5
Research Needs
Despite the rapid pace of research on coral biology and conservation that is occurring on a global scale, there are many gaps and unresolved issues that need to be addressed in the short and long terms. The committee’s review of the state of science in its first report (NASEM, 2019) illuminated many information gaps regarding the risks, benefits, and feasibility of the coral interventions. It is important to improve understanding of the potential interventions to inform their implementation. The best practices described in Chapter 3 and the description of the model in Chapter 4 identify priority areas where knowledge needs to be improved to create a detailed, reef-specific decision support tool. Generally, these include improved ways to identify, measure, and monitor fitness parameters of corals; greater understanding of factors that contribute to stress tolerance and associated tradeoffs for corals; and measuring the impact of interventions on demographic processes within reef ecosystems. With improved understanding, models can better predict outcomes of various reef management approaches. The committee divided research needs supporting this work into four broad categories (summarized in Box 5.1): (1) research on fundamental coral reef biology, (2) site-specific research relevant to a proposed intervention, (3) research to inform specific interventions, and (4) research to improve risk assessment and decision models. The committee ends this section by emphasizing the structural needs relevant to supporting community-based research efforts.
The decision process itself is a tool for identifying and prioritizing research needs. Articulation by stakeholders of preferred objectives and
management options may narrow the scope of research needs by clarifying the relevant modeling approaches and the most pertinent reef dynamics to resolve. Structured decision analyses can also be applied to the design of research and development strategies with the objective of delivering interventions that can more effectively support reef persistence and resilience under climate change. This can start, for example, with the identification of management strategies (using strategy-generation tables, e.g., Howard, 1988) and their ties to the research needs in Box 5.1.
RESEARCH ON FUNDAMENTAL CORAL REEF BIOLOGY
Coral reefs are intensively studied ecosystems, and much progress has been made in understanding coral ecosystem dynamics through monitoring of key reef species (see overviews in Jackson et al., 2014; Wilkinson, 2008). A focus on discovery and applied science to improve the understanding, management, and resilience of individual coral reefs, as well as the broad ecosystem, will continue (Lam et al., 2017). However, effective restoration approaches for reefs require an improved understanding of what factors underpin coral health and how these lead to reef-scale
resilience. Though these topics are inherently broad, there are a number of research areas that can be prioritized to aid the decision analysis and implementation of current and future restoration approaches identified in the committee’s work.
1. Identify the cellular mechanisms of bleaching and how these pathways are influenced by recent thermal history, host genetics, symbiont type, and microbiome.
The processes that lead to expulsion of the endosymbiotic algae (Symbiodiniaceae) partner that result in the characteristic signs of bleaching are still poorly resolved at the cellular level. The current state of knowledge of these cellular mechanisms was comprehensively discussed in the first committee report (NASEM, 2019). Active ejection of the symbionts from coral cells is likely, combined with impairment of symbiont photosynthesis (Baird et al., 2009; Downs et al., 2002; Weis, 2008). Definitive identification of the cellular mechanism of how bleaching occurs is critical to assessing the thresholds of bleaching across different species. This information is also central to refining the modeling parameters for heat stress sensitivity of corals and evaluating potential mitigation outcomes provided by intervention strategies. The cellular triggers of coral bleaching can be influenced by historic exposures, and intervention strategies such as pre-exposure rely on low-level heat stress to increase resilience to later high temperature exposures. Defining how the host genotype, algal symbionts, and even the associated microbiome interact to trigger bleaching across both historic and current temperature thresholds is key to minimizing uncertainty associated with most interventions.
2. Identify underlying causes of coral diseases and develop biomarkers of coral health, heat susceptibility, and disease diagnosis as well as ecosystem health.
Some measure of coral health both before and after an intervention is implemented is required to evaluate the need for and impact of the intervention. The underlying molecular, cellular, and physiological processes that facilitate a healthy functioning coral remain poorly resolved due to the complex interplay among the host, its algal symbionts (Symbiodiniaceae), and other microbial partners. Biomarkers (attributes and substances indicative of a biological state/condition) are critical tools in biomedical research and clinical practice for assessing the health status of patients. However, in corals there are currently no universally accepted biomarkers for healthy or stressed individuals, and homeostatic baselines are also generally unavailable. Multiple studies have used a range
of approaches to link cellular physiological functions to stress and, ultimately, the health of individual corals including growth (Edinger et al., 2000), fecundity (Linton and Warner, 2003), productivity (Scheufen et al., 2017), calcification (Anthony et al., 2008; De’ath et al., 2009), protein assays (Downs et al., 2000), lipid and fatty acid constituents (Bachok et al., 2006), gene expression (Louis et al., 2017), and metabolite chemistry (Farag et al., 2018; Sogin et al., 2016), among others. However, the cellular response of coral holobionts to stress is still unclear because it is complex and driven by numerous pathways that interconnect and are likely different among coral species. Furthermore, biomarkers or even simple physiological tests for differences in heat or disease resistance are generally lacking or just being developed. Morikawa and Palumbi (2019) tested known biomarkers, environmental sensing, heat mapping, and physiological profiling as predictors of tolerance to bleaching in nursery corals. Muller et al. (2018) measured heat tolerance and disease tolerance along with their associated tradeoffs among clones of one Caribbean coral species. However, further work and refinement across species are clearly needed.
In addition, teasing apart the biomarkers associated with the coral host and the algal partner can be challenging because understanding of biomarkers of stress for the symbiont lags behind that of the host. Continued efforts to identify and develop easy-to-use biomarkers and physiological assessment of coral health and stress resistance for species targeted in restoration efforts is a priority area that can aid restoration approaches in the future. Rapidly advancing technologies in the areas of “omics,” nanotechnologies, sensor development, and imaging are being applied to corals and will no doubt aid and accelerate filling this knowledge gap.
Disease and increased incidence of disease(s) in a coral population are obvious indicators of declining coral health and potential stress on the reef ecosystem. Effective field-based monitoring and reporting systems are established in many regions to identify and respond to disease outbreaks (Beeden et al., 2012). However, the underlying causative agents, either biotic or abiotic, of many coral diseases have been difficult to identify because the drivers of outbreaks may be linked to multifaceted interacting effects of environmental stress, reduced host fitness, destabilized symbiosis within the coral holobiont, and increased abundance or virulence of microbial pathogens (Bourne et al., 2009; Burge et al., 2014; Mera and Bourne, 2018; Woodley et al., 2016). These difficulties are highlighted by the recent extensive mortality of a number of coral species due to an ongoing disease epidemic in southeastern Florida for which the underlying causes still remain unknown (Precht et al., 2016; Rippe et al., 2019; Walton et al., 2018). Because many restoration approaches are dependent on disease-free corals before intervention (e.g., managed breeding, assisted gene flow) in addition to avoiding losses due to disease
after intervention (e.g., algal symbiont manipulation), or spreading disease agents across geographically dispersed areas (e.g., managed relocation with introduction to new areas), research into coral diseases, their vectors, and treatment remains an ongoing priority.
