2
Emerging Insights (Physiological)
DIETARY NEEDS IN AGING POPULATIONS
Katherine Tucker, Professor of Nutritional Epidemiology at the University of Massachusetts Lowell, opened her presentation on dietary needs in aging populations by saying that she would focus her talk on a brief review of many of the vitamins and minerals of concern. She noted that diet and exercise are the major things that will help everyone live active and healthy lives without chronic diseases.
Tucker opened her review by noting changing dietary needs that occur in aging, including
- Lower energy requirements resulting from a slower metabolism,
- Less efficient absorption and utilization of many nutrients, and
- Chronic conditions and medication that affect nutrient requirements.
She then listed a number of specific age-related changes that influence nutrient requirements to reinforce the point that a major nutrition goal for older adults is to achieve a nutrient-dense diet. At the same time that older adults have higher nutrient requirements, they are dealing with issues that can make it harder to choose the right foods and to eat enough of them. Many older people experience loss of appetite, changes in taste and smell, problems with eating, dentures and oral health issues, mobility constraints, and income constraints.
Tucker then reviewed specific nutrients of particular importance to older adults (see Box 2-1). Tucker summed up this review by stating that
overall, older adults are not getting enough of a wide spectrum of nutrients. Each nutrient has its own functions and many of them protect against chronic conditions of great concern, such as heart disease, osteoporosis, diabetes, and cognitive decline.
Relationship Between the Quality of the Overall Dietary Pattern and Health
An emerging trend in nutritional science is to look at the quality of the overall dietary pattern and its relationships to health and disease outcomes. For example, scores on the Mediterranean Diet Score, a measure of adherence to the Mediterranean diet, have a very strong linear relationship with many mental or cognitive functions. Scores on the Healthy Eating Index, an index developed by the U.S. Department of Agriculture (USDA) that is a measure of adherence to Dietary Guidelines for Americans recommendations, also have a strong relationship with cognitive functions. The message here, noted Tucker, is that we need to think about the whole diet in order to ensure sufficient intakes of all of these nutrients.
Tucker then summarized healthy diet recommendations for older adults:
- First and foremost, fruits and vegetables must play a central role in the diet.
- More nuts and legumes for proteins, vitamin B6, magnesium, and dietary fiber.
- More fish.
- More low-fat dairy products, which provide protein, magnesium, calcium, potassium, vitamin B12, and vitamin D.
- More whole grains, which contribute vitamin B6, magnesium, and dietary fiber. In some cases, fortified breakfast cereals are a good source of vitamin B12.
- Fewer calories1 and limited refined foods and foods high in sugars, solid fats, and sodium.
AGING, BODY MASS INDEX, AND MORTALITY OUTCOMES
Gordon Jensen, Senior Associate Dean for Research, Professor of Medicine and Nutrition, University of Vermont, and Chair of the Planning Committee on Meeting the Dietary Needs of Older Adults, began his talk by saying that he has become very interested in how body mass index (BMI) relates to health and mortality outcomes in older adults. He stated that one focus of his talk would be to examine whether, within this framework, obesity has any benefits. Most importantly, he noted that he would be talking about the effects of confounding variables.
Research on the Effects of Body Weight on Overall Health
How does body weight affect health? Jensen explained that obesity clearly does affect the duration of life through its association with premature disease, chronic diseases, and functional decline, especially mobility limitations. At the other end of the spectrum, the Nurses’ Health Study found the lowest mortality in those weighing 15 percent below the national average (Manson et al., 1995). Jensen added that the National Institute on Aging (NIA) has been very interested in studying dietary energy restriction and metabolic outcomes and ultimately longevity.
Early studies looking at BMI and mortality outcomes were confounded by smoking and disease burden, explained Jensen. In general, however, there is a J-shaped mortality curve in relation to BMI, with elevated mortality at either end of the BMI spectrum. Obesity is associated with mortality and life expectancy, and importantly, it is a much stronger predictor of mortality at younger ages (i.e., young adults and middle-aged adults). Some recent projections have suggested that because of the current epidemic of obesity, today’s children may not ultimately live as long as projected for current adults.
Obesity also is related to a host of significant comorbidities, including cardiovascular disease, hypertension, sleep apnea, diabetes, dyslipidemia, metabolic syndrome, some cancers, osteoarthritis, gout, and other disorders. Jensen added that historically, health care focused on underweight,
___________________
1 This may not apply to all older adults. Some older adults may be advised by their health care professional to consume more calories.
frail older adults. Those who work with older people today, whether in the community, acute care, or skilled nursing facilities, know that that has changed and that there are many obese frail older persons. This obesity may be associated with profound functional decline, even to the point of becoming homebound.
BMI and Mortality
Continuing, Jensen stated that a number of papers in the scientific literature have looked at the relationship between BMI and mortality outcomes in older persons. One study examined cause-specific excess deaths associated with underweight, overweight, and obesity (Flegal et al., 2007). Using National Health and Nutrition Examination Survey (NHANES) data combined with vital statistics data, these analyses found that the BMI-mortality association varied by cause of death:
- 6,859 persons with 571,042 person years follow-up
- BMI <18.5—more noncancer, noncardiovascular disease deaths
- BMI 25-29.9—fewer noncancer, noncardiovascular disease deaths
- BMI ≥30—more cardiovascular disease deaths, more cancers considered to be obesity-related
Jensen then described another study conducted in 2007 of more than 2,000 older adults who underwent exercise tolerance testing on a treadmill. BMI and body composition were measured and the outcome was 12-year follow-up all-cause mortality (Sui et al., 2007). The key finding was that fitness was a significant mortality predictor independent of adiposity.
Being fit had a dramatic mortality benefit, explained Jensen, and it is likely related to disease status and body composition, particularly lean body mass.
In a 2008 study, Wildman and colleagues used NHANES data to categorize individuals according to their metabolic risk (e.g., hypertension, lipids, fasting glucose, C-reactive proteins) (Wildman et al., 2008). The investigators found that half of the overweight and one-third of the obese were metabolically healthy while 23.5 percent of the normal weight individuals had abnormal metabolic indicators. The conclusion, noted Jensen, is that it may not be “all or nothing.” A person can be overweight or moderately obese and be metabolically healthy or be underweight and metabolically unhealthy.
The “Obesity Paradox”
Jensen explained that the data he presented were a backdrop to the “obesity paradox” in older persons. Oreopoulos et al. (2009) highlighted that in the elderly, obesity is paradoxically associated with a lower, not a higher, mortality risk. An interesting 2009 study of hospitalized older adults found that fat mass was associated with reduced mortality and complications, and there was no relationship with lean mass or appendicular muscle mass (Bouillanne et al., 2009). Jensen then showed another analysis, published in 2013, which he said triggered considerable controversy (Flegal et al., 2013). This was a systematic review and meta-analysis of 97 studies, with nearly 3,000,000 individuals and 270,000 deaths. The analysis found that compared to the desirable National Institutes of Health (NIH) BMI category (i.e., 18.5 to 24.9), those with class 2 and 3 obesity had significantly greater all-cause mortality, while class 1 obesity was not associated with higher mortality. Moreover, overweight was associated with significantly lower mortality. Another way of putting this, noted Jensen, is that the lowest mortality for older persons shifts toward higher BMI with increasing age (Childers and Allison, 2010). The lowest mortality appears not to be in the desirable BMI range (see Figure 2-1). Jensen noted that a host of studies have now shown similar results (e.g., Ford et al., 2014).
Communicating Research Findings to the Public and Practitioners
Jensen raised an important concern: How to appropriately and accurately communicate the meaning of these findings to the public and the practitioners. A central factor in these findings, stated Jensen, appears to be body composition. It appears that overweight or obesity may be protective only in patients with intact lean body mass. He noted that reduced lean body mass is likely to be a proxy for chronic disease, inflammation, and relative immobility. A study from an NIA research group showed the highest mortality in those with low muscle mass in both normal and overweight study participants (Murphy et al., 2014). In addition, the normal weight participants had an elevated mortality risk compared to those who were overweight. This paradox was partly mediated by muscle size.
