Continuous Learning and Improvement in Health Care
KEY SPEAKER POINTS
- Michael McGinnis noted that, when asked to reflect on their experiences, virtually all successful corporate CEOs in the country would speak to the centrality of continuous learning to their effectiveness and efficiency—even to their survival.
- “We are aware that among the needs that our systems have from research at this point are speed, relevance, precision, and methods that really fit the kinds of questions, decisions, and actions that are needed by us as delivery system leaders, as patients, and as clinicians,” said Raymond Baxter.
- Baxter also noted that PCORnet’s success will not only accelerate the pace of change, but it will also change the culture of research for the better by bringing patients, clinicians, and delivery system leaders into the research enterprise.
- Rainu Kaushal said that she applauds the foresight of large health care system CEOs who, despite the fragmentation and competitive pressures, see that sharing data is the right thing to do for their health systems, for clinical care, and for populations.
- Kaushal said, too, that there is a tension between research interests and organizational priorities and that she believes that the key to resolving those tensions rests with good communication and alignment of priorities.
As was noted in the previous chapter, the vision of the Roundtable on Value & Science-Driven Health Care—which is in line with the goals of PCORI—is that the nation will develop a continuously learning health system in which science, informatics, incentives, and culture are aligned for continuous improvement and innovation. In a session designed to serve as a backdrop for further discussion, Michael McGinnis, senior scholar at the IOM, provided a brief introduction to the concepts of a learning health system, and Joe Selby, PCORI’s executive director, gave an overview of PCORnet. The workshop also heard presentations describing two of the CDRNs that are participating in PCORnet. Raymond Baxter, senior vice president for community benefit research and health policy at Kaiser Permanente, and Elizabeth McGlynn, director of Kaiser Permanente’s Center for Effectiveness and Safety Research, spoke about the Patient Outcomes Research to Advance Learning (PORTAL) Network, while Rainu Kaushal, Chair of Healthcare Policy and Research at Weill Cornell Medical College, discussed the New York City Clinical Data Research Network (CDRN). Afterward Eric Larson, vice president for research at Group Health, executive director of the Group Health Research Institute, and the workshop planning committee chair, moderated an open discussion.
To start his short overview of the IOM and work that has been done to date to conceptualize and advance a continuously learning health care system, McGinnis recounted a discussion that the workshop planning committee had had concerning whom it might ask to provide a perspective on how other sectors view a learning system in comparison with how the health care system operates today. He suggested, for example, that Jeff Bezos from Amazon might reflect on what his company’s situation might be if it captured as little of its customers’ experience—and of its own experience—as is often the case in health care. Or Mary Barra, the new CEO of General Motors, could have something to say about the importance in a competitive market of investing in an employee culture of continuous quality and the consequences when that does not happen. Michael Huerta, the administrator of the Federal Aviation Administration, could reflect on what airline safety might look like if the airline industry had the level of nonstandard work procedures in air safety that often exists in health care. William Dudley, CEO of the construction giant Bechtel, could comment on how well his company would operate if the plumbers, architects, carpenters, and electricians were all working from different blueprints. And Dee Hock, founder and former CEO of the Visa Credit Card Association, could reflect on the likely condition of the credit card industry if it suffered the same scale of electronic disconnect as currently exists in health care.
Virtually all successful corporate CEOs in the country, McGinnis said, would speak to the centrality of continuous learning to their effectiveness and efficiency—even to their survival. With this in mind, McGinnis said he would provide an overview of the possibilities and concepts for continuously learning health care and ask the workshop participants to imagine the kinds of changes that can occur in a truly transformed health system.
The perspectives that the above-mentioned CEOs might have provided were noted in the opening pages of the 2012 IOM report Best Care at Lower Cost: The Path to Continuously Learning Health Care in America, which, McGinnis said, “looked very carefully at the state of play with respect to quality, cost, science, technology, and culture in health care and said, in effect, we’re well past the time when health care should be lagging so far behind the best practices in other sectors and even in our own sector.” McGinnis added, “We can do much better in terms of delivering the efficiency and effectiveness that the American people deserve.” According to McGinnis, the committee that authored the report was saying that the health care system needs to transition away from a linear system in which learning opportunities are substantively lost (see Figure 2-1) to one characterized by a continuous feedback loop in which science yields evidence that is then applied to care and that care experience is captured to generate new knowledge.
