5
Late Precambrian Bilaterians: Grades and Clades
James W. Valentine
The record of the first appearance of living phyla, classes, and orders can best be described in Wright's (1949) term as "from the top down" (Figure 1). Nearly all of the durably skeletonized (i.e., easily preservable) phyla appear in the Early Cambrian, body plans already in place so far as can be told, and then radiate into numbers of classes, and these into orders, so that the diversity peak of each lower taxonomic rank is shifted towards the present (Valentine, 1969, Erwin et al., 1987). Far more evolution devoted to the rise of body plans and subplans is recorded during the Early Cambrian than during any subsequent geologic Epoch, producing a burst of novelty, termed the Cambrian explosion, that created the "tops." However, there must have been a buildup to those body plans at some time during the preceding Epochs, when the taxa were built ''from the bottom up." Origination and extinction rates of families, genera, and species were highest early in metazoan history, but diversities then were low (Sepkoski, 1984, 1992, 1993; Valentine et al., 1991). The relatively few body fossils known from the late Precambrian do not throw light on the sequence of evolutionary advances that led to the Cambrian taxa. The purpose of this paper is to characterize the evolution of metazoan body plans during the late Precambrian and Early Cambrian, with evidence drawn chiefly from Phanerozoic fossils and from living forms.
James W. Valentine is professor of integrative biology at the University of California, Berkeley.
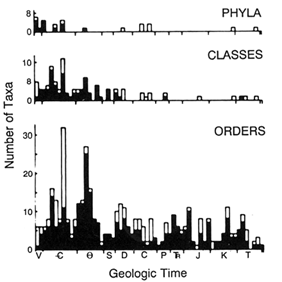
Figure 1 Histograms of first appearances of metazoan phyla, classes, and orders in the marine fossil record. Filled bars, durably skeletonized taxa; open bars, poorly or non-skeletonized taxa. Bar widths equal 10 million years (my). [Reprinted with permission, from Valentine, 1969 (copyright Society for the Study of Evolution).]
Appearance of Phyla in the Fossil Record
Ages of First Appearances. Figure 2 depicts the ages associated with the sequence of Stages and Series of the late Precambrian and Early Cambrian, with most terminology based on the Russian sequence (Bowring et al., 1993). The past couple of decades have witnessed intense work on the early faunas, and during most of that time the base of the Tommotian has been taken as the base of the Cambrian. However, within the last few years new criteria have been developed and now the lowest Cambrian boundary is commonly based on the earliest appearance of the trace fossil Phycodes pedum (see Narbonne et al., 1987). Choosing this boundary has lowered the base of the Cambrian, enlarging that Period by about one half (Figure 2). Despite this expansion of the Cambrian, new absolute age estimates have caused the length of time believed to be available for the Cambrian explosion to be shortened (Bowring et al., 1993). The relationship of the dates given in Figure 2 to the boundaries of the Stages of the Lower Cambrian remains a difficult stratigraphic problem, but it is likely that the most critical Stages, the Tommotian and Atdabanian, are probably only 8–10 my in
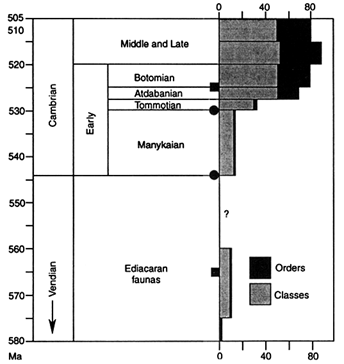
Figure 2 Geochronological time scale for the late Precambrian and Cambrian, with stratigraphic terms derived from the Russian geological column and with recorded metazoan diversity indicated by orders and classes (the earliest records are poorly constrained in time). Dates are from U-Pb zircon ages assigned to the stratigraphic levels indicated by filled circles or squares. [Reprinted with permission, from Bowring et al., 1993 (copyright American Association for the Advancement of Science); diversity data from Sepkoski, 1992.]
duration; over 50 metazoan orders first appear in the record during that interval (Valentine et al., 1991).
Vendian Faunas. The earliest fossils that may be metazoans are preserved in two modes. One is as body fossils, chiefly as impressions in this case. Most of these fossils somewhat resemble cnidarians but many cannot easily be assigned to living classes, and their affinities even as to kingdom are in dispute. Seilacher (1989) has suggested that most of them are not metazoans at all but represent a separate, extinct multicellular clade; they are suggested to have fed via symbionts as chemautotrophs (Seilacher, 1989), phototrophs, or osmotrophs (McMenamin, 1993). However, especially well-preserved specimens of "medusoid"
forms show tentacles, which suggest prey capture, and there are other indications that feeding was by ingestion (Fedonkin, 1994). Other frond-like forms resemble sea pens; Conway Morris (1993) has studied similar forms from the Burgess Shale, wherein preservation is better, and has concluded that both Vendian and Burgess Shale forms are cnidarians. It seems likely on present evidence that most of the late Precambrian forms do have cnidarian affinities. Even so, the late Precambrian body fossils do not represent direct ancestors of any of the higher metazoans and do not help to resolve the puzzle of the origin of the remaining phyla. A few other body fossils may represent bilaterians (see below), but such an assignment is disputed also.
The other mode of preservation of late Precambrian forms is as trace fossils—markings made by animal activities (Glaessner, 1969; Fedonkin, 1985b; Crimes, 1989). Some traces are of winding, rather featureless, trails, but others display transverse rugae and contain pellets that can be interpreted as of fecal origin. The bilaterian nature of these traces is not in dispute. Furthermore, such traces must have been made by worms, some of which had lengths measured in centimeters, with through guts, which were capable of displacing sediment during some form of peristaltic locomotion, implying a system of body wall muscles antagonized by a hydrostatic skeleton. Such worms are more complex than flatworms, which cannot create such trails and do not leave fecal strings (Fedonkin and Runnegar, 1992; Fedonkin, 1994). It is among the trace makers that the ancestors of the Cambrian clades are most likely to be represented.
