2
Approaches to Developing Exposure Values
Development of an occupational exposure level (OEL) involves reviewing the toxicological and epidemiological information available on a chemical, determining the critical health end points of concern, performing dose-response analyses, and considering uncertainties to calculate a health protective exposure value. The approach proposed by the U.S. Department of Defense (DOD) for developing an OEL for trichloroethylene (TCE) is reviewed conceptually in this chapter by comparing it with approaches used by other agencies and organizations for developing exposure guidelines to protect workers or the general public. Individual elements of DOD’s approach are reviewed in more detail in Chapters 3, 4, and 5.
DEVELOPMENT OF OCCUPATIONAL EXPOSURE GUIDELINES
Many regulatory agencies and non-regulatory organizations worldwide develop OELs (Deveau et al. 2015). In the United States, the Occupational Safety and Health Administration (OSHA) establishes regulatory permissible exposure limits (PELs), while the American Conference of Governmental Industrial Hygienists (ACGIH), the National Institute for Occupational Safety and Health (NIOSH), the National Academies of Sciences, Engineering, and Medicine, and other organizations develop voluntary guidelines. Entities reviewing similar literature databases can develop different OELs for the same chemical. A specific OEL is determined by the goals and intended uses of the OELs by the agency or organization, as well as by the process used to determine the values. For example, an OSHA PEL may not be fully health protective because OSHA regulations require that a PEL be technically achievable and cost feasible, whereas ACGIH’s threshold limit value–time weighted average (TLV®-TWA) is solely health-based, and does not consider technical or economic feasibility.
DOD describes its OEL for TCE as being “similar in scope to the OELs published by the ACGIH” (Sussan et al. 2019, p. 4), but used a more comprehensive process for developing an OEL (described later in this chapter and in Chapters 4 and 5). The TLV®-TWA is a concentration for a conventional 8-hour workday and 40-hour workweek, to which nearly all workers may be repeatedly exposed, day after day, for a working lifetime without adverse effect (ACGIH 2019). Workers are assumed to be exposed in a traditional workplace setting
where a chemical is intentionally used for a specific task or in a certain process. DOD’s TCE OEL is intended to also protect workers repeatedly exposed 8 hours per day, 5 days per week, including workers who might be exposed incidentally because of vapor intrusion.
ACGIH appoints volunteer committee members to develop TLV®s in accordance with procedures described in its operations manual (ACGIH 2015) and on its website (www.acgih.org). The ACGIH documentation is not intended to be a comprehensive review of the literature on a chemical, but a short (approximately 10 pages) and integrated analysis of the most pertinent studies for determining a TLV®. General procedures for considering existing literature reviews, searching the literature, evaluating studies, determining the critical effects, determining notations (e.g., potential dermal absorption, respiratory or dermal sensitizer, carcinogen), selecting key studies, determining a point of departure, and handling uncertainties are provided in the manual to help ensure that TLV®s are developed in a consistent manner.
The National Academies has also assisted federal agencies with developing OELs for agency-specific occupational scenarios. The National Academies supported the U.S. Navy between 1984 and 2009 by developing emergency and continuous exposure guidelines for airborne contaminants found aboard submarines (e.g., NRC 1984, 1986, 2009a), and the National Aeronautics and Space Administration between 1992 and 2016 with developing exposure guidelines for air and water contaminants found aboard spacecraft (e.g., NRC 1992, 1994, 2000, 2008; NASEM 2016a). For both agencies, guidance documents established procedures for performing narrative reviews and for determining and documenting how the exposure guidelines were developed (NRC 1986, 1992, 2000; NASEM 2016a), which included factors to consider because of the specialized environmental and physiological conditions and uncertainties of working and living continuously in a submarine or spacecraft. The National Academies provided guidance on the types of evidence to consider, selection of studies, determinations about points of departure, approaches to extrapolate data, exposure adjustments, and consideration of uncertainties. These methods helped to ensure consistency in how the values were determined and that the exposure values met each agency’s needs. The National Academies also evaluated whether OSHA standards for lead exposure were health protective for DOD firing range personnel (NRC 2013).
