3
Disease-Induced Loss of Function and Tissue Regeneration
Session 3 was moderated by Alex Kemper, Chief of the Division of Ambulatory Pediatrics at the Nationwide Children’s Hospital, and a Planning Committee member. In the first presentation Christopher Duggan, Director of the Center for Nutrition at the Boston Children’s Hospital, discussed examples of gastrointestinal (GI) dysfunction and nutrient malabsorption in intestinal failure. The next speaker, Sarah Jane Schwarzenberg, Chief of Pediatric Gastroenterology, Hepatology, and Nutrition at the University of Minnesota Masonic Children’s Hospital, also provided an overview of GI dysfunction and nutrient malabsorption, focusing on cystic fibrosis (CF). The third speaker was Dale Lee, Assistant Professor of Pediatrics at the Seattle Children’s Hospital. He presented an overview of nutritional requirements for inflammatory bowel disease (IBD). In the following presentation, Martha Field, Assistant Professor in the Division of Nutritional Sciences at Cornell University, discussed blood–brain barrier dysfunction and resulting brain nutrient deficiencies. The final presentation, which covered macronutrient and micronutrient homeostasis in the setting of chronic kidney disease (CKD), was provided by Alp Ikizler, Professor of Medicine at the Vanderbilt University School of Medicine. A moderated panel discussion and question and answer session concluded the session.
EXAMPLES OF GASTROINTESTINAL DYSFUNCTION AND MALABSORPTION OF NUTRIENTS: INTESTINAL FAILURE1
Intestinal failure is characterized by a reduction of functional intestinal mass necessary for adequate digestion and absorption to meet nutrient, fluid, and growth requirements. By definition, Duggan explained, the condition requires specialized nutritional support through either the enteral or parenteral route. Intestinal failure has three physiological classifications: short bowel syndrome, GI motility disorders, and intestinal epithelial defects. Duggan stated that his presentation would focus mainly on surgical short bowel syndrome, a condition resulting from massive resection of portions of the GI tract.
Duggan noted that short bowel syndrome has been defined as intestinal loss due to acquired or congenital disease leading to dependence on parenteral nutrition for more than 90 days. He noted that this 90-day cutoff is admittedly arbitrary but seems to correlate with the occurrence of a number of side effects of long-term parenteral nutrition so is considered to be clinically reasonable. Some papers have used shorter cut-off times (e.g., 60 days) or anatomical definitions.
Duggan explained that until relatively recently, more than one-half of pediatric patients with short bowel syndrome either died or underwent multivisceral transplantation, which involves transplantation of three or more abdominal organs (Squires et al., 2012). As care of patients with intestinal failure has improved, the survival rate has continued to increase, to the point that some centers are publishing 90 percent or near 100 percent survival rates, even among those with chronic, refractory disease (Duggan and Jaksic, 2017).
Patients with intestinal failure have massive bowel loss or intestinal dysfunction which, depending on extent and location of resection, leads to malabsorption of all three classes of macronutrients and many micronutrients. Therefore, the mainstay of intestinal failure treatment, Duggan explained, is early and aggressive parenteral nutrition, with gradually increasing amounts of enteral nutrition as the bowel adapts after massive resection. Micronutrient supplementation is an important therapy as well. A number of medical and surgical therapies for underlying complications from intestinal failure are being developed, including new hormone analogs for glucagon-like peptide-2, surgical therapies that taper and lengthen the bowel, and multivisceral transplantation. The underlying and perhaps most effective therapy, however, remains supporting the body’s natural process of intestinal adaptation (i.e., the process wherein residual bowel becomes more efficient at absorbing nutrients).
___________________
1 This section summarizes information presented by Christopher Duggan.
In addition to the malabsorption of nutrients, complications of intestinal failure include liver disease and fat malabsorption due to pancreatic insufficiency. Duggan argued that intestinal failure-associated liver disease seems to be related largely to toxicity of additives in long-term parenteral nutrition, as opposed to nutrient deficiencies. These findings, he said, raise the issue of whether the route of administration of nutrients needs to be considered when determining nutrient requirements and toxicities.
Nutritional requirements in patients with intestinal failure are different from those of a healthy population. “Total” parenteral nutrition, however, is a misnomer, in that it does not meet all nutrient requirements. In addition, Duggan said, nutritional therapy for intestinal failure is a supportive, not curative, therapy per se (unless a patient has severe acute malnutrition or important micronutrient deficiencies). A retrospective review of children with intestinal failure at Boston Children’s Hospital showed a high prevalence of various micronutrient deficiencies, even while the patient was receiving parenteral nutrition at least several days per week at a center that takes special care to maintain children on micronutrient supplementations as they are weaning from parenteral nutrition (Yang et al., 2011; see Figure 3-1).
Duggan went on to say that recommendations for pediatric parenteral nutrition intakes from groups such as the American Society for Parenteral and Enteral Nutrition have shifted over time depending on the nutrient under study, that these recommendations have often been based on expert opinion, and that they are often derived from clinical observations of nutrient deficiency states, and/or by extrapolation from adult or animal data.
Supporting data for different nutritional requirements for patients who are dependent on intravenous nutrition or transitioning from parenteral to enteral nutrition are limited and often derived from empiric observations, such as lack of weight gain or lack of height gain in children and infants, and laboratory data in others. Requirements for many nutrients have been demonstrated when the omission of important vital nutrients resulted in clinically apparent deficiencies. Data for conditionally essential nutrients in the context of intestinal malabsorption or dependence on parenteral nutrition are also weak. Glutamine supplementation offers a perfect example of these issues, Duggan explained. In this case, animal and other data on the requirement for glutamine in the study of GI diseases are quite voluminous, but when the nutrient was examined carefully in large randomized trials, no positive effect was seen from parenteral glutamine supplementation among premature infants.
