5
Environmental Interventions
While mitigation of global carbon emissions is the obvious global solution to climate change, even limiting human-induced surface warming to 2°C is unlikely to protect most coral reefs from an increased frequency and severity of bleaching events (Frieler et al., 2013). Even if biological and ecological interventions are able to enhance the thermal tolerance of some coral species by 1°C, predicted emissions scenarios in the coming decades may still lead to temperatures that exceed the thermal tolerance of most species, even under the Representative Concentration Pathway 2.6 by the Intergovernmental Panel on Climate Change (IPCC) (e.g., Anthony et al., 2017; Lough et al., 2018). Therefore, strategies that employ a portfolio of coordinated interventions including local cooling, shading, and amelioration of ocean acidification (OA) in combination with biological and ecological interventions may be necessary for building climate resilience in coral reefs.
A number of candidate interventions may provide such local, and in places regional, relief of climate-driven stress on reef ecosystems. These operate on a scale from microhabitats (meters) to potentially subregions (hundreds of kilometers) and serve a key purpose: reduce temperature and light stress during summer warming events where coral bleaching risk is significant (Eakin et al., 2009; Hoegh-Guldberg, 1999) and alter the chemical environment around coral reefs to reduce acidification impacts. Because OA can exacerbate thermal stress (Albright et al., 2016; Anthony et al., 2008; Kroeker et al., 2013; Mcleod et al., 2013; Reynaud et al., 2003), efforts to alleviate OA locally may also indirectly lower stress from
climate change. These interventions include atmospheric, sea surface, and water column reflection or absorption of light, advection of cooler reef waters around reefs, and lowering of water acidity and increasing water acid-buffering capacity. While the biological and ecological interventions considered in previous chapters act to enhance stress tolerance, the environmental interventions considered here all act to reduce or prevent stress exposure directly. In theory, if these environmental interventions can be made sufficiently effective at scale, then they can buy time for biological and ecological interventions to be developed further.
Here we review such interventions in light of projected climate change scenarios. Local cooling and shading interventions are in essence engineering solutions constrained by logistics, resources, and efficacy. Abiotic interventions to address OA fall in the category of engineering solutions using physical and/or chemical interventions that directly alter the ocean carbon chemistry toward a higher pH and aragonite saturation state (Ωa). Biotic interventions take advantage of the natural abilities of some marine species or biological communities to draw down carbon dioxide (CO2) and/or shift the seawater chemistry in a direction that ameliorates OA. The strengths of these interventions are in targeted asset protection, rather than broadscale prevention, at least without up-scaling of effort and potential risks. Such local- and regional-scale interventions are distinct from geoengineering initiatives that manipulate the atmosphere in an attempt to regulate the Earth’s global climate (e.g., Crutzen, 2006; Ricke et al., 2010; Robock et al., 2008a, 2008b).
SHADING OF CORAL REEFS
What It Is
Shading interventions are those that reduce the exposure of coral reefs to solar radiation, with the purpose of reducing light incidence and/or lowering water temperatures. Pacific coral reefs that were under cloud cover in the summer of 1998 avoided bleaching (Mumby et al., 2001). Bleaching risk has also been lowered by volcanic clouds (Gill et al., 2006) and marine turbidity (Cacciapaglia and van Woesik, 2016; Oxenford and Vallès, 2016). Engineered options for shading coral reefs may occur either in the atmosphere or in the water column over the reef. Induced cloud formation and/or brightening has been proposed as a means for atmospheric shading. Marine shading has been suggested to include increased turbidity, shading layers, microbubble ocean whitening, and wave lensing reduction. For each of the shading interventions that are not based on a fixed structure, the advection of the substance or effect into and out of the target area needs to be considered when evaluating scale and effectiveness.
Benefit and Goals
Shading of the atmosphere and ocean surface on or near coral reefs during warm summers can reduce bleaching risk by alleviating both heat stress and light pressure. The role that both heat and light stress play in coral bleaching is described in Chapter 1. Shading may cool the water column, and thereby cool the water around reef organisms. For example, if 30% of the sun’s irradiance can be absorbed or scattered in the atmosphere over a coral reef at noon during summer, it would lower the energy in the system by around 10 MJ m–2 d–1 (e.g., Masiri et al., 2008). Additionally, because light is a co-factor in the coral bleaching process (e.g., Lesser and Farrell, 2004), shading will directly lower coral bleaching risk. This is the rationale for the National Oceanic and Atmospheric Administration’s (NOAA’s) light stress product,1 which helps account for the total environmental stress that causes coral bleaching.