DEVELOPMENT OF PUBLIC HEALTH EXPOSURE GUIDELINES
The National Academies and others have developed exposure guidelines for protecting the general public. For example, standing operating procedures for developing acute exposure guideline levels (AEGLs) were established in 2001 (NRC 2001a), and were used to produce 20 volumes on hazardous chemicals to which the public could be exposed in emergencies (e.g., NRC 2001b; NASEM 2016b). The U.S. Environmental Protection Agency (EPA) has also developed standard operating procedures for the development of Provisional Advisory Levels for hazardous
agents (Young et al. 2009). For non-emergency situations, reviews have been performed for EPA’s Integrated Risk Information System (IRIS) program, which conducts toxicological assessments that include hazard and dose-response characterizations to support the ultimate derivation of toxicity values that are used to protect the general public (e.g., NRC 2006, 2010, 2011, 2014). These reviews for EPA emphasized the need for ensuring consistent, comprehensive, balanced, and reproducible results, and has led to the creation of a more structured process to provide better documentation and transparency about how the assessments are performed (NASEM 2018). Concepts and procedures developed by the IRIS program could be used by DOD as a guide for the performance of either narrative or systematic reviews as part of the OEL development process. Figure 2-1 illustrates the process, which has been expanded to include steps for performing dose-response assessment and derivation of an OEL.
To determine the potential hazards from a chemical, an evaluation of the scientific literature is undertaken in which relevant studies are identified from the body of literature on a chemical. Studies are subsequently assessed and integration of different lines of evidence is used to make a determination about the potential hazards. It has been a long standing practice to conduct the literature review using a narrative approach with qualitative assessment of studies. Ideally, an analysis plan is developed to guide the narrative review. More recently, EPA and other agencies have begun to use systematic reviews in response to recommendations to provide greater transparency and rigor in toxicological assessments (NRC 2011,
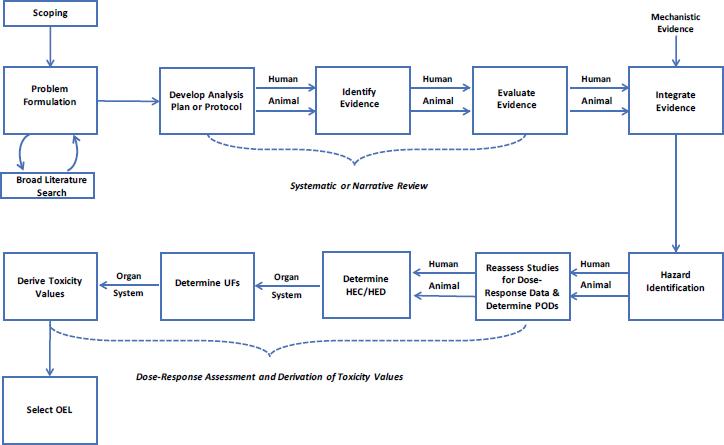
2014). Systematic review is “a scientific investigation that focuses on a specific question and uses explicit, pre-specified scientific methods to identify, select, assess, and summarize the findings of similar but separate studies” (IOM 2011, p. 1). The National Toxicology Program’s Office of Health Assessment and Translation method (NTP 2019) and the Navigation Guide (Woodruff and Sutton 2014) are two established methods for incorporating systematic review methodology into answering environmental health questions for the purposes of hazard identification. A critical element of systematic review is that the methods and evaluation criteria for performing the review are specified in a protocol before the assessment is undertaken. Preferably the protocol is peer-reviewed and published or made publicly available prior to the start of the systematic review.
After hazards are identified, dose-response relationships for the critical end points are characterized to help determine the basis (point of departure) for an exposure value. Studies with appropriate information on dose-response relationships are identified, and consideration is given to the need for exposure adjustments, the need for extrapolation, uncertainties in the evidence base, and other factors to determine a protective exposure value (EPA 1994, 2002; NRC 2009b). Cancer concerns have traditionally been evaluated separately from but concurrently with noncancer effects (e.g., EPA 2005).
