3
Process Redesign
This National Academies of Sciences, Engineering, and Medicine (the National Academies) committee considers that process redesign for updating the Dietary Guidelines for Americans (DGA)1 can improve transparency and stakeholder engagement, accelerate access to expertise and experience, promote independence in decision making, and enhance scientific rigor. If successfully implemented, these changes collectively have the potential to improve trustworthiness of the process to update the DGA. Redesign can also improve the agility of the process and provide for continuity of focus in key operational and strategic areas over multiple DGA cycles. For example, the DGA objective to promote health requires the engagement of many resources and an expanded set of multidisciplinary experts. Redesign that allows for on-demand acquisition of such resources can improve the efficiency of the process (e.g., specialized expertise in behavioral and implementation science, data science, technology, complex systems methods). Redesign can also address needs for improved continuity between DGA cycles in operational areas such as real-time monitoring and curation of new evidence, and maintaining a focus on strategic objectives that may span multiple DGA cycles.
A more agile and effective process can improve the relevance and usefulness of the DGA recommendations. The DGA cycle time has been 5 years per the National Nutrition Monitoring and Related Research
___________________
1 Refer to Chapter 1, Box 1-1, for an explanation of how the term DGA is used throughout this National Academies report.
Act. However, the process to update the DGA has occurred over a 3-year time period: 2 years for the work of the Dietary Guidelines Advisory Committee (DGAC), and 1 year for the generation of the DGA Policy Report. The remaining 2 years have been voids before the 3-year process is repeated. This National Academies committee believes that using the entire 5 years would provide the opportunity for redesigning the DGA process to become more agile, flexible, and effective. The model discussed below accomplishes these objectives by reducing the administrative and operational tasks of the DGAC. This is achieved by redistributing DGAC tasks to provide more time and dedicated expertise to focus on each task in the process.
Recommendation 1. The secretaries of the U.S. Department of Agriculture (USDA) and the U.S. Department of Health and Human Services (HHS) should redesign the Dietary Guidelines for Americans (DGA) process to prioritize topics to be reviewed in each DGA cycle, and redistribute the current functions of the Dietary Guidelines Advisory Committee to three separate groups:
- Dietary Guidelines Planning and Continuity Group to monitor and curate evidence generation, to identify and prioritize topics for inclusion in the DGA, and to provide strategic planning support across DGA cycles;
- Technical expert panels to provide content and methodological consultation during evaluation of the evidence; and
- Dietary Guidelines Scientific Advisory Committee to interpret the scientific evidence and draw conclusions.
MODEL PROCESS REDESIGN
The following process redesign model retains the components and subprocesses used for the 2015–2020 DGA, and reflects elements of the process instituted for the review of evidence targeted to pregnancy and infancy that began in 2012 (USDA, 2017). However, the proposed redesign redistributes the tasks among a revised set of groups instead of having all tasks supporting the scientific assessment being conducted by a single group (DGAC). Compared to the current process, separating the tasks allows for more targeted, dedicated expertise to complete a specific task, higher-quality inputs into the synthesis of evidence, and more time for deliberations, stakeholder engagement, and transparency-related activities. This redesign also permits much of the context setting and evidence development to be accomplished early in the process.
In the process redesign model, a Dietary Guidelines Planning and Continuity Group (DGPCG) is established to monitor new relevant scientific evidence, to identify topics and questions for review by the Dietary Guidelines Scientific Advisory Committee (DGSAC), as well as to provide support for DGSAC alignment with any strategic objectives that may span multiple cycles. Subcommittees would be convened as needed to address specific topic areas. The new DGPCG is envisioned to operate continually across DGA cycles, but would act primarily in the period before a DGSAC is convened and after the DGA Policy Report is updated. The redesign also creates an additional framework to improve support for the scientific needs of the process: technical expert panels (TEPs). The synthesis and interpretation of evidence and the development of conclusions would be the primary focus of the DGSAC. Each of these three entities is discussed in detail below. It will be important for some level of coordination to occur among the group. However, this National Academies committee believes that in order for the proposed redesign to be successfully implemented, specific details (e.g., how each entity operates and coordinates with each other) needs to be left to the secretaries of USDA and HHS and/or the entities themselves to decide.
The model process redesign is shown in Figure 3-1. It displays the new redesigned process as well as the current process on the 5-year DGA update timeline. This process redesign model will be referred to throughout the description of the proposed structure and workflow.
The following sections present the roles of each group. Composition of each group is also proposed and is summarized in Table 3-1.