Today, however, McGinnis noted, the translation from evidence to care is often lacking with respect to the extent of its application, and the care experience itself is, for the most part, poorly captured, which limits the opportunity for feedback that could further inform and improve care. What the nation needs instead is to bring the practice and culture of science into the networked age and create a virtuous cycle of learning, one that
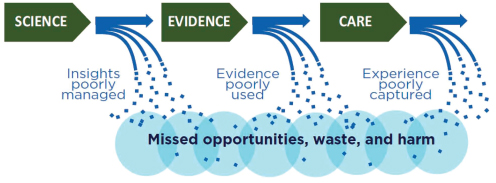
FIGURE 2-1 Learning opportunities are lost in today’s linear system.
SOURCE: IOM, 2012.
aligns science, informatics, incentives, and culture to produce continuous improvement and innovation, and one that embeds best practices seamlessly into the delivery process and that captures new knowledge as an integral by-product of the delivery experience. McGinnis said that many of the workshop participants could testify to the value of creating such a system.
McGinnis also reminded the workshop participants that the Best Care at Lower Cost report had been revisiting the ground laid by two earlier IOM reports: To Err Is Human: Building a Safer Health System and Crossing the Quality Chasm: A New Health System for the 21st Century, which were issued in 1999 and 2001, respectively. The 2012 report, McGinnis said, provided an update on these two earlier reports in the context of the increasing complexity and cost of health care and also in the context of the new tools and levers that had become available for continuous quality improvement, through the provisions of the Health Information Technology for Economic and Clinical Health Act and the Affordable Care Act, as well as in the context of the overall increase in learning capacity stemming from advances in research methods and in the nation’s digital infrastructure. The 2012 report called for ramping up real-world and real-time learning strategies that employ data science to engage both structured and spontaneous learning. The report noted that work that is patient participatory in nature is an untapped resource for the learning process. The goal should be to create a learning infrastructure that is research-ready, multiuse, focused on both quality and knowledge generation, and patient accessible.
McGinnis mentioned, too, that the context of the 2012 report also reflected the 15 reports that the Roundtable had developed over the previous 7 years. These reports explore various elements of the multiple issues involved in creating a continuously learning health care system, such as effectiveness research, the implications of the growing complexity of care, the value proposition, and the digital platform needed for a learning health system. McGinnis also noted the work that the IOM has done with the Office of the National Coordinator for Health Information Technology in laying out the vision for the digital platform for continuous learning in health and health care (see Figure 2-2).
McGinnis then noted several assumptions underlying the structure of the workshops. The first assumption was that the workshop participants did not need to be convinced about the value of a learning health care system; these are CEOs who are leading the field on behalf of continuous learning. The second assumption was that developing a learning health system is fundamentally not a technical problem. Yes, there are technical complexities, McGinnis acknowledged, but he said that experts in the software and hardware development arenas do not see these complexities as fundamentally impossible technical challenges. The third assumption was that culture need not be an obstacle, given that patients understand the
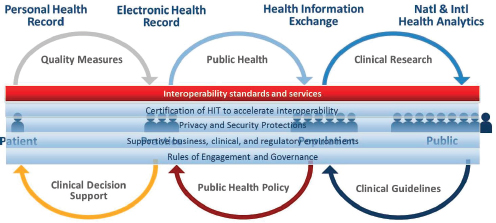
FIGURE 2-2 The Office of the National Coordinator’s vision of a learning health system.
NOTE: HIT = health information technology.
SOURCE: Reprinted with permission from Douglas Fridsma.
value of research, that clinicians believe that knowledge generates pride, and that CEOs want a culture that creates value at speed and allows them to stay ahead of the curve. The fourth assumption was that this is not an economic problem at heart, given the sizable investment in health information technologies, which is estimated to be some $34.5 billion in 2014. The fifth and final assumption was that this is not a political problem: Democrats and Republicans agree on the importance of developing an evidence-based learning health care system. What this is, McGinnis said in closing, “is fundamentally a commons problem,” or one in which the behavior of individual actors is contrary to the best interest of the group.
As a preamble to his remarks about PCORnet, Joe Selby commented that in his experience, sometimes randomized interventions are the only way to learn, with enough certainty to justify implementation, whether something works better than an alternative. However, he said there are concerns within the community of people working toward the development of a learning health care system that, on the one hand, many interesting questions concerning effectiveness are not amenable to randomization, and on the other hand, observational study designs are limited and can be biased. Additionally, there is also some concern that joining a network that includes multiple institutions or a network of networks such as PCORnet will retard
rather than accelerate local progress. In addressing those concerns, Selby said that networks such as the CDRNs and PCORnet will enable observational studies that “look for all the world like randomized trials,” as well as drive innovation in the use of randomized approaches. One of the keys features of PCORnet, he said, is that consolidating data from multiple systems and multiple clinical units within those systems provides enough variation in practice to provide statistical power and rigor and to reduce the chances of bias. “So I would say one advantage of PCORnet as it matures could in fact be to have a broader range of variation in practice that could be studied,” he said.
