7
Toward a High-Quality Clinical Eye and Vision Service Delivery System
Encouraging high-quality eye and vision care is one component of a comprehensive population health approach to reduce vision impairment in the United States. As defined by the Institute of Medicine (IOM), quality care must be safe, timely, effective, efficient, equitable, and patient-centered (IOM, 2001). Currently, the clinical eye and vision service delivery system faces several challenges to achieving these goals. Multiple and sometimes conflicting clinical practice guidelines create different standards from which to measure and improve quality and clear, consistent public messaging about what care is needed when. Limited integration among and between clinical and public health services, combined with insufficient cross-disciplinary training of the workforce, may negatively affect diagnosis and follow-up care. Inadequate health care services research on the vision care system further hampers the ability to improve care quality through application of continuous quality improvement programs.
This chapter focuses on improving the quality and consistency of eye and vision care in the United States. The first section discusses the importance of consistent evidence-based guidelines to inform care seeking and providing behaviors, especially in the context of vision screenings and comprehensive eye examinations. The second section examines the role of continuous quality improvement initiatives in promoting high-quality eye and vision care. The third section explores potential integrated models of care to promote detection and diagnosis of vision problems and subsequent referral to eye care providers. The state of, and need for, cross-disciplinary education of the public health, eye care, and broader clinical workforce is described in the final section, with a focus on training to promote cultural
competency, leadership, teamwork, and awareness of the interrelations between eye and vision health, general health, and population health.
THE NEED FOR EVIDENCE-BASED GUIDELINES: ESTABLISHING THE BASELINE
Evidence-based guidelines are an important foundational element to anchor a population health approach that advances eye and vision health. First, they provide guidance based on sound, objective evidence that a variety of care providers can use to improve the uniformity of messaging about, and the quality of, patient care. Second, they establish a baseline from which to measure improvement in care processes and patient health outcomes. Third, they promote a culture of accountability by enabling performance comparisons and encouraging the uniform adoption of best practices.
To promote clear and consistent messaging about whom needs what care and when, it is important that a single set of evidence-based guidelines be available to the public, especially in the context of vision screenings and comprehensive eye examinations. Evidence-based guidelines released by the American Academy of Ophthalmology and the American Optometric Association (AOA), which are based on a review of the literature, serve to guide the members of the respective organizations in the treatment and management of eye diseases.1 These organizations use some of the same literature from which to develop their guidelines and, in many cases, offer consistent guidelines or recommendations. However, there are important differences, especially on topics related to the frequency of exams, specific types of treatments, and the use of screenings. These differences may be due to a confluence of factors, including guideline development processes, evidentiary standards, and professional emphasis of optometrists and ophthalmologists.
This section is intended to describe some of the current inconsistencies and challenges related to clinical practice guidelines in eye and vision health and to reiterate the standards to which clinical practice guidelines should be held. This section does not endorse any particular set of professional guidelines nor make conclusions about the quality of evidence supporting any specific guideline.
___________________
1 Clinical practice guidelines developed by AAO are referred to as “Preferred Practice Pattern® guidelines,” whereas clinical practice guidelines developed by AOA are referred to as “Clinical Practice Guidelines” or “Optometric Clinical Practice Guidelines.” Throughout this report, AAO and AOA clinical practice guidelines will be referred to as “guidelines,” or, wherever the distinction is pertinent, as “evidence-based guidelines” or “consensus-based guidelines.”
Distinguishing Comprehensive Eye Examinations from Vision Screenings
A comprehensive eye examination is a dilated eye examination that may include a series of assessments and procedures to evaluate the eyes and visual system, assess eye and vision health and related systemic health conditions, characterize the impact of disease or abnormal conditions on the function and status of the visual system, and provide treatment and follow-up options (see Chapter 1). Eye examinations can detect incipient eye disease before the onset of visual symptoms. Eye examinations can also detect chronic conditions, such as diabetes and multiple sclerosis (Crews et al., 2016; Frohman et al., 2008). Schaneman and colleagues (2010) conducted a retrospective, claims-based analysis of employed adults living in the United States, and found that individuals who detected a chronic disease through an eye examination had lower first-year health plan costs and fewer missed work days and were less likely to terminate employment than individuals who did not detect chronic conditions early through eye examinations.
Eye examinations are performed by ophthalmologists and optometrists and usually include taking a patient history, assessing the patient’s visual functioning (e.g., visual acuity, field of vision, eye movements), observing indicators of eye health (e.g., intraocular pressure), and examining the state of the pupil, iris, cornea, lens, optic nerve, retina, and other parts of the eye after dilation (AAO, 2012a). Eye providers may also perform the procedures listed in Box 7-1, along with cover tests, color blindness tests, depth perception tests, and other supplementary tests. Comprehensive eye examinations may last 45 to 90 minutes, and may require the use of photoropters, keratometers, tonometers, gonioscopy, diagnostic testing, and anesthetic and pupillary dilating eye drops (AAO, 2012a, 2016c; AOA, 2015d).
Vision screenings are tools that allows for the possible identification but not diagnosis of eye disease and conditions. Screenings are available for a variety of diseases and conditions, such as refractive error, eye problems in children, diabetic retinopathy, and age-related macular degeneration (AMD) (AAPOS, 2014; Garg and Davis, 2009; Jain et al., 2006). Vision screenings can be used by both optometrists and ophthalmologists, as well as by other health care professionals, as a public health tool in community settings to identify potential vision problems early and to assist in the collection of evidence-based population vision health data (AAPOS, 2014). Vision screenings are used to identify issues with visual functioning or symptoms suggestive of an eye disease or condition. Box 7-2 lists a number of examples of common vision screening tests.
There is significant debate among professional and advocacy organizations about the role of vision screenings in clinical eye and vision care. Comprehensive eye examinations and vision screenings have different
strengths and weaknesses, and each serves a different role in the promotion of eye and vision health. In general, the findings of eye examinations are more complete, accurate, precise, and broader in scope than the results of vision screening. A comprehensive eye exam is more sensitive and specific and can precisely measure the extent—and identify the cause—of decreased visual acuity and the presence of eye disease and disorders and conditions of the eye and visual system, in addition to providing other assessments of eye health and functioning. The costs of comprehensive eye examinations are briefly mentioned in Chapter 6.
On the other hand, vision screenings have the potential to improve eye and vision health through potentially less expensive and resource-intensive
means of identifying specific vision problems, especially in children. For example, visual acuity tests using a letter or symbol chart, autorefractor, or photoscreener can be performed in a few minutes by a school nurse at no cost to the patient, or by primary care providers as part of a comprehensive physical examination. However, the effectiveness of vision screening as a diagnostic tool varies among patient populations, and depends on the screening tools used and the diseases or conditions targeted by screening (Chou et al., 2016a,b; USPSTF, 2011). Furthermore, studies have reported low or inconsistent rates of referral and follow-up care for individuals with abnormal screening results (Hartmann et al., 2006; Hered and Wood, 2013; Kemper et al., 2011).
The cost-effectiveness of screening can be sensitive to the eye disease or conditions and vision impairment risk profile of the targeted population, the screening interval, the diagnostic accuracy of screening tools, the staffing models used in a screening program, the rate of follow-up after abnormal screening results, the efficacy of available clinical treatments, and numerous other factors. Research on these and other issues is needed, but remains limited (Azuara-Blanco et al., 2016; Blumberg et al., 2014; Burr et al., 2007; Ladapo et al., 2012). The cost-effectiveness of vision screenings and comprehensive eye examinations is considered further in Chapter 6.
Conflicting study results and relatively limited research on the cost-effectiveness of vision screenings for specific eye diseases and conditions and comprehensive eye examinations for asymptomatic patients can also impede efforts to align existing guidelines (see, e.g., AHRQ, 2012; Burr et al., 2007; Gangwani et al., 2014; Jones and Edwards, 2010; Karnon et al., 2008; Rein et al., 2012a,b).
Advancing Evidence-Based Guidelines
Guidelines for Comprehensive Eye Examinations
Although comprehensive eye examinations are generally accepted as the gold standard in clinical vision care to most accurately identify and diagnose eye and vision problems, different professional groups often disagree on the age and frequency at which different patient groups should specific services. Both the American Academy of Ophthalmology and AOA recommend that at-risk populations receive more frequent eye exams. For example, the American Academy of Ophthalmology and AOA guidelines and/or policy statements both indicate that the frequency of age-related eye examinations should increase with advancing age, but AOA supports shorter intervals for testing at each age (AAO, 2015a; AOA, 2015d). In addition, AOA recommends that persons ages 18 to 39 without symptoms or risk factors be seen at least every 2 years, whereas the American
Academy of Ophthalmology states that “a routine comprehensive annual adult eye examination in individuals under age 40 unnecessarily escalates the cost of eye care” and is not indicated without specific risk factors or symptoms (AAO, 2015a; AOA, 2015d). Table 7-1 compares AAO and AOA guidelines on the frequency of comprehensive eye examinations for patients by age group with and without risk factors or symptoms.
TABLE 7-1 Comparison of AAO and AOA Guidelines for Frequency of Comprehensive Eye Examinations for Adults
| AAO | AOA | ||||
|---|---|---|---|---|---|
| Ages | Without Risk Factors or Symptomsa | Higher Risk Groupsb | Ages | Without Risk Factors or Symptomsc | At-Risk Groupsd |
| Adults under age 40 | 5–10 yearse | Every 1–2 years | 18–39 | At least every 2 yearsf | At least annually or as recommended |
| 40–54 | Every 2–4 yearsg | Every 1–3 years | 40–64 | At least every 2 yearsh | At least annually or as recommended |
| 55–64 | Every 1–3 years | Every 1–2 years | |||
| 65 and older | Every 1–2 years | Every 1–2 years | 65 and older | Annuallyh | At least annually or as recommended |
a Intervals in this column apply to individuals who lack “symptoms or other indications following the initial comprehensive medical eye evaluation.” These intervals account for “the relationship between increasing age and the risk of asymptomatic or undiagnosed disease” (AAO, 2016c).
b Intervals in this column apply to individuals with glaucoma risk factors. The recommended frequency of eye exams varies among eye disease risk factors (AAO, 2016c).
c Intervals in this column apply to “asymptomatic, low risk” individuals (AOA, 2015d).
d Intervals in this column apply to “[p]ersons who notice vision changes, those at higher risk for the development of eye and vision problems, and individuals with a family history of eye disease.” AOA states that “adult patients should be advised by their doctor to seek eye care more frequently than the recommended re-examination interval if new ocular, visual, or systemic health problems develop” (AOA, 2015d).
e AAO states that “routine comprehensive annual adult eye examination in individuals under the age of 40 unnecessarily escalates the cost of eye care” and is not indicated without specific risk factors or symptoms (AAO, 2015a).
f AOA Consensus-Based Action Statement (AOA, 2015d).
g AAO states that “[a]dults with no signs or risk factors for eye disease should receive a comprehensive medical eye evaluation at age 40 if they have not previously received one” (AAO, 2016c).
h AOA Evidence-Based Action Statement (AOA, 2015d).
SOURCES: AAO, 2015a, 2016c; AOA, 2015d.
Guidelines for pediatric populations may also differ. For example, AOA guidelines on pediatric eye and vision examinations recommend comprehensive eye examinations for both asymptomatic/risk-free and at-risk pediatric patients. Table 7-2 details AOA recommendations on the frequency of pediatric eye examinations. By comparison, the American Academy of Ophthalmology states that “[c]omprehensive eye examinations are not necessary (but can be performed) for healthy asymptomatic children who have passed an acceptable vision screening test, have no subjective visual symptoms, and have no personal or familial risk factors for eye disease” (AAO, 2012b). It further recommends eye examinations for children who “fail a vision screening, are untestable, have a vision complaint or an observed abnormal visual behavior, or are at risk for the development of eye problems” and, where appropriate, for children with learning disabilities to rule out the presence of eye and vision problems, and for children with intellectual disabilities, neuropsychological conditions, or behavioral issues that cause them to be otherwise untestable (AAO, 2012b).
The American Academy of Ophthalmology and AOA guidelines for the evaluation and treatment of specific eye diseases may also vary in terms of the recommended frequency of eye care. For example, the American Academy of Ophthalmology guidelines on primary open-angle glaucoma
TABLE 7-2 AOA Recommended Frequency of Comprehensive Eye Examinations for Pediatric Patients
| Age | Asymptomatic/Risk-Free | At-Risk |
|---|---|---|
| Birth to 24 months | At 6 months of age | At 6 months of age or as recommended |
| 2–5 years | At 3 years of age | At 3 years of age or as recommended |
| 6–18 years | Before first grade and every 2 years thereafter | Annually or as recommended |
NOTES: AOA states that “[t]he extent to which a child is at risk for the development of eye and vision problems determines the appropriate re-evaluation schedule. Individuals with ocular signs and symptoms require prompt examination. Furthermore, the presence of certain risk factors may necessitate more frequent examinations, based on professional judgement” (AOA, 2002). According to AOA, the factors placing an infant, toddler, or child at significant risk for visual impairment include “prematurity, low birth weight, prolonged supplemental oxygen, or grade III or IV intraventricular hemorrhage; a family history of retinoblastoma, congenital cataracts, or metabolic or genetic disease; infection of mother during pregnancy (e.g., rubella, toxoplasmosis, venereal disease, herpes, cytomegalovirus, or human immunodeficiency virus); difficult or assisted labor, which may be associated with fetal distress or low Apgar scores; high refractive error; strabismus; anisometropia; known or suspected central nervous system dysfunction evidenced by developmental delay, cerebral palsy, dysmorphic features, seizures, or hydrocephalus” (AOA, 2002).
SOURCE: AOA, 2002.
recommend follow-up evaluations for glaucoma patients from every 1 to 2 months to at least once every 12 months, depending on the duration of control of intraocular pressure (IOP), the extent of progression of glaucomatous damage, and whether the patient’s target IOP is reached (AAO, 2015b). By comparison, the frequency of follow-up glaucoma evaluations recommended by AOA varies by patient status and the stability and severity of disease, ranging from weekly or biweekly evaluations for new glaucoma patients or patients with unstable IOP, progressing optic nerve damage, or visual field loss, to once every 6 to 12 months for suspected cases of glaucoma, depending on a particular patient’s risk (AOA, 2011).2
Vision Screening Guidelines
Guidelines specific to vision screenings may also differ and, in some instances, even contradict one another. In a joint policy statement on pediatric vision screenings in community, school, and primary care settings, the American Academy of Ophthalmology and the American Association for Pediatric Ophthalmology and Strabismus (AAPOS) stated that “routine comprehensive professional eye examinations performed on normal asymptomatic children have no proven medical benefit” (AAO, 2013, p. 3). Instead, the American Academy of Pediatrics (AAP), the American Association of Certified Orthoptists (AACO), AAPOS, and the American Academy of Ophthalmology jointly recommended that pediatricians assess the visual system beginning in infancy and continuing at regular intervals throughout childhood and adolescence. They also suggested that serial visual system screenings in the medical home, using validated techniques, could provide an effective mechanism for the detection and subsequent referral of potentially treatable visual system disorders (AAP et al., 2016). Table 7-3 details the joint pediatric vision screening recommendations.
Conversely, AOA does not generally recommend vision screenings (AOA, 2015d, 2016c). AOA argues that screening is not effective in detecting eye and vision health problems (AOA, 2016c). Moreover, AOA argues
___________________
2 According to the American Academy of Ophthalmology and AOA guidelines on primary open-angle glaucoma, care of patients with glaucoma includes initial and follow-up glaucoma evaluations (AAO, 2015b; AOA, 2011). Initial glaucoma evaluations include many of components of a comprehensive eye examination in addition to procedures or tests specific to diagnosis of glaucoma. For follow-up glaucoma evaluations, the American Academy of Ophthalmology guidelines state that evaluation involves “clinical examination of the patient, including optic nerve head assessment (with periodic color stereophotography or computerized imaging of the optic nerve and retinal nerve fiber layer structure) and visual field assessment” (AAO, 2015b, p. 76). AOA guidelines state that follow-up evaluations are similar to the initial evaluation and may include patient history, visual acuity, blood pressure and pulse, biomicroscopy, tonometry, gonioscopy, optic nerve assessment, nerve fiber layer assessment, fundus photography, and automated perimetry, among other procedures (AOA, 2011).
TABLE 7-3 AAP, AAO, AAPOS, and AACO Joint Recommendations on Frequency of Visual System Assessment in Asymptomatic Infants, Children, and Young Adults by Pediatricians
| Assessment | Newborn to 6 Months | 6–12 Months | 1–3 Years | 4–5 Years | 6 Years and Older |
|---|---|---|---|---|---|
| Ocular history | X | X | X | X | X |
| External inspection of lids and eyes | X | X | X | X | X |
| Red reflex testing | X | X | X | X | X |
| Pupil examination | X | X | X | X | X |
| Ocular motility assessment | X | X | X | X | |
| Instrument-based screening when available | Xa | X | X | Xb | |
| Visual acuity fixate and follow response | Xc | X | X | ||
| Visual acuity age-appropriate optotype assessment | Xd | X | X |
a AAO recommends instrument-based screening at age 6 months. However, the rate of false-positive results is high for this age group, and the likelihood of ophthalmic intervention is low. A future AAO policy statement will likely reconcile what appears to be a discrepancy.
b Instrument-based screening at any age is suggested if the care provider is unable to test visual acuity monocularly with age-appropriate optotypes.
c The development of fixating on and following a target should occur by 6 months of age; children who do not meet this milestone should be referred.
d Visual acuity screening may be attempted in cooperative 3-year-old children.
