1
Introduction
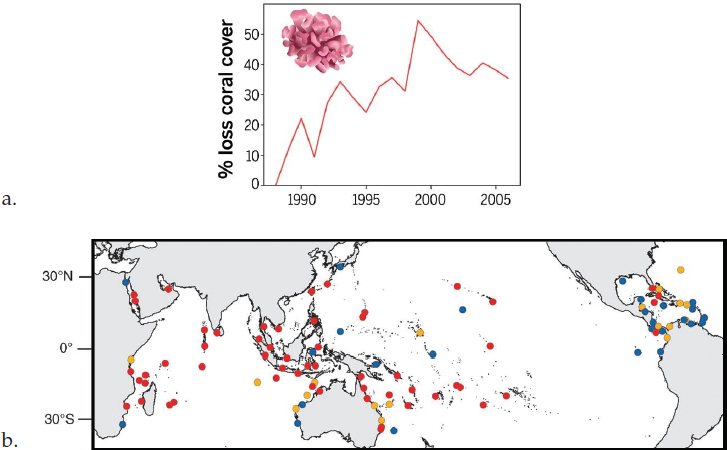
Coral reef declines have been recorded for all major tropical ocean basins since the 1980s, averaging approximately 30%-50% reductions in reef cover globally, with spikes after the 1998 global coral bleaching event (Selig et al., 2012; see Figure 1.1a). These losses derive from a host of problems experienced by coral reefs around the world, including coastal habitat loss, pollution, overfishing, and climate change. Recent losses of coral cover have increased dramatically following the multiple global bleaching events from 2014 to 2017 (Hughes et al., 2017a, 2017b, 2018; see Figure 1.1b), driven by an extensive and prolonged ocean warming event (NOAA, 2018). Because coral reefs are integrated ecosystems, declines of reef-building corals and their associated three-dimensional complexity tend to also lead to declines of other species that depend on healthy reefs (Bellwood et al., 2006), such as a large number of coral reef fish (Graham et al., 2007), reducing the ecosystem services provided by coral reefs. For the hundreds of millions of people who depend on reefs for food or employment (Cinner et al., 2012; Moberg and Folke, 1999), the thousands of communities that depend on reefs for wave protection (Ferrario et al., 2014; Sheppard et al., 2005), and the many economies that depend on reefs for fisheries or tourism (Costanza et al., 2014; Hoegh-Guldberg et al., 2015), the health and maintenance of this major global ecosystem is crucial.
Anthony et al. (2015) divided the discussion of the ways coral reefs are impacted by humans into press and pulse stressors. Press stressors include pollution, sedimentation, overfishing, ocean warming, and acidification,
as these factors exert long-term steady pressure on coral growth, survival, and reproduction. Pulse stressors include storms, bleaching events, or disease outbreaks that are more acute, periodic, and destructive over short time periods. Some press stressors are local (e.g., pollution), and some affect large regions, driven by global climate change (chronic warming and acidification). An effort to sustain coral reefs in the future calls for conservation and management strategies that can underpin the biological and ecological processes that build tolerance and resilience to multiple stressors (Anthony et al., 2017).
GLOBAL CLIMATE STRESSORS
Increasing changes in the global climate and ocean chemistry threaten the persistence of coral reefs. An understanding of these impacts is important for identifying the processes that interventions may target. Additionally, variable responses of coral to stressors provide insight into potential natural resilience to these stressors. It should be noted that mitigation of
global stressors is important for preserving coral reefs and, compared to novel interventions that target the resilience of coral reefs, has high certainty in effectiveness. Mitigation of global stressors will also inevitably increase the likelihood of any local intervention succeeding.
Temperature Stress
By far the most attention has been paid to the global effects of ocean warming on coral health. The link between temperature and corals derives largely from the bleaching response of coral cells to higher-than-normal temperatures (see Box 1.1). Recent increases in ocean temperature have led to a significant increase in exposure of corals to high-temperature events (see Figure 1.2) and have caused severe coral loss globally (reviewed in Hughes et al., 2018). Comparative analysis of bleaching patterns and temperatures shows that some corals are more resistant to bleaching than others (especially species in the genus Porites; see Carpenter et al., 2008; Marshall and Baird, 2000). Comparisons also show that the temperature at which bleaching occurs is relative to the temperature regime at a specific location, even for the same species (Glynn, 1996). These observations have
led to a widespread rule of thumb that bleaching starts after temperatures exceed the maximum average summer temperature for a region. The longer these high temperatures persist, the more likely bleaching becomes. Thus, bleaching susceptibility typically is linked to the sum of excess degrees of heat over weeks of exposure—measured by the National Oceanic and Atmospheric Administration’s (NOAA’s) Coral Reef Watch program as Degree Heating Weeks (DHWs). Bleaching events can be driven or amplified locally or regionally by ephemeral atmospheric systems that promote calm and clear conditions (e.g., doldrums conditions) that favor ponding of warm water masses and deep light penetration (which aggravate the effects of high temperature).
Recovery from bleaching depends on the severity of the heating event (duration and degree of temperature increase) and the condition of the corals. During strong temperature pulses, or when colonies are less able to capture food, coral death is common (Grottoli et al., 2006). In milder bleaching events, or when corals have abundant energy reserves
(Connolly et al., 2012), recovery can be widespread. Even in these cases, full recovery of physiological homeostasis, growth, and reproduction lags behind recovery of symbiont density for months (Grotolli et al., 2006; Thomas et al., 2018). Following long recovery periods, the ability of colonies to survive another bleaching event is curtailed and may also result in subsequent increased disease within the population. Back-to-back El Niño and La Niña events from 2015 to 2017 led to widespread coral loss, especially among colonies impacted by the second event but not yet recovered from the first (Hughes et al., 2018; NOAA, 2018).