A variety of potential benefits have been attributed to obesity, including reduced osteoporosis, better survival in heart failure, myocardial infarction, peripheral vascular disease surgery, and nonbariatric general surgery. However, Jensen went on to state that, when confounding factors are adequately controlled, most of these advantages of being obese disappear. Is the researcher talking about individuals or a population? How is body composition measured? What measures of adiposity are used? Have age, gender, race/ethnicity, disease burden, and smoking been considered?
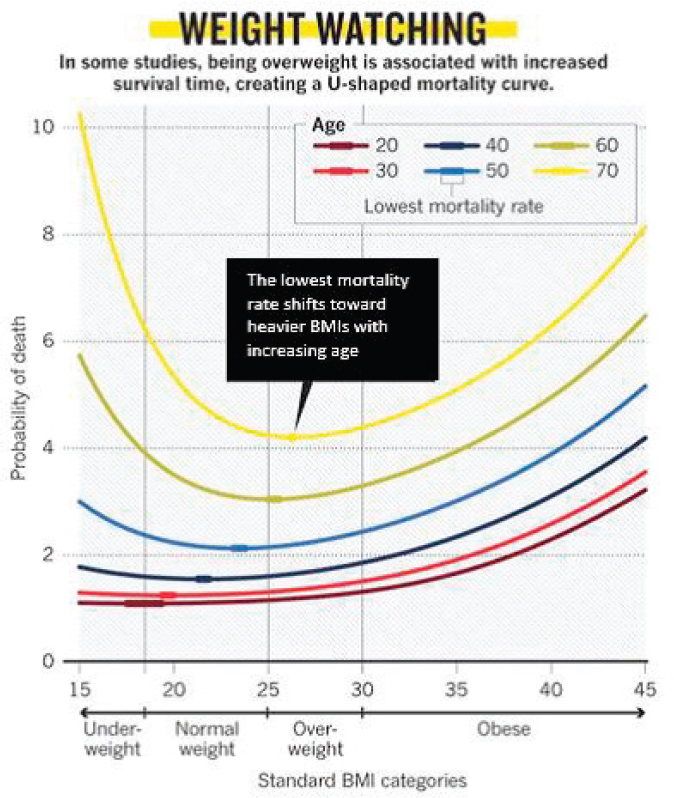
NOTE: BMI = body mass index.
SOURCES: Presented by Gordon Jensen on October 28, 2015 (adapted from Childers and Allison, 2010).
Jensen concluded his talk with several summary points:
- Obesity does not generally confer mortality or health benefits.
- The current use of the NIH BMI guidelines warrant reevaluation for older adults. Jensen noted that it is not unusual for staff in skilled nursing facilities to tell overweight older adults that they need to lose weight when, for many, that may be the wrong advice.
- Disease burden and body composition are likely key factors in the relationship between BMI and mortality in older adults.
- Now that we are on the verge of better understanding the obesity paradox in older adults, Jensen said, there is a need to determine how best to interpret these findings for health professionals and for the public.
MUSCLE HEALTH: PROTEIN
Wayne Campbell, Professor in the Department of Nutrition Science at Purdue University, began his talk on muscle health and protein by encouraging workshop participants to recognize that dietary recommendations cannot be made based on a single organ. His talk would be focused on muscle, but recommendations are intended for a whole body response not just for muscle.
The Recommended Dietary Allowance (RDA) for protein established by the Institute of Medicine (IOM) in the early 2000s is 0.8 g/kg for all adults, both men and women, young and old (IOM, 2005). Recommendations for higher protein intake (25 percent and 50 percent higher than the RDA) have been made by other groups (e.g., American College for Sports Medicine, PROT-AGE Study Group) for athletes and older individuals.
Origins of Current RDAs for Protein Intake
To understand the current recommendation, Campbell stated that it is important to realize that the current RDAs are not based on any functional outcome but on nitrogen balance data. The next question then is whether these data apply to older adults. Campbell explained that the largest study, which was of the nitrogen balance type, confirmed that there are no apparent differences in protein requirements for young men and young women versus older men and older women and that the data are largely consistent with the current Estimated Average Requirement (EAR) (Campbell et al., 2008). An alternative way of looking at these data, however, is based not so much on new data as much as different mathematics. Campbell noted that Professor Rand, the author of the original analyses used for the current RDAs, stated that a biologically more realistic equation may be a better approach (Rand et al., 2003). Using a biologically realistic approach from essentially the same data results in an EAR about 50 percent higher. The RDA therefore goes up by about 25 percent simply by the mathematics.
But, noted Campbell, none of this is physiological. New data are emerging from an indicator of amino acid oxidation intake. This work, which has been done initially in young men, involved feeding people differing amounts of amino acids and determining that the amount of protein
intake that meets their need for synthesizing proteins and limiting oxidation of proteins is about 0.93 g/kg (Humayun et al., 2007). Nitrogen balance and isotope kinetics data consistently show that aging does not affect daily dietary protein needs, but these newer data bring into question the accuracy of the current EAR and RDA for adults of all ages. This idea, Campbell stated, is often lost in the discussion regarding protein intake recommendations.
Campbell continued by saying that age-associated differences may occur specifically with muscle. He showed data from a recent analyses of acute feeding studies where the investigators were looking at the amount of protein in a meal versus the rate at which proteins were synthesized in skeletal muscle (Campbell et al., 2008). They found that, compared to younger adults, older adults required greater single meal protein intakes to maximize muscle protein synthesis. These data show that there is no effect of aging on the maximum rate of protein synthesis and muscles, but that it might take an older adult a little more protein in the meal to get there, explained Campbell.
Alternative Considerations for Assessing Protein Requirements
Campbell then noted that current recommendations are based on how to prevent deficiencies and inadequacies but not necessarily how to optimize functional outcomes, such as health-related outcomes, balance, gait, strength and endurance, and ability to carry out activities of daily living. Those outcomes can relate to body composition and muscle size and strength.
Investigators have looked at the genetic profiles of skeletal muscle of younger and older men and found amounts of protein that span the range of adequacy, from 0.5 g to 0.75 g, which is very close to the RDA or 25 percent higher than the RDA, at 1 g (Thalacker-Mercer et al., 2010). The results from a cluster of 159 gene transcripts that are associated with protein catabolism or breakdown show that the expression of those transcripts goes up markedly when a person eats an inadequate amount of protein.
The body catabolizes muscle when it does not have enough of a substrate to draw on. In contrast, a higher protein diet promotes expression of genes related to stress responses, stimuli responses, muscle function, and organ development. Data show that achieving the RDA for protein prevents deficiency but is not necessarily an optimal amount for functional outcomes such as body composition, muscle size, strength, balance, stress response, and weight-loss. Data from a study of postmenopausal women indicate that eating about 56 percent of the RDA for protein results in a loss of lean body mass, muscle, muscle size, and strength (Castaneda et al.,
1995, 2000). One could conclude, stated Campbell, that inadequate protein intake will compromise muscle function.
Campbell then presented data from a 14-week controlled feeding metabolic balance study (Campbell et al., 2001, 2002). All the participants consumed the RDA for protein. Some of the participants were sedentary and others performed resistance exercise training. Results showed that when all of the participants met the RDA while maintaining energy balance, they did not lose weight; they gained body fat and lost fat-free mass. While consuming the RDA for protein, the sedentary participants lost skeletal muscle. However, this level of protein intake did not prevent those participants who were performing resistance training to actually increase the muscles that were trained.
Campbell also presented data from a prospective study of several thousand older adults, which concluded that participants in the highest quintile of protein intake lost 40 percent less lean mass and appendicular lean mass than did those in the lowest quintile of protein intake (Houston et al., 2008). It could be, noted Campbell, that those who were eating the lowest quintiles were actually eating less than the RDA levels. So while more protein might be better, the data also show that inadequate protein is not enough to maintain lean mass and appendicular lean mass. Indeed, Campbell continued, protein intake had no effect on changes in appendicular lean mass for the participants who were weight stable. It was only when participants were stressed with an energy deficit and were eating below the RDA that they actually were losing muscle. In contrast, participants who were gaining weight, were gaining lean mass only when they were at the highest quintile of protein intake (1.1 g/kg).
