3
The Spectrum of Aging and Health Over the Lifespan
Session 2 was moderated by Catherine Kwik-Uribe, global director of applied scientific research and scientific and regulatory affairs, Mars Symbioscience. In the first presentation, Janet King, senior scientist, Children’s Hospital Research Institute, emphasized the strong connection between in utero nutrition and outcomes later in life. She described highlights of the research in this area and speculated on the mechanistic role of DNA methylation in the link between early nutrition and early health. Next, Luigi Ferrucci, scientific director, National Institute on Aging, National Institutes of Health, differentiated between chronological and biological aging. He focused most of his talk on homeostatic dysregulation, specifically dysregulation of inflammation, which he said can have “devastating” effects, including impaired mobility. Finally, Gordon Jensen, senior associate dean for research and professor of medicine and nutrition, University of Vermont Larner College of Medicine, emphasized that obesity is a much stronger predictor of all-cause mortality at younger than at older ages, and spoke about the so-called obesity paradox—that mild obesity in the elderly is paradoxically associated with lower, not higher, mortality risk.
EARLY-LIFE ORIGINS OF METABOLIC DISEASE AND AGING1
King emphasized the strong connection between in utero nutrition and outcomes later in life. She began by describing what she termed the “maternal nutrition–offspring metabolic disease cycle,” whereby prepregnancy
___________________
1 This section summarizes information presented by Dr. King.
maternal nutrition affects fetal metabolism and growth and secondarily also affects child growth during the preschool years. This segment of the life cycle is, in turn, linked to the early adult years, when chronic oxidative stress and inflammation begin to manifest. Chronic oxidative stress and inflammation in young adults are in turn linked to an increased risk of metabolic disease at older ages.
The Link Between Fetal Undernutrition and Increased Risk of Metabolic Disease Later in Life
The first evidence linking fetal nutrition to later health came from a series of studies on the Dutch famine during World War II, which, King explained, serves as a natural study of severe food deprivation. From December 1944 through April 1945, Amsterdam was embargoed, and the food supply in the city was providing only around 400-800 calories per day per person, with limited amounts of protein (30-40 grams/day). When the city was liberated on May 5, 1945, the food supply immediately increased to about 2000 calories per day. During the famine, King said, pregnant women experienced severe maternal starvation, and they experienced it at different stages of pregnancy. This event therefore enables study of the effects of starvation at specific stages of pregnancy—for example, around the time of conception, during midpregnancy, or in late pregnancy.
King described how epidemiologists Mervyn Susser and Zena Stein spent many years reviewing data on the Dutch famine and found that not only did maternal starvation have an effect later in life but outcomes varied depending on when during fetal development the starvation was induced (Susser and Stein, 1994). Specifically, King reported, they found that starvation during periconception was associated with decreased fertility, but if a woman did conceive, there was an increased risk of neural tube defects and effects on brain function (i.e., increased schizophrenia and antisocial personalities in later years of life). Starvation during the first trimester of pregnancy was associated with increases in preterm births, stillbirths, and first-week deaths, noted King. Starvation during early pregnancy was also associated with increased obesity later in life. Finally, starvation induced during the third trimester was associated with low maternal weight gain, reduced birth weight in both the first and next generations, and increased neonatal mortality (0-3 months). Additionally, King explained, in contrast with what was observed with first-trimester starvation, third-trimester starvation was associated with a decrease in obesity later in life. King remarked that these data from the Dutch famine show strongly that fetal development is highly dependent on nutrition at all stages of reproduction and that changes induced are irreversible.
While Susser and Stein were examining data on the Dutch famine, King continued, epidemiologist David Barker began reviewing data that had been collected in England and Wales in the 1920s and evaluating the effect of fetal or infant nutrition during that time on health outcomes later in life. In the 1920s, King noted, England and Wales were quite thorough in collecting as much data as possible on mothers during pregnancy, and they followed children’s growth very carefully as well. Barker and colleagues found that malnutrition in the womb changes the structure and function of the body for life, making the individual vulnerable to heart disease, diabetes, and stroke later in life (“fetal programming”). This work eventually led to formation of the International Society for Developmental Origins of Health and Disease (DOHaD) in 2004. In 2010, DOHaD expanded its scope to include gestational exposure to overnutrition, as well as to stress.
Barker’s initial paper, which was published in the Lancet (Barker and Osmond, 1986), was an analysis of large birth cohort studies from different regions of England and Wales. King reported that the authors found a strong geographic relationship between mortality rates from ischemic heart disease (IHD) and infant mortality 40 years earlier, such that higher regional infant mortality rates in the 1920s were associated with elevated IHD mortality rates in those same regions in the 1960s. Additionally, she noted, they found that poor, rural areas had higher rates of infant mortality and IHD mortality relative to urban areas. The relationships between infant mortality and other disease mortality rates (e.g., respiratory diseases, cancers) were not as strong, she observed. Barker and Osmond (1986) concluded that poor nutrition in early life increases later susceptibility to the effects of an affluent diet.
