BRCA1 and BRAC2 and associated proteins in the maintenance of genomic stability. Oncogene 25(43):5864-5874.
Guth, U., D. J. Huang, A. Schotzau, and E. Wight. 2010. Is the current concept of recurrent ovarian carcinoma as a chronic disease also applicable in platinum resistant patients? Archives of Gynecology and Obstetrics 281(2):339-344.
Hacker, N. F. 2011. Quality control in ovarian cancer surgery. Annals of Oncology 22(Suppl 8):viii19-viii22.
Hacker, N. F. 2013. State of the art of surgery in advanced epithelial ovarian cancer. Annals of Oncology 24(Suppl 10):x27-x32.
Hall, G. D., J. M. Brown, R. E. Coleman, M. Stead, K. S. Metcalf, K. R. Peel, C. Poole, M. Crawford, B. Hancock, P. J. Selby, and T. J. Perren. 2004. Maintenance treatment with interferon for advanced ovarian cancer: Results of the Northern and Yorkshire gynaecology group randomised phase III study. British Journal of Cancer 91(4):621-626.
Hampel, H., R. L. Bennett, A. Buchanan, R. Pearlman, G. L. Wiesner, and the Guideline Development Group of the American College of Medical Genetics and Genomics Professional Practice and Guidelines Committee and the National Society of Genetic Counselors Practice Guidelines Committee. 2015. A practice guideline from the American College of Medical Genetics and Genomics and the National Society of Genetic Counselors: Referral indications for cancer predisposition assessment. Genetics in Medicine 17(1):70-87.
Harlan, L. C., L. X. Clegg, and E. L. Trimble. 2003. Trends in surgery and chemotherapy for women diagnosed with ovarian cancer in the United States. Journal of Clinical Oncology 21(18):3488-3494.
Harter, P., A. du Bois, M. Hahmann, A. Hasenburg, A. Burges, et al. 2006. Surgery in recurrent ovarian cancer: The Arbeitsgemeinschaft Gynaekologische Onkologie (AGO) DESKTOP OVAR trial. Annals of Surgical Oncology 13(12):1702-1710.
Harter, P., J. Sehouli, A. Reuss, A. Hasenburg, G. Scambia, et al. 2011. Prospective validation study of a predictive score for operability of recurrent ovarian cancer: The Multicenter Intergroup Study DESKTOP II. A project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. International Journal of Gynecological Cancer 21(2):289-295.
Harter, P., B. Beutel, P. F. Alesina, D. Lorenz, A. Boergers, F. Heitz, R. Hils, C. Kurzeder, A. Traut, and A. du Bois. 2014. Prognostic and predictive value of the Arbeitsgemeinschaft Gynaekologische Onkologie (AGO) score in surgery for recurrent ovarian cancer. Gynecologic Oncology 132(3):537-541.
Hayes, D. F., J. Allen, C. Compton, G. Gustavsen, D. G. Leonard, R. McCormack, L. Newcomer, K. Pothier, D. Ransohoff, R. L. Schilsky, E. Sigal, S. E. Taube, and S. R. Tunis. 2013. Breaking a vicious cycle. Science Translational Medicine 5(196): 196cm6.
Herzog, T. J., D. K. Armstrong, M. F. Brady, R. L. Coleman, M. H. Einstein, B. J. Monk, R. S. Mannel, J. T. Thigpen, S. A. Umpierre, J. A. Villella, and R. D. Alvarez. 2014. Ovarian cancer clinical trial endpoints: Society of Gynecologic Oncology white paper. Gynecologic Oncology 132(1):8-17.
Hess, L. M., M. Benham-Hutchins, T. J. Herzog, C. H. Hsu, D. C. Malone, G. H. Skrepnek, M. K. Slack, and D. S. Alberts. 2007. A meta-analysis of the efficacy of intraperitoneal cisplatin for the front-line treatment of ovarian cancer. International Journal of Gynecological Cancer 17(3):561-570.
Hey, S. P., and J. Kimmelman. 2015. Are outcome-adaptive allocation trials ethical? Clinical Trials (London, England) 12(2):102-106.
HHS (U.S. Department of Health and Human Services). 2013. Increase the proportion of women with a family history of breast and/or ovarian cancer who receive genetic counseling. http://www.healthypeople.gov/2020/topics-objectives/topic/genomics/objectives (accessed September 15, 2015).
Hicklin, D. J., and L. M. Ellis. 2005. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. Journal of Clinical Oncology 23(5):1011-1027.
Hirte, H., S. Lheureux, G. F. Fleming, A. Sugimoto, R. Morgan, J. Biagi, L. Wang, S. McGill, S. P. Ivy, and A. M. Oza. 2015. A phase 2 study of cediranib in recurrent or persistent ovarian, peritoneal or fallopian tube cancer: A trial of the Princess Margaret, Chicago and California Phase II Consortia. Gynecologic Oncology 138(1):55-61.
Hodi, F. S., S. J. O—Day, D. F. McDermott, R. W. Weber, J. A. Sosman, et al. 2010. Improved survival with ipilimumab in patients with metastatic melanoma. New England Journal of Medicine 363(8):711-723
Hoffmann-La Roche. 2015. An observational study of avastin (bevacizumab) in patients with advanced/metastatic epithelial ovarian cancer, fallopian tube cancer or primary peritoneal cancer. Bethesda, MD: National Library of Medicine. https://clinicaltrials.gov/ct2/show/NCT01932125 (accessed December 18, 2015).
Horowitz, N. S., A. Miller, B. Rungruang, S. D. Richard, N. Rodriguez, M. A. Bookman, C. A. Hamilton, T. C. Krivak, and G. L. Maxwell. 2015. Does aggressive surgery improve outcomes? Interaction between preoperative disease burden and complex surgery in patients with advanced-stage ovarian cancer: An analysis of GOG 182. Journal of Clinical Oncology 33(8):937-943.
Horstmann, E., M. S. McCabe, L. Grochow, S. Yamamoto, L. Rubinstein, T. Budd, D. Shoemaker, E. J. Emanuel, and C. Grady. 2005. Risks and benefits of phase 1 oncology trials, 1991 through 2002. New England Journal of Medicine 352(9):895-904.
Hoskins, W. J., W. P. McGuire, M. F. Brady, H. D. Homesley, W. T. Creasman, M. Berman, H. Ball, and J. S. Berek. 1994. The effect of diameter of largest residual disease on survival after primary cytoreductive surgery in patients with suboptimal residual epithelial ovarian carcinoma. American Journal of Obstetrics and Gynecology 170(4):974-979; discussion 979-980.
Howell, E. A., N. Egorova, M. P. Hayes, J. Wisnivesky, R. Franco, and N. Bickell. 2013. Racial disparities in the treatment of advanced epithelial ovarian cancer. Obstetrics and Gynecology 122(5):1025-1032.
Hunsberger, S., Y. Zhao, and R. Simon. 2009. A comparison of phase II study strategies. Clinical Cancer Research 15(19):5950-5955.