Elucidating underlying coral disease etiology to allow for rapid identification of the underlying causes and implementing measures to manage, prevent, or mitigate spread is a priority. For interventions such as antibiotic treatment, knowledge of the specific pathogen(s) and their mode of action is critical to provide informed choices for which antibiotics will be effective, where to target application, and when to apply the treatments. Similarly, phage therapy treatments can only progress when the target host bacterial population is identified, and effective application is dependent on knowledge of the infection process. Disease is generally an interplay between the causative agent (biotic or abiotic), the host, and the surrounding environment. Therefore, at the heart of disease causation is the host response to challenge, and in the case of corals, their immune system is surprisingly complex for an organism with a relatively simple tissue organization (Mydlarz, 2019; Mydlarz et al., 2010; Palmer, 2018; Palmer and Traylor-Knowles, 2012; Toledo-Hernández and Ruiz-Diaz, 2014). Recent studies have significantly improved understanding of the coral immune system (Mydlarz, 2019; Palmer and Traylor-Knowles, 2012; van de Water et al., 2016); however, identifying the response of this immune system to disease onset is a significant knowledge gap. In undertaking restoration of reef ecosystems, preventing or mitigating effects of disease will be paramount and aided greatly by improving the knowledge of the underlying factors contributing to disease and the corals’ response at all life stages (planktonic larvae, juveniles, and adults).
Finally, the goal for many intervention programs is likely to be the maintenance of coral reef structure and function as it is associated with the sustainable delivery of associated ecosystem services. Monitoring whether intervention programs achieve this goal will require indicators of ecosystem “health.” These indicators can also inform prioritization of locations for testing and deploying interventions based on the degree of reef degradation as described in Chapter 2 (Smith et al., 2008). A large array of possible indicators ranges in ecological scale (e.g., the coral community, the associated fish and invertebrate community, the social system) and metrics (e.g., cover or biomass, rugosity, species richness, evenness, functional diversity). A research challenge is then determining which indicators best predict larger-scale processes, such as systemwide resistance to and recovery from disturbance to maintain a coral-dominated state (e.g., McClanahan et al., 2012), that connect to the ecosystem health goals identified by stakeholders.
3. Determine functional roles of, and tradeoffs among, members of coral reef communities at multiple ecological scales from coral-associated symbionts and microbiomes up to the composition of coral species in reef communities.
The symbiosis established between the algal dinoflagellates (Symbiodiniaceae) and the coral host underpins the health and resilience of coral reefs, and this intimate association has been the focus of a large current and historical body of research (e.g., Cowen, 1988; Falkowski et al., 1984, 2008; Suggett et al., 2017). The manipulation of the symbiotic associations has garnered much interest due to the potential ability of these symbionts to increase stress tolerance (van Oppen et al., 2017). While shuffling or switching the algal symbionts is currently possible, the effects are temporary; fast-tracking efforts aimed at filling the knowledge gaps would help facilitate successful future restoration activities and reduce risks associated with symbiont manipulations at this pre-intervention stage.
The other microbial communities associated with corals (bacteria, archaea, fungi, and viruses) also play important roles in maintaining coral health, but understanding of this symbiosis lags behind that of the algal–coral association. Whereas there has been extensive research determining the diversity of the coral microbiome, understanding is still limited regarding the function that the microbiome plays in the holobiont and which interactions are essential to health and stress resistance. This includes if there are shared metabolic pathways for the coral, algal, and microbial partners, and the spatial integration (niche microhabitat localization) of these communities within the coral animal as a whole. Manipulation of these other microbial symbionts has been proposed as an additional means of conferring stress tolerance to corals, and the research gaps overlap somewhat with Symbiodiniaceae manipulation questions.
The major research priorities for manipulation of algae and other microbial groups in symbiosis with coral include identifying the suite of taxa that can provide the required conferred traits of tolerance to the host (including tolerance to varying environmental stress plus disease resistance) and how these associations can be managed. For these populations, identifying their required abundance, their specific functions, and the mechanisms that confer benefit to the coral holobiont is an ongoing research priority. The specificity and flexibility of these associations over multiple symbiont types and coral species need to be established with a focus on corals targeted for restoration. In addition, elucidating the direct versus the indirect effects of algal and microbiome symbiont manipulation on the coral host is needed. For example, does adding or shifting a microbial community member(s) benefit the coral host by a specific inferred trait (i.e., scavenging of reactive oxygen species) or is it simply stimulating other processes, such as adding nutrients that stimulate a microbial loop
and hence coral heterotrophic feeding (i.e., no association is established with the coral host)? Additionally, the longevity of association of introduced symbionts within the coral host, and if these associations can be maintained across generations, needs to be determined to guide when and how often interventions to shift the symbionts and/or microbiome to increase resilience (or more temporary stress tolerance) are required. Lastly, improved understanding and assessment of tradeoffs for each individual coral species/symbiont manipulation is essential because this will inform us of the associated risks of algal and microbiome symbiont manipulation.
Finally, the genetic, reproductive, physiological, population, and community interventions require identification of a target coral species or set of species. Informed prioritization of target species to achieve a goal of maintaining coral reef ecosystem function will rely on identifying the functional role of different coral species (e.g., Darling et al., 2012) and the vulnerability of different functional groups and life history types (e.g., Darling et al., 2013; McClanahan et al., 2014; van Woesik et al., 2012). Such prioritization can further rely on an understanding of the tradeoffs between tolerance to different stressors as well as between different life history types and functional roles. This includes the relative roles of the most stress-tolerant types (Côté and Daring, 2010), as compared to a diversity of functional types (Baskett et al., 2014), in overall reef persistence in a high-stress future.
4. Identify population structure, determine evidence for local adaptation, and define relevant management units for population recovery.
Understanding the standing genetic variation in corals is critical to defining the genotypic diversity, distribution, and connectivity between populations within a species range. Such information is central to many interventions that involve the collection, outplanting, and translocation of individuals. Ideally, this information would be gathered through population genetic studies and measures of ecological connectivity across metapopulations, such as the range of larval dispersal and recruitment and the speed of any range shifts that are naturally occurring. Additional information is also required on the extent of local adaptation across the species range, and the performance of different genotypes across a range of environments that emulate future climate projections. This information might be obtained through common-garden experiments that use a broad range of genotypes representing each of the populations or environments compared. Similarly, the population genetic structure of critical symbionts, such as Symbiodiniaceae, may need to be considered both in terms
of their functional role (Parkinson et al., 2015) and as part of translocations (Grupstra et al., 2017) given that they introduce novel variation to local populations and may invoke many of the same concerns considered for coral host populations.
Information on population structure is relevant to the hierarchy and scale at which interventions should be conducted. Risk assessment requires an understanding of the effect an intervention might have on rebuilding within populations, as well as the impact that interventions might have on metapopulation structure as a whole. Risk assessments might also seek to understand how specific interventions impede or enhance adaptability of coral reefs into the future—for example, whether individuals translocated across different populations enhance or reduce persistence. In this case, biologically relevant information on local adaptation will help identify optimal source and target populations. It is also worth estimating the genetic variability, genetic load, and fitness of edge versus central populations because the former may be the target for conservation actions (e.g., whether assisted gene flow from the center of a species distribution might be an effective restoration approach).