Early Cambrian Faunas. During the Manykaian Stage mineralized skeletons begin to appear, and during the Tommotian and Atdabanian they increase spectacularly in numbers and diversity (Bengtson and Conway Morris, 1992). Many of these fossils are dissociated sclerites that give few clues as to the nature of their scleritomes or of the animals that bore them. In a few cases, however, sclerites have been found in life associations or preserved with soft-bodied remains to provide indications of a body plan (Conway Morris and Peel, 1990; Hinz et al., 1990; Ramsköld and Hou, 1991). Of living phyla, skeletons of brachiopods, mollusks, arthropods (trilobites), and echinoderms appear in the Tommotian and Atdabanian Stages, and nearly all durably skeletonized phyla are known by the end of the Early Cambrian. The exceptions are chordates (Middle Cambrian) and bryozoans (Early Ordovician); however, the body plans of neither of those phyla require a mineralized skeleton and both may have been present well before they appear as fossils. Body fossils of some phyla or subphyla lacking mineralized skeletons (priapulans, onychophoran relatives, etc.) are known from
rocks of probable Atdabanian age from the Baltic Shield (Dzik and Lendzion, 1988 and refs. therein), China (Hou et al., 1991), and Greenland (Conway Morris et al., 1987), together with a variety of animals that cannot be assigned with any confidence to living phyla (Dinomischus, paleoscolicids, halkieriids, etc.).
Trace fossils in the Early Cambrian are greater in abundance and diversity than in the late Precambrian (Crimes, 1992). Penetrating vertical burrows, exceedingly rare and small earlier, become larger, longer, and more common, and bioturbation increases in depth and intensity in increasingly younger sediments (Crimes and Droser, 1992; Droser and Bottjer, 1993). The increased biological activity indicated by traces is consistent with that indicated by the explosion of body fossil types.
Middle Cambrian Faunas. The rate of appearance of novel body plans slows greatly during the Middle Cambrian with the exception of the fauna of the Burgess Shale and its correlatives, which create a diversity "spike" (Whittington, 1985; Conway Morris, 1992). The Burgess Shale fauna is exceptionally diverse and brings to light many of the less easily preserved members of the Cambrian fauna. The Chinese Atdabanian fauna is also exceptionally preserved and contains numbers of taxa in common with the Burgess Shale, suggesting that many of the Burgess Shale forms, or at least the higher taxa to which they belong, would be found to have originated in the Early Cambrian if that fauna were better known.
Summary. It is consistent with the fossil record that all of the body plans now ranked at the phylum level originated by the close of the Early Cambrian, although some that are not easily fossilized do not appear until later, and indeed some (platyhelminths, gnathostomulids, gastrotrichs, acanthocephalans, loriciferans, and kinorhynchs) are unknown as body fossils. As the very first bearers of any given body plan are unlikely to be found, any corrections for a smearing out of first appearances towards the Recent may add to the abruptness of the Cambrian explosion. The first appearances of fossil groups whose relationships are problematic but that have very distinctive skeletal or body plans and that may be phyla or subphyla are also concentrated during the Early Cambrian. The Cambrian explosion was geologically abrupt and taxonomically broad.
The Rise of Body-Plan Complexity
Metazoans evidently originated from unicellular (perhaps colonial) choanoflagellates or their allies (Wainright et al., 1993). By the time of the Cambrian explosion, some metazoan bodies were as complex as primi-
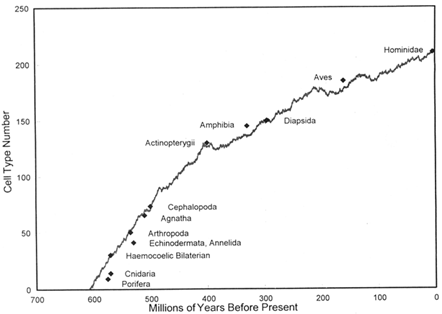
Figure 3 Estimated somatic cell-type number (except nerve cells) required by the body plans of various taxa, plotted against the time of origin of those taxa indicated by the fossil record. The irregular curved line represents the upper bound of cell-type number generated by a stochastic model based on passive increase or decrease of cell-type number within each lineage during diversification of lineages from 1 to 2000; the line is an average of five runs. [Reproduced with permission, from Valentine et al., 1994 (copyright Paleobiology).]
Cambrian explosion, some metazoan bodies were as complex as primitive arthropods and other higher invertebrates. This rise in complexity is obscured in the fossil record. Perhaps the best practical index of body-plan complexity is cell-type number (Bonner, 1965; Sneath, 1964; Raff and Kaufman, 1983), which may be taken to have begun as two in metazoans and to have increased through time as more complex bodies evolved. Figure 3 depicts the estimated cell-phenotype numbers of the more complex body plans known at a given time during the history of the metazoans, plotted against the times of their first appearances as judged by the fossil record (Valentine et al., 1994). A curve embracing the points should approximate the upper bound of body-plan complexity. Complexity increases may be forced, perhaps by natural selection, or may be passive, resulting from random opportunities to become either more or less complex (Fisher, 1986; Gould, 1988; McShea, 1991,
1993). In Figure 3, a computer model of random change in cell-type number in diversifying lineages over 4000 steps produced the upper bound shown by the shaded line (Valentine et al., 1994). In that model the number of lineages began at one and increased logistically to 2000; the addition or subtraction of a cell type in each lineage in any step was treated as a Markov process. The model curve is scale to geological time by being pinned to landmarks at 30 and 210 cell types but is free elsewhere.
The chief caveats in interpreting this model relate to the estimates of cell-phenotype numbers indicated on Figure 3. The estimates represent a sampling of cell types and are made with a lumper's approach, and nerve cell types are not distinguished (Valentine et al., 1994). For the simpler organisms the numbers may be fairly accurate, but for increasingly complex forms the figures increasingly underestimate true cell-type numbers. However, the intent is not to measure an increasing developmental complexity, or an increasing information content of metazoan genomes, but simply to reflect the gross morphological complexity of the body plans. Nevertheless, that portion of the curve represented by chordates should be treated with caution. As the early portion of the curve should be on the firmer footing, it is worth considering its implications for the early history of the metazoans and for the Cambrian explosion.
Although the model is not meant to replicate the history of metazoan complexity, the behavior of the upper bound suggests that no forcing mechanism may be necessary to account for the empirical complexity increase in early metazoan body plans. The relatively rapid initial increase in complexity, created partly by a "floor" of two cell types and partly by the diversification of clades, may have been a feature of real clades even in the absence of forcing agents. The origin of metazoans may thus be hypothesized to have been near 600 my ago, more recently than is usually supposed. The rise in complexity is parabolic in the model. At present there is no evidence of a major step in body-plan complexity during the Cambrian explosion. The implication is that when animals with, say, 45 cell types appear during the explosion, there were ancestors of that clade with 44 cell types (or in that general region) that we don't see, and complexities should dwindle, perhaps at an increasing rate as we go back farther in time. Again, the ancestors may be known today only by the late Precambrian traces.