A few years later, King reported, Barker extended his studies using data from Helsinki and examined, in addition to nutrition of the fetus, nutrition of the child during the growing years (Barker et al., 2002). He and his colleagues also had access to data on adult disease. They found that disease risk increased with low birth weight combined with a marked increase in childhood body mass index (BMI) from ages 3 to 11 years (Barker et al., 2002)—in other words, King explained, a child born small but growing rapidly during the preschool and early school-age years. Specifically, compared with infants with normal birth weight and childhood growth, infants born small but growing rapidly showed a 57 percent increase in type 2 diabetes later in life; a 25 percent increase in hypertension; and 25 and 63 percent increases in coronary heart disease for men and women, respectively. Thus, King emphasized, rapid growth in early childhood does have an impact on chronic disease later in life.
Since Barker’s initial studies, King noted, more than 100 additional studies involving half a million cases have shown consistent associations
between low birth weight and metabolic disorders later in life (i.e., type 2 diabetes, hypertension, dyslipidemia, central obesity, insulin resistance, and cardiovascular disease). They have shown inconsistent associations between low birth weight and respiratory, immunity, and psychiatric illnesses and cancer. It appears, King suggested, that brain growth is “protected” at the expense of lung, heart, and kidney.
In summary, King said, the work of Susser, Stein, Barker, and others has shown that undernutrition during fetal development increases risk for metabolic disease later in life and that this increased risk for metabolic disease is exacerbated by overnutrition during early childhood.
The Link Between Maternal Overnutrition and Later Health Outcomes
Evidence also indicates, King continued, that overnutrition in utero combined with overnutrition in childhood increases the risk for metabolic disease later in life. She summarized some of this evidence, based on a review paper published in 2015 (Gaillard, 2015). Among obese mothers, she said, fetal outcomes include increases in stillbirths, neonatal deaths, congenital anomalies, large size for gestational age, neonatal hypoglycemia, and referrals to the intensive care unit. Childhood outcomes among children born to obese mothers include obesity, adverse body composition, increased blood pressure, adverse lipid profile, increased inflammatory markers, and impaired insulin/glucose homeostasis. Adult outcomes include obesity, increased blood pressure, adverse lipid profile, impaired insulin/glucose homeostasis, and premature mortality.
Of interest, according to King, maternal prepregnancy obesity has a stronger association than gestational weight gain with adverse fetal outcomes. She interprets this to mean that efforts should be focused on trying to help obese women achieve a normal weight before they conceive. Additionally, it is now known, according to King, that maternal obesity alters the placental structure and function in a way that leads to an increased disease risk later in life. She noted that the placenta used to be viewed as being highly adaptable to many environmental conditions the mother might be experiencing. But now, she said, it is recognized that obesity can alter placental structure and function in a way that influences fetal development. Additionally, excessive first trimester weight gain is now known to be a key risk factor for a later adverse cardiometabolic profile in the offspring. Taken together, King said, these findings mean, “You need to intervene prior to pregnancy and achieve a normal body weight.”
With respect to the relationship between breastfeeding and later health, King remarked that the literature in this area is not strong for several reasons. First, she suggested, it is very difficult to determine breastfeeding exposures accurately. Many women do not breastfeed exclusively, she noted,
and the amount of formula provided is not well documented. Moreover, she added, the data that do exist are primarily from higher-income countries. Nonetheless, she reported, some preliminary conclusions have been drawn: that in high-income populations, breastfeeding is associated with reductions in blood pressure and blood cholesterol and a lower risk of obesity and diabetes in adulthood (Robinson and Fall, 2012), and that early exclusive breastfeeding (4-6 weeks) is associated with longer telomeres at 4-5 years of age (Wojcicki et al., 2016).
Mechanisms Linking Early Nutrition to Later Health
Among the variety of mechanisms that King suspects play a role in the link between early nutrition and early health, she focused on DNA methylation, which she suggested may increase disease risk through gene silencing. She cited two ways in which DNA methylation in the fetus could increase in utero. One possible mechanism, she suggested, is the micronutrient intake of the mother, with folate, methionine, B12, choline, B6, and riboflavin all influencing DNA methylation. In addition, she said, data are beginning to accumulate indicating that the mother’s microbiota may produce metabolites with DNA methylation potential in the fetus. She added that because a mother’s microbiome is related to her diet, these two potential mechanisms are interrelated.