Management units for target species are central to conservation planning. In the United States, the definition of management units relies on information on population structure and on evidence of adaptation (Waples, 2006). The latter is often challenging to define and might include proxies such as selective regimes (differences in ecological and environmental variation) and phenotypic or life history diversity. The definition of management units informs the development of appropriate status reviews (Brainard et al., 2011), risk assessments (Boyd et al., 2017), and recovery planning (NOAA, 2015). They also inform the context of decision making by defining the jurisdictional boundaries and identifying the stakeholders and decision makers who will be engaged in the recovery process. Definitions of conservation units have been prioritized in corals broadly (Brainard et al., 2011), but require fine-scale development within key indicator species.
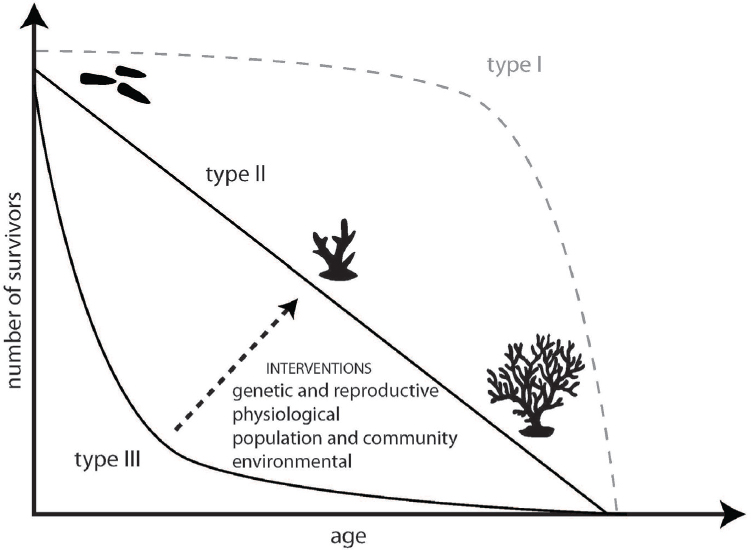
5. Develop methods to improve survivorship for corals that are released, planted, relocated, or settled on reefs at the reef scale.
Increasing the survival of corals that are reared, planted, relocated, or settled on reefs is essential for ecologically and financially effective restoration efforts (dela Cruz and Harrison, 2017; Raymundo and Maypa, 2004). For interventions that rely on sexual reproduction, larval recruitment, and juvenile grow-out, success at ecological scales requires shifting the survival curve from type III (low survivorship at early life stages) to higher age-specific survival probabilities in early and middle life stages
(see Figure 5.1). Population rearing and release will likely be conducted not only for genetic and reproductive interventions, but also physiological and ecological interventions that rely on introduction of treated gametes, larvae, or adults into the water. Rearing should seek to enhance survivorship in culture, while retaining or creating as high amount of genetic diversity as possible and minimizing risks posed by pathogens. Supportive breeding, hybridization, and managed selection rely on rearing methods that reduce the impact of the culture environment as well as on release strategies that improve survivorship in the wild. Certain physiological interventions require appropriate rearing environments for pre-exposure or delivery, which in turn rely on culture environments that are conducive to such methods.
The development of appropriate release strategies for larvae should aim to improve settlement cues and optimize benthic substrates that enhance coral larval recruitment and subsequent survival (e.g., managing algal turf; Arnold et al., 2010). These factors have been studied for only a few coral species; universal settlement cues and optimized substrate conditioning is an important pre-intervention step for many restoration
approaches. Post-release survival can be low (50-100% mortality can occur in the first 5 years; Young et al., 2012) such that an understanding of the relative and interactive effects of different abiotic and biotic drivers of near-term survival of outplanted corals (e.g., degree of wave shelter, prevalence of corallivorous snails and fireworms, presence of herbivores that can mitigate competition with macroalgae) can inform methods for improved release success (e.g., identification of locations for release, corallivore clearing, herbivore reintroduction; Idjadi et al., 2010; Young et al., 2012). For interventions that rely on propagating individuals both within populations (from high-stress environments) or between populations (managed relocation), improvement in methods of transportation, banking of large numbers of individuals, and increased survivorship of the relevant life history stage (such as coral fragments or gametes, including cryopreserved gametes) are required. Resultant technologies need to be adapted for both large-scale aquaculture operations that supply coral for outplanting, as well as performing in situ reef-scale applications. Finally, while cryopreservation approaches are successful for many coral species (Daly et al., 2018), developing approaches for egg storage and egg revival that allows subsequent fertilization and large-scale supply will greatly help the approaches detailed above.
6. Develop extensive, freely available databases on coral communities, hosts, symbionts, and microbiomes to support studies on genotype–phenotype relationships, population structure, and community dynamics.
Genetic markers for target species will play a central role in fundamental coral biology research relevant to interventions and in ongoing monitoring for risk assessments. Such markers are required for defining population structure; for identifying genotypes held in captive breeding facilities and determining strategies for outcrossing; and for determining the survivorship of individuals in technologies that rely on gamete and larval seeding, sexual reproduction, and relocation. Advanced genomic and other “omic” approaches can assist investigations into the extent of local adaptation, although this work needs to be substantially extended from present efforts. Genetic manipulation requires extensive characterization of the genotype–phenotype relationship of corals and symbionts in different environments in order to identify genes that may be manipulated to confer resilience. Interventions that rely on population structure and monitoring recruitment success require fewer markers than those that depend on a greater understanding of the genome and its interaction with the environment. The development of whole genome resources for target species will benefit all efforts.
Given the high value of coral genetics and genomics data for the design of intervention strategies, significant effort needs to be expended in this area, targeting a variety of ecologically important species. The panoply of “omics” methods (e.g., genomics, transcriptomics, proteomics, metabolomics, methylomics) available in modern scientific research holds great promise in uncovering the genetic basis of phenotypes, and with the advent of genome editing methods such as CRISPR/Cas9 (Cleves et al., 2018), the opportunity to validate and/or enhance gene function. These methods have been applied to corals and their dinoflagellate symbionts, and a variety of key genes and pathways related to stress responses have been identified (Guzman et al., 2018; Helmkampf et al., 2019). However, given that typical corals contain in excess of 20,000 genes and responses to stressors such as elevated temperature can lead to the differential expression of hundreds to thousands of genes (Shumaker et al., 2019), identifying individual targets for genetic manipulation remains a major hurdle in this field. This complex biological system would benefit from a rapidly expanding arsenal of bioinformatic methods referred to as multi-omics. These approaches aim to integrate multiple lines of “omics” evidence and other forms of data such as protein–protein interaction networks to explain phenotype (e.g., Huang et al., 2017; Kim and Tagkopoulos, 2018). Shared network hubs derived from different “omics” datasets can provide strong evidence for genes that underlie a particular phenotype. Data or network integration provides, therefore, the framework to move beyond analyzing single data types. These methods require, however, that inferences are grounded in the biological world and have ecological importance rather than being exhaustive catalogues of species or gene expression and metabolite data (Vilanova and Porcar, 2016). Multi-omics is poorly developed in corals, and once such data are produced and made freely available in public databases, it will still require extensive testing in natural conditions to identify the major players (genes, pathways) that underlie coral stress tolerance and resilience. However, once these types of data are available (e.g., as in the mouse model; Li et al., 2018), then the choice of regulatory genes for targeted manipulation using genetics (i.e., through under- or overexpression) will have a firmer foundation. Equally important will be the collection of abiotic data at the level of individual coral genotypes to allow the discovery of loci that may confer specific adaptive traits. The current use of reef or broader site-level measurements will therefore need to be scaled down to the level of colonies with sensors that are both inexpensive and powerful (e.g., Sekli Belaïdi et al., 2019).