Campbell then showed data from his lab that looked at weight loss in older men and women (Kim et al., 2016). He noted that on average, as older adults lose weight, about 75 percent comes from fat mass and about 25 percent comes from lean mass. The question is, is it possible to influence that change in body composition by changing the amount of protein that is consumed? His data show that people who were eating protein at about the RDA were actually losing about 30 percent of their lean mass, whereas people who were eating about 1.4 or 1.5 g/kg actually cut their lean mass loss by one-third. Additional data support this finding that protein intake above 1 g/kg influences body composition responses to weight loss.
Protein Requirements and Exercise
Campbell presented data from a compilation of a number of studies that his group has conducted over the years. These studies have looked at how much protein older men and women were eating chronically while they performed 12 weeks of resistance training (Campbell and Leidy, 2007).
What his group observed was that at intakes of anything less than about 1 g/kg, changes in fat-free mass, at least on the linear scale, were neutral or negative. People needed to be consuming more than 1 g/kg to get a net anabolic response. He also reported on a newly published study from his research group, which assessed the effects of protein supplementation on changes in body composition and metabolic syndrome indexes in overweight and obese middle-aged adults who performed exercise training for 9 months (Campbell et al., 2015). They observed that when total protein intakes were less than 1 g/kg, the participants actually gained weight. The groups who consumed different amounts of protein did not differ in their capacity to respond to the exercise from a muscle enlargement perspective, but in their body fat. Campbell added that the study showed no advantage to eating more than 1.2 g/kg of protein compared to 1.0 to 1.2 g/kg.
Challenges in Developing Protein Intake Recommendations
With his last few minutes, Campbell stated that he would like to focus on the challenges of establishing recommendations for protein intake for older adults. For example, several groups over the past couple of years have gathered scientific experts in the field to look at issues related to protein needs and overall health. The PROT-AGE Study Group (Bauer et al., 2013) and the European Food Safety Authority (EFSA, 2012) looked at essentially the same data, that is, outcomes related to muscle in body composition, insulin sensitivity, and bone mineral density, but they approached the conclusions differently. The PROT-AGE group recommended that protein intakes greater than the RDA would lower the risk of frailty and improve muscle mass and function while the European Food Safety Authority found that the data were not scientifically strong enough to change the current recommendations. Campbell concluded that these differing conclusions are a good indication of the current state of the field.
Using a recent review article on protein intake and the effects on muscle in hospitalized older adults, Campbell quoted the authors’ conclusion that “focused efforts on using nutrition to maintain muscle health during hospitalization of older adults should be geared toward the quantity (30 g high-quality protein; 3 g leucine) and quality of protein (fast absorbing, high leucine content), equality meal protein distribution (30:30:30) and timing of protein supplementation (between meals)” (Thalacker-Mercer and Drummond, 2014). Indeed, noted Campbell, the scientific foundation for that recommendation is that the amount of protein in a meal is going to determine a person’s responses to synthesizing protein in muscles. Theoretically, different amounts of protein at each meal would result in different amounts of protein synthesis at those times. Rebalancing protein intake and including a protein supplement would maximize muscle protein
synthesis throughout the day. However, Campbell noted that the evidence at this stage is still mixed and it is not known whether redistributing protein throughout the day will help to maintain muscle mass in older adults.
Campbell concluded his presentation by stating that the current RDA for protein, which is based on short-term nitrogen balance studies, prevents aggressive muscle catabolism in older adults, but does not maximally promote muscle anabolism. He added that protein metabolism, muscle health, and functional outcomes data that are slowly becoming available should be critically and systematically evaluated to update the Dietary Reference Intakes (DRIs) for protein. Finally, Campbell noted, enthusiasm for prescriptive use of protein to promote the retention of muscle mass and function in older adults must be supported by high-quality longitudinal research, not just theories, hypotheses, or extrapolations from short-term studies.
AGING, VITAMIN D, AND PHYSICAL FUNCTION
Denise Houston, Associate Professor in the Department of Internal Medicine, Section on Gerontology and Geriatric Medicine at the Wake Forest School of Medicine, opened her presentation by explaining that she would focus on the relationship of vitamin D and physical function although she recognized that vitamin D affects many other health outcomes. It has long been acknowledged that vitamin D plays an important role in intestinal calcium and phosphorus absorption as well as skeletal health. Over the past two decades, a number of studies also have shown that vitamin D is associated with an assortment of different outcomes in many conditions that are prevalent in older adults, such as depression, high blood pressure, cardiovascular disease, diabetes, muscle weakness, osteoporosis, and osteoarthritis, and some cancers. Vitamin D’s association with the majority of these health outcomes has been observed in observational studies, with the exception of osteoporosis and skeletal health.
DRIs for Vitamin D and Current Intakes
In 2010, the IOM updated the DRIs for vitamin D and set the RDA based on a 25(OH)D (25 hydroxy vitamin D) concentration of 20 ng/mL. The RDA was set at 600 International Units (IU)/day for adults ages 51 to 70 and 800 IU/day for adults ages 71 and older (IOM, 2010). The IOM committee’s recommendations were based on bone outcomes, as few randomized controlled studies had been conducted on other health outcomes, and thus evidence was insufficient to suggest that other intake levels would be necessary for other health outcomes.
Using the 20 ng/mL cut-point, Houston showed a pie chart with the
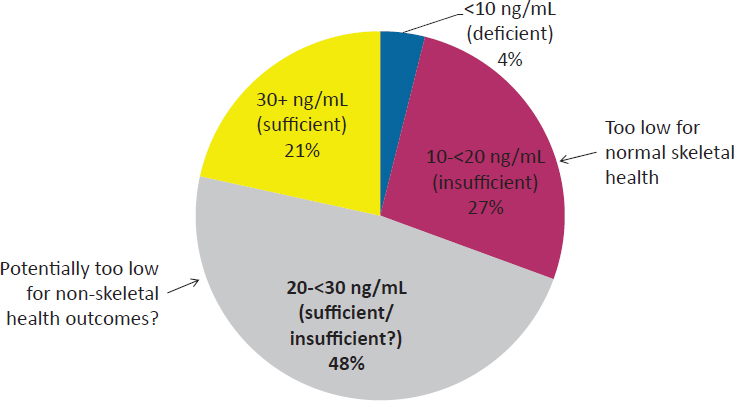
SOURCES: Presented by Denise Houston on October 28, 2015 (NHANES 2000-2004; Looker et al., 2008).
prevalence of vitamin D deficiency and insufficiency among older adults in the United States in 2000-2004 (Looker et al., 2008) (see Figure 2-2).
About one-third of older adults have vitamin D concentrations that are either insufficient or deficient (<20 ng/mL). However, controversy exists about whether the 25(OH)D cut-point of 20 ng/mL is actually sufficient for nonbone health outcomes. In that case, an additional 50 percent of older adults may be at risk for vitamin D insufficiency. However, as noted above, data for randomized controlled trials are not currently available to establish whether higher amounts are needed.
Some risk factors for vitamin D deficiency and insufficiency are specific to older adults as well as common in the population. Decreased exposure to sunlight is a problem in homebound and institutionalized older adults. A second problem is that older adults have a decreased capacity of the skin to produce vitamin D. Compared to younger adults, older adults produce about one quarter the amount of vitamin D with exposure to the same amount of sunlight. Third, inadequate dietary intake of vitamin D is common among all adults, primarily because it is found naturally in few foods. Fortified foods are the major source of vitamin D in the United States.
The most recent NHANES data on vitamin D intake from 2005-2006 show the amount of vitamin D coming from diet or food sources, and from supplements (Bailey et al., 2010). In both men and women across the adult age categories, approximately 200 IU/day of vitamin D comes from the diet.
Vitamin D coming from supplements is higher among older age groups. The average intake of vitamin D from both diet and supplements in adults older than age 70 is only about half the current RDA.
Vitamin D and Physical Function
Houston discussed the potential roles of vitamin D and physical function. Direct effects on physical function include muscle contraction, muscle cell proliferation and differentiation, and muscle growth. Vitamin D may affect muscle fiber composition by increasing type II muscle fibers which may account for the association between vitamin D and fall risk. As far as some of the indirect pathways, Houston continued, vitamin D has a role in neuromodulation and has been associated with cognitive function and, in particular, executive function, which also have been shown to be associated with physical function. Vitamin D has been shown to play a role in the inflammatory response as well. In addition, vitamin D has been associated with many of the same chronic conditions that are related to the development of functional limitations, such as diabetes, heart disease, hypertension, and osteoarthritis.