The prevailing view in Washington, Selby said, is that the nation’s clinical research system is in trouble—that it is well-intentioned but flawed. According to this view, the current system is not generating the evidence needed to support most clinical decisions, there are health outcomes and disparities that are not by and large improving, and the current clinical research system is too slow and too expensive and it does not answer the questions that worry most patients, clinicians, and health system leaders. PCORnet envisions a “community of research” that addresses these concerns by uniting systems, patients, clinicians, and research in a national infrastructure for patient-centered clinical research. Toward that end, PCORnet has created a network of networks comprising 11 CDRNs, clinical data networks spanning at least two health systems, and 18 PPRNs, disease-specific networks focused on the collection and use of patient-generated data, that are distributed across all 50 states and the District of Columbia (see Figure 2-3). Selby said that he sees the patient-powered networks as “the really radical portion of PCORnet, those groups of activated patients who are charged to grow and to interact with the CDRNs.”
The overall goal of PCORnet, Selby explained, is to achieve a single functional research network through the following actions:
- Create a secure national research resource that will enable teams of health researchers, patients, and their partners to work together on studying questions of shared interest.
- Utilize multiple rich data sources, such as EHRs, insurance claims data, and data reported directly by patients, to support research.
- Engage patients, clinicians, and health system leaders throughout the research cycle from idea generation to implementation.
- Support observational and interventional research studies that compare how well different treatment options work for different people.
- Enhance the use of clinical data to advance the learning health care system.
- Enable external partners to collaborate with PCORI-funded networks.
- Sustain PCORnet resources for a range of research activities supported by PCORI and other sponsors.
A steering committee consisting of a representative of each of the CDRNs and PPRNs along with patient advocates and representatives of medical product and device manufacturers, various agencies in the U.S. Department of Health and Human Services, and PCORI oversees a coordinating center that manages 11 task forces, each of which in turn oversees a mission-critical activity (see Figure 2-4). PCORnet, Selby said, needs to phase in those 11 activities over the 15 months remaining in Phase 1 of this initiative. He said, too, that agencies such as the NIH, the U.S. Food and Drug Administration (FDA), the Agency for Healthcare Research and Quality (AHRQ), and the Centers for Disease Control and Prevention (CDC) as well as the pharmaceutical and devices industries could prove to be interesting partners for and funders of future research. “This research would bring together delivery systems, payers, the research community, the
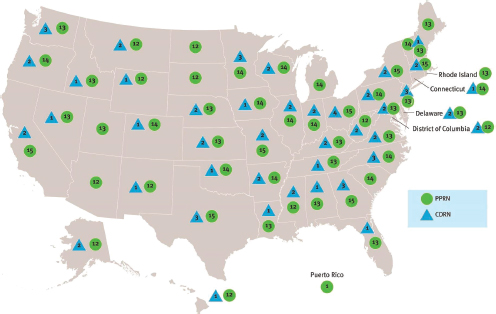
FIGURE 2-3 Geographic coverage of the PPRNs and CDRNs that have joined PCORnet.
SOURCE: Reprinted with permission from the Patient-Centered Outcomes Research Institute.
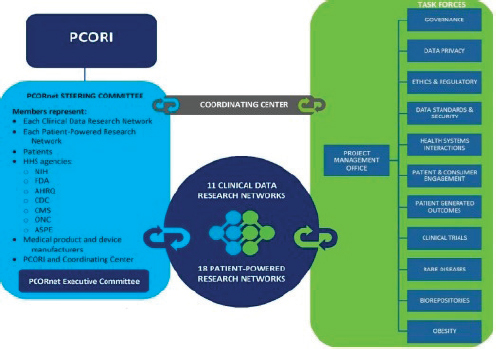
FIGURE 2-4 PCORnet’s operational structure.
SOURCE: Reprinted with permission from the Patient-Centered Outcomes Research Institute.
sponsors of the research, and patients to fund research that is of interest to all groups,” Selby said. “So we have a hope that we will find research being funded through PCORnet that brings these stakeholders in the enterprise together at the beginning, not at the end of the research,” when someone may bring in a result that could upset the system.
