Summary1
This is the second and final report to examine and recommend ways to improve the process used to update the Dietary Guidelines for Americans (DGA). What foods should Americans eat to promote their health, and in what amounts? What is the scientific evidence that supports specific recommendations for dietary intake to reduce the risk of multifactorial chronic disease? These questions are critically important because dietary intake has been recognized to have a role as a key determinant of health. Some relationships between diet and health, such as under- or overconsumption of certain micronutrients, have been well established. For example, an individual whose diet lacks iron can develop iron-deficiency anemia. However, through years of scientific investigation in nutrition and health, an understanding that there are complex relationships between dietary intake and the risk of developing multifactorial chronic disease has been developing. Poor dietary habits have been associated with the increased prevalence of chronic diseases such as type 2 diabetes and cardiovascular disease in the United States. Likewise, poor-quality diets that result in energy imbalance can increase the risk of obesity. Diet is a multidimensional exposure, and metabolic responses to diet are varied. While the presence of a relationship between dietary habits and multifactorial chronic disease can be identified, the precise relationship between dietary patterns and health is complex, involving dynamic inter-
___________________
1 This summary does not include references. Citations for the findings presented in the summary appear in subsequent chapters of the report.
actions among physical, social, behavioral, genetic, environmental, and other determinants of health. Because of this complexity, the responses to the questions of what Americans should eat and the supporting scientific evidence are not always simple ones.
As the primary federal source of consistent, evidence-based information on dietary practices for optimal nutrition, the DGA have the promise to empower Americans to make informed decisions about what and how much they eat to improve health and reduce the risk of chronic disease. In addition, the DGA serve as the basis for the types and composition of food provided in government food programs such as the National School Lunch Program and the Special Supplemental Nutrition Program for Women, Infants, and Children. Additionally, the DGA can be used as a basis for the development of more healthful products by food manufacturers. The individual and population uses of the DGA have the combined potential to improve population health. Unfortunately, most Americans do not consume a diet fully consistent with the DGA.
The adoption and widespread translation of the DGA require that they be universally viewed as valid, evidence-based, and free of bias and conflicts of interest to the extent possible. This has not routinely been the case. The DGA have been challenged, with critics questioning the validity of the evidence assessments. This has raised concerns in Congress about the trustworthiness of the DGA. This report recommends changes to the DGA process to reduce and manage sources of bias and conflicts of interest, improve timely opportunities for engagement by all interested parties, enhance transparency, and strengthen the science base of the process.
To help Americans make healthful food choices, the U.S. Department of Agriculture (USDA) and the U.S. Department of Health and Human Services (HHS) are mandated by Congress in the National Nutrition Monitoring and Related Research Act of 1990 to jointly review and author the guidelines every 5 years through a multistep process to reflect “the preponderance of the scientific and medical knowledge which is current at the time the report is prepared.” The process to create the guidelines is not simple and has been modified as the science has evolved. In previous editions of the DGA, the process has begun with an assessment of relevant scientific data by a federal advisory committee selected and convened by USDA and HHS (see Figure S-1). This panel of nationally recognized experts, known as the Dietary Guidelines Advisory Committee (DGAC), independently evaluates the scientific evidence and makes recommendations to the departments about how the previous DGA could be revised. The conclusions of the DGAC are submitted to the secretaries of USDA and HHS in the form of a scientific report and are only advisory; they do not constitute draft policy. The DGAC Scientific Report serves as the scientific basis for the next edition of the DGA.

NOTES: Timeline based on the 2015–2020 DGA. “Month” values indicate the approximate number of months after release of the previous edition of the DGA. HHS = U.S. Department of Health and Human Services; USDA = U.S. Department of Agriculture.