Hurria, A., S. G. Mohile, and W. Dale. 2012. Research priorities in geriatric oncology: Addressing the needs of an aging population. Journal of the National Comprehensive Cancer Network 10(2):286-288.
Hwang, W. T., S. F. Adams, E. Tahirovic, I. S. Hagemann, and G. Coukos. 2012. Prognostic significance of tumor-infiltrating T cells in ovarian cancer: A meta-analysis. Gynecologic Oncology 124(2):192-198.
Hylander, B., E. Repasky, P. Shrikant, M. Intengan, A. Beck, D. Driscoll, P. Singhal, S. Lele, and K. Odunsi. 2006. Expression of Wilms tumor gene (WT1) in epithelial ovarian cancer. Gynecologic Oncology 101(1):12-17.
IOM (Institute of Medicine). 2008. Retooling for an aging America: Building the health care workforce. Washington, DC: The National Academies Press.
IOM. 2009. Ensuring quality cancer care through the oncology workforce: Sustaining care in the 21st century: Workshop summary. Washington, DC: The National Academies Press
IOM. 2010. Evaluation of biomarkers and surrogate endpoints in chronic disease. Washington, DC: The National Academies Press.
IOM. 2012. Evolution of translational omics: Lessons learned and the path forward. Washington, DC: The National Academies Press.
IOM. 2013. Delivering high-quality cancer care: Charting a new course for a system in crisis. Washington, DC: The National Academies Press.
IOM and NRC (National Research Council). 1999. Ensuring quality cancer care. Washington, DC: National Academy Press.
Ivy, S. P., L. L. Siu, E. Garrett-Mayer, and L. Rubinstein. 2010. Approaches to phase 1 clinical trial design focused on safety, efficiency, and selected patient populations: A report from the Clinical Trial Design Task Force of the National Cancer Institute Investigational Drug Steering Committee. Clinical Cancer Research 16(6):1726-1736.
Jaaback, K., N. Johnson, and T. A. Lawrie. 2011. Intraperitoneal chemotherapy for the initial management of primary epithelial ovarian cancer. Cochrane Database of Systematic Reviews 11:CD005340.
Jordan, S., C. Steer, A. DeFazio, M. Quinn, A. Obermair, M. Friedlander, J. Francis, S. O—Brien, G. Goss, D. Wyld, Australian Ovarian Cancer Study Group, and P. Webb for the Ovarian Cancer Patterns of Care Study Group. 2013. Patterns of chemotherapy treatment for women with invasive epithelial ovarian cancer: A population-based study. Gynecologic Oncology 129(2):310-317.
Joslin, C. E., K. C. Brewer, F. G. Davis, K. Hoskins, C. E. Peterson, and H. A. Pauls. 2014. The effect of neighborhood-level socioeconomic status on racial differences in ovarian cancer treatment in a population-based analysis in Chicago. Gynecologic Oncology 135(2):285-291.
Kalos, M., B. L. Levine, D. L. Porter, S. Katz, S. A. Grupp, A. Bagg, and C. H. June. 2011. T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Science Translational Medicine 3(95):95ra73.
Katsumata, N., M. Yasuda, F. Takahashi, S. Isonishi, T. Jobo, D. Aoki, H. Tsuda, T. Sugiyama, S. Kodama, E. Kimura, K. Ochiai, K. Noda, and Japanese Gynecologic Oncology Group. 2009. Dose-dense paclitaxel once a week in combination with carboplatin every 3 weeks for advanced ovarian cancer: A phase 3, open-label, randomised controlled trial. Lancet 374(9698):1331-1338.
Kaye, S. B., J. Lubinski, U. Matulonis, J. E. Ang, C. Gourley, B. Y. Karlan, A. Amnon, K. M. Bell-McGuinn, L. M. Chen, M. Friedlander, T. Safra, I. Vergote, M. Wickens, E. S. Lowe, J. Carmichael, and B. Kaufman. 2012. Phase II, open-label, randomized, multicenter study comparing the efficacy and safety of olaparib, a poly (ADP-ribose) polymerase inhibitor, and pegylated liposomal doxorubicin in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer. Journal of Clinical Oncology 30(4): 372-379.
Kehoe, S., and M. Nankivell. 2015. Primary chemotherapy versus primary surgery for ovarian cancer—Authors’ reply. Lancet 386(10009):2143.
Kehoe, S., J. Hook, M. Nankivell, G. C. Jayson, H. Kitchener, T. Lopes, D. Luesley, T. Perren, S. Bannoo, M. Mascarenhas, S. Dobbs, S. Essapen, J. Twigg, J. Herod, G. McCluggage, M. Parmar, and A. M. Swart. 2015. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): An open-label, randomised, controlled, non-inferiority trial. Lancet 386(9990):249-257.
Kim, G., G. Ison, A. E. McKee, H. Zhang, S. Tang, et al. 2015. FDA approval summary: Olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clinical Cancer Research 21(19):4257-4261.
Kobel, M., S. E. Kalloger, N. Boyd, S. McKinney, E. Mehl, C. Palmer, S. Leung, N. J. Bowen, D. N. Ionescu, A. Rajput, L. M. Prentice, D. Miller, J. Santos, K. Swenerton, C. B. Gilks, and D. Huntsman. 2008. Ovarian carcinoma subtypes are different diseases: Implications for biomarker studies. PLoS Medicine 5(12):e232.
Kohn, E. C. 2014. Molecular profiling and commercial predication assays in ovarian cancer: Still not ready for prime time? http://meetinglibrary.asco.org/content/114000139-144 (accessed October 1, 2015).
Korn, E. L., and B. Freidlin. 2011a. On the usefulness of outcome-adaptive randomization—author reply. Journal of Clinical Oncology 29(13):E393.
Korn, E. L., and B. Freidlin. 2011b. Outcome-adaptive randomization: Is it useful? Journal of Clinical Oncology 29(6):771-776.
Korn, E. L., and B. Freidlin. 2012. Methodology for comparative effectiveness research: Potential and limitations. Journal of Clinical Oncology 30(34):4185-4187.
Korn, E. L., and B. Freidlin. 2015. Evaluation of chemoresponse assays as predictive markers. British Journal of Cancer 112(4):621-623.
Korn, E. L., B. Freidlin, and J. S. Abrams. 2011. Overall survival as the outcome for randomized clinical trials with effective subsequent therapies. Journal of Clinical Oncology 29(17):2439-2442.
Korn, E. L., B. Freidlin, J. S. Abrams, and S. Halabi. 2012. Design issues in randomized phase II/III trials. Journal of Clinical Oncology 30(6):667-671.
Korn, R. L., and J. J. Crowley. 2013. Overview: Progression-free survival as an endpoint in clinical trials with solid tumors. Clinical Cancer Research 19(10):2607-2612.
Kummar, S., R. Kinders, L. Rubinstein, R. E. Parchment, A. J. Murgo, J. Collins, O. Pickeral, J. Low, S. M. Steinberg, M. Gutierrez, S. Yang, L. Helman, R. Wiltrout, J. E. Tomaszewski, and J. H. Doroshow. 2007. Compressing drug development timelines in oncology using phase “0” trials. Nature Reviews: Cancer 7(2):131-139.