DOD’S CONCEPTUAL APPROACH TO DERIVING AN OEL
Assessment of a data-rich chemical such as TCE is a challenge given the large number of studies that are available. To facilitate this endeavor, DOD created its own process for deriving an OEL for TCE (see Figure 2-2). The starting point for the analysis was an IRIS toxicological assessment of TCE developed by EPA (2011) to support determining reference values for the chemical. DOD’s assessment framework appears to incorporate selected elements of systematic review, narrative review, and evidence integration techniques to update EPA’s literature search. Figure 2-2 illustrates that a quality scoring technique was created and used to evaluate controlled in vivo studies, and the scores were considered in conjunction with other factors, such as evidence of a dose-response relationship, to determine the subset of studies that could form the basis of an OEL value. The diamonds in the figure show that evidence from human and mechanistic studies were evaluated separately and were used to corroborate the animal studies to determine the toxic end points of concern. After points of departure were identified from relevant studies, DOD performed physiologically based pharmacokinetic (PBPK) modeling to calculate human equivalent concentrations from the animal data. A Bayesian statistical approach was used to address uncertainties (Sussan et al. 2019).
Using this conceptual approach, DOD derived candidate OELs for six noncancer end points, including neurological, liver, kidney, immunological, reproductive, and developmental effects (see Table 2-1). DOD then selected the lowest candidate OEL, which was based on immunological effects, as the overall OEL
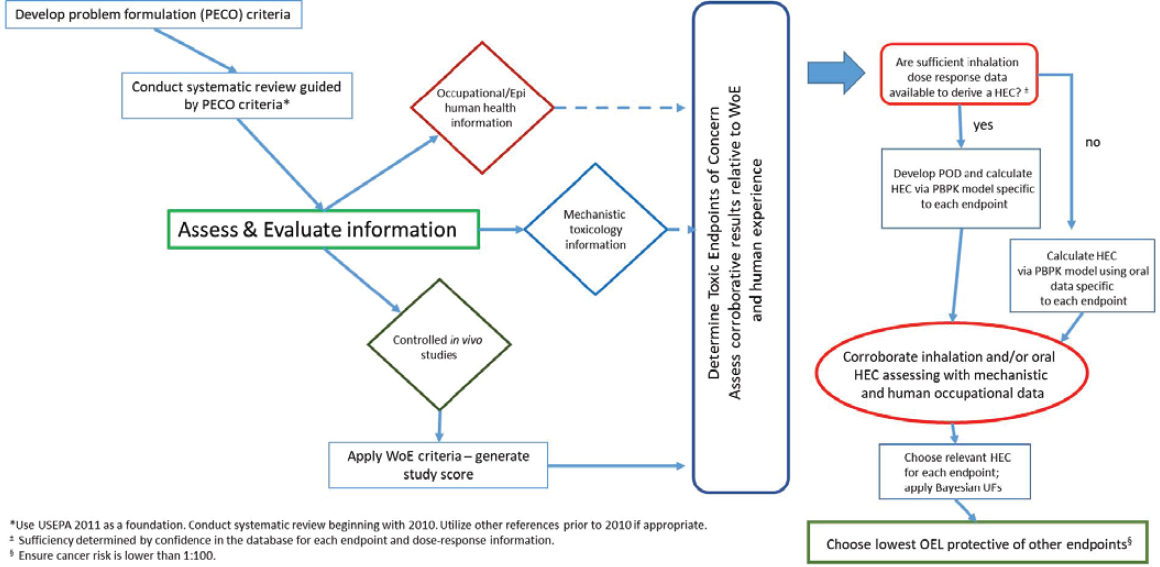
TABLE 2-1 Candidate OELs Developed by DOD
| Toxicity End Point | OEL, ppm | Critical Effect | Key Study | Study Design |
|---|---|---|---|---|
| Kidney | 16 | Increased relative kidney weight | Woolhiser et al. 2006 | Rat, inhalation (6 h/d; 5 d/wk; 4 wk) |
| Liver | 6.9 | Increased relative liver weight | Kjellstrand et al. 1983 | Mouse, inhalation (continuous 30 d) |
| Reproductive | 5.1 | Impaired in vitro fertilization of oocytes from exposed females | Wu et al. 2007 | Rat, oral drinking water (4 d, 5 d, 2 wk) |
| Neurological | 3.8 | Decreased wakefulness | Arito et al. 1994 | Rat, inhalation (8 h/d; 5 d/wk; 2, 4, 6 wk) |
| Developmental | 2.9 | Resorptions, fetal weight | Healey et al. 1982 | Rat, inhalation (4 h/d, gestation days 8-21) |
| Immunological | 0.9 | Effects on cell-mediated sheep red blood cell response and bone marrow stem cells | Sanders et al. 1982 | Mouse, oral drinking water (4, 6 mo) |
SOURCE: Adapted from Sussan et al. 2019.