Roles and Composition of the Dietary Guidelines Planning and Continuity Group (DGPCG)
The DGPCG is envisioned as a group of nonfederal experts and several federal staff convened to do the following:
- Provide the secretaries of USDA and HHS with planning support that assures alignment with long-term strategic objectives spanning multiple DGA cycles;
- Identify and prioritize topics for the DGSAC to evaluate in subsequent DGA cycles; and
- Oversee monitoring and surveillance for new evidence.
Strategic planning is needed across DGA cycles to introduce new, relevant topics while also ensuring that all DGA recommendations remain based on appropriate scientific evidence. As discussed in Chapter 2, not all topics need to be fully reevaluated every 5 years. The DGPCG would
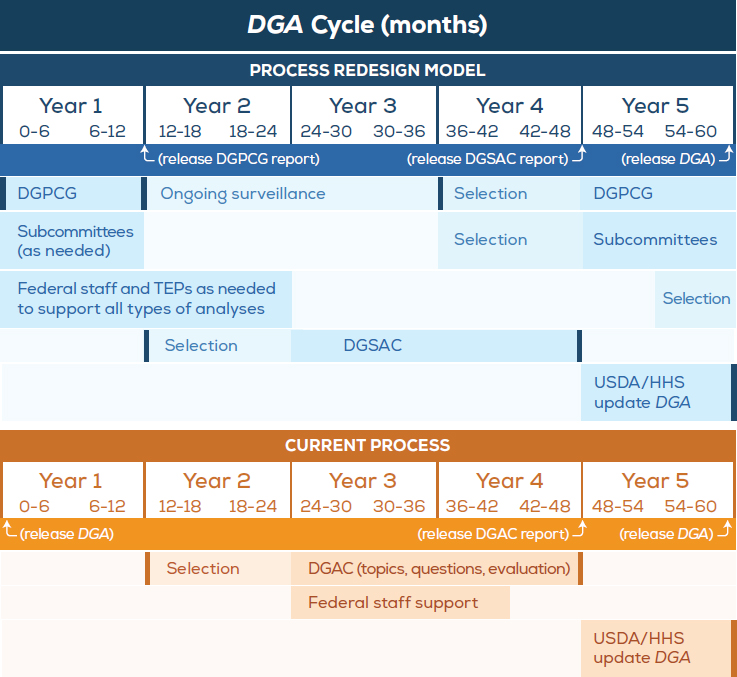
NOTES: Dark bars indicate opportunities for public comment and explanation of key decisions made. Darker shaded boxes indicate most active periods, while lighter shaded boxes denote potential times of less active engagement as needed. “Selection” refers to the selection of members for the respective groups. “Federal staff” includes those providing technical support such as the Nutrition Evidence Library staff and those conducting food pattern modeling and descriptive data analyses. DGA = Dietary Guidelines for Americans; DGAC = Dietary Guidelines Advisory Committee; DGPCG = Dietary Guidelines Planning and Continuity Group; DGSAC = Dietary Guidelines Scientific Advisory Committee; HHS = U.S. Department of Health and Human Services; TEP = technical expert panel; USDA = U.S. Department of Agriculture.
be responsible for publicly disclosing in a brief report the criteria and logic for how it prioritized topics and the associated research questions recommended. The DGPCG would also help to make the process agile. For example, by helping oversee activities to monitor the scientific and public health literature, the DGPCG can assist with determining when
TABLE 3-1 Composition of Groups in the Proposed Process to Update the DGA
| Group | Function | General Types of Expertise | Specific Types of Proficiencies |
|---|---|---|---|
| Dietary Guidelines Planning and Continuity Group (DGPCG) |
|
|
|
| Subcommittees as needed |
|
|
|
| Technical expert panels (TEPs) | |||
| Systematic reviews |
|
|
|
| Other types of analyses |
|
|
|
| Dietary Guidelines Scientific Advisory Committee (DGSAC) |
|
|
|
NOTE: CVD = cardiovascular disease; HHS = U.S. Department of Health and Human Services; NEL = Nutrition Evidence Library; P/B–24 = pregnant women and children from birth to 24 months; PICO = population, intervention/exposure, comparator, outcome of interest; USDA = U.S. Department of Agriculture.
enough new evidence has been developed on a specific topic to warrant review for potential inclusion in a future DGA cycle. The threshold for what constitutes “enough new evidence” ought to be an initial task of the DGPCG and updated as needed. The functions of supporting strategic planning and overseeing monitoring and surveillance for new evidence require that the DGPCG not be time limited and that it operate across DGA cycles.