Kummar, S., A. M. Oza, G. F. Fleming, D. M. Sullivan, D. R. Gandara, et al. 2015. Randomized trial of oral cyclophosphamide and veliparib in high-grade serous ovarian, primary peritoneal, or fallopian tube cancers, or BRCA-mutant ovarian cancer. Clinical Cancer Research 21(7):1574-1582.
Kurzrock, R., and R. S. Benjamin. 2005. Risks and benefits of phase 1 oncology trials, revisited. New England Journal of Medicine 352(9):930-932.
Kwak, E. L., Y. J. Bang, D. R. Camidge, A. T. Shaw, B. Solomon, et al. 2010. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. New England Journal of Medicine 363(18):1693-1703.
Lataifeh, I., D. E. Marsden, G. Robertson, V. Gebski, and N. F. Hacker. 2005. Presenting symptoms of epithelial ovarian cancer. Australian and New Zealand Journal of Obstetrics and Gynaecology 45(3):211-214.
Ledermann, J. A., and F. El-Khouly. 2015. PARP inhibitors in ovarian cancer: Clinical evidence for informed treatment decisions. British Journal of Cancer 113(Suppl 1):S10-S16.
Ledermann, J. A., A. Hackshaw, S. Kaye, G. Jayson, H. Gabra, I. McNeish, H. Earl, T. Perren, M. Gore, M. Persic, M. Adams, L. James, G. Temple, M. Merger, and G. Rustin. 2011. Randomized phase II placebo-controlled trial of maintenance therapy using the oral triple angiokinase inhibitor BIBF 1120 after chemotherapy for relapsed ovarian cancer. Journal of Clinical Oncology 29(28):3798-3804.
Ledermann, J., P. Harter, C. Gourley, M. Friedlander, I. Vergote, G. Rustin, C. Scott, W. Meier, R. Shapira-Frommer, T. Safra, D. Matei, E. Macpherson, C. Watkins, J. Carmichael, and U. Matulonis. 2012. Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. New England Journal of Medicine 366(15):1382-1392.
Lengyel, E. 2010. Ovarian cancer development and metastasis. American Journal of Pathology 177(3):1053-1064.
Lesnock, J. L., K. M. Darcy, C. Tian, J. A. Deloia, M. M. Thrall, C. Zahn, D. K. Armstrong, M. J. Birrer, and T. C. Krivak. 2013. BRCA1 expression and improved survival in ovarian cancer patients treated with intraperitoneal cisplatin and paclitaxel: A Gynecologic Oncology Group study. British Journal of Cancer 108(6):1231-1237.
Lewis, J. H., M. L. Kilgore, D. P. Goldman, E. L. Trimble, R. Kaplan, M. J. Montello, M. G. Housman, and J. J. Escarce. 2003. Participation of patients 65 years of age or older in cancer clinical trials. Journal of Clinical Oncology 21(7):1383-1389.
Liang, M. I., A. C. El Naggar, S. Nekkanti, D. M. O—Malley, E. M. Hade, L. J. Copeland, J. M. Fowler, R. Salani, F. J. Backes, and D. E. Cohn. 2015. Setting the bar: Compliance with ovarian cancer quality indicators at a National Cancer Institute-designated comprehensive cancer center. Gynecologic Oncology 138(3):689-693.
Linnemann, C., M. M. van Buuren, L. Bies, E. M. Verdegaal, R. Schotte, J. J. Calis, S. Behjati, A. Velds, H. Hilkmann, D. E. Atmioui, M. Visser, M. R. Stratton, J. B. Haanen, H. Spits, S. H. van der Burg, and T. N. Schumacher. 2015. High-throughput epitope discovery reveals frequent recognition of neo-antigens by CD4+ T cells in human melanoma. Nature Medicine 21(1):81-85.
Liu, J. F., W. T. Barry, M. Birrer, J. M. Lee, R. J. Buckanovich, G. F. Fleming, B. Rimel, M. K. Buss, S. Nattam, J. Hurteau, W. Luo, P. Quy, C. Whalen, L. Obermayer, H. Lee, E. P. Winer, E. C. Kohn, S. P. Ivy, and U. A. Matulonis. 2014a. Combination cediranib and olaparib versus olaparib alone for women with recurrent platinum-sensitive ovarian cancer: A randomised phase 2 study. Lancet Oncology 15(11):1207-1214.
Luvero, D., A. Milani, and J. A. Ledermann. 2014. Treatment options in recurrent ovarian cancer: Latest evidence and clinical potential. Therapeutic Advances in Medical Oncology 6(5):229-239.
Lyman, G. H. 2009. Comparative effectiveness research in oncology: The need for clarity, transparency and vision. Cancer Investigation 27(6):593-597.
Mackay, H. J., D. Provencheur, M. Heywood, D. Tu, E. A. Eisenhauer, A. M. Oza, and R. Meyer. 2011. Phase II/III study of intraperitoneal chemotherapy after neoadjuvant chemotherapy for ovarian cancer: NCIC CTG OV.21. Current Oncology 18(2):84-90.
Mali, P., L. Yang, K. M. Esvelt, J. Aach, M. Guell, J. E. DiCarlo, J. E. Norville, and G. M. Church. 2013. RNA-guided human genome engineering via Cas9. Science 339(6121):823-826.
Markman, M., B. N. Bundy, D. S. Alberts, J. M. Fowler, D. L. Clark-Pearson, L. F. Carson, S. Wadler, and J. Sickel. 2001. Phase III trial of standard-dose intravenous cisplatin plus paclitaxel versus moderately high-dose carboplatin followed by intravenous paclitaxel and intraperitoneal cisplatin in small-volume stage III ovarian carcinoma: An intergroup study of the Gynecologic Oncology Group, Southwestern Oncology Group, and Eastern Cooperative Oncology Group. Journal of Clinical Oncology 19(4):1001-1007.
Markman, M., P. Y. Liu, S. Wilczynski, B. Monk, L. J. Copeland, R. D. Alvarez, C. Jiang, D. Alberts, G. Southwest Oncology, and G. Gynecologic Oncology. 2003. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy: A Southwest Oncology Group and Gynecologic Oncology Group trial. Journal of Clinical Oncology 21(13):2460-2465.
Mauge, L., M. Terme, E. Tartour, and D. Helley. 2014. Control of the adaptive immune response by tumor vasculature. Frontiers in Oncology 4:61.
McGuire, W. P., W. J. Hoskins, M. F. Brady, P. R. Kucera, E. E. Partridge, K. Y. Look, D. L. Clarke-Pearson, and M. Davidson. 1996. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. New England Journal of Medicine 334(1):1-6.
McShane, L., and S. Hunsberger. 2015. An overview of phase II clinical trial designs with biomarkers. In Design and analysis of clinical trials for predictive medicine. Boca Raton, FL: Chapman and Hall/CRC. Pp. 71-87.
McShane, L. M., and M. Y. Polley. 2013. Development of omics-based clinical tests for prognosis and therapy selection: The challenge of achieving statistical robustness and clinical utility. Clinical Trials (London, England) 10(5):653-665.