This raises the question, noted Houston, of whether 25(OH)D concentrations that are sufficient to maintain bone health are adequate for optimal physical function in older adults. One of the largest original studies that examined this question used data from NHANES 2000-2004 (Bischoff-Ferrari et al., 2004). It showed that older adults with lower 25(OH)D concentrations had lower walk speeds and lower sit-to-stand times (see Figure 2-3). Most of the difference comes in a range below the 20 ng/mL cut-point.
Other studies, such as the Health Aging, Body, and Composition (ABC) Study, looking at this same question showed that older adults with low 25(OH)D concentrations had lower physical performance scores and slower gait speeds (Houston et al., 2012). Houston noted that there appears to be a dose–response relationship, suggesting a possible advantage for physical function of having higher 25(OH)D. Similar to other studies, data from the Cardiovascular Health Study All-Stars Study show an association between low 25(OH)D concentrations and weaker grip strength (Houston et al., 2011). Somewhat of a dose response also occurred in this study, suggesting that higher concentrations may be optimal for function.
These results lead to the question of whether a 25(OH)D concentration threshold for physical function exists. Houston explained that some investigators have suggested that a concentration of 30 to 32 ng/mL may be optimal for various health outcomes (Bischoff-Ferrari et al., 2006). Early data from NHANES III show that most of the increase in walk speed and sit-to-stand time occurred in individuals with 25(OH)D concentrations between 9 and 16 ng/mL (Bischoff-Ferrari et al., 2004). Other studies have

SOURCES: Presented by Denise Houston on October 28, 2015 (adapted from Bischoff-Ferrari et al., 2004).
shown a threshold at higher 25(OH)D concentrations (Houston et al., 2012; Janssen et al., 2013; Sohl et al., 2013, 2015; Wicherts et al., 2007). Data from several cohorts from The Netherlands showed a threshold of 26 ng/mL for the short physical performance battery, which includes balance, short walks distances, and repeated chair stands (Sohl et al., 2013). They found similar results for grip strength but not knee extension strength. Houston stated that her group applied piecewise regression models to the Health ABC cohort to determine 25(OH)D thresholds across different aspects of physical performance (Houston et al., 2012). The group found that the thresholds ranged from 26 ng/mL for grip strength to 33 ng/mL for the 20 m gait speed. At concentrations below these thresholds, they found significant positive associations between 25(OH)D levels and these performance measures. However, above these concentrations, they found no association between 25(OH)D levels and physical performance and strength measures, suggesting no additional benefit of having 25(OH)D levels above the thresholds. They did not observe a 25(OH)D threshold for knee strength. Houston summarized by saying that data from the Health ABC and other studies suggest that the 25(OH)D concentration threshold for physical performance and muscle strength is between 24 and 32 ng/mL.
Houston then addressed the question of whether low 25(OH)D concentrations are associated with greater decline in physical function in older adults. She noted that longitudinal studies are mixed, showing both no effect (Bartali et al., 2008; Faulkner et al., 2006; Houston et al., 2012; Michael et al., 2011; Verreault et al., 2002) and greater declines in muscle strength and physical performance with deficient/insufficient 25(OH)D concentrations (Dam et al., 2009; Sohl et al., 2013; Visser et al., 2003; Wicherts et al., 2007). Data from the Health ABC study show that older adults with low 25(OH)D concentrations had poorer physical performance over a 4-year follow-up period, but lower concentrations were not associated with a faster rate of decline (Houston et al., 2012). In contrast, data from the Rancho Bernardo study showed that women in the lowest quartile of 25(OH)D concentration had an accelerated rate of decline in physical function (Dam et al., 2009). Importantly, noted Houston, the lowest quartile started at less than 32 ng/mL, which is a very replete vitamin D population. Houston noted that some possible explanations for the inconsistent results from the observational studies are differences in study populations, different methods to measure serum 25(OH)D and physical performance, different cut-points used to define 25(OH)D deficiency/insufficiency, differences in adjustment for potential confounders, and differences in duration of follow-up.
Houston then posed the question of whether 25(OH)D status alters the disability trajectory. Data from the Health ABC study show no difference in the rate of decline. However, if people with lower 25(OH)D concentrations start out having worse performance, then potentially they will cross the disability threshold earlier in life than those who have higher 25(OH)D concentrations. In Health ABC, those who had vitamin D insufficiency, whether using a cut-point of 20 or 30 ng/mL, had about a 30 percent greater risk of developing mobility limitations over 3 years of follow-up (Houston et al., 2013). Mobility limitation was assessed every 6 months based on self-report of having difficulty walking a quarter-mile or climbing a flight of stairs.
The next question Houston considered was whether vitamin D supplements improve physical function. She noted that results of randomized controlled trials are inconsistent, showing both no effect (Brunner et al., 2008; Kenny et al., 2003; Lips et al., 2010) as well as improvements in muscle strength and physical performance (Bischoff et al., 2003; Bunout et al., 2006; Dhesi et al., 2004; Pfeifer et al., 2000, 2009). A 2011 meta-analysis on the effects of vitamin D supplementation on balance or body sway showed about a 20 percent improvement in those who took vitamin D compared to a placebo (Muir and Montero-Odasso, 2011). No difference was apparent for muscle strength. In a subset of the Women’s Health Initiative randomized controlled trial of calcium (1,000 mg/d) and vitamin
D (400 IU/d), vitamin D supplementation was not found to be beneficial in protecting older women against declines in physical performance or strength over a 7-year follow-up (Brunner et al., 2008). Possible explanations for the inconsistent results in randomized controlled trials include small sample sizes, insufficient vitamin D dose, or type and frequency of vitamin D supplementation.
Potential Knowledge Gaps and Research Priorities
Houston summarized the following knowledge gaps and research priorities:
- Observational data suggest that 25(OH)D concentrations greater than those needed for skeletal health may be optimal for nonskeletal outcomes. But questions remain:
- Is 25(OH)D an appropriate biomarker?
- How do physiological factors (e.g., age, BMI) affect the 25(OH)D response to dietary intake or endogenous production of vitamin D?
- Are similar associations observed in minorities?
- Will increasing 25(OH)D concentrations above the current IOM cut-point of 20 ng/mL improve physical function?
- Convincing evidence for nonskeletal health outcomes from sufficiently powered randomized controlled trials is lacking.
- Large randomized controlled trials under way include primary outcomes related to physical function (DO-HEALTH, n = 2,158; STURDY, n = ~1,200; EVIDENCE, n = ~200), as well as secondary outcomes related to physical function (VITAL, n = 25,874).
BRAIN HEALTH: B VITAMINS AND OMEGA-3 FATTY ACIDS
Irwin Rosenberg, Senior Scientist at the Jean Mayer USDA Human Nutrition Research Center on Aging, opened his talk by saying that he welcomed the opportunity to discuss multimicronutrient and nutrient interactions. He stated that he hoped he could persuade workshop participants that some of these interactions may be even more important than the effects of individual nutrients alone.
August D, the “poster child of Alzheimer’s disease,” was the subject of Alois Alzheimer’s 1907 report in which he described the pathology of pre-senile dementia, focusing on never-before-described depositions of plaques and tangles in the brain of the seriously demented woman. The rediscovery of this study six decades later changed the entire nomenclature about senile dementia or pre-senile dementia, which had been associated with hardening
of the arteries. In fact, Rosenberg noted, he considers that relationship to the vascular system appropriate.
Rosenberg then showed a recent article published by colleagues from Oxford that reported an association between omega-3 fatty acids and B vitamins status. In this randomized controlled trial, a B vitamin intervention had a beneficial effect on the cognitively impaired elderly and to some extent on atrophy. In a retrospective analysis, this effect was particularly observed in people who had a deficiency of long chain fatty acids (Jernerén et al., 2015). Rosenberg noted that these results suggest interactions between nutrients, and that some of those interactions involve the vascular system.