The DGPCG will need to be composed of nongovernmental experts together with federal staff from USDA and HHS to fulfill its mission. Nongovernmental experts would include generalists in nutrition, experts in relevant stages of the life cycle, and experts in core competency areas such as research methods, public health, medicine, implementation science, and food production. The federal staff provide the governmental context and knowledge of the requirements of the federal food and nutrition programs. These core competencies can be supplemented by additional expertise required at any point through various mechanisms, ranging from membership on the DGPCG to advisors. It is envisioned that DGPCG members would serve across 5-year DGA cycles, rotating through staggered terms that could begin or end in the middle of a DGA cycle. It is the intent of this National Academies committee to leave the secretaries of USDA and HHS the flexibility to determine the size, expertise, member tenure, and ad hoc mechanisms for supplementing DGPCG intelligence needs, as well as the roles of the members themselves. However, the composition of the DGPCG should be selected based on the values and processes delineated in this National Academies committee’s first report, including identification and management of potential financial and nonfinancial conflicts of interest (see Boxes 1-4 and 2-3 for further discussion about identifying and managing biases and conflicts of interest).
Supplementing DGPCG Expertise
It is likely that additional expertise will be needed during the deliberations of the DGPCG. For example, fully vetting topic considerations may require expertise not covered by DGPCG members. The DGPCG could seek supplemental expertise in a number of ways, including commissioned papers, invited speakers, consultants or advisors, roundtables, or subcommittees, depending on the breadth and complexity of the topic. Full public access to any form of additional expertise solicited will be needed, and individuals providing such expertise would not be allowed to partake in DGPCG deliberations and decision making. A good model to consider for identifying questions related to topics with broad subject matter is the project to expand the DGA to include women who are pregnant and infants and children from birth to 24 months (P/B–24). Specifi-
cally, the P/B–24 project engaged with a broad number of stakeholders through both face-to-face workshops and online interactions to identify and develop topics and questions for systematic reviews. The work of the P/B–24 project separated the topic identification process from the evidence evaluation (see Chapter 5 for details).
The extension of the DGA to include recommendations for P/B–24 introduced a subpopulation for specific attention and a need for the DGPCG to obtain expertise in these domains on an ongoing basis. The DGPCG will have to consider a broad array of subjects for this demographic group ranging from the developmental needs of infants and varying nutritional requirements of children 0 to 24 months, to feeding behaviors and the roles of caretakers in feeding practices. Because the number of seats on the DGPCG itself will be limited to allow it to be operationally efficient, having experts in each P/B–24 subject is not feasible. A small number of experts in P/B–24 would be members of the DGPCG given the breadth of the subject matter. Additional expertise related to P/B–24 will be required to supplement the DGPCG using any of the aforementioned mechanisms, preferably through appointment of a subcommittee.
Roles and Composition of Technical Expert Panels (TEPs)
TEPs, including nonfederal and federal experts with a diversity of expertise and viewpoints, are proposed by this National Academies committee as a flexible mechanism to supplement the technical insights in beginning stages of any type of evidence analysis. The number and timing of such TEPs will vary based on needs during each DGA cycle. It is important to note that TEP members would provide their input on an individual basis; no set of collective advice from the TEP would be prepared. As such, TEPs would not need to convene in person. TEP members would be domain experts well versed in the specific research method being considered. Domain experts are people who are authorities on a specific area or topic. TEP members would be identified by USDA and HHS, and their selection could include consideration of the list of nominees suggested for membership on the DGPCG. Rules for bias and conflicts of interest, as well as scientific positions and views, would need to be created and made publicly available prior to identification.
TEPs would provide content and methodological consultation. For example, a TEP would work with the Nutrition Evidence Library (NEL) in the initial phase of conducting a systematic review. The TEPs would operationalize the research questions formulated by the DGPCG by helping to set the eligibility criteria for the literature review and to clarify systematic review question elements (i.e., population, intervention, comparison, and outcomes). TEPs could also help the NEL with techni-
cal issues during the review of the literature by the NEL staff, such as understanding the nuances of measurements, tests, and definitions. A TEP may be convened to provide input on additional systematic review questions identified by a newly constituted DGSAC. TEPs would not be responsible for conducting the systematic review or assessing the quality of the studies. TEP members could also be included in the peer review of the draft systematic review, but if they are, they would only be part of a much larger number of peer reviewers. The use of TEPs is modeled after the inclusion of domain expertise in the Agency for Healthcare Research and Quality (AHRQ) Evidence-based Practice Centers Program and the non-DGAC systematic reviews conducted by the NEL (see Chapter 6 for a full description of how technical experts are used in these processes2). In the AHRQ Evidence-based Practice Centers Program, TEPs typically interact with the systematic review team through one to three teleconferences over several weeks to 1 month.