Cellular mechanisms of coral bleaching are much less understood than ecological patterns of bleaching. Bleaching induced by temperature appears to largely involve active ejection of symbionts from coral cells, rather than strict loss of symbiont-containing coral cells or death of coral cells (Weis, 2008). Bleaching is widely associated with impairment of symbiont photosynthesis, especially disruption of the electron transport chain delivering the energy of captured photons to carbon-fixing centers in the chloroplast. This breakdown occurs at about the same time as reactive oxygen species (ROS) are released in the cells, either through production by malfunctioning chloroplasts or in mitochondria (Baird et al., 2009a; Downs et al., 2002). Thus, high temperature promotes the dysfunction of cellular proteins involved in photosynthesis. Additionally, light (specifically, photosynthetically active radiation at wavelengths that promote photosynthesis) induces electron flow in symbiont chloroplasts that causes a buildup of ROS that can impair cell function (photo-oxidation; Lesser, 2011). In addition, components of solar radiation that do not contribute to photosynthesis, such as ultraviolet light, have also been implicated in bleaching (Gleason and Wellington, 1993; Lesser, 1997). Light is therefore a co-factor in initiating the coral bleaching response (Baker et al., 2008). Coral cells undergo a range of responses, including production of heat shock and chaperone proteins that refold proteins damaged by heat (Barshis et al., 2013; DeSalvo et al., 2008, 2010; Meyer et al., 2011), proteins that remove reactive oxygen (Lesser, 1997; Weis, 2008), and a large number of transcription factors that serve in cellular signaling (Traylor-Knowles et al., 2017).
Despite these detailed studies, the stress trigger that induces coral cells to initiate ejection of the symbiont remains unknown. Some indications are that the cellular mechanism controlling programmed cell death (apoptosis) may be involved because of the changes in apoptosis-related genes during bleaching (Weis, 2008). Alternatively, other mechanisms including cell detachment (Gates et al., 1992) might be at the heart of this phenomenon. The unfolded protein response is a basic eukaryotic cellular mechanism to increase protein folding in times of stress (Oakley et al., 2017), but it may induce cell death after severe heat stress and
bleaching (Ruiz-Jones and Palumbi, 2017). Such fundamental cellular stress responses could underlie coral bleaching if the switching mechanism used for the unfolded protein response was linked to the bleaching threshold (Ruiz-Jones and Palumbi, 2017).
The cellular mechanism that causes bleaching appears to have different environmental thresholds for corals of different species, and for corals housing different species of internal symbiotic algae. Within species, even those with the same symbionts, different coral colonies can have different bleaching tolerances. Field observations, transplants, and laboratory studies have shown that some of the variation among colonies is due to the widespread ability of corals to acclimate to local or seasonal heat conditions (Ainsworth et al., 2016; Palumbi et al., 2014). Other shifts in susceptibility to bleaching are associated with changes in the symbiont after bleaching events (reviewed by Baker et al., 2004; Silverstein et al., 2015). A strong source of variation among colonies also resides in the genetic or epigenetic differences between populations selected for living in different microhabitats. The genetic underpinnings of these differences are not well known, but appear to include many genes. Dixon et al. (2015) showed that hybrids of warm- and cool-water-adapted corals from the Great Barrier Reef exhibited intermediate heat tolerance. Bay and Palumbi (2015) used transcriptome scans to uncover more than 100 genes associated with populations living in a warm water, back reef pool in Samoa. By contrast, Jin et al. (2016) showed strong effects at a single genetic locus for antioxidant capacity. Although detailed study of the genetic mechanisms of coral bleaching or heat tolerance is still in progress, little evidence currently exists to suggest that there are a few master genes of large effect controlling heat tolerance in many species.
Ocean Acidification
A second global stressor related to climate change is the gradual acidification of the oceans because of increasing carbon dioxide (CO2) levels in the atmosphere (e.g., Doney et al., 2009). CO2 emitted into the atmosphere from fossil fuel combustion forms carbonic acid when it dissolves in the ocean. This causes a decrease in pH and decline in carbonate saturation (Caldeira and Wickett, 2003), resulting in a lower aragonite saturation state (Ωa) (Kleypas et al., 1999, 2006; Raven et al., 2005). Lower pH, increasing CO2, and decline in Ωa have complex effects on reef-building corals. Long-term exposure to low pH reduces calcification and growth rates (Albright et al., 2018; Chan and Connolly, 2013; Kleypas and Yates, 2009; Langdon et al., 2000; Pandolfi et al., 2011), with a reduction in calcification of 15%-22% per unit decrease in the Ωa of seawater.