King went on to describe efforts by Andrew Prentice and colleagues, working in The Gambia, to investigate the relationship between maternal nutrition and DNA methylation. In their first study (Cooper et al., 2012), Prentice and colleagues wanted to see whether they could influence DNA methylation in the newborn by providing women with a supplement of micronutrients prior to conception and through the first trimester. To this end, King reported, they randomized Gambian women to multimicronutrient or placebo supplementation from prepregnancy to the end of the first trimester. They found that periconceptional nutrition did indeed influence DNA methylation in the newborn, and that the genes that were methylated differed between boys and girls. King speculated that this finding might explain why women are more susceptible than men to heart disease later in life. Finally, she noted that Prentice and colleagues found that methylation occurred independently of the season of the year. (The Gambia, she explained, has a rainy season, which is called the “hunger season,” and a dry season, when food is more plentiful.) In sum, she said, these researchers showed that maternal diet can influence the methylation of genes in the newborn (Cooper et al., 2012).
King explained that Prentice and colleagues conducted two additional studies (Dominguez-Salas et al., 2013, 2014) in which they examined blood biomarkers of methylation in mothers in conjunction with their diet and
methylation of genes in infants. It is during the rainy, or “hunger,” season, she said, that the intake of methyl-donor micronutrients is expected to be lower. However, what the researchers found was opposite to what they expected. They found that the blood biomarkers of methylation (folate, methionine, riboflavin, and SAM/SAH ratio [a measure of the amount of methylation possible, King explained]) were higher during the rainy season. Additionally, they found that offspring of mothers who conceived during the rainy season had higher levels of leukocytes and hair DNA methylation. Of interest, King noted, they also found that women with slightly higher BMIs had less methylation, indicating perhaps that they were in a better nutritional state. The conclusion to be drawn from these two studies, she asserted, is that poor maternal nutrition at conception enhances gene silencing in newborns.
The big picture that is beginning to emerge, King summarized, is that DNA methylation increases as a result of maternal diet in utero and that this increased silencing, in turn, may lead to inflammation and oxidative stress. She called attention to the large literature on the relationship between DNA methylation and inflammation and oxidative stress, noting that inflammation and oxidative stress may, in turn, be precursors of aging-related metabolic diseases.
Breaking the Cycle
The question for King is whether this increased risk of metabolic disease can be reduced—for example, by adding micronutrients to the diet. She mentioned Bruce Ames’s “triage theory,” which predicts that micronutrient deficiencies lead to essential nutrients being “triaged” to support critical functions (Ames, 2006). According to the theory, this shift reduces the availability of micronutrients to prevent oxidative stress and inflammation (i.e., because under normal conditions, the essential nutrients being triaged would be preventing oxidative stress and inflammation).
King decided to test Ames’s triage theory using zinc. She and her colleagues fed 18 men a low-zinc diet for 2 weeks (6 mg per day) (“depletion”) and then 10 mg per day for 4 weeks (“repletion”) and measured oxidative stress by examining DNA damage (i.e., using the comet assay, which measures both double and single DNA strand breaks). They found that during depletion, DNA damage increased, while during repletion, DNA damage declined back to baseline. King described this change in zinc level as a short, acute change and remarked that she did not expect to see this kind of shift in DNA damage with such a small change.
BIOMARKERS OF AGING2
Ferrucci differentiated between chronological and biological aging. The metrics of aging, he explained, change very quickly early in life as newborns grow into young children. This early rapid phenotypic change over time then stabilizes for a while as individuals live independently and are able to interact with their environments. Then at some later point, which Ferrucci emphasized is very different for different individuals, phenotype change over time begins to accelerate again. This period of accelerated aging, he explained, is when all the compensatory strategies in human physiology that have been selected to maintain the body’s stability start failing, and the body is unable to respond to the challenges destabilizing it.
What Is Biological Aging?
Ferrucci asked members of the audience to imagine walking in Piazza del Camp, Siena, Italy, and coming across a man sitting on the ground, leaning back, his legs stretched out in front of him, his arms braced behind him, holding his torso up, and then wondering how old this man is. There are some discrepancies in the man’s appearance. Ferrucci described the man as someone who is clearly enjoying his life. It is a spring day. He is looking up to the sky and smiling, with a pleasant expression on his face. He appears to be past middle age. He has lost some musculature. He is dressed like a teenager, in shorts and sneakers, but, Ferrucci said, he is in fact probably wearing the sneakers because of some pain in his feet. So when asked how old the man is, Ferrucci said, you will formulate a number based on all of this information and say, “This person is probably x.” He did not ask the workshop audience to estimate the age of the man in the photo, but Ferrucci said that if he did, the range of answers would be wide. The point he wanted to make was that until recently, scientists were not much better than this at estimating someone’s biological age, especially in old age.
If the goal is to develop interventions to slow aging, Ferrucci argued, one needs to be able to measure aging. Otherwise, how will it be possible to demonstrate that an intervention has worked? Based on a review of epidemiological studies, he and his colleagues identified four domains of aging: (1) changes in body composition, (2) energy imbalance (production/utilization), (3) homeostatic dysregulation, and (4) aging of the brain (e.g., neurodegeneration, neural dysfunction, loss of plasticity) (Ferrucci and Studenski, 2011). In his opinion, energy is key and is probably the most understudied and most important parameter for understanding aging.