Phylogenetic Models and Body Plans
The Molecular Phylogenetic Model. Expectations as to the body plans of late Precambrian bilaterians depend partly upon phylogenetic models.
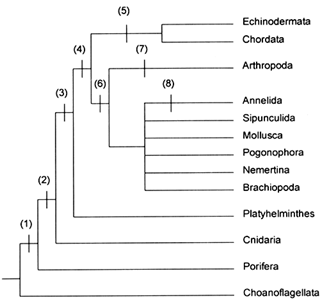
Figure 4 Branching sequence of lineages leading to some modern phyla as indicated by some of the more robust 16S and 18S rRNA molecular phylogenies, with a hypothesized sequence of introduction of some design elements in their body plans as indicated. Branch lengths or spacings are not scaled. Design elements are 1, collagen, tissues; 2, gastrulation, enteron; 3, mesoderm; 4, blood-vascular system; 5, oligomerous coelom; 6, hemocoel; 7, arthropodan segmentation; 8, annelidan segmentation. If the unresolved rRNA branching events leading to various protostomes occurred within proto-mollusks, it may not be possible in principle to characterize them by synapomorphies that are germane to the derived features of living phyla. (Molecular data from various sources but chiefly Wainright et al., 1993; Field et al., 1988; Lake, 1990; Turbeville et al., 1991; Turbeville et al., 1992; Raff, 1994; design element sequence chiefly from Valentine, 1992.)
There is hardly a scheme of relationships that hasn't been suggested by one worker or another; the models that have been most important have been reviewed by Willmer (1990). Comparative studies of 16S and 18S rRNA sequences from living phyla have provided new evidence as to ancestral branching patterns (e.g., Field et al., 1988; Lake, 1990; Turbeville et al., 1991, 1992; Wainright et al., 1993; Wheeler et al., 1993; Raff, 1994). While these findings are still provisional and involve some contradictions, most can be rationalized with morphologic and developmental evidence and accord well with the fossil evidence, so they will be used here as the basis of a phylogenetic scenario. Figure 4 summa-
rizes some of the more robust findings by listing some phyla from which rRNAs were studied to the right and the inferred branching patterns of the rRNAs on the left, with the implied appearance of some design elements in metazoan body plans indicated. A few of the design elements require comment. For present purposes, the unqualified terms "hemocoel" and "coelom" refer to fluid-filled spaces usually used as hydrostatic skeletons—the former being developed from the topological position of the blastocoel, the latter being developed within mesoderm. Spaces devoted only to serving organs (commonly as ducts or as buffering against solid tissues) are termed "organ hemocoels" or ''organ coeloms." "Seriation" refers to longitudinal repetition of organs, muscles, or other features but not necessarily in a correlated fashion, while "segmentation" refers to serial repetition with correlation among organ systems.
Many of the metazoan relationships indicated in Figure 4 are quite conventional, such as the sequence of choanoflagellates/poriferans/cnidarians/platyhelminths, the sister-group relationship of platyhelminths with higher metazoans, and the sister-group relationship of protostomes with deuterostomes. Less conventional, though hardly shocking, is the sister-group relationship of the arthropods with the unresolved cluster of protostomes, which includes mollusks and annelids. This is a particularly important branching, because it suggests that the last common ancestor of the protostomes was hemocoelic and that the coelom(s) of the deuterostomes arose independently of the coelom(s) of the protostomes. This branching also suggests that arthropod and annelid segmentation arose independently. The next deeper branching, between protostomes and deuterostomes, suggests that while their last common ancestor was not coelomic and need not have been hemocoelic, it did have a blood-vascular system. Other interpretations are possible but they are less parsimonious.
Late Precambrian Body Plans. The list of phyla on the right of Figure 4 indicates the body plans of the living organisms studied but certainly does not indicate the body plans of the common ancestors deeper within the branching pattern. On present evidence all of the branching between the choanoflagellate/poriferan and the arthropod/unresolved protostome branches occurred during the Precambian. The bilaterian traces of the Vendian must have been made by descendants of the platyhelminth/higher metazoan branch. The simple earliest trails require active worms with some hydrostatic system, possibly a tissue skeleton, or fluid in part; the relief indicated by these traces implies a non-flat body and that, in turn, a blood-vascular system. Such a worm, a vascularized "roundish flatworm," acoelomate (except possibly for organ spaces), probably
appropriate ancestor for both protostomes and deuterostomes. Today the nearest approximations to a worm of this grade are found among the mollusks—aplacophorans and chitons—although these living groups have derived features that would have been absent in the Vendian. The more complex horizontal trails or shallow burrows appearing later imply a more efficient hydrostatic system and thus probably an ample hemocoel. Again, worms of this grade would probably be regarded as simplified mollusks, were they to be found alive and shoehorned into a living phylum.
If the sequence of introductions of design elements was truly parsimonious, the ancestral arthropods branched from this sort of proto-mollusk. A plausible scenario would begin with a seriated form, probably preferring hard grounds and supplementing a fundamentally peristaltic creeping locomotion by lateral body projections that served as accessory gripping mechanisms. Sclerotization may have begun as protection in such a habitat, since burrowing would not be possible, but flexibility of the body wall and peristaltic efficiency would have been sacrificed as it became heavier. Locomotory functions shifted to the lateral appendages, and a segmented anatomy evolved as series of muscles, nerves, and blood vessels developed to serve them. Flexibility of the trunk was maintained by jointing of the integument; this permitted vertical flexure when creeping over uneven substrates and perhaps lateral flexure to enhance locomotory power as needed through the use of the body wall musculature (Valentine, 1989). The jointing enhanced the segmented architecture. Body spaces continued to be developed on the site of the blastocoel: a hemocoel to provide hydrostatic functions and an enlarged organ hemocoel to bathe the heart. Jointing of the appendages occurred as they lengthened within a rigidifying exoskeleton. By the Tommotian, arthropod-type appendages were sufficiently well developed to permit their use in furrowing the substrate, presumably in search of food, which created a characteristic type of trace fossil (Crimes, 1989), probably reflecting an evolutionary radiation of jointed body types onto soft substrates.