The projected fall in pH and Ωa as CO2 levels increase (e.g., Figure 1.3) suggests long-term declines of coral reefs (Chan and Connolly, 2013;
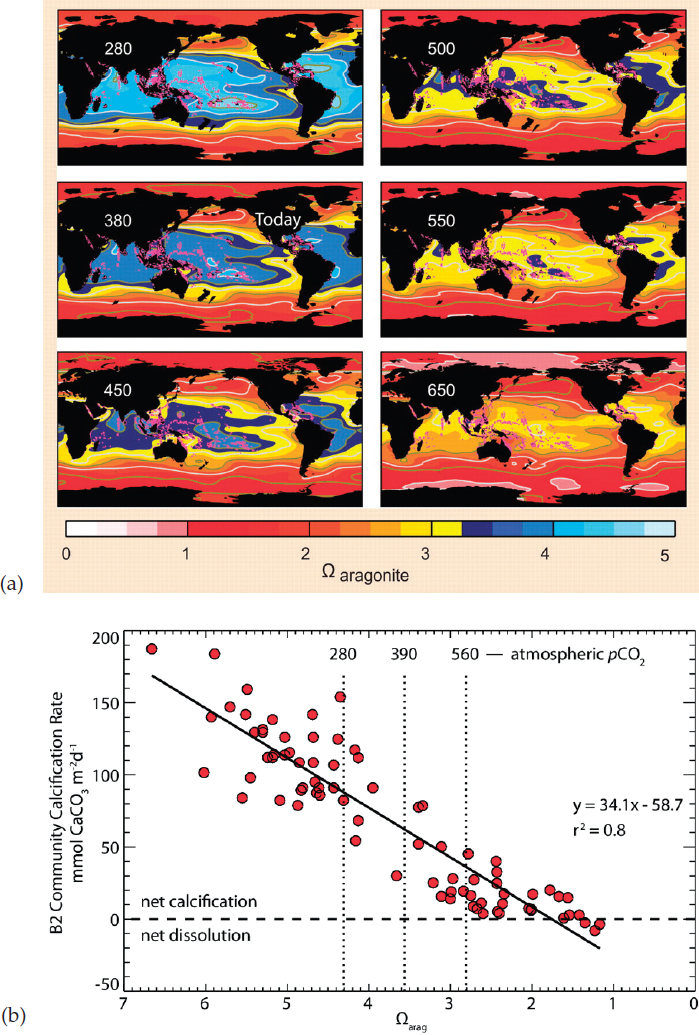
Hoegh-Guldberg et al., 2007) as CO2 emissions build up in ocean waters. However, unlike thermal anomalies, ocean acidification (OA) is a gradual buildup of a chronic stress that does not manifest as acute events. Also, coral reefs modify their seawater carbon chemistry through calcification (Kleypas et al., 2011), complicating the attribution of local impacts to ocean acidification. During the Anthropocene, the mean pH of the global ocean has dropped by about 0.1 units, lowering the mean Ωa in coral reef waters by more than half a unit (Cao and Caldeira, 2008). If atmospheric CO2 were allowed to double relative to pre-industrial levels (560 ppm), mean Ωa in coral reef waters would drop another full unit. Recent work on the southern Great Barrier Reef demonstrates that OA has already caused an approximately 6% drop in reef calcification (Albright et al., 2016). Other consequences of OA for reef corals include reduced coral fertilization and recruitment (Albright and Langdon, 2011), increased fragility of coral skeletons (Tambutté et al., 2015), and increased risk of reef wave damage by storms (Madin et al., 2008), the latter in part caused by accelerated bioerosion of the reef substrate (DeCarlo et al., 2015; Wisshak et al., 2012). As OA directly impacts two key fitness components of corals—growth and reproduction (Anthony, 2016)—any interventions that can counteract OA will directly support reef resilience.
The reactions of corals to increased CO2 levels and decreased pH varies among species. A study of coral reefs near natural CO2 vents in Papua New Guinea and Mexico shows that species of Porites tend to be dominant nearest the vent locations (Fabricius et al., 2011), with colonies exhibiting reduced calcification rates (Crook et al., 2013). At other sites such as Nikko Bay in Palau, high concentrations of CO2 combined with high temperatures result in reef areas dominated by Porites and a few other genera that maintain high growth rates. Laboratory studies also show increased ability of these corals to calcify under high CO2 levels (Shamberger et al., 2014). The ability of some corals to calcify under a variety of pH levels has been traced to their ability to maintain high pH at the site of calcification. For example, as external seawater pH dropped from 8.1 to 7.2, the pH of fluids at the local sites of calcification in Porites corals dropped only from 8.5 to 8.2 (McCulloch et al., 2012). Other studies suggest that ample external food supply allows coral calcification to be maintained despite higher metabolic costs of calcification under low pH conditions (Drenkard et al., 2013).
DISEASE
Increased reports of coral disease outbreaks have been associated with warming events as well as local stressors (Carpenter et al., 2008; Harvell et al., 2007). In the Caribbean specifically, disease outbreaks affecting both corals (often white-band disease initially) and a key reef herbivore, the
urchin Diadema antillarum, were instrumental in driving declines in coral cover (Aronson and Precht, 2001a, 2001b; Lessios, 2016). The rapid loss of corals in the Caribbean from disease outbreaks is unprecedented in the geological records (Aronson and Precht, 2001a; Gardner et al., 2003), altering both the total abundance and diversity of coral species (Weil, 2004). Because disease agents often infect the frame-building corals, the destruction of the reef habitat due to disease also affects other members of the coral reef ecosystem (Nugues, 2002).
Disease outbreaks within ecosystems often occur when thermal thresholds are surpassed, making corals among the most susceptible due to a very narrow thermal threshold for optimal coral health (Harvell et al., 2002). Randall and van Woesik (2015) linked increase of sea surface temperatures (SSTs; both thermal minima and maxima) with the spread of white-band disease, which contributed to the region-wide declines of the dominant Caribbean reef-building coral, Acropora palmata and A. cervicornis. Bleaching extent has been found to correlate with disease incidence (Brandt and McManus, 2009). In other regions, similar links between thermal stress and coral disease outbreaks have been reported (Bruno et al., 2007). Importantly, future climate scenarios have been projected to result in increased disease outbreaks, impacting coral populations globally (Fabricius, 2005; Harvell et al., 1999, 2002; Maynard et al., 2015). Factors such as nutrient enrichment and increased sedimentation linked with rapid urbanization and coastal development can also exacerbate disease impacts on reefs (Bruno et al., 2003; Pollock et al., 2014; Vega Thurber et al., 2014). For example, field and laboratory experiments have demonstrated that moderate increases in nutrient concentrations (phosphorus, nitrate, and ammonium) can substantially increase the severity of some diseases, through both higher prevalence and increased lesion progression rates (Bruno et al., 2003; Vega Thurber et al., 2014; Voss and Richardson, 2006; Wang et al., 2018). Similarly, by stressing corals, sediments may make the corals more susceptible to infections by microbial pathogens and may also act as disease reservoirs (Voss and Richardson, 2006). Coinciding with the emergence of coral diseases, many coral reefs have experienced dramatic increases in benthic macroalgae (Ceccarelli et al., 2018; Edmunds, 2002; Hughes, 1994; McCook, 1999). Elevated dissolved inorganic carbon levels derived from algal exudates can accelerate the growth rate of microbes in the coral’s surface mucopolysaccharide layer by an order of magnitude, which impacts coral health through disruption in the balance (dysbiosis) between the coral and its associated microbiota (Kline et al., 2006). Macroalgae may also act as disease reservoirs as was found in the Caribbean, where corals that were in contact with the algae Halimeda opuntia developed disease symptoms (Nugues et al., 2004).