Expert panels could also be employed to supplement the expertise of groups conducting other types of analyses such as food pattern modeling and descriptive data analyses. TEPs for these analyses are envisioned as supporting efforts such as verifying key assumptions in the development of food pattern and systems models or helping refine research questions related to data analyses.
Roles and Composition of the Dietary Guidelines Scientific Advisory Committee (DGSAC)
In the process redesign model, the DGSAC is charged with synthesizing and interpreting scientific evidence, as well as developing conclusions for USDA and HHS to consider in the DGA update (see Box 4-2 for descriptions of terminology). Specifically, the DGSAC would be charged with integrating all data inputs such as systematic reviews, food pattern modeling, and descriptive data analyses to develop its conclusions regarding diet and its relationship to health. To evaluate the science, the DGSAC will need to determine a priori the standards of evidence it will consider. As needed, the DGSAC would also be able to identify and request a limited number of new analyses and develop research recommendations for consideration by the DGPCG and the larger research community. The DGSAC would deliver a DGSAC scientific report that would serve as the scientific foundation for the DGA Policy Report. The DGSAC would also be charged with identifying topics where more evidence is needed, and
___________________
2 Groups of technical experts are used in the AHRQ Evidence-based Practice Centers Program and the non-DGAC NEL systematic review process. These experts are consulted for their subject-matter expertise and are not considered authors of the final publication.
suggesting those topics for future DGA cycles. Owing to the critical nature of this work and the need to ensure integrity of the process, it is essential that the DGSAC provide timely and ample opportunities and forums for stakeholders to provide insights and to engage in transparent and credible ways. However, the DGSAC will act independently in its interpretation of the scientific evidence and in its final conclusions.
As with previous DGACs, members of the DGSAC would include experts in domain subject matters to be reviewed to provide relevant knowledge and context for reviewing the evidence. The DGSAC will also need to include experts in the methods being considered for use in that particular DGA cycle (e.g., systematic reviews, food pattern modeling). It will be helpful for DGSAC members to understand best practices for producing guidelines even though the DGSAC will not be crafting the DGA recommendations themselves. This knowledge can facilitate creation of effective DGSAC conclusions for the federal DGA writing team’s consideration in its development of the DGA Policy Report. Like the DGPCG, DGSAC members would be selected through the process recommended in this National Academies committee’s first report (see Boxes 1-4 and 2-3 for further discussion about identifying and managing biases and conflicts of interest).
Regarding the inclusion of the P/B–24 population, because it is unlikely that this process redesign model could be made fully operational for the 2020–2025 DGA update, an interim enhancement to the existing process could be developed to add P/B–24-specific expertise to support the DGSAC’s synthesis and interpretation of the evidence. To accomplish this, systematic reviews being conducted for the P/B–24 project ought to be peer reviewed, and at least one person with general expertise in the science of P/B–24 and experience with systematic reviews needs to be involved with the DGSAC.
PROPOSED WORKFLOW
At a minimum, this National Academies committee believes the criteria, process, and logic for topic selection and evidence grading must be clearly articulated and fully disclosed. The public, including consumers and stakeholders, need to be provided timely opportunities to engage at key points in the process: (1) nominating topics; (2) responding to a list of selected topics; (3) nominating experts to the DGPCG and DGSAC; (4) commenting on a list of provisional appointees; (5) providing feedback on the DGSAC report; and (6) commenting on the DGA Policy Report. While it will not be feasible to engage the public at every task because public comment periods can be lengthy and adds time to the overall process, care should be taken to help build trust in the overall process. The
major tasks of the proposed process include topic identification, selection, and prioritization; data collection and evaluation; data synthesis, interpretation, and integration; and the update of the DGA.
Topic Identification, Selection, and Prioritization
A critical task in the process to update the DGA is deciding on the topics to be reviewed, which can be controversial. To that end, transparency and appropriate opportunities for stakeholder participation are needed to help develop credibility in the final list of topics. A number of procedural decisions will need to be made. For example, will there be an explicit process for making decisions such as putting a voting structure in place? Will methods such as value of information analysis be considered for ranking,3 or will the process for making decisions be more subjective (i.e., committee discussion and consensus)? Regardless of the process used, it will be necessary to clearly articulate to the public at the onset of the process how topics are identified, selected, and prioritized; the criteria against which topics are considered (see Figure 3-2 for examples); and how the criteria are operationalized. The process for identifying, selecting, and prioritizing topics could be modeled after a number of other processes such as AHRQ’s process for comparative effectiveness reviews, Healthy People, and the World Health Organization (Andrews, 2013; HHS, 2008; WHO, 2017).