How to Do It
Below we present briefly the technical capability and challenges of each category of intervention. Generally, shading can occur in the atmosphere, at the water surface, or in the water column, at diminishing scale, respectively.
Atmospheric Shading
Clouds and aerosols in the atmosphere absorb and scatter solar radiation (Lacis and Hansen, 1974). Natural aerosols resulting in part from ocean primary productivity and ocean surface spray are key drivers of cloud formation and cloud albedo (McCoy et al., 2015). Artificially induced shading can be accomplished by introducing reflective particles in the upper atmosphere (above the troposphere) thereby inducing low-altitude cloud formation (Latham et al., 2012). Stratospheric cooling can be accomplished by injection of sulfur aerosols, akin to the natural process of volcanism (Crutzen, 2006), and this has been suggested as a possible strategy to mitigate coral bleaching (Zhang et al., 2018).
Low-altitude cloud formation above the air-sea boundary layer can also be promoted by seeding with aerosolized seawater particles near the marine surface (Latham et al., 2012). On a global scale, the resultant brightening, and hence reflection of solar radiation, can reduce globally averaged incoming radiant energy by up to 4.8 W m–2 at the top of the atmosphere (Alterskjær et al., 2012). This can produce a cooling effect
___________________
1 See https://coralreefwatch.noaa.gov/satellite/lsd/index.php.
sufficient to mitigate warming equivalent to a doubling of the atmospheric CO2 concentration relative to pre-industrial levels (Latham et al., 2013). However, sky brightening could potentially be targeted to specific reef areas to promote cooling (Latham et al., 2012) and reduce incident light during thermal anomalies. Previous efforts to model sky brightening have focused on areas where low marine clouds are abundant and would interact with injected particles (indirect brightening; Latham et al., 2012). However, more recent modeling suggests that injected particles themselves have a strong brightening effect (direct brightening; Ahlm et al., 2017), suggesting that even regions without abundant marine clouds, or when marine clouds are absent during bleaching conditions, could benefit from low atmosphere particle injection. Although typically referred to as “marine cloud brightening,” this means that “marine sky brightening” is a more appropriate term for sea spray climate or reef protection engineering (Ahlm et al., 2017).
Marine Shading
Induced turbidity Suspended particulate matter, generally referred to as turbidity, filters light in the water column, hence reducing benthic irradiance (Devlin et al., 2008; Falkowski and Raven, 1997). High turbidity on coral reefs is generally considered a stress factor as it can lead to a negative energy balance for autotrophic organisms, and settling particles may smother coral reef organisms (Fabricius, 2011; see Chapter 1 for further discussion). However, coral bleaching risk may be reduced by low or moderate turbidity (Anthony et al., 2007; Cacciapaglia and van Woesik, 2016) and potentially other reflective surfaces in marine waters (Seitz, 2011). Also, because suspended particulate matter may represent an alternative nutrient and energy source, it has the potential to lower mortality risk associated with bleaching (Anthony et al., 2009; Connolly et al., 2012).
Shading layers Both fixed physical shades, such as surface shade cloths (Rau et al., 2012), and temporary surface films have been used or proposed to reduce incident light on corals. Shade cloths have been shown to reduce bleaching in Acropora muricata, Pocillopora damicornis, and Porites cylindrical in American Samoa (Coelho et al., 2017). Shade cloths were used at a small scale (tens of meters) to reduce light stress on high-value reefs on the Great Barrier Reef (Rau et al., 2012). Also on the Great Barrier Reef, a project is trialing a reflective polymer surface film as a potential tool to shade coral reefs during summer doldrums.2 The buoyant, monolayer
___________________
2 See announcement at https://www.barrierreef.org/latest/news/reef-sun-shield-trialsshow-promise-to-prevent-coral-bleaching.
polymer film consists primarily of calcium carbonate (CaCO3) and is reported to be biodegradable and innocuous. Results of early trials in the Sea Simulator at the Australian Institute of Marine Science indicate that the film can reduce surface penetration of solar irradiance by up to 30% in calm sea states.