Duggan concluded his presentation by noting that although patients with intestinal failure have taught the nutrition community a substantial
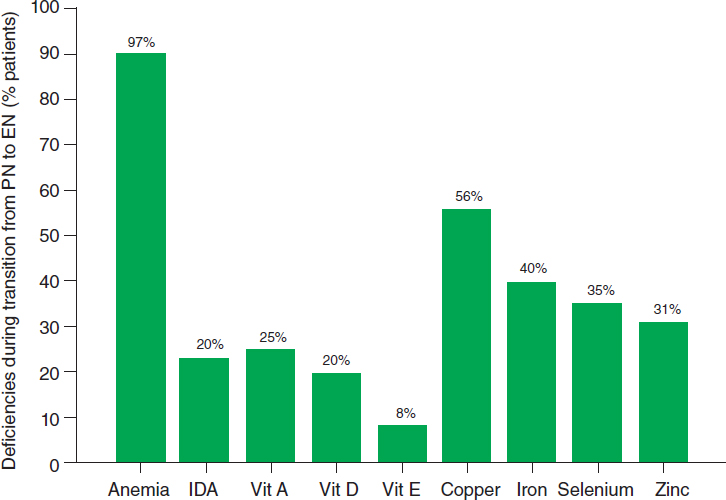
NOTE: EN = enteral nutrition; IDA = iron deficiency anemia; PN = parenteral nutrition.
SOURCES: As presented by Christopher Duggan, April 2, 2018; Yang et al., 2011. Reprinted from Journal of Pediatrics, volume 159, issue 1, Yang, C. F., D. Duro, D. Zurakowski, M. Lee, T. Jaksic, and C. Duggan, High prevalence of multiple micronutrient deficiencies in children with intestinal failure: A longitudinal study, pages 39–44, Copyright 2011, with permission from Elsevier. https://www.sciencedirect.com/journal/the-journal-of-pediatrics (accessed June 6, 2018).
amount about nutrient needs in health and disease, it is difficult to study nutrient needs in this condition because of the
- rarity of the condition;
- small size of infants and concomitant limited availability of blood and other biologic specimen volumes;
- lack of valid biomarkers for nutritional status;
- heterogeneity of intestinal absorptive capacity among subjects; and
- limited ability to perform cross-over studies because the majority of patients are growing children and are difficult to consider in steady state.
EXAMPLES OF GASTROINTESTINAL DYSFUNCTION AND MALABSORPTION OF NUTRIENTS: CYSTIC FIBROSIS2
CF occurs in about 1 in 3,500 individuals in the United States and Europe, and is one of the more common genetic diseases. It is caused by loss-of-function mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. Approximately 1,000 genetic mutations have been demonstrated in the CFTR gene. Many of them lead to clinical disease with variable consequences.
The CFTR protein mediates the secretion of chloride and bicarbonate as well as water across the epithelial layer. Loss of this function results in progressive obstructive lung disease that generally leads to death in the fourth decade. Management of CF generally involves maintaining pulmonary function and controlling infections.
In the United States, care for about 85 percent of CF patients is provided through Cystic Fibrosis Foundation care centers, which also includes a patient registry with data on infections, nutrition, liver disease, pulmonary function, and other information. These data allow care centers to compare survival, body mass index (BMI), 1-second forced expiratory volume, and many other aspects of CF.
Nutrition-Related Features of Cystic Fibrosis
Schwarzenberg explained that although CF is predominantly a lung disease, the disease also has the following significant impact on nutrition:
- People with CF require more calories than average because of increased pulmonary effort and chronic and recurrent inflammation. At the same time, they often have poor appetite and intake.
- People with CF often have abnormalities of motility, diminished sense of smell, abdominal pain and depression, malabsorption related to pancreatic insufficiency, and small bowel overgrowth. They also have a unique form of diabetes and a unique form of liver disease, each of which affects nutrition. CF-related diabetes affects about 50 percent of people with CF over the lifespan, mainly as the result of the loss of islet function in the pancreas.
- Most individuals with CF are pancreatic insufficient at birth, and 85 percent will be pancreatic insufficient by age 1. Clinicians try to offset this through pancreatic enzyme replacement therapy, but even at its most successful, this therapy allows patients to digest about 95 percent of the fat ingested.
___________________
2 This section summarizes information presented by Sarah Jane Schwarzenberg.
- Chronic intestinal inflammation, a less well recognized problem than pancreatic insufficiency, often occurs. This leads to thick and dehydrated intestinal mucus and to bacterial growth against the intestinal lining. The resulting dysbiosis is exacerbated by the repeated antibiotic use, ending in intestinal lesions with symptoms such as diarrhea, bloating, nausea, abdominal pain, and distention.
- Intestinal bicarbonate in enterocytes and pancreas is decreased. This is important for absorption of fat (absorption of fat requires the production of mixed micelles with bile acids, and bile acids do not remain in solution at low pH) and for the activity of the enteric-coated pancreatic enzymes used today (they will not dissolve in acidic pHs).
- Patients with CF can develop a unique form of liver disease. This focal biliary fibrosis and multilobular cirrhosis, which can occur with or without portal hypertension, is the third leading cause of death in CF. The main issue for people with CF and nutrition is the reduction in the production of bile salts associated with liver disease.
- Dysbiosis results in intestinal dysmotility and gastroparesis. Gastroparesis affects about one-third of individuals with CF, resulting in nausea, early satiety, persistent dyspepsia, and abdominal pain.
- Sinus disease is also common in CF and manifests through nasal polyps that impair the sense of smell, contributing to the lack of appetite and thick retropharyngeal mucus.
Evidence since the 1980s has indicated that maintenance of weight greater than the 50th percentile for age and sex is associated with longer survival. Good nutritional management may result in an additional decade of life, which is quite significant in CF. For example, a prospective observational study using data from the Cystic Fibrosis Foundation registry (see Figure 3-2) showed that pulmonary function was much lower in CF patients with a weight-for-age percentile less than 10 percent at age 4. This outcome tracked through 18 years of life. The main takeaway, noted Schwarzenberg, is that clinicians should try to keep patients on the same weight-for-age percentile throughout life. Many healthy children grow on the 10th percentile, but CF patients must be at the 50th percentile or greater in order to achieve the longest life possible.
Cystic Fibrosis Nutrition Interventions
CF-specific nutritional interventions are driven by quality improvement projects and guidelines developed by the Cystic Fibrosis Foundation. It is well established that children who obtain newborn screening
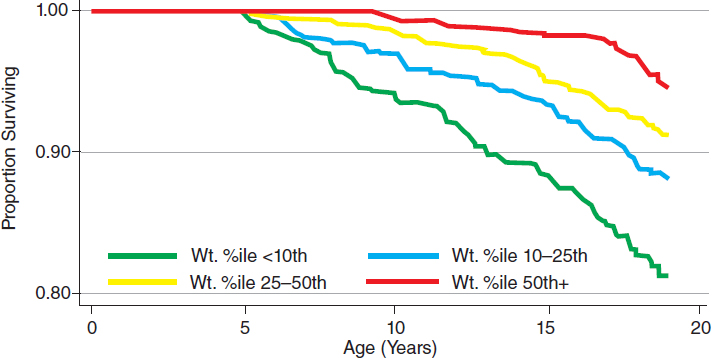
NOTE: Survival was highest in patients with better weight-for-age percentiles at 4 years of age.