of 0.9 parts per million (ppm). Selecting the most sensitive OEL was consistent with DOD’s process (see Figure 2-2). DOD’s proposed OEL is more conservative than existing OELs established by OSHA of 100 ppm (OSHA 2019), NIOSH of 25 ppm (NIOSH 2007), and the ACGIH of 10 ppm (ACGIH 2017). DOD intends the OEL to represent an
OEL where nearly all workers may be repeatedly exposed, usually for 8 hours per day for 5 days per week, without adverse health effects. This value is intended to be protective of sensitive populations; but may not be protective of all sensitive individuals; therefore, this analysis is intended to be similar in scope to the OELs published by the ACGIH. (Sussan et al. 2019, p. 4)
DOD’s procedures for deriving the OEL for TCE are evaluated in the next three chapters.
FINDINGS
DOD created a process for developing an OEL that appears specifically designed for TCE. This contrasts with the development of other occupational and public health exposure guidelines, where methods were developed to be applicable to any chemical. If DOD intends to develop OELs for other chemicals, it should consider establishing a generic set of methods in advance of performing the assessments to ensure that OELs are developed in a consistent manner. If only an OEL for TCE is needed, it would have been prudent of DOD to pre-specify the parameters and methods that were used for the evaluation (see Chapters 3 and 4).
REFERENCES
ACGIH (American Conference of Governmental Industrial Hygienists). 2015. Operations Manual: Threshold Limit Values for Chemical Substances Committee. Cincinnati, OH: ACGIH [online]. Available: https://www.acgih.org/tlv-bei-guidelines/policies-procedures-presentations/tlv-bei-committee-operations-manuals [accessed July 1, 2019].
Deveau, M., C-P. Chen, G. Johanson, D. Krewski, A. Maier, K.J. Niven, S. Ripple, P.A. Schulte, J. Silk, J.H. Urbanus, D.M. Zalk, and R.W. Niemeier. 2015. The global landscape of occupational exposure limits—Implementation of harmonization principles to guide limit selection. J. Occup. Environ. Hyg. 12(Suppl. 1):S127-S144.
EPA (U.S. Environmental Protection Agency). 1994. Methods for Derivation of Inhalation Reference Concentrations and Application of Inhalation Dosimetry. EPA/600/8-90/006F. Washington, DC: Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, Office of Research and Development, EPA. October 1994 [online]. Available: https://www.epa.gov/risk/methods-derivation-inhalation-reference-concentrations-and-application-inhalation-dosimetry [accessed May 7, 2019].
EPA. 2002. A Review of the Reference Dose and Reference Concentration Process. EPA/630/P-02/002F. Risk Assessment Forum, EPA, Washington, DC. December 2002 [online]. Available: https://www.epa.gov/risk/review-reference-dose-and-reference-concentration-processes-document [accessed May 7, 2019].
EPA. 2005. Guidelines for Carcinogen Risk Assessment. EPA/630/P-03/001F. Risk Assessment Forum, EPA, Washington, DC. March 2005 [online]. Available: https://www.epa.gov/risk/guidelines-carcinogen-risk-assessment [accessed May 7, 2019].
EPA. 2011. Toxicological Review of Trichloroethylene (CAS No. 79-01-6) in Support of Summary Information on the Integrated Risk Information System (IRIS), September 11, 2011. EPA/635/R-09/011F. Washington, DC: EPA [online]. Available: https://www.epa.gov/iris/supporting-documents-trichloroethylene [accessed April 24, 2019].