Microbubble ocean whitening Seitz (2011) has proposed the use of very fine bubbles or “highly dilute micron-radius hydrosols” to brighten surface waters and reflect light away from the seabed. In principle, this is the water-column equivalent to cloud brightening, and at fine local scale. The technique is proposed to reduce the amount of energy reaching the seabed by up to 100 W m–2 and at relatively low cost (Seitz, 2011).
Wave-lensing reduction Wave lensing is the focusing and defocusing of incident light on the ocean’s surface by the complex surface topography caused by waves. Wave lensing causes highly transient light flickering (within seconds) on shallow ocean floors, with order-of-magnitude increases and decreases in irradiance (Veal et al., 2010). This can be reduced over small areas by breaking up the water surface with techniques such as seawater sprinklers. The effect is only a modest decrease in mean down-welling photosynthetically active radiation and ultraviolet irradiance, but reduces irradiance variability. However, Veal et al. (2010) concluded from experiments that shallow-water corals are not negatively impacted by wave lensing at bleaching temperatures; therefore, this potential intervention will not be discussed further.
Current Feasibility
Techniques that shade coral reefs to cool water and lower photo stress are at various stages of feasibility that largely relate to their intended or potential scale. Atmospheric approaches (i.e., marine sky brightening) at the regional and local scales still need further technical development to be ready for implementation (Latham et al., 2012). Increased turbidity, shading layers, and ocean whitening could all technically be implemented now on small scales (less than 1 kilometer). However, while their effectiveness can be derived theoretically, they are untested as operational solutions.
Potential Scale
One advantage to marine shading manipulations for near-term thermal stress (see Frieler et al., 2013) is that they can be targeted in time and space to areas under acute thermal stress. However, the cost-benefit ratio of some techniques may be too high to scale up to regions or whole reefs.
An example is shading screens or structures; while they are effective at, for example, the scale of a tourism pontoon, the infrastructure required to scale up becomes both cost prohibitive and logistically infeasible. Some techniques may also be limited in spatial and temporal scale because of the dissipation of the effect after application. For example, surface polymer films may be advected away from a reef by currents after application and therefore require monitoring and maintenance during a potential bleaching event. Atmospheric shading from marine sky brightening and marine whitening, on the other hand, could be scaled up to local and regional spatial scales with few constraints. The limiting steps here are integrated systems for coordinated application, ideally linked to satellite mapping of cloud covers, computer models of winds and weather, and network control of dosing arrays.
Risk
A key risk from shading is light reduction to photosynthetic organisms, including corals. In deeper or turbid waters, phototrophic organisms may already be near the compensation point where photosynthesis is balanced by respiration. This can cause shallowing of the depth limit of corals and phototrophic organisms, such as seagrasses, under the area shaded (Latham et al., 2013) or complete restriction of some organisms from areas that are turbid (Bessell-Browne et al., 2017). For any strategy apart from fixed shading structure, it would be difficult to control the boundaries of the affected area in turbulent atmospheric and oceanic waters because of advection. Sedimentation is a common stressor on coral reefs and its use as a shading intervention would need to consider any unintended side effects such as enrichment with particulate nutrients or the smothering of benthic organisms as particles settle onto the seafloor. Bubbling would alter the gas balance, including CO2, in surface waters and such alteration of water chemistry could have risks if, for example, surface waters were enriched with CO2. Importantly, however, the downside risks of any of these shading techniques need to be considered in the context of the damage they may prevent during an acute heat wave. In other words, if shading can be kept short, their downside relative to upside risks may be both low and manageable.
Other risks from aerosol injection in the atmosphere include the impact of settling salt particles and changes in precipitation on adjacent habitats (e.g., including commercially and socially valuable areas such as agricultural lands). This review does not focus on global climate engineering, but more locally on atmospheric engineering specifically protective to coral reefs. However, the risks of shading may apply equally to global or regional/local strategies.
Another risk of shading (and also cooling, described in the next section), could be the reduced scope for natural adaptation to thermal stress. In other words, lowering local thermal stress by shading may limit a pathway for acclimation. If shading or cooling interventions cannot be sustained, unhardened coral fauna could face rapid onset of thermal stress. Effectively, once a cooling or shading regime is implemented in an area or a region, the effort requires sustained commitment. Last, and importantly, interventions to reduce heat and light exposure and support persistence are only capable of buying time while global mitigation efforts seek to stabilize the climate.