In reviewing the activities of the 11 issue-focused task forces that form part of the PCORnet coordinating center, Selby explained that the governance task force is concerned with issues such as who owns the data collected by the members of the networks, who can use the data, who can access the data, what is required to be a member of PCORnet, and what the expectations are regarding members securing additional funding to support PCORnet activities. Privacy is a critical activity, as it could prove to be either PCORnet’s Achilles’ heel or, as Selby put it, “a real triumph if we are able to bring patients, patient organizations, and delivery systems together with ethicists to discuss the benefits and the potential risks of big data and using big data to address questions of importance to patients.”
The ethics and regulatory oversight task force is looking at issues concerning informed consent and how to balance the concerns of institutional
review boards (IRBs) and the need to streamline and accelerate research. The data standards task force is not concerned with establishing a new set of standards but is looking at ways of encouraging electronic health record vendors to make greater and more consistent use of the nationally agreed-upon standards that already exist. The task force on health systems interactions is working to maintain, strengthen, and nurture PCORnet’s relationships with health care delivery systems, while the patient and consumer engagement task force is doing the same for patient groups. The patient-generated outcomes task force is working from the premise that getting patient-generated outcomes is another area that could greatly enhance clinical care and could also enhance clinical and outcomes research, Selby said.
Regarding clinical trials, Selby said that PCORnet’s philosophy is that they will be done only when needed. Occasionally, he said, it will be necessary to run small, randomized trials to find small differences in effectiveness. “I’d totally agree though that most of the time you can’t do randomization and you’d prefer not to,” he said. Rare diseases will be something that PCORnet tackles because the network will have data from more than 25 million people, and, in fact, the legislation that created PCORI specifically charges the institute with studying rare diseases. The goal for the biorepository task force is to work to strengthen the biorepositories associated with the CDRNs and PPRNs and to bring them into clinical outcomes research.
Shelby explained that the CDRNs (see Table 2-1) include a number
TABLE 2-1 The 11 Clinical Data Research Networks
| CDRN Name | Lead Organization | Principal Investigator |
| ADVANCE | Oregon Community Health Information Network | Jennifer DeVoe |
| CAPriCORN | The Chicago Community Trust | Terry Mazany |
| Great Plains Collaborative | University of Kansas Medical Center | Lemuel Waitman |
| Louisiana Clinical Data Research Network | Louisiana Public Health Institute | Thomas Carton |
| Mid-South CDRN | Vanderbilt University | Russell Rothman |
| NYC-CDRN | Weill Medical College of Cornell University | Rainu Kaushal |
| PEDSNet | The Children’s Hospital of Philadelphia | Christopher Forrest |
| PORTAL | Kaiser Fiundation Research Institute | Elizabeth McGlynn |
| pSCANNER | University of California, San Diego | Lucila Ohno-Machado |
| P2ATH | University of Pittsburgh | Rachel Hess |
| SCIHLS | Harvard University | Kenneth Mandl |
SOURCE: Reprinted with permission from the Patient-Centered Outcomes Research Institute.
of networks of two or more health care systems, networks of nonprofit integrated health systems, networks of federally qualified health centers, and networks that have leveraged previous NIH and AHRQ investments. At least 54 of the 61 Clinical and Translational Science Award winners are involved in one of PCORnet’s 11 CDRNs, and many of them include large populations of underserved individuals. The Louisiana CDRN, Selby said, is built substantially on the state’s health information exchange. Among the PPRNs in PCORnet (see Tables 2-2 and 2-3), whose focus is the collection and use of patient-generated information, about half are devoted to rare diseases.
Selby concluded his introduction to PCORnet by recapping some early observations. “We are convinced that we’ve got to establish priorities that patients, clinicians, health systems, payers, manufacturers—as well as researchers—share and consider important,” he said. “We think if we can find that sweet spot that the findings are much more likely to be listened to, taken up, and implemented and to change practice.” PCORnet has a great deal of work to do, much of it around governance and issues about data ownership and privacy, to facilitate the trust that will support collaborations between networks.
Selby said that it is going to be critical to embed research, including randomized research, as intimately as possible within the practice setting
TABLE 2-2 The Patient-Powered Research Networks Devoted to Common Diseases
| Organization | PI | Condition | Population Size |
| Accelerated Cure Project for Multiple Sclerosis | Robert McBurney | Multiple sclerosis | 20,000 |
| American Sleep Apnea Association | Susan Redline | Sleep apnea | 50,000 |
| Cincinnati Children’s Hospital Medical Center | Peter Margolis | Pediatric Crohn’s disease and ulcerative colitis | 15,000 |
| COPD Foundation | Richard Mularski | Chronic obstructive pulmonary disease | 50,000 |
| Crohn’s and Colitis Foundation of America | R. Balfour Sartor | Inflammatory bowel disease (Crohn’s disease and ulcerative colitis) | 30,000 |
| Global Healthy Living Foundation | Seth Ginsberg | Arthritis (rheumatoid arthritis; spondyloarthritis), musculoskeletal disorders (osteoporosis), and inflammatory conditions (psoriasis) | 50,000 |
| Massachusetts General Hospital | Andrew Nierenberg | Major depressive disorder and bipolar disorder | 50,000 |
| University of California, San Francisco | Mark Pletcher | Cardiovascular health | 100,000 |
| University of South Florida | Rebecca Sutphen | Hereditary breast and ovarian cancer | 17,000 |
NOTE: PI = primary investigator.