Disease is a natural part of any ecosystem; interactions between the host and the disease agent are a constant arms race that help shape
patterns of species evolution (Rosenberg et al., 2007). The surrounding environment has a strong influence on disease dynamics within animal populations, either through reducing the resilience of the host or promoting the pathogenicity/virulence of a causative agent (Plowright et al., 2008). An increase in disease prevalence within coral populations is therefore an indicator of stress on the reef ecosystem. Ocean warming or human-driven environmental perturbation affect basic biological and physiological properties of coral, thus influencing the balance between opportunistic pathogens and the coral’s ability to prevent pathogen infection or overcome abiotic challenges (Ben-Haim et al., 2003; Harvell et al., 2002; Rosenberg and Ben-Haim, 2002). Currently, understanding of the interactions at the cellular level between the disease agents, the coral immune system, and environmental factors is poor and inhibits attribution of disease causation (Mera and Bourne, 2018). For successful management of disease outbreaks and therefore ensuring the long-term resilience of coral populations in both natural ecosystems and artificially reared coral systems, a more thorough understanding of the underlying biotic, abiotic, host, and environmental factors leading to disease onset is still required. Nevertheless, the clear links between anthropogenic stresses on reefs and higher disease prevalence highlights that as we move to climate regimes outside previously experienced thresholds, the potential for diseases to impact coral populations at broad ecosystem scales increases as well.
LOCAL STRESSORS AND STRESS PREVENTION
Because of the high severity and widespread occurrence of local human pressures on coral reefs, there have been many studies, and decades of effort, to measure the impact and mitigate the effect of human activities on reef ecosystems. Despite the impact of bleaching on coral cover (Hughes et al., 2017b), historically the majority of coral losses have been ascribed to non-climate-related coral reef changes, such as local habitat destruction, overgrowth by algae after overfishing, pollution, and sedimentation (Bellwood et al., 2004; Pandolfi et al., 2003). It is not the purpose of this report to summarize these many papers and decades of work. However, the role of local stressors on reefs interacts with the role of climate (e.g., Carilli et al., 2009; Hughes et al., 2017a), and so local efforts to support reef ecosystems will always be a substantial part of any global reef protection strategy.
A recent summary for the Great Barrier Reef, for example, shows that local effects of human activity on reefs include strong impacts by overfishing, habitat destruction, pollution from land, and sedimentation, which may result from coastal development, transportation, aquaculture, and other activities (Uthicke et al., 2016; see Table 1.1). Similar conclusions
TABLE 1.1 Characterization of Stressors to Coral on the Great Barrier Reef
| Pressure/Threat | Local (L) Versus Global (G) Causes | Acute (A)/Chronic (C) | Risk to Ecosystem |
|---|---|---|---|
| Ocean warming | G | C/A | Very High |
| Ocean acidification | G | C | Very High |
| Cyclones/altered weather patterns | L (G) | A/C | Very High |
| Illegal fishing and poaching | L | C | Very High |
| Incidental catch of species of conservation concern | L | A | Very High |
| Nutrient runoff | L | C/A | Very High |
| Outbreak of Crown of Thorns Seastars | L | A | Very High |
| Sediment runoff | L | C/A | Very High |
| Coastal habitat modification | L | C | Very High |
| Sea level rise | G | C | Very High |
| Pesticide pollution | L (G) | C/A | High |
| Barriers to flow | L | C | High |
| Discarded catch | L | A | High |
| Extraction of predators | L | A/C | High |
| Disposal of dredge material | |||
| Marine debris | L (G) | A/C | High |
| Extraction from spawning aggregators | L | A/C | High |
| Outbreak of disease | L (G) | A | High |
SOURCE: Recreated from Uthicke et al., 2016.
have been made for the Caribbean (Jackson et al., 2014). Because of the importance of local stressors on corals, and the links between local impacts and global climate change, some key findings about local impacts are briefly summarized here. In each case, interventions that address the source of local stressors have been identified, and in some cases, stressors have been mitigated. Despite decades of understanding the nature and extent of these local stressors in causing coral reef health declines (e.g., Russ et al., 2015), it remains challenging for local jurisdictions to solve these problems (Aswani et al., 2015). Also, the recent pan-tropical bleaching events from 2014 to 2017 showed that remote coral reefs under minimal influence from pollution and overfishing bleached as severely as reefs
exposed to multiple pressures (Hughes et al., 2017b). Therefore, while local- and regional-scale management of local stressors will continue to be critical under climate change because it reduces chronic mortality and facilitates recovery from pulse stressors, these efforts on their own are inadequate in the face of ongoing climate change (Anthony, 2016).