Beyond “omic” databases, support for databases of life history, phylogenetic, and biogeographic data (such as the Coral Traits Database; Madin et al., 2016) will be central to informing multiple decisions around interventions. Life history and phylogenetic data can inform identification
of target species based on possible stakeholder priorities such as phylogenetically unique species and species with particularly vulnerable or invulnerable life histories (see Research Need #3). Phylogenetic data can also inform the drivers of species ranges and local adaptation within those ranges, which will be particularly important to informing decisions on translocation-based interventions (see Research Need #4). Finally, all data types can lead to more rapid development and parameterization of models that inform the decision-making process as described in Chapters 3 and 4: biogeographic data can identify the relevant species to consider modeling, life history data can provide parameters for those species, and phylogenetic data can help identify parameters for closely related species when data for a given target species are not available.
7. Identify species-specific threshold responses of corals to changes in temperature, light incidence, and ocean pH, as well as reef-scale threshold responses to disturbance and environmental change.
Interventions are most necessary in areas where corals will experience environmental conditions in which they have a low likelihood of natural adaptation or acclimation. Just as predictions of future environmental conditions are important for assessing the benefit of deploying an intervention, so is understanding the response of corals to these conditions. Increases in average temperature of 1-2°C can lead to coral bleaching (Donner et al., 2005; Frieler et al., 2013; Hoegh-Guldberg, 1999; Sheppard, 2003; van Hooidonk et al., 2013, 2014), although the threshold varies by species. Heat tests based on long-term exposure (multiple days to weeks) to low levels of heating (1-2°C above ambient) are common in the bleaching literature (Ainsworth et al., 2016; Jokiel and Coles, 1990; Lesser, 1997; Oliver and Palumbi, 2011). In addition, more rapid acute heat tests have been used comparatively among species and individuals within species (Barshis et al., 2018; Morikawa and Palumbi, 2019; Parkinson et al., 2018). However, for mapping heat tolerance (and sensitivity) to improve the parameters for modeling and reduce uncertainties, the methods for assessing bleaching thresholds need to be standardized across coral species and conducted for corals inhabiting specific target reefs. Development of a standardized, inexpensive, low-maintenance, and reliable temperature control and experimental system that can deliver a variety of temperature profiles would be a key goal.
Harder to quantify is the potential amount and rate of coral adaptation to increased thermal stress through the interactive effect of multiple mechanisms, which is necessary to understand the impact of various climatologies on coral survival. Therefore, improving measures of species susceptibility and mechanisms of increased thermal tolerance that can
be integrated into the modeling approaches and reconciled with physio-chemical metrics such as degree heating weeks will inform prediction of differential loss of species due to temperature-driven ecosystem stress. Because there are multiple mechanisms that could lead to increased tolerance over ecological timescales, it is also important to investigate how these mechanisms could interact to constrain or facilitate adaptive evolution (e.g., Ghalambor et al., 2015; Kronholm and Collins, 2016). Species-specific susceptibility and adaptability metrics across other environmental stresses (i.e., nutrients, light, pH) would further resolve the predictive value of modeling efforts. Being able to quantify these coral species-specific responses to environmental change would importantly assist in identifying when environmental interventions, which reduce stress experienced by coral, would have an impact on coral survival or increase the benefit of other coral interventions.
In addition to physiological response and adaptive capacity to environmental stress, a mechanistic understanding of population- and community-level response to environmental stress will inform a strategy of protecting overall reef resilience (Anthony et al., 2015; Mcleod et al., 2019). Understanding the mechanisms that support the maintenance of a coral-dominated state during and after disturbance (e.g., relative roles of factors such as diversity, connectivity, and the degree of different local stressors) can (a) inform modeling and selection of local management actions that might increase reef persistence and therefore intervention success and (b) inform where and how much interventions might alter overall resilience. Coral population- and community-level response to disturbance might depend on thresholds in coral diversity and cover necessary for reef-level persistence. On the population level, thresholds in coral population density and genetic diversity might affect successful reproduction (where diversity could affect reproductive success for obligate outcrossing species; Baums, 2008; Miller et al., 2018). On the community level, thresholds in overall coral cover and community composition might affect the maintenance of a coral-dominated reef state with high herbivory on macroalgae (Bellwood et al., 2004; Mumby et al., 2007). The potential for community-level thresholds is particularly controversial and unresolved given the long timescale of observations necessary to discern their relevancy to ecological systems (Mumby et al., 2013; Petraitis and Dudgeon, 2004; Żychaluk et al., 2012). Discerning whether population- and community-level thresholds are relevant, and if so, what the threshold values are, is vital to identifying locations where interventions are more likely to be successful and the scale of interventions that might be necessary for success (see also Research Need #9).
SITE-SPECIFIC RESEARCH AND ASSESSMENT TO DETERMINE WHETHER INTERVENTION IS NEEDED OR POSSIBLE
Development of appropriate ecological models and identification of relevant management objectives and goals require site-specific information. The following research areas include the research and assessments that should be conducted to inform robust decision-making processes including identifying the problem, developing management objectives and evaluation metrics, and modeling risks and benefits. The goal of such research is to evaluate which interventions might be appropriate in a particular setting (if any), how they would be implemented, and what objectives they could be expected to achieve.
8. Identify local stressors that influence population recovery and determine whether stressors are likely to influence the success of the intervention.
As the committee illustrates in its model in Chapter 4, it is important to integrate the impact of local stressors (and management of these stressors) into a decision framework to evaluate the use of the more novel coral interventions as part of a comprehensive management strategy. Reef degradation is often the result of multiple stressors on the ecosystem acting synergistically (Gardner et al., 2003; Hughes and Connell, 1999; Hughes et al., 2017b). The effects of local impacts such as poor water quality, coastal development, and overfishing are well documented (Fabricius, 2005; Jackson et al., 2001; Mora, 2008), and unless these stressors are managed effectively the benefits of any proposed intervention may be minimal. For example, recent work identified that the resilience of coral populations was impacted by poor water quality because the recovery following a disturbance is much slower and populations are more susceptible to other localized impacts such as higher predation and disease (MacNeil et al., 2019). Similarly, the appearance of the stony coral tissue loss disease in Florida and parts of the central and northeastern Caribbean (NOAA, 2019) highlights important considerations when assessing which interventions may or may not be an option. In this case, managed relocation may not be a viable option (without additional strict quarantine measures). Hence, knowledge of the historical, current, and predicted future local stressors is required to assess if any proposed intervention can be successful and meet the goals of the project.