CHARGE TO THE NATIONAL ACADEMIES OF SCIENCES, ENGINEERING, AND MEDICINE
More than 29,000 public comments were submitted in response to the Scientific Report of the 2015 DGAC both in support of and against the conclusions made. The predominant topic addressed in the public comments was added sugars, with suggestions ranging from overall limitations to “a focus on total calories and portion sizes.” The 2015 DGAC’s inclusion of sustainability concerns was also controversial. In response to these criticisms, Congress mandated that the National Academies of Sciences, Engineering, and Medicine (the National Academies) evaluate the entire
process used to develop the DGA. Specifically, the Consolidated Appropriations Act, 2016, calls for a review of the following (see Box 1-3 for the full Statement of Task):
- How the advisory committee selection process can be improved to provide more transparency, eliminate bias, and include committee members with a range of viewpoints;
- How the Nutrition Evidence Library (NEL) is compiled and utilized, including whether NEL reviews and other systematic reviews and data analysis are conducted according to rigorous and objective scientific standards;
- How systematic reviews are conducted on long-standing DGA recommendations, including whether scientific studies are included from scientists with a range of viewpoints; and
- How the DGA can better prevent chronic disease, ensure nutritional sufficiency for all Americans, and accommodate a range of individual factors, including age, gender, and metabolic health.
The National Academies appointed 14 members to the Committee to Review the Process to Update the Dietary Guidelines for Americans. Specifically, the task is to assess the process used to develop the guidelines and not evaluate the substance or use of the guidelines. A response to question 1, “How the advisory committee selection process can be improved to provide more transparency, eliminate bias, and include committee members with a range of viewpoints?” was published for the purpose of informing the 2020 cycle in a first short report, Optimizing the Process for Establishing the Dietary Guidelines for Americans: The Selection Process. This second report responds to the remaining questions through a comprehensive review of the process to update the DGA.
FINDINGS AND CONCLUSIONS
Upon evaluating the current process used to update the DGA, this National Academies committee found that the process has become more evidence-based over its more than 30-year history as demonstrated by the formal integration of food pattern modeling and the ability to conduct original systematic reviews. However, the entire process has not been comprehensively reconsidered in a manner that effectively allows it to adapt to changes in food diversity and chronic disease prevalences, while also protecting the integrity of the process. Specific to the process, the findings and conclusions of this National Academies committee are as follows:
- The purpose and target audiences of the DGA have not been consistently interpreted, giving rise to confusion.
- The juxtaposition of the 5-year DGA cycle and the 2-year DGAC term imposed by the National Nutrition Monitoring and Related Research Act and the Federal Advisory Committee Act constrain the overall system (e.g., time to complete tasks, structure). Additionally, because the DGAC has conducted all tasks of the scientific review, opportunities for a truly deliberative process with the nutrition community, technical experts, and the public are limited.
- Transparency of the overall process to update the DGA needs improvement. For example, what standards are used to translate the evidence into recommendations and why the final DGA deviate from the conclusions of the DGAC Scientific Report have not been clearly explained. The current process also lacks a diversity of viewpoints and mechanisms to be responsive to topics of high public interest.
- The methodological approaches to evaluating the scientific evidence require increased rigor to better meet current standards of practice. The 2010 and 2015–2020 DGA were based on four types of analyses: (1) original systematic reviews; (2) existing systematic reviews, meta-analyses, and reports; (3) food pattern modeling;2 and (4) descriptive data analyses. The basic steps for conducting each analysis are generally reasonable; however, there are many ways in which the analyses need to be strengthened. For example, the NEL systematic review protocol lacks a clear separation of functions between the primary actors: the DGAC and the NEL. Additionally, the procedure by which the NEL protocol is updated to take into account advances in systematic review methods is not clear.
- Several aspects of the current approach to the scientific review would benefit from revision. First, the long duration between systematic reviews on a topic under the current system often does not keep pace with the emerging science; thus, ongoing surveillance of the literature needs to be instituted. Second, food pattern modeling is generally well designed for the questions it is intended to answer related to the average American diet, but its applicability to those who follow a different consumption pattern has been limited. Lastly, the processes to identify nutrients
___________________
2 Food pattern modeling refers to analyses that incorporate various data inputs, constraints, goals, and assumptions to assess the nutrient content of various possible eating patterns based on typical choices within food groups.