McShane, L. M., S. Hunsberger, and A. A. Adjei. 2009. Effective incorporation of biomarkers into phase II trials. Clinical Cancer Research 15(6):1898-1905.
Menear, K. A., C. Adcock, R. Boulter, X. L. Cockcroft, L. Copsey, et al. 2008. 4-[3-(4-cyclopropanecarbonylpiperazine-1-carbonyl)-4-fluorobenzyl]-2H-phthalazin-1-one: A novel bioavailable inhibitor of poly(ADP-ribose) polymerase-1. Journal of Medicinal Chemistry 51(20):6581-6591.
Michels, J., I. Vitale, M. Saparbaev, M. Castedo, and G. Kroemer. 2014. Predictive biomarkers for cancer therapy with PARP inhibitors. Oncogene 33(30):3894-3907.
Mobus, V. J., R. P. Baum, M. Bolle, R. Kreienberg, A. A. Noujaim, B. C. Schultes, and C. F. Nicodemus. 2003. Immune responses to murine monoclonal antibody-B43.13 correlate with prolonged survival of women with recurrent ovarian cancer. American Journal of Obstetrics and Gynecology 189(1):28-36.
Modesitt, S. C. and J. R. van Nagell, Jr. 2005. The impact of obesity on the incidence and treatment of gynecologic cancers: A review. Obstetrical and Gynecological Survey 60(10):683-692.
Mohell, N., J. Alfredsson, A. Fransson, M. Uustalu, S. Bystrom, J. Gullbo, A. Hallberg, V. J. Bykov, U. Bjorklund, and K. G. Wiman. 2015. APR-246 overcomes resistance to cisplatin and doxorubicin in ovarian cancer cells. Cell Death & Disease 6:e1794.
Monk, B. J., A. Poveda, I. Vergote, F. Raspagliesi, K. Fujiwara, et al. 2014. Anti-angiopoietin therapy with trebananib for recurrent ovarian cancer (TRINOVA-1): A randomised, multicentre, double-blind, placebo-controlled phase 3 trial. Lancet Oncology 15(8):799-808.
Morrison, J., K. Haldar, S. Kehoe, and T. A. Lawrie. 2012. Chemotherapy versus surgery for initial treatment in advanced ovarian epithelial cancer. Cochrane Database of Systematic Reviews 8:CD005343.
Moy, B., T. W. Flaig, H. B. Muss, B. Clark, W. Tse, and T. C. Windham. 2014. Geriatric oncology for the 21st century: A call for action. Journal of Oncology Practice 10(4):241-243.
Munoz, M., P. T. Ramirez, C. Echeverri, L. G. Alvarez, M. A. Palomino, and L. R. Pareja. 2012. Gastrointestinal stromal tumors as an incidental finding in patients with a presumptive diagnosis of ovarian cancer. Journal of Gynecologic Oncology 23(1):48-52.
Murthy, V. H., H. M. Krumholz, and C. P. Gross. 2004. Participation in cancer clinical trials: Race-, sex-, and age-based disparities. Journal of the American Medical Association 291(22):2720-2726.
Mutch, D. G., and J. Prat. 2014. 2014 FIGO staging for ovarian, fallopian tube and peritoneal cancer. Gynecologic Oncology 133(3):401-404.
Nagle, C. M., S. C. Dixon, A. Jensen, S. K. Kjaer, F. Modugno, et al. 2015. Obesity and survival among women with ovarian cancer: Results from the Ovarian Cancer Association Consortium. British Journal of Cancer 113(5):817-826.
NCCN (National Comprehensive Cancer Network). 2015. Ovarian cancer including fallopian tube cancer and primary peritoneal cancer (version 1.2015). http://www.nccn.org/professionals/physician_gls/pdf/ovarian.pdf (accessed June 3, 2015).
NCI (National Cancer Institute). 2015. NCI-molecular analysis for therapy choice (NCI-match) trial. http://www.cancer.gov/about-cancer/treatment/clinical-trials/nci-supported/nci-match (accessed December 18, 2015).
Neijt, J. P., W. W. ten Bokkel Huinink, M. E. van der Burg, A. T. van Oosterom, R. Vriesendorp, C. D. Kooyman, A. C. van Lindert, J. V. Hamerlynck, M. van Lent, J. C. van Houwelingen, et al. 1984. Randomised trial comparing two combination chemotherapy regimens (Hexa-CAF vs CHAP-5) in advanced ovarian carcinoma. Lancet 2(8403):594-600.
Nick, A. M., R. L. Coleman, P. T. Ramirez, and A. K. Sood. 2015. A framework for a personalized surgical approach to ovarian cancer. Nature Reviews: Clinical Oncology 12(4):239-245.
NIH (National Institutes of Health) Consensus Development Panel on Ovarian Cancer. 1995. NIH consensus conference. Ovarian cancer. Screening, treatment, and follow-up. Journal of the American Medical Association 273(6):491-497.
Nolen, B. M., and A. E. Lokshin. 2013. Biomarker testing for ovarian cancer: Clinical utility of multiplex assays. Molecular Diagnosis and Therapy 17(3):139-146.
Noujaim, A. A., B. C. Schultes, R. P. Baum, and R. Madiyalakan. 2001. Induction of CA125-specific B and T cell responses in patients injected with MAb-B43.13—Evidence for antibody-mediated antigen-processing and presentation of CA125 in vivo. Cancer Biotherapy and Radiopharmaceuticals 16(3):187-203.
NRG Oncology. 2015. Comprehensive patient questionnaires in predicting complications in older patients with gynecologic cancer undergoing surgery. Bethesda, MD: National Library of Medicine. https://clinicaltrials.gov/ct2/show/NCT02315469?term=NCT02315469 (accessed January 29, 2016).
Odunsi, K., A. A. Jungbluth, E. Stockert, F. Qian, S. Gnjatic, J. Tammela, M. Intengan, A. Beck, B. Keitz, D. Santiago, B. Williamson, M. J. Scanlan, G. Ritter, Y. T. Chen, D. Driscoll, A. Sood, S. Lele, and L. J. Old. 2003. NY-ESO-1 and LAGE-1 cancer-testis antigens are potential targets for immunotherapy in epithelial ovarian cancer. Cancer Research 63(18):6076-6083.
Odunsi, K., F. Qian, J. Matsuzaki, P. Mhawech-Fauceglia, C. Andrews, E. W. Hoffman, L. Pan, G. Ritter, J. Villella, B. Thomas, K. Rodabaugh, S. Lele, P. Shrikant, L. J. Old, and S. Gnjatic. 2007. Vaccination with an NY-ESO-1 peptide of HLA class I/II specificities induces integrated humoral and T cell responses in ovarian cancer. Proceedings of the National Academy of Sciences of the United States of America 104(31):12837-12842.