In the topic identification phase, all stakeholders would nominate topics for potential inclusion in the DGA. This National Academies committee believes nominations need to be fielded from a broad group of interested parties, including the public; professional organizations; food sector organizations; researchers; and state, federal, and local governments. Nomination statements ought to have a standard format for purposes of clarity and organization to facilitate selection of potential topics, such as (1) why the topic is important, (2) how the implementation of recommended changes may improve health outcomes, (3) several specific, key questions to explore within the topic, and (4) supporting references as applicable. Topics could be collected by USDA and HHS and then filtered based on explicit criteria, in accordance with a transparent and documented process. In addition, the DGPCG could review topics of public interest even if not specifically nominated, for example through search engine analytics as discussed in Chapter 2. A list of nominated and identi-
___________________
3 Value of information analysis can be used as a tool to set research priorities. It is “an approach to research prioritization which uses Bayesian methods to estimate the potential benefits of gathering further information (through more research) before making a decision” (Myers et al., 2012).
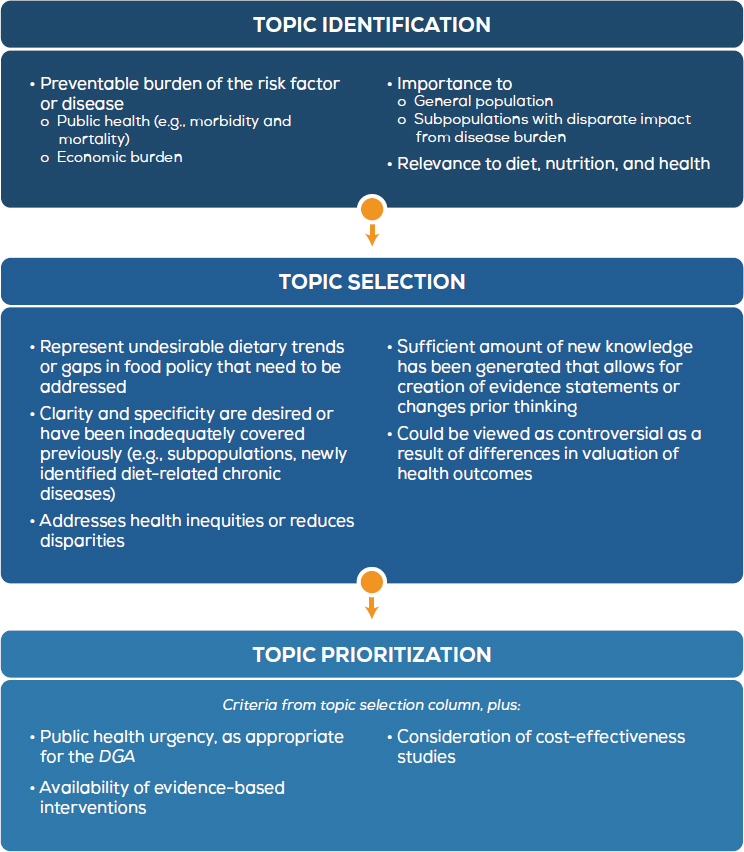
NOTES: These criteria were derived from a number of other efforts at organizations, including the World Health Organization, the Patient-Centered Outcomes Research Institute, the Agency for Healthcare Research and Quality Evidence-based Practice Centers Program, the Institute of Medicine, Cochrane, the U.S. Preventive Services Task Force, the Guide to Community Preventive Services, and the 2015 DGAC. The criteria from each of these organizations were modified to fit the needs of the process to update the DGA. The darkest blue box indicates any stakeholder as primary actor; the lighter blue boxes indicate DGPCG as the actor.
fied topics deemed not to be relevant could then be made publicly available. All nominated topics that meet the topic identification criteria ought to be reviewed with the qualification that topics identified as being of strong public interest be of current, not historical, interest. Owing to time and resource constraints, and available evidence, not all topics could be included in each cycle. Topics could be accepted continuously, but cutoff dates could be established for a topic to be considered for each DGA cycle.
Topic selection refers to the process for narrowing the list of all relevant nominations to a set of topics eligible to be addressed in the upcoming cycle. In this stage, the DGPCG would consider each identified topic based on a clear set of criteria and would publish a prioritization of selected topics, as well as explanations for why excluded topics or categories of topics were deferred.
The last stage, topic prioritization, refers to the process for choosing the order in which topics are evaluated for inclusion in the DGA. The DGPCG could prioritize the topics from the topic selection stage based on its expert opinions and a predetermined set of criteria. The final prioritization would be made publicly available along with a statement of why some topics or a tier of topics were designated as being of lower priority. A lower priority would be designated for the purpose of making evidence gathering feasible, not to indicate that topics be disregarded.