SOURCE: Reprinted with permission from the Patient-Centered Outcomes Research Institute.
TABLE 2-3 The Patient-Powered Research Networks Devoted to Rare Diseases
| Organization | PI | Condition | Population Size |
| ALD Connect, Inc. | Florian Eichler | Adrenoleukodystrophy | 3,000 |
| Arbor Research Collaborative for Health | Bruce Robinson | Primary nephrotic syndrome; focal segmental glomerulosclerosis; minimal change disease; and membranous nephropathy multiple sclerosis | 1,250 |
| Duke University | Laura Schanberg | Juvenile rheumatic disease | 9,000 |
| Epilepsy Foundation | Janice Beulow | Aicardi syndrome; Lennox-Gastaut syndrome; Phelan-McDermid syndrome; hypothalamic hamartoma; Dravet syndrome; tuberous sclerosis | 1,500 |
| Genetic Alliance, Inc. | Sharon Terry | Alström syndrome; dyskeratosis congenital; Gaucher disease; hepatitis; inflammatory breast cancer; Joubert syndrome; Klinefelter syndrome and associated conditions; psoriasis; metachromatic leukodystrophy; pseudoxanthoma elasticum | 50 to 50,000 |
| Immune Deficiency Foundation | Kathleen Sullivan | Primary immunodeficiency diseases | 1,250 |
| Parent Project Muscular Dystrophy | Holly Peay | Duchenne and Becker muscular dystrophy | 4,000 |
| Phelan-McDermid Syndrome Foundation | Megan O’Boyle | Phelan-McDermid syndrome | 737 |
| University of Pennsylvania | Peter Merkel | Vasculitis | 500 |
SOURCE: Reprinted with permission from the Patient-Centered Outcomes Research Institute.
without disrupting clinical operations. In order to achieve this, he said that it will be crucial to develop ethical oversight procedures and human subject procedures that protect patients while minimizing redundancy, wasted effort, and the barriers that prevent participation. It is also apparent that PCORnet will have to engage individuals and organizations beyond the initial awardees, and, toward that end, Phase 2 of the effort will open the network to broader participation. Finally, Selby said, PCORnet must strive for simplicity in a very complex and multilayered environment.
PATIENT OUTCOMES RESEARCH TO ADVANCE LEARNING (PORTAL) NETWORK
In the first of two presentations highlighting specific PCORNet CDRNs, Raymond Baxter and Elizabeth McGlynn described the PORTAL network developed by Kaiser Permanente and its principal partners Group Health Cooperative, HealthPartners, and Denver Health. These organizations, Baxter said, share the aim of being true learning health care organizations and see research as a critical part of the strategy to realize that aim rather than merely an interesting sideline activity. All four organizations are also
grappling not only with the transformation of care but also with the transformation of research to serve that transformation of care, he added. “We are aware that among the needs that our systems have from research at this point are speed, relevance, precision, and methods that really fit the kinds of questions, decisions, and actions that are needed by us as delivery system leaders, as patients, and as clinicians,” Baxter said. “At least for Kaiser Permanente, that translates into a sense that we now see the need for a broad continuum of research and analytic capabilities in the organization, in order to answer the critical questions that we have.” Among the other members of the PORTAL CDRN are three patient groups—the Adult Congenital Heart Association, Fight Colorectal Cancer, and Smart Patients—that Baxter said play a critical role in driving the activities of the PORTAL network.
PORTAL members see their network as being able to contribute quickly to PCORnet, thanks to the internal resources that PORTAL puts at their disposal, the connection between their delivery systems, and their research and analytic activities. They also see that belonging to PCORnet will help with the work that they each are doing in their individual organizations. “PCORnet represents a model and a discipline that we think can help us push faster the changes we need to make within our own systems in terms of the relationships between research and analytics and care delivery change,” Baxter explained. “It also offers us a network that gives us the opportunity not only to share what we are learning, not only to share our data and our capabilities, but to understand what we think we have been learning is actually generalizable in a broader way to the field. We also see PCORnet as something that will be self-reinforcing for us, if it is successful.”