SOURCE: AAP et al., 2016.
that vision screening promotes a “false sense of security” and delays treatment for individuals with eye diseases or conditions (AOA, 2016c). Draft guidelines on pediatric eye and vision examination currently available on AOA’s website for comment state that “age-appropriate examination strategies should be used” for infants and toddlers, whereas preschool-age and school-age children are more cooperative with traditional eye and vision tests, although some modifications may still be necessary (AOA, 2016, p. 13). AOA guidelines on the care of patients with amblyopia state that “screening for causes of form deprivation amblyopia should be conducted by the infant’s primary care physician within the first 4 to 6 weeks after birth, and children at risk should be monitored yearly throughout the sensitive developmental period (birth to 6 to 8 years of age)” (AOA, 2004, p. 12). AOA is currently updating its clinical practice guidelines, many of which are almost 15 years old, to better reflect recent research.3 Again, the import of these examples is not to endorse or criticize a particular set of
___________________
3 Personal communication, R. Peele, American Optometric Association, September 6, 2016.
guidelines, but rather to highlight conflicting information available to the public and health care providers.
Lack of guideline consistency is exacerbated by separate guidelines for primary care providers and related evidentiary standards. The U.S. Preventive Services Task Force (USPSTF) issues evidence-based recommendations that assess the balance of benefits and harms of preventative services provided to asymptomatic patients in a primary care setting or by a primary care clinician (USPSTF, 2016a).4 In 2013, the USPSTF concluded that current evidence “was insufficient to assess the balance of benefits and harms of screening for primary open-angle glaucoma (POAG) in adults,” citing the inadequacy of available evidence (USPSTF, 2013).5 USPSTF found no studies that directly evaluated the impact of glaucoma screening on the prevention of visual field loss, vision impairment, or worsened quality of life, nor any direct evidence demonstrating screening-related harm. Evidence on the accuracy of glaucoma screening was inadequate, and the risks of inaccurate diagnosis associated with glaucoma screening were recognized (Moyer, 2013; USPSTF, 2013). Similar conclusions were reached regarding visual acuity screening of asymptomatic adults ages 65 and older (USPSTF, 2016a). The USPSTF concluded that “evidence [was] insufficient to assess the balance of benefits and harms of screening for impaired visual acuity in older adults” (USPSTF, 2016c, p. 908). In particular, although evidence supporting the benefits of early treatment of refractive error, cataracts, and AMD was deemed adequate, three randomized controlled trials (RCTs) found no association between visual acuity screening of adults ages 65 and older and improved clinical outcomes (Chou et al., 2016b; USPSTF, 2016c).
In contrast, visual acuity screening by a primary care provider of all children ages 3 to 5 years to detect amblyopia or amblyopia risk factors received a B grade recommendation (USPSTF, 2011). The USPSTF found adequate evidence that the early treatment of amblyopia is associated with improved vision outcomes and that vision screening tools are reasonably
___________________
4 USPSTF recommendations are graded according to the strength of evidence identified by literature review. Grade A recommendations are used when “[t]here is high certainty that the net benefit is substantial.” Grade B recommendations are used when “[t]here is high certainty that the net benefit is moderate or there is moderate certainty that the net benefit is moderate to substantial.” Grade C and D recommendations are used when there is moderate certainty that the net benefit is small or there is evidence of harm or lack of benefit (USPSTF, 2016b). The Patient Protection and Affordable Care Act of 2010 permits the secretary of the U.S. Department of Health and Human Services to authorize Medicare coverage of preventative services that receive an A or B grade from the USPSTF. Therefore, although the USPSTF does not account for the cost of a service when assessing its benefits and harms, its recommendations nevertheless have consequences for Medicare payment policy and care access (Lesser et al., 2011).
5 USPSTF I statements are used when “[e]vidence is lacking, of poor quality, or conflicting, and the balance of benefits and harms cannot be determined” (USPSTF, 2016b).
accurate at detecting refractive error, strabismus, and amblyopia. There was limited evidence on the harms of screening. Although a literature review found limited direct evidence supporting the comparative benefit of screening over not screening in pediatric populations, USPSTF noted that good evidence supporting the accuracy of screening methods and the effectiveness of treatments suggests that screening is more likely to lead to improved eye health than no screening (Chou et al., 2011; USPSTF, 2011). USPSTF I statements include information on clinical considerations (e.g., potential preventable burden, potential harm of the intervention, costs, and current practice) in an effort to provide guidance to primary care providers in the absence of a recommendation (Petitti et al., 2009).
Some experts have criticized the USPSTF methodology, especially when rigorous evidentiary standards contribute to the proliferation of I statements that provide limited clinical guidance. For example, Lee (2016) noted that the methodological and financial challenges of conducting an RCT on the impact of visual acuity screening among asymptomatic older adults poses a barrier to the production of evidence that could lead to a conclusive recommendation. Parke and colleagues (2016), commenting on the same recommendation, noted that the review discounted strong evidence on the negative health consequences of vision impairment. Others have noted that the high standards for evidence quality and the narrow focus of questions informing USPSTF literature reviews limit the scope of recommendations and their value as tools to guide clinical decisions (Donahue and Ruben, 2011; Sommer, 2016).
Developing a unified set of evidence-based clinical and rehabilitation guidelines could serve to guide payment policies and address inconsistencies, coverage gaps, and duplicative or wasteful spending. Recent initial attempts by the American Academy of Ophthalmology, the American Society of Cataract and Refractive Surgery, and the American Academy of Optometry to foster such unification through joint educational initiatives or integrated care delivery have highlighted the opportunity for providers to engage one another and promote quality, efficient care (AAO/AAO, 2013; Bailey, 2013).
Assessing the Quality of Guidelines for Eye and Vision Care
In its 2011 report Clinical Practice Guidelines We Can Trust, the IOM highlighted the need for a set of standards that clinical guidelines must meet in order to be trustworthy and serve as a framework for provider decision making (IOM, 2011b). This will require systematic reviews of the evidence, including research question identification, adherence to evidentiary standards, and a compilation of all findings that meets the standard (IOM, 2011a,b). According to the IOM, the evidence should be used to
establish guidelines, which are reviewed and updated every 3 to 5 years (IOM, 2011a). This will require an ongoing commitment to a rigorous evidence-based guideline development process and greater collaboration among professional groups. Box 7-3 includes a list of specific standards from which to assess guideline quality.
Unfortunately, limited research suggests that many existing guidelines, in general, do not meet these standards. For example, a 2012 review of 114 guidelines found that only 49.1 percent met more than half of 18 selected IOM standards (Kung et al., 2012). Other assessment tools, such as the Appraisal of Guidelines for Research and Evaluation II instrument, can be used to assess the quality of guidelines in terms of their scope and purpose, stakeholder involvement, rigor of development, clarity of presentation, applicability, and editorial independence.6 Several peer-reviewed evaluations of the quality of AAO guidelines are available (Wu et al., 2015a,b,c,d; Young et al., 2015). The committee did not find any similar assessments of AOA guidelines in the published literature. Although the committee was not constituted to evaluate or identify the most effective tools for evaluating guidelines for eye and vision health, it is important that the development of evidence-based guidelines adhere to particular standards, to the extent possible, to ensure robust and comprehensive support for recommended actions.
___________________
6 The Appraisal of Guidelines for Research and Evaluation (AGREE) and AGREE II instruments are frequently used in clinical practice guideline (CPG) evaluations. All CPG evaluations referred to in this report use AGREE or AGREE II. See Brouwers et al. (2010) for a description and comparison of the tools.
The committee acknowledges efforts within the eye and vision field to improve professional guidelines. The American Academy of Ophthalmology and AOA have made efforts to improve and ensure the quality of their respective guidelines. According to Lum and colleagues (2016), the American Academy of Ophthalmology guidelines are “based on the best available scientific data as interpreted by panels of knowledgeable physicians and methodologists”—in cases where data are not persuasive, guideline development groups must “rely on both their collective experience and the evaluation of current evidence” (p. 928). The American Academy of Ophthalmology guideline development process includes the identification, limitation, and management of conflicts of interest through an adherence to codes set by the Council of Medical Specialty Societies, and review by several medical societies and relevant patient organizations. The American Academy of Ophthalmology guidelines use standardized methods to grade the quality of individual studies, the quality of the body of evidence supporting recommendations, and the strength of recommendations.7 Barring revisions, the American Academy of Ophthalmology guidelines are valid for 5 years after their release date (AAO, 2016e). As of May 2016, 22 AAO Preferred Practice Pattern® guidelines were listed on the AAO website (AAO, 2016a).
AOA has developed 18 consensus-based guidelines and two evidence-based guidelines, Eye Care of the Patient with Diabetes Mellitus, and the Comprehensive Adult Eye and Vision Examination (AOA, 2014, 2015d).8 AOA has created a 14-step process for developing evidence-based guidelines and states that its Evidence-Based Optometry Committee is revising optometric guidelines in response to the IOM standards for trustworthy guidelines (AOA, 2015c, 2016a). AOA evidence-based guidelines use scales to grade the strength of evidence and recommendations and offer a guideline development process that includes steps to manage conflicts of interest and allow for peer and public review (AOA, 2014, 2015d).9 AOA evidence-based guidelines state that the guidelines should be revised every 2 to 5 years (AOA, 2014, 2015d). Unfortunately, statements in AOA consensus-
___________________
7 AAO uses the Scottish Intercollegiate Guideline Network and the Grading of Recommendations Assessment, Development and Evaluation methods for grading evidence quality and recommendation strength. For details, see the Methods and Key to Ratings section of an AAO Preferred Practice Pattern guideline.
8 At the time of writing, a third clinical practice guideline (Comprehensive Pediatric Eye and Vision Examination) was under revision, and a draft version of the document was available for peer and public review.
9 The evidence and recommendation grading tools as described in the AOA evidence-based guidelines on Eye Care of the Patient with Diabetes Mellitus and on Comprehensive Adult Eye and Vision Examination are not identical. For details, see the How to Use This Guideline sections of these guidelines.
based guidelines, such as the guidelines on Pediatric Eye and Vision Examination, do not always provide information on the graded strength of this evidence or of the statements it supports (AOA, 2002). Similarly, the AOA consensus guidelines on Pediatric Eye and Vision Examination do not include information on the guideline development process, including methods of literature review, management of conflicts of interest, or external review (AOA, 2002). AOA consensus-based guidelines, such as the guidelines on Pediatric Eye and Vision Examination, state that the guidelines will be reviewed periodically and revised as needed (AOA, 2002).10
Improving the Consistency of Eye and Vision Care Guidelines
Coming to consensus on recommended care is critical to create clear messaging that targets both patients and non–eye care providers about the need for regular eye care services. From a population health perspective, prioritizations for guideline development should be influenced by the number of people affected, the severity and reversibility of vision loss that can occur, the diversity with which the condition is currently managed, the number of health care professionals who typically engage with patients about a specific disease or condition, and the breadth of the literature currently available. This might be of most relevance for guidance on the frequency of eye examinations and may be supported by federal guidance on what factors (e.g., frequency, severity, preventability, treatability, difference between current and optimal practice) should be used in consensus-based and evidence-based recommendations.
A collaborative and inclusive working group would be useful to establish a single set of guidelines that are coherent, comprehensive, and clear about what services are required at what intervals, and how best to connect patients to necessary follow-up care. The process for conducting a systematic review of existing evidence and distilling guidelines from that evidence is a lengthy process, and collaboration throughout the eye and vision care field (including federal and state governmental entities that focus on eye and vision health) will be paramount. This collaborative process can also help identify research gaps to promote a shared research agenda.
___________________
10 The AOA consensus-based guideline on Care of the Patient with Learning Related Vision Problems states that the guideline “will be reviewed periodically” (AOA, 2008, p. ii). All other consensus-based guidelines state that the guideline will be “will be reviewed periodically and revised as needed.”
ASSESSING QUALITY AND IMPROVEMENT INITIATIVES
Focusing on quality improvement as an overarching goal can help standardize and promote high-quality care; in turn, high-quality care can address factors that contribute to poor and inequitable eye and vision health care. Continuous quality improvement (CQI) is a “process-based, data-driven approach to improving the quality of a product or service through iterative cycles of action and evaluation” (RWJF, 2012, p. 1). Rather than a short-term, single-issue quality improvement and assurance initiative, CQI focuses on correcting the root causes of systemic issues through an iterative quality improvement process. This process complements the public health wheel (see Chapter 1), which depicts population health as an iterative process of assessment, policy development, and assurance.
Figure 7-1 presents a common CQI framework adapted for eye and vision care. In this figure, the CQI process is a five-step process: guideline development, practice change, performance monitoring, surveillance and data analysis, and identification of opportunities for improvement. An adherence to established guidelines provides the baseline from which to
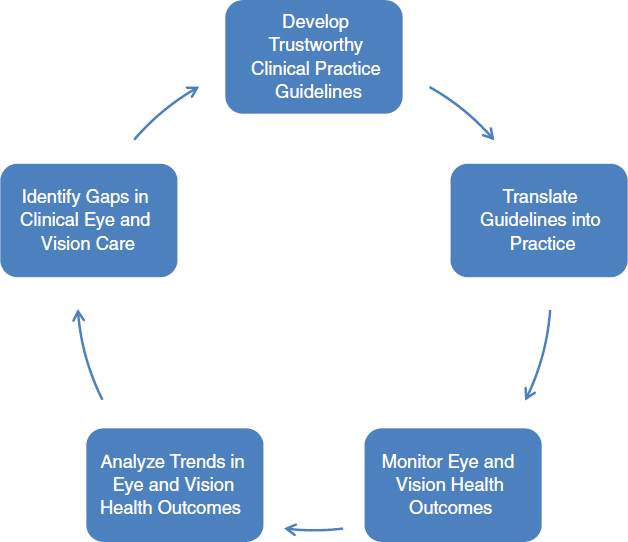
SOURCE: Adapted from Kneib, 2009.
measure impact on health outcomes and improvement over time. Development of evidence-based guidelines requires sufficient research and data. The rapid and accurate translation of guidelines into daily practice may require the development and provision of educational materials and training programs informed by translational and implementation science. Tracking patient outcomes and process and performance measures of the health care system requires a broad set of surveillance and monitoring tools, including electronic health records (EHRs). Data analysis may require the expertise of statisticians, epidemiologists, health informaticians, and other public health and quality improvement specialists. Identifying opportunities for improved patient outcomes and health care system performance can then inform revisions of evidence-based guidelines, which can help accelerate policy changes that better support eye and vision health.
An effective CQI process requires organizational champions and committed leadership. A number of government agencies, educational organizations, and professional groups are involved in CQI activities or efforts that could facilitate a CQI process, to improve the quality of eye and vision care. The Health Resources and Services Administration (HRSA) is one of a number of agencies and organizations that provide guidance and tools to help stakeholders design, implement, sustain, and spread quality improvement within a health care system (HRSA, 2011). Toolkits have been developed to promote CQI in eye and vision health (Brien Holden Vision Institute, 2016). AOA and the American Academy of Ophthalmology have launched patient registries as part of larger quality improvement efforts (AAO, 2016b; AOA, 2015b).11 Patient registries can promote improvements in care quality by enabling providers to assess the effectiveness of treatment, analyze their performance for improvement opportunities, and monitor patient outcomes. Data from patient registries may also be used to inform policy, monitor the effectiveness of treatments across populations, and identify public health issues (Kent, 2015). The U.S. Department of Veterans Affairs (VA) has employed its Quality Enhancement Research Initiative to identify opportunities for improving eye care and preventing vision loss among veterans with diabetes (Krein et al., 2008). The Diabetes Recognition Program of the National Committee for Quality Assurance (NCQA) recognizes physicians for complying with diabetes care quality measures, which include the appropriate referral of diabetic patients for eye examination (NCQA, 2016b). In January 2016, HRSA announced a funding opportunity for rural providers to participate in a Small Health Care Provider Quality Improvement Program, and it listed eye exams for diabetic patients as an optional quality measure (HRSA, 2016b). Although evidence
___________________
11 See Chapter 4 for further detail on the AOA and American Academy of Ophthalmology registries.
on the impact of these efforts and programs on eye care quality and patient outcomes is limited, their existence points to an increased emphasis on CQI in eye and vision care. These efforts can be used as a foundation from which to implement subsequent CQI initiatives.
Quality improvement is also being explicitly built into grants that support eye and vision health in communities. For example, the National Institute for Children’s Health Quality has partnered with the National Center for Children’s Vision & Eye Health at Prevent Blindness on a 3-year, HRSA-funded project to support the development of comprehensive and coordinated approaches to children’s vision and eye health in five states (NICHQ, 2015). The project will employ quality improvement (QI) principles and practices to strengthen statewide partnerships and stakeholder coordination, increase accessibility of eye care in remote communities, increase early detection and treatment of eye diseases, establish state-level surveillance, and implement accountability measures (NICHQ, 2015). As more eye and vision professionals participate in accountable care organizations or other pay-for-performance initiatives in the value-driven health care landscape, CQI will play an increasingly important role in clinical eye and vision care.