___________________
2 This section summarizes information presented by Dr. Ferrucci.
Aging-Related Inflammation and Implications for Mobility
Ferrucci focused the remainder of his talk on homeostatic dysregulation,3 specifically dysregulation of inflammation. The development of a mild, proinflammatory state, he said, is a universal characteristic of every aging organism. He noted that aging has been associated with up-regulation of immune function genes not just in humans but in every living organism studied (e.g., frogs, fish, mice, nonhuman primates). “In every living organism,” he said, “dysregulation of inflammation is one of the culprits of aging.” Although there are hints as to why this is the case, however, the mechanisms are not well understood, he observed.
Importantly, Ferrucci continued, dsyregulation of inflammation has implications for mobility. He described results from a case cohort study he and colleagues conducted showing that interleukin (IL)-6 levels above a certain threshold (2.5 picograms/milliliter) are associated with higher levels of mobility disability 4 years later (Ferrucci et al., 1999) (see Figure 3-1). “I’m not talking about disease,” he said. “I’m talking about something that is really devastating. Development of a mobility disability and the inability to move without help is a “tragic, drastic change in the condition of your life.”
Ferrucci explained that normally, inflammation is a “good” thing. An inflammatory response, he noted, is how the body responds to viral and other attacks. When “fighting a war,” he said, maintenance and repair are not important. It does not matter during inflammation that one’s muscles, for example, cannot synthesize proteins or that erythrocytes cannot be produced or that neurogenesis is blocked (Zonis et al., 2015). It is more important, he emphasized, that the body does what it needs to do to fight off the attack. Then when the “war” is over, he continued, those resources can be used to “rebuild.” So when inflammation is transient, he said, “it is fine.” Otherwise, the accumulation of damage leads to chronic disease.
Obesity is known to be proinflammatory, Ferrucci explained, especially when fat does not deposit in the normal place, that is, subcutaneously. It therefore becomes strongly proinflammatory because of the infiltration of macrophages and also, probably, he said, because of cell senescence (the loss of a cell’s power of division and growth) (Zhu et al., 2009). Yet, while obesity is proinflammatory, he reported, evidence from bariatric surgery and liposuction and the disconnect between rapid weight loss and IL-6 levels in the blood following these procedures suggests that “fat cannot be the only thing” (Klein et al., 2004). Following bariatric surgery, he noted, inflammation drops on the second day, much before any substantial reduction in weight, and following liposuction, inflammation actually goes up.
___________________
3 Disruption or impairment in the ability to maintain a stable equilibrium among interdependent elements within body systems or physiological processes.
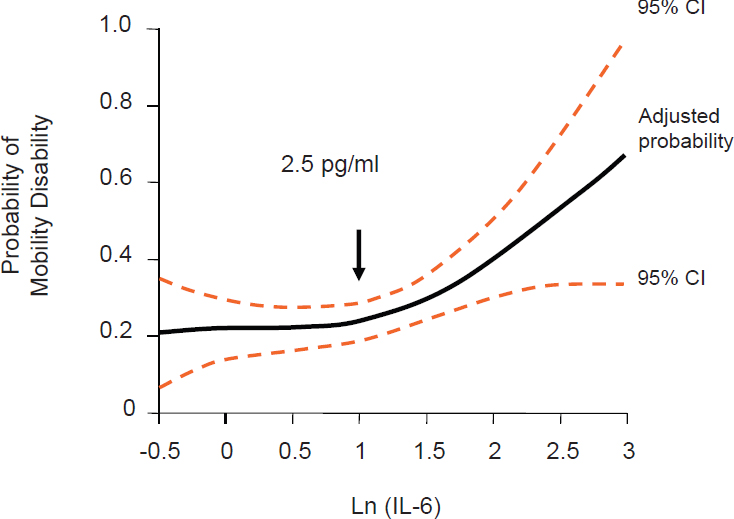
NOTE: CI = confidence interval.
SOURCES: Presented by L. Ferrucci, September 13, 2016. Reprinted with permission from Wiley: Ferrucci et al., 1999.
Beyond obesity, Ferrucci wondered whether inflammation might be related to the absorption of food. He and colleagues conducted what he called “the fast food study,” in which they mimicked a fast food meal (a 1,000-calorie meal with saturated fat instead of polyunsatured fat) and provided study participants with either the fast food–like meal or a healthy meal. During digestion of this meal (within 2 hours of eating), the researchers found a much higher elevation of IL-6 among those who had eaten the saturated fat–filled, fast food–like meal (see Figure 3-2). In other, still unpublished experiments, Ferrucci and colleagues conducted ultracentrifugation of lipids and found that at 2 hours, that is, when IL-6 levels were elevated, there was an increase in oxidized lipoprotein. This finding suggested, he said, that perhaps administering an antioxidant exactly 2 hours after eating a meal high in saturated fat might reduce the negative effect of this meal.