-
of concern would benefit from further standardization.3 Further integration of biochemical and health-related data in a systematic and consistent manner in the classification of nutrients of public health concern is also important.
- There is a lack of mechanistic data to support incorporation of newly identified diet-related health conditions in future DGA. Analytic frameworks also are needed to guide topic selection and evidence review toward the synthesis and interpretation of analyses.
Collectively, these findings and conclusions suggest the integrity of the DGA is compromised and the ability to develop a full body of evidence on a continuous basis over time is limited. The process to update the DGA should be comprehensively redesigned to allow it to adapt to changes in needs, evidence, and strategic priorities.
VALUES OF AN EFFECTIVE PROCESS TO UPDATE THE DGA
Based on the key findings, this National Academies committee identified five values to improve the integrity of a process to develop credible and trustworthy guidelines:
- Enhance transparency.
- Promote diversity of expertise and experience.
- Support a deliberative process.
- Manage biases and conflicts of interest.
- Adopt state-of-the-art processes and methods.
The process to update the DGA is also time- and resource-intensive. To the extent possible, a more efficient use of resources ought to be considered to minimize duplication of efforts and simplify the DGA without endangering its integrity. Implementing these values in the process to update the DGA will require that significant changes be made, necessitating a commitment from both USDA and HHS to ultimately achieve sustained performance.
Transparency is vital to engendering trust in the process, and it provides assurance that decisions were made free of undue influences. Each step of the process needs to be documented and updated, and such documentation needs to be readily available to the public. Opportuni-
___________________
3 Nutrients of concern are nutrients that may be a substantial public health concern and are determined by evaluating the prevalence of nutrient inadequacies and excesses in the U.S. population and select population groups.
ties for meaningful public participation and engagement will need to be enhanced. However, in the steps of the process where public participation would be inappropriate, such as decision making, it will be critical to explain why key decisions were made.
Trustworthiness of the process can also be enhanced by increasing opportunities for stakeholder participation, particularly by involving a broad range of expertise and experience, at appropriate times throughout the process by which the DGA are produced. Encouraging participation from stakeholders who represent a wide variety of perspectives—including the public, academia and researchers, advocacy groups, professional organizations, the food sector, and federal agencies—is critical to fostering diversity. All stakeholders could provide input into the process; however, only experts as appointed by the secretaries of USDA and HHS meeting bias and conflict-of-interest criteria ought to be involved in decision-making processes.
A more deliberative approach can help a process adapt to dynamic shifts in the system in which it operates. Characteristics of a deliberative process include supporting adaptability, continuity, and continuous learning. The breadth and content of each required report could vary such that not all topics may require a detailed review every 5 years; only those topics with enough new data to generate a full review would be considered for in-depth evaluation in the next DGA cycle. Second, to facilitate a deliberative process, the DGA cycles need to be considered as a continuous activity to foster learning across cycles. Continuity also allows a strategic approach be developed to accomplish the goals and vision of the DGA. Third, the DGA process itself needs to continuously evolve and improve dynamically in response to advances in science.
An effective process also needs to ensure independence in decision making. The process redesign will need to align the roles and responsibilities at each step of the process with appropriate experts involved in decision making. Actual or perceived conflicts of interest—both financial and nonfinancial—will need to be eliminated to the extent possible or their effects minimized and managed.
Finally, scientific rigor needs to be maximized. The process by which the science is evaluated can be strengthened by (1) using validated, standardized processes and methods; and (2) using the most up-to-date data. Processes and actions ought to be based on the best available evidence, requiring that the types of analysis used be continuously improved and advanced.
BROADENING THE SCOPE OF THE DGA
A fundamental shift is required such that future DGA focus on the general public across the entire life span, and not just healthy Americans ages 2 years and older. Given the range of metabolic health and the prevalence of chronic diseases in the population, as well as the importance of nutrition to pregnant women and children from birth to 24 months,4 it is essential that the DGA be developed for all Americans whose health could benefit by improving diet. Numerous organizations have developed or endorsed population- or disease-specific guidelines. However, the DGA are not designed to adjudicate among them. Confusion regarding which guidelines to follow could be reduced by identifying areas of consistency among guidelines developed in a manner in line with the methods used in the DGA.