9. Develop appropriate metrics and recovery goals that assess the effects of the intervention on ongoing tolerance, health, fitness, and recruitment within the target management unit as well as on connected reefs.
Risk assessments and monitoring efforts depend on accurate measures of community composition and population demographic data, such as survivorship and recruitment, within the specific environmental context. Therefore, accurate baselines and appropriate metrics need to be established before the interventions are implemented. Data collection specifically targeted at characterizing community composition and interactions is essential, because not all corals will respond to interventions in the same way. Defining demographic patterns for target species within the local context will also inform programmatic decisions on whether interventions are effective in meeting recovery goals, especially in the context of a changing environment. Therefore, it is important to determine which community measures and species-specific traits are relevant to population recovery parameters—for example, reef structural complexity as it relates to multiple reef functions (Graham and Nash, 2013), algal cover, and multiple coral attributes, such as diversity, cover, growth rate, reproductive rate, disease susceptibility, and thermal tolerance.
As noted in Research Need #7, the success of interventions that involve outplanting, such as managed breeding and managed relocation, might depend on population- and community-level thresholds in values such as genetic diversity (Baums, 2008; Miller et al., 2018) and coral cover (Mumby et al., 2007) necessary for reproductive success and reef-level persistence. Quantifying such thresholds can identify the appropriate targets for restoration of ecosystem processes (Mumby et al., 2007; Suding and Hobbs, 2009; Suding et al., 2004). Therefore, the development of metrics should take threshold effects into account where they are expected to be relevant.
Finally, it is also important to understand how an intervention on one reef may impact the demographic properties of adjacent reefs. Setting target population sizes for species of interest is part of this effort; as population size increases, so may recruitment on adjacent reefs. Therefore, site-specific research should be conducted to determine connectivity between a target reef and adjacent reefs. Understanding the degree of connectivity from other reefs to a target reef can also inform evaluation of whether or not relocation (assisted migration and assisted gene flow) can improve on naturally occurring dispersal and range shifts.
10. Evaluate whether population recovery at a specific site can be achieved through managed relocation or managed breeding, and if so, which intervention is most appropriate for recovery.
In cases when the objective is to supplement reef populations with more individuals, the preferred intervention strategy will depend on a balance between the risk and the likelihood for recovery. In all cases, assessing the impact of broodstock collection (whether through fragmentation or collection of gametes and larvae) on wild or captive source populations is necessary. This information might be gathered through site-specific mapping of coral distribution, measurement of genotype diversity across the reef, and assessment of population size. Similarly, the risk of releases or managed relocations from such programs on target populations should be evaluated by measuring the degree of genetic divergence between the source and target populations and estimating their effective population sizes. The risk to local populations may increase with greater genetic divergence of introduced individuals (whether through inadvertent changes due to domestication or to deliberate manipulation), especially following reproduction between the source and target populations. However, this risk should be balanced against decreased genetic divergence and adaptability in the target population, especially given a rapidly changing environment.
11. Identify host, symbiont, and microbial populations at restoration sites to ensure that treatments or manipulations aimed at improving coral physiological performance can achieve recovery goals.
Physiological response of coral populations to environmental conditions can be attributed to the host, the symbiont, and/or the microbiome. Therefore, identification of available symbionts (including the microbiome) and their potential beneficial traits is needed at each site. This includes establishing the diversity, specificity, and flexibility of these associations over multiple symbiont types, microbiome populations, and coral species. Having a good understanding of the host–symbiont dynamics through a multispecies genetic modeling framework will help to disentangle the role of diversity in the efficacy of interventions and improve the predictions of the responsiveness of the coral to interventions. For example, pre-exposure treatment aimed at conferring some degree of additional tolerance to subsequent re-exposure requires understanding of the response of populations and the potential mechanisms that contribute to increased tolerance across the host, symbiont, and microbiome. Similarly, for other physiological treatments including antibiotics, antioxidants, nutritional supplementation, and phage therapy, assessment of effects on the host and changes in community structure across the symbiont and
microbiome populations before and after treatment is required and needs to be linked to individual and reef-wide coral health metrics.
Relatively little is known about the diversity, community structure, and abundance of free-living Symbiodiniaceae in reef waters and sediments, or the degree to which corals acquire symbionts from these sources or exchange them with other noncoral hosts. Changing environmental conditions may shift the relative abundance of different symbionts, and local declines or extinctions of coral hosts will likely also affect the availability of appropriate symbionts. This in turn may affect recovery and repopulation dynamics and potentially limit the capacity of reef corals to respond to changing conditions. Consequently, a greater understanding of the population dynamics and genetic diversity of algal symbionts among different hosts and free-living life stages is required.
12. Assess in a site-specific manner the benefits, risks, and chances of success for implementing environmental interventions.
Each reef site or region needs to be assessed for its physical suitability for the proposed intervention. All environmental interventions are dependent on a set of bathymetric, geomorphologic, oceanographic, and/or atmospheric conditions to be met for the intervention to have a chance of success. Without meeting these defined geophysical parameters, the risk of failure and unintended consequences for the intervention increases.
The environmental interventions can operate over different scales. Most proposed interventions are at the reef scale (increased turbidity, shading layers, microbubble ocean whitening, mixing of cool waters, abiotic and biotic ocean acidification interventions) potentially focused on high-value reefs, though atmospheric shading (marine cloud/sky brightening) can operate over regional scales. Infrastructure costs associated with implementation are a major barrier for many of these intervention strategies (i.e., shading using physical structures, mixing of cool water, or marine sky brightening). For some reef-scale interventions (increased turbidity, polymer shading layers, microbubble ocean whitening, mixing of cool waters, chemical additions), modeling that provides detailed water residence and flow dynamics across the reef is required. Similarly, for the use of seagrass meadows and macroalgal beds to reduce ocean acidification, the foremost limitation is identification of geographic setting, reef type, and oceanography that allows it to be effective. Hence, site-specific and robust, linked biogeochemical, biophysical, and hydrodynamic models are required on a site-by-site basis to assess suitability and the probability of success of the intended intervention (e.g., cloud brightening by Alterskjær et al., 2012; biotic ocean acidification interventions by Anthony et al., 2013).
13. Identify the most appropriate site-specific, synergistic management and intervention strategies that together provide greater chance of success and reduced risks than the sum of the impacts of each intervention alone.