Limitations
The shading approaches are largely limited by scales of operation. Shade sails at high-value dive sites on the Great Barrier Reef were highly localized (meters), and the idea has not been pursued because their cost-benefit ratio is too high. Other techniques might be limited by their efficacy. For example, marine polymer films can reduce incident light by 30%, but shading experiments suggest that even at 50% and 75% light reduction they do not protect corals from bleaching during severe heat waves (Coelho et al., 2017). Another consideration is direction and rates of atmospheric and marine fluid flow, which limits the area affected and the residence time of the effect (Mark Baird, presentation to committee, 2018).
Infrastructure
The infrastructure needed to support marine shading is labor intensive and potentially costly. Small-scale interventions, such as physical barriers and microbubble generators, require boats for installation of equipment and potentially in-water manipulation by divers. Microbubble generators and methods to increase turbidity (e.g., sediment resuspension) require energy to drive mechanical pumps, agitators, or similar tools and this generates logistical challenges in the marine environments. Local, regional, or larger-scale sky brightening requires boats with pumping and dispersant systems. These boats may be autonomous with periodic human maintenance or constantly manned vessels.
Monitoring for efficacy and control of negative environmental impacts requires a different set of infrastructure. At the reef level, established methods of in-water direct and autonomous monitoring of corals will be needed to assess the efficacy of the intervention. Monitoring will also be needed for maintenance of the intervention itself. For spatially fixed interventions, such as shade cloths, this requires some onsite or remote monitoring. For interventions that advect in water or sky, monitoring is
needed to assess where to continue application to alleviate heat stress. Polymer films and microbubbles may require water sampling to establish the dispersal and lifetime of the polymer and bubble particles, unless the film can be detected from airborne sensors. Sky brightening may be detected from satellites or atmospheric sensors, but there is an issue of a standard from which to judge efficacy (Robert Wood, presentation to committee, 2018).
MIXING OF COOL WATER
What It Is
Thermal reduction can be accomplished by a process that replaces or dilutes warm water with cooler water. Active methods of bringing in cooler waters to thermally stressed reefs are possible in areas with access to deeper, cooler waters. These methods include mechanical pumps or self-sustaining processes (such as the differential in temperature between the depths and surface waters) to create artificial upwelling to replace shallow and warm with deeper and cooler water.
Benefit and Goals
The goal of cool water mixing is thermal stress reduction sufficient to prevent or lower the risk of coral bleaching. In natural systems, this is analogous to reduction of thermal stress by upwelling and enhanced vertical mixing (Glynn, 1996; Riegl and Piller, 2003).
How to Do It
Most of the experimental work on artificial upwelling has been done in the context of aquaculture and ocean fertilization (Pan et al., 2016). Pulling up of deep water can be accomplished through active methods, such as pumps and air lifts driven by nonrenewable energy (municipal electricity and fossil fuels) and renewable energy (e.g., ocean wave energy), or passive methods that take advantage of thermal and salinity differences between layers to generate a self-sustaining flow of water.
Current Feasibility
While technically feasible to create mechanisms for artificial upwelling, the efficacy of these approaches has not yet been tested fully. Underwater fans have recently been deployed in a trial phase on the Great
Barrier Reef under the Reef Havens project.3 The intent of the program is to bring up cooler water from deep to shallow waters and under doldrum conditions to provide local relief of peak sea surface temperature. This program targets selected, high-value reefs used by tour operators. Only one small-scale example of bringing up cool water to alleviate bleaching has been demonstrated.4
Potential Scale
Current initiatives such as the Reef Havens fan project are at the scale of one to a few hectares only. While it is conceivable that larger pump or fan systems can be made operational in places where there is access to cooler water at depth, the ability to scale up will be limited by infrastructure requirements. Like shading, cool water mixing is a temporary stress relief that can be focused during periodic times of environmental stress.
Risk
One of the technical risks associated with the pumping of deep, cooler waters to coral reefs in shallow water is that such artificial upwelling potentially leads to both nutrient and CO2 enrichment, exacerbating algal growth and OA (e.g., Feely et al., 2008; Leichter et al., 2003; Manzello, 2010).