PROCESS REDESIGN
This National Academies committee concluded that process redesign for updating the DGA can improve transparency, promote diversity of expertise and experience, support a deliberative process, promote independence in decision making, and strengthen scientific rigor. If successfully implemented, these modifications collectively have the potential to help improve the credibility of the DGA and trustworthiness of the process. Redesign can also improve the agility of the process and promote continuity of focus in key areas. Redesign that allows for the on-demand acquisition of many resources and an expanded set of multidisciplinary experts can improve the efficiency of the process. Redesign can also address needs for improved continuity between DGA cycles in areas such as real-time monitoring and curation of new evidence, with a consistent focus on strategic objectives. A more agile, efficient, and effective process can improve the relevance and usefulness of the DGA, which may improve adherence to the guidelines.
This National Academies committee considers that the 5-year cycle time can be leveraged more effectively by redistributing the tasks of the DGAC (the aforementioned group of experts appointed to assess the science) to other entities. While separation of tasks adds additional components and potentially cost to the overall process, more targeted expertise can be dedicated to completing a specific task, resulting in higher-quality inputs into the synthesis of evidence, and more time for deliberations, stakeholder engagement, and transparency-related activities. This pro-
___________________
4 The Agricultural Act of 2014 mandates that pregnant women and children from birth to 24 months be included in the 2020–2025 DGA.
posed process redesign model also permits much of the context setting and evidence development to be accomplished early on in the process.
Recommendation 1. The secretaries of the U.S. Department of Agriculture (USDA) and the U.S. Department of Health and Human Services (HHS) should redesign the Dietary Guidelines for Americans (DGA) process to prioritize topics to be reviewed in each DGA cycle, and redistribute the current functions of the Dietary Guidelines Advisory Committee to three separate groups:
- Dietary Guidelines Planning and Continuity Group to monitor and curate evidence generation, to identify and prioritize topics for inclusion in the DGA, and to provide strategic planning support across DGA cycles;
- Technical expert panels to provide content and methodological consultation during evaluation of the evidence; and
- Dietary Guidelines Scientific Advisory Committee to interpret the scientific evidence and draw conclusions.
The redesign incorporates continuity across 5-year cycles, with some activities spanning across DGA cycles (see Figure S-2). A Dietary Guidelines Planning and Continuity Group (DGPCG) identifies topics and questions for review, as well as provides help with strategic planning. Subcommittees would be convened as needed to address specific topic areas. The redesign also creates an additional framework that would support the scientific needs of the process: technical expert panels (TEPs). The synthesis and interpretation of evidence, as well as development of conclusions, would be the primary focus of a Dietary Guidelines Scientific Advisory Committee (DGSAC). The secretaries of USDA and HHS would oversee the entire process.
The DGPCG is envisioned as a group of federal staff and nonfederal experts convened to perform the following three functions:
- Provide the secretaries of USDA and HHS with planning support that assures alignment with long-term strategic objectives spanning multiple DGA cycles.
- Identify and prioritize topics for the DGSAC to evaluate in subsequent DGA cycles.
- Oversee monitoring and surveillance for new evidence.