Overfishing Fish impact coral health through many mechanisms (Hixon, 2015; Mora et al., 2011; Mumby et al., 2007). A primary role is that of herbivorous fish in preventing fast-growing algae from over-growing corals (Hughes, 1994; Hughes et al., 2010; Jackson, 1997). When herbivory declines, macroalgae quickly grow, overwhelming corals (e.g., McCook, 1999) and potentially preventing settlement of coral larvae (Davies et al., 2013). As a result, loss of herbivores through local fishing pressure can result in a phase shift from coral-dominated to algal-dominated reefs (Hughes, 1994). The role of some herbivorous species such as parrotfish has been so great that their abundance has been suggested as a major indicator of reef health (Jackson et al., 2014; Mumby et al., 2007). However, a series of studies suggests that the relationship between herbivory and coral health is contingent on other environmental factors, such as the species involved, the location (Adam et al., 2015; Russ et al., 2015), and nutrient load (e.g., Burkepile et al., 2013).
Beyond traditional fisheries management strategies of effort and size limit controls, two broad categories of management approaches can reduce the likelihood of overfishing: marine protected areas (MPAs) and rights-based fisheries. MPAs address the ecosystem impacts of fishing by restricting or eliminating fishing in designated areas (McClanahan et al., 2006; Pikitch et al., 2004; Soler et al., 2015). On average, MPAs lead to increases in density, biomass, body size, and diversity compared to before MPA establishment or outside MPAs (Lester et al., 2009), and MPA networks on the Great Barrier Reef support coral resilience through a suite of processes (Mellin et al., 2016). Meta-analyses have suggested key features of MPA management that increase their effectiveness in nearshore fisheries recovery including implementation of no-take areas, effective enforcement, and engagement of local fishers in planning and implementation (Di Franco et al., 2016; Edgar et al., 2014). Rights-based fisheries management can incentivize sustainable resource use and environmental stewardship by providing ownership over a share of the total fishery quota (catch shares or individual transferable quotas [ITQs]; Copes, 1986, Costello et al., 2008) or ownership over a region through membership in a small collective (territorial user rights for fishing [TURFs]; Gelcich et al., 2010; Jentoft et al., 1998). Observed benefits of ITQs include reduced variability in fishing and less discards (Essington, 2010), and observed benefits of TURFs include greater abundance and size of harvested species as well as greater biodiversity (Gelcich et al., 2010).
Habitat destruction Direct impacts of fishing on coral reefs include blast fishing (McManus et al., 1997), trawling (Hall-Spencer et al., 2002), or pull seining (McClanahan et al., 1997) that cause physical damage to the reef structure via detonation of explosives or dragging of nets through reefs. Reefs are also destroyed for facilities development (e.g., ports and airports) or to provide building materials. In addition, indirect reef destruction occurs through increases in sedimentation associated with coastal development, pollution, and other kinds of deleterious human impacts. Habitat destruction can also occur from overharvesting colonies for the aquarium trade or nursery construction, and from damage from tourism (Davenport and Davenport, 2006). Protecting local reefs from habitat destruction can be an important way to protect ecosystem diversity (White et al., 2000).
Water quality Growth, survival, reproduction, and recruitment of corals are strongly influenced by water quality including turbidity, sediments, nutrients, and toxic pollutants. Particular attention has been paid to nutrient enrichment, sewage, petroleum products, and metals (Dubinsky and Stambler, 1996; Fabricius et al., 2005), and many other local pollutants have been shown to negatively affect corals (Duprey et al., 2016; Loya and Rinkevich, 1980; Walker and Ormond, 1982; Wenger et al., 2016; Wilkinson, 1999). Sources of pollution range from human sewage systems, farm runoff, golf courses, urban development, waste deposition, oil and gas leakage, livestock pens, and many other human activities, even use of sunscreens (Downs et al., 2016; Pandolfi et al., 2005). Effective mitigation of pollution, typically at a local level, is key to reducing or reversing these problems (e.g., Wear and Vega Thurber, 2015).
Sediments broadly affect corals through smothering, especially when sediment loads are high after storms or coastal construction (reviewed in Fabricius, 2005; Richmond et al., 2007). There is also evidence that sediments can reduce egg-sperm interactions, thereby decreasing fertilization success (Humanes et al., 2017), inhibit settlement (Babcock and Smith, 2000; Hodgson, 1990; Perez et al., 2014), and decrease the likelihood of juvenile coral survival (Babcock and Smith, 2000; Richmond et al., 2018). Sediment and turbidity may reduce light reaching symbiotic zooxanthellae and increase prevalence of coral disease and other indicators of poor coral health (Pollock et al., 2014).
Nutrients in the sediment that dissolve in seawater as well as from other effluent sources can contribute to eutrophication and algal growth, primarily when algae are otherwise nutrient-limited. However, the effect of eutrophication on coral is not strictly deleterious, and its impact will be ecosystem-dependent (D’Angelo and Wiedenmann, 2014; McCook, 1999; McCook et al., 2001). Turbidity due to sedimentation also decreases
the ratio of photosynthesis to respiration (Junjie et al., 2014; Philipp and Fabricius, 2003; Telesnicki and Goldberg, 1995). However, because particulate matter is a source of both nutrients and carbon in addition to providing shade, moderate turbidity can potentially provide an energetic and light reduction benefit for corals during or after bleaching (e.g., Anthony et al., 2007, 2009). Erosion-prevention techniques to reduce turbidity include halting deforestation and planting seagrass or scrub brush on dunes. Construction, farming, and coastal use practices that minimize erosion have also been successful (Gibson et al., 1998).
Storms Tropical cyclones or hurricanes are a natural part of the disturbance regime on coral reefs (Connell, 1978; Connell et al., 1997; Rogers, 1993). Recovery from storm damage has classically been a way that coral reef communities have been thought to maintain high levels of diversity in the face of competition for space. Tropical summer storms often follow repeated storm tracks, and so certain reef areas experience more frequent and more severe storm damage (Wolff et al., 2016). In some cases, typhoons during warm water events can reduce bleaching (e.g., Bernardo et al., 2017; Manzello et al., 2007). However, storms can cause massive damage and long-term losses of coral when recovery is low (Gardner et al., 2005; Woodley et al., 1981).