SOURCES: As presented by Sarah Jane Schwarzenberg, April 2, 2018; Yen et al., 2013. Reprinted from Journal of Pediatrics, volume 162/issue 3, Yen, E. H., H. Quinton, and D. Borowitz, Better nutritional status in early childhood is associated with improved clinical outcomes and survival in patients with cystic fibrosis, pages 530–535, Copyright 2013, with permission from Elsevier. https://www.sciencedirect.com/journal/the-journal-of-pediatrics (accessed June 6, 2018).
and who are introduced to good nutrition before the age of 2 or 3 months have better health outcomes than children whose diagnosis is made even a short time later. Children are asked to eat a high-fat diet to increase calories, and to eat three meals and three snacks every day. Schwarzenberg noted that this is very hard for many patients. Patients are given water-miscible fat-soluble vitamin supplementation because they generally cannot adequately absorb ordinary vitamin supplements.
Schwarzenberg then briefly mentioned some interventions that improve the function of some gene mutations in CF. One in particular is ivacaftor, which makes gating mutations less injurious. These mutations occur in about 4 to 6 percent of people with CF, but people on ivacaftor experience improved lung function and reduced sweat chloride and they gain weight, perhaps due to the release of bicarbonate into the GI tract.
Research Gaps
Schwarzenberg admitted that the research gaps in CF and nutrition are numerous, but she noted that the following questions highlight key gaps:
- What is the cause of dysbiosis and dysmotility in CF, and how is it best managed?
- What is the appropriate fat type for people with CF to ensure adequate essential fatty acids without increasing inflammatory products?
- How should nutrition be addressed as patients age and new complications occur?
- What effect will new modulators of CFTR have on nutrition?
Schwarzenberg concluded her presentation by summarizing that in CF, better early childhood nutrition is associated with better height growth, better lung function, and improved survival into adulthood. However, optimal nutrition in CF is not a simple matter of increased calories. Clinicians must pay attention to a complex disordered matrix of digestion, absorption, and intestinal function, and that is really the challenge in this disease.
NUTRITIONAL REQUIREMENTS FOR INFLAMMATORY BOWEL DISEASE3
IBD is a chronic inflammatory condition with symptoms including abdominal pain, diarrhea, and blood in the stool, and systemic manifestations such as fever, body rashes, linear growth delay, and poor weight gain. IBD has two types. Crohn’s disease involves any portion of the GI tract, from mouth to anus, and ulcerative colitis is confined to the colon. IBD arises in the setting of a genetic predisposition, but numerous environmental insults can occur to trigger a perpetuating cycle of inflammation. At least 200 known genes are associated with IBD risk, explained Lee. The known environmental risk factors include antibiotic exposures, certain dietary exposures, and infections. When these environmental exposures occur in the setting of a genetic risk, a breach of the epithelial barrier of the GI tract can occur. Translocation of microbial products may occur, which stimulates the immune system to react and develop an inflammatory reaction. The chronic inflammation in IBD can lead to complications, including scarring and infections in the GI tract and increased risk of intestinal cancers.
Lee stated that epidemiological studies have shown that diet and nutrients are important for IBD. Immigration studies also have implicated the role of environment and diet. In addition, it is known that exclusion diets can be effective in treating IBD. Although several risk factors (e.g., total saturated fat, total polyunsaturated fatty acids, omega-6 poly-
___________________
3 This section summarizes information presented by Dale Lee.
unsaturated fatty acid, and meats) and protective dietary factors (e.g., fiber, fruits, vegetables, and omega-3 polyunsaturated fatty acids) have been identified, it is unclear whether these are actually risk and protective factors, or whether individuals at risk of developing IBD merely have different nutrient requirements.
Nutrient Deficiencies Associated with Inflammatory Bowel Disease
It is known that individuals with systemic inflammation and IBD have anorexia, so they have decreased global intake of macro- and micronutrients. Individuals with IBD also often have malabsorption and resultant diarrhea and deficiency of fat-soluble vitamins and zinc. Intestinal blood loss with IBD can lead to iron deficiency. The chronic systemic inflammation that occurs in IBD can lead to anemia and activated 1,25 hydroxy vitamin D deficiency. With Crohn’s disease in particular, the most common area of IBD inflammation is the ileum, or the distal small bowel, and as a result, patients can develop vitamin B12 deficiencies.
Role of Nutrient Supplementation
In general, the current approach to treating IBD is to focus on controlling the active inflammation, which can help improve nutrient status. As an adjunct, supplementation with specific nutrients, such as vitamin D, iron, vitamin B12 injections, zinc, and a variety of other B vitamins, may be recommended.
Lee elaborated by discussing vitamin D and the risk of developing IBD, using the Harvard Nurses’ Health Study to illustrate. Among study participants, the risk of developing Crohn’s disease in the highest quartile of serum vitamin D appeared to be lower than for the lowest quartile. When a similar study was done analyzing for ulcerative colitis, investigators did not find significant associations, suggesting that Crohn’s and ulcerative colitis, though both IBDs, have a different pathogenesis. Some trials that have explored the role of vitamin D supplementation in IBD have suggested that increased vitamin D is associated with a decreased risk of disease relapse, and in children, that markers of inflammation are reduced. In terms of iron deficiency, production of hepcidin, a protein produced by the liver that causes decreased uptake of iron from the GI tract and decreased mobilization of iron from the liver and spleen, can be induced in IBD.
Treatment
Switching gears, Lee described the strategies for treating IBD. The mainstream treatment is the use of immunosuppressive medications, such
as corticosteroids, immunomodulators, and biological medications. In terms of nutritional therapy, exclusive enteral nutrition (EEN) therapy is a formula-based approach. A number of specific exclusion diets used for IBD have stimulated provocative, relatively large-scale studies.