These functions are consistent with the conclusion that not all topics need to be fully reevaluated every 5 years. Strategic planning is needed
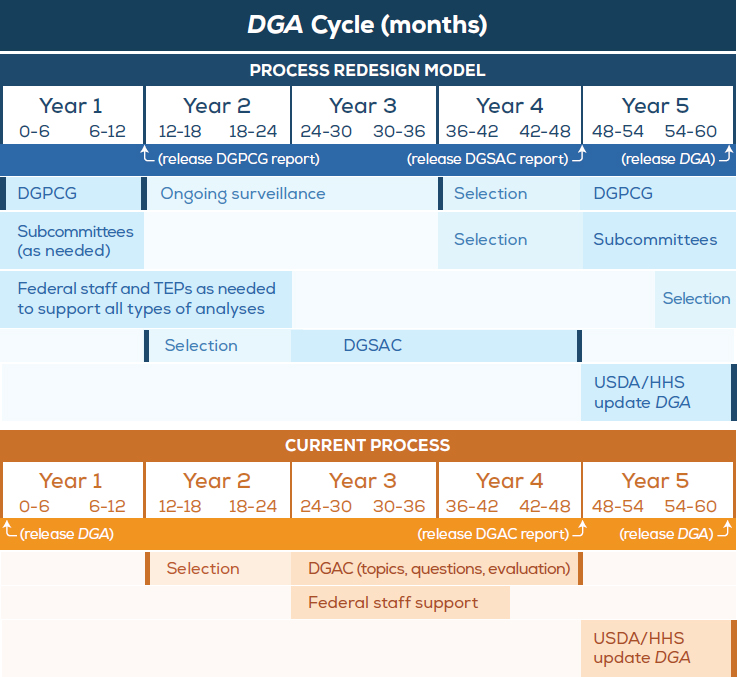
NOTES: Dark bars indicate opportunities for public comment and explanation of key decisions made. Darker shaded boxes indicate most active periods, while lighter shaded boxes denote potential times of less active engagement as needed. “Selection” refers to the selection of members for the respective groups. “Federal staff” includes those providing technical support such as the Nutrition Evidence Library staff and those conducting food pattern modeling and descriptive data analyses. DGA = Dietary Guidelines for Americans; DGAC = Dietary Guidelines Advisory Committee; DGPCG = Dietary Guidelines Planning and Continuity Group; DGSAC = Dietary Guidelines Scientific Advisory Committee; HHS = U.S. Department of Health and Human Services; TEP = technical expert panel; USDA = U.S. Department of Agriculture.
across DGA cycles. With respect to identifying and prioritizing topics, the DGPCG would be responsible for disclosing in a brief report the criteria and logic for the list of topics and associated research questions recommended. The DGPCG would also help oversee activities to monitor the scientific and public health literature to determine when enough
new evidence has been developed on a specific topic, or when a topic is of high enough public interest, to warrant review for potential inclusion in a future DGA cycle. Functions 1 and 3 require that the DGPCG not be time limited and that it operate across DGA cycles. It is likely that additional expertise will be needed during the deliberations of the DGPCG. For example, fully vetting topic considerations may require expertise not covered by DGPCG members. The DGPCG could seek supplemental expertise in a number of ways including commissioned papers, invited speakers, consultants, advisors, roundtables, or subcommittees, depending on the breadth and complexity of the topic. A good model to consider for identifying questions for topics with broad subject matter is the project to expand the DGA to include women who are pregnant and children from birth to 24 months. That project engaged with a broad number of stakeholders with specialized expertise to identify and develop topics and questions for systematic reviews, while separating the topic identification process from the evidence evaluation.
TEPs, inclusive of nonfederal and federal experts in the domains of relevant methodologies with a diversity of scientific viewpoints, are proposed by this National Academies committee as a mechanism to supplement the technical insights in the beginning stages of any type of analysis. The number of such TEPs will vary based on needs during each DGA cycle.
In the process redesign model, the DGSAC is charged with developing evidence-based conclusions for USDA and HHS to consider in the next DGA update. Specifically, the DGSAC would be charged with integrating all data inputs such as systematic reviews, food pattern modeling, and descriptive data analyses in order to develop its conclusions regarding diet and its relationship to health. The DGSAC will need to determine a priori standards for evidence it will consider. As needed, the DGSAC would also be able to identify and request a limited number of new analyses. The DGSAC would deliver a scientific report that would serve as the scientific foundation for the DGA Policy Report. The DGSAC would also be charged with identifying topics where more evidence is needed, and suggesting those topics for future DGA cycles. Members of the DGSAC would include subject matter experts, as well as experts in the methods being considered for use in that particular DGA cycle (e.g., systematic reviews, food pattern modeling). Collectively, the experts involved with the DGPCG, TEPs, and DGSAC would represent a wide range of expertise and experience.