While USDA and HHS proposed criteria for the 2015 DGAC to consider,4 it is not clear how the DGAC used the criteria. It is this National Academies committee’s opinion that specific criteria be clearly defined for each stage and the process by which the criteria are considered be made transparent.
___________________
4 The 2015 DGAC was to consider the following draft topic selection criteria: (1) target populations; (2) potential effect on food and nutrition-related outcomes of public health concern, such as health outcomes and diet-related behaviors, and (3) likelihood of informing recommendations, whether it be to suggest new guidance, inform a revision to current guidance, or address urgent public health concerns. Suggested criteria for prioritization included (1) a review of the current evidence on the topic may inform the development of new dietary guidance for Americans ages 2 years and older; (2) a review of the current evidence on the topic may result in a change or elaboration in existing recommendations; (3) the topic represents important uncertainty or a knowledge gap for decision makers; (4) the topic addresses a dilemma in public health nutrition; (5) the topic represents an area where there is a degree of urgency for guidance (e.g., significant area of public health concern, emerging area for public health action); (6) the topic addresses a common practice in public health nutrition for which there is no government guidance; and (7) the topic has the potential to inform the development of dietary guidance that is public health oriented (i.e., the promotion of health and the prevention of disease at the population/community level) and not the development of clinical guidelines to use for the treatment and care of individuals with specific diseases and conditions (see Chapter 5 for details).
Data Collection and Evaluation
As soon as the DGPCG prioritizes topics for a particular DGA cycle and the secretaries of USDA and HHS affirm the list, the next task of collecting and evaluating data would begin. Original and existing systematic reviews, food pattern modeling, and descriptive data analyses would be conducted by federal methodologists (e.g., NEL staff, nutritional epidemiologists, respectively). TEPs would work with the federal teams as needed. Analyses ought to be conducted with the goal of providing final peer-reviewed results to the DGSAC when it first convenes (see Chapter 4). Some analyses are performed each cycle, such as identifying nutrients of concern, and could be produced before the DGPCG concludes its work, depending on data availability. If new data became available over the course of the DGA cycle that might lead to significant changes in results, it would be prudent to rerun analyses as needed.
Data Synthesis, Interpretation, and Integration
The final tasks in evaluating the body of evidence are data synthesis, interpretation, and integration, which would all be conducted by the DGSAC. This National Academies committee strongly believes that the DGSAC, as an independent arbiter of the state of the science, needs to be separated from data collection and evaluation to the greatest extent possible. While it will be necessary to work with the federal teams responsible for conducting systematic reviews, food pattern modeling, and descriptive data analyses, the role of the DGSAC needs to be clear, resulting in a different relationship than recent DGACs have had (see Chapter 4 for more details). By having more independence from the federal teams performing the analyses than in the current process, the DGSAC would be able to evaluate the evidence and develop conclusions without being able to unduly influence the process of data collection and evaluation.
The DGSAC would submit its final evaluation of the body of evidence to the secretaries of USDA and HHS in a scientific report. This scientific report ought to be open for public comment, similar to the public review of the current DGAC Scientific Report.
Update the DGA
Upon release of the DGSAC’s scientific report, USDA and HHS would consider the DGSAC’s conclusions in its update and review of the DGA, similar to what has been done with the 2005, 2010, and 2015 DGAC reports. Aside from selection of DGPCG, TEP, and DGSAC members, this final step of updating the DGA is the first place in the process policy makers should be involved in substantive decision making.
This National Academies committee believes that USDA and HHS should decide how the update is conducted, while making sure the process is transparent (see Chapter 5 for a full description and assessment of how USDA and HHS consider the DGAC Scientific Report).5 For example, the federal writing team—the group that updates the DGA based on the DGAC Scientific Report—needs to adhere to explicit and transparent standards for developing evidence-based recommendations. Multiple sets of standards exist that could serve as models, with the understanding that the process for developing DGA recommendations does not follow typical guidelines development processes because the experts assessing the evidence do not write the guidelines and recommendations themselves.6 As part of following these standards, it will be important to review the potential biases and conflicts of interest for writing team members, and ensure external reviewers represent a diverse set of viewpoints. As standards for the guidelines development process evolve, changes ought to be adopted by the federal writing team to keep the DGA process current with best practices in the field.
To enhance the integrity of the process, the DGA Policy Report should disclose what decisions were made about the DGSAC’s conclusions and why any conclusions were not acted upon or modified.