Limitations
The surface mixing of cool deep water and warm surface water needs to be considered to understand the size of the water mass that needs to be cooled. The cooling load is the amount of water at a lower temperature and density needed to reduce the temperature of another warmer water mass at a given temperatures and density, and this then needs to be scaled to a reef (Mark Baird, presentation to committee, 2018). For the method to be effective, the reef cooling load needs to be determined a priori. This means taking account of spatial variability in processes that determine advection and residence time of the cooled water mass, such as local wind, distance to reef edge, and depth. Also, depending on the geographical and oceanographic setting on and around coral reefs, very deep water (more than 30-50 meters) may need to be pumped to achieve more than 1°C cooler water at the surface (Furnas and Mitchell, 1996). Thus, the cooling effect is dependent on a high degree of reef-specific characteristics
___________________
3 See http://rrrc.org.au/reef-havens.
4 See http://www.climatefoundation.org/coral-reef-cooling.html.
and may need to be applied on a reef-to-reef basis. In addition, because the present mechanisms to generate upwelling are small-scale and constrained by source water, the application of water cooling will likely be localized and not regional.
Infrastructure
The infrastructure needed for reef water cooling includes boats, pumps, and divers. Reducing the environmental impact of deep installations will need to take into account deeper water ecosystems and, thus, in many locations will require in situ biological assessments.
ABIOTIC OCEAN ACIDIFICATION INTERVENTIONS
What It Is
Abiotic OA interventions at the local reef scale act directly on the carbon chemistry of the seawater flowing over reefs. Interventions can be either chemical, involving the addition of a strong base to elevate pH or by interacting with the reef limestone, or physical, for example stripping CO2 from the water column.
Benefit and Goals
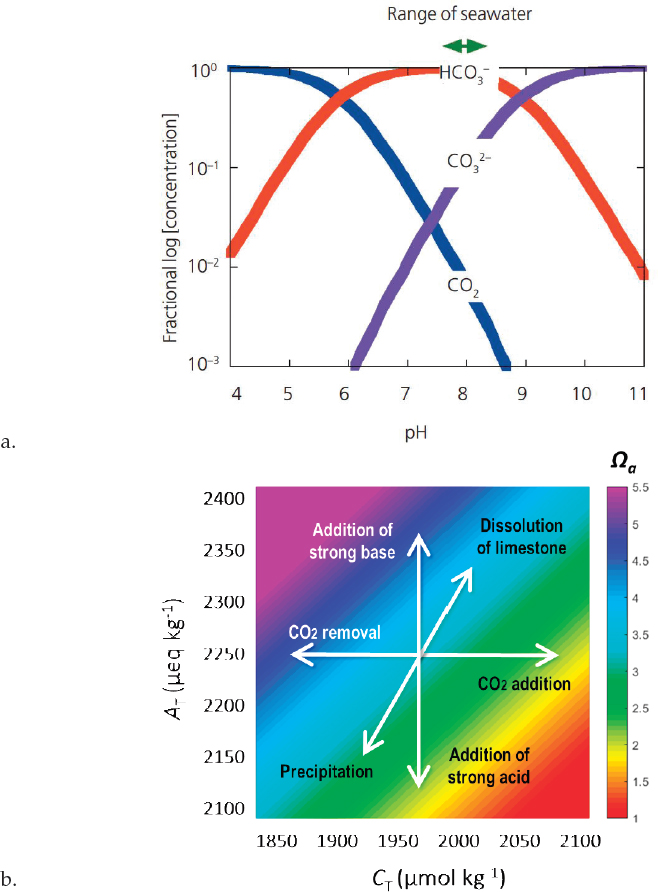
The fundamental goal of any OA amelioration technique is to shift the seawater carbon chemistry toward an increasing pH level (Raven et al., 2005; see Figure 5.1a) and more saturated aragonite saturation state (Kleypas et al., 1999). While pH is a key indicator of OA, the range of biological processes affected by changes in the ocean carbon chemical system are more fully represented by Ωa, which is a measure of the amount of carbonate ions in solution (Kleypas et al., 2006). This is critical because some OA manipulation techniques will affect pH but may only have limited effect on the carbonate system (Gattuso and Lavigne, 2009; see Figure 5.1b).
How to Do It
A number of different physical and chemical approaches show varying promise. We provide five examples to illustrate physical, chemical, and combined techniques.
Bubble stripping The use of bubble streams with low CO2 partial pressure to remove CO2 from seawater builds on the principle that CO2 in air equilibrates with CO2 dissolved in seawater (Zeebe and Wolf-Gladrow,
2001). Modeled experiments indicate that CO2 stripping using plumes of fine bubbles in a shallow-water environment can lower the CO2 partial pressure by around 20% over a 2-hour period (Koweek et al., 2016). Importantly, the effectiveness of CO2 removal is a direct function of the CO2 partial pressure in the bubble stream and the size distribution of bubbles. Challenges associated with this technique are adequate CO2 stripping of the bubble stream and the fact that CO2 is displaced from ocean to atmosphere.