Lee explained that EEN therapy for IBD involves consumption of a defined formula (by mouth, delivered by nasogastric tube, or delivered by a gastrostomy tube) that accounts for the patient’s entire nutritional needs. Studies have demonstrated that the type of formula is not important, in that it can have intact protein or protein hydrolysate with different carbohydrate and fat content. The formula supplements calories and controls the inflammation in Crohn’s disease. Studies in children with Crohn’s have demonstrated that EEN is associated with about 80 percent induction of clinical remission. In one Italian study, comparing a group using EEN and controls, both groups achieved fairly good clinical remission rates (greater than 60 percent), but the EEN group had greater than 70 percent mucosal healing, whereas the corticosteroid group had only 30 percent mucosal healing (Borrelli et al., 2006). These results, noted Lee, suggest that the luminal delivery of nutrition was superior to broad immunosuppression of the corticosteroids.
Lee also described a second study, the PLEASE study, in which children with active Crohn’s disease were divided into three groups—partial enteral nutrition, EEN, and a biological medication (Lee et al., 2015). The outcomes examined were clinical disease activity and fecal calprotectin, a surrogate marker of intestinal inflammation that can be measured in the stool. Results showed outcomes for the EEN group were clearly superior to the partial enteral nutrition group for both outcomes.
Elaborating on the point that appropriate delivery of nutrition is important, Lee noted that studies have looked at allowing nothing by mouth and delivering nutrition solely through the intravenous route. Results showed that for individuals with IBD, this approach would greatly benefit overall nutritional status but would not affect the eventual need for surgery for these patients. Clearly, Lee noted, the contents in the lumen of the GI tract are important to intestinal health. It is known, for example, that fiber is fermented to short-chain fatty acids and short-chain fatty acids provide an energy source to intestinal epithelial cells, and they also have a role in promoting immune tolerance.
Knowing that a formula-based treatment approach works, the logical next question, noted Lee, is whether food-based dietary therapy for IBD also works. A few food-based dietary approaches have been gaining in traction, including a specific carbohydrate diet, the Crohn’s diseases exclusion diet, semi-vegetarian diets, and a variety of different anti-inflammatory diets. Each of these diets involves the restriction of certain food groups. The most commonly avoided food groups are bread and
gluten and also “processed foods,” a broad, poorly defined category. On the other hand, restricted diets are of some concern because they eliminate entire food groups from the diet, which can be problematic for nutritional adequacy and emotional well-being of individuals.
The specific carbohydrate diet, which was initially proposed as a dietary therapy for celiac disease, has an extremely broad following for a variety of different GI illnesses. However, rigorous studies need to be conducted to determine their usefulness in this context. Some studies, mostly case series, conducted on the effect of the specific carbohydrate diet on clinical disease activity indices as well as laboratory parameters suggest that patients experienced some improvement. To help answer some questions regarding the carbohydrate diets, the results of two ongoing large, multi-center trials will be helpful. An N-of-1 study with children is exploring the specific carbohydrate diet. (For more information on N-of-1 studies, see Nicholas Schork’s presentation in Chapter 5.) In the adult arena, the University of Pennsylvania has designed a multi-center study looking at the specific carbohydrate diet versus the Mediterranean diet. This study will look at objective markers of inflammation, clinical disease activity, and the microbiome.
Complexities of Inflammatory Bowel Disease and Nutrients
Lee then summarized the following complexities with IBD and nutrients:
- Nutritional therapy is used differently for Crohn’s and ulcerative colitis and the formula-based approach (i.e., EEN) is effective only for Crohn’s disease.
- Genetic polymorphisms are known to be associated with the efficacy of the formula-based approach. The NOD2 gene is involved with bacterial product recognition. Polymorphisms of this gene have been associated with efficacy of EEN therapy.
- In active inflammation versus quiescent disease, vitamin D and iron may be processed differently. For vitamin D, individuals with active inflammation have a decreased parathyroid hormone activity and decreased 1-alpha-hydroxylation of 25-hydroxy vitamin D at the renal level.
Gaps in Knowledge
Finally, Lee noted the following gaps in current understanding about IBD and nutrition:
- The role of specific dietary components on IBD pathogenesis needs to be better elucidated. These components go beyond macro- and micronutrients to include food additives and products generated during cooking.
- Better methodologies, specifically biomarkers, are needed to assess dietary exposures.
- A better understanding of the interplay among diet, the intestinal microbiome, and the metabolome is needed, as these data will help inform individual-focused interventions.
BLOOD–BRAIN BARRIER DYSFUNCTION AND RESULTING BRAIN NUTRIENT DEFICIENCIES4
Field began her remarks by showing a figure that illustrates the relationships between disease etiology, physiological impact on nutrients and function, and impact on human nutrition and on biomarkers (see Figure 3-3).
Field noted that the framework shown in the figure provides the context to examine how several disease-related factors, including inflammation, genetic predisposition, autoimmunity, and mitochondrial dysfunction, specifically affect the blood–brain barrier (BBB) and then influence cerebral folate deficiency (CFD).
Cerebrospinal fluid (CSF) serves as a nourishing fluid for the brain and a way to remove waste products. The epithelial and endothelial barriers, which are components of the BBB, are critical for maintaining brain nutritional status as they serve to concentrate nutrients through a variety of mechanisms (e.g., vitamin C and folate are concentrated fourfold in the CSF relative to the serum; many B vitamins are concentrated, some as much as 50-fold). BBB function, however, declines as a result of disease (e.g., Alzheimer’s disease, multiple sclerosis, human immunodeficiency virus infection), inflammation, or aging. This dysfunction can occur due to a loss of transport protein expression or erosion of the tight junctions in the BBB that prevent nutrients or other molecules from leaking out of the brain.