Ferrucci reiterated that during inflammation the ability of muscle to regenerate protein is clearly impaired. He mentioned two experiments in animal models showing that after a meal muscles absorb amino acids for
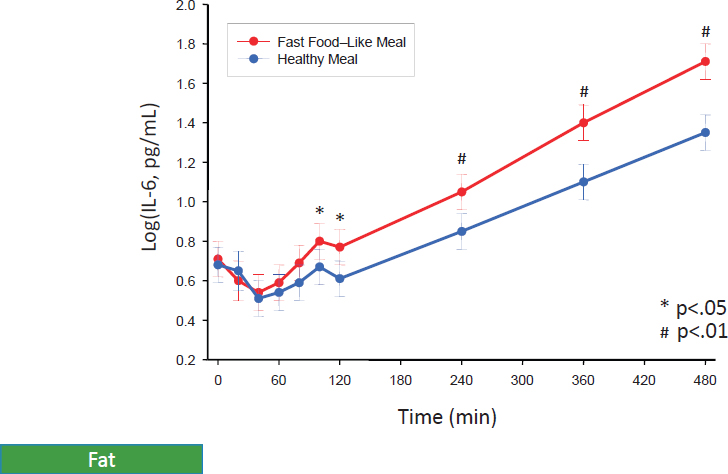
SOURCES: Presented by L. Ferrucci, September 13, 2016. Reprinted with permission.
30-35 minutes, depending on age. In a strongly inflammatory state (IL-6 greater than 3.5 pg/mL), he explained, this postmeal effect disappears. Similarly, in a state of chronic inflammation, the increase in protein synthesis that normally occurs with exercise does not occur. Ferrucci and colleagues found in a longitudinal observational study with humans that in individuals with inflammation, the amount of protein required to maintain stability of muscle mass was much greater relative to the amount required by individuals without inflammation. Moreover, he noted, those with high inflammation, regardless of their protein intake, always showed a decline in muscle mass.
One “quick” strategy for addressing aging-related inflammation, Ferrucci concluded, is to treat periodontal disease, which has been associated with a higher brain amyloid load in older individuals (Kamer et al., 2015). He encouraged examining other “details” of aging, such as the atrophy of fungiform papillae in the tongue that occurs in older individuals (with consequences for insulin secretion), as ways to address not just inflammation but other burdens of aging as well.
OVERWEIGHT AND OBESITY IN OLDER PERSONS: IMPACT ON HEALTH AND MORTALITY OUTCOMES4
Jensen recalled that when he first started his aging studies, in the 1990s, he would see patients with severe obesity in the clinic, but that the focus in the field at that time was on frailty and undernutrition. Today, he said, while some frail, tiny individuals will still be encountered in any hospital or chronic care facility, many frail and overweight or obese persons will be encountered as well. During his presentation, he said, he would “wade into an area of tremendous controversy,” that is, the so-called obesity paradox, whereby mild obesity in the elderly is paradoxically associated with lower, not higher, mortality risk.
Health and Mortality Outcomes Associated with Obesity
Jensen said, “I sometimes think you are born, you start to have inflammation, and then it progresses the rest of your life.” That said, he continued, there are many adverse health outcomes specifically associated with obesity as one ages, including premature disease, functional decline, and shortened duration of life. The earliest studies on obesity and duration of life that did not show a relationship, he noted, were confounded by important covariates, including smoking. It is safe to say, in his opinion, that obesity generally does not confer health benefits. He observed that the classic mortality curve in relation to BMI is J-shaped: mortality increases at either end of the BMI spectrum. Yet while obesity is strongly associated with all-cause mortality, he said, a wealth of data now exist to indicate that it is a much stronger predictor at younger than at older ages. That is, he explained, while obesity in childhood or young adulthood has been clearly associated with decreased life expectancy, the relationship between obesity and mortality in older adults is more complex. In addition, he noted, obesity is associated with a long list of comorbidities, including cardiovascular disease, diabetes, metabolic syndrome, and several malignancies.
The Obesity Paradox
Jensen remarked that the scientific literature on the obesity paradox has been growing for more than a decade and referred workshop participants to what he described as a helpful review by Oreopoulos and colleagues (2009). Although unanswered questions remain about why the obesity paradox exists, he expressed the hope that some of what he would discuss is beginning to provide some clarity. It has been suggested, he said, that some degree of
___________________
4 This section summarizes information presented by Dr. Jensen.
overweight may provide a metabolic reserve for surviving illness and injury, and he believes “there may be something to that.”