Addition of strong bases Manipulating pH directly with the addition of a strong base (e.g., NaOH) forces a shift in the relative distribution of pCO2 and the carbonate ion concentration (Albright et al., 2016; Riebesell et al., 2010; see Figure 5.1a). While pH is increased, no CO2 is removed from the system. Again, exact dosing to match the environmental pH is challenging in a variable environment and sufficient dosing is a supply issue.
Addition of limestone The dissolution of CaCO3 powder has been proposed as a mechanism to enhance CO2 uptake by the global ocean, and further as an avenue for limiting ocean acidification (Harvey, 2008). The technique builds on the principle that added CaCO3 ions consume hydrogen ions, thereby elevating pH and aragonite saturation state. It does so primarily by elevating the total alkalinity (see Figure 5.1b). Two key challenges of the technique at any scale are (1) to match the limestone dosing with environmental chemistry, and (2) to source material adequately and sustainably.
Accelerated weathering of limestone This technique is a variant of the addition of limestone, but involves the use of CO2 to create a local environment of low pH around a CaCO3 source. The dissolution of limestone in turn leads to increased total alkalinity. The outcome is a net increase in aragonite saturation state (Rau et al., 2007) along the “dissolution of limestone” axis in Figure 5.1b. This operates at a small scale in the calcium reactors used in maintaining alkalinity levels in aquariums.
Electrochemical splitting of calcium carbonate Here, CaCO3 (for example, using deposits from rubble banks) is split between a cathode and an anode, forming calcium dihydroxide (Ca(OH)2) at the cathode and carbonic acid (H2CO3) at the anode. Reactions between Ca(OH)2 and dissolved CO2 then produce dissolved calcium bicarbonate (Ca(HCO3)2) (Rau, 2008). While the method shows promise, questions arise around the economics and scalability of electrochemical approaches.
Current Feasibility
All five methods above are feasible now at small scale. Aeration is already being used to remove pollutants in wetlands (Ouellet-Plamondon et al., 2006). Logistical constraints here would be the ability to pump enough CO2-stripped air into the water body upstream of the target coral reef. Injection of air with atmospheric-level CO2 will not ameliorate ocean acidification on reefs exposed to oceanic currents but may in areas where metabolic processes temporarily lower pH and Ωa (Koweek et al., 2016). Similar logistical constraints would apply to the other techniques, but rather than injecting air at the target site, the dosing of limestone powder, strong bases, CO2, or power present further complications and risks.
Potential Scale
All methods above operate at the local scale only—kilometers at best. The reality is that ocean acidification is an escalating global phenomenon. Furthermore, some coral reefs are exposed to vast volumes of oceanic water every day. CO2 extraction or chemical conversion would require consistent and homogeneous dosing of large amounts of CO2-stripped air, limestone, or strong bases. Even at such a small local scale, the resourcing, infrastructure, and maintenance required to consistently counteract ocean acidification would likely be unsustainable.
Risk
Bubble streams may carry the least risk because only air is injected into the marine environment. The modeling study by Koweek et al. (2016) was applied to a shallow, coastal embayment where metabolic processes can drive pCO2 levels to more than twice that of the atmospheric concentration during the night (Albright et al., 2015; Shaw et al., 2012). Under such circumstances, bubbling with unmodified atmospheric air can lower pCO2 in the marine environment with relatively low risk. However, if CO2 first needs to be removed from the airstream using chemical techniques (House et al., 2011) then environmental risks (e.g., spills) and cost are likely to increase.
Direct dosing with limestone or strong bases could represent high risk. For example, the spatial and temporal variability of the physiochemical environment of reef environments (e.g., Mongin et al., 2016b) means dosing in space and time will need to be anticipatory as well as responsive.
Limitations
Scale, logistics, resources, and infrastructure represent major constraints. In general, OA amelioration is limited to local intervention where the environmental setting (oceanography, hydrodynamics, and bathymetry) is such that flow direction is predictable, depth is shallow, and/or reefs are in an embayment where the seawater carbon chemistry can be modified cost efficiently. As carbon-removal techniques become more effective and cost-efficient (House et al., 2011), local control of seawater carbon chemistry over coral reefs might become more feasible, but will still be limited to the local scale and to a subset of oceanographic settings.