Storm damage varies dramatically on a local scale but is not due to local stressors. The number and severity of strong storms is thought to be increasing with climate change, but such links are difficult to apply to any given storm (NASEM, 2016b). While infrastructure has been developed to protect human communities from storms, few interventions have been documented for protecting coral reefs. It is worth noting here that healthy coral reefs provide natural coastal protection against storm waves (Ferrario et al., 2014), an ecosystem service worth trillions of dollars on a global scale (Beck et al., 2018; Costanza et al., 2014).
Invasive species Traditionally, marine invasive species have been considered to be more of a problem in low-diversity environments such as estuaries, rather than on coral reefs. Nevertheless, some high-profile cases of invasive species on coral reefs have raised awareness of this threat. When invasive species cause major physical, chemical, or biological changes, they set in motion the establishment of fundamentally novel communities. Most notorious is the lionfish (Pterois spp.) invasion of the tropical Atlantic; what started with a few observations in Florida has in less than two decades resulted in the establishment of populations ranging from the northwest Atlantic, the Gulf of Mexico, and the Caribbean to Brazil and from shallow waters to mesophotic reefs. They are considered a major problem because they are voracious predators of juvenile reef fish (Hixon
et al., 2016), which is of particular concern because of the widespread prevalence of overfishing in these regions.
However, lionfish are not the only invasive species on reefs. It has been estimated that 23% of the species reported for Pearl Harbor in Hawaii are potentially non-native (Coles et al., 1999). Invasive seaweed species have become established in a variety of locations including Florida (Lapointe and Bedford, 2010) and Hawaii (Martinez et al., 2012), and the eastern Caribbean has been invaded by a non-native seagrass (Scheibling et al., 2018). There are several species of invasive Tubastrea corals in the Atlantic (e.g., Luz and Kitahara, 2017), and the now globally distributed, temperature-hardy symbiont Durusdinium trenchii (formerly Symbiodinium clade D) is thought to be an invader in the Caribbean (Pettay et al., 2015). Recently, even terrestrial invaders have been shown to harm coral reefs; rat-plagued islands in the Indian Ocean have fewer seabirds, which in turn results in a variety of impacts on the surrounding reefs, including fewer nutrients and fewer algae-eating fish (Graham et al., 2018).
In the context of restoration, invasive species, once established, are essentially impossible to eradicate. Efforts to mitigate the effects of invasive species often rely on volunteer efforts, are labor intensive, are expensive (e.g., Neilson et al., 2018), and require constant effort (e.g., Malpica-Cruz et al., 2016). Moreover, the possible movement of organisms as part of human-assisted migration to reduce the impact of climate change (see Chapter 4) brings with it the risk of unintentionally introducing other potentially invasive species, including non-native pathogens. The origin of disease outbreaks is difficult to determine, but for example, it has been argued that the epidemic that decimated the Caribbean keystone herbivore Diadema antillarum was due to a pathogen introduction via ballast water (Bak et al., 1984).
FUTURE GLOBAL CLIMATE PROJECTIONS
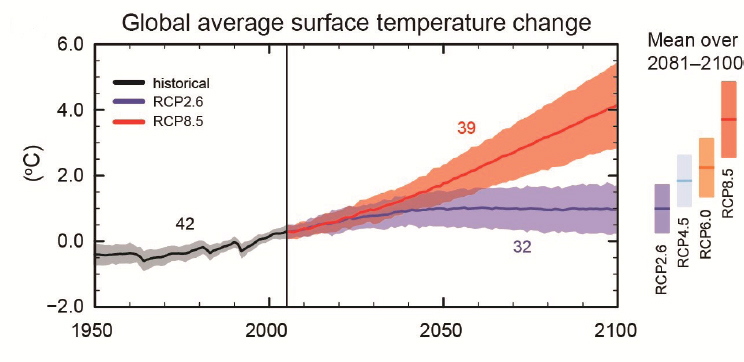
Global climate models provide the basis for future projections of coral health in response to climate change and the conditions that interventions will need to offset. The current suite of Intergovernmental Panel on Climate Change (IPCC) models (AR5) includes four emissions scenarios: a strong mitigation scenario (representative concentration pathway [RCP], 2.6), two intermediate scenarios (RCP4.5 and 6.0), and a “worst-case” business-as-usual scenario (RCP8.5). Global average surface temperature is projected to increase 2.6°C-4.8°C by 2100 under RCP8.5 and 0.3°C-1.7°C under RCP2.6 (Collins et al., 2013; see Figure 1.4). Even if all emissions were curtailed today, an increase of 0.3°C-0.7°C is still expected to occur by 2035 due to “committed” warming driven by the long residence time of CO2 in the atmosphere (Collins et al., 2013). While there are
limitations in how these models are applied to predict corals’ responses to climate change, they provide the best predictions of future SSTs. A fixed temperature-based threshold of 1°C-2°C above the historical summertime maximum is typically used in combination with future SSTs to predict bleaching (Donner et al., 2005; Frieler et al., 2013; Hoegh-Guldberg, 1999; Sheppard, 2003; van Hooidonk et al., 2013, 2014). The DHW cumulative stress index provides the basis for this empirically derived threshold (Strong et al., 2006).