Odunsi, K., J. Matsuzaki, J. Karbach, A. Neumann, P. Mhawech-Fauceglia, A. Miller, A. Beck, C. D. Morrison, G. Ritter, H. Godoy, S. Lele, N. duPont, R. Edwards, P. Shrikant, L. J. Old, S. Gnjatic, and E. Jager. 2012. Efficacy of vaccination with recombinant vaccinia and fowlpox vectors expressing NY-ESO-1 antigen in ovarian cancer and melanoma patients. Proceedings of the National Academy of Sciences of the United States of America 109(15):5797-5802.
Odunsi, K., J. Matsuzaki, S. R. James, P. Mhawech-Fauceglia, T. Tsuji, A. Miller, W. Zhang, S. N. Akers, E. A. Griffiths, A. Miliotto, A. Beck, C. A. Batt, G. Ritter, S. Lele, S. Gnjatic, and A. R. Karpf. 2014. Epigenetic potentiation of NY-ESO-1 vaccine therapy in human ovarian cancer. Cancer Immunology Research 2(1):37-49.
Olsen, C. M., J. Cnossen, A. C. Green, and P. M. Webb. 2007. Comparison of symptoms and presentation of women with benign, low malignant potential and invasive ovarian tumors. European Journal of Gynaecological Oncology 28(5):376-380.
Omura, G., J. A. Blessing, C. E. Ehrlich, A. Miller, E. Yordan, W. T. Creasman, and H. D. Homesley. 1986. A randomized trial of cyclophosphamide and doxorubicin with or without cisplatin in advanced ovarian carcinoma. A Gynecologic Oncology Group study. Cancer 57(9):1725-1730.
Omura, G. A., B. N. Bundy, J. S. Berek, S. Curry, G. Delgado, and R. Mortel. 1989. Randomized trial of cyclophosphamide plus cisplatin with or without doxorubicin in ovarian carcinoma: A Gynecologic Oncology Group study. Journal of Clinical Oncology 7(4):457-465.
O—Quigley, J., M. Pepe, and L. Fisher. 1990. Continual reassessment method: A practical design for phase 1 clinical trials in cancer. Biometrics 46(1):33-48.
Oza, A. M., D. Cibula, A. O. Benzaquen, C. Poole, R. H. Mathijssen, G. S. Sonke, N. Colombo, J. Spacek, P. Vuylsteke, H. Hirte, S. Mahner, M. Plante, B. Schmalfeldt, H. Mackay, J. Rowbottom, E. S. Lowe, B. Dougherty, J. C. Barrett, and M. Friedlander. 2015. Olaparib combined with chemotherapy for recurrent platinum-sensitive ovarian cancer: A randomised phase 2 trial. Lancet Oncology 16(1):87-97.
Ozols, R. F., B. N. Bundy, B. E. Greer, J. M. Fowler, D. Clarke-Pearson, R. A. Burger, R. S. Mannel, K. DeGeest, E. M. Hartenbach, R. Baergen, and Gynecologic Oncology Group. 2003. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: A Gynecologic Oncology Group study. Journal of Clinical Oncology 21(17):3194-3200.
Parmar, M. K., J. A. Ledermann, N. Colombo, A. du Bois, J. F. Delaloye, G. B. Kristensen, S. Wheeler, A. M. Swart, W. Qian, V. Torri, I. Floriani, G. Jayson, A. Lamont, C. Trope, and ICON and AGO collaborators. 2003. Paclitaxel plus platinum-based chemotherapy versus conventional platinum-based chemotherapy in women with relapsed ovarian cancer: The ICON4/AGO-OVAR-2.2 trial. Lancet 361(9375):2099-2106.
Pavelka, J. C., R. S. Brown, B. Y. Karlan, I. Cass, R. S. Leuchter, L. O. Lagasse, and A. J. Li. 2006. Effect of obesity on survival in epithelial ovarian cancer. Cancer 107(7):1520-1524.
Perren, T. J., A. M. Swart, J. Pfisterer, J. A. Ledermann, E. Pujade-Lauraine, et al. 2011. A phase 3 trial of bevacizumab in ovarian cancer. New England Journal of Medicine 365(26):2484-2496.
Pfisterer, J., M. Plante, I. Vergote, A. du Bois, H. Hirte, et al. 2006. Gemcitabine plus carboplatin compared with carboplatin in patients with platinum-sensitive recurrent ovarian cancer: An intergroup trial of the AGO-OVAR, the NCIC CTG, and the EORTC GCG. Journal of Clinical Oncology 24(29):4699-4707.
Piccart, M. J., K. Bertelsen, K. James, J. Cassidy, C. Mangioni, et al. 2000. Randomized intergroup trial of cisplatin-paclitaxel versus cisplatin-cyclophosphamide in women with advanced epithelial ovarian cancer: Three-year results. Journal of the National Cancer Institute 92(9):699-708.
Plamadeala, V., J. L. Kelley, J. K. Chan, T. C. Krivak, M. J. Gabrin, S. L. Brower, M. A. Powell, T. J. Rutherford, and R. L. Coleman. 2015. A cost-effectiveness analysis of a chemoresponse assay for treatment of patients with recurrent epithelial ovarian cancer. Gynecologic Oncology 136(1):94-98.
Polley, M. Y., B. Freidlin, E. L. Korn, B. A. Conley, J. S. Abrams, and L. M. McShane. 2013. Statistical and practical considerations for clinical evaluation of predictive biomarkers. Journal of the National Cancer Institute 105(22):1677-1683.
Polsky, D., K. A. Armstrong, T. C. Randall, R. N. Ross, O. Even-Shoshan, P. R. Rosenbaum, J. H. Silber. 2006. Variation in chemotherapy utilization in ovarian cancer: The relative contribution of geography. Health Services Research 41(6):2201-2218.
Powell, C. B., R. Littell, E. Hoodfar, F. Sinclair, and A. Pressman. 2013. Does the diagnosis of breast or ovarian cancer trigger referral to genetic counseling? International Journal of Gynecological Cancer 23(3):431-436.
Prat, A., M. Parera, B. Adamo, S. Peralta, M. A. Perez-Benavente, A. Garcia, A. Gil-Moreno, J. M. Martinez-Palones, J. Baselga, and J. M. del Campo. 2009. Risk of recurrence during follow-up for optimally treated advanced epithelial ovarian cancer (EOC) with a low-level increase of serum CA-125 levels. Annals of Oncology 20(2):294-297.
Pujade-Lauraine, E., F. Hilpert, B. Weber, A. Reuss, A. Poveda, G. Kristensen, R. Sorio, I. Vergote, P. Witteveen, A. Bamias, D. Pereira, P. Wimberger, A. Oaknin, M. R. Mirza, P. Follana, D. Bollag, and I. Ray-Coquard. 2014. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: The AURELIA open-label randomized phase III trial. Journal of Clinical Oncology 32(13):1302-1308.
Raja, F. A., N. Counsell, N. Colombo, J. Pfisterer, A. du Bois, M. K. Parmar, I. B. Vergote, A. Gonzalez-Martin, D. S. Alberts, M. Plante, V. Torri, and J. A. Ledermann. 2013. Platinum versus platinum-combination chemotherapy in platinum-sensitive recurrent ovarian cancer: A meta-analysis using individual patient data. Annals of Oncology 24(12):3028-3034.