A number of organizations, including the National Quality Forum (NQF), Physician Consortium for Performance Improvement, NCQA, and Agency for Healthcare Research and Quality, have identified and developed, or evaluated and endorsed, quality improvement measures and metrics that can be used for CQI activities (AHRQ, 2016a; AMA, 2016; NCQA, 2016a; NQF, 2016c). The NQF has endorsed several quality measures related to counseling and eye examinations for patients with AMD, eye examination and follow-up care for patients with diabetic retinopathy, examination of the optic nerve head and treatment outcomes for patients with glaucoma, and complications and outcomes after cataract surgery.12 Unfortunately, NQF has not endorsed—and the National Quality Measures Clearinghouse does not list—other measures related to eye and vision care, including those pertaining to vision screening and the subsequent referral and follow-up of adult patients, referral to vision rehabilitation and support services for patients with irreversible vision impairment, correction of identified
___________________
12 NQF-endorsed, eye care–related quality measures include Primary Open-Angle Glaucoma: Optic Nerve Evaluation (NQF-0086); Age-Related Macular Degeneration: Dilated Macular Examination (NQF-0087); Diabetic Retinopathy: Documentation of Presence or Absence of Macular Edema and Level of Severity of Retinopathy (NQF-0088); Diabetic Retinopathy: Communication with the Physician Managing Ongoing Diabetes Care (NQF-0089); Primary Open-Angle Glaucoma: Reduction of Intraocular Pressure by 15% or Documentation of a Plan of Care (NQF-0563); Cataracts: Complications Within 30 Days Following Cataract Surgery Requiring Additional Surgical Procedures (NQF-0564); Cataracts: 20/40 or Better Visual Acuity Within 90 Days Following Cataract Surgery (NQF-0565); and Age-Related Macular Degeneration: Counseling on Antioxidant Supplement (NQF-0566).
refractive error, or patient counseling on modifiable risk factors for eye disease. To support the CQI process, research to build the evidence base informing these and other quality these measures will need to be pursued.
Pursuing a CQI research agenda will require collaboration among investigators working in multiple areas, as well as the dedication of funding, tools, and facilities. It will also require a population health surveillance system capable of collecting data on myriad facets of the vision care system and the populations and communities it serves (see Chapter 4). These surveillance data are necessary for the implementation of a successful CQI program to improve the performance of the vision care system.
PROMOTING DIAGNOSIS AND FOLLOW-UP CARE TRANSITIONS THROUGH INTEGRATION
Identifying who needs what care at what time is only part of the equation to promoting appropriate eye care. In many cases, especially in the context of vision screening, additional follow-up care will be necessary to provide prescription lenses or other types of clinical treatments or monitoring. Despite advances in the clinical treatments for major eye diseases that have dramatically improved population eye and vision health, many barriers to care delivery remain. Structural separations between optometry, ophthalmology, and primary care may contribute to inconsistent referral practices, poor communication, inappropriate or delayed referrals, and interruptions in care continuity, as reported in studies assessing the state of primary care–specialty referrals (Mehrotra et al., 2011; Wiggins et al., 2013). The integration of primary care and eye and vision care, as modeled in the patient-centered medical home and accountable care organizations, holds potential as a strategy for improving coordination and communication between providers in primary care and those in eye and vision care. EHRs and other health informatics tools can contribute to integration by enabling secure data sharing among providers.
Referrals Within and Across Professional Lines
According to the World Health Organization (WHO), collaborative practice occurs when “multiple health workers from different professional backgrounds provide comprehensive services by working with patients, their families, caregivers, and communities to deliver the highest quality of care across settings” (WHO, 2010, p. 13). Beyond simply raising public awareness about the roles, competencies, and services of eye and vision care professionals and ensuring an adequate workforce to meet patient needs (see Chapter 6), it is important to recognize that eye and vision health is the domain of all types of health care providers. For example, eye and
vision care providers can help identify risk factors as well as detect and help manage many chronic diseases. For example, comprehensive dilated eye exams can detect retinal vascular changes that may suggest hypertension or diabetes, reveal cholesterol plaques within retinal arteries that indicate risk of stroke, and detect tumors, among other things (Chous and Knabel, 2014). Similarly, by providing vision screenings and referring patients as appropriate to eye and vision care providers, primary care providers can help identify potentially vision-threatening problems and refer patients to an ophthalmologist or optometrist for a comprehensive eye examination to make a definitive diagnosis, establishing productive and ongoing professional relationships. An investigation of the referral patterns of 136 family physicians found that referrals to ophthalmologists were more likely than referrals to other medical specialties to result in long-term (as compared to short-term) referrals or consultations and in the transfer of patient management (Starfield et al., 2002). However, the study also found that family physicians referred patients with diabetes to ophthalmologists in only 27 of 56 cases, with remaining referrals directed primarily to endocrinologists and nutritionists.
Eye and vision health is also relevant to health care providers beyond primary care physicians. Often, vision impairment is a manifestation or consequence of a disease that requires the expertise of other health care specialists involved in the coordination of care. For example, visual impairment is a key symptom of multiple sclerosis (Balcer et al., 2015). The American Academy of Neurology’s current practice parameter on the diagnostic assessment of children with cerebral palsy notes that vision impairment and disorders of ocular motility occur in 28 percent of children with cerebral palsy and recommends that this patient population receive vision screenings (Ashwal et al., 2004). Endocrinology is another specialty that frequently intersects with vision care, particularly in diabetes management.
Physician assistants and nurse practitioners provide at least 11 percent of all outpatient medical services in the United States, and are more likely to practice in rural areas than primary care physicians (16 percent versus 11 percent) (AHRQ, 2014a; Hooker and Everett, 2012). The inclusion of nurse practitioners and physician assistants in patient care is associated with decreased health care costs, higher-quality care, and improved patient outcomes (Hooker and Everett, 2012; Reuben et al., 2013; Roblin et al., 2004). However, research indicates that nurse practitioners or physician assistants seldom practice in ophthalmology. The 2013 Annual Survey Report of the American Academy of Physician Assistants found that only 10 out of 15,798 responding physician assistants worked in ophthalmology as a primary specialty (AAPA, 2014). A report by the American Association of Nurse Practitioners (AANP) on nurse practitioner practice environments did not include ophthalmology as a subspecialty in which nurse practitioners actively
practiced (AANP, 2015). In addition, specialty certification and postgraduate training focused on ophthalmology is not available for nurse practitioners or physician assistants. The AANP Certification Program does not offer certification in ophthalmic care (AANPCP, 2016). The National Commission on Certification of Physician Assistants offers specialty certifications, but not in ophthalmology (NCCPA, 2016). The Association of Postgraduate Physician Assistant Programs compiles a list of current postgraduate programs; although none of these programs focuses specifically on ophthalmology, several programs include clinical or surgical rotations in ophthalmology (APPAP, 2016).13 For registered nurses specializing in ophthalmic care, certification through the National Certifying Board for Ophthalmic Registered Nurses is available but not required (ASORN, 2016).
Unfortunately, referrals to ophthalmologists and optometrists from other health care professionals remain suboptimal, for various reasons. Holley and Lee (2010) interviewed focus groups of nurse practitioners, physician assistants, and rural and academic primary care physicians in order to identify barriers to referring patients to eye care. The three most commonly cited barriers were “no/little feedback from eye care providers” (27.5 percent of comments), followed by “patient’s lack of finances/insurance coverage” (25.5 percent), and “difficulty in scheduling ophthalmology appointments” (15.7 percent) (p. 1867).14 The most common suggestions for improving referral to eye care involved implementing shared electronic medical records (26.5 percent of comments), improving eye care provider communication and feedback (22.4 percent), and having ophthalmologists in primary care clinics on an intermittent basis (18.4 percent) (Holley and Lee, 2010).15 Thus, there is an opportunity for both eye care professionals and the medical establishment to capitalize on shared interests and concern for patients. By considering referrals to be part of whole patient care, health care providers contribute to practices that may lead to improvements across multiple measures of health.
___________________
13 The Emergency Medicine physician assistant (PA) residency at Johns Hopkins University, the PA Postgraduate Fellowship in Emergency Medicine at Albany Medical Center, the Emergency Medicine PA Residency at the University of California, San Francisco (UCSF) Fresno, the Surgery Physician Assistant Fellowship at the Texas Children’s Hospital, and the Emergency Medicine PA/Nurse Practitioner (NP) Residency Program at Yale New Haven Hospital all include clinical or surgical rotations in ophthalmology (APPAP, 2016).
14 Percentages are calculated. Out of 51 total comments, 14 were categorized as “No/little feedback from eye care providers” [(14/51) × 100 = 27.5 percent], 13 were categorized as “Patient’s lack of finances/insurance coverage” [(13/51) × 100 = 25.5 percent], and 8 were categorized as “Difficulty in scheduling ophthalmology appointment” [(8/51) × 100 = 15.7 percent].
15 Percentages are calculated. Out of 49 total comments, 13 were categorized as “Implement electronic medical records” [(13/49) × 100 = 26.5 percent], 11 were categorized as “Better communication/feedback from ECPs” [(11/49) × 100 = 22.4 percent], and 9 were categorized as “Have ophthalmologists in primary care clinic on certain days” [(9/49) × 100 = 18.4 percent].
Models of Integrated Eye and Vision Care in the United States
Integrated models of care improve efficiencies within the medical establishment to facilitate coordinated, patient-centered care across multiple providers in a time when the increasing prevalence of chronic conditions, such as chronic vision impairment, and age-related diseases require more value-conscious health care systems. For purposes of this report, the term “integrated care” refers to any model of care designed to promote collaborative practice for the purposes of improving eye and vision care access and quality.
Unfortunately, the research on the distribution, frequency, cost, effectiveness, and cost-effectiveness of models of integrated vision care in the United States is limited. A number of studies have documented international efforts to improve referral patterns between general practitioners, ophthalmologists, and optometrists and to implement referral-only diagnostic services for patients with common eye diseases in order to improve the referral process and reduce costs and patient load on secondary and tertiary services. These efforts have met with some success (Bourne et al., 2010; Jamous et al., 2015; Mandalos et al., 2012; Voyatzis et al., 2014). Several of these studies have highlighted the need for new or additional training to effectively implement referral centers. Although these studies can be instructional in identifying factors to consider when designing interventions within the United States, they have limited applicability in the United States because of the differences in how health care systems are structured.
Several recent international investigations have highlighted the success of integrated care models at decreasing the costs of health care, increasing the efficiency of health systems, and improving patient outcomes. For example, an RTC conducted in the Netherlands compared different methods of monitoring glaucoma patients and found that eye care provided to stable glaucoma patients and patients at risk of glaucoma by ophthalmic technicians and optometrists working in hospital-based glaucoma follow-up units was equal in quality and lower in cost than care provided by hospital-based residents and ophthalmologists specializing in glaucoma care (Holtzer-Goor et al., 2010).16 Compared to monitoring of stable glaucoma patients and patients at-risk of glaucoma by glaucoma specialists (i.e., ophthalmologists specializing in glaucoma) and residents, monitoring of these patients by a
___________________
16 Care quality was measured in terms of provider compliance to a predetermined care protocol; multiple indicators of patient satisfaction; the stability of the patient according to practitioner, as measured by changes in the duration of intervals between patient visits; difference between IOP at baseline and at study conclusion; examination results; and the number of treatment changes. There were no statistically significant differences between the care provided by glaucoma specialists or residents and the care provided by optometrists and ophthalmic technicians for any of the care quality measures. Patients randomized to receive care from the glaucoma follow-up unit were seen by an ophthalmologist every third visit, or sooner if necessary.
glaucoma follow-up unit staffed by optometrists and ophthalmic technicians was associated with a high probability of reduced costs of care to the patient (78 percent probability of reduced costs of care), to the hospital (98 percent probability of reduced costs of care), to the health care system (87 percent probability of reduced costs of care), and to society as a whole (84 percent to 89 percent probability of reduced costs of care) (Holtzer-Goor et al., 2010). A subsequent study of the same integrated care model found that care provided in the glaucoma follow-up unit adhered closely to treatment protocols and was preferred by patients (Holtzer-Goor et al., 2016).
A study performed in Belgium compared the efficiency and efficacy of “lean” care pathways for cataract surgery and perioperative care, where the management of uncomplicated cases was shared by ophthalmologists, optometrists, and nurses, to the efficiency and efficacy of traditional pathways, where ophthalmologists managed nearly all elements of care (van Vliet et al., 2010). In the traditional care pathway, the eye examination and pre-assessment (e.g., health check, patient-reported medical history) were performed during separate patient visits to the hospital, and the formulation of the surgical care plan and the next-day post-surgery review required visits by all patients. In the “lean” care pathway, the eye examination and pre-assessment were performed in a single patient visit to the hospital, and the formulation of the surgical plan and next-day post-surgery review did not require patient visits in cases where ocular comorbidities and perioperative complications were not present. The minimum number of patient visits to the hospital decreased from five in the traditional care pathway to three in the lean care pathway. Compared to patients in the traditional pathway, those in the “lean” care pathway required on average significantly fewer hospital visits. Compared to the traditional care pathway, the “lean” care pathway also allowed ophthalmologists to treat significantly more patients in the same amount of time, and the authors suggest that further gains in efficiency might be achieved through greater adherence to the design of the “lean” care pathway (van Vliet et al., 2010).
In the United Kingdom, the National Health Service’s Chronic Eye Care Services Programme supported eight pilot projects that sought to improve care pathways for the treatment of glaucoma, AMD, and vision impairment through better integration of the eye care workforce (McLeod et al., 2006). Together, these findings suggest that the integration of clinical eye and vision care services holds promise for lowering costs, improving patient satisfaction, promoting adherence to guidelines, and ensuring efficient and effective care. It is important to note that while these studies can serve as examples, they may have limited applicability to the U.S. system because of differences in the health care systems, in the scopes of practices of eye care providers, and in the patient populations in these countries compared to those in the United States. The committee was unable to identify
peer-reviewed studies that described how U.S.-based integrated optometrist-ophthalmologist models are organized or operated, their associated costs and cost-effectiveness, or their impact on eye and vision health care and patient outcomes. Existing articles in trade publications only acknowledge that these models exist and are viable from a business perspective, although evidence supporting these claims may be insufficient. This is a much needed area of research. One potential strategy for developing an evidence base would be to conduct demonstration projects testing different models of eye and vision care organization, operation, and payment.
Incorporating Vision and Eye Health into Emerging Medical Models of Care
According to the American College of Physicians, the patient-centered medical home (PCMH) is a “care delivery model whereby patient treatment is coordinated through their primary care physician to ensure they receive the necessary care when and where they need it, in a manner they can understand” (ACP, 2016). The model emphasizes services that are comprehensive, patient-centered, coordinated, accessible, safe, and of high quality (AHRQ, 2016b). A review of peer-reviewed studies, government reports, industry studies, and independent federal program evaluations found that PCMH programs have been associated with reductions in cost and in the unnecessary use of health care services, and may also lead to improvements in patient satisfaction, quality-of-care metrics, and access to primary care services (Nielsen et al., 2016). Examples of PCMH programs that have resulted in improved patient health and care quality, reduced readmissions rates, reduced care costs, and/or short-term return on investments include the New York-Presbyterian Regional Health Collaborative, the Pennsylvania Chronic Care Initiative, and Illinois Medicaid’s Illinois Health Connect and Your Healthcare Plus program (Carrillo et al., 2014; Friedberg et al., 2014; Phillips et al., 2014).
The Veterans Health Administration (VHA) has developed a unique PCMH model called the Patient Aligned Care Team (PACT). The provider team includes a primary care provider, a nurse care manager, a clinical associate, and an administrative clerk. Specialty care, including vision care, is provided through referral (VA, 2016). Studies of the PACT program suggest that PACT implementation is associated with significant improvements in the proportion of acute care patients contacted within 2 days of hospital discharge (Werner et al., 2014). PACT implementation has also been associated with a significant increase in the overall rate of telephone-based encounters between providers and patients, in the proportion of patients seen within 7 days of desired appointment date, and in the proportion of same day appointment requests accommodated (Rosland et al.,
2013). Among 913 VHA primary care clinics, greater progress in the PACT implementation process was associated with higher patient satisfaction, lower staff burnout rates, lower hospital admission rates for some ambulatory conditions, lower emergency department usage rates, and improved performance on 41 of 48 clinical quality measures (Nelson et al., 2014).