He went on to illustrate the dimensions of the problem of cognitive impairment by showing a graph of the growth in the number of people with dementia in low-, middle-, and high-income countries (see Figure 2-4).
To find ways within nutrition and related activities to decrease the risk and decrease the trajectory of important problems related to aging and chronic disease could make an enormous impact on public health, Rosenberg said.
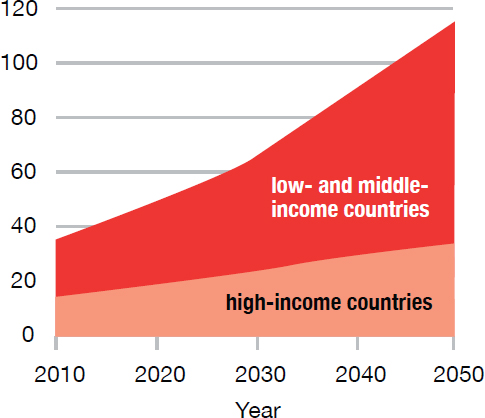
SOURCES: Presented by Irwin Rosenberg on October 28, 2015 (Alzheimer’s Disease International, 2010).
Vitamin B Metabolism, Homocysteine, and Risk of Dementia
Rosenberg pointed out that homocysteine is at a key juncture that influences at least three B vitamins, including vitamin B6, and vitamin B12. He then presented data from the Framingham Study showing that individuals with homocysteine levels in the highest quartile had a higher cumulative incidence of dementia over more than a decade. Individuals with levels in the lower quartiles had a significantly lower rate of onset of incident dementia. These data suggest that homocysteine is a risk factor for dementia, although Rosenberg noted, whether it is a factor directly in the pathogenesis, or as a reflection of its integration of B vitamin metabolism continues to be a matter for discussion. Rosenberg then showed data from the Nutrition, Aging, and Memory in the Elderly (NAME) Study that Tucker referred to earlier, pointing out that the data show a clear decline in cognitive function with higher homocysteine levels. However, this relationship is seen only in individuals with evidence of cerebrovascular disease on magnetic resonance imaging. Also, homocysteine concentration was not related to measures of memory or attention in either group (see Figure 2-5).
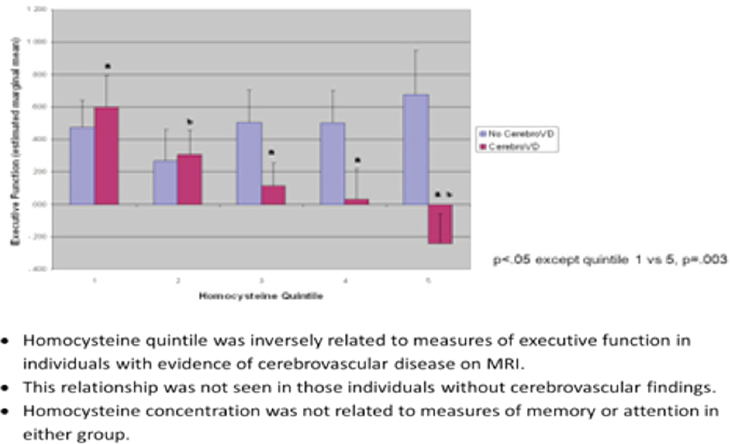
SOURCE: Presented by Irwin Rosenberg on October 28, 2015.
He noted that this observation points to an important role of the cerebrovascular system in cognitive decline and loss of brain health.
The Role of Lipids
The brain is particularly enriched with lipids as compared to the other organs, and these lipids play an essential role in neuronal plasticity, mitochondrial activity, and myelin integrity. Lipids also are important structural components of brain membranes and signaling molecules. Rosenberg called attention to one of the lipids in particular—phosphatidylcholine—which has a methyl group that is dependent on methylation reactions involving B vitamins.
Additional evidence of this relationship is shown in another study that examined red blood cell omega-3 fatty acid levels and markers of accelerated brain aging. Lower levels of fatty acids and high homocysteine were associated with similar white matter hyperintensities, which are believed to be associated with small vessel disease and to correlate with cognitive decline (Tan et al., 2012). Some evidence suggests that the process by which medium chain fatty acids are desaturated and elongated to be incorporated into the phospholipids of the brain is less efficient in older adults. He noted that this issue would be important as scientists look at the interaction of fatty acids and brain health, and reminded workshop participants that phospholipids are very important parts of key brain membranes, including the myelin sheath that surrounds neurons. Some work has been done to look at the ability of B vitamins or omega-3 fatty acids to affect the trajectory of cognitive loss of brain volume and loss of specific focus in memory. Studies have showed that multicomponent interventions had a greater effect than any of the individual interventions. Rosenberg concluded that we will be hearing this message increasingly in the coming years.
Rosenberg ended his talk by pointing out that higher levels of a nutrient are not always better and that dose–response effects and safety aspects should probably be considered much more in interventions. For example, considerable evidence indicates, at least in brain health, that there may be a U-shaped curve in which moderate folate intake may be helpful in decreasing the onset of disease of the brain but there may be an inflection point at which high folate intake could have an adverse effect. This may be a concern for older adults, noted Rosenberg, because considerable amounts of folic acid are being added to the food supply.
GUT HEALTH: DIETARY FIBER AND THE MICROBIOME
Lita Proctor is the Coordinator of the Human Microbiome Project for the NIH’s Common Fund. She opened her presentation by noting that the
microbiome is the subject of considerable public attention today, as well as confusion about its diverse roles in the human body. It is a young field that is growing fast. She explained that she would walk through the natural history of the microbiome to show how it develops in the body and the roles it plays throughout the lifespan. This set the stage for additional comments about the role of the microbiome in the older population.
The microbiome includes bacteria, fungi, and all manner of microscopic life that turn out to be essential to health. Humans acquire their genes from their parents and they acquire their microbiome anew each generation, much like human genes. To explain, Procter showed a cluster analysis in which investigators measured the mother’s microbiome in her mouth, vagina, and skin and then looked at the baby’s delivery mode—vaginal or cesarean birth. They found that babies born vaginally acquire their mother’s microbiome primarily from the vagina. In fact, over the baby’s 9-month gestation period, the microbiome of the pregnant woman’s vagina changes as it waits for the baby to pass through to receive this initial inoculation. Even when babies are born by cesarean delivery and do not come into any contact with the vaginal microbiome, they still acquire microbes primarily from the skin of their mothers and those (medical staff, family members) who handle the child. Over the first couple of years of life, microbes move in and colonize every epithelial surface of the baby (the gut lining, the skin, mouth, anus) and the microbiome continues to change and mature, becoming like the adult’s microbiome.
At the same time, a person’s immune system develops over the first few years of life. A baby first acquires passive immunity by receiving antibodies from the mother, and then develops adaptive immunity through producing antibodies themselves. This development of the microbiome and the immune system must occur together. Proctor explained that humans have co-evolved with the microbes that form the microbiome. The immune system cannot mature without specific bacteria. Some microbes play a critical role in maturation of the immune system because they induce pro-inflammatory responses to protect the body against invading pathogens. Other microbes induce host anti-inflammatory responses to restore immune system balance. The co-evolution of humans with their microbiome is not a random process. The gut has specific receptors that are required to be present in order for microbes to be able to colonize the human host. In fact, Proctor noted, some recent studies have demonstrated that human genetics play a major role in development of the microbiome. A study conducted in the United Kingdom comparing dizygotic twins against other members of the population found that the twins had a more similar microbiome than did other siblings pairs in the population (Goodrich et al., 2014).
The Microbiome’s Role in Regulating Human Health
Proctor then summarized the various ways in which microbiota and hosts interact to regulate human health. They:
- “Educate” the immune system to recognize self from nonself.
- Digest the “indigestibles” (e.g., plant material in diet, host cells, mucus).
- Produce energy substrates for host cells (e.g., short-chain fatty acids for epithelial cells).
- Metabolize drugs/detoxify drugs.
- Produce beneficial compounds (e.g., vitamins, antimicrobials).
- Produce signaling molecules that interact with the host.
- Communicate with the brain.