CFD, which can result from BBB dysfunction, affects the metabolic functions of folate within the brain. Folates carry and chemically activate one-carbon units for biosynthetic reactions. More specifically, folate is needed to synthesize purines and thymidylate and for the remethylation of homocysteine to methionine. Methionine is a precursor of S-adenosylmethionine, which is required for methylation of deoxyribonucleic acid (DNA), and histones, and in synthesis of neural transmitters. Folate is transported across the BBB
___________________
4 This section summarizes information presented by Martha Field.
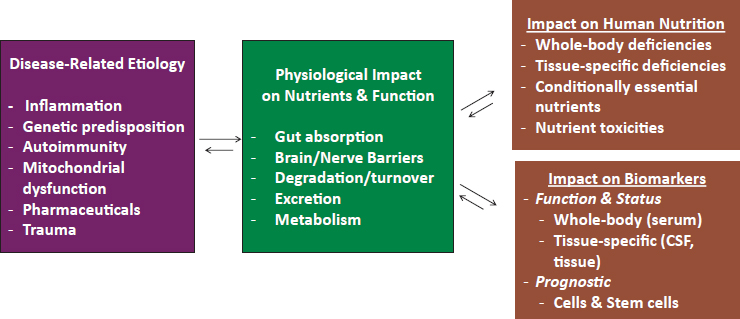
NOTE: CSF = cerebrospinal fluid.
SOURCE: As presented by Martha Field, April 2, 2018.
to the CSF at the choroid plexus by receptor-mediated endocytosis, and this process is mediated by folate receptor alpha in an adenosine triphosphate (ATP)-dependent manner. Because of the difference in folate concentration in the CSF relative to the blood, CSF folate levels indicate brain folate status independent of indicators of whole-body folate status.
Inborn Errors of Metabolism and Cerebral Folate Deficiency
Field noted that a number of studies demonstrate that individuals with inborn errors of metabolism in the folate receptor or with genetic disorders that cause mitochondrial DNA dysfunction—in other words, either a mutation in the receptor or a loss of ability to generate ATP—develop CFD and exhibit CSF folate levels much lower than normal (around 100 nanomolar). Successful intervention for CFD requires high-dose folinic acid, a reduced form of folate. Folic acid, a synthetic form of folate, is highly bioavailable and chemically stable but not as effective as folinic acid or methyl folate (a B vitamin form in fruits and vegetables) at improving CSF folate levels, possibly due to the ability of folic acid to bind tightly to the receptor, impairing transport of methyl folate across the BBB. In fact, at the very high folic acid doses that are shown to be effective in addressing CFD, folic acid can be associated with some toxicity.
Field noted that the relationship between impairments in whole body folate status and brain disease or, more specifically, depression, have been recognized by authoritative bodies, including the American Psychiatric Association, the Institute of Medicine, and the World Health Organization
(APA, 2010; de Benoist, 2008; IOM, 1998). She then went on to explain how this association between whole body folate status and depression parallels with inborn errors of metabolism and a tissue-specific nutrient deficiency. A genetic component to the link between CFD or between low folate status and depression exists. One common genetic variant, a single nucleotide change in the MTHFR gene (MTHFR C677T), affects the functioning of folate-mediated one-carbon metabolism and folate status. Neural tube defect risk is doubled for carriers of the TT genotype, which is also associated with increased risk of miscarriage. Interestingly, in folate-sufficient adults, the TT genotype is protective against colon cancer.
Because of its effect on lowering folate status, the MTHFR T allele has been associated with increased risk of developing both depression and schizophrenia, and up to 70 percent of patients with either depression or schizophrenia have some form of the polymorphism (i.e., one or two T alleles). On the other hand, major depressive disorder, projected to be the second leading global disease burden by 2020, is responsive to standard medication in less than one-half of all patients. The etiology of major depressive disorder is poorly understood but is likely to involve a complex interplay of sensitivities to stressors and alterations in metabolic pathways. Some evidence suggests that BBB integrity is compromised in depressed patients. Several informative clinical trials to examine folate-related treatments for depression suggest that both the form of folate and the dose influence the efficacy for folate in improving depression-related outcomes (Alpert et al., 2002; Papakostas et al., 2012, 2014). When describing results from the trials, Field emphasized that the doses used are very different than requirements in healthy individuals, by more than an order of magnitude. She also noted that although the origins of CFD are very different than those caused by inborn errors of metabolism, the treatments that result in clinical improvement are essentially the same.
Field then provided the following summary of her presentation:
- Brain-specific folate deficiency can occur in the absence of whole body deficiency in inborn errors of metabolism and in adult disease, although whole body deficiency could exacerbate the condition.
- BBB folate transport defects can be overcome with high levels of folate. Folate form or composition matters for efficacy. Reduced folates are efficacious; folic acid is not.
- This evidence clearly indicates a different nutritional requirement in these conditions than for healthy population.
- The disease states are responsive to intervention, but some individuals are responders and others are not. There does seem to be evidence of dose response.
- Understanding these relationships requires an understanding of biomarkers and rigorous, well-designed controlled trials.
Field concluded her presentation by reiterating that several nutrients, not only folate, are concentrated within the brain, and that little is currently known about how these nutrients are affected by disruptions in BBB activity. It is also not well understood whether repairing the barrier can reverse the nutritional deficiency and/or whether restoring brain nutrient levels can augment BBB repair.
MACRO- AND MICRONUTRIENT HOMEOSTASIS IN THE SETTING OF CHRONIC KIDNEY DISEASE5
In contrast to most of the conditions discussed in previous presentations, Ikizler stated that kidney disease is very common. About 20 million individuals in the United States have CKD. The mortality rate in the setting of end-stage renal disease is about 20 percent per year. Ikizler also noted that, as with any other chronic disease state, nutrition is one of the most important, if not the most important, predictors of outcomes.
Turning his attention to the nutrition aspects of chronic kidney disease, Ikizler noted that protein energy wasting (PEW) is highly prevalent in this patient population (about 12 to 18 percent of all patients with CKD not yet on maintenance dialysis and 30 to 50 percent for patients on dialysis). Kidney disease leads to PEW for multiple reasons. Ikizler observed that in developed countries, PEW, sarcopenia, wasting syndrome, and malnutrition predispose patients to significant complications that are associated with PEW, such as infection, cardiovascular disease, and frailty, that in aggregate or individually put CKD patients at increased mortality risk.
Evidence of Unique Nutritional Requirements in Chronic Kidney Disease
A number of studies have established the need for more or less of the nutrients that are usually given to healthy individuals6 in various disease states, including CKD (see Table 3-1).
In describing this table, Ikizler noted that protein requirements are significantly altered at different stages of kidney disease and different from the needs of the general population. Dietary requirements (e.g., sup-
___________________
5 This section summarizes information presented by Alp Ikizler.
6 For Dietary Reference Intake (DRI) values, see http://nationalacademies.org/HMD/Activities/Nutrition/SummaryDRIs/DRI-Tables.aspx (accessed June 6, 2018).