Redman, M. W. 2012. Early stopping of clinical trials. In Handbook of statistics in clinical oncology, 3rd ed., edited by J. Crowley and A. Hoering. Boca Raton, FL: Chapman and Hall/CRC. Pp. 211-228.
Rizvi, N. A., M. D. Hellmann, A. Snyder, P. Kvistborg, V. Makarov, et al. 2015. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348(6230):124-128.
Robbins, P. F., R. A. Morgan, S. A. Feldman, J. C. Yang, R. M. Sherry, et al. 2011. Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. Journal of Clinical Oncology 29(7):917-924.
Robboy, S., Kraus, FT, Kurman, RJ. 2002. Gross description, processing and reporting of gynecologic and obstetric specimens. In Blaustein’s pathology of the female genital tract, edited by R. Kurman. New York: Springer. Pp. 1319-1345.
Robert, C., A. Ribas, J. D. Wolchok, F. S. Hodi, O. Hamid, et al. 2014. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: A randomised dose-comparison cohort of a phase 1 trial. Lancet 384(9948):1109-1117.
Rosenberg, S. A., N. P. Restifo, J. C. Yang, R. A. Morgan, and M. E. Dudley. 2008. Adoptive cell transfer: A clinical path to effective cancer immunotherapy. Nature Reviews: Cancer 8(4):299-308.
Rosenberger, W. F., and J. M. Lachin. 1993. The use of response-adaptive designs in clinical trials. Controlled Clinical Trials 14(6):471-484.
Rubinstein, L. V. 2000. Therapeutic studies. Hematology/Oncology Clinics of North America 14(4):849-876.
Rubinstein, L. V., E. L. Korn, B. Freidlin, S. Hunsberger, S. P. Ivy, and M. A. Smith. 2005. Design issues of randomized phase II trials and a proposal for phase II screening trials. Journal of Clinical Oncology 23(28):7199-7206.
Rubinstein, L., J. Crowley, P. Ivy, M. Leblanc, and D. Sargent. 2009. Randomized phase II designs. Clinical Cancer Research 15(6):1883-1890.
Rustin, G. J. 2011. Follow-up with CA125 after primary therapy of advanced ovarian cancer has major implications for treatment outcome and trial performances and should not be routinely performed. Annals of Oncology 22(Suppl 8):viii45-viii48.
Rustin, G. J., M. E. van der Burg, C. L. Griffin, D. Guthrie, A. Lamont, G. C. Jayson, G. Kristensen, C. Mediola, C. Coens, W. Qian, M. K. Parmar, and A. M. Swart, for the MRC OV05, EORTC 55955 investigators. 2010. Early versus delayed treatment of relapsed ovarian cancer (MRC OV05/EORTC 55955): A randomised trial. Lancet 376(9747):1155-1163.
Rutherford, T., J. Orr, Jr., E. Grendys, Jr., R. Edwards, T. C. Krivak, R. Holloway, R. G. Moore, L. Puls, T. Tillmanns, J. C. Schink, S. L. Brower, C. Tian, and T. J. Herzog. 2013. A prospective study evaluating the clinical relevance of a chemoresponse assay for treatment of patients with persistent or recurrent ovarian cancer. Gynecologic Oncology 131(2):362-367.
Sabbatini, P., T. Tsuji, L. Ferran, E. Ritter, C. Sedrak, et al. 2012. Phase I trial of overlapping long peptides from a tumor self-antigen and poly-ICLC shows rapid induction of integrated immune response in ovarian cancer patients. Clinical Cancer Research 18(23):6497-6508.
Sabbatini, P., P. Harter, G. Scambia, J. Sehouli, W. Meier, et al. 2013. Abagovomab as maintenance therapy in patients with epithelial ovarian cancer: A phase III trial of the AGO OVAR, COGI, GINECO, and GEICO—the MIMOSA study. Journal of Clinical Oncology 31(12):1554-1561.
Sahin, U., O. Tureci, H. Schmitt, B. Cochlovius, T. Johannes, R. Schmits, F. Stenner, G. Luo, I. Schobert, and M. Pfreundschuh. 1995. Human neoplasms elicit multiple specific immune responses in the autologous host. Proceedings of the National Academy of Sciences of the United States of America 92(25):11810-11813.
Salani, R., F. J. Backes, M. F. Fung, C. H. Holschneider, L. P. Parker, R. E. Bristow, and B. A. Goff. 2011. Posttreatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: Society of Gynecologic Oncologists recommendations. American Journal of Obstetrics and Gynecology 204(6):466-478.
Sargent, D. J., B. A. Conley, C. Allegra, and L. Collette. 2005. Clinical trial designs for predictive marker validation in cancer treatment trials. Journal of Clinical Oncology 23(9):2020-2027.
Sato, E., S. H. Olson, J. Ahn, B. Bundy, H. Nishikawa, F. Qian, A. A. Jungbluth, D. Frosina, S. Gnjatic, C. Ambrosone, J. Kepner, T. Odunsi, G. Ritter, S. Lele, Y. T. Chen, H. Ohtani, L. J. Old, and K. Odunsi. 2005. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proceedings of the National Academy of Sciences of the United States of America 102(51):18538-18543.
Sawyers, C. L., and L. J. van ’t Veer. 2014. Reliable and effective diagnostics are keys to accelerating personalized cancer medicine and transforming cancer care: A policy statement from the American Association for Cancer Research. Clinical Cancer Research 20(19):4978-4981.
Scaletta, G., F. Plotti, A. Aloisi, and R. Angioli. 2015. Primary chemotherapy versus primary surgery for ovarian cancer. Lancet 386(10009):2142.
Scanlan, M. J., N. K. Altorki, A. O. Gure, B. Williamson, A. Jungbluth, Y. T. Chen, and L. J. Old. 2000. Expression of cancer-testis antigens in lung cancer: Definition of bromodomain testis-specific gene (BRDT) as a new CT gene, CT9. Cancer Letters 150(2): 155-164.
Schiphorst A. H. W., D. Ten Bokkel Huinink, R. Breumelhof, J. P. J. Burgmans, A. Pronk, and M. E. Hamaker. 2015. Geriatric consultation can aid in complex treatment decisions for elderly cancer patients European Journal of Cancer Care (Epub ahead of print).
Scott, C. L., M. A. Becker, P. Haluska, and G. Samimi. 2013. Patient-derived xenograft models to improve targeted therapy in epithelial ovarian cancer treatment. Frontiers in Oncology 3:295.
Scully, R. E., D. E. Henson, M. L. Nielsen, and S. G. Ruby. 1996. Practice protocol for the examination of specimens removed from patients with ovarian tumors: A basis for checklists. Cancer Committee, College of American Pathologists. Gynecologic Oncology 63(2):276-289.
SGO (Society of Gynecologic Oncology). 2015. Quality indicators. https://www.sgo.org/quality-outcomes-and-research/quality-indicators (accessed December 17, 2015).
Shalowitz, D. I., A. G. Smith, M. C. Bell, and R. K. Gibb. 2015. Teleoncology for gynecologic cancers. Gynecologic Oncology 139(1):172-177.