Recommendation 2. The secretaries of USDA and HHS should provide the public with a clear explanation when the DGA omit or accept only parts of conclusions from the scientific report.
CONSIDERATION OF OPTIONS FOR IMPLEMENTATION
The proposed redesign model will not be easy to implement, but it is a necessary step to provide the process with agility and flexibility. This National Academies committee considered how the process redesign model could be implemented, while conforming to the National Nutrition Monitoring and Related Research Act and the Federal Advisory Committee Act; none of the recommended changes in this report would require a revision to either act.
This National Academies committee envisions the DGSAC as a federal advisory committee, like the DGAC has been. Constitution as a federal advisory committee would allow the DGSAC to provide con-
___________________
5 The major steps include assembling a writing team of USDA and HHS staff, incorporating evidence, external peer review, and federal reviews and approvals.
6 Organizations that have developed standards for clinical practice guidelines include the AGREE next steps consortium, the GRADE working group, the Guidelines International Network, the Institute of Medicine, and the World Health Organization (Brouwers et al., 2010; Guyatt et al., 2008; IOM, 2011; Oxman et al., 2006; Schünemann et al., 2012, 2013, 2014).
clusions to the secretaries of USDA and HHS based on the members’ consensus. TEPs could be convened on an ad hoc basis by USDA and HHS. However, to allow for the DGPCG to operate continually under the constraint of the aforementioned laws, this National Academies committee has identified three options.
The first option is to establish the DGPCG as a federal advisory committee whose charter would be renewed every 2 years, the maximum length of time allowed under the Federal Advisory Committee Act. This option would allow the DGPCG to provide independent consensus-based recommendations to the secretaries in a transparent fashion. With the ability to be functionally continuous, the DGPCG would be able to help oversee efforts to monitor the literature. As a federal advisory committee, the DGPCG would recommend to the secretaries that its prioritized list of topics and questions be the set of topics reviewed by the DGSAC. USDA and HHS could informally share ownership of the process and operational costs, by each establishing a federal advisory committee (for example, USDA establish the DGPCG and HHS establish the DGSAC), thereby not overburdening any particular agency.7 However, this option adds a degree of complexity to the current process that will require a number of handoffs between groups, necessitating coordination by USDA and HHS. Establishment of the DGPCG by a single agency could result in questions related to the independence of the DGPCG, as USDA’s involvement in the DGA has been criticized in the past (Mozzaffarian, 2016). These concerns could be minimized if the checks in the process redesign model (e.g., more public comment periods, increased transparency at each step) were instituted.
A second option considered was establishing the DGPCG as a federal group consisting of both federal and nonfederal members, but not governed by the Federal Advisory Committee Act. As a federal group, the DGPCG could meet continually but could not issue consensus-based recommendations to the secretaries of USDA and HHS. This option could be seen as limiting transparency and jeopardizing the trustworthiness of the process based on the perception that USDA and HHS would have too much influence over the steps of monitoring new evidence and topic identification and prioritization. Other potential benefits and challenges regarding governance, funding, member composition and selection processes, and interactions with the DGSAC would depend in large part on
___________________
7 Generally, only one agency can establish a discretionary, time-limited federal advisory committee so as to comply with federal restrictions related to the use of appropriated funds. Maintaining the current practice of rotating leadership and corresponding operational costs between the two agencies would require congressional authorization or presidential directive. This level of authority has not been needed for the current DGAC because it has not been acting in an ongoing manner.
how the federal group would be created and implemented. If this option was selected, creation of a federal group would need to be completed in a transparent manner.
A third option calls for a nongovernmental organization to convene the DGPCG. Term limits and other rules imposed by the Federal Advisory Committee Act would no longer apply and would also limit potential criticisms of USDA and HHS’s roles. However, it is unclear whether advice from a nongovernmental organization would be as influential as options 1 and 2, particularly with respect to federal programs, or whether its processes would be transparent. It is also unlikely that a nongovernmental organization would have the funds and capacity to convene the DGPCG on its own. If this option were considered, organizations with the necessary breadth of expertise and experience would need to be identified.
Weighing the relative advantages and disadvantages of each option, this National Academies committee recognizes that no perfect option exists. Although option 1 adds layers of complexity with establishment of two federal advisory committees, it is the only option that features built-in mechanisms to ensure objectivity and transparency. Option 2 would potentially be the easiest to implement, but it is most likely to face criticism regarding its ability to perform the tasks of the DGPCG in a transparent and independent manner. Option 3 has great potential to ensure continuity and transparency, but it is dependent on numerous unknowable factors, most important of which is that an influential, nonpartisan organization with the necessary experience and expertise would need to be identified. Given these options, this National Academies committee believes establishing the DGPCG as a federal advisory committee to be the most likely option to yield a trustworthy, dependable evaluation of the science, without causing undue burden on any particular agency.