Despite the success of PACT and other PCMH models at reducing health care costs and improving some measures of care quality and access, it is not clear what impact these models have had on eye health, as research on the impact of the PCMH model on eye care is limited. However, studies on the impact of the PCMH model on comprehensive diabetes care—a component of which is annual dilated eye exams—can provide clues to the quality of eye care received within a PCMH. Kern and colleagues (2014) compared quality of patient care provided by physicians practicing in PCMHs established over the course of a 3-year study to that provided by physicians not practicing in a PCMH and who used either paper-based patient health records or an EHR. Quality of care was assessed using 10 quality measures, including provision of dilated eye examinations for patients with diabetes. At baseline, no significant differences in the percentage of patients with diabetes who were receiving dilated eye examinations existed between groups. By the end of the study, the proportion of patients with diabetes receiving dilated eye examinations was significantly higher among those receiving care from physicians practicing in a PCMH compared to those receiving care from physicians not practicing in a PCMH. Over the course of the study, performance on the dilated eye examination quality measure was 3–4 percent higher for physicians practicing in PCMHs than for physicians not practicing in PCMHs (Kern et al., 2014). A follow-up study found that although the overall care quality was similar between physician groups, patients with diabetes who were receiving care from physicians in PCMHs were still more likely to have eye exams than patients with diabetes who were receiving care from other physicians (Kern et al., 2016). Another study found that diabetic patients of primary care physicians who did not formally practice within a PCMH but who adhered closely to key features of the PCMH model were more likely to have received an eye exam in the previous year than diabetic patients of primary care physicians who adhered less closely to the PCMH model (Stevens et al., 2014).17 These studies demonstrate the positive impact of the PMCH model of care on the provision of diabetic eye exams, and suggest the possibility that the PCMH model’s emphasis on coordinated, team-based care
___________________
17 Physician adherence to features of PCMH was based on patient responses to the Primary Care Assessment Tools (PCAT) Adult Expanded. A one-point increase in a physician’s overall PCAT score was associated with 1.88 higher odds of his or her patients having received an eye exam in the previous year (Stevens et al., 2014).
that is comprehensive, patient-centered, and high-quality could also benefit other aspects of eye care.
The PCMH model may not always promote the quality of eye and vision care. Among a core set of clinical quality measures recommended by the Patient-Centered Medical Home Evaluators’ Collaborative for evaluating and comparing the effectiveness of PCMH programs, the sole vision-related measure was the percentage of patients ages 18 to 75 with type 1 or 2 diabetes who received a retinal eye exam as a component of comprehensive diabetes care (Rosenthal et al., 2012).18 The 2014 Standards and Guidelines for NCQA’s Patient-Centered Medical Home, which provide guidance for assessing the quality of care offered by practices using the PCMH model, do not explicitly require the collection of vision-related clinical data, the inclusion of vision-specific components within the patient health assessment, or the use of vision screenings except where recommended by major public health agencies or organizations (NCQA, 2014b). Thus, while the PCMH model of care represents an opportunity for improved integration of eye and primary care, the inclusion of more eye care services and additional eye care professionals on the team should be studied to determine how it would affect the quality of eye and vision care in PCMH programs.
Providing high-quality, accessible, and patient-centered care requires coordination between primary care providers practicing in the PCMH and clinical and nonclinical providers and services operating outside the medical home.19 The concept of the “medical neighborhood” was developed to account for the role that this broader set of services plays in achieving the goals of the PMCH, and the NCQA Patient-Centered Specialty Practice (PCSP) Recognition Program was developed to recognize and support specialty practices that seek to improve the quality of specialty care through coordination with primary care, performance measurement, and
___________________
18 In addition to the core set of clinical quality measures, the collaborative also recommended that PCMH program evaluators select a group of clinical quality measures from a list that included measures related to adolescent well-child visits, well-child visits in the first 15 months of life, and well-child visits in the third, fourth, fifth, and sixth years of life. American Academy of Pediatrics guidelines require eye care to be included as part of the physical exam component of these visits (AAP, 2008a,b,c).
19 AHRQ has conceived the medical neighborhood as a “PCMH and the constellation of other clinicians providing health care services to patients within it, along with community and social service organizations and State and local public health agencies” (AHRQ, 2011, p. 5). NCQA has identified specialty practices, accountable care organizations, behavioral health, public health, work site and retail clinics, and pharmacies as members of the medical neighborhood (NCQA, 2014a).
other means (Fisher, 2008; Huang and Rosenthal, 2014; NCQA, 2016d).20 Ophthalmologists, but not optometrists, are eligible to participate in the PCSP Recognition Program (NCQA, 2016c).21 Excluding optometrists from participating in the PCSP program may eliminate the contribution of a critical cohort of eye care providers and may limit the degree to which eye and vision care can be easily incorporated into specific PCMH models and medical neighborhoods.
Accountable care organizations (ACOs) offer another path to integration of primary care and specialty medical services. The Centers for Medicare & Medicaid Services (CMS) describes ACOs as “groups of doctors, hospitals, and other health care providers, who come together voluntarily to give coordinated high quality care” (CMS, 2015b). Initiatives such as the Medicare Shared Savings Program and the Pioneer and Next Generation ACO Models seek to improve the quality of patient care while lowering health care costs (CMS, 2015c,d, 2016b). Achieving these twin goals may require extensive data sharing, transitioning to value-based payment policies, using quality measures to monitor provider performance and patient outcomes, and the coordination of providers and services, among other strategies.
The fact that there is a limited amount of research on the impacts of ACOs on eye and vision health may in part be the consequence of limitations in the reporting requirements for ACOs. ACOs participating in the Medicare Shared Savings Program are required to report quality data, and they must meet established quality performance standards set by CMS in order to be eligible to receive shared savings (CMS, 2016a). Since performance year 2012, quality measures related to diabetes care have been included among the quality performance measures CMS requires ACOs to report; however, a measure related to the provision of diabetic eye exams was not included among these until performance year 2015 (CMS, 2011, 2012, 2014, 2015a, 2016a). For performance years 2015 and 2016, ACO performance on the provision of diabetic eye exams will be reported as a composite measure with ACO performance on control of hemoglobin A1c
___________________
20 The objectives of the PCSP Recognition Program are to enhance coordination between primary care and specialty care, strengthen relationships between primary care clinicians and clinicians outside the primary care specialties, improve the experience of patients accessing specialty care, align requirements with processes demonstrated to improve quality and eliminate waste, encourage practices to use performance measurement and results to drive improvement; and identify requirements appropriate for various specialty practices seeking recognition for excellent care integration within the medical home (NCQA, 2016e).
21 Other clinicians who are eligible to participate in the PCSP Recognition Program include nurse practitioners, physician assistants, and certified nurse midwives, as well as doctoral or master’s-level psychologists, social workers, and marriage and family counselors who are state licensed or certified (NCQA, 2016c).
levels among diabetic patients.22 Data for ACO performance on this composite measure were not available at the time of writing. The addition of eye care–related quality measures to the set of measures that CMS requires ACOs to report could provide useful data on how ACOs affect eye care quality and access.
As the population ages, and chronic eye disease and other vision problems become increasingly prevalent, demand for models of care capable of meeting national eye and vision health needs will continue to grow. More research is needed to understand how the medical field can work as a cohesive and coordinated unit, achieving better value and health outcomes. For example, it is important to determine how PCMHs, ACOs, and other integrated models of care that promote collaborative practice—as well as the policies that inform their organization, adoption, and performance monitoring and quality improvement activities—can be adapted to best meet current and future eye and vision health needs. Strategies and actions to better support integration of clinical eye and vision services with vision rehabilitation, social services, public health departments, and other stakeholders are also needed. More health services research and evaluation of existing programs will be essential to guide these efforts.
Role of Health Informatics
Advances in health information technology, particularly the implementation of EHRs, have the potential to optimize care delivery, enhance quality and safety in a number of ways, and ultimately improve patient outcomes (Blumenthal and Tavenner, 2010). Implementing EHR systems can ensure that providers have access to up-to-date, relevant clinical patient information at the point of care (Chiang et al., 2011). It can also facilitate information exchange across care settings, thereby improving communication and coordination between primary care providers and eye care providers (e.g., optometrists, ophthalmologists) or other specialists treating comorbidities. This would theoretically reduce the duplication of various laboratory tests or imaging and lessen the risk of treatment errors. The data in EHRs can also be used to prioritize, justify, and analyze public health activities by identifying and tracking eye disease and vision impairment—and interventions to prevent and reduce the same—at the population level.
___________________
22 CMS-required quality performance measures on diabetic eye exams and control of hemoglobin A1c levels are based on NQF-endorsed quality measures. NQF-0055: “The percentage of members 18 to 75 years of age with diabetes (type 1 and type 2) who had an eye exam (retinal) performed” (NQF, 2016a). NQF-0059: “The percentage of patients 18–75 years of age with diabetes (type 1 and type 2) whose most recent HbA1c level during the measurement year was greater than 9.0% (poor control) or was missing a result, or if an HbA1c test was not done during the measurement year” (NQF, 2016b).
For this reason, EHRs can be powerful tools for informing public health practice and improving public health.
EHR use has grown considerably in recent years. From 2001 to 2014 the proportion of office-based physicians who used any type of EHR system increased from 18.0 percent to 82.8 percent, and the proportion of office-based physicians who reported having an EHR system that met the criteria for a basic EHR system increased significantly from 11.0 percent in 2006 to 50.5 percent in 2014 (CDC, 2015; Hsiao, 2014).23 However, EHR use among ophthalmologists is low compared with most other medical specialties, with only 15.0 percent and 34.7 percent of ophthalmologists reporting EHR use in 2003 and 2010, respectively, compared with 13.7 percent and 64.2 percent in 2003 and 2010 for general or family medicine. In multivariate analysis, ophthalmologists had lower odds of using EHRs than any other of 13 specialties except psychiatry and dermatology (Kokkonen et al., 2013). A 2012 survey of 492 ophthalmologists found that 32 percent of ophthalmology practices in the United States had adopted EHR systems, and another 31 percent planned to do so within 2 years (Boland et al., 2013). A 2015 survey by AOA found that 17 percent and 49 percent of responding optometrists planned to achieve criteria for, respectively, stage 1 and stage 2 meaningful use in 2015. The survey also found that 66 percent of responding optometrists used complete EHRs, as defined by the AOA (AOA, 2015a).24 Adoption among both groups is increasing: EHR adoption among surveyed ophthalmologists doubled from 2007 to 2011 and increased by 3 percent from 2014 to 2015 among surveyed optometrists (AOA, 2015a; Boland et al., 2013).
The adoption of EHRs by eye care specialists may be impeded by concerns about its effects on the quality of clinical documentation. Sanders and colleagues (2013) compared the effects of a paper-based health record system to those of an EHR system on the clinical documentation habits of ophthalmologists assessing patients with AMD, glaucoma, and pigmented choroidal lesions. For all three diseases, the paper-based system was associated with significantly fewer complete examination findings and critical
___________________
23 According to Hsiao (2014), a basic EHR system is one that has each of the following features: “patient history and demographics, patient problem lists, physician clinical notes, [a] comprehensive list of patients’ medications and allergies, computerized orders for prescriptions, and [the] ability to view laboratory and imaging results electronically.”
24 Complete EHRs were defined as those that included both practice management and patient health information systems. Practice management systems were defined as “electronic software packages that track and maintain information such as: patient demographics, scheduling, billing, insurance, and recall.” Patient health information systems were defined as “electronic software packages that maintain health information such as: exam data, testing, images and prescriptions” (AOA, 2015a, p. 1).
clinical findings (Sanders et al., 2013).25Sanders and colleagues (2014) found that EHR implementation in an ophthalmic surgical practice was associated with significant short-term increases in the portion of total procedure time spent on the documentation of cataract, vitreoretinal, and extraocular procedures and with significant increases in the number of circulating operating room nurses present for cataract and vitreoretinal procedures. These values returned to baseline after 3 months for most, but not all, procedures (Sanders et al., 2014).26Chan and colleagues (2013) investigated the impact of three different methods (i.e., keyboard-based, mouse-based, and paper-based documentation) of recording patient data on the accuracy and speed of clinical documentation of ophthalmology residents and fellows and found that sensitivity was highest for the keyboard-based method, while paper-based documentation performed better than the other methods in terms of positive ratio and documentation speed.27 Other studies have found associations between EHR implementation in some eye care practices and significantly longer nonclinical documentation times, as well as increases in the amount of time that eye care providers spend on clinical examinations (Chiang et al., 2013; Pandit and Boland, 2013). Finally, transitioning to an EHR system can have negative impacts on the ability of eye care providers to include drawings in patient records. The rate of drawing in EHRs is low, and EHR adoption can dramatically decrease the proportion of clinical records that include drawings (Lim et al., 2015; Sanders et al., 2013). Ophthalmologists have reported difficulties in using existing EHR drawing programs and have identified the inability to include drawings in patient records as a moderate to significant barrier to EHR adoption (Boland et al., 2013; Chiang et al., 2008, 2011).
Research also suggests that EHR adoption among eye care practices is associated with measurable, but rarely significant, economic impacts. A large multispecialty ophthalmic practice saw only nonsignificant changes in mean net monthly revenue and mean patient visits per month after
___________________
25 Complete examination includes general examination, slit-lamp examination, and fundus examination. Critical clinical findings were defined as the subset of ophthalmic examination elements believed to be required documentation for clinical evaluation of that disease.
26 Circulating operating room nurses and documentation time as a percentage of total procedure time remained elevated for cataract procedures, and the total documentation time remained significantly higher for all but the cornea- and glaucoma-related procedures.
27 Sensitivity was calculated by dividing the number of findings identified by subject that were truly present in the actual case by the total number of actual findings in the case. Positive ratio was calculated by dividing the number of findings identified by subject that were truly present in the actual case by the number of positive findings reported by subject. Documentation speed was calculated by dividing the documentation time of the entire case by the number of examination findings identified by subject (Chan et al., 2013).
transitioning to EHRs (Singh et al., 2015).28 EHR implementation in an academic medical center resulted in nonsignificant changes in the average number of ophthalmic surgical procedures per month and in operating room turnover time (Read-Brown et al., 2013).29 Other studies confirm the absence of statistically significant changes in clinical volume associated with EHR implementation in ophthalmology practices (Chiang et al., 2013; Redd et al., 2014). Lim and colleagues (2015) reported that EHR implementation in an academic ophthalmology practice resulted in a significant decrease in annual transcription costs and related cost-savings but that changes to annual clinical revenue per provider and to the number of patients per provider per year were nonsignificant, leading the authors to conclude that the study had not demonstrated a clear financial gain associated with EHR implementation.30
According to a national survey, a majority of physicians consider the large initial costs and uncertain return on investment (ROI) to be barriers to EHR adoption (DesRoches et al., 2008). These concerns are not unfounded. For example, the costs associated with implementation of a customized EHR system at the Cole Eye Institute included $1,571,864 in capital costs as well as $1,160,694 in personnel and ongoing costs and $1,514,334 in operating costs in 2011 (Chiang et al., 2013). Adler-Milstein and colleagues (2013) found that just 27 to 33 percent of specialty practices participating in the Massachusetts eHealth Collaborative projected a positive 5-year ROI; on average, the participating specialty practices expected to lose $50,722 per physician over 5 years. However, ROI varied considerably with practice size and type, with primary care practices showing smaller losses, practices with six or more physicians showing positive ROI, and ROI improving for all practices over longer periods. Other studies have found that EHR implementation in general and also the particular functions of an EHR system (e.g., computerized provider order entry), are associated with long-term cost-savings, improvements in medication safety, reductions in medical error, and benefits to society (Charles et al., 2014; Forrester et al., 2014; Grieger et al., 2007; Kaushal et al., 2006; Nuckols et al., 2014; Patil et al., 2008).
Increasingly, EHR systems designed for general use by health care institutions are becoming able to support the unique requirements of vision care, which previously hindered widespread implementation (Chiang et al.,
___________________
28 Total net fiscal revenue declined by $44,372 per month (median value) over the 24-month study period. Total patient volume increased by 217 visits per month (mean value).
29 Ophthalmic surgical procedures per month decreased after EHR implementation (14.9 to 14.2 among 25 stable providers), while operating room turnover time decreased (17.3 minutes to 15.6 minutes for the 4- to 12-month period after implementation).
30 Reduced transcription costs resulted in cost-savings of $188,951 over a 4-year post-EHR implementation period.
2011). For example, some current EHR systems can incorporate images or add sketches of the eye, which many ophthalmologists and optometrists rely on in assessing patient records. Health information technology can also simplify care delivery and patient adherence to treatment protocols. For example, one study found that embedding a referrals tool into EHR systems can improve the quality and frequency of referral-related communications between primary care providers and specialists (Gandhi et al., 2008). It can potentially encourage at-risk patients to seek preventive eye care and adhere to recommended guidelines for receiving comprehensive eye examinations. There are also other strategies for improving patient care; for example, electronic dosing aids with audiovisual reminders, provided as part of a multifaceted intervention, can increase patient adherence to glaucoma medication regimens, and telephone and text message reminders can improve adherence to follow-up care (Lin and Wu, 2014; Okeke et al., 2009).
Although health informatics has the potential to improve the delivery of eye care and thus improve patient outcomes, a number of challenges have limited the adoption of these tools, especially in solo practices. Key among these is the limited interoperability among EHRs and other sources of patient data.31 The Office of the National Coordinator for Health Information Technology (ONC) has reported that claimed barriers to interoperability include the “use of different technical standards, lack of business incentives that can lead vendors and providers to block the transmission of health information to other vendors and providers, deficits in trust, and differences in state laws and regulations that make it difficult to share health information across state lines” (ONC, 2015). Other barriers to interoperability can include negative impacts of EHR use on provider workflow, and challenges in maintaining the security of patient information (HITPC, 2015). The potential of interoperable EHRs to enhance care coordination and collaboration may hold special value for eye and vision care, where several providers—including ophthalmologists and optometrists, primary care providers, multidisciplinary vision rehabilitation teams, and public health workers—can contribute to a single patient treatment plan. Research is needed to identify strategies to promoting interoperability of EHR systems and other sources of patient data used by providers involved in eye and vision care.