Infrastructure
The exact dosing of chemicals, air, or limestone to reefs in such a way that the marine carbon chemistry can be kept within a range that favors coral survival, growth, and reproduction requires precision technology on a massive scale. Even technologies that attempt this at the scale of tens of meters in aquarium, field, or large laboratory settings demand automated sensing, control, and dosing equipment typically at the scale of hundreds to millions of dollars (Dove et al., 2013; Kline et al., 2012).
SEAGRASS MEADOWS AND MACROALGAL BEDS
What It Is
Seagrass meadows and macroalgal (commonly known as seaweed) beds have the potential to draw down CO2 concentrations and elevate Ωa in shallow-water environments on or adjacent to coral reefs, in effect serving a localized carbon-sequestration function. The drawdown of CO2 by benthic primary producers is part of the carbon cycle in the oceans and may also play a role in global carbon sequestration (Krause-Jensen and Duarte, 2016).
Benefit and Goals
The motivation behind this inquiry into seagrasses and macroalgae as OA management tools is that waters over some seagrass beds have significantly higher pH and Ωa than oceanic waters during periods of high productivity, suggesting that natural OA refugia exist, at least temporarily (Manzello et al., 2012). These observations raise the idea that active promotion or spatial conservation planning of seagrass meadows or macroalgal beds near coral reefs could be an opportunity to identify
or develop such refugia as part of an intervention strategy to help sustain coral reefs under climate change.
Seagrass meadows may be more effective in elevating Ωa than macroalgal beds (Manzello et al., 2012; Unsworth et al., 2012). This is partly because seagrass rhizomes stimulate carbonate dissolution in the sediment pore water (Burdige and Zimmerman, 2002), hence elevating seawater alkalinity and, as a consequence, Ωa. Seagrasses can also store carbon for longer periods than macroalgae in their rhizomes, which macroalgae lack. Seagrasses therefore both draw down CO2 and increase alkalinity, thus driving up Ωa analogous to the combined macroalgae-sand scenario above (see Figure 5.2e,f). Also, because seagrasses are carbon-limited, whereas macroalgae are not, the effectiveness of seagrasses in elevating Ωa may increase with OA (Palacios and Zimmerman, 2007).
Contrary to seagrasses, some macroalgae are a food commodity used in aquaculture or farming (Msuya et al., 2007) and biofuel production (Wei et al., 2013). Macroalgal farming in areas adjacent to coral reefs may both generate a revenue stream that more than pays for the cost of the intervention, and maximize net Ωa increase by removing the fixed carbon before it breaks down. Without removal, decomposing macroalgae increase community respiration (Falter et al., 2011), reintroducing CO2 into the system, thereby lowering the efficacy of the approach.
How to Do It
Local reversal of OA using benthic primary producers works on a set of simple processes and objectives. Specifically, the aim is to increase Ωa, or counteract its decline, locally. This can be achieved via two key avenues. The first is to increase the ratio of benthic photosynthesis to respiration in areas upstream of the target coral reef (Anthony et al., 2011; Kleypas et al., 2011). The second avenue is by promoting net CaCO3 dissolution, as opposed to calcification, in upstream areas or on the reef itself (Anthony et al., 2013). The results of these biotic processes bear resemblance to the abiotic processes discussed above. The first process can be achieved directly by macroalgal beds or seagrass areas upstream of the target coral reef.
The extent to which these processes will work to achieve the objective (elevating Ωa of coral reef waters) depends on the extent to which photosynthesis exceeds respiration and dissolution exceeds calcification in the upstream area. A scenario that maximizes Ωa is captured by the following hypothetical: A coral reef is situated directly downstream of a large shallow area of a dense macroalgal bed (or seagrass meadow) during the day, and directly downstream of a large shallow area of reef sand with minimal content of organic matter during the night. The day/night shift
in upstream benthic type is here assumed to be driven by tides and/or a shift in wind direction.