In concluding his comments, Baxter said that if PORTAL can contribute to PCORnet and if PORTAL’s members are able to integrate findings from PCORnet into their own care delivery systems, that would serve as a proof point not only for the importance of research but also for the importance of engaging with others in a collaborative manner to speed the development and adoption of knowledge on a broad scale. In his opinion, he said, PCORnet’s success will not only accelerate the pace of change, but it will also change the culture of research for the better by bringing patients, clinicians, and delivery system leaders into the research enterprise.
McGlynn then described some of PORTAL’s accomplishments over its first 3 months of operation. Though PORTAL is relatively new, this is not Kaiser Permanente’s first experience in working within the structure of a network (see Figure 2-5). In 1994, she said, her organization joined the HMO Research Network, which demonstrated the importance of developing a common data model and strong governance provisions. “What we have learned and honed and perfected is the ability to write code in one place and distribute it and run it in lots of different places with really minor

FIGURE 2-5 Kaiser Permanente’s experience in working with data networks.
SOURCE: Reprinted with permission from Ray Baxter and Elizabeth McGlynn.
tweaks,” she said. “The opportunity to answer more questions and move more quickly is really facilitated by this common data model.” She added that given the size, complexity, and multitude of stakeholders involved, PCORnet has the components of a great social experiment, noting that, as such, strong governance will be critical for its success.
From its experience as part of the CDC’s Vaccine Safety Datalink and the FDA’s Mini-Sentinel networks, Kaiser Permanente learned the value of listening to its customers and how to do surveillance more efficiently with standardized methods. Through its interactions with these two networks, McGlynn said, she and her colleagues also learned the value of not reinventing the wheel as it joined each of these networks. These lessons informed Kaiser Permanente’s investment in its Center for Effectiveness and Safety Research, which the organization established in 2009 to answer delivery system questions and to help stabilize the organization’s data infrastructure so that it could answer a wide range of questions without having to rework the organization’s central data model for every new research project. PCORnet is the “roof on the house that we are building,” McGlynn said. “For us, the real opportunity here is learning how to more effectively engage patients and system leaders in the conversation in the development of questions.”
Stakeholders are at the heart of PORTAL’s organizational structure, McGlynn said. A patient engagement council includes representatives from the three patient groups that are PORTAL members as well as individual patients from the four member health systems. In addition, PORTAL is developing a large online community using the Smart Patients online platform, which allows the network to invite everyone who would be eligible from one of its cohorts to participate in an online community conversation with an even broader reach than its patient engagement council can provide. PORTAL is also working through its community benefit group to reach out to community-based organizations, which will enable it to have conversations with stakeholders who, as McGlynn said, “may not be as online savvy and that represent some of the underserved populations that we don’t always hear from.”
PORTAL’s clinician engagement council aims to broadly engage physicians in the conversation and to help determine the best ways to integrate research and research findings with the care delivery system. An operational engagement council brings together the people whom McGlynn described as the ones who have to get the work done: the information technology groups, the compliance groups, and those who have to manage workflows within these large health care systems. “The idea here is that we are building infrastructure that lets us develop systems, so that we are not making these things up each and every time we set out to do a research project,” she said. “We are exercising them with the PCORnet dollars to find out how to make them work and then improve them as we go along.” Toward that end, she said, “I think that having this kind of engagement with our critical stakeholders really at the center of what we are doing is going to be critical.”
PORTAL’s contract with PCORnet calls for it to focus its efforts on three specific groups of its 11 million patients across its delivery systems: colorectal cancer patients, with an emphasis on treatment and how patients navigate survivorship; adolescents and adults with severe congenital heart disease, a rare condition that affects about 330 individuals across the network, with an emphasis on transitions in care as these patients not only live to be adults, but thrive; and obesity in adults. Together, these three projects aim to enhance the PCORnet health systems’ ability to embed research in routine care delivery, and McGlynn listed several factors that she thinks will be key to the success of this effort. The first is the ability to ask the right questions—in particular, questions identified by the people who will be implementing the resulting answers. The second is engaging clinical champions who can help advocate for some of the changes that will have to occur to make research possible within the routine care delivery system. The third is engaging the operational teams that will do the work and make these projects happen so that, McGlynn said, “as we develop systems for
care delivery, we are thinking about how those can be used for research, as opposed to having separate and parallel systems.”