TABLE 3-1 Summary of Nutrient Requirements in Chronic Kidney Disease
| Non-Dialysis CKD | Hemodialysis | Peritoneal Dialysis | |
|---|---|---|---|
| Energy | 30–35 kcal/kg/day | 35 kcal/kg/day | 35 kcal/kg/day including kcal from dialysate |
| Protein | 0.6–0.8 g/kg/day Illness 1.0 g/kg | >1.2 g/kg/day | >1.2 g/kg/day Peritonitis >1.5 g/kg |
| Sodium | 80–100 mmol/day | 80–100 mmol/day | 80–100 mmol/day |
| Potassium | <1 mmol/kg if elevated | <1 mmol/kg if elevated | Not usually an issue |
| Phosphorus | 800–1,000 mg and binders if elevated | 800–1,000 mg and binders if elevated | 800–1,000 mg and binders if elevated |
NOTE: CKD = chronic kidney disease.
SOURCES: As presented by Alp Ikizler, April 2, 2018; Cano et al., 2006; Fouque et al., 2007; NKF/KDOQI, 2000, 2002.
plementation, limitations, or restrictions) must be determined based on each patient’s background and level of kidney function. Studies suggest that higher protein intakes, greater than 1.0 or 1.2 grams per kilogram per day, could be associated with progression of kidney disease. In contrast, patients with end-stage renal disease experience the syndrome of protein wasting and they have substantial need for additional protein intake. Other evidence suggests that patients with obesity who have end-stage renal disease do better than patients with normal weight. In the setting of CKD, however, patients with obesity actually experience much faster progression of their kidney disease than patients with normal weight.
Nutrition Issues in Chronic Kidney Disease
Ikizler continued by explaining that CKD has three nutrition issues of relevance: changes in requirements for protein intake, hemodialysis-associated catabolism, and the effect of inflammation and how it influences the metabolic profile of people with kidney disease.
He reminded workshop participants that the hallmark of uremia (or uremic syndrome, that is, urea in the blood due to an excess of amino acid and protein metabolism into end-products in the blood that would be normally excreted in the urine) is anorexia. The initiation of dialysis in CKD patients with uremia occurs when they became anorexic and can-
not eat adequately. This decrease in nutrient intake, especially protein, progresses as the glomerular filtration rate (GFR) goes down from greater than 60 to 30 or less. The initiation of dialysis reverses this problem to some extent, but dialysis in itself is inadequate to replace original kidney function, and the initial improvement subsides over the course of 3 to 4 years. Restricting protein in earlier stages of kidney disease limits the amount of protein being metabolized and the uremic toxins that are produced through this process are decreased, which results in less nausea and other uremic symptoms.
Another problem with maintenance dialysis is that in addition to filtering out undesirable molecules, it also eliminates other small molecules, such as essential amino acids. The resulting diminished amino acid pool leads to increased catabolism and decreased protein synthesis, which results in robust net catabolism. Results from a study that examined protein turnover using stable isotopes show a significant drop in the protein synthesis and a significant increase in protein breakdown, resulting in an almost doubling of the net catabolic process (Ikizler et al., 2002).
Dialysis itself causes a loss of protein and the catabolic process continues after dialysis is completed, probably because the complement system and subsequently the inflammatory pathway (interleukin [IL]-6 and other inflammatory cytokines) become activated. In kidney disease, the average C-reactive protein or IL-6 concentrations are threefold or higher than for healthy individuals, leading to poor outcomes.
Responsiveness of Chronic Kidney Disease to Nutritional Intervention
CKD responds to specific nutritional interventions, Ikizler explained, but like many other aspects of this disease, it is complex. Limiting protein intake to less than 0.6 g/kg/day is desirable in CKD for the following reasons:
- It delays the initiation of uremic syndrome.
- It preserves kidney function by decreasing the load on remaining nephrons.
- It improves proteinuria/albuminuria.
- It improves metabolic profiles (i.e., decreased inflammation and oxidative stress, and increased insulin sensitivity) of CKD patients.
In fact, protein restriction may delay GFR decline by an average of 0.5 mL/min/year. However, Ikizler stated, the opposite is true for dialysis patients. That is, providing additional protein to individuals during dialysis at the setting with catabolic effect can completely reverse
the process and initiate a robust anabolic response (Deger et al., 2017). A number of randomized controlled trials (RCTs) suggest that benefits to the intermediate surrogate outcomes of nutritional state, such as serum albumin, pre-albumin, body weight, lean body mass, bone density, and physical function, occur with protein supplementation. A recently completed study examined the effects of nutritional supplementation using a dual glucose-amino acid clamp procedure on changes in muscle protein signaling (Gamboa et al., 2016). In contrast to healthy controls, who showed an anabolic response, dialysis patients had no response. An examination under electron microscopy showed significant problems in the muscle tissue mitochondria that limits the full benefits of this nutritional supplementation in the setting of CKD. No information is available from RCTs on fat intake in the setting of CKD. Current recommendations follow those for the general population: low consumption of saturated fat and possible inclusion of omega-3 fatty acids. In terms of vitamins, the recommendation is that people with kidney disease should pay attention to intake of water-soluble vitamins because they are usually lost in dialysis, even though no evidence suggests that stores of these vitamins are extremely depleted. On the other hand, care must be taken with fat-soluble vitamins because they accumulate in the setting of kidney disease, especially vitamin A. Usually only vitamin D is supplemented. No information is available regarding trace elements in this context.
Gaps in Knowledge
Ikizler ended his presentation with a very brief roundup of current gaps in knowledge, including
- specific role of uremic toxins and the human microbiome;
- how to improve the dialysis process;
- optimal level of supplementation;
- the role of anabolic or anti-catabolic strategies; and
- obesity in CKD.
MODERATED PANEL DISCUSSION AND Q&A
Kemper initiated the discussion with a question to Duggan about whether it would ever be possible to have a scientific approach to determining what supplements an individual with intestinal failure needs. For example, would it be possible to take into account factors such as a patient’s age, weight, how much residual bowel they have left, and comorbidities and determine a formula to figure out how much of these various nutrients they need? Duggan replied that that kind of prognostic
equation would require much larger sample sizes and detailed intake data than are currently available but that it is a useful question to ponder.