Proctor concluded this portion of her talk by emphasizing that although the human microbiome is a fixed feature, it is also a variable trait that changes between generations, throughout the lifespan and in disease versus health (see Figure 2-6). The mutability of the microbiome is the most important trait that can be exploited to support health and influence disease. It is the primary mediator between the body and the environment.
To illustrate this point, Proctor noted that diet is a very important environmental factor that regulates the microbiome. She showed data from the South Dakota Hutterites, a closed religious communal society that raises all of their own food (Davenport et al., 2014). For example, they eat fresh food in the summer and canned foods in the winter. An analysis of
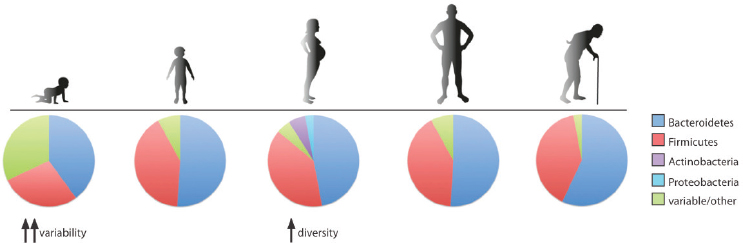
SOURCE: Presented by Lita Proctor on October 28, 2015 (NIH Human Microbiome Project).
their microbiome clearly shows how it changes with the seasons, reflecting changes in their diet between the seasons.
Older Adults and the Microbiome
After laying this foundation, Proctor then shifted to a discussion of the microbiome and older adults. As a point of discussion, she used the ELDERMET project, a University College Cork longitudinal cohort study that is measuring the microbiome of older adults in Ireland. Investigators are correlating the participants’ microbiome with health, diet, and lifestyle indicators to develop dietary recommendations in order to improve health and to provide evidence for future studies of health, environment, and the microbiome in the elderly. Proctor presented a cluster analysis from the ELDERMET study showing clear differences in the microbiomes of healthy older adults living freely in the community versus frail older adults living in nursing facilities (Claesson et al., 2012). In fact, Proctor pointed out, the microbiomes of the healthy community-dwelling older adults were similar to young healthy study participants, who served as controls for this study.
The microbiome could be an excellent marker for, or an integrator of, various aspects of health in the elderly, said Proctor. For example, the ELDERMET study showed differences in the microbiome depended on differences in diet composition, particularly fiber intake (Claesson et al., 2012). Proctor continued, saying that fiber is a topic of interest as a growth substrate for microbes, and investigators are trying to be more precise in their definition of various diet components that specifically support microbial growth. The classification of dietary fiber is now expanding and is being included into a larger class of microbial growth compounds called prebiotics. Prebiotics are defined as “a non-digestible compound which, through microbial metabolism, modulates the composition, and/or activity of gut microbiota. This change in microbial composition or activity confers health benefit to the host” (Bindels et al., 2015). Proctor expanded on the definition by stating that it shifts the focus on the role of the gut microbiota from the effect on the host to the direct effect of the compound on the microbiota. Prebiotics are no longer strictly defined as being carbohydrates (e.g., fiber); they are compounds that provide some type of beneficial physiologic effect on the microbiota and subsequently the host.
In closing, Proctor reiterated that although the human microbiome is a fixed feature of the host, it is also a variable trait and can be exploited to support health and treat disease. She also noted that one indicator of the growing interest in the microbiome is that in 2015, President Obama chartered a committee of government scientists to conduct an analysis of microbiome research across the federal government. The Fast-Track Action Committee on Mapping the Microbiome, which was formed by the Office
of Science and Technology Policy, includes representatives from 16 agencies in 6 departments, including the USDA, the U.S. Department of the Interior, the U.S. Department of Energy, the U.S. Department of Defense, the U.S. Department of Health and Human Services as well as the Environmental Protection Agency, National Aeronautics and Space Administration, National Science Foundation, and the Smithsonian Institution. The committee was charged with conducting a portfolio analysis of federal microbiome research and reporting on the gaps and challenges to advance the field. That report is intended to inform a Microbiome Initiative in the president’s fiscal year (FY) 2017 budget request.
QUESTION AND ANSWER SESSION
Following the Session 2 presentations, the floor was opened for questions and general discussion. In response to a question about the magnitude of funding for microbiome research and future research directions, Proctor stated that researchers are increasingly looking at microbiome components and their functions. She noted that she could not quote exact numbers but that the amounts of funding are increasing significantly. One reason why the field is growing so fast is because of new methods that are allowing researchers to conduct studies that were not possible before.
A participant asked Campbell whether researchers are looking at actual body weight or at ideal body weight in estimating protein requirements. Campbell replied that recommendations are truncated at a person’s upper limit of their ideal body weight. Considering people’s actual body weight, the proportion of older adults who are consuming an inadequate amount of protein is twofold. He went on to say that one of the things that becomes a challenge, especially with older adults, is when older people are consuming a low amount of energy and they try to restrict calories to lose weight. Even though they may be eating enough protein as a percentage of calories and think that they are within the acceptable range, they are below the RDA and closer to the EAR.
This question generated discussion about the importance of new thinking about nutrient needs among the older adult population, especially in the context of managing chronic disease, and of encouraging the consumption of nutrient-dense diets to help older adults come closer to meeting nutrient recommendations.
A final question for Campbell dealt with the best measure of a person’s protein status. He replied that one of the biggest challenges in the protein field is that it does not have biomarkers. That is why studies still rely heavily on nitrogen balance and isotope kinetics. Determining protein status requires serial measurements of a blood urea nitrogen, which is responsive
to differences in protein intake. However, that also is influenced by many factors that affect older people, function in particular.
REFERENCES
Alzheimer’s Disease International. 2010. World Alzheimer Report 2010. The global economic impact of dementia. http://www.alz.co.uk/research/world-report-2010 (accessed February 22, 2016).
Bailey, R. L., K. W. Dodd, J. A. Goldman, J. J. Gahche, J. T. Dwyer, A. J. Moshfegh, C. T. Sempos, and M. F. Picciano. 2010. Estimation of total usual calcium and vitamin D intakes in the United States. Journal of Nutrition 140(4):817-822.
Bartali, B., E. A. Frongillo, J. M. Guralnik, M. H. Stipanuk, H. G. Allore, A. Cherubini, S. Bandinelli, L. Ferrucci, and T. M. Gill. 2008. Serum micronutrient concentrations and decline in physical function among older persons. JAMA 299(3):308-315.
Bauer, J., G. Biolo, T. Cederholm, M. Cesari, A. J. Cruz-Jentoft, J. E. Morley, S. Phillips, C. Sieber, P. Stehle, D. Teta, R. Visvanathan, E. Volpi, and Y. Boirie. 2013. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. Journal of the American Medical Directors Association 14(8):542-559.
Bindels, L. B., N. M. Delzenne, P. D. Cani, and J. Walter. 2015. Towards a more comprehensive concept for prebiotics. Nature Reviews. Gastroenterology & Hepatology 12(5):303-310.
Bischoff, H. A., H. B. Stähelin, W. Dick, R. Akos, M. Knecht, C. Salis, M. Nebiker, R. Theiler, M. Pfeifer, B. Begerow, R. A. Lew, and M. Conzelmann. 2003. Effects of vitamin D and calcium supplementation on falls: A randomized controlled trial. Journal of Bone Mineral Research 18(2):343-351.
Bischoff-Ferrari, H. A., T. Dietrich, E. J. Orav, F. B. Hu, Y. Zhang, E. W. Karlson, and B. Dawson-Hughes. 2004. Higher 25-hydroxyvitamin D concentrations are associated with better lower-extremity function in both active and inactive persons aged > or =60 y. American Journal of Clinical Nutrition 80(3):752-758.
Bischoff-Ferrari, H. A., E. Giovannucci, W. C. Willett, T. Dietrich, and B. Dawson-Hughes. 2006. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. American Journal of Clinical Nutrition 84(1):18-28.
Bouillanne, O., C. Dupont-Belmont, P. Hay, B. Hamon-Vilcot, L. Cynober, and C. Aussel. 2009. Fat mass protects hospitalized elderly persons against morbidity and mortality. American Journal of Clinical Nutrition 90(3):505-510.