A landmark paper in the field was a systematic review and meta-analysis of all-cause mortality among different BMI categories (Flegal et al., 2013). This was a large analysis, Jensen said, covering nearly 3 million people and 270,000 deaths. The key finding, he reported, was that, compared with desirable BMI (defined as 18.5-24.9), while class II obesity and class III obesity (severe obesity) were associated with significantly greater all-cause mortality, class I obesity was not associated with higher mortality, and overweight was associated with significantly lower mortality. Among individuals aged 65 and older, the hazard ratios for all-cause mortality were 0.90 for those with a BMI of 25-30, 0.87 for those with a BMI of 30-35, and 1.20 for those with a BMI of 35 or greater. The Flegal et al. (2013) paper, Jensen said, rather than settling the paradox, started a “raging controversy,” with a key issue for discussion being how to interpret its findings for health professionals and for the public (Hughes, 2013). Jensen also mentioned another review by Childers and Allison (2010), who showed that the lowest mortality rate in the classic J-shaped mortality curve in relation to BMI generally shifts toward a higher BMI for older persons.
Sarcopenic Obesity and the Obesity Paradox
Evidence from studies on sarcopenic obesity (obesity accompanied by reduced lean body mass), Jensen continued, suggest that the obesity paradox may be explained in part by body composition and that overweight in older adults is protective only in those with high muscle mass (see the discussion of sarcopenia in Chapter 4). One of these studies was Murphy et al. (2014), a modest-sized cohort study (N = 637) of individuals aged 66-96 with diabetes. Jensen reported that the thigh muscle mass of participants was assessed by computed tomography scan. The interesting finding from this study, he said, was that the highest mortality was among those with low muscle mass who were either normal weight or overweight, and that normal-weight participants had a greater mortality risk than overweight individuals, a paradox explained partly by muscle mass.
In another study of sarcopenic obesity, Jensen continued, Bea and colleagues (2015) examined the risk of mortality in relation to BMI and body composition among a large sample (N = 10,525) of women who underwent DEXA (dual-energy X-ray absorptiometry) scans and were followed for 13.6 years. They found that, overall, high BMI (greater than or equal to 35) was associated with increased mortality, while total body fat and lean body mass were not. However, Jensen reported, among women aged 50-59, those with greater total body fat had increased mortality, while those with greater lean body mass had decreased mortality. These findings were
reversed for women aged 70-79. These results reflect what Jensen described as a “very complex scenario,” with body composition, age, and sex, as well as the population being studied, all being potential factors.
BMI, Metabolic Health Status, and All-Cause Mortality Among Older Adults
Jensen’s own research group recently conducted two studies on the relationship between BMI and all-cause mortality, both released just prior to the workshop. Both studies, he explained, involved subsamples of the cohort of rural, older Pennsylvanians with a mean age of 74 at baseline that he and his research team have been following for more than 20 years. What remains of the cohort, he said, is about 2,500 surviving people all well over the age of 80.
In the first study, Cheng and colleagues (2016a) examined BMI and all-cause mortality among more than 4,500 members of this cohort with a mean BMI of 25.9 at baseline. Jensen explained that they followed this subset for almost 11 years. They analyzed three models: one that adjusted for age and sex; a second that adjusted for both of those plus smoking status and alcohol use; and a third that adjusted for all of the above plus blood glucose, diabetic medication, triglycerides, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, cholesterol medication, diastolic blood pressure, hypertension medication, and disease burden. According to Jensen, the multiple models represented an attempt to control systematically for as many confounding variables as possible. The researchers found that, compared with the reference BMI (18.5-24.9), all-cause mortality was greatest among individuals with BMIs below 18.5, which Jensen suggested may be explained by some of these individuals having underlying disease. In contrast, he noted, all-cause mortality among individuals with BMIs of 25-29.9 and 30-34.9 was lower than that among the reference BMI group, for all three models. At the higher end of the BMI spectrum, he added, the hazard ratios for all-cause mortality increased again. In sum, he said, individuals who were underweight had a significantly greater risk of all-cause mortality, while those with overweight or mild obesity had a significantly lower risk. Thus, he explained, consistent with results of other studies, these researchers found a U-shaped relationship between BMI and all-cause mortality, with the lowest mortality risk seen among those with a BMI of 28-30 (see Figure 3-3).
In the second study, Jensen continued, Cheng and colleagues (2016b) used the same data set to take the analysis one step further and examine metabolic health status (using the Adult Treatment Panel III criteria, with metabolic health being defined as having fewer than two risk factors) and its relationship to all-cause mortality and BMI. Covariates
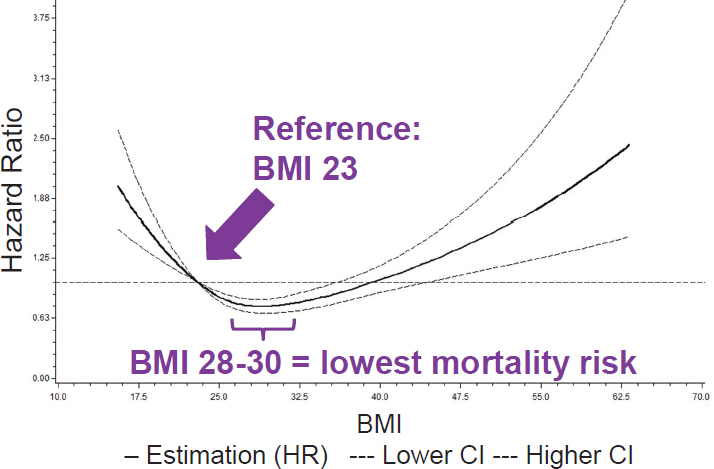
NOTE: CI = confidence interval; HR = hazard ratio.