The benefits and risks associated with interventions to increase the resilience of corals and reef ecosystems have been addressed individually (NASEM, 2019). However, it is clear that many interventions are synergistic; the benefits of implementing two or more approaches increase the chances of success of each and cumulatively provide even greater potential reef resilience benefits. Note, however, that some combinations of interventions may have antagonistic outcomes, and these risks need to also be assessed. The modeling framework in Chapter 4 details an example of a dynamic coral reef model to illustrate how one might use the output in a decision-making process. For this example, traditional management of herbivores was assessed with assisted gene flow and atmospheric shading. However, empirical data that identify the synergies between both traditional management and the array of novel intervention strategies are a necessity and to date have not been collected at any scale. Some reef systems might need specific combinations of interventions to achieve success (i.e., active algal removal prior to larval capture and release). Nevertheless, research that specifically assesses these synergistic benefits (as well as potential antagonistic interactions) between traditional management approaches in conjunction with novel intervention strategies is vital to reduce uncertainty associated with the outputs of the modeling approaches.
RESEARCH TO IMPROVE ASSESSMENT OF THE BENEFITS, EFFICACY, AND RISKS OF SPECIFIC INTERVENTIONS
Research is needed to stage interventions from laboratory experiments to full-scale management strategies so that the safety, efficacy, and cost-efficiency of interventions can be improved. Implementation of a research program around risky approaches may benefit from a phased testing program, where decision points occur that require stakeholder-based criteria to progress in a stepwise manner from the laboratory to the field (such as is recommended for gene drive research in NASEM, 2016, based on WHO, 2014). Other approaches may have more well-understood risks and benefits but require a development program to design effective implementation. For example, while interventions such as probiotics (microbiome manipulation), antibiotics, and nutritional supplementation are possible, they lack specificity because of limited understanding.
14. Develop protocols for control of pathogens (biosecurity and quarantine).
Biosecurity and quarantine protocols are important for identifying, isolating, and removing pathogenic microbes. Such protocols have been developed for many jurisdictions and countries (Hathaway and Fisher, 2010; Hewitt and Campbell, 2007) and are a priority for industries such as the marine aquarium trade (Morrisey et al., 2011) and high-value aquaculture farms (Pali et al., 2015). However, similar robust biosecurity and quarantine protocols need to be adapted to meet the requirements of the expanding reef restoration activities, incorporating the additional challenges that arise from the emerging intervention strategies highlighted across this report. Many coral restoration approaches rely on assessing the pathogen loads and virulence potential within coral populations and preventing their further spread. Large-scale aquaculture operations can promote increased disease occurrence due to high population densities and suboptimal environmental conditions. Managed breeding and managed relocation interventions also have the potential to spread disease. Through improved understanding of the underlying etiology of coral diseases, development of rapid diagnostic assays for the identification of pathogens and quantification of their abundance and/or virulence is possible and should be a long-term goal of disease studies to reduce risks associated with intervention approaches (Pollock et al., 2011).
Corals are also host to a wide range of other cryptic organisms that interact across the mutualism–parasitism continuum (Bronstein, 1994). These organisms, which may include but are not limited to acoels, digeneans, polyclads, gastropods, decapods, copepods, and pyrgomatids, can be hosted by corals in low abundance in natural environments but become problematic under captive conditions or when moved to environmental ranges that do not also host natural biocontrol organisms. Hence, a more comprehensive understanding of the cryptic species associated with corals, their symbiotic interactions, and their potential for causing detrimental effects on individual corals or the ecosystem more widely is warranted.
15. Develop effective approaches to modify symbiotic algal and/or microbiome populations.
Though conferred beneficial stress-tolerance traits have been detected for some algal symbiont populations associated with coral (Suggett et al., 2017) and potentially for the microbiome (Rosado et al., 2018), there are currently no effective approaches to modify these algal and/or microbiome populations or shift the abundance of members of these populations at a scale that would help confer benefits to multiple coral species. Having approaches that are effective at shifting populations, are targeted
to specific populations (host, symbiont, and microbiomes), and do not result in detrimental effects on the target corals (known as dysbiosis), is critical to reduce associated risks for these interventions. The longevity of the symbiont populations and their associated conferred traits (including across generations) with the coral host needs to be established to inform whether manipulation and therefore delivery of the modified symbiotic communities is a periodic intervention during times of stress. If manipulations are undertaken during coral settlement and juvenile grow-out in aquaculture facilities, the long-term maintenance of a modified symbiont community and the ongoing conferred traits needs to be established in situ.
16. Develop effective approaches to determine whether corals that are released, planted, relocated, or settled on reefs contribute to recovery goals while reducing risk to ongoing adaptation and ecological processes.
Risk assessment based on evolutionary principles requires the development of models to assess the impact of gene flow from introduced corals on the genetic diversity of established populations, which in turn influences population demographic processes (Baskett and Waples, 2013; Ford, 2002; Lynch and O’Hely, 2001; Tufto, 2017). Benefits and risks associated with gene flow relate to changes in effective population size, rates of inbreeding, and fitness. Risks associated with the first and second factors might occur through large releases of individuals with little genetic diversity, or by limited recruitment. The third may result following introduction of maladapted individuals and interbreeding between such individuals and native corals. Each of the interventions within this category will benefit from research that evaluates the genetic gains versus risks associated with each activity, as well as the associated changes in population growth and recruitment.
Supportive breeding propagates a portion of a population using local broodstock and releases these individuals back into the natural environment. Genetic gains may be accrued by increasing the effective population size, increasing genetic diversity through reproduction, and improving the population’s ability to adapt. However, if insufficient diversity is captured by the breeding program, then genetic change might occur through a decrease in effective population size and inbreeding or through domestication selection. This change may in turn compromise the health of the wild populations. Therefore, research should evaluate the genetic diversity within the captive and wild populations and monitor long-term fitness changes in the supplemented population. Managed selection, outcrossing between populations, and hybridization between species are
intended to increase genetic variation and fitness of released individuals, especially in changing environments. However, research in this area is limited to a few case studies, and often to laboratory settings. Scaling these interventions to the natural environment depends on concerted efforts to evaluate offspring performance in the target environments. Risk assessments need to evaluate whether released individuals pose an ecological or genetic risk to existing populations, and whether they contribute to population growth. Relocated corals, or corals with novel genotypes, may interbreed with individuals in target populations or with themselves. Fitness benefits seen in one generation may not be transmitted to the next, which in turn may affect population growth and survival. This information is thus relevant to risk assessments that evaluate recovery goals across generations, especially when nonnative or domesticated genotypes are introduced.
Finally, it is necessary to investigate how the number of introduced individuals affects intra- and interspecies interactions and ecosystem functioning. Risk assessments that combine genetic models with demographic models may include density dependence, competitive interactions, and changes in community composition through interactions with other species such as symbionts, fish, and macroalgae.