Brunner, R. L., B. Cochrane, R. D. Jackson, J. Larson, C. Lewis, M. Limacher, M. Rosal, S. Shumaker, R. Wallace, and the Women’s Health Initiative Investigators. 2008. Calcium, vitamin D supplementation, and physical function in the Women’s Health Initiative. Journal of the American Dietetic Association 108(9):1472-1479.
Bunout, D., G. Barrera, L. Leiva, V. Gattas, M. P. de la Maza, M. Avendano, and S. Hirsch. 2006. Effects of vitamin D supplementation and exercise training on physical performance in Chilean vitamin D deficient elderly subjects. Experimental Gerontology 41(8):746-752.
Campbell, W. W., and H. J. Leidy. 2007. Dietary protein and resistance training effects on muscle and body composition in older persons. Journal of the American College of Nutrition 26(6):696S-703S.
Campbell, W. W., T. A. Trappe, R. R. Wolfe, and W. J. Evans. 2001. The recommended dietary allowance for protein may not be adequate for older people to maintain skeletal muscle. The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 56(6):M373-M380.
Campbell, W. W., T. A. Trappe, A. C. Jozsi, L. J. Kruskall, R. R. Wolfe, and W. J. Evans. 2002. Dietary protein adequacy and lower body versus whole body resistive training in older humans. Journal of Physiology 542(Pt2):631-642.
Campbell, W. W., C. A. Johnson, G. P. McCabe, and N. S. Carnell. 2008. Dietary protein requirements of younger and older adults. American Journal of Clinical Nutrition 88(5):1322-1329.
Campbell, W. W., J. E. Kim, A. F. Amankwaah, S. L. Gordon, and E. M. Weinheimer-Haus. 2015. Higher total protein intake and change in total protein intake affect body composition but not metabolic syndrome indexes in middle-aged overweight and obese adults who perform resistance and aerobic exercise for 36 weeks. Journal of Nutrition 145(9):2076-2083.
Castaneda, C., J. M. Charnley, W. J. Evans, and M. C. Crim. 1995. Elderly women accommodate to a low-protein diet with losses of body cell mass, muscle function, and immune response. American Journal of Clinical Nutrition 62(1):30-39.
Castaneda, C., P. L. Gordon, R. A. Fielding, W. J. Evans, and M. C. Crim. 2000. Marginal protein intake results in reduced plasma IGF-I levels and skeletal muscle fiber atrophy in elderly women. Journal of Nutritional Health and Aging 4(2):85-90.
Childers, D. K., and D. B. Allison. 2010. The “obesity paradox”: A parsimonious explanation for relations among obesity, mortality rate and aging? International Journal of Obesity 34(8):1231-1238.
Claesson, M. J., I. B. Jeffery, S. Conde, S. E. Power, E. M. O’Connor, S. Cusack, H. M. Harris, M. Coakley, B. Lakshminarayanan, O. O’Sullivan, G. F. Fitzgerald, J. Deane, M. O’Connor, N. Harnedy, K. O’Connor, D. O’Mahony, D. van Sinderen, M. Wallace, L. Brennan, C. Stanton, J. R. Marchesi, A. P. Fitzgerald, F. Shanahan, C. Hill, R. P. Ross, and P. W. O’Toole. 2012. Gut microbiota composition correlates with diet and health in the elderly. Nature 488(7410):178-184.
Dam, T. T., D. von Mühlen, and E. L. Barrett-Connor. 2009. Sex-specific association of serum vitamin D levels with physical function in older adults. Osteoporosis International 20(5):751-760.
Davenport, E. R., O. Mizrahi-Man, K. Michelini, L. B. Barreiro, C. Ober, and Y. Gilad. 2014. Seasonal variation in human gut microbiome composition. PLoS ONE 9(3):e90731.
Dhesi, J. K., S. H. Jackson, L. M. Bearne, C. Moniz, M. V. Hurley, C. G. Swift, and T. J. Allain. 2004. Vitamin D supplementation improves neuromuscular function in older people who fall. Age and Ageing 33(6):589-595.
EFSA (European Food Safety Authority). 2012. Scientific opinion on Dietary Reference Values for protein. EFSA Journal 10(2):2557.
Faulkner, K. A., J. A. Cauley, J. M. Zmuda, D. P. Landsittel, A. B. Newman, S. A. Studenski, M. S. Redfern, K. E. Ensrud, H. A. Fink, N. E. Lane, and M. C. Nevitt. 2006. Higher 1,25-dihydroxyvitamin D3 concentrations associated with lower fall rates in older community-dwelling women. Osteoporosis International 17(9):1318-1328.
Flegal, K. M., B. I. Graubard, D. F. Williamson, and M. H. Gail. 2007. Cause-specific excess deaths associated with underweight, overweight, and obesity. JAMA 298(17):2028-2037.
Flegal, K. M., B. K. Kit, H. Orpana, and B. I. Graubard. 2013. Association of all-cause mortality with overweight and obesity using standard body mass index categories: A systematic review and meta-analysis. JAMA 309(1):71-82.
Ford, D.W., T. J. Hartman, C. Still, C. Wood, D. C. Mitchell, P. Erickson, R. Bailey, H. Smiciklas-Wright, D. L. Coffman, and G. L. Jensen. 2014. Body mass index, poor diet quality, and health-related quality of life are associated with mortality in rural older adults. Journal of Nutrition in Gerontology and Geriatrics 33(1):23-34.
Goodrich, J. K., J. L. Waters, A. C. Poole, J. L. Sutter, O. Koren, R. Blekhman, M. Beaumont, W. Van Treuren, R. Knight, J. T. Bell, T. D. Spector, A. G. Clark, and R. E. Ley. 2014. Human genetics shape the gut microbiome. Cell 159(4):789-799.
Hannan, M. T., K. L. Tucker, B. Dawson-Hughes, L. A. Cupples, D. T. Felson, and D. P. Kiel. 2000. Effect of dietary protein on bone loss in elderly men and women: The Framingham Osteoporosis Study. Journal of Bone Mineral Research 15(12):2504-2512.
Houston, D. K., B. J. Nicklas, J. Ding, T. B. Harris, F. A. Tylavsky, A. B. Newman, J. S. Lee, N. R. Sahyoun, M. Visser, S. B. Kritchevsky, and the Health ABC Study. 2008. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The Health, Aging, and Body Composition (Health ABC) Study. American Journal of Clinical Nutrition 87(1):150-155.
Houston, D. K., J. A. Tooze, C. C. Davis, P. H. Chaves, C. H. Hirsch, J. A. Robbins, A. M. Arnold, A. B. Newman, and S. B. Kritchevsky. 2011. Serum 25-hydroxyvitamin D and physical function in older adults: The Cardiovascular Health Study All Stars. Journal of the American Geriatrics Society 59(10):1793-1801.
Houston, D. K., J. A. Tooze, R. H. Neiberg, D. B. Hausman, M. A. Johnson, J. A. Cauley, D. C. Bauer, P. M. Cawthon, M. K. Shea, G. G. Schwartz, J. D. Williamson, F. A. Tylavsky, M. Visser, E. M. Simonsick, T. B. Harris, S. B. Kritchevsky, and the Health ABC Study. 2012. 25-hydroxyvitamin D status and change in physical performance and strength in older adults: The Health, Aging, and Body Composition Study. American Journal of Epidemiology 176(11):1025-1034.
Houston, D. K., R. H. Neiberg, J. A. Tooze, D. B. Hausman, M. A. Johnson, J. A. Cauley, D. C. Bauer, M. K. Shea, G. G. Schwartz, J. D. Williamson, T. B. Harris, S. B. Kritchevsky and the Health ABC Study. 2013. Low 25-hydroxyvitamin D predicts the onset of mobility limitation and disability in community-dwelling older adults: The Health ABC Study. The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 68(2):181-187.
Humayun, M. A., R. Elango, R. O. Ball, and P. B. Pencharz. 2007. Reevaluation of the protein requirement in young men with the indicator amino acid oxidation technique. American Journal of Clinical Nutrition 86(4):995-1002.
IOM (Institute of Medicine). 2005. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients). Washington, DC: The National Academies Press.
IOM. 2010. Dietary reference intakes for calcium and vitamin D. Washington, DC: The National Academies Press.