SOURCES: G. Jensen, September 16, 2016. Reprinted with permission from Wiley: Cheng et al., 2016a (modified).
addressed in the analysis, he explained, included age, sex, smoking, alcohol, metabolic laboratory test results, medications, and disease burden. Jensen did not have time to present each analysis, but listed the multiple sensitivity analyses conducted in an attempt to bring a high level of rigor to the analysis, which included analysis of a fully adjusted model with never-smokers with no disease burden; metabolically unhealthy individuals, including all individuals meeting any diabetes criterion; metabolically healthy individuals, including only those with no metabolic risk factors; never-smokers, excluding individuals who died during the first 5 years of follow-up; all individuals, excluding those who became metabolically unhealthy during follow-up; and all individuals, excluding those who used cholesterol-lowering, diabetes, and blood pressure medications.
Over the almost 11-year study period, Jensen reported, there were 2,294 deaths among the study participants. The main finding was that metabolically healthy overweight or obesity was not associated with a sig-
nificantly greater mortality risk. This finding, Jensen said, was unchanged by the exclusions tested in the sensitivity analyses. Additionally, the researchers found that the association between BMI and mortality was more pronounced in individuals with fasting blood glucose levels greater than 100 mg/dL or diabetes diagnoses.
Although metabolically healthy overweight or obesity was not associated with a greater mortality risk, Jensen noted, the proportion of metabolically unhealthy individuals still increased at higher levels of obesity—from about 65 percent among individuals with “desirable” BMIs to 90 percent among those with class II or III obesity. In a comparison of all metabolically healthy and metabolically unhealthy individuals (N = 4,551), the shift toward an increased risk for being metabolically unhealthy across all BMI categories is “dramatic,” Jensen said. The same shift occurs, he observed, in a comparison of only metabolically healthy and metabolically unhealthy never-smokers with no identified chronic disease burden (N = 1,046 remaining for analysis), and again in a comparison of never-smokers excluding individuals that died during the first 5 years of follow-up (N = 2,367 remaining for analysis).
According to Jensen, the results of this second study suggest that there is a subset of what he described as “pretty robust healthy overweight and mildly obese older people.” The obesity paradox in previous studies may be partially explained, he said, by the inclusion of metabolically healthy overweight and mildly obese people who did not have an elevated mortality risk. Of course, he added, there are many older adults who are overweight or mildly obese who are metabolically unhealthy and who do have an increased mortality risk.
In Jensen’s opinion, an important hypothesis to test in the future is whether these same metabolically healthy overweight and mildly obese individuals are also to some degree protected from chronic inflammation and sarcopenic obesity. He added that the rigorous sensitivity analyses conducted as part of this study show that methodological issues cannot fully explain the obesity paradox.
Conclusion: Benefits Versus Risks
In closing, Jensen stated that obesity generally does not confer mortality or health benefits. However, he believes the current use of the National Institutes of Health BMI guidelines warrants reevaluation for older persons. He said he is concerned when he sees a frail older person in a chronic care facility who is overweight or mildly obese placed on a restricted diet and encouraged to lose weight. With respect to the protective effect of elevated BMI, he believes that both disease burden and body composition are key factors, and that elevated BMI may have no protective effect in the pres-
ence of reduced muscle mass (sarcopenic obesity). Finally, he reiterated the importance of determining how to interpret findings on the obesity paradox for health professionals and the public. He said the message cannot be that people all should go out and eat what they want or that there is no need to engage in exercise.
DISCUSSION WITH THE AUDIENCE
Questions About the Obesity Paradox
Following Jensen’s talk, he, King, and Ferrucci participated in an open discussion with the audience. The majority of the discussion revolved around Jensen’s presentation on the obesity paradox. Catherine Kwik-Uribe asked whether obesity is more of a spectrum condition and whether Jensen or others have examined the onset of overweight or obesity and its relationship to mortality risk. Jensen replied that answering this question would require a large enough cohort of people who have become overweight or obese later in life, and that at present, there are not enough data available to address the question. He suspects that many of the older adults that he and his group have been following were overweight or obese in middle age or younger. He noted the many changes that occur in postmenopausal women in particular.
Jensen does think that obesity encompasses a spectrum of nutrition concerns. He pointed to the range in nutritional status among overweight and obese persons as an example. He and his research team have documented multiple micronutrient deficiencies in overweight and obese older individuals. It is very possible, he said, to be large, mobility-limited, and homebound and undernourished at the same time.