The number of independent lineages that reached the arthropod condition has been in dispute. An analysis of the branching order of 12S rRNA within the arthropods (Ballard et al., 1992) suggests that ancestors of the myriapods branched most deeply and were followed by branchings that produced the ancestors of onychophorans (a lobopodal group that does not have jointed appendages) and then of the chelicerates, crustaceans, and hexapods in that order. However, an 18S rRNA tree (Wheeler et al., 1993) suggests that the onychophorans are sisters to a monophyletic arthropod clade, a view supported by a consensus tree
that includes morphological and Ubiquitin sequence data. A number of fossil types that can be interpreted as lobopods are known from the Early (possibly Tommotian) and Middle Cambrian (Ramsköld and Hou, 1991; Bengtson et al., 1986; Budd, 1993). Living lobopods have hemocoels but flexible body walls (and can therefore squeeze through narrow openings). These data leave open the question of the body plans associated with the branch points. Perhaps the most likely possibility is that the onychophoran and arthropodan ancestors branched from a hemocoelic, seriated form before jointed, sclerotized exoskeletons appeared.
Among the Vendian body fossils of uncertain afinity are a few that have sometimes been interpreted as segmented bilaterians, including sprigginids and vendomiids (Glaessner, 1984; Fedonkin, 1985a). These forms are serially constructed and, if bilaterians, have cephalic shields but lack appendages; they have also been reconstructed as frond-like forms (Seilacher, 1989; Bergström, 1989). In some of them at least, the putative jointing alternates across the midline, so that "segments" are laterally offset rather than continuous; however, offsetting of serial structures is known in many living bilaterians (e.g., muscle blocks in cephalochordates) (Jeffries, 1986). If these fossils are bilaterians, then they may belong to the grade of segmented, hemocoelic organisms postulated to have given rise to arthropods (Valentine, 1989, 1992).
Ancestral annelids may also have branched from proto-molluscan grade ancestors, but the formation of the annelid body plan involved the origin of the famous compartmented coelom, widely interpreted as a hydrostatic skeleton to aid in peristaltic burrowing (Clark, 1964) or horizontal plowing. Undoubted annelidan body fossils have not been found in rocks older than Middle Cambrian; the earliest penetrating burrows may have been made by so-called pseudocoelomates such as priapulids or paleoscolecids, both of which are known in the Lower Cambrian. Long tubes presumably constructed by vermiform organisms, the sabelliditids, occur in the Manykaian and are sometimes considered to be pogonophorans, but no body fossils of these forms are known. Like arthropods, annelids possess larval intramesodermal spaces. In various arthropod taxa the spaces are occluded, or become organ coeloms, or are incorporated into the blood-vascular system during development; in annelids they are lost during metamorphosis, although the adult coelom develops (by schizocoely) within tissues that are derived from larval mesoderm (Anderson, 1973). If the larval coeloms in these phyla are homologous they are presumably plesiomorphic. In the larvae of some phyla, an intramesodermal space serves as a nephridium (Ruppert and Balser, 1986), and possibly this was the original function in this case as well. The segmentation in annelids is not
restricted to the locomotary system, although it does involve the parapodia and their musculature and its vascularization and innervation. Unlike the arthropods, annelidan gonads and nephridia are also seriated in concert with the coelomic compartmentalization. Annelidan and arthropodan segmentation, though associated with locomotion in each case, operate on different principles and involve some different organ systems including the fluid skeletons; there is little reason to regard them as homologous. Eernisse et al. (1992) have presented a cladistic analysis that arrives at similar conclusions (but see Backeljau et al., 1993).
Brachiopods are commonly regarded as allied to phoronids and bryozoans, as all three phyla possess similarly regionated coeloms and lophophores; this relationship has not been corroborated by molecular techniques at this writing. According to the 18S rRNA tree, the brachiopods may have arisen from the last common arthropod—annelid ancestor and therefore from a proto-molluscan grade in the present scenario. It has been suggested (Valentine, 1992) that an unsegmented coelom was evolved within an ancestral worm clade for locomotion in soft sediments; direct peristalsis, which requires an unsegmented coelom, is a common locomotory technique in flocculent muds, for example (Elder, 1980). Radiation of this clade onto firmer substrates may then have produced sessile forms with regionated coeloms to serve both a trunk and a tentacular crown, the feeding lophophore. Perhaps annelids have also descended from a small-bodied ancestor with an unregionated coelom, with segmentation evolving for efficiency in locomotion.
Mollusks exhibit the body plan that is least derived from that of the postulated protostome ancestor, and most molluscan classes have only organ coeloms. However the cephalopods, which form the most advanced molluscan class and which appear only in the Upper Cambrian, have a well-developed coelomic space (a gonocoel?) that can be regarded as a novel evolutionary feature. The nemertine rhynchocoel, which has been demonstrated to be a coelom histologically (Turbeville, 1991), probably was derived independently.
Late Vendian and Manykaian bilaterian body plans are thus visualized as consisting of an array of vermiform types, including flatworms and "round flatworms" with blood-vascular systems, many with hemocoels or "pseudocoels," and some with seriation of one organ system or another (Valentine, 1989, 1990; Bergström, 1989). Organ coeloms were doubtless present in many lineages. Possibly some of these worms were incipiently segmented in the arthropod style. It seems likely that some worms had unregionated perivisceral coeloms, but it is doubtful that any had annelid-style coelomic segmentation. In this scenario, the body plans of these worms were based primarily upon adaptations to loco-
motory requirements on a variety of substrates, radiating from at least two proto-molluscan body plans, one with a simple blood vascular system and the other with a hemocoel. Once a successful new locomotory system was evolved on one substrate type, invasion and reinvasion of other substrates became possible and the diversity of types increased further. These worms have left us an array of trace fossils and evidently little else, except their descendants, among whom we number.
Tempo and Mode and Body Plans
The more complex of the late Precambrian worms must have been higher invertebrates in every sense, with, by invertebrate standards, sophisticated organ systems consisting of appropriately specialized tissues and these in turn composed of differentiated cell phenotypes that, judging by the body plans of living organisms, probably numbered in the 40s. Other worms were clearly less complex. For those worms that gave rise to Phanerozoic clades, it is expected that their primitive members were only minimally more complex than their ancestors, and it is plausible that the ancestral body plans were precursory to their Phanerozoic descendants. These considerations, and the evidence reviewed above, imply that many of the branching events required of metazoan diversifications occurred well before the onset of the Cambrian explosion. After the branchings, complexity continued to increase within many clades. For example, proto-mollusks gave rise to several distinctive hemocoelic and coelomic body plans, and early mollusks gave rise to the relatively complex cephalopods. The pattern of branching during the Vendian appears as a more-or-less orderly accumulation of a series of derived features. This has been the usual interpretation of the metazoan diversification pattern, more-or-less faute de mieux, though occasionally it has been challenged by proponents of a bush or grass-like pattern of many parallel lineages rising from a common ancestral clade (Nursall, 1962), which in the latest version is composed of flatworms (Willmer, 1990; Barnes et al., 1993). It appears more likely that the body plans from which numbers of phyla evolved were proto-molluscan.