The structure of the process redesign model allows each group to focus on a major task of the proposed process: topic identification, selection, and prioritization; data collection and evaluation; data synthesis, interpretation, and integration; and the update of the DGA. Because the
goals of topic identification, selection, and prioritization are different, it is this National Academies committee’s opinion that specific criteria need to be defined for each stage, and that the public ought to participate in topic nomination. As soon as the DGPCG prioritizes topics for a particular DGA cycle and the secretaries of USDA and HHS affirm the list, the next task of collecting and evaluating data would begin. Teams of federal methodologists would work with TEPs to begin conducting systematic reviews, food pattern modeling, and descriptive data analyses (e.g., NEL staff, nutritional epidemiologists, respectively), with the goal of having final results available for the DGSAC when it first convenes. The DGSAC would then independently evaluate the evidence and develop conclusions, consulting with appropriate methodologists as needed to understand the evidence.
The federal writing team—the group that updates the DGA based on the scientific report—needs to adhere to explicit and transparent standards for developing evidence-based guidelines and recommendations. These standards ought to be incorporated into the DGA process and updated to align with best practices in the field. To enhance the integrity of the process, every effort needs to be made to ensure that the DGA Policy Report is transparent about what decisions were made about the DGSAC’s conclusions, and the secretaries should explain why any deviations exist.
Recommendation 2. The secretaries of USDA and HHS should provide the public with a clear explanation when the DGA omit or accept only parts of conclusions from the scientific report.
This National Academies committee envisions the DGSAC as a federal advisory committee and the TEPs as ad hoc groups. Three options were considered for establishing the DGPCG: a federal advisory committee, a federal group, and a nongovernmental organization. Weighing the advantages and disadvantages reveals no perfect option. Establishing the DGPCG as a federal advisory committee ensures some mechanisms for objectivity and transparency but adds layers of complexity. A federal group would likely be the easiest to implement; however, it may not be viewed as independent. Although a nongovernmental organization could ensure transparency, it depends on identification of an influential, nonpartisan organization. This National Academies committee believes establishing the DGPCG as a federal advisory committee to be the most likely option to yield a trustworthy, dependable evaluation of the science. Regardless of which option is selected, the redesign will need to include experts with a diversity of scientific viewpoints.
STRENGTHENING ANALYSES AND ADVANCING METHODS USED
The DGA have to be based on the highest standards of scientific data and analyses to reach the most robust recommendations. The DGA require multiple sources of evidence to address the breadth of its scope. Data will need to come from varying study designs, such as randomized trials and observational studies. These aggregate data, analyzed with the most current methodology, provide complementary evidence to answer different inferential questions and inform various parts of the evidence base. Properly evaluating and calibrating results from a variety of data sources and methodological approaches is critical to understanding and interpreting the body of evidence to arrive at appropriate conclusions, as all study designs have innate limitations and can be susceptible to different types of bias. The dual challenge faced in developing the DGAC Scientific Report, and subsequently the DGA recommendations, is to properly assess the quality and interpret the results of studies available, and to use them appropriately in drawing conclusions about the body of evidence. Taking the limitations of evidence sources into account is crucial for building guidelines that are based on the totality of scientific evidence. Strengthening the current analyses depends on using the best data and the most rigorous processes and methods available. Advancing the evidence underpinning the DGA will also require integrating newer methods that help better elucidate and represent the complex systems involved.
Strengthening the NEL process for conducting de novo systematic reviews and identifying appropriate existing systematic reviews will require a multipronged approach. Clearly delineating the roles of the DGSAC and the NEL staff, as well as incorporating formal peer review, would ensure appropriate methods are used and would minimize the risk of bias in conducting systematic reviews. It is also critical to incorporate the appropriate expertise at specific steps in the protocol.