Shankaran, V., H. Ikeda, A. T. Bruce, J. M. White, P. E. Swanson, L. J. Old, and R. D. Schreiber. 2001. IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410(6832):1107-1111.
Simon, R., B. Freidlin, L. Rubinstein, S. G. Arbuck, J. Collins, and M. C. Christian. 1997. Accelerated titration designs for phase I clinical trials in oncology. Journal of the National Cancer Institute 89(15):1138-1147.
Smits, A., E. Smits, A. Lopes, N. Das, G. Hughes, A. Talaat, A. Pollard, F. Bouwman, L. Massuger, R. Bekkers, and K. Galaal. 2015a. Body mass index, physical activity and quality of life in ovarian cancer survivors: Time to get moving? Gynecologic Oncology 139(1):148-154.
Smits, A., A. Lopes, N. Das, A. Kumar, W. Cliby, E. Smits, R. Bekkers, L. Massuger, and K. Galaal. 2015b. Surgical morbidity and clinical outcomes in ovarian cancer: The role of obesity. BJOG: An International Journal of Obstetrics and Gynaecology. (Epub ahead of print).
Snyder, A., V. Makarov, T. Merghoub, J. Yuan, J. M. Zaretsky, et al. 2014. Genetic basis for clinical response to CTLA-4 blockade in melanoma. New England Journal of Medicine 371(23):2189-2199.
Spannuth, W. A., A. K. Sood, and R. L. Coleman. 2008. Angiogenesis as a strategic target for ovarian cancer therapy. Nature Clinical Practice: Oncology 5(4):194-204.
Sparreboom, A., A. C. Wolff, R. H. J. Mathijssen, E. Chatelut, E. K. Rowinsky, J. Verweij, and S. D. Baker. 2007. Evaluation of alternate size descriptors for dose calculation of anticancer drugs in the obese. Journal of Clinical Oncology 25(30):4707-4713.
Stewart, J. H., A. G. Bertoni, J. L. Staten, E. A. Levine, and C. P. Gross. 2007. Participation in surgical oncology clinical trials: Gender-, race/ethnicity-, and age-based disparities. Annals of Surgical Oncology 14(12):3328-3334.
Storey, D. J., R. Rush, M. Stewart, T. Rye, A. Al-Nafussi, A. R. Williams, J. F. Smyth, and H. Gabra. 2008. Endometrioid epithelial ovarian cancer: 20 years of prospectively collected data from a single center. Cancer 112(10):2211-2220.
Stuart, G. C., H. Kitchener, M. Bacon, A. duBois, M. Friedlander, J. Ledermann, C. Marth, T. Thigpen, E. Trimble, participants of 4th Ovarian Cancer Consensus Conference (OCCC), and Gynecologic Cancer Intergroup. 2011. 2010 Gynecologic Cancer Intergroup (GCIG) consensus statement on clinical trials in ovarian cancer: Report from the fourth Ovarian Cancer Consensus Conference. International Journal of Gynecological Cancer 21(4):750-755.
Suvarna, V. 2010. Consort 2010: A standard for reporting clinical trials revised anew? Perspectives in Clinical Research 1(3):87-89.
Symeonides, S., and C. Gourley. 2015. Ovarian cancer molecular stratification and tumor heterogeneity: A necessity and a challenge. Frontiers in Oncology 5:229.
Sznol, M. 2010. Reporting disease control rates or clinical benefit rates in early clinical trials of anticancer agents: Useful endpoint or hype? Current Opinion in Investigational Drugs 11(12):1340-1341.
Talarico, L., G. Chen, and R. Pazdur. 2004. Enrollment of elderly patients in clinical trials for cancer drug registration: A 7-year experience by the U.S. Food and Drug Administration. Journal of Clinical Oncology 22(22):4626-4631.
Teplinsky, E., and F. Muggia. 2015. EGFR and HER2: Is there a role in ovarian cancer? Translational Cancer Research 4(1):107-117.
Tewari, D., J. J. Java, R. Salani, D. K. Armstrong, M. Markman, T. Herzog, B. J. Monk, and J. K. Chan. 2015. Long-term survival advantage and prognostic factors associated with intraperitoneal chemotherapy treatment in advanced ovarian cancer: A Gynecologic Oncology Group study. Journal of Clinical Oncology 33(13):1460-1466.
Thrall, M. M., H. J. Gray, R. G. Symons, N. S. Weiss, D. R. Flum, and B. A. Goff. 2011. Trends in treatment of advanced epithelial ovarian cancer in the Medicare population. Gynecologic Oncology 122(1):100-106.
Tian, W. J., D. S. Chi, J. Sehouli, C. G. Trope, R. Jiang, A. Ayhan, G. Cormio, Y. Xing, G. P. Breitbach, E. I. Braicu, C. A. Rabbitt, H. Oksefjell, C. Fotopoulou, H. G. Meerpohl, A. Du Bois, J. S. Berek, R. Y. Zang, and P. Harter. 2012. A risk model for secondary cytoreductive surgery in recurrent ovarian cancer: An evidence-based proposal for patient selection. Annals of Surgical Oncology 19(2):597-604.
Torres, M. L., L. C. Hartmann, W. A. Cliby, K. R. Kalli, P. M. Young, A. L. Weaver, C. L. Langstraat, A. Jatoi, S. Kumar, A. Mariani. 2013. 129(3):548-553.
Tumeh, P. C., C. L. Harview, J. H. Yearley, I. P. Shintaku, E. J. Taylor, et al. 2014. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515(7528):568-571.
Ulanday, K. T., K. K. Ward, C. A. Macera, M. Ji, and S. C. Plaxe. 2014. Regional variation in surgical assessment of lymph nodes for staging among women with early-stage epithelial ovarian cancer. Gynecologic Oncology 132(2):411-415.
van de Laar, R., P. L. Zusterzeel, T. Van Gorp, M. R. Buist, W. J. van Driel, K. N. Gaarenstroom, H. J. Arts, J. C. van Huisseling, R. H. Hermans, J. M. Pijnenborg, E. M. Schutter, H. M. Pelikan, J. H. Vollebergh, M. J. Engelen, J. Inthout, R. F. Kruitwagen, and L. F. Massuger. 2014. Cytoreductive surgery followed by chemotherapy versus chemotherapy alone for recurrent platinum-sensitive epithelial ovarian cancer (SOCceR trial): A multicenter randomised controlled study. BMC Cancer 14:22.
Van den Eynde, B. J., and T. Boon. 1997. Tumor antigens recognized by T lymphocytes. International Journal of Clinical and Laboratory Research 27(2):81-86.
van der Bruggen, P., C. Traversari, P. Chomez, C. Lurquin, E. De Plaen, B. Van den Eynde, A. Knuth, and T. Boon. 1991. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 254(5038):1643-1647.
Vang, R., M. Shih Ie, and R. J. Kurman. 2009. Ovarian low-grade and high-grade serous carcinoma: Pathogenesis, clinicopathologic and molecular biologic features, and diagnostic problems. Advances in Anatomic Pathology 16(5):267-282.