Internal Factors. The mode of evolution that might account for the observed explosive appearance of body plans in the fossil record must have involved changes in genetic regulation within many lineages, including repatternings of gene expression and respecifications of cell fates and movements, but perhaps with little increase in levels of cell differentiation. Evidence of the nature of the transcriptional regulation of these processes is beginning to accumulate. It is clearly of interest to study the similarities and differences among the regulators that mediate
body-plan formation in metazoans, in the context of the molecular phylogenetic tree and of the sequence of morphological complexities outlined above, but such comparative data are still scarce. Many of the relevant data involve homeobox genes, best known for their roles in determining developmental patterns in Drosophila and mice (Akam, 1987; Shashikant et al., 1991). It appears that these genes are widely (probably universally) distributed in metazoan phyla (McGinnis, 1985; Kenyon and Wang, 1991; Schierwater et al., 1991; Webster and Mansour, 1992; Oliver et al., 1992; Schummer et al., 1992; Bartels et al., 1993; Chalfie, 1993); they are highly conserved, and some are found in conserved clusters, such as the homeotic Hox/HOM genes. The anterior boundaries of the expression of these genes occur in a sequence along the anteroposterior body axis of bilaterians in the conserved order in which the genes occur in the cluster.
Most nonchordate phyla are thought to have only a single Hox/HOM cluster. Relative to higher metazoans, few Hox/HOM genes have been found in platyhelminths (Bartels et al., 1993) and there may be only a small cluster in nematodes, although they have perhaps 60 homeobox genes (Chalfie, 1993; Bürglin and Ruvkun, 1993). So far as is known, most higher invertebrates have a single cluster; cephalochordates are inferred to have two clusters, agnathans to have three (or four), and mammals have four (Pendleton et al., 1993). Most or all of the Hox/HOM genes within clusters evidently arose by duplications beginning with an ancestral homeobox gene, while multiple clusters such as found in chordates arose later by cluster duplication, homologous genes commonly being more similar among clusters than within them (Kappen et al., 1989). Cartwright et al. (1993) have produced evidence from a survey of Hox/HOM genes in the chelicerate Limulus polyphemus that it may have four clusters. A form interpreted as an ancestral chelicerate (it lacks chelicerae) is described from the Middle Cambrian Burgess Shale (Briggs and Collins, 1988); whether any cluster duplication had occurred by then is unknown.
In addition to the homeotic genes, a cascading series of pattern-formation genes is expressed earlier in metazoan development and mediates the progressive regionation and differentiation of body form; in Drosophila these include maternal genes and gap, pairrule, and segment-polarity genes, expressed in that order (Akam, 1987; Ingham, 1988). Some of these genes, such as the segment-polarity gene engrailed, are known to be represented by homologs in, among other organisms, mice (see Patel et al., 1989), and thus were presumably present in a common protostome–deuterostome ancestor. The segment-polarity gene hedgehog (hh) has been shown to have a homolog in chordates, Sonic hedgehog (Shh), that mediates polarity in the developing central
nervous system, trunk, and limbs of various members of that phylum, activating Hox genes (Riddle et al., 1993; Echelard et al., 1993; Krauss et al., 1993). It is an obvious hypothesis that a gene ancestral to hh and Shh was present and mediated polarizing activity in the last common protostome—deuterostome ancestor and is probably much more ancient.
It is possible to speculate on aspects of the general course of evolution of metazoan body plans, assuming that the preceding picture of early metazoan evolution is approximately correct. Homeobox genes presumably arose within protistans, but took on a role in the specification of cell fates, movements, and patterns as cell differentiation accompanied the rise of multicellular organisms. During the first few tens of millions of years of metazoan history a regulatory cascade was assembled, mediating the transcription of a growing morphological complexity. If the rise of complexity was as rapid as hypothesized here, it seems likely that many of these regulatory pathways were already present in protistans. The number of transcriptional regulators may have grown hand in hand with the upper bound of metazoan complexity. Bilaterians became increasingly differentiated along their anteroposterior body axes, and this trend may be reflected in the growth of the Hox/HOM cluster. The numbers of homeobox genes besides those in this cluster (and of other regulatory gene types) must have increased as well. At some point during this trend, bilaterians became able to displace sediment and thus to produce trace fossils, and they entered the fossil record of the Late Vendian. By the time that the last common ancestor of deuterostomes and protostomes evolved, presumably still during the Vendian, a large Hox/HOM cluster was present in that lineage. The Cambrian explosion, then, may have occurred largely or entirely within organisms that had a single Hox/HOM cluster. However, the major increase in the upper bound of complexity during the Phanerozoic may be associated with a series of duplications of the cluster.
The postulated branching of lineages at proto-molluscan grades that led eventually to a variety of descendants with distinctive body plans requires that the details of pattern formation responsible for the derived features of each of those body plans evolved independently. In this view the Hox/HOM cluster had a responsibility for anteroposterior differentiation in the proto-mollusk (and earlier) and retained that role, coming to be associated with the development of segmentation in arthropods, in annelids, and with anteroposterior structures in vertebrates, even though those features are quite different and evolved independently. Many early developmental steps also were greatly modified after the ancestral branching that led to separate phyla (Davidson, 1990, 1991). In other words, nearly all of the body architectures, and many of the
developmental steps that characterize the phyla, evolved after the pattern formation and selector genes were assembled. The Cambrian explosion in metazoan body plans may not have involved any great expansion of the gene regulatory apparatus but probably could not have occurred if the regulatory systems were not already sophisticated, and thus able rapidly to create novel morphologies, as the morphological innovations were evidently achieved through modes of regulatory evolution (Britten and Davidson, 1971; Valentine and Campbell, 1975; Jacobs, 1990; Valentine, 1994).