The presented model is one example of a new process that achieves the values and goals articulated in Chapter 2. This National Academies committee recognizes that the secretaries will need flexibility in implementing the proposed redesign, as there is no single best process to use. One reason for the need for flexibility is that a detailed exploration of the costs of the proposed redesign model could not be weighed in this report owing to a lack of information available regarding current resource use. This National Academies committee believes the operational costs would likely increase in the short term as a result of needing to set up and support the DGPCG, TEPs, and DGSAC. However, because the current DGAC tasks would be reallocated among the various groups, and over time the proposed process will likely simplify the number of questions being studied within each DGA cycle, it is this National Academies committee’s judgment that costs may decrease in the long term. This National Academies committee believes the benefits and outcomes will justify any additional costs.
REFERENCES
Andrews, J. 2013. AHRQ methods for effective health care. In Prioritization criteria methodology for future research needs proposals within the effective health care program: PiCMeprioritization criteria methods. Rockville, MD: Agency for Healthcare Research and Quality.
Brouwers, M., M. E. Kho, G. P. Browman, J. S. Burgers, F. Cluzeau, G. Feder, B. Fervers, I. D. Graham, J. Grimshaw, S. Hanna, P. Littlejohns, J. Makarski, and L. Zitzelsberger, for the AGREE Next Steps Consortium. 2010. AGREE II: Advancing guideline development, reporting and evaluation in healthcare. Canadian Medical Association Journal 182(18):E839-E842.
Guyatt, G. H., A. D. Oxman, R. Kunz, Y. Falck-Ytter, G. E. Vist, A. Liberati, and H. J. Schünemann. 2008. Going from evidence to recommendations. BMJ 336(7652):1049-1051.
HHS (U.S. Department of Health and Human Services). 2008. The Secretary’s Advisory Committee on National Health Promotion and Disease Prevention objectives for 2020: Phase I report recommendations for the framework and format of Healthy People 2020. https://www.healthypeople.gov/sites/default/files/PhaseI_0.pdf (accessed July 14, 2017).
IOM (Institute of Medicine). 2011. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
Mozzaffarian, D. 2016 (unpublished). Comments presented at USDA Dietary Guidelines for Americans listening sessions. Washington, DC, February 19, 2016.
Myers, E., A. J. McBroom, L. Shen, R. E. Posey, R. Gray, and G. D. Sanders. 2012. Value-of-information analysis for patient-centered outcomes research prioritization. Durham, NC: Duke Evidence-based Practice Center.
Oxman, A. D., A. Fretheim, and H. J. Schünemann. 2006. Improving the use of research evidence in guideline development: Introduction. Health Research Policy and Systems 4(1):12.
Schünemann, H. J., M. Woodhead, A. Anzueto, A. S. Buist, W. Macnee, K. F. Rabe, and J. Heffner. 2012. A guide to guidelines for professional societies and other developers of recommendations: Introduction to integrating and coordinating efforts in COPD guideline development. An official ATS/ERS workshop report. Proceedings of the American Thoracic Society 9(5):215-218.
Schünemann, H., J. Brožek, G. Guyatt, and A. Oxman, editors. 2013. Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. The GRADE Working Group. http://gdt.guidelinedevelopment.org/app/handbook/handbook.html#h.33qgws879zw (accessed August 3, 2017).
Schünemann, H., W. Wiercioch, I. Etxeandia, M. Falavigna, N. Santesso, R. Mustafa, M. Ventresca, R. Brignardello-Petersen, K.-T. Laisaar, S. Kowalski, T. Baldeh, Y. Zhang, U. Raid, I. Neumann, S. L. Norris, J. Thornton, R. Harbour, S. Treweek, G. Guyatt, P. Alonso-Coello, M. Reinap, J. Brožek, A. Oxman, and E. A. Akl. 2014. Guidelines 2.0: Systematic development of a comprehensive checklist for a successful guideline enterprise. Canadian Medical Association Journal 186(3):E123-E142.
USDA (U.S. Department of Agriculture). 2017. Pregnancy and birth to 24 months project. https://www.cnpp.usda.gov/birthto24months (accessed January 8, 2017).
WHO (World Health Organization). 2017. Normative work in nutrition at WHO: Priority setting for guideline development. Geneva, Switzerland: World Health Organization.
This page intentionally left blank.


