IOM (Institute of Medicine). 2011. Finding What Works in Health Care: Standards for Systematic Reviews. Washington, DC: The National Academies Press.
NASEM (National Academies of Sciences, Engineering, and Medicine). 2016a. Refinements to the Methods for Developing Spacecraft Exposure Guidelines. Washington, DC: The National Academies Press.
NASEM. 2016b. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 20. Washington, DC: The National Academies Press.
NASEM. 2018. Progress Toward Transforming the Integrated Risk Information System (IRIS) Program: A 2018 Evaluation. Washington, DC: The National Academies Press.
NRC (National Research Council). 1984. Emergency and Continuous Exposure Limits for Selected Airborne Contaminants: Volume 1. Washington, DC: National Academy Press.
NRC. 1986. Criteria and Methods for Preparing Emergency Exposure Guidance Level (EEGL), Short-term Public Emergency Guidance Level (SPEGL), and Continuous Exposure Guidance Level (CEGL) Documents. Washington, DC: National Academy Press.
NRC. 1992. Guidelines for Developing Spacecraft Maximum Allowable Concentrations for Space Station Contaminants. Washington, DC: National Academy Press.
NRC. 1994. Spacecraft Maximum Allowable Concentrations for Selected Airborne Contaminants: Volume 1. Washington, DC: National Academy Press.
NRC. 2000. Methods for Developing Spacecraft Water Exposure Guidelines. Washington, DC: National Academy Press.
NRC. 2001a. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
NRC. 2001b. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 1. Washington, DC: National Academy Press.
NRC. 2006. Assessing the Human Health Risks of Trichloroethylene: Key Scientific Issues. Washington, DC: The National Academies Press.
NRC. 2008. Spacecraft Water Exposure Guidelines for Selected Contaminants: Volume 3. Washington, DC: The National Academies Press.
NRC. 2009a. Emergency and Continuous Exposure Guidance Levels for Selected Submarine Contaminants: Volume 3. Washington, DC: The National Academies Press.
NRC. 2009b. Science and Decisions: Advancing Risk Assessment. Washington, DC: The National Academies Press.
NRC. 2010. Review of the Environmental Protection Agency’s Draft IRIS Assessment of Tetrachloroethylene. Washington, DC: The National Academies Press.
NRC. 2011. Review of the Environmental Protection Agency’s Draft IRIS Assessment of Formaldehyde. Washington, DC: The National Academies Press.
NRC. 2013. Potential Health Risks to DOD Firing-Range Personnel from Recurrent Lead Exposure. Washington, DC: The National Academies Press.
NRC. 2014. Review of EPA’s Integrated Risk Information System (IRIS) Process. Washington, DC: The National Academies Press.
NTP (National Toxicology Program). 2019. Handbook for Conducting a Literature-Based Health Assessment Using OHAT Approach for Systematic Review and Evidence Integration. Research Triangle Park, NC: Office of Health Assessment and Translation, Division, National Toxicology Program, National Institute of Environmental Health Sciences. March 4, 2019 [online]. Available: https://ntp.niehs.nih.gov/pub-health/hat/review/index-2.html [accessed July 3, 2019].
Sussan, T.E., G.J. Leach, T.R. Covington, J.M. Gearhart, and M.S. Johnson. 2019. Trichloroethylene: Occupational Exposure Level for the Department of Defense. January 2019. U.S. Army Public Health Center, Aberdeen Proving Ground, MD.
Woodruff, T.J., and P. Sutton. 2014. The Navigation Guide systematic review methodology: A rigorous and transparent method for translating environmental health science into better health outcomes. Environ. Health Perspect. 122(10):1007-1014.
Young, R.A., C.B. Bast, C.S. Wood, and F. Adeshina. 2009. Overview of the Standing Operating Procedure (SOP) for the development of Provisional Advisory Levels (PALs). Inhal. Toxicol. 21(Suppl. 3):1-11.