External Factors. The abrupt appearance of higher taxa in the Cambrian has stimulated a search for possible environmental changes that might have produced this evolutionary outburst; there has been no shortage of suggestions (Valentine et al., 1991; Signor and Lipps, 1992). However, the geological record has not revealed unequivocal evidence of unique Cambrian events that might be held accountable. One of the more common suggestions has been that oxygen levels rose in the late Precambrian to values that could sustain metazoans and thus permitted evolution to produce increasingly active organisms through time. Recent studies have indeed revealed series of late Precambrian—Cambrian geochemical excursions that can be plausibly interpreted as involving CO2 and oxygen levels and as being associated with the biogeochemical cycling of the time (Knoll, 1994).
It is not clear that an external trigger was needed to produce the Cambrian explosion. A continuous expansion of an already moderately complex fauna as various lineages acquired evolutionary access to broadening arrays of marine habitats, with the resulting enhancement of ecological interactions within that fauna, may be all that was required to produce the record we have, insofar as the origin of phyla is concerned. However, continuing geochemical and other studies will surely lead to a better understanding of the nature of environmental change during the Cambrian explosion, and then we shall be better able to judge the possible role of physical triggers.
Summary
A broad variety of body plans and subplans appear during a period of perhaps 8 million years (my) within the Early Cambrian, an unequaled explosion of morphological novelty, the ancestral lineages represented chiefly or entirely by trace fossils. Evidence from the fossil record can be combined with that from molecular phylogenetic trees to suggest that the last common ancestor of (i) protostomes and deuterostomes was a roundish worm with a blood vascular system and (ii) of arthropods and
annelids was similar, with a hydrostatic hemocoel; these forms are probably among trace makers of the late Precambrian. Cell-phenotype numbers in living phyla, and a model of cell-phenotype number increase, suggest an origin of metazoans near 600 my ago, followed by a passive rise in body-plan complexity. Living phyla appearing during the Cambrian explosion have a Hox/HOM gene cluster, implying its presence in the common ancestral trace makers. The explosion required a repatterning of gene expression that mediated the development of novel body plans but evidently did not require an important, abrupt increase in genomic or morphologic complexity.
I thank Dave Jablonski (University of Chicago) and Allen Collins and Chris Meyer (University of California, Berkeley) for valuable discussions and reviews of the manuscript, and Clint Turbeville for insights into coelom development. This research was based on work supported by Grant EAR-9196068, National Science Foundation, and by Faculty Research grants, University of California, Berkeley.
References
Akam, M. (1987) The molecular basis for metameric pattern in Drosophila embryos. Development 101, 1–22.
Anderson, D. T. (1973) Embryology and Phylogeny of Annelids and Arthropods (Pergamon, Oxford).
Backeljau, T., Winnepenninckx, B. & De Bruyn, L. (1993) Cladistic analysis of metazoan relationships: a reappraisal. Cladistics 9, 167–181.
Ballard, J. W. O., Olsen, G. J., Faith, D. P., Odgers, W. A., Rowell, D. M. & Atkinson, P. W. (1992) Evidence from 12S ribosomal RNA sequences that onychophores are modified arthropods. Science 258, 1345–1348.
Barnes, R. S. K., Callow, P. & Olive, P. J. W. (1993) The Invertebrates, 2nd. Ed. (Blackwell Sci., Oxford).
Bartels, J. L., Murtha, M. T. & Ruddle, F. H. (1993) Multiple Hox/HOM -class homeobox genes in Platyhelminthes. Mol. Phylog. Evol. 2, 143–151.
Bengtson, S. & Conway Morris, S. (1992) Early radiation of biomineralizing phyla. In Origin and Early Evolution of the Metazoa, eds. Lipps, J. H. & Signor, P. W. (Plenum, New York), pp. 448–481.
Bengtson, S., Matthews, S. C. & Missarzhevsky, V. V. (1986) The Cambrian netlike fossil Microdictyon. In Problematic Fossil Taxa, Hoffman, A. & Nitecki, M. H. (Oxford Univ. Press, Oxford), pp. 97–113.
Bergström, J. (1989) The origin of animal phyla and the new phylum Procoelomata. Lethaia 22, 259–269.
Bonner, J. T. (1965) Size and Cycle (Princeton Univ. Press, Princeton, NJ).
Bowring, S. A., Grotzinger, J. P., Isachsen, C. E., Knoll, A. H., Pelechaty, S. M. & Kolosov, P. (1993) Calibrating rates of Early Cambrian evolution. Science 261, 1293–1298.
Briggs, D. E. G. & Collins, D. (1988) A Middle Cambrian chelicerate from Mount Stephen, British Columbia. Palaeontology 31, 779–798.
and a speculation on the origins of evolutionary novelty. Quart. Rev. Biol. 46, 111–133.
Budd, G. (1993) A Cambrian gilled lobopod from Greenland. Nature 364, 709–711.
Bürglin, T. R. & Ruvkun, G. (1993) The Caenorhabditis elegans homeobox gene cluster. Curr. Opin. Genet. Develop. 3, 615–620.
Cartwright, P., Dick, M. & Buss, L. W. (1993) HOM/Hox type homeoboxes in the chelicerate Limulus polyphemus. Mol. Phyl. Evol. 2, 185–192.
Chalfie, M. (1993) Homeobox genes in Caeonorhabditis elegans. Curr. Opin. Genet. Develop. 3, 275–277.
Clark, R. B. (1964) Dynamics in Metazoan Evolution (Clarendon Press, Oxford).
Conway Morris, S. (1992) Burgess Shale-type faunas in the context of the "Cambrian explosion": a review. Jour. Geol. Soc. London 149, 631–636.
Conway Morris, S. (1993) Ediacaran-like fossils in Cambrian Burgess Shale-type faunas of North America. Palaeontology 36, 593–635.
Conway Morris, S. & Peel, J. S. (1990) Articulated halkieriids from the Lower Cambrian of north Greenland. Nature 345, 802–805.
Conway Morris, S., Peel, J. S., Higgins, A. K., Soper, N. J. & Davis, N. C. (1987) A Burgess Shale-like fauna from the Lower Cambrian of north Greenland. Nature 326, 181–183.
Crimes, T. P. (1989) Trace Fossils. In The Precambrian-Cambrian Boundary , eds. Cowie, J. W. & Brazier, M. D. (Clarendon Press, Oxford), pp. 166–185.
Crimes, T. P. (1992) Changes in the trace fossil biota across the Proterozoic–Phanerozoic boundary. Jour. Geol. Soc. London 149, 637–646.
Crimes, T. P. & Droser, M. L. (1992) Trace fossils and bioturbation: the other fossil record. Ann. Rev. Ecol. Syst. 23, 339–360.