On a question related to the response to nutrients in short bowel syndrome, when the metabolic homeostasis has been altered because of tissue loss, Duggan responded that a common etiology of gut resection in neonates is necrotizing enterocolitis, wherein resection preferentially occurs in the ileum and colon, which is exactly where intestinal L cells secrete glucagon peptide 2. An analog of this peptide has shown promise in adults but only limited data are available in children. Thinking more broadly, Duggan stated that the issue of whether the malabsorption of various nutrients can have a deleterious effect on intestinal adaptation is a little less clear. Although animal models have shown that vitamin A deficiency, for instance, reduces epithelial turnover, Duggan has not seen any data to suggest that high-dose vitamin A improves intestinal adaptation. Zinc is an important trace element as well for intestinal adaptation, but the animal model suggesting that profound zinc deficiency leads to decreased adaptation has not been well addressed in human trials, to his knowledge. Duggan also commented that some animal data suggest that bacterial overgrowth seems to relate to non-alcoholic fatty liver disease, acknowledging that valid methods to diagnose bacterial overgrowth in humans has been the main conundrum there.
Kemper noted a number of common elements, including the importance of determining not only the right nutrient or supplement to give but considering the right form and delivery system, and the important effect that inflammation seems to have on special nutritional requirements. Although levels of substances are generally measured in blood, plasma, or serum, levels in tissues are what is really desired. However, some tissues, such as brain, are difficult to sample. Field replied that the starting point with respect to tissues that are difficult to sample are mechanistic studies in animal models that provide a good understanding of the biology followed by studies of surrogate biomarkers.
Special Nutrient Requirements for Metabolic Problems Not Directly Involving a Nutrient
An enquiry was raised on whether it is possible to have a special nutrient requirement in a metabolic problem that does not directly involve a nutrient. That is, can one just account for an absorption problem, or is there another way of thinking about the special nutrient requirement as it is affected by the metabolic state? Schwarzenberg replied that at least for CF the problem is more complex than just malabsorption. Although it is important management in CF to take into account the digestive and
absorptive matrix abnormalities in the GI, nutrients are also being affected by the CFTR abnormality itself and by inflammation.
Ikizler addressed a related question regarding the role of inflammation in the diseases discussed during this session. From the kidney disease perspective, controversy exists over whether inflammation is the root cause of death in kidney disease patients. Although this can be determined only by examining the effect of anti-inflammatory interventions in the target outcome, he admitted that these studies are extremely complex because of the many outcomes involved that are interrelated and that might influence other outcomes or the disease process as a whole.
Evidence on Nutritional Factors That Trigger or Help the Disease Process
To a question related to whether any evidence exists that separates nutritional factors that trigger the disease versus those involved in treating or managing the disease, Lee responded that the evidence about risk and protective factors comes from large epidemiological studies that do not provide high-quality, detailed assessment of dietary data. The epidemiological studies cited in his presentation probably reflect both risk and protective factors and are probably pertinent for inception of disease and treatment. Lee added that when people think about nutrients, they generally think of the classically described macronutrients and micronutrients. However, food products consumed today include many other components that are not classified as nutrients and that need to be better understood.
The importance of studying not just single nutrients but patterns of dietary intake and how foods interact with each other was also raised. Schwarzenberg added that the dimension of time is also important to consider. An acute problem may be resolved, but the underlying pathologic process may continue for some time.
Conversion of Folate
A question was raised regarding the metabolic or environmental difficulties in the conversion of folate to folinic acid. Field replied that the fact that conversion of folic acid to the reduced form is saturable makes it very hard to get the high blood folate levels (above the Tolerable Upper Intake Level) needed to overcome the energetic barrier. These high levels cause GI toxicity and nephrotoxicity. However, Field continued, even at these high levels she would not consider folate to be a drug, because the nutrient requirement is a result of the disease itself (e.g., inflammation or Alzheimer’s disease) and the breakdown of the BBB.
Effect of Nutritional Therapies on Cellular Composition of the Gut
The panel was asked whether any of the nutritional therapies for the conditions discussed during the session actually change the cellular composition of the intestine, for example goblet cell concentrations. According to Lee, studies have shown that changing the diet dramatically causes alterations in the microbiome as well as improvement in inflammation, although the exact causality is not easy to decipher. Ikizler noted that some ongoing research in CKD patients is also showing changes in the gut that may be due to inflammation or uremic toxins. The discussion continued with a question about whether it was possible to formulate a food matrix that contains specific functional proteins, pectins, or other components that interact with the microbiome to develop short-chain fatty acids for certain conditions. Lee responded that this is a critically important issue, and work is under way to better understand pathophysiology and how dietary changes affect the microbiome and how that microbiome interacts with the healthy host versus the inflamed diseased host.
Protein Dietary Reference Intakes and Chronic Kidney Disease
A comment was made on how potential increases in protein Dietary Reference Intakes might affect prevention of CKD in a population with a high prevalence of overweight or obesity.7 This is an important dilemma, Ikizler replied, illustrating the balance between recommendations for the public when many people in that population have conditions, or risk factors for conditions, that would necessitate more tailored nutrition recommendations. Ikizler stated that in the era of precision medicine, individuals must be considered rather than just creating a generalized one-size-fits-all approach. There was general agreement among panel members that reducing calories overall and maintaining an appropriate distribution of macronutrients would be sufficient for individuals with obesity who might be at risk of diabetes or other chronic diseases.
Defining Malnutrition
The panel was asked to comment on how the lack of a definition of malnutrition hampers the ability to think about special nutrient requirements. Schwarzenberg responded that she was not certain that an overarching definition that would describe malnutrition in every chronic disease or every major disease is possible. Lee added that some very general
___________________
7 For DRI values, see http://nationalacademies.org/HMD/Activities/Nutrition/SummaryDRIs/DRI-Tables.aspx (accessed June 6, 2018).
criteria are available to diagnose malnutrition, such as weight for length Z scores, BMI Z scores, and weight loss over time. Astute clinicians and dieticians thinking about the appropriate trajectory for children will be needed in efforts to identify malnutrition and appropriate supplementation. Understanding the disease process is also critical.