___________________
31 Interoperability is “the ability of two or more systems to exchange information and the ability of those systems to use the information that has been exchanged without special effort” (HITPC, 2015, p. 4).
WORKFORCE TRAINING AND EDUCATION TO PROMOTE EYE AND VISION HEALTH
Improving access to high-quality care requires well-educated workforces across a variety of fields that are familiar with each other’s expertise and capabilities. Eye care providers must be able to communicate with, educate, counsel, and treat a racially, ethnically, and culturally diverse patient population whose members speak multiple languages, occupy various socioeconomic positions, have different professional and educational backgrounds, possess diverse beliefs about health and health care, and suffer from eye diseases and comorbid conditions of varying complexity and severity. Unfortunately, a lack of trust, communication, and coordination among different primary medical personnel and ophthalmologists and optometrists may lead to miscommunication, poor health care decisions, medical errors, the unnecessary duplication of services, lost continuity, excess costs, low-quality (e.g., inappropriate and untimely) care, and suboptimal outcomes in health care (Elhauge, 2010; Enthoven, 2009; IOM, 2001).
In order to cultivate the practices and knowledge needed to meet the multidisciplinary needs of eye care patients and address fragmentation of the vision care system, eye care providers, medical professionals working in primary care and medical specialties, allied health professionals, social workers, and members of the public health workforce must all be aware of the services offered by other health care and public health disciplines and must engage cooperatively with professionals in those disciplines on the development and implementation of treatment plans. Achieving high levels of health care system performance and improved patient outcomes through changes to the culture, practices, and processes of the vision care system and workforce will also require exceptional leadership, ongoing and effective teamwork, and commitment to the principles and purpose of quality improvement. Thus, positive change demands a workforce that is culturally competent and diverse, trained in cross-disciplinary and integrative approaches to care, predisposed to both teamwork and leadership, and inclined to lead quality improvement efforts.
Interprofessional Education
Optometrists, ophthalmologists, and primary care clinicians who can collaborate with one another and with other professionals in allied health, public health, social services, and other relevant fields, are essential to integrated models of eye and vision care. Interprofessional education (IPE) is one strategy for developing a workforce with the competencies necessary to meet the demands of integrated care. According to WHO,
“[i]nterprofessional education occurs when students from two or more professions learn about, from and with each other to enable effective collaboration and improve health outcomes” (WHO, 2010, p. 10). This training is a “key step in moving health systems from fragmentation to a position of strength” (WHO, 2010, p. 10). IPE can take place during formal professional training, as part of continuing education or professional development or quality programs.
A review of studies comparing IPE modules in health professional education programs found a wide range of instructional models, objectives, and reported outcomes (Abu-Rish et al., 2012). Reeves and colleagues (2010) performed a synthesis of systematic reviews and found that, despite heterogeneity among interventions, IPE was frequently well received by students and had a positive impact on care quality patient outcomes. A systematic review found limited evidence that IPE improves patient outcomes, provider adherence to clinical guidelines, patient satisfaction, or clinical processes. However, the differences among interventions and a limited number of high-quality studies made it impossible to make broader conclusions about the value of IPE (Reeves et al., 2013). Studies on the impacts of IPE on students or practitioners of optometry or ophthalmology are very limited, but they indicate that IPE can have positive impacts on attitudes toward collaborative practice (Sheppard et al., 2015).
Despite uncertainty regarding their optimal design and implementation, IPE programs are broadly supported by numerous stakeholders in health care. The IOM report Health Professions Education: A Bridge to Quality asserts that interdisciplinary education is a key to fostering interdisciplinary practice (IOM, 2003a).32 WHO and the American Public Health Association (APHA) have identified interprofessional education as essential to promoting collaborative practice (APHA, 2008; WHO, 2010). IPE programs have been implemented by HRSA, the VA, and several medical centers (Bridges et al., 2011; Remington et al., 2006). The Liaison Committee on Medical Education (LCME) requires the curricula of M.D. programs to include training in “interprofessional collaborative skills” (LCME, 2015, p. 11). 33 The Council on Education for Public Health requires public health
___________________
32 Working in interdisciplinary teams was described as involving cooperation, collaboration, and integration of care. Interdisciplinary education was defined as “a group of students from the health-related occupations with different educational backgrounds learn[ing] and interact[ing] together during certain periods of their education in order to collaborate in providing health-related services” (IOM, 2003a, p. 79).
33 LCME Standard 7.9: “The faculty of a medical school ensure that the core curriculum of the medical education program prepares medical students to function collaboratively on health care teams that include health professionals from other disciplines as they provide coordinated services to patients. These curricular experiences include practitioners and/or students from the other health professions” (LCME, 2015, p. 11).
schools to “function as a collaboration of disciplines” and to “provide a special learning environment that supports interdisciplinary communication” (CEPH, 2011, p. 2). The Accreditation Council on Optometric Education (ACOE) does not explicitly require O.D. programs to include training in interprofessional education or other competencies to support collaborative practice (ACOE, 2014). Inclusion of training in interprofessional education in ACOE accreditation standards for O.D. programs could potentially foster collaboration in the eye care workforce.
Eye and Vision Health in Medical and Population Health Education
All physicians are generally trained in the fundamentals of ophthalmic diseases and examination. A recent survey by Shah and colleagues reported that 95 percent, or 104 of the 109 osteopathic and allopathic schools of medicine who responded to the survey, conduct preclinical didactics within the first 2 years and 84 percent, or 92 schools, provide specific training related to the ophthalmic examination (Shah et al., 2014). However, additional training beyond this initial exposure varies with only 18 percent of schools requiring a clinical rotation in ophthalmology (Shah et al., 2014).
There is documented erosion in ophthalmic training in general medical education (Quillen et al., 2005; Shah et al., 2014). The accrediting body for schools of medicine, the Liaison Committee on Medical Education, does not specify ophthalmic training in its guidelines, making it difficult for ophthalmologists to increase their presence in the curriculum. The Centers for Disease Control and Prevention (CDC) notes a need to emphasize ophthalmic education in the medical curriculum (CDC, 2009). A survey conducted by residency program directors in primary care fields found that 64 percent of directors in family practice, 87.5 percent in internal medicine, and 86.3 percent in pediatrics believed that additional training in ophthalmology should be incorporated into their residency programs (Stern, 1995). Additionally, 90 percent of directors believed that less than 50 percent of entering residents met the minimum standard on medical student education set by the Association of University Professors of Ophthalmology (AUPO) (Stern, 1995). See Box 7-4 for AUPO policy on medical student education. Requiring a rotation in ophthalmology during medical school training could be proposed as a solution.
Some medical and dental schools are incorporating coursework with a focus on interprofessional competencies into their curriculums. For example, dental students at Harvard University participate in a primary care medical rotation, dental and medical students at the University of Michigan can elect to take interprofessional education courses, and dental and medical students at the University of Connecticut and Harvard Medical School follow identical curriculums during their first 2 years of study (HSDM,
2016; Kirk, 2011; University of Michigan, 2015a). To support integration efforts, many schools have also developed programs on interprofessional collaboration and education (e.g., University of Michigan, 2015a).
Because the public health workforce is responsible for a range of activities to support eye and vision health (see Chapter 5), public health professionals need to be knowledgeable about eye and vision health. To support efforts to improve eye and vision health, public health professionals need to be able to ascertain and communicate information about community health to providers, understand the roles that different eye care specialists and other health care providers play in advancing vision and eye health, and have a reasonable appreciation for the types of effective treatments or interventions that are available.
Population Health Training in Clinical Medicine
Over the past decade, the general landscape affecting health within communities has changed dramatically, emphasizing the health of individuals and populations. In 2008, Berwick and colleagues at the Institute for Healthcare Improvement (IHI) introduced the “Triple Aim,” which altered thinking about how to improve the U.S. health care system by suggesting three interdependent goals: improving the experience of care, improving the health of populations, and reducing per-capita costs of health care (Berwick et al., 2008). To achieve the “Triple Aim,” health care professionals
will need to understand the types of patient experiences and data that are relevant to population health activities and population health practitioners will need to understand the demands placed on health care providers and what they need in order to provide high-quality care. In terms of eye health, this means that health care providers need to understand what types of eye diseases and conditions pose the greatest risk of vision impairment within their communities, how to eliminate the most significant risk factors and foster the most significant protective factors (including health literacy and social determinants of health), and how this shapes patient interactions.
Some medical schools and other health professional curricula and practicums already expose students to population health theory and practices. For example, seven schools in 2003 received grants to become regional medicine-public health education centers and are expected to partner with local or state public health departments to improve population health and public health practice for current medical students (Maeshiro, 2008). The program has since expanded with a second cohort of medical schools in 2006 and then into other graduate medical education programs in 2008 (AAMC, 2015). Numerous medical schools offer dual degree programs in medicine and public health; a program in public health ophthalmology at Johns Hopkins University seeks to enable eye care professionals to apply public health concepts to blindness prevention (Johns Hopkins Medicine, 2016).
HRSA supports interprofessional education through grants to consortiums or partnerships identified in the Public Health Service Act (HRSA, 2016a). Grant-winning programs are supported by the Coordinating Center for Interprofessional Education and Practice, which serves as a focal point for efforts to increase collaborative, team-based care through program coordination, research, and data collection (HRSA, 2016a). Funded by the HRSA grant program, the National Center for Interprofessional Practice and Education (National Center) collects and offers educational resources on interprofessional education; manages a network of researchers, educators, and practitioners engaged in interprofessional education; and partners with researchers across the country to develop and evaluate interprofessional education programs (NCIPE, 2016a). Currently, only a few of the research efforts and educational materials available through the National Center concern the interprofessional education of eye care providers (NCIPE, 2016b,c). An increased emphasis by the National Center or other HRSA grant awardees on the education of the vision care workforce could provide valuable support for actions to improve the quality of eye care through workforce integration.
There are also growing links among educational institutions in public health, optometry, and ophthalmology. The schools and colleges of optometry include various aspects of public health (e.g., epidemiology,
biostatistics, health policy, ethics) in their curricula, and public health content is included on Part II of the National Board of Examiners in Optometry exam. In addition, education on public health is included in the curricula of medical schools and ophthalmology residencies. The American Board of Ophthalmology includes content about public health on the written and oral examinations of ophthalmologists for board certification. APHA’s Vision Care Section, whose members include optometrists, ophthalmologists, and other public health practitioners, was established to promote health and well-being with an emphasis on vision and eye health through interdisciplinary partnerships (APHA, 2016).
The American Optometric Student Association has partnered with Salus University to offer a scholarship to optometry students seeking to earn a master’s degree in public health at Salus University, and the Massachusetts College of Pharmacy and Health Sciences offers a dual degree program in optometry and public health (AOSA, 2016; MCPHS, 2016). The University of California, Los Angeles (UCLA), provides the opportunity for ophthalmology residents to obtain a doctorate in the UCLA Fielding School of Public Health during residency, and Johns Hopkins University offers a program in public health ophthalmology. Additionally, the emergence of initiatives in support of IPE signal new opportunities for enhancing interprofessional collaboration and improving population health.
Cultivating Leadership and Teamwork
Beyond knowledge of clinical and population health management, health care personnel and public health practitioners will need leadership abilities that allow them to cultivate trust between different groups and sectors and the capacity to form and articulate a shared vision to unite different stakeholders. Leadership programs exist for public health workers, optometrists, and ophthalmologists. For example, the Center for Health Leadership and Practice promotes leadership within the public health workforce through its National Leadership Academy programs (CHLP, 2016). AOA and the American Academy of Ophthalmology both offer professional leadership programs or training (AAO, 2016d; AOA, 2016b). The Robert Wood Johnson Foundation’s Interdisciplinary Research Leaders program provides interdisciplinary research teams with the leadership and technical skills necessary to pursue research to improve community health and promote health equity (RWJF, 2016). The successful cultivation of leadership and teamwork skills can positively impact patient and population health outcomes, by developing a workforce with the competencies necessary to pursue the difficult work of change and improvement at the level of health care systems.
Promoting Cultural Competency
Cultural competency helps build concordance between patients and health care providers by challenging providers to think outside of their strict biomedical constructs and respond to the cultural barriers that are inherent in their patients’ diverse belief systems and views about health, health care, and health care providers. For example, religious and spiritual beliefs, culturally traditional healing rituals, denial, and conflicting perspectives about the etiology of disease and the efficacy of long-term therapies can create roadblocks to the biomedical management of clinical conditions. The use of community health centers has been linked to improved health outcomes, through addressing health needs with culturally and linguistically appropriate services (Torres et al., 2014).34
The Association of American Medical Colleges, the Association of Schools and Colleges of Optometry, and the Association of Schools of Public Health have made efforts to incorporate cultural competency components into the curricula of member institutions. The Liaison Committee on Medical Education has required the faculty and students of medical schools to “demonstrate an understanding of the manner in which people of diverse cultures and belief systems perceive health and illness and respond to various symptoms, diseases, and treatments,” and it requires medical students to “recognize and appropriately address gender and cultural biases in health care delivery, while considering first the health of the patient” (AAMC, 2005, p. 1). Similarly, the Association of Schools and Colleges of Optometry have developed guidelines for culturally competent eye and vision care (ASCO, 2008); the Interprofessional Education Collaborative Expert Panel identified “embrac[ing] the cultural diversity and individual differences that characterize patients, populations, and the health care team” as a specific competency in health professional education (Interprofessional Education Collaborative Expert Panel, 2011, p. 19); and the Expert Panel on Cultural Competence Education for Students in Medicine and Public Health developed a set of common cultural competencies that medical and public health schools can use to “standardize curricula, benchmark student performance, and better prepare graduates for culturally competent practice” (Expert Panel on Cultural Competence Education for Students in Medicine and Public Health, 2012, p. 1).
Despite these broader gains, many gaps in the cultural competency of eye care remain. For example, only limited attempts have been made
___________________
34Torres et al. (2014) describe community health workers (CHWs) as front-line health workers who are members of the communities and provide community outreach programs or practices. The authors state that “CHWs are considered to have a deep understanding of the issues faced by these communities in accessing health and social services, and are able to offer linguistically and culturally appropriate assistance” (Torres et al., 2014, p. 75).
to tailor vision interventions to the unique cultural characteristics of the patient. Typically, these efforts in the past have been integrated into other programs. For example, a study utilizing the Spoken Knowledge in Low Literacy in Diabetes survey measured baseline knowledge of low-literate Hispanic-speaking individuals with diabetes. Diabetes baseline knowledge was weak in understanding the relationship to eye health, with only 31.5 percent of the study cohort correctly identifying the importance of seeing an eye doctor (Pena-Purcell and Boggess, 2014). The use of a 5-week diabetes education series conducted with pictorial materials and spoken Spanish significantly improved awareness, with 74.6 percent of participants identifying the importance of seeing an eye doctor post-intervention (Pena-Purcell and Boggess, 2014).
The continuing development, implementation, and evaluation of cultural competence programs, training modules, and educational tools designed to improve the affective dimensions of communication and clinical behavior along with increasing the diversity in the health care workforce can help increase patient–provider concordance, reduce implicit bias, and not only address the public health challenge of eliminating ethnic and cultural disparities, but also address contributors to the health outcomes, affecting the health of vulnerable populations (Elam and Lee, 2013; IOM, 2003b; Sabin and Greenwald, 2012). Including training in cultural competency in all medical, optometric, allied health, and public health educational programs could be an effective strategy for improving health system quality across all specialties and professions. Finally, while this discussion has focused on the development and training of the clinical eye and vision care workforce, similar interventions may be necessary to develop a patient-centered and culturally proficient public health workforce.
CONCLUSION
A comprehensive population health approach to reducing vision impairment and promoting eye and vision health requires, among other important components, the ability to deliver and measure high-quality care. Establishing clear messaging about who should receive what care and when is essential to educating not only the public, but also health care and population health fields. A single set of evidence-based guidelines, especially in the context of vision screenings and comprehensive eye examinations, that adhere to specific development standards can improve the uniformity and quality of patient care, establish a consistent baseline from which to measure improvement, and promote accountability for eye and vision health outcomes and care processes.
Quality improvement initiatives are important to help standardize care and address factors that contribute to unnecessary vision impairment and
inequitable health outcomes. Numerous governmental and health care entities are engaged in various CQI activities, but the evidence generated thus far has only led to a handful of nationally endorsed quality measures for specific diseases and conditions. Other measures related to vision screenings, general eye examinations, and vision rehabilitation are still lacking. Addressing the key research gaps and opportunities (see Box 7-5) could lead to additional improvements in the quality of eye and vision care. However, this research will require a broad set of surveillance and monitoring tools, which are currently limited (see Chapter 4).
Integrated models of care have the potential to improve detection and diagnosis of vision problems and subsequent referral to eye care providers. Patient-centered medical homes, ACOs, and other integrated care models provide lessons in collaboration and coordination that can inform efforts to integrate vision care, medicine, and public health. Investment in emerging technologies may also increase the accessibility of vision care for underserved populations. There is a need for cross-disciplinary education and training in the public health, eye and vision care, and broader clinical workforces, emphasizing cultural competency, leadership, teamwork, and awareness of the interrelations between eye and vision health, general health, and population health. The alignment of professional guidelines for eye and vision care, investment in CQI activities, and the integrated delivery of eye and vision care with the general field of medicine will require collaboration and commitment from a wide range of complementary, yet sometimes competing, stakeholders. Although this will be challenging, it
can be done and is necessary to improve the quality of eye and vision care and to promote the overall health of populations in the United States.