Davidson, E. H. (1990) How embryos work: a comparative view of diverse modes of cell fate specification . Development 108, 365–389.
Davidson, E. H. (1991) Spatial mechanisms of gene regulation in metazoan embryos. Development 113, 1–26.
Droser, M. & Bottjer, D. J. (1993) Trends and patterns of Phanerozoic ichnofabrics. Ann. Rev. Earth Planet. Sci. 21, 205–225.
Dzik, J. & Lendzion, K. (1988) The oldest arthropods of the East European Platform. Lethaia 29–38.
Echelard, Y., Epstein, D. J., St-Jacques, B., Shen, L., Mohler, J., McMahon, J. A. & McMahon, A. P. (1993) Sonic hedgehog, a member of a family of putative signalling molecules, is implicated in the regulation of CNS polarity. Cell 75, 1417–1430.
Eernisse, D. J., Albert, J. S. & Anderson, F. E. (1992) Annelida and Arthropoda are not sister taxa: a phylogenetic analysis of spiralian metazoan morphology. Syst. Biol. 41, 305–330.
Elder, H. Y. (1980) Peristaltic mechanisms. In Aspects of Animal Movement , eds. Elder, H. Y. and Trueman, E. R. (Cambridge Univ. Press, Cambridge), pp. 71–92.
Erwin, D. H., Valentine, J. W. & Sepkoski, J. J., Jr. (1987) A comparative study of diversification events: the early Paleozoic versus the Mesozoic. Evolution 41, 1177–1186.
Fedonkin, M. A. (1994) Vendian body fossils and trace fossils. In Early Life on Earth, ed. Bengtson, S. (Columbia Univ. Press, New York), pp. 370–388.
Fedonkin, M. A. (1985a) Systematic description of Vendian Metazoa. In The Vendian System Vol. 1, eds. Sokolov, B. S. & Ivanovsky, A. B. (Nauka, Moscow), Vol. 1, pp. 70–106 (in Russian).
Fedonkin, M. A. (1985b) Systematic description of Vendian Metazoa. In The Vendian
Fedonkin, M. A. (1985b) Systematic description of Vendian Metazoa. In The Vendian System Vol. 1, eds. Sokolov, B. S. & Ivanovsky, A. B. (Nauka, Moscow), Vol. 1, pp. 112–117 (in Russian).
Fedonkin, M. A. & Runnegar, B. (1992) Proterozoic metazoan trace fossils. In The Proterozoic Biosphere, eds. Schopf, J. W. & Klein, C. (Cambridge Univ. Press, Cambridge), pp. 389–395.
Field, K. G., Olsen, G. J., Lane, D. J., Giovannoni, S. J., Ghiselin, M. G., Raff, E. C., Pace, N. R. & Raff, R. A. (1988) Molecular phylogeny of the animal kingdom. Science 239, 748–753.
Fisher, D. C. (1986) Progress in organismal design. In Patterns and Processes in the History of Life, eds. Raup, D. M. & Jablonski, D. (Springer, Berlin), pp. 99–117.
Glaessner, M. (1969) Trace fossils from the Precambrian and basal Cambrian. Lethaia 2, 369–393.
Glaessner, M. F. (1984) The Dawn of Animal Life (Cambridge Univ. Press, Cambridge).
Gould, S. J. (1988) Trends as changes in variance: a new slant on progress and directionality in evolution. Paleontol. 62, 319–329.
Hinz, I., Kraft, P., Mergl, M. & Müller, K. J. (1990) The problematic Hadimopanella, Kaimenella, Milaculum and Utahphospha identified as sclerites of Paleoscolecida. Lethaia 23, 217–221.
Hou, X.-G., Ramsköld, L. & Bergström, J. (1991) Composition and preservation of the Chengjiang fauna—a Lower Cambrian soft-bodied biota. Zool. Scr. 20, 395–411.
Ingham, P. (1988) The molecular genetics of embryonic pattern formation in Drosophila. Nature 335, 25–34.
Jacobs, D. K. (1990) Selector genes and the Cambrian radiation of the bilateria. Proc. Natl. Acad. Sci. USA 87, 4406–4410.
Jefferies, R. P. S. (1986) Ancestry of the Vertebrates (Cambridge Univ. Press, Cambridge).
Kappen, C., Schughart, K. & Ruddle, F. H. (1989) Two steps in the evolution of Antennapedia-class vertebrate homeobox genes. Proc. Natl. Acad. Sci. USA 86, 5459–5463.
Kenyon, C. & Wang, B. (1991) A cluster of Antennapedia-class homeobox genes in a nonsegmented animal. Science 253, 516–517.
Knoll, A. H. (1994) Neoproterozoic evolution and environmental change. In Early Life on Earth, ed. Bengtson, S. (Columbia Univ. Press, New York), pp. 439–449.
Krauss, S., Concordet, J.-P., & Ingham, P. W. (1993) A functionally conserved homolog of the Drosophia segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75, 1431–1444.
Lake, J. A. (1990) Origin of the multicellular animals. Proc. Natl. Acad. Sci. USA 87, 763–766.
McGinnis, W. (1985) Homeo box sequences of the Antennapedia class are conserved only in higher animal genomes. Cold Spring Harbor Symp. Quant. Biol. 50, 263–270.
McMenamin, M. (1993) Osmotrophy in fossil protoctists and early animals. Invertebr. Repro. Develop. 23, 165–166.
McShea, D. W. (1991) Complexity and evolution: what everybody knows. Biol. Philosph. 6, 303–324.
McShea, D. W. (1993) Evolutionary change in the morphological complexity of mammalian vertebral column. Evolution 47, 730–740.
Narbonne, G. M., Myrow, P., Landing, E. & Anderson, M. M. (1987) A candidate
stratotype for the Precambrian-Cambrian boundary, Fortune Head, Burin Peninsula, southeastern Newfoundland. Can. Earth Sci. 24, 1277–1293.
Nursall, J. R. (1962) On the origins of the major groups of animals. Evolution 16, 118–123.
Oliver, G., Vispo, M., Mailhos, A., Martinez, C., Sosa-Pineda, B., Fielitz, W. & Ehrlich, R. (1992) Homeoboxes in flatworms. Gene 121, 337–342.
Patel, N. H., Martin-Blanco, E., Coleman, K. G., Poole, S. J., Ellis, M. C., Kornberg, T. B. & Goodman, C. S. (1989) Expression of engrailed proteins in arthropods, annelids and chordates. Cell 58, 955–968.