The goal for each of the three projects is to produce information that will help patients and doctors make decisions together in real time. McGlynn said that the ideal would be to integrate information from research into care delivery at the point of care in a way that makes it easy for doctors and patients alike to use that information. McGlynn concluded her remarks by noting that the tools and technology pieces are largely in place and that the challenge now is getting people to collaborate in the service of answering critical questions for patients, the health care system, and the nation.
NEW YORK CITY CLINICAL DATA RESEARCH NETWORK
In providing background information for her presentation on the New York City CDRN, Rainu Kaushal said that while there are 8 million residents in New York City, the number of people who come to the city for clinical care is easily double the permanent population. There are six academic medical centers in the city, and in any given year some 40 percent of one center’s inpatients will either use an emergency department or be hospitalized at another academic medical center that is also in New York City. “When you start thinking about the degree of fragmentation, including the ambulatory side of things,” she said, “it is not unusual for a Medicare beneficiary in New York City, for example, to be seeing 10 or 12 different providers. That amount of fragmentation and the very tight operating margins that our health systems are under causes tremendous complexities for providing the best possible clinical care and getting the right treatment to the right person at the right time. It makes population health management exceedingly challenging.”
That kind of fragmentation also creates significant challenges for sharing data among multiple institutions and for creating a learning health system, Kaushal said. As a result, she explained, when the opportunity arose to build the infrastructure to support clinical research and care and to manage health on a population basis, the 22 partners in the New York City CDRN jumped at the chance to participate, she said. The partners in this CDRN include six academic medical centers, one practice-based research network, six consumer partners, and six research infrastructure organizations, including the New York Genome Center, several information exchanges, and Cornell’s new technology campus. Kaushal applauded the foresight being shown by the CEOs of the large health care systems who, despite the fragmentation and competitive pressures, see that sharing data is the right thing to do for their health systems, for clinical care, and for populations.
The New York City CDRN has four goals, Kaushal said. The first is
ensuring security and privacy. “Because of the fragmentation in our market, we are forced to centralize our data in order to perform de-duplication and then de-identification,” she said. “In that context, privacy and security are our guardrails, and we are putting an incredible amount of energy right now into figuring out those guardrails and making them as secure as we possibly can.” The second goal is to build a research infrastructure populated by complete comprehensive longitudinal data on a minimum of 1 million patients, although, given the populations of New York City, she said she expects that number will be higher by the end of the project. Kaushal said that the plan is to incorporate clinical data, claims data, patient-reported survey data, biospecimen data, and patient-generated data into the database. The project’s third goal is to integrate the viewpoints of patients and practicing clinicians.
All of this activity has the express goal of embedding research into health care delivery so that a physician can provide the appropriate care to meet each patient’s individual needs. Kaushal noted the tension between the research interests and organizational priorities and said she believes that the key to resolving those tensions rests with good communication. She also said that the New York City CDRN is putting a great deal of time into patient consent and is borrowing and sharing from the best practices across the network in order to streamline the process and make research more effective across the network.
Kaushal spoke about the value that she believes the network brings to the health systems that belong to the New York City CDRN. First and foremost is the ability to generate actionable, patient-centered knowledge that improves the delivery of health care and the conduct of research. Another contribution will lie in assuring quality and safety when quality databases are integrated with clinical and claims databases. Enabling population management in the New York City area will also be a huge contribution, she said. “If we could really understand our attributed Medicare beneficiaries, what their costs are, how their costs are accruing across our health care systems, and what the quality of care that we, as a system, are providing to them, that would be huge,” she said. Another contribution will be working toward creating a citywide learning health care system.
The New York City CDRN has enjoyed tremendous support from senior leadership, Kaushal said, and partner institutions have been generous in terms of providing infrastructure and other resources. She concluded her presentation by showing a picture of her 19-month-old twins and saying that they are the reason she is involved in this project. “I feel an imperative to work on this network and to be part of PCORnet,” she said, “because when I take them to a pediatrician, I want them to get antibiotics because they actually need them for an infection, or if they need a tympanostomy tube, I want that to be placed because it is surgically indicated. Those are
the types of studies that, given the scope of PCORnet, could be answered with trivial amounts of energy. I want to see those things.”
In opening the discussion period, Selby said that the other nine CDRNs that are part of PCORnet could have told equally compelling stories. He also said that the original intention was to fund eight CDRNs but that the decision was made to fund 11 because the applications were so strong.