REFERENCES
AAMC (Association of American Medical Colleges). 2005. Cultural competence education. Washington, DC: AAMC. https://www.aamc.org/download/54338/data/culturalcomped.pdf (accessed June 21, 2016).
———. 2015. Regional medicine-public health education centers. https://www.aamc.org/initiatives/diversity/portfolios/cdc/aamcbased/rmphec (accessed April 7, 2016).
AANP (American Association of Nurse Practitioners). 2015. 2013–14 national nurse practitioner practice site census. https://www.aanp.org/press-room/press-releases/166-pressroom/2015-press-releases/1694-2013-14-national-nurse-practitioner-practice-site-census (accessed June 21, 2016).
AANPCP (AANP Certification Program). 2016. AANP certification program: Program description. http://www.aanpcert.org/ptistore/control/certs/program (accessed April 27, 2016).
AAO (American Academy of Ophthalmology). 2012a. Eye exams 101. http://www.aao.org/eye-health/tips-prevention/eye-exams-101 (accessed March 29, 2016).
———. 2012b. Pediatric eye evaluations Preferred Practice Pattern® guidelines. http://www.aao.org/Assets/7364f939-97b3-4c2e-948f-e6c3d78cb45d/636009832100970000/pediatric-eye-evaluations-ppp-pdf (accessed July 18, 2016).
———. 2013. Vision screening for infants and children. http://www.aao.org/clinical-statement/vision-screening-infants-children--2013 (accessed April 7, 2016).
———. 2015a. Policy statement: Frequency of ocular examinations. http://www.aao.org/clinical-statement/frequency-of-ocular-examinations--november-2009 (accessed June 21, 2016)
———. 2015b. Primary open-angle glaucoma Preferred Practice Pattern® guidelines. http://www.aaojournal.org/article/S0161-6420(15)01276-2/pdf (accessed July 18, 2016).
———. 2016a. AAO guidelines. http://www.aao.org/guidelines-browse?filter=preferredpracticepatternsguideline (accessed April 7, 2016).
———. 2016b. About the IRIS® registry. http://www.aao.org/iris-registry/about (accessed April 7, 2016).
———. 2016c. Comprehensive adult medical eye evaluation Preferred Practice Pattern® guidelines. http://www.aaojournal.org/article/S0161-6420(15)01269-5/pdf (accessed July 18, 2016).
———. 2016d. Leadership development program. http://www.aao.org/about/leadershipdevelopment/overview (accessed April 7, 2016).
———. 2016e. Preferred Practice Pattern® guidelines. http://www.aao.org/about-preferred-practice-patterns (accessed May 16, 2016).
AAO/AAO (American Academy of Ophthalmology/American Academy of Optometry). 2013. American Academy of Ophthalmology and American Academy of Optometry to explore educational opportunities to advance quality eye care. December 4. http://www.aao.org/newsroom/news-releases/detail/american-academy-of-ophthalmology-optometry-to-exp (accessed June 15, 2016).
AAP (American Academy of Pediatrics). 2002. Use of photoscreening for children’s vision screening. Pediatrics 109(3):524–525.
———. 2008a. Bright futures guidelines: Adolescence 11 to 21 years. https://brightfutures.aap.org/Bright%20Futures%20Documents/18-Adolescence.pdf (accessed July 18, 2016).
———. 2008b. Bright futures guidelines: Early childhood 1 to 4 years. https://brightfutures.aap.org/Bright%20Futures%20Documents/16-Early_Childhood.pdf (accessed July 18, 2016).
———. 2008c. Bright futures guidelines: Middle childhood 5 to 10 years. https://brightfutures.aap.org/Bright%20Futures%20Documents/17-Middle_Childhood.pdf (accessed July 18, 2016).
———. 2008d. Red reflex examination in neonates, infants, and children. Pediatrics 122(6): 1401–1404.
AAP, Committee on Practice and Ambulatory Medicine, Section on Ophthalmology, American Association of Certified Orthoptists, American Association for Pediatric Ophthalmology and Strabismus, and American Academy of Opthalmology. 2016. Visual system assessment in infants, children, and young adults by pediatricians. Pediatrics 137(1):1–3. http://pediatrics.aappublications.org/content/pediatrics/early/2015/12/07/peds.2015-3596.full.pdf (accessed June 20, 2016).
AAPA (American Academy of Physician Assistants). 2014. 2013 AAPA annual survey report. https://www.aapa.org/WorkArea/DownloadAsset.aspx?id=2902 (accessed June 21, 2016).
AAPOS (American Association for Pediatric Ophthalmology and Strabismus). 2014. Vision screening. http://www.aapos.org/terms/conditions/107 (accessed March 29, 2016).
Abu-Rish, E., S. Kim, L. Choe, L. Varpio, E. Malik, A. A. White, K. Craddick, K. Blondon, L. Robins, P. Nagasawa, A. Thigpen, L. L. Chen, J. Rich, and B. Zierler. 2012. Current trends in interprofessional education of health sciences students: A literature review. Journal of Interprofessional Care 26(6):444–451.
ACOE (Accreditation Council on Optometric Education). 2014. Accreditation manual: Professional optometric degree programs. St. Louis, MO: ACOE. http://www.aoa.org/Documents/students/od_manual_08_2014.pdf (accessed July 19, 2016).
ACP (American College of Physicians). 2016. What is the patient-centered medical home? https://www.acponline.org/practice-resources/business/payment/models/pcmh/understanding/what-pcmh (accessed June 21, 2016).
Adler-Milstein, J., C. E. Green, and D. W. Bates. 2013. A survey analysis suggests that electronic health records will yield revenue gains for some practices and losses for many. Health Affairs 32(3):562–570.
AHRQ (Agency for Healthcare Research and Quality). 2011. Coordinating care in the medical neighborhood: Critical components and available mechanisms. Washington, DC: AHRQ.
———. 2012. Screening for glaucoma: Comparative effectiveness. Washington, DC: AHRQ.
———. 2014a. Primary care workforce facts and stats no. 3. http://www.ahrq.gov/research/findings/factsheets/primary/pcwork3/index.html (accessed January 8, 2016).
———. 2014b. Research Initiative in Clinical Economics. http://www.ahrq.gov/professionals/clinicians-providers/resources/rice/index.html#Projects (accessed April 7, 2016).
———. 2016a. AHRQuality indicators—Introduction. http://www.qualityindicators.ahrq.gov (accessed May 5, 2016).
———. 2016b. Defining the PCMH. https://pcmh.ahrq.gov/page/defining-pcmh (accessed June 21, 2016).
AMA (American Medical Association). 2016. About the PCPI. https://www.ama-assn.org/ama/pub/physician-resources/physician-consortium-performance-improvement/about-pcpi.page? (accessed May 5, 2016).
AOA (American Optometric Association). 2002. Pediatric eye and vision examination. https://www.aoa.org/documents/CPG-2.pdf (accessed June 21,2016).
———. 2004. Care of the patient with amblyopia. http://www.aoa.org/documents/CPG-4.pdf (accessed July 18, 2016).
———. 2008. Care of the patient with learning related vision problems. http://www.aoa.org/documents/optometrists/CPG-20.pdf (accessed July 18, 2016).
———. 2011. Care of the patient with open angle glaucoma. http://www.aoa.org/documents/optometrists/CPG-9.pdf (accessed July 18, 2016).
———. 2014. Eye care of the patient with diabetes mellitus.http://www.aoa.org/Documents/EBO/EyeCareOfThePatientWithDiabetesMellitus%20CPG3.pdf (accessed July 18, 2016).
———. 2015a. 2015 AOA new technology & EHR survey. https://www.aoa.org/Documents/ric/2015%20EHR%20Executive%20Summary_FINAL.pdf (accessed July 18, 2016).
———. 2015b. AOA MORE. http://www.aoa.org/more?sso=y (accessed April 7, 2016).
———. 2015c. AOA optometric clinical practice guidelines. http://www.aoa.org/patients-andpublic/caring-for-your-vision/clinical-practice-guidelines?sso=y (accessed May 18, 2016).
———. 2015d. Comprehensive adult eye and vision examination. https://www.aoa.org/documents/CPG-1.pdf (accessed June 21, 2016).
———. 2016a. AOA’s 14 steps to evidence-based clinical practice guideline development. http://www.aoa.org/Documents/EBO/APPENDIX%20A-1%20AOA%2014%20Steps%20form.pdf (accessed June 1, 2016).
———. 2016b. Education & training. http://www.aoa.org/optometrists/education-andtraining?sso=y (accessed April 7, 2016).
———. 2016c. Limitations of Vision Screening Programs. http://www.aoa.org/patients-andpublic/caring-for-your-vision/comprehensive-eye-and-vision-examination/limitations-ofvision-screening-programs?sso=y (accessed July 18, 2016).
AOSA (American Optometric Student Association). 2016. Optometry education and public health. http://theaosa.org/about/optometry-education-and-public-health (accessed April 7, 2016).
APHA (American Public Health Association). 2008. Promoting interprofessional education. http://www.apha.org/policies-and-advocacy/public-health-policy-statements/policydatabase/2014/07/23/09/20/promoting-interprofessional-education (accessed June 21, 2016).
———. 2016. Vision care. https://www.apha.org/apha-communities/member-sections/vision-care (accessed June 21, 2016).
APPAP (Association of Postgraduate Physician Assistant Programs). 2016. Post graduate PA programs listings. http://appap.org/post-graduate-pa-programs/programs (accessed April 27, 2016).
ASCO (Association of Schools and Colleges of Optometry). 2008. Guidelines for culturally competent eye and vision care. Diversity Task Force, Association of Schools and Colleges of Optometry. http://opted.org/files/Guidelines_CulturallyCompetentEyeAndVisionCare.pdf (accessed June 21, 2016).
Ashwal, S., B. S. Russman, P. A. Blasco, G. Miller, A. Sandler, M. Shevell, and R. Stevenson. 2004. Practice parameter: Diagnostic assessment of the child with cerebral palsy: Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 62(6):851–863.
ASORN (American Society of Ophthalmic Registered Nurses). 2016. Why Certification for Registered Nurse in Ophthalmology (CRNO)? http://www.asorn.org/certification (accessed April 27, 2016).
Azuara-Blanco, A., K. Banister, C. Boachie, P. McMeekin, J. Gray, J. Burr, R. Bourne, D. Garway-Heath, M. Batterbury, R. Hernandez, G. McPherson, C. Ramsay, and J. Cook. 2016. Automated imaging technologies for the diagnosis of glaucoma: A comparative diagnostic study for the evaluation of the diagnostic accuracy, performance as triage tests and cost-effectiveness (GATE study). Health Technology Assessment 20(8):1–168.
Bailey, G. M. 2013. Certain ODs welcome to participate in new ASCRS integrated program. http://ophthalmologytimes.modernmedicine.com/ophthalmologytimes/news/user-defined-tags/ophthalmology/certain-ods-welcome-participate-new-ascrs-in (accessed May 17, 2016).
Balcer, L. J., D. H. Miller, S. C. Reingold, and J. A. Cohen. 2015. Vision and vision-related outcome measures in multiple sclerosis. Brain 138(1):11–27.
Berwick, D. M., T. W. Nolan, and J. Whittington. 2008. The triple aim: Care, health, and cost. Health Affairs 27(3):759–769.
Blumberg, D. M., R. Vaswani, E. Nong, L. Al-Aswad, and G. A. Cioffi. 2014. A comparative effectiveness analysis of visual field outcomes after projected glaucoma screening using SD-OCT in African American communities. Investigative Ophthalmology & Visual Science 55(6):3491–3500.
Blumenthal, D., and M. Tavenner. 2010. The “meaningful use” regulation for electronic health records. New England Journal of Medicine 363(6):501–504.
Boland, M. V., M. F. Chiang, M. C. Lim, L. Wedemeyer, K. D. Epley, C. A. McCannel, D. E. Silverstone, and F. Lum. 2013. Adoption of electronic health records and preparations for demonstrating meaningful use: An American Academy of Ophthalmology survey. Ophthalmology 120(8):1702–1710.
Bourne, R. R., K. A. French, L. Chang, A. D. Borman, M. Hingorani, and W. D. Newsom. 2010. Can a community optometrist-based referral refinement scheme reduce false-positive glaucoma hospital referrals without compromising quality of care? The Community and Hospital Allied Network Glaucoma Evaluation Scheme (CHANGES). Eye (London) 24(5):881–887.
Bridges, D. R., R. A. Davidson, P. S. Odegard, I. V. Maki, and J. Tomkowiak. 2011. Interprofessional collaboration: Three best practice models of interprofessional education. Medical Education Online 16:6035. doi: 10.3402/meo.v16i0.6035.
Brien Holden Vision Institute. 2016. Eye & vision care toolkit. https://academy.brienholdenvision.org/browse/resources/courses/eye-toolkit (accessed June 21, 2016).
Brouwers, M. C., M. E. Kho, G. P. Browman, J. S. Burgers, F. Cluzeau, G. Feder, B. Fervers, I. D. Graham, J. Grimshaw, S. E. Hanna, P. Littlejohns, J. Makarski, and L. Zitzelsberger. 2010. GREE II: Advancing guideline development, reporting and evaluation in health care. Canadian Medical Association Journal 182(18):E839–E842.
Burr, J. M., G. Mowatt, R. Hernandez, M. A. Siddiqui, J. Cook, T. Lourenco, C. Ramsay, L. Vale, C. Fraser, A. Azuara-Blanco, J. Deeks, J. Cairns, R. Wormald, S. McPherson, K. Rabindranath, and A. Grant. 2007. The clinical effectiveness and cost-effectiveness of screening for open angle glaucoma: A systematic review and economic evaluation. Health Technololgy Assessment 11(41):iii–iv, ix–x, 1–190.
Carrillo, J. E., V. A. Carrillo, R. Guimento, J. Mucaria, and J. Leiman. 2014. The New York-Presbyterian Regional Health Collaborative: A three-year progress report. Health Affairs 33(11):1985–1992.
CDC (Centers for Disease Control and Prevention). 2009. A public health approach to vision loss. http://www.cdc.gov/visionhealth/about/approach.htm (accessed October 7, 2015).
———. 2015. 2014 NEHRS data on electronic health record adoption and use among office-based physicians in the U.S., by state. http://www.cdc.gov/nchs/data/ahcd/nehrs/2015_web_tables.pdf (accessed July 18, 2016).
CEPH (Council on Education for Public Health). 2011. Accreditation criteria: Schools of public health. Silver Spring, MD: CEPH. http://ceph.org/assets/SPH-Criteria-2011.pdf (accessed June 21, 2016).
Chan, P., P. J. Thyparampil, and M. F. Chiang. 2013. Accuracy and speed of electronic health record versus paper-based ophthalmic documentation strategies. American Journal of Ophthalmology 156(1):165–172, e162.
Charles, K., M. Cannon, R. Hall, and A. Coustasse. 2014. Can utilizing a computerized provider order entry (CPOE) system prevent hospital medical errors and adverse drug events? Perspectives in Health Information Management 11:1b.
Chiang, M. F., M. V. Boland, J. W. Margolis, F. Lum, M. D. Abramoff, and P. L. Hildebrand. 2008. Adoption and perceptions of electronic health record systems by ophthalmologists: An American Academy of Ophthalmology survey. Ophthalmology 115(9):1591–1597; quiz 1597, e1591–e1595.
Chiang, M. F., M. V. Boland, A. Brewer, K. D. Epley, M. B. Horton, M. C. Lim, C. A. McCannel, S. J. Patel, D. E. Silverstone, L. Wedemeyer, and F. Lum. 2011. Special requirements for electronic health record systems in ophthalmology. Ophthalmology 118(8):1681–1687.
Chiang, M. F., S. Read-Brown, D. C. Tu, D. Choi, D. S. Sanders, T. S. Hwang, S. Bailey, D. J. Karr, E. Cottle, J. C. Morrison, D. J. Wilson, and T. R. Yackel. 2013. Evaluation of electronic health record implementation in ophthalmology at an academic medical center. Transactions of the American Ophthalmological Society 111:70–92.
CHLP (Center for Health Leadership & Practice). 2016. Home page: Center for Health Leadership & Practice. http://healthleadership.org (accessed August 15, 2016).
Chou, R., T. Dana, and C. Bougatsos. 2011. Screening for visual impairment in children ages 1–5 years: Update for the USPSTF. Pediatrics 127(2):e442–e479.
Chou, R., T. Dana, C. Bougatsos, S. Grusing, and I. Blazina. 2016a. Screening for impaired visual acuity in older adults: A systematic review to update the 2009 U.S. Preventive Services Task Force recommendation. Rockville, MD: AHRQ.
———. 2016b. Screening for impaired visual acuity in older adults: Updated evidence report and systematic review for the U.S. Preventive Services Task Force. JAMA 315(9):915–933.
Chous, L. M., and T. L. Knabel. 2014. Impact of eye exams in identifying chronic conditions. https://www.uhc.com/content/dam/uhcdotcom/en/Employers/PDF/EyeExamsChronicConditions.pdf (accessed July 18, 2016).
Cleveland Clinic. 2015. Diseases and conditions. https://my.clevelandclinic.org/health/diseases_conditions/hic_ocular_health/hic_visual_field_testing (accessed March 31, 2016).