Globally scaled models suggest that annual or biannual severe bleaching will occur by the 2050s under all emissions scenarios assuming no adaptation of corals (Donner et al., 2005; Frieler et al., 2013; van Hooidonk et al., 2013; see Figure 1.5). This finding is similar to the earliest bleaching regional models applied to the Great Barrier Reef and Indian Ocean (Hoegh-Guldberg, 1999; Sheppard, 2003). To avoid this outcome, the majority of the world’s coral reefs would require an adaptation rate of approximately 0.2°C-0.3°C per decade, with rates of up to 0.5°C-1.0°C per decade in certain regions (Donner et al., 2005). Recent studies have tested algorithms that explicitly account for historical thermal variability (Donner et al., 2011; Logan et al., 2012; Teneva et al., 2012) or incorporate specific mechanisms of adaptation and acclimation to rising temperatures (Baskett et al., 2009; Bay et al., 2017; Logan et al., 2014; Matz et al., 2018). These models generally find that even bleaching algorithms that include
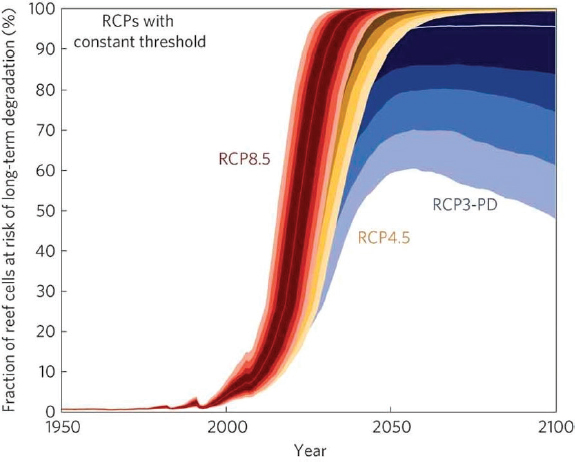
adaptive responses to warming predict corals only survive through 2100 under the lower emission scenarios (Bay and Palumbi, 2017).
The effects of OA on future bleaching frequencies (Frieler et al., 2013; van Hooidonk et al., 2014), the consequences of rising SSTs on coral disease (Maynard et al., 2015), and the effects of sea level rise (Perry et al., 2018) have also been modeled at a global scale. While higher latitude areas may have a relatively lower frequency of future bleaching, corals in these “refugia” may encounter lower calcification rates driven by greater decreases in aragonite saturation state (Guionotte et al., 2003; Kleypas et al., 1999; van Hooidonk et al., 2014). Maynard et al. (2015) found that disease is as likely to result in coral mortality as bleaching in the next few decades. Perry et al. (2018) modeled expected reef accretion rates under projected sea level rise for tropical western Atlantic and Indian Ocean reefs and found that most reefs are expected to experience a mean water depth increase of more than 0.5 meters by 2100 under RCP8.5. This change is projected to increase coastal flooding risk and change nearshore sediment dynamics. In addition to these global or multiocean basin forecasts, many regionally specific models have included coral communities with different thermal sensitivities, ecological interactions, evolutionary
processes, and multiple stressors to predict future coral population sizes, extent of coral cover, and/or bleaching frequencies (e.g., Anthony et al., 2011; Baskett et al., 2009; Bay et al., 2017; Matz et al., 2018). Ongoing development of ecologically and evolutionary mechanistic models may better help predict how coral communities will change in the future.
Applying IPCC-based global circulation model (GCM) projections to predictions of coral distributions incurs inherent uncertainties at local and regional spatial scales and interannual and decadal timescales (Donner et al., 2005; Logan et al., 2014; Stock et al., 2011; van Hooidonk et al., 2014, 2016). However, near-term climate matching bleaching predictions may be more reliable using dynamically and statistically downscaled model projections for specific regions (e.g., Caribbean corals, van Hooidonk et al., 2015). Nevertheless, increases in thermal stress on coral reefs are presently so rapid at the global scale that using the next generation of GCMs is unlikely to change general bleaching projections (Donner et al., 2018).
STUDY TASK AND APPROACH
This study committee has been tasked with reviewing and summarizing the state of science on genetic, ecological, and environmental interventions meant to enhance the persistence and resilience of coral reefs. In this interim report, the committee provides a review and summary of currently available information on the range of interventions present in the scientific literature. As part of this review, the committee has been asked to evaluate the state of readiness of the interventions. To that end, the committee has attempted to identify the current feasibility, potential scale, risks, limitations, and infrastructure needs for implementation for each intervention. The committee did not prioritize or make recommendations regarding implementation of the interventions; the use of one or more interventions is a complex decision dependent on social, political, and environmental factors as well as the progress of future research. The committee’s final report will build on this review to develop a science-based environmental risk assessment and decision framework to help inform actions to be taken by coral reef managers. As described in the committee’s task, this report addresses item 1, with the remaining items to be covered in a subsequent report (see Box 1.2). This study was requested and sponsored by NOAA with additional support from Paul G. Allen Family Foundation.
This report is informed by a review of current literature and public information-gathering efforts. The committee acknowledges that this is a fast-moving field, and has made efforts to consider all currently available information, but due to new and upcoming research efforts, this may not be an exhaustive reference. Strong attention has been paid to similar
efforts under way in other countries that are home to extensive reefs and strong research capacity, particularly in Australia. To explore this component of their task with the research and management community, the committee held workshops on May 31, 2018, in Miami, Florida, and August 28, 2018, in Honolulu, Hawaii, as well as a virtual webinar on August 2, 2018. The agendas and panelists can be found in Appendix B. During these sessions, panelists discussed with the committee the risks, benefits, and feasibility of implementing novel interventions, as well as the underlying science driving their potential for effectiveness.