Recommendation 3. The secretary of USDA should clearly separate the roles of USDA Nutrition Evidence Library (NEL) staff and the Dietary Guidelines Scientific Advisory Committee (DGSAC) such that
- The NEL staff plan and conduct systematic reviews with input from technical expert panels, perform risk of bias assessment of individual studies, and assist the DGSAC as needed.
- The NEL systematic reviews are externally peer reviewed prior to being made available for use by the DGSAC.
- The DGSAC synthesizes and interprets the results of systematic reviews and draws conclusions about the entire body of evidence.
The NEL should also maintain state-of-the-art systematic review methods. By instituting ongoing training and collaboration, as well as a supportive methodological infrastructure to cultivate practitioners of systematic review with a nutrition focus, the NEL has the opportunity to become a leading evidence source for the nutrition community. One opportunity to review implementation of methods is to invite experts in systematic review methodology to periodically review the NEL process. The NEL can learn from other organizations in particularly challenging steps of systematic reviews, such as implementation of grading criteria and evaluation of evidence. Another opportunity for collaboration and alignment with best practices is in synthesizing and interpreting the body of evidence. These are subjective processes and require experience and expertise. Thus, standard and up-to-date approaches are necessary to account for the strengths and the limitations of included studies and to formulate high-quality, evidence-based conclusions.
Recommendation 4. The secretary of USDA should ensure all Nutrition Evidence Library (NEL) systematic reviews align with best practices by
- Enabling ongoing training of the NEL staff,
- Enabling engagement with and learning from external groups on the forefront of systematic review methods,
- Inviting external systematic review experts to periodically evaluate the NEL’s methods, and
- Investing in technological infrastructure.
Using high-quality systematic reviews from the literature whenever possible maximizes limited time and resources, as well as reduces duplication of efforts. However, this will require ongoing surveillance of the literature to ensure systematic reviews are up to date while at the same time leveraging resources.
Diet constitutes an extremely complex system of exposure that is known to influence health, and modeling can help to make sense of that complex system. More advanced food pattern modeling can increase the ability of the DGA to account for the complex systems involved and the variabilities in food composition and consumption. Food pattern models will be most useful as methods are strengthened to adapt to new areas of science, a better appreciation of the systems involved is formed,
more systems science methods become available, and technology becomes increasingly more sophisticated.
Recommendation 5. The secretaries of USDA and HHS should enhance food pattern modeling to better reflect the complex interactions involved, variability in intakes, and range of possible healthful diets.
The accuracy and efficiency of data analyses could be improved by standardizing and validating the processes used, both within and between DGA cycles to identify nutrients of concern. Standardization would lead to consistent development of quantitative thresholds of inadequacy or excess and the integration of other supportive evidence to identify a nutrient of concern. This consistency would facilitate comparisons of descriptive data analyses over time, benefiting practitioners, consumers, and the food sector.
Recommendation 6. The secretaries of USDA and HHS should standardize the methods and criteria for establishing nutrients of concern.
The questions asked by previous DGACs have been, by necessity, limited by the types of evidence, data, and methods available. Advancing the evidence base will require not only strengthening existing data and types of analyses but also including new sources of evidence.
A systems approach is recommended to account for and understand the interrelated factors at play in both population and individual health. The DGA can play a key role in advancing the understanding of the role of diet within the larger body of evidence on factors that affect health. Constructing systems maps can lead to new insights and advance knowledge of the pathways connecting diet and health. Systems thinking, when fully integrated into the DGA process and supported with systems mapping and modeling, has the potential to influence the DGA recommendations based on comprehensive knowledge of the relationships of interest between diet and health. Systems thinking can also inform the translation of the guidelines to maximize impact and identify relevant connections across stakeholders.
Recommendation 7. The secretaries of USDA and HHS should commission research and evaluate strategies to develop and implement systems approaches into the DGA. The selected strategies should then begin to be used to integrate systems mapping and modeling into the DGA process.
