CMS (Centers for Medicare & Medicaid Services). 2011. Accountable care organization 2012 program analysis: Quality performance standards narrative measure specification: Final report. https://www.cms.gov/medicare/medicare-fee-for-service-payment/sharedsavingsprogram/downloads/aco_qualitymeasures.pdf (accessed June 21, 2016).
———. 2012. Accountable care organization 2013 program analysis quality performance standards narrative measure specifications. https://www.cms.gov/medicare/medicarefee-for-service-payment/sharedsavingsprogram/downloads/aco_qualitymeasures.pdf (accessed June 21, 2016).
———. 2014. Accountable care organization 2014 program analysis quality performance standards narrative measure specifications. http://www.healthreform.ct.gov/ohri/lib/ohri/work_groups/quality/2014-09-03/aco_narrative_measures_specs_1.pdf (accessed June 21, 2016).
———. 2015a. Accountable care organization 2015 program analysis quality performance standards narrative measure specifications. https://www.cms.gov/medicare/medicare-feefor-service-payment/sharedsavingsprogram/downloads/ry2015-narrative-specifications.pdf (accessed June 21, 2106).
———. 2015b. Accountable care organizations (ACO). https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ACO/index.html?redirect=/Aco (accessed March 29, 2016).
———. 2015c. Next generation ACO model. https://innovation.cms.gov/initiatives/Next-Generation-ACO-Model (accessed April 7, 2016).
———. 2015d. Shared Savings Program. https://www.cms.gov/Medicare/Medicare-Fee-forService-Payment/sharedsavingsprogram/index.html (accessed March 29, 2016).
———. 2016a. Accountable care organization 2016 program quality measure narrative specifications. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/sharedsavingsprogram/Downloads/2016-ACO-NarrativeMeasures-Specs.pdf (accessed June 21, 2016).
———. 2016b. Pioneer ACO models. https://innovation.cms.gov/initiatives/Pioneer-ACOModel/index.html (accessed April 29, 2016).
Crews, J. E., C. F. Chou, M. M. Zack, X. Zhang, K. M. Bullard, A. R. Morse, and J. B. Saaddine. 2016. The association of health-related quality of life with severity of visual impairment among people aged 40–64 years: Findings from the 2006-2010 Behavioral Risk Factor Surveillance System. Ophthalmic Epidemiology 23(3):145–153.
DesRoches, C. M., E. G. Campbell, S. R. Rao, K. Donelan, T. G. Ferris, A. Jha, R. Kaushal, D. E. Levy, S. Rosenbaum, A. E. Shields, and D. Blumenthal. 2008. Electronic health records in ambulatory care—A national survey of physicians. New England Journal of Medicine 359(1):50–60.
Donahue, S. P., and J. B. Ruben. 2011. U.S. Preventive Services Task Force vision screening recommendations. Pediatrics 127(3):569–570.
Elam, A. R., and P. P. Lee. 2013. High-risk populations for vision loss and eye care underutilization: A review of the literature and ideas on moving forward. Survey of Ophthalmology 58(4):348–358.
Elhauge, E. 2010. Why we should care about health care fragmentation and how to fix it. In The fragmentation of U.S. health care: Causes and solutions, edited by E. Elhauge. New York: Oxford University Press.
Enthoven, A. C. 2009. Integrated delivery systems: The cure for fragmentation. American Journal of Managed Care 15(10 Suppl):S284–S290.
Expert Panel on Cultural Competence Education for Students in Medicine and Public Health. 2012. Cultural competence education for students in medicine and public health: Report of an expert panel. Washington, DC: Association of American Medical Colleges and Association of Schools of Public Health.
Fisher, E. S. 2008. Building a medical neighborhood for the medical home. New England Journal of Medicine 359(12):1202–1205.
Forrester, S. H., Z. Hepp, J. A. Roth, H. S. Wirtz, and E. B. Devine. 2014. Cost-effectiveness of a computerized provider order entry system in improving medication safety ambulatory care. Value Health 17(4):340–349.
Friedberg, M. W., E. C. Schneider, M. B. Rosenthal, K. G. Volpp, and R. M. Werner. 2014. Association between participation in a multipayer medical home intervention and changes in quality, utilization, and costs of care. JAMA 311(8):815–825.
Frohman, E. M., J. G. Fujimoto, T. C. Frohman, P. A. Calabresi, G. Cutter, and L. J. Balcer. 2008. Optical coherence tomography: A window into the mechanisms of multiple sclerosis. Nature Clinical Practice Neurology 4(12):664–675.
Gandhi, T. K., N. L. Keating, M. Ditmore, D. Kiernan, R. Johnson, E. Burdick, and C. Hamann. 2008. Improving referral communication using a referral tool within an electronic medical record. In Advances in patient safety: New directions and alternative approaches, edited by K. Henriksen, J. B. Battles, M. A. Keyes, and M. L. Grady. Rockville, MD: Agency for Healthcare Research and Quality.
Gangwani, R., W. W. Lai, R. Sum, S. M. McGhee, C. W. Chan, A. J. Hedley, and D. Wong. 2014. The incidental findings of age-related macular degeneration during diabetic retinopathy screening. Graefe’s Archive for Clinical and Experimental Ophthalmology 252(5):723–729.
Garg, S., and R. M. Davis. 2009. Diabetic retinopathy screening update. Clinical Diabetes 27(4):140–145.
Grieger, D. L., S. H. Cohen, and D. A. Krusch. 2007. A pilot study to document the return on investment for implementing an ambulatory electronic health record at an academic medical center. Journal of the American College of Surgeons 205(1):89–96.
Hartmann, E. E., G. E. Bradford, P. K. Chaplin, T. Johnson, A. R. Kemper, S. Kim, and W. Marsh-Tootle. 2006. Project Universal Preschool Vision Screening: A demonstration project. Pediatrics 117(2):e226–e237.
Hered, R. W., and D. L. Wood. 2013. Preschool vision screening in primary care pediatric practice. Public Health Reports 128(3):189–197.
HITPC (Health IT Policy Committee). 2015. Report to congress: Challenges and barriers to interoperability. https://www.healthit.gov/facas/sites/faca/files/HITPC_Final_ITF_Report_2015–12-16%20v3.pdf (accessed July 19, 2016).
Holley, C. D., and P. P. Lee. 2010. Primary care provider views of the current referral-to-eye-care process: Focus group results. Investigative Ophthalmology & Visual Science 51(4):1866–1872.
Holtzer-Goor, K. M., E. van Sprundel, H. G. Lemij, T. Plochg, N. S. Klazinga, and M. A. Koopmanschap. 2010. Cost-effectiveness of monitoring glaucoma patients in shared care: An economic evaluation alongside a randomized controlled trial. BMC Health Services Research 10(1):1–11.
Holtzer-Goor, K. M., E. J. van Vliet, E. van Sprundel, T. Plochg, M. A. Koopmanschap, N. S. Klazinga, and H. G. Lemij. 2016. Shared care in monitoring stable glaucoma patients: A randomized controlled trial. Journal of Glaucoma 25(4):e392–e400.
Hooker, R. S., and C. M. Everett. 2012. The contributions of physician assistants in primary care systems. Health & Social Care in the Community 20(1):20–31.
HRSA (Health Resources and Services Administration). 2011. Redesigning a system of care to promote QI. http://www.hrsa.gov/quality/toolbox/methodology/redesigningasystemofcare (accessed June 21, 2016).
———. 2016a. Interprofessional Education Coordinating Center for Interprofessional Education and Collaborative Practice. http://bhpr.hrsa.gov/grants/interprofessional/index.html (accessed April 7, 2016).
———. 2016b. Small Health Care Provider Quality Improvement Program. Funding opportunity number: HRSA-16-019. http://www.hrsa.gov/ruralhealth/programopportunities/fundingopportunities/?id=b5c3a786-4ffc-4e0a-bb2e-e7e3cc6b29a6 (accessed June 21, 2016).
HSDM (Harvard School of Dental Medicine). 2016. Case for action. http://oralhealth.hsdm.harvard.edu/home (accessed July 18, 2016).
Hsiao, C-J., E. Hing. 2014. Use and characteristics of electronic health record systems among office-based physician practices: United States, 2001–2013. NCHS Data Brief No. 143, January. http://www.cdc.gov/nchs/products/databriefs/db143.htm (accessed June 21, 2016).
Huang, X., and M. B. Rosenthal. 2014. Transforming specialty practice—The patient-centered medical neighborhood. New England Journal of Medicine 370(15):1376–1379.
Interprofessional Education Collaborative Expert Panel. 2011. Core competencies for interprofessional collaborative practice: Report of an expert panel. Washington, DC: Interprofessional Education Collaborative. https://www.aamc.org/download/186750/data/core_competencies.pdf (accessed July 18, 2016).
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
———. 2003a. Health professions education: A bridge to quality. Washington, DC: The National Academies Press.
———. 2003b. Unequal treatment: Confronting racial and ethnic disparities in health care. Washington, DC: The National Academies Press.
———. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
———. 2011b. Report brief: Clinical practice guidelines we can trust. http://www.nationalacademies.org/hmd/~/media/Files/Report%20Files/2011/Clinical-Practice-GuidelinesWe-Can-Trust/Clinical%20Practice%20Guidelines%202011%20Report%20Brief.pdf (accessed April 7, 2016).
Jain, S., S. Harnada, W. L. Membrey, and V. Chong. 2006. Screening for age-related macular degeneration using nonstereo digital fundus photographs. Eye 20(4):471–475.
Jamous, K. F., M. Kalloniatis, M. P. Hennessy, A. Agar, A. Hayen, and B. Zangerl. 2015. Clinical model assisting with the collaborative care of glaucoma patients and suspects. Clinical & Experimental Ophthalmology 43(4):308–319.
Johns Hopkins Medicine. 2016. Public Health Ophthalmology (PHO) program. http://www.hopkinsmedicine.org/wilmer/education/dana.html (accessed April 7, 2016).
Jones, S., and R. T. Edwards. 2010. Diabetic retinopathy screening: A systematic review of the economic evidence. Diabetic Medicine 27(3):249–256.
Karnon, J., C. Czoski-Murray, K. Smith, C. Brand, U. Chakravarthy, S. Davis, N. Bansback, C. Beverley, A. Bird, S. Harding, I. Chisholm, and Y. C. Yang. 2008. A preliminary model-based assessment of the cost-utility of a screening programme for early age-related macular degeneration. Health Technology Assessment 12(27):iii–iv, ix–124.
Kaushal, R., A. K. Jha, C. Franz, J. Glaser, K. D. Shetty, T. Jaggi, B. Middleton, G. J. Kuperman, R. Khorasani, M. Tanasijevic, and D. W. Bates. 2006. Return on investment for a computerized physician order entry system. Journal of the American Medical Informatics Association 13(3):261–266.
Kellogg Eye Center. 2015. Strabismus (misaligned eyes, crossed eyes, or wall eyes). http://www.kellogg.umich.edu/patientcare/conditions/strabismus.html (accessed March 29, 2016).
Kemper, A. R., A. Helfrich, J. Talbot, N. Patel, and J. E. Crews. 2011. Improving the rate of preschool vision screening: An interrupted time-series analysis. Pediatrics 128(5):e1279–e1284.
Kent, C. 2015. The IRIS registry: The first 18 months. The Review of Ophthalmology. November 5. http://www.reviewofophthalmology.com/content/c/58132 (accessed June 21, 2016).
Kern, L. M., A. Edwards, and R. Kaushal. 2014. The patient-centered medical home, electronic health records, and quality of care. Annals of Internal Medicine 160(11):741–749.
———. 2016. The patient-centered medical home and associations with health care quality and utilization: A 5-year cohort study. Annals of Internal Medicine 164(6):395–405.
Kirk, N. 2011. The curriculum: “A model for other schools to follow.” UConn Today, April 25. http://today.uconn.edu/2011/04/the-curriculum-a-model-for-other-schools-to-follow (accessed July 18, 2016).
Kneib, B. A. 2009. AOA Continuous Quality Improvement Process.
Kokkonen, E. W. J., S. A. Davis, H.-C. Lin, T. S. Dabade, S. R. Feldman, and A. B. Fleischer. 2013. Use of electronic medical records differs by specialty and office settings. Journal of the American Medical Informatics Association 20(e1):e33–e38.
Krein, S. L., S. J. Bernstein, C. E. Fletcher, F. Makki, C. L. Goldzweig, B. Watts, S. Vijan, and R. A. Hayward. 2008. Improving eye care for veterans with diabetes: An example of using the QUERI steps to move from evidence to implementation: QUERI series. Implementation Science 3:18.
Kung, J., R. R. Miller, and P. A. Mackowiak. 2012. Failure of clinical practice guidelines to meet Institute of Medicine standards: Two more decades of little, if any, progress. Archives of Internal Medicine 172(21):1628–1633.
Ladapo, J. A., S. M. Kymes, J. A. Ladapo, V. C. Nwosu, and L. R. Pasquale. 2012. Projected clinical outcomes of glaucoma screening in African American individuals. Archives of Ophthalmology 130(3):365–372.
LCME (Liaison Committee on Medical Education). 2015. Functions and structure of a medical school: Standards for accreditation of medical education programs leading to the M.D. degree. http://umsc.org.uic.edu/documents/LCME_standards.pdf (accessed June 21, 2016).
Lee, P. 2016. Visual acuity screening among asymptomatic older adults. JAMA 315(9):875–876.
Lesser, L. I., A. H. Krist, D. B. Kamerow, and A. W. Bazemore. 2011. Comparison between U.S. Preventive Services Task Force recommendations and Medicare coverage. Annals of Family Medicine 9(1):44–49.
Lim, M. C., R. P. Patel, V. S. Lee, P. D. Weeks, M. K. Barber, and M. R. Watnik. 2015. The long-term financial and clinical impact of an electronic health record on an academic ophthalmology practice. Journal of Ophthalmology 2015:329819.
Lin, H., and X. Wu. 2014. Intervention strategies for improving patient adherence to followup in the era of mobile information technology: A systematic review and meta-analysis. PLoS ONE 9(8):e104266.
Lowry, E. A., W. Wang, and O. Nyong’o. 2015. Objective vision screening in 3-year-old children at a multispecialty practice. Journal of the American Association for Pediatric Ophthalmology and Strabismus 19(1):16–20.
Lum, F., R. S. Feder, S. D. McLeod, and D. W. Parke, II. 2016. The preferred practice pattern guidelines in ophthalmology. Ophthalmology 123(5):928–929.
Maeshiro, R. 2008. Responding to the challenge: Population health education for physicians. Academic Medicine 83(4):319–320.
Mandalos, A., R. Bourne, K. French, W. Newsom, and L. Chang. 2012. Shared care of patients with ocular hypertension in the Community and Hospital Allied Network Glaucoma Evaluation Scheme (CHANGES). Eye 26(4):564–567.
McLeod, H., H. Dickinson, I. Williams, S. Robinson, and J. Coast. 2006. Evaluation of the chronic eye care services programme: Final report. Birmingham, UK: Health Services Management Centre, University of Birmingham. www.eyecare.nhs.uk/uploads/ChronicEyecareServicesProgramme.doc (accessed June 21, 2016).
MCPHS (Massachusetts College of Pharmacy and Health Sciences). 2016. Optometry/public health (O.D./M.P.H.). http://www.mcphs.edu/Academics/Programs/Optometry%20OD%20MPH (accessed April 7, 2016).
Medline Plus. 2015a. Extraocular muscle function testing. https://www.nlm.nih.gov/medlineplus/ency/article/003397.htm (accessed April 20, 2016).
———. 2015b. Refraction test. https://www.nlm.nih.gov/medlineplus/ency/article/003844.htm (accessed April 20, 2016).
———. 2015c. Slit-lamp exam. https://www.nlm.nih.gov/medlineplus/ency/article/003880.htm (accessed April 20, 2016).
———. 2015d. Tonometry. https://www.nlm.nih.gov/medlineplus/ency/article/003447.htm (accessed April 20, 2016).
———. 2015e. Visual acuity test. https://www.nlm.nih.gov/medlineplus/ency/article/003396.htm (accessed April 20, 2016).
———. 2015f. Visual field. https://www.nlm.nih.gov/medlineplus/ency/article/003879.htm (accessed April 20, 2016).
Mehrotra, A., C. B. Forrest, and C. Y. Lin. 2011. Dropping the baton: Specialty referrals in the United States. The Milbank Quarterly 89(1):39–68.
Moyer, V. A. 2013. Screening for glaucoma: U.S. Preventive Services Task Force recommendation statement. Annals of Internal Medicine 159(7):484–489.
NCCPA (National Commission on Certification of Physician Assistants). 2016. Specialty Certificates of Added Qualification (CAQs). http://www.nccpa.net/Specialty-CAQs (accessed April 27, 2016).
NCIPE (National Center for Interprofessional Practice and Education). 2016a. Home page: National Center for Interprofessional Practice and Education. https://nexusipe.org (accessed June 21, 2016).
———. 2016b. Interprofessional professionalism collaborative. https://nexusipe.org/informing/resource-center/interprofessional-professionalism-collaborative (accessed June 21, 2016).
———. 2016c. University of Alabama at Birmingham: The role of interprofessional faculty development in improving collaborative practice behavior competencies. https://nexusipe.org/advancing/nexus-innovations-network/university-alabama-birmingham-role-interprofessional-faculty (accessed June 21, 2016).