REFERENCES
Alpert, J. E., D. Mischoulon, G. E. Rubenstein, K. Bottonari, A. A. Nierenbert, and M. Fava. 2002. Folinic acid (Leucovorin) as an adjunctive treatment for SSRI-refractory depression. Annals of Clinical Psychiatry 14(1):33–38.
APA (American Psychiatric Association). 2010. Practice guideline for the treatment of patients with major depressive disorder. Washington, DC: American Psychiatric Association.
Borrelli, O., L. Cordischi, M. Cirulli, M. Paganelli, V. Labalestra, S. Uccini, P. M. Russo, and S. Cucchiara. 2006. Polymeric diet alone versus corticosteroids in the treatment of active pediatric Crohn’s disease: A randomized controlled open-label trial. Clinical Gastroenterology amd Hepatology 4(6):744–753.
Cano, N., E. Fiaccadori, P. Tesinsky, G. Toigo, W. Druml, DGEM (German Society for Nutritional Medicine), M. Kuhlmann, H. Mann, W. H. Horl, and ESPEN (European Society for Parenteral and Enteral Nutrition). 2006. ESPEN Guidelines on Enteral Nutrition: Adult renal failure. Clinical Nutrition 25(2):295–310.
de Benoist, B. 2008. Conclusions of a WHO technical consultation on folate and vitamin B12 deficiencies. Food and Nutrition Bulletin 29(2 Suppl.):S238–S244.
Deger, S. M., A. M. Hung, J. L. Gamboa, E. D. Siew, C. D. Ellis, C. Booker, F. Sha, H. Li, A. Bian, T. G. Stewart, R. Zent, W. E. Mitch, N. N. Abumrad, and T. A. Ikizler. 2017. Systemic inflammation is associated with exaggerated skeletal muscle protein catabolism in maintenance hemodialysis patients. JCI Insight 2(22):e95185.
Duggan, C. P., and T. Jaksic. 2017. Pediatric intestinal failure. New England Journal of Medicine 377(7):666–675.
Fouque, D., M. Vennegoor, P. Ter Wee, C. Wanner, A. Basci, B. Canaud, P. Haage, K. Konner, J. Kooman, A. Martin-Malo, L. Pedrini, F. Pizzarelli, J. Tattersall, J. Tordoir, and R. Vanholder. 2007. EBPG guideline on nutrition. Nephrology Dialysis Transplantation 22(Suppl. 2):ii45–ii87.
Gamboa, J. L., F. T. Billings, M. T. Bojanowski, L. A. Gilliam, C. Yu, B. Roshanravan, L. J. Roberts, J. Himmelfarb, T. A. Ikizler, and N. J. Brown. 2016. Mitochondrial dysfunction and oxidative stress in patients with chronic kidney disease. Physiological Reports 4(9):e12780.
Ikizler, T. A., L. B. Pupim, J. R. Brouillette, D. K. Levenhagen, K. Farmer, R. M. Hakim, and P. J. Flakoll. 2002. Hemodialysis stimulates muscle and whole body protein loss and alters substrate oxidation. American Journal of Physiology-Endocrinology and Metabolism 282(1):E107–E116.
IOM (Institute of Medicine). 1998. Dietary Reference Intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. Washington, DC: National Academy Press.
Lee, D., R. N. Baldassano, A. R. Otley, L. Albenberg, A. M. Griffiths, C. Compher, E. Z. Chen, H. Li, E. Gilroy, L. Nessel, A. Grant, C. Chehoud, F. D. Bushman, G. D. Wu, and J. D. Lewis. 2015. Comparative effectiveness of nutritional and biological therapy in North American children with active Crohn’s disease. Inflammatory Bowel Diseases 21(8):1786–1793.
NKF/KDOQI (National Kidney Foundation/Kidney Disease Outcomes Quality Initiative). 2000. KDOQI Clinical Practice Guidelines for nutrition in chronic renal failure.http://kidneyfoundation.cachefly.net/professionals/KDOQI/guidelines_nutrition/doqi_nut.html (accessed May 14, 2018).
NKF/KDOQI. 2002. Clinical Practice Guidelines for chronic kidney disease: Evaluation, classification and stratification.https://www.kidney.org/sites/default/files/docs/ckd_evaluation_classification_stratification.pdf (accessed May 11, 2018).
Papakostas, G. I., R. C. Shelton, J. M. Zajecka, B. Etemad, K. Rickels, A. Clain, L. Baer, E. D. Dalton, G. R. Sacco, D. Schoenfeld, M. Pencina, A. Meisner, T. Bottiglieri, E. Nelson, D. Mischoulon, J. E. Alpert, J. G. Barbee, S. Zisook, and M. Fava. 2012. L-methylfolate as adjunctive therapy for SSRI-resistant major depression: Results of two randomized, double-blind, parallel-sequential trials. American Journal of Psychiatry 169(12):1267–1274.
Papakostas, G. I., R. C. Shelton, J. M. Zajecka, T. Bottiglieri, J. Roffman, C. Cassiello, S. M. Stahl, and M. Fava. 2014. Effect of adjunctive L-methylfolate 15 mg among inadequate responders to SSRIs in depressed patients who were stratified by biomarker levels and genotype: Results from a randomized clinical trial. Journal of Clinical Psychiatry 75(8):855–863.
Squires, R. H., C. Duggan, D. H. Teitelbaum, P. W. Wales, J. Balint, R. Venick, S. Rhee, D. Sudan, D. Mercer, J. A. Martinez, B. A. Carter, J. Soden, S. Horslen, J. A. Rudolph, S. Kocoshis, R. Superina, S. Lawlor, T. Haller, M. Kurs-Lasky, S. H. Belle, and the Pediatric Intestinal Failure Consortium. 2012. Natural history of pediatric intestinal failure: Initial report from the Pediatric Intestinal Failure Consortium. Journal of Pediatrics 161(4):723–728.
Yang, C. F., D. Duro, D. Zurakowski, M. Lee, T. Jaksic, and C. Duggan. 2011. High prevalence of multiple micronutrient deficiencies in children with intestinal failure: A longitudinal study. Journal of Pediatrics 159(1):39–44.
Yen, E. H., H. Quinton, and D. Borowitz. 2013. Better nutritional status in early childhood is associated with improved clinical outcomes and survival in patients with cystic fibrosis. Journal of Pediatrics 162(3):530–535.
This page intentionally left blank.
