Varga, A., S. A. Piha-Paul, P. A. Ott, J. M. Mehnert, D. Berton-Rigaud, E. A. Johnson, J. D. Cheng, S. Yuan, E. H. Rubin, and D. E. Matei. 2015. Antitumor activity and safety of pembrolizumab in patients (pts) with PD-L1 positive advanced ovarian cancer: Interim results from a phase Ib study. Journal of Clinical Oncology 33(Suppl):abstract 5510.
Vaughan, S., J. I. Coward, R. C. Bast, Jr., A. Berchuck, J. S. Berek, et al. 2011. Rethinking ovarian cancer: Recommendations for improving outcomes. Nature Reviews: Cancer 11(10):719-725.
Vergote, I., C. G. Trope, F. Amant, G. B. Kristensen, T. Ehlen, et al. 2010. Neoadjuvant chemotherapy or primary surgery in stage IIIc or IV ovarian cancer. New England Journal of Medicine 363(10):943-953.
Vergote, I., A. du Bois, F. Amant, F. Heitz, K. Leunen, and P. Harter. 2013. Neoadjuvant chemotherapy in advanced ovarian cancer: On what do we agree and disagree? Gynecologic Oncology 128(1):6-11.
Verleye, L., P. B. Ottevanger, G. B. Kristensen, T. Ehlen, N. Johnson, M. E. van der Burg, N. S. Reed, R. H. Verheijen, K. N. Gaarenstroom, B. Mosgaard, J. M. Seoane, J. van der Velden, R. Lotocki, W. van der Graaf, B. Penninckx, C. Coens, G. Stuart, and I. Vergote. 2011. Quality of pathology reports for advanced ovarian cancer: Are we missing essential information? An audit of 479 pathology reports from the EORTC-GCG 55971/NCIC-CTG OV13 neoadjuvant trial. European Journal of Cancer 47(1):57-64.
Vernooij, F., P. Heintz, E. Witteveen, and Y. van der Graaf. 2007. The outcomes of ovarian cancer treatment are better when provided by gynecologic oncologists and in specialized hospitals: A systematic review. Gynecologic Oncology 105(3):801-812.
Wagner, U., C. Marth, R. Largillier, J. Kaern, C. Brown, M. Heywood, T. Bonaventura, I. Vergote, M. C. Piccirillo, R. Fossati, V. Gebski, and E. P. Lauraine. 2012. Final overall survival results of phase III GCIG CALYPSO trial of pegylated liposomal doxorubicin and carboplatin vs paclitaxel and carboplatin in platinum-sensitive ovarian cancer patients. British Journal of Cancer 107(4):588-591.
Ward, B. G., S. J. Mather, L. R. Hawkins, M. E. Crowther, J. H. Shepherd, M. Granowska, K. E. Britton, and M. L. Slevin. 1987. Localization of radioiodine conjugated to the monoclonal antibody HMFG2 in human ovarian carcinoma: Assessment of intravenous and intraperitoneal routes of administration. Cancer Research 47(17):4719-4723.
Washam, C. 2012. How obesity complicates cancer treatment. Oncology Times 34(4):15-17.
Wehland, M., J. Bauer, N. E. Magnusson, M. Infanger, and D. Grimm. 2013. Biomarkers for anti-angiogenic therapy in cancer. International Journal of Molecular Sciences 14(5):9338-9364.
Weisberger, A. S., B. Levine, and J. P. Storaasli. 1955. Use of nitrogen mustard in treatment of serous effusions of neoplastic origin. Journal of the American Medical Association 159(18):1704-1707.
Welsh, J., J. Thomas, D. Shah, P. K. Allen, X. Wei, K. Mitchell, S. Gao, P. Palter, R. Komaki, J. Y. Chang. 2011. Obesity increases the risk of chest wall pain from thoracic stereotactic body radiation therapy. International Journal of Radiation Oncology*Biology*Physics 81(1):91-96.
Wick, D. A., J. R. Webb, J. S. Nielsen, S. D. Martin, D. R. Kroeger, K. Milne, M. Castellarin, K. Twumasi-Boateng, P. H. Watson, R. A. Holt, and B. H. Nelson. 2014. Surveillance of the tumor mutanome by T cells during progression from primary to recurrent ovarian cancer. Clinical Cancer Research 20(5):1125-1134.
Wimberger, P., N. Lehmann, R. Kimmig, A. Burges, W. Meier, A. Du Bois, and G. Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group. 2007. Prognostic factors for complete debulking in advanced ovarian cancer and its impact on survival. An exploratory analysis of a prospectively randomized phase III study of the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group (AGO-OVAR). Gynecologic Oncology 106(1):69-74.
Winter-Roach, B. A., H. C. Kitchener, and T. A. Lawrie. 2012. Adjuvant (post-surgery) chemotherapy for early stage epithelial ovarian cancer. Cochrane Database of Systematic Reviews 3:CD004706.
Wolchok, J. D., A. Hoos, S. O—Day, J. S. Weber, O. Hamid, C. Lebbe, M. Maio, M. Binder, O. Bohnsack, G. Nichol, R. Humphrey, and F. S. Hodi. 2009. Guidelines for the evaluation of immune therapy activity in solid tumors: Immune-related response criteria. Clinical Cancer Research 15(23):7412-7420.
Wood, A. J., T. W. Lo, B. Zeitler, C. S. Pickle, E. J. Ralston, A. H. Lee, R. Amora, J. C. Miller, E. Leung, X. Meng, L. Zhang, E. J. Rebar, P. D. Gregory, F. D. Urnov, and B. J. Meyer. 2011. Targeted genome editing across species using ZFNs and TALENs. Science 333(6040):307.
Wright, A. A., A. Cronin, D. E. Milne, M. A. Bookman, R. A. Burger, D. E. Cohn, M. C. Cristea, J. J. Griggs, N. L. Keating, C. F. Levenback, G. Mantia-Smaldone, U. A. Matulonis, L. A. Meyer, J. C. Niland, J. C. Weeks, and D. M. O—Malley. 2015. Use and effectiveness of intraperitoneal chemotherapy for treatment of ovarian cancer. Journal of Clinical Oncology 33(26):2841-2847.
Yeon, C. H., and M. D. Pegram. 2005. Anti-erbB-2 antibody trastuzumab in the treatment of HER2-amplified breast cancer. Investigational New Drugs 23(5):391-409.
Yuan, Y., and G. Yin. 2011. On the usefulness of outcome-adaptive randomization. Journal of Clinical Oncology 29(13):e390-392; author reply e393.
Zhang, L., J. R. Conejo-Garcia, D. Katsaros, P. A. Gimotty, M. Massobrio, G. Regnani, A. Makrigiannakis, H. Gray, K. Schlienger, M. N. Liebman, S. C. Rubin, and G. Coukos. 2003. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. New England Journal of Medicine 348(3):203-213.


































