Pendleton, J. W., Nagai, B. K., Murtha, M. T. & Ruddle, F. H. (1993) Expansion of the Hox gene family and the evolution of chordates. Proc. Natl. Acad. Sci. USA 90, 6300–6304.
Raff, R. A. (1994) Developmental mechanisms in the evolution of animal form: origins and evolvability of body plans. In Early Life on Earth , ed. Bengtson, S. (Columbia Univ. Press, New York), pp. 489–500.
Raff, R. A. & Kaufman, T. C. (1983) Embryos, Genes and Evolution (Macmillan, New York).
Ramsköld, L. & Hou, X.-G. (1991) New Early Cambrian animal and onychophoran affinities of enigmatic metazoans. Nature 351, 225–227.
Riddle, R. D., Johnson, R. L., Laufer, E. & Tabin, C. (1993) Sonic hedgehog mediates the polarizing activity of the ZPA. Cell 75, 1401–1416.
Ruppert, E. E. & Balser, E. J. (1986) Nephridia in the larvae of hemichordates and echinoderms. Biol. Bull. 171, 188–196.
Schierwater, B., Murtha, M., Dick, M., Ruddle, F. H. & Buss, L. W. (1991) Homeoboxes in cnidarians. Exper. Zool. 260, 413–416.
Schummer, M., Scheurlen, I., Schaller, C. & Falliot, B. (1992) HOM/Hox homeobox genes are present in hydra (Chlorohydra viridissima) and are differentially expressed during regeneration. EMBO 11, 1815–1823.
Seilacher, A. (1989) Vendozoa: organismic construction in the Proterozoic biosphere. Lethaia 22, 229–239.
Sepkoski, J. J., Jr. (1984) A kinetic model of Phanerozoic taxonomic diversity. III. Post-Paleozoic families and mass extinctions. Paleobiology 10, 246–267.
Sepkoski, J. J., Jr. (1992) Proterozoic–Early Cambrian diversification of metazoans and metaphytes. In The Proterozoic Biosphere, eds. Schopf, J. W. & Klein, C. (Cambridge Univ. Press, Cambridge, U.K.), pp. 553–564.
Sepkoski, J. J., Jr. (1993) Ten years in the library: new data confirm paleontological patterns. Paleobiology 19, 43–51.
Shashikant, C. S., Utset, M. F., Violette, S. M., Wise, T. L., Einat, P., Einat, M., Pendleton, J. W., Schugart, K. & Ruddle, F. H. (1991) Homeobox genes in mouse development. Crit. Rev. Eukaryotic Gene Expression 1, 207–245.
Signor, P. W. & Lipps, J. H. (1992) Origin and early radiation of the Metazoa. In Origin and Early Evolution of the Metazoa, eds. Lipps, J. H. & Signor, P. W. (Plenum, New York), pp. 3–23.
Sneath, P. H. A. (1964) Comparative biochemical genetics in bacterial taxonomy. In Taxonomic Biochemistry and Serology, ed. Leone, C. A. (Ronald, New York), pp. 565–583.
Turbeville, J. M. (1991) Nemertinea. In Microscopic Anatomy of Invertebrates: Platyhelminthes and Nemertinea, eds. Harrison, F. W. & Bogitsh, B. J. (Wiley-Liss, New York), Vol. 3, pp. 285–328.
Turbeville, J. M., Pfeifer, D. M., Field, K. G. & Raff, R. A. (1991) The phylogenetic
status of arthropods, as inferred from 18S rRNA sequences. Mol. Biol. Evol. 8, 669–686.
Turbeville, J. M., Field, K. G. & Raff, R. A. (1992) Phylogenetic position of Phylum Nemertini, inferred from 18S rRNA sequences: Molecular data as a test of morphological character homology. Mol. Biol. Evol. 9, 235–249.
Valentine, J. W. (1969) Patterns of taxonomic and ecological structure of the shelf benthos during Phanerozoic time. Palaeontology 12, 684–709.
Valentine, J. W. (1989) Bilaterians of the Precambrian-Cambrian transition and the annelid-arthopod relationship. Proc. Natl. Acad. Sci. USA 86, 2272–2275.
Valentine, J. W. (1990) Molecules and the early fossil record. Paleobiology 16, 94–95.
Valentine, J. W. (1992) The macroevolution of phyla. In Origin and Early Evolution of the Metazoa, eds. Lipps, J. H. & Signor, P. W. (Plenum, New York), pp. 525–553.
Valentine, J. W. (1994) The Cambrian explosion. In Early Life on Earth, ed. Bengtson, S. (Columbia Univ. Press, New York), pp. 401–411.
Valentine, J. W. & Campbell, C. A. (1975) Genetic regulation and the fossil record. Am. Sci. 63, 673–680.
Valentine, J. W., Collins, A. G. & Meyer, P. C. (1994) Morphological complexity increase in metazoans. Paleobiology 20, 131–142.
Valentine, J. W., Awramik, S. M., Signor, P. W. & Sadler, P. M. (1991) The biological explosion at the Precambrian-Cambrian boundary. Evol. Biol. 25, 279–356.
Valentine, J. W., Tiffney, B. & Sepkoski, J. J., Jr. (1991) Evolutionary dynamics of plants and animals: a comparative approach. Palaios 6, 81–88.
Wainright, P. O., Hinkle, G., Sogin, M. L. & Stickel, S. K. (1993) Monophyletic origins of the Metazoa: an evolutionary link with fungi. Science 260, 340–342.
Webster, P. J. & Mansour, T. E. (1992) Conserved classes of homeodomains in Schistosoma mansoni, an early bilateral metazoan. Mech. Dev. 38, 25–32.
Wheeler, W. C., Cartwright, P. & Hayashi, C. Y. (1993) Arthropod phylogeny: a combined approach. Cladistics 9, 1–39.
Whittington, H. B. (1985) The Burgess Shale (Yale Univ. Press, New Haven, CT).
Willmer, P. (1990) Invertebrate Relationships (Cambridge Univ. Press, Cambridge, U.K.).
Wright, S. (1949) Adaptation and selection. In Genetics, Paleontology and Evolution, eds. Jepson, G. L., Simpson, G. G. & Mayr, E. (Princeton Univ. Press, Princeton, NJ), pp. 365–389.
| This page in the original is blank. |






