Study Scope: Interventions That Increase Persistence and Resilience
The ability to resist or recover from disturbance, as well as the rate and pathway taken toward that recovery, are collectively termed resilience (see Box 1.3). As a concept, resilience can be applied to different levels of ecosystems. Individual organisms can show physiological or growth resilience via survival, sustained growth, and/or reproduction (fitness), for example. Populations can show resilience through the ability to recruit new individuals after a disturbance. Communities can show resilience in ecosystem traits such as productivity, diversity, trophic linkages, or sustained biomass through shifts in species composition. A key goal for any intervention is that corals become more resilient in the field than they would have been without it. This goal is a scale issue as well—individual corals placed on reefs might be more resilient, but they might not be common enough to provide resilience to the reef itself or ecosystem services that the reef provides. This report is structured to address the interventions that have the potential to increase resilience at each of these scales. Additional measures are considered in this report that may increase the persistence of coral in deteriorating conditions, although they do not bestow resilience to the coral and in fact, by reducing exposure to stress, may hinder natural adaptation. However, they are important to consider as part of a toolkit of options for increasing coral survival.
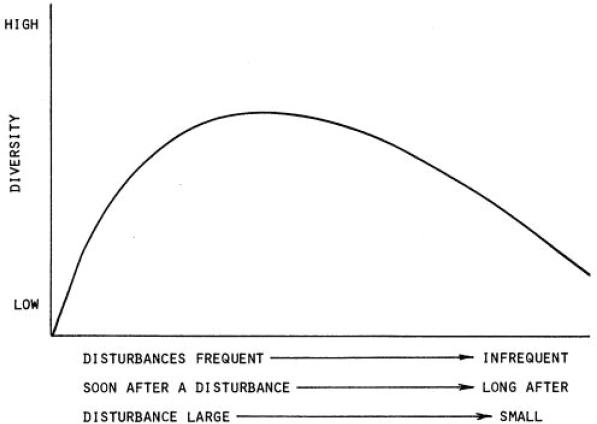
While a focus on enhancing stress-tolerant genotypes or species is inherent to many of the interventions described in this report as a way to maintain overall coral cover, maintenance of diversity of genotypes or species is another consideration for supporting the goal of increasing resilience of coral reefs and their associated ecosystem services. Traditionally, the complex diversity of coral reef ecosystems has been described as being driven by a balance between natural disturbance from storms, competition, predation, and other stressors and recovery. Joseph Connell developed the classic idea that complex communities are at their most diverse at an intermediate level of disturbance (Connell, 1978), and he
illustrated this in part with examples from coral reefs (see Figure 1.6). Additional anthropogenic disturbance upsets this balance, often driving the community toward lower diversity, and affecting species in different ways than do natural disturbances. Yet, even in the face of human disturbance, reef corals and the ecosystem they create can recover. Such recovery occurs at the colony level when corals grow back from bleaching, at the population level when coral recruits settle in a disturbed area, and at the community level when disturbed species assemblages regenerate some ecosystem functioning.
Diversity can increase population-level adaptive capacity and community-level resilience to disturbance (Levin and Lubchenco, 2008). At the population level, greater genetic diversity leads to greater expected adaptive capacity to future stress such as temperature stress (Baskett et al., 2009). In addition, diversity might enhance reproductive success (Baums, 2008; Miller et al., 2018), which might be particularly important at high-stress conditions given the potential for stressors to reduce genetic diversity within populations of corals (Baums et al., 2013; Meyer et al., 2009). At the community level, diversity is an important attribute that determines the resilience and robustness of marine ecosystems, in addition to redundancy, modularity, and strong feedback loops (Levin and
Lubchenco, 2008). For example, having a combination of slow-growing stress-tolerant species and fast-growing stress-susceptible species can theoretically increase the long-term likelihood of maintaining a coral-dominated state, especially under high disturbance levels (i.e., greater reversibility as defined in Box 1.3; Baskett et al., 2014). In addition to adaptive capacity or resilience to a single stressor such as thermal stress, having a diversity of genotypes or species might increase the likelihood of reef persistence when exposed to multiple stressors (e.g., disease, pollution, sedimentation, acidification, sea-level rise), especially under an uncertain future about the degree of change across stressors (Putnam and Gates, 2015) and uncertainty in the potential for tradeoffs in tolerance to different stressors. Furthermore, a diversity of corals and therefore reef structure might best support coral-associated assemblages (Graham et al., 2006; Gratwicke and Speight, 2005a; Wilson et al., 2006) and full ecosystem function and services (Bellwood et al., 2004). Ultimately, the decision to focus on increasing the cover of tolerant genotypes or species and/or to focus on maintaining diversity will be driven by reef management goals and values at the local level. Similarly, whether these goals are accomplished regardless of species composition or whether native diversity is a priority to maintain is a choice that reef managers will make in selection and implementation of interventions.
This study focuses on interventions that improve the persistence and resilience of coral species to global stressors linked to climate change, particularly ocean warming and increased frequency of bleaching events, as well as ocean acidification. Disease prevalence is expected to be augmented by environmental stress, and also increases the vulnerability of coral to the stresses from changing climatic conditions; thus, resilience to disease is also integral to this study. Mechanisms at the colony, population (e.g., increased recruitment), and community (e.g., support of species diversity) levels are considered. As directed by the statement of task, the committee does not emphasize management of local stressors. This is not because these stressors are unimportant, but rather because a great deal of attention and thought has already been paid to them, and control of these stressors is more manageable than control of global climate change.
Because corals are complex individuals with internal symbionts and a rich microbiome, we also review interventions that operate on these major features of coral biology. In particular, we consider interventions into the genetic makeup, the physiology, and the diversity of three different parts of the coral holobiome: the corals themselves, their algal symbionts, and the other members of their microbiome.
Aspects of environmental manipulation are included in the study for their potential ability to increase coral persistence by decreasing stress. Although these may not act on the resilience of the coral themselves, they may be an important tool in conserving reefs overall.
