17. Develop and test genome-editing methods in a wide variety of ecologically important coral species.
As with all multicellular organisms that house a complex microbiome, targets for genetic manipulation in corals are not only difficult to identify, but the outcomes of genetic changes are challenging to predict. Nonetheless, the first trials of CRISPR/Cas9 methods have been completed in coral animals (Cleves et al., 2018), resulting in mosaic creation of tissues containing gene deletions or knockouts. There is as of yet no evidence provided of an altered phenotype or the inheritance of edited genes in adult corals. These technical issues may be addressed in the near future, but developing or identifying faster growing (e.g., shorter generation time) corals would also be a major advance in this field. However, even if the incorporation and complete or near complete inheritance of edited genes can be proven in adult coral animals using CRISPR or other methods (e.g., transcription activator-like effector nucleases, TALENs), it is unclear how effective these methods will be in field trials. Nonetheless, given the high promise of genome-editing methods to enhance coral resilience through stress tolerance, there is a clear need to develop and test these methods in a wide variety of ecologically important coral species for deployment in the future.
In contrast, no genome editing or genetic transformation methods have yet been published for the algal symbionts of corals. Although the microbial component of the coral holobiont (the microbiome) is known to play an important role in reef health, genetic manipulation of these microbes has also not yet been attempted. A major research need in this area is identifying the major genes and pathways involved in coral holobiont resilience under fluctuating environmental conditions. Identifying the phenotypes most useful for genetic selection that are relevant to real-world conditions is also a priority. In addition, development of more robust genetic modification techniques for the coral animal to efficiently spread adaptive genes into natural populations is required alongside accelerating the life history of corals to allow more generations to be produced and studied prior to outplanting. Finally, developing genetic tools for the algal symbionts of corals should be facilitated.
18. Develop methods of delivery for nutrients, probiotics, antibiotics, phage therapy, and antioxidants at reef scales.
Some proposed interventions that promote coral resilience can be combined, provided that appropriate reef-scale delivery approaches are developed. For example, nutritional supplementation of corals can be linked to provision of probiotic coral microbes, antioxidants, antibiotics, and phage therapy. Developing coral-specific diets that promote increased fitness through enhanced heterotrophic feeding can also contain antioxidants and symbionts (algal and microbial probiotics) that confer benefits to the coral host. Scale of production for such supplements is easily achieved, but what is required is improved knowledge of the nutritional requirements of coral (Conlan et al., 2017) and conferred benefits for each coral species that increase resilience. Constraints on heterotrophic feeding exist for different coral species, with some corals possessing large polyps that are well suited to capturing prey to supplement their diets while other coral species have small polyps and rely heavily on photosynthesis-derived nutrients rather than heterotrophic feeding. Hence, for coral species targeted for restoration, feeding dynamics and nutrient acquisition and energy budgets need to be refined.
Formulation of diets, inclusive of probiotics, is well developed in the animal and human therapeutics industries and possibly transferable to corals. For example, encapsulation of probiotics and antioxidants into digestible formulations is a global multi-billion-dollar industry (Sapkale et al., 2012; Shinde et al., 2014). Adapting these approaches to develop formulations that can deliver nutritional supplements, probiotics, antibiotics, and antioxidants to corals is a research priority provided that the benefits are demonstrable. Delivery of such formulations is easily achieved in
experimental systems or large-scale coral aquaculture facilities. However, delivery onto reefs and at the ecosystem scale is much more challenging. While it is conceivable to deliver encapsulated formulations onto a reef, ensuring these bundles target corals specifically and are ingested or taken up is far more challenging and associated with risks such as enhancing nutrients entering the reef and stimulating growth of undesirable reef communities such as macroalgae.
19. Assess feasibility, potential benefits, costs, limitations, and risks associated with environmental interventions.
Currently, the effectiveness and potential benefits of the proposed environmental interventions can only be derived theoretically because there are few operational examples that conclusively demonstrate an increase in coral resilience over any scale. Though a small field trial is being conducted on the Great Barrier Reef for bringing deep water to the surface to cool reefs (Reef Havens Project1), there is still insufficient information from this project and for all current proposed environmental interventions. Hence, further small-scale experimental and field trials are a priority in these areas to assess feasibility, potential benefits, costs, limitations, and risks.
Many of the environmental interventions require significant technology development that can be adapted for application in reef ecosystems. For interventions such as atmospheric shading and cool water mixing, the challenges lie in the design, engineering, and deployment of equipment and infrastructure to ensure that they are feasible and effective at their desired scale of impact. Cool water mixing can be targeted at the reef scale, whereas atmospheric shading is expected to have impacts at a regional scale. Shading that uses thin-layer barriers needs demonstration of the effectiveness of these formulations to ensure that they are biodegradable and safe, yet also reduce light enough to benefit corals. For all environmental interventions, the general requirements of research, development, and testing is still a priority to determine their effectiveness, control, and technical aspects of scaling up.
RESEARCH TO INFORM RISK ASSESSMENTS AND MODELING
The adaptive management cycle requires monitoring and evaluating the results of a management action based on an established monitoring program in order to iteratively gain information to support decision
___________________
making. Thus, continuous improvements to a structured decision-making process require efforts in the areas of monitoring and feedback into model improvements. As described in Chapter 3, there is also a feedback in the reverse direction, where structured decision frameworks can help prioritize areas of research.
20. Use targeted monitoring to evaluate performance, improve benefits, and minimize or manage risks.
Though there are a large number of unknowns related to coral interventions, this does not mean action must be delayed. Action can take place based on the best available science and then monitoring can focus on these key unknowns to resolve them and learn in an adaptive management process (Holling, 1973; Walters, 1986; Walters and Holling, 1990). Any intervention that is conducted for coral reefs will rely on monitoring coral populations and broader reef ecosystems to assess effectiveness of the approach and to identify any unintended consequences. For effective comparisons, baseline information on reef habitats needs to be established at the sites of interventions, and best practices for long-term monitoring regimes need to be implemented. Across the spectrum of all interventions, current established metrics of reef health are important to assess their impact on reef species and the ecosystem more broadly. These metrics relate to management objectives as reflected by the decision criteria, and can include such attributes as coral cover, species diversity, genetic diversity, productivity, disease prevalence, bleaching thresholds, fecundity and recruitment data, and coral growth rates. Additional decision criteria may include social or economic factors.
Often, measures of ecosystem impact and effectiveness of the intervention strategies may be long term. Therefore, establishing intermediate indicators or proxy indicators for whole ecosystem response and impacts is important. Development of robust biomarkers of coral health has been identified previously as a research priority (Research Need #2), and furthermore, technology development that allows incorporation of these biomarkers into small, inexpensive, and portable tools to assess stress tolerance would be highly valuable. Indicator taxa can also be a valuable tool, for example, tracking abundance and cover of macroalgae. Monitoring microbial communities and the tipping points that shift the community can be valuable for earlier detection of ecosystem changes. For example, studies show that anthropogenic stress on reefs can shift microbial communities and their metabolic pathways from autotrophic communities to more inefficient heterotrophic communities that change the nutrient dynamics and flows in the ecosystem (Dinsdale and Rohwer, 2011; Haas et al., 2013; McDole et al., 2012). Metrics such as microbialization scores
have been proposed as one approach to assess changes in microbial function within the reef that directly affect reef health and resilience (Haas et al., 2016; McDole et al., 2012).