Russell Rothman of Vanderbilt University, who heads the Mid-South CDRN, said that the two preceding presentations had embodied what each of the CDRNs are doing, and he added that at this point in the PCORnet grant cycle, much of what is happening at his and other CDRNs is infrastructure development and stakeholder engagement, a point with which Selby agreed. “I just want to emphasize the point that this is a work in progress,” Rothman said. “We very much want as much input as we can at this point in the process.” Rothman also said that there is a grand opportunity in bringing together 11 CDRNs and 18 PPRNs under the PCORnet umbrella to collaborate and ask questions at a level that cannot be done by any single CDRN. This is particularly true for rare diseases, he said. Baxter said that another benefit of belonging to PCORnet has been that it forced him and his colleagues to make decisions and set up a structure quickly rather than debating exactly what to do for the next several years. “That is a very tangible benefit for us as a system,” he said.
Steven Lipstein of BJC Health care asked Kaushal how the New York City CDRN handles IRB approvals involving multiple institutions in the network and how it handles patient recruitment, given that there may be competition among institutions and clinical trials. The New York City CDRN, Kaushal replied, has a centralized IRB that three of the member institutions had already been sharing for a decade and that all of the other partners have agreed to use. Concerning patient recruitment, she said that she and her colleagues have been thinking about this because they want to avoid multiple contacts of a single patient for research studies. Their solution, she said, has been to develop a centralized de-identified database and “use attribution logic to attribute each patient as a primary for a given health system.”
Jeffrey Grossman from the University of Wisconsin Medical Foundation asked if PCORI had a vision for studying the outcomes of its research. Selby replied, “PCORI has a real sense of urgency about evaluating the impact of its research. We have a date with the [Government Accounting Office] in 2018 where we are really going to have to have some of that evidence for Congress to look at. We have an evaluation plan that specifi-
cally looks at very critically [whether] all of this engagement [is] making any difference.”
Jonathan Tobin of the Clinical Directors Network and the Rockefeller University Center for Clinical and Translational Science asked if there was an opportunity to use PCORnet to phenotype organizations in the same way that patients are phenotyped. In other words, would it be possible to look at different delivery care models, different types of clinician groups, and other system variables and look at the associations between those different models and the participants in the system and answer questions about uptake, implementation, effectiveness, and outcome? Selby answered that that type of question was something that PCORnet could address.
Petra Kaufmann from NIH said that the National Center of Advancing Translational Sciences, where she works, recently received an IOM report reviewing the Clinical and Translational Science Awards program, and one of its recommendations was to strengthen the capacity of the program to function as a network and, in particular, to leverage existing information technology systems. A key to doing that, she said, will be to ensure that the data systems of its grantees are interoperable with those of the PCORnet members. Toward that end, she said, her center will work closely with PCORI to make sure that there is synergy between the two.
Robert Kaplan from AHRQ asked if PCORnet is giving any thought to how to recognize scholars that are now going to be working as part of large networks and to how that recognition might impact career choices. Selby said that younger researchers are excited about the opportunities that come from working within a large network and about the access to large amounts of data that provides. Young researchers, he said, want to be part of an effort in which their research can be embedded in the care environment, and he predicted that “we will see M.D. and Ph.D. trained researchers working from bases within systems to conduct research that shows up in the New England Journal of Medicine.” McGinnis added that there is a need for a different type of researcher, one who is comfortable talking to leadership and to researchers from outside of their disciplines.
Continuing on that theme, John Gallin of the NIH Clinical Center asked if as much thought has gone into the training of tomorrow’s physicians to interact with this type of system as has gone into what information should be recorded in a patient’s medical record. He also asked how to engage patients so that they not only expect to receive something from the system but contribute to it as well. Selby replied that one of the underlying principles for PCORnet is that there has to be something in it for everybody. “If clinicians are really engaged in using these data to improve care,” he said, “I think it’s a natural consequence that they will begin to take more responsibility and care in putting the data in. If they can see when bad data lets you down and when good data enlightens you, they will take more
care when they feel that they can use these data ultimately to ask questions on their minds. I think the same thing goes for patients. You can talk to patients about their responsibilities vis-à-vis EHRs and PCORnet once you’ve given them a seat at the table and once they feel that this is their enterprise as well.”
Darrell Kirch of the Association of American Medical Colleges said that the challenge for trainees is that they often have to deal with multiple medical record systems as they move through different rotations during their years of training. He also said that medical education needs to change to reflect the fact that EHRs will have uses far beyond billing. “We have a long way to go to create the infrastructure and the attitudes so that the students and residents, who are very willing to use the electronic record, can shift their focus away from reimbursement to creating the research database,” he said.
This page intentionally left blank.




