Janssen, H. C., M. H. Emmelot-Vonk, H. J. Verhaar, and Y. T. van der Schouw. 2013. Vitamin D and muscle function: Is there a threshold in the relation? Journal of the American Medical Directors Association 14(8):627.
Jernerén, F., A. K. Elshorbagy, A. Oulhaj, S. M. Smith, H. Refsum, and A. D. Smith. 2015. Brain atrophy in cognitively impaired elderly: The importance of long-chain ω-3 fatty acids and B vitamin status in a randomized controlled trial. The American Journal of Clinical Nutrition 102(1):215-221.
Kenny, A. M., B. Biskup, B. Robbins, G. Marcella, and J. A. Burleson. 2003. Effects of vitamin D supplementation on strength, physical function, and health perception in older, community-dwelling men. Journal of the American Geriatric Society 51(12):1762-1767.
Kim, J. E., L. E. O’Connor, L. P. Sands, M. B. Slebodnik, and W. W. Campbell. 2016. Effects of dietary protein intake on body composition changes after weight loss in older adults: A systematic review and meta-analysis. Nutrition Reviews 74(3):210-224.
Lips, P., N. Binkley, M. Pfeifer, R. Recker, S. Samanta, D. A. Cohn, J. Chandler, E. Rosenberg, amd D. A. Papanicolaou. 2010. Once-weekly dose of 8400 IU vitamin D(3) compared with placebo: Effects on neuromuscular function and tolerability in older adults with vitamin D insufficiency. American Journal of Clinical Nutrition 91(4):985-991.
Looker, A.C., C. M. Pfeiffer, D. A. Lacher, R. L. Schleicher, M. F. Picciano, and E. A. Yetley. 2008. Serum 25-hydroxyvitamin D status of the US population: 1988-1994 compared with 2000-2004. American Journal of Clinical Nutrition 88(6):1519-1527.
Manson, J. E., W. C. Willett, M. J. Stampfer, G. A. Colditz, D. J. Hunter, S. E. Hankinson, C. H. Hennekens, and F. E. Speizer. 1995. Body weight and mortality among women. New England Journal of Medicine 333(11):677-685.
Michael, Y. L., E. Smit, R. Seguin, J. D. Curb, L. S. Phillips, and J. E. Manson. 2011. Serum 25-hydroxyvitamin D and physical performance in postmenopausal women. Journal of Women’s Health 20(11):1603-1608.
Muir, S. W. and M. Montero-Odasso. 2011. Effect of vitamin D supplementation on muscle strength, gait and balance in older adults: A systematic review and meta-analysis. Journal of the American Geriatric Society 59(12):2291-2300.
Murphy, R. A., I. Reinders, M. E. Garcia, G. Eiriksdottir, L. J. Launer, R. Benediktsson, V. Gudnason, P. V. Jonsson, T. B. Harris; and Age, Gene/Environment Susceptibility-Reykjavik Study (AGES-Reykjavik). 2014. Adipose tissue, muscle, and function: potential mediators of associations between body weight and mortality in older adults with type 2 diabetes. Diabetes Care 37(12):3213-3219.
Oreopoulos, A., K. Kalantar-Zadeh, A. M. Sharma, and G. C. Fonarow. 2009. The obesity paradox in the elderly: Potential mechanism and clinical implications. Clinics in Geriatric Medicine 25(4):643-659.
Pfeifer, M., B. Begerow, H. W. Minne, C. Abrams, D. Nachtigall, and C. Hansen. 2000. Effects of a short-term vitamin D and calcium supplementation on body sway and secondary hyperparathyroidism in elderly women. Journal of Bone Mineral Research 15(6):1113-1118.
Pfeifer, M., B. Begerow, H. W. Minne, K. Suppan, A. Fahrleitner-Pammer, and H. Dobnig. 2009. Effects of a long-term vitamin D and calcium supplementation on falls and parameters of muscle function in community-dwelling older individuals. Osteoporosis International 20(2):315-322.
Rand, W. M., P. L. Pellett, and V. R. Young. 2003. Meta-analysis of nitrogen balance studies for estimating protein requirements in healthy adults. American Journal of Clinical Nutrition 77(1):109-127.
Scott, T. M., I. Peter, K. L. Tucker, L. Arsenault, P. Bergethon, R. Bhadelia, J. Buell, L. Collins, J. F. Dashe, J. Griffith, P. Hibberd, D. Leins, T. Liu, J. M. Ordovas, S. Patz, L. L. Price, W. Q. Qiu, M. Sarnak, J. Selhub, L. Smaldone, C. Wagner, L Wang, D. Weiner, J. Yee, I. Rosenberg, and M. Folstein. 2006. The Nutrition, Aging, and Memory in Elders (NAME) study: Design and methods for a study of micronutrients and cognitive function in a homebound elderly population. International Journal of Geriatric Psychiatry 21(6):519-528.
Sohl, E., R. T. de Jongh, A. C. Heijboer, K. M. Swart, E. M. Brouwer-Brolsma, A. W. Enneman, C. P. de Groot, N. van der Velde, R. A. Dhonukshe-Rutten, P. Lips, and N. M. van Schoor. 2013. Vitamin D status is associated with physical performance: The results of three independent cohorts. Osteoporosis International 24(1):187-196.
Sohl, E., R. T. de Jongh, M. W. Heymans, N. M. van Schoor, and P. Lips. 2015. Thresholds for serum 25(OH)D concentrations with respect to different outcomes. Journal of Clinical Endocrinology and Metabolism 100(6):2480-2488.
Sui, X. M., J. LaMonte, J. N. Laditka, J. W. Hardin, N. Chase, S. P. Hooker, and S. N. Blair. 2007. Cardiorespiratory fitness and adiposity as mortality predictors in older adults. JAMA 298(21):2507-2516.
Talegawkar, S. A., E. J. Johnson, T. Carithers, H. A. Taylor, Jr., M. L. Bogle, and K. L. Tucker. 2007. Total alpha-tocopherol intakes are associated with serum alpha-tocopherol concentrations in African American adults. Journal of Nutrition 137(10):2297-2303.
Tan, Z. S., W. S. Harris, A. S. Beiser, R. Au, J. J. Himali, S. Debette, A. Pikula, C. Decarli, P. A. Wolf, R. S. Vasan, S. J. Robins, S. Seshadri. 2012. Red blood cell ω-3 fatty acid levels and markers of accelerated brain aging. Neurology 78(9):658-664.
Thalacker-Mercer, A. E., and M. J. Drummond. 2014. The importance of dietary protein for muscle health in inactive, hospitalized older adults. Annals of the New York Academy of Sciences 1328:1-9.
Thalacker-Mercer, A. E., J. C. Fleet, B. A. Craig, and W. W. Campbell. 2010. The skeletal muscle transcript profile reflects accommodative responses to inadequate protein intake in younger and older males. Journal of Nutritional Biochemistry 21(11):1076-1082.
Verreault, R., R. D. Semba, S. Volpato, L. Ferrucci, L. P. Fried, and J. M. Guralnik. 2002. Low serum vitamin D does not predict new disability or loss of muscle strength in older women. Journal of the American Geriatric Society 50(5):912-917.
Visser, M., D. J. Deeg, P. Lips, and the Longitudinal Aging Study Amsterdam. 2003. Low vitamin D and high parathyroid hormone levels as determinants of loss of muscle strength and muscle mass (sarcopenia): The Longitudinal Aging Study Amsterdam. Journal of Clinical Endocrinology and Metabolism 88(12):5766-5772.
Wicherts, I. S., N. M. van Schoor, A. J. Boeke, M. Visser, D. J. Deeg, J. Smit, D. L. Knol, and P. Lips. 2007. Vitamin D status predicts physical performance and its decline in older persons. The Journal of Clinical Endocrinology and Metabolism 92(6):2058-2065.
Wildman, R. P., P. Muntner, K. Reynolds, A. P. McGinn, S. Raipathak, J. Wylie-Rosett, and M. R. Sowers. 2008. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999-2004). Archives of Internal Medicine 168(15):1617-1624.






