NCQA (National Committee for Quality Assurance). 2014a. The future of patient-centered medical homes. Foundation for a Better Health Care System. https://www.ncqa.org/Portals/0/Public%20Policy/2014%20Comment%20Letters/The_Future_of_PCMH.pdf (accessed June 21, 2016).
———. 2014b. Standards and guidelines for NCQA’s patient-centered medical home (PCMH) 2014. http://www.acofp.org/acofpimis/Acofporg/Apps/2014_PCMH_Finals/Tools/1_PCMH_Recognition_2014_Front_Matter.pdf (accessed June 21, 2016).
———. 2016a. About NCQA. http://www.ncqa.org/about-ncqa (accessed May 5, 2016).
———. 2016b. Diabetes Recognition Program (DRP). http://www.ncqa.org/tabid/139/Default.aspx (accessed April 7, 2016).
———. 2016c. Patient-centered specialty practice (PCSP) clinician eligibility.http://www.ncqa.org/programs/recognition/practices/patient-centered-medical-home-pcmh/during-earn-itpcmh/other-pcmh-resources/pcsp-clinician-eligibility (accessed March 29, 2016).
———. 2016d. Patient-centered specialty practice recognition. http://www.ncqa.org/programs/recognition/practices/patient-centered-specialty-practice-pcsp (accessed April 29, 2016).
———. 2016e. Standards and guidelines for PCSP 2016 recognition. Washington, DC: NCQA.
NEI (National Eye Institute). 2016. What is a comprehensive dilated eye exam? https://nei.nih.gov/healthyeyes/eyeexam (accessed March 29, 2016).
Nelson, K. M., C. Helfrich, H. Sun, P. L. Hebert, C. F. Liu, E. Dolan, L. Taylor, E. Wong, C. Maynard, S. E. Hernandez, W. Sanders, I. Randall, I. Curtis, G. Schectman, R. Stark, and S. D. Fihn. 2014. Implementation of the patient-centered medical home in the Veterans Health Administration: Associations with patient satisfaction, quality of care, staff burnout, and hospital and emergency department use. JAMA Internal Medicine 174(8):1350–1358.
NICHQ (National Institute for Children’s Health Quality). 2015. Reducing vision impairment in young children is focus of new initiative. http://www.nichq.org/news%20and%20events/news/vision_impairment_project (accessed April 7, 2016).
Nielsen, M., L. Buelt, K. Patel, and L. M. Nichols. 2016. The patient-centered medical home’s impact on cost and quality: Annual review of evidence 2014-2015. Washington, DC: Patient-Centered Primary Care Collaborative.
NQF (National Quality Forum). 2016a. Comprehensive diabetes care: Eye exam (retinal) performed. http://www.qualityforum.org/QPS/0055 (accessed April 29, 2016).
———. 2016b. Comprehensive diabetes care: Hemoglobin a1c (hba1c) poor control (>9.0%). http://www.qualityforum.org/QPS/0059 (accessed April 29, 2016).
———. 2016c. Improving healthcare quality. http://www.qualityforum.org/Setting_Priorities/Improving_Healthcare_Quality.aspx (accessed May 5, 2016).
Nuckols, T. K., C. Smith-Spangler, S. C. Morton, S. M. Asch, V. M. Patel, L. J. Anderson, E. L. Deichsel, and P. G. Shekelle. 2014. The effectiveness of computerized order entry at reducing preventable adverse drug events and medication errors in hospital settings: A systematic review and meta-analysis. Systematic Reviews 3:56.
Okeke, C. O., H. A. Quigley, H. D. Jampel, G. S. Ying, R. J. Plyler, Y. Jiang, and D. S. Friedman. 2009. Interventions improve poor adherence with once daily glaucoma medications in electronically monitored patients. Ophthalmology 116(12):2286–2293.
ONC (Office of the National Coordinator for Health Information Technology). 2015. Federal health IT strategic plan 2015–2020. https://www.healthit.gov/sites/default/files/9-5-federalhealthitstratplanfinal_0.pdf (accessed July 18, 2016).
Pandit, R. R., and M. V. Boland. 2013. The impact of an electronic health record transition on a glaucoma subspecialty practice. Ophthalmology 120(4):753–760.
Parke, D. W., Ii, M. X. Repka, and F. Lum. 2016. The US Preventive Services Task Force recommendation on vision screening in older adults: A narrow view. JAMA Ophthalmology 134(5):485–486.
Patil, M., L. Puri, and C. M. Gonzalez. 2008. Productivity and cost implications of implementing electronic medical records into an ambulatory surgical subspecialty clinic. Urology 71(2):173–177.
Pena-Purcell, N. C., and M. M. Boggess. 2014. An application of a diabetes knowledge scale for low-literate Hispanic/Latinos. Health Promotion Practice 15(2):252–262.
Petitti, D. B., S. M. Teutsch, M. B. Barton, G. F. Sawaya, J. K. Ockene, and T. DeWitt. 2009. Update on the methods of the U.S. Preventive Services Task Force: Insufficient evidence. Annals of Internal Medicine 150(3):199–205.
Phillips, R. L., M. Han, S. M. Petterson, L. A. Makaroff, and W. R. Liaw. 2014. Cost, utilization, and quality of care: An evaluation of Illinois’ Medicaid primary care case management program. Annals of Family Medicine 12(5):408–417.
Quillen, D. A., R. A. Harper, and B. G. Haik. 2005. Medical student education in ophthalmology: Crisis and opportunity. Ophthalmology 112(11):1867–1868.
Read-Brown, S., D. S. Sanders, A. S. Brown, T. R. Yackel, D. Choi, D. C. Tu, and M. F. Chiang. 2013. Time-motion analysis of clinical nursing documentation during implementation of an electronic operating room management system for ophthalmic surgery. AMIA Annual Symposium Proceedings 2013:1195–1204.
Redd, T. K., S. Read-Brown, D. Choi, T. R. Yackel, D. C. Tu, and M. F. Chiang. 2014. Electronic health record impact on pediatric ophthalmologists’ productivity and efficiency at an academic center. Journal of the American Association for Pediatric Ophthalmology and Strabismus 18(6):584–589.
Reeves, S., J. Goldman, A. Burton, and B. Sawatzky-Girling. 2010. Synthesis of systematic review evidence of interprofessional education. Journal of Allied Health 39(Suppl 1):198–203.
Reeves, S., L. Perrier, J. Goldman, D. Freeth, and M. Zwarenstein. 2013. Interprofessional education: Effects on professional practice and healthcare outcomes (update). Cochrane Database of Systematic Reviews 3:Cd002213.
Rein, D. B., J. S. Wittenborn, X. Zhang, T. J. Hoerger, P. Zhang, B. E. Klein, K. E. Lee, R. Klein, and J. B. Saaddine. 2012a. The cost-effectiveness of Welcome to Medicare visual acuity screening and a possible alternative Welcome to Medicare eye evaluation among persons without diagnosed diabetes mellitus. Archives of Ophthalmology 130(5):607–614.
Rein, D. B., J. S. Wittenborn, X. Zhang, M. Song, and J. B. Saaddine. 2012b. The potential cost-effectiveness of amblyopia screening programs. Journal of Pediatric Ophthalmology and Strabismus 49(3):146–155; quiz 145, 156.
Remington, T. L., M. A. Foulk, and B. C. Williams. 2006. Evaluation of evidence for interprofessional education. American Journal of Pharmaceutical Education 70(3):66.
Reuben, D. B., D. A. Ganz, C. P. Roth, H. E. McCreath, K. D. Ramirez, and N. S. Wenger. 2013. The effect of nurse practitioner co-management on the care of geriatric conditions. Journal of the American Geriatrics Society 61(6):857–867.
Roblin, D. W., D. H. Howard, E. R. Becker, E. Kathleen Adams, and M. H. Roberts. 2004. Use of midlevel practitioners to achieve labor cost savings in the primary care practice of an MCO. Health Services Research 39(3):607–626.
Rosenthal, M. B., M. K. Abrams, A. Bitton, and The Patient Centered Medical Home Evaluators’ Collaborative. 2012. Recommended core measures for evaluating the patient-centered medical home: Cost, utilization, and clinical quality. http://www.commonwealthfund.org/~/media/Files/Publications/Data%20Brief/2012/1601_Rosenthal_recommended_core_measures_PCMH_v2.pdf (accessed June 20, 2016).
Rosland, A. M., K. Nelson, H. Sun, E. D. Dolan, C. Maynard, C. Bryson, R. Stark, J. M. Shear, E. Kerr, S. D. Fihn, and G. Schectman. 2013. The patient-centered medical home in the Veterans Health Administration. American Journal of Managed Care 19(7): e263–e272.
Rupp, J. D. 2016. The 8 point eye exam. American Academy of Ophthamology, May 24. http://www.aao.org/young-ophthalmologists/yo-info/article/how-to-conduct-eight-point-ophthalmology-exam (accessed April 20, 2016).
RWJF (Robert Wood Johnson Foundation). 2012. Improving the science of continuous quality improvement program and evaluation: An RWJF national program. http://www.rwjf.org/content/dam/farm/reports/program_results_reports/2012/rwjf73230 (accessed July 18, 2016).
———. 2016. Interdisciplinary research leaders. http://www.rwjf.org/en/library/fundingopportunities/2016/interdisciplinary-research-leaders.html (accessed April 7, 2016).
Sabin, J. A., and A. G. Greenwald. 2012. The influence of implicit bias on treatment recommendations for 4 common pediatric conditions: Pain, urinary tract infection, attention deficit hyperactivity disorder, and asthma. American Journal of Public Health 102(5):988–995.
Sanders, D. S., D. J. Lattin, S. Read-Brown, D. C. Tu, D. J. Wilson, T. S. Hwang, J. C. Morrison, T. R. Yackel, and M. F. Chiang. 2013. Electronic health record systems in ophthalmology: Impact on clinical documentation. Ophthalmology 120(9):1745–1755.
Sanders, D. S., S. Read-Brown, D. C. Tu, W. E. Lambert, D. Choi, B. M. Almario, T. R. Yackel, A. S. Brown, and M. F. Chiang. 2014. Impact of an electronic health record operating room management system in ophthalmology on documentation time, surgical volume, and staffing. JAMA Ophthalmology 132(5):586–592.
Schaneman, J., A. Kagey, S. Soltesz, and J. Stone. 2010. The role of comprehensive eye exams in the early detection of diabetes and other chronic diseases in an employed population. Population Health Management 13(4):195–199.
Schmidt, P., M. Maguire, V. Dobson, G. Quinn, E. Ciner, L. Cyert, M. T. Kulp, B. Moore, D. Orel-Bixler, M. Redford, and G. S. Ying. 2004. Comparison of preschool vision screening tests as administered by licensed eye care professionals in the Vision in Preschoolers Study. Ophthalmology 111(4):637–650.
Shah, M., D. Knoch, and E. Waxman. 2014. The state of ophthalmology medical student education in the United States and Canada, 2012 through 2013. Ophthalmology 121(6):1160–1163.
Sheppard, K. D., C. R. Ford, P. Sawyer, K. T. Foley, C. N. Harada, C. J. Brown, and C. S. Ritchie. 2015. The interprofessional clinical experience: Interprofessional education in the nursing home. Journal of Interprofessional Care 29(2):170–172.
Singh, R. P., R. Bedi, A. Li, et al. 2015. The practice impact of electronic health record system implementation within a large multispecialty ophthalmic practice. JAMA Ophthalmology 133(6):668–674.
Sommer, A. 2016. The USPSTF position on vision screening of adults—Seeing is believing? JAMA Internal Medicine 176(4):438–439.
Starfield, B., C. B. Forrest, P. A. Nutting, and S. von Schrader. 2002. Variability in physician referral decisions. Journal of the American Board of Family Practice 15(6):473–480.
Stern, G. A. 1995. Teaching ophthalmology to primary care physicians. The Association of University Professors of Ophthalmology Education Committee. Archives of Ophthalmology 113(6):722–724.
Stevens, G. D., L. Shi, C. Vane, and A. L. Peters. 2014. Do experiences consistent with a medical-home model improve diabetes care measures reported by adult Medicaid patients? Diabetes Care 37(9):2565–2571.
Stevens, S., C. Gilbert, and N. Astbury. 2007. How to measure intraocular pressure: Applanation tonometry. Community Eye Health 20(64):74–75.
Torres, S., R. Labonté, D. L. Spitzer, C. Andrew, and C. Amaratunga. 2014. Improving health equity: The promising role of community health workers in Canada. Healthcare Policy 10(1):73–85.
UnitedHealthcare. 2016. UnitedHealthcare vision plan. https://www.opm.gov/healthcareinsurance/healthcare/plan-information/plan-codes/2016/brochures/UnitedHealth.pdf (accessed April 21, 2016).
University of Michigan. 2015a. Interprofessional education. https://interprofessional.umich.edu/about/courses/ (accessed April 7, 2016).
———. 2015b. Vision tests. http://www.uofmhealth.org/health-library/hw235693#aa24248 (accessed March 31, 2016).
USPSTF (U.S. Preventive Services Task Force). 2011. Visual impairment in children ages 1-5: Screening. http://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/visual-impairment-in-children-ages-1-5-screening (accessed April 7, 2016).
———. 2013. Final recommendation statement: Glaucoma screening.http://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/glaucomascreening (accessed April 25, 2016).
———. 2016a. About the USPSTF. http://www.uspreventiveservicestaskforce.org/Page/Name/about-the-uspstf (accessed September 1, 2016).
———. 2016b. Grade definitions: Grade definitions after July 2012. http://www.uspreventiveservicestaskforce.org/Page/Name/grade-definitions (accessed July 19, 2016).
———. 2016c. Screening for impaired visual acuity in older adults: U.S. Preventive Services Task Force recommendation statement. JAMA 315(9):908–914.
VA (U.S. Department of Veterans Affairs). 2016. Team-based care: PACT. http://www.va.gov/HEALTH/services/primarycare/pact/team.asp (accessed June 30, 2016).
van Vliet, E. J., W. Sermeus, C. M. van Gaalen, J. C. Sol, and J. M. Vissers. 2010. Efficacy and efficiency of a lean cataract pathway: A comparative study. Quality & Safety in Health Care 19(6):e13.
Voyatzis, G., H. W. Roberts, J. Keenan, and M. S. Rajan. 2014. Cambridgeshire cataract shared care model: Community optometrist-delivered postoperative discharge scheme. British Journal of Ophthalmology 98(6):760–764.
Werner, R. M., A. Canamucio, J. A. Shea, and G. True. 2014. The medical home transformation in the Veterans Health Administration: An evaluation of early changes in primary care delivery. Health Services Research 49(4):1329–1347.
WHO (World Health Organization). 2010. Framework for action on interprofessional education & collaborative practice. Geneva: WHO. Available at http://apps.who.int/iris/bitstream/10665/70185/1/WHO_HRH_HPN_10.3_eng.pdf?ua=1 (accessed June 21, 2016).
Wiggins, M. N., R. D. Landes, S. D. Bhaleeya, and S. H. Uwaydat. 2013. Primary care physician’s knowledge of the ophthalmic effects of diabetes. Canadian Journal of Ophthalmology 48(4):10.1016/j.jcjo.2013.1003.1011.
Wu, A. M., C. M. Wu, B. K. Young, D. J. Wu, A. Chen, C. E. Margo, and P. B. Greenberg. 2015a. Evaluation of primary open-angle glaucoma clinical practice guidelines. Canadian Journal of Ophthalmology 50(3):192–196.
Wu, A. M., C. M. Wu, B. K. Young, D. J. Wu, C. E. Margo, and P. B. Greenberg. 2015b. Critical appraisal of clinical practice guidelines for age-related macular degeneration. Journal of Ophthalmology 2015:1–5.
Wu, C. M., A. M. Wu, B. K. Young, D. Wu, A. Chen, C. E. Margo, and P. B. Greenberg. 2015c. An evaluation of cataract surgery clinical practice guidelines. British Journal of Ophthalmology 99(3):401–404.
Wu, C. M., A. M. Wu, B. K. Young, D. J. Wu, C. E. Margo, and P. B. Greenberg. 2015d. An appraisal of clinical practice guidelines for diabetic retinopathy. American Journal of Medical Quality 31(4):370–375.
Ying, G. S., M. T. Kulp, M. Maguire, E. Ciner, L. Cyert, and P. Schmidt. 2005. Sensitivity of screening tests for detecting vision in preschoolers-targeted vision disorders when specificity is 94%. Optometry & Vision Science 82(5):432–438.
Ying, G. S., M. Maguire, G. Quinn, M. T. Kulp, and L. Cyert. 2011. ROC analysis of the accuracy of noncycloplegic retinoscopy, retinomax autorefractor, and suresight vision screener for preschool vision screening. Investigative Ophthalmology & Visual Science 52(13):9658–9664.
Young, B. K., C. M. Wu, A. M. Wu, C. E. Margo, and P. B. Greenberg. 2015. Are clinical practice guidelines for cataract and glaucoma trustworthy? American Journal of Medical Quality 30(2):188–190.
This page intentionally left blank.
























