Across all intervention strategies, improved measurement of reef demographic processes will be critical to assess benefits, identify off-ramps where the intervention may have unintentional consequences, and measure the overall success or failure of the approach. Traditional metrics of changes in coral cover, species diversity, genetic diversity, invasive species, and disease abundance will continue to be effective and central to assessing these demographic processes. However, new ways to collect these metrics are being developed and should be embraced; supplementing expensive and laborious diving operations with advanced imaging technologies from satellites, remotely operated vehicles, and underwater drones combined with high-powered computing, machine learning, and autonomous technologies is a priority.
21. Use iterative model design to reduce uncertainties and improve model predictions to increase confidence in the decision support framework.
As Chapters 3 and 4 describe and illustrate, model construction, parameterization, and validation for a specific location and set of interventions are intensive but highly valuable endeavors. The previous research items described in this chapter will inform development of the management objectives and provide the data for evaluating management decisions, but further efforts toward model development are required that involve feedback between research/monitoring and model improvement. Information collected in monitoring programs can reduce uncertainties in biophysical system models by improving parameterization, and by illuminating relationships between parts of the system and between the system and management actions (model structure). The modeling approaches also highlight the uncertainty and the transferability of many interventions from the laboratory and small-scale experimental systems into the field. Therefore, many of the proposed interventions require extensive small- to medium-scale trials to assess the benefit, risks, and scalability, thus informing the knowledge gaps and reducing the uncertainties. This process can require an extensive period of testing and trialing as some approaches need to be assessed over generational times for coral populations. These periods can be as short as 2 years and as long as 10 years for many target coral species. Additionally, the effect of synergistic and antagonistic interactions between interventions, or between interventions and conventional management, on the outcomes of a management strategy will be harder to parameterize in a model until field trials have been
undertaken. Hence, to fast-track decision processes around restoration, obtaining relevant and informative direct and proxy metrics that can parameterize these decision models is a priority.
Conducting a sensitivity analysis would identify which parameters to prioritize to better constrain the models. Comparing realized outcomes to model predictions informs the adaptive management cycle (see Figure 3.2). This is essential to assess whether the expectations that went into the model resulted in the predicted outcomes. Value of information techniques provide monetized estimates of the value of collecting additional data to reduce uncertainties across model inputs. These techniques can be used to assist in refining monitoring programs and to identify key biophysical outcomes for which uncertainty is large and having more information would potentially alter the decision.
There is also a critical need to improve or develop higher-resolution oceanographic models for target areas of interventions. The model presented in this report is based on coarse-resolution sea surface temperatures (SSTs; 100 km2, monthly) from a global climate model designed to capture large-scale characteristics of the global climate system (Stock et al., 2011), whereas SSTs most relevant to coral reefs vary at spatial scales of less than 1 kilometer and temporal scales of hours to weeks. Global climate model output should not be applied to make predictions about a specific reef cell. To more accurately represent future coral reef dynamics, global climate models can be statistically or dynamically downscaled or bias-corrected for particular regions (Stock et al., 2011), but additional research is necessary to make linkages between globally modeled SSTs, observed satellite SST measurements, and in situ temperature data. In addition, the thermal environment below the surface of the ocean within the depth ranges of coral reefs is often decoupled from that of the surface, limiting prediction of heat stress with depth (Neal et al., 2014; Smith et al., 2016).
RESEARCH CAPACITY
The committee identified research priorities across four broad themes (see Box 5.1). Many are currently established research projects that need to be supported to fast-track the goal of developing realistic interventions for reefs faced with multiple pressures. Others require significant research and development within the biology and engineering fields to demonstrate or develop their efficacy for application to reef ecosystems. These research priorities hold significant potential to advance the safe, cost-effective, and scalable approaches to build resilience in coral populations and reef ecosystems more broadly. No doubt the urgent requirement for solutions to the dire current trends in reef ecosystem health, combined
with the scale at which intervention has to occur, is the greatest challenge to overcome for reef restoration approaches to succeed. To achieve scale, novel engineering solutions need to be developed alongside the urgent need to fill the critical research knowledge gaps for each proposed intervention and restoration strategy. Some approaches are achievable at the experimental scale (algal symbiont manipulation, shading) or with individual colonies in field settings (antibiotic treatment), but developing the technologies to allow delivery at the reef and ecosystem scales will require considerable investment in human and infrastructure capital.
Improved coordination among federal and nonfederal entities and across jurisdictional boundaries is important for addressing the long-term research needs. The recent establishment of the Coral Bleaching Research Coordination Network (RCN) is one such attempt to accelerate efforts to understand coral bleaching by bringing research across interdisciplinary backgrounds together and standardize sampling and experimental protocols. This effort promotes the sharing and use of the highest-value data by the community for management or other decisions. While the Coral Bleaching RCN is primarily focused on sampling, experimental, and data assimilation protocols, extending efforts to globally coordinate bleaching observations and establish a consistent monitoring framework for thermal stress tolerance and bleaching susceptibility across coral species would be highly valuable. Development of stable, long-lived databases and high-performance computing resources that house such coral reef data would support analyses focused on resilience and restoration. The Coral Trait Database is a good example of a valuable resource that consolidates information pertaining to coral life history, phylogenetic, and biogeographic data; it is freely available to scientists and reef managers throughout the world (Madin et al., 2016). Expanding to coordinate bleaching observations, monitoring frameworks, and thermal stress thresholds, guided by an international program, would ensure that these goals are reached. Such an effort would benefit from the leadership of core partner federal and international agencies, which together could serve as the primary entity for coordination of research nationally and internationally and for information dissemination.
Research and development activities can be streamlined through ensuring open access of data and information and facilitated through creation of coordinated and integrated databases. Such integration of data has been captured in existing collaborative projects such as the eReefs platform2 that incorporates a vast array of water quality, remote sensing, and hydrodynamic modeling to allow predictive forecasting of environmental parameters across the Great Barrier Reef. Building a similar
___________________
2 See https://ereefs.org.au.
resource for the Caribbean region would be highly valuable. Databases of management programs and relevant scientific studies (e.g., Conservation Evidence3) can facilitate knowledge transfer, development of best practices, and meta-analyses that quantify larger-scale outcomes and resolve context dependencies. Similarly, databases that report bleaching events, such as ReefBase, can significantly enhance understanding of ecosystem responses to changing environments and inform management actions. Development of standardized protocols and open data dissemination allows for rapid assessment of the strengths or weakness of datasets.
Training of a workforce that is highly skilled across the range of disciplines from field ecology, to engineering, to laboratory research and bioinformatics will facilitate the generation and dissemination of knowledge to a wide international audience. This workforce will need to include local, culturally connected scientists, managers, and practitioners. Workforce and infrastructure capacity are important to develop within coral reef jurisdictions around the world. These far-reaching communities would need to be connected through well-developed outreach and communications that share scientific information and serve as a bridge to management and policy decisions.
___________________
This page intentionally left blank.






