Simin Meydani, a workshop participant, asked whether Jensen would still recommend that older adults stay within a BMI range of 18.5-24.9, given that it appears that only a small percentage of older people are overweight or obese and metabolically healthy. Jensen replied that it is an important observation that some 30 percent of older adults who are overweight or have class I obesity are metabolically healthy, compared with 10 percent of those with class II or III obesity. He clarified that he was not suggesting that public health guidance regarding BMI be changed; rather, he said, “we might want to rethink how we apply it.” Instead of suggesting to some older adults who are overweight and metabolically healthy that they lose weight, he observed, perhaps the focus should be on how best to preserve their quality of life with respect to flexibility, transfer, strength, and function.
Meydani then asked whether the metabolically healthy overweight and obese individuals in Jensen’s studies were physically active. Jensen replied
that the researchers did examine physical activity levels, but only through a single, self-report question. He agreed that this is an important question to revisit. He mentioned again that in his opinion, an important hypothesis to test in the future is whether metabolically healthy overweight and obese individuals are also to some degree protected from chronic inflammation and reduced lean body mass. Physical activity could be examined in that context, he noted.
Several questions were raised about whether and how obesity affects health outcomes in different populations. The population with end-stage renal disease is very large in the United States, according to Johanna Dwyer, workshop participant, with many people on dialysis. While individuals in this population have chronic inflammation and are not metabolically healthy, she noted, the advantage of overweight is quite substantial for them with respect to mortality as well as morbidity. She wondered whether that population warrants a different standard. Jensen reiterated that many factors influence the relationship between obesity and all-cause mortality. He described the association as “very population-specific.” He recognizes that his own cohort studies focus on community-dwelling, older individuals, although many are significant health care consumers. Regarding inflammation in people on dialysis, he wondered whether dialysis itself was proinflammatory in some way. Given that many of these people are in a state of chronic inflammation and are metabolically unhealthy, he said, it would not surprise him to learn that being overweight or obese provides some kind of metabolic reserve to enable people to get through what would otherwise be a deteriorating course. He added that the relationship between overweight and obesity and mortality not only is population-specific but also is likely individual-specific as well.
Ferrucci remarked that in longitudinal studies loss of weight is an extremely strong predictor of mortality. When people begin losing weight without trying, he said, this can be an indication that they could die within a few years. He wondered whether the effect being observed in studies of the obesity paradox might be a biomarker of less severe disease (i.e., that people are not going to die within a few years) rather than an actual protective factor.
Finally, the panelists were asked whether there were any distinctions between the sexes with respect to the obesity paradox. The most glaring sex difference, Jensen replied, is that in populations of older, community-dwelling adults, there are not many obese men. He then referred to the several sex differences he had noted during his presentation. Ferrucci added that there are sex differences in inflammation and in the relationship between inflammation and obesity. Women have much lower inflammation than men, he explained, but also in women, a small change in inflammation
has a more powerful effect on outcome than it does in men. In women, visceral obesity is a stronger predictor of inflammation than in men.
Questions About Biomarkers of Aging
Biomarkers of aging were another key focus of the discussion. In response to Kwik-Uribe’s asking the panelists whether they thought any specific biomarkers warranted further evaluation, Ferrucci commented on the emergence of robust biomarkers that track chronological age, such as DNA methylation markers. By analyzing the DNA from blood, he said, one can tell the age of a person within 1.5 years, but when these biomarkers are applied to biological age, they do not work. In his opinion, inflammation is the best available marker for biological age, but even so, he believes it is a much better predictor of short-term, not long-term, mortality. He suspects that many biomarkers of aging that have been studied in animals and are now being examined in humans, such as the number of senescence cells in the skin, the ability to repair DNA, and the functioning of mitochondria in the muscle, eventually will not only emerge as predictors of biological aging but also reveal which mechanisms in a specific individual need to be addressed to slow aging.
Finally, when asked to name a function biomarker of aging, Ferrucci replied, “walking speed.” He considers walking speed to be the strongest available predictor of mortality, disability, and declining health.
Miscellaneous Comments
In response to King’s having mentioned some of the benefits of exclusive breastfeeding, an audience member stressed keeping in mind as well the importance of nutrition and the quality of energy and calories during the third and fourth years of life. She mentioned what she called the “Mountain Dew mouth study,” which showed that children in West Virginia who consume large quantities of calories from Mountain Dew grow up to have high incidences of oral cavity cancer, neck cancer, and brain cancer.
This audience member also commented on some of what Ferrucci had discussed about inflammation and obesity. In her opinion, obesity is not the origin of inflammation; rather, the origin of inflammation is the level of oxygen in the blood. Glucose sucks the oxygen from the blood, she said, which causes not just inflammation, but also an accumulation of plaque in the arteries.


