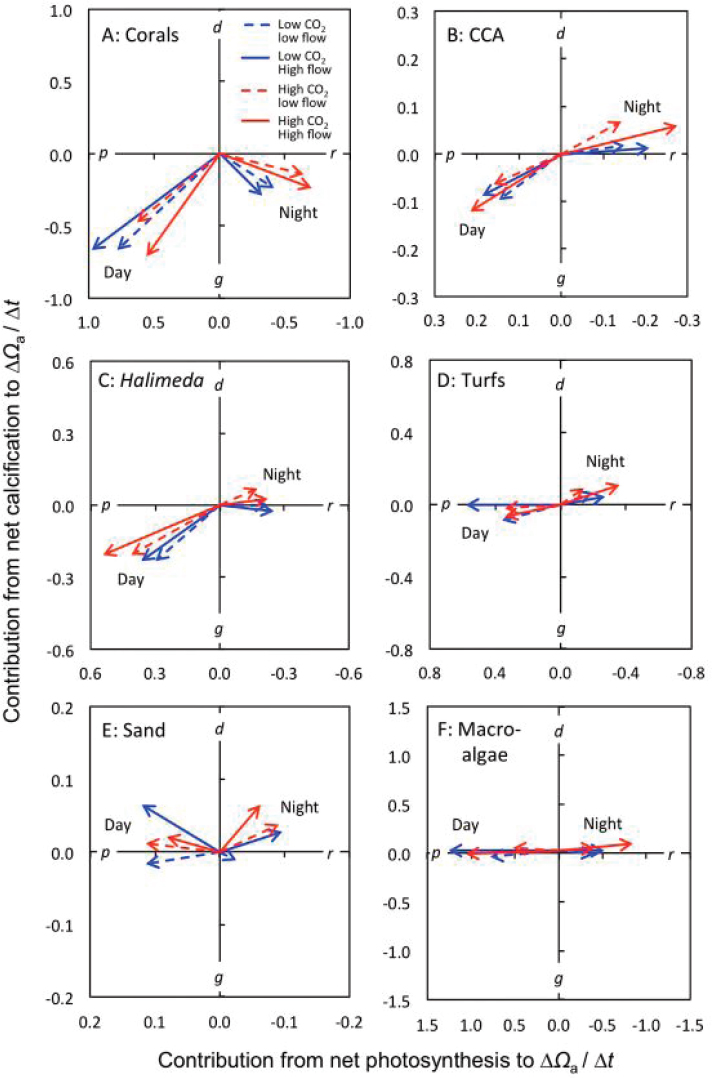
This near-optimal situation can be demonstrated by the results of flume experiments by Anthony et al. (2013) that examined carbon fluxes in six different benthic groups from shallow reef environments around Heron Island on the southern Great Barrier Reef (see Figure 5.2). The dense macroalgal bed in the experimental flume increased Ωa by nearly one unit per hour (at 1-meter depth) under high-CO2 concentrations and high-flow conditions and half a unit under low flow (see Figure 5.2f). Half of this gain was offset by algal respiration at night. The daytime gain by macroalgae could, in this experiment, fully offset the Ωa drop due to coral calcification during the day (see Figure 5.2a). Calcifying macroalgae such as Halimeda are less effective in elevating Ωa than noncalcifying macroalgae because their calcification also draws down alkalinity (see Figure 5.2c; see also Chisholm and Gattuso, 1991). At night, dissolution of crustose coralline algae (see Figure 5.2b) and coral sand (see Figure 5.2e) contributed most positively to Ωa under the acidified scenario. However, respiration by organic matter in the sand used in this experiment offset the Ωa benefit from dissolution. In summary, the example illustrates that a dense upstream macroalgal bed the size of the coral reefs (areas are constant across benthic groups in Figure 5.2) can fully offset coral-driven Ωa decline during the day. Also, water flowing from a large area of sand (e.g., a lagoon) at night may offset night-time Ωa decline driven by nighttime coral calcification and respiration. See Kleypas et al. (2011) for an additional case study.
The environmental context of the use if seagrass meadows or macroalgae as an OA management intervention is important to consider in implementing the strategy:
- Water depth. The drawdown of seawater CO2 will only be effective in shallow water because the ratio of photosynthesis to respiration, and hence CO2 fixation versus release, generally declines with water depth (Barnes and Chalker, 1990), specifically below the point of photo-inhibition (Hoogenboom et al., 2006, 2009).
- Water residence time. While fast-moving water promotes the exchange of CO2 (and other chemical species) and the biota (Mass et al., 2010; Shashar et al., 1996), slow-moving water allows for a longer period of CO2 exchange between the benthic primary producers and the overlying water column.
- Seagrass or macroalgal density. Optimal density will be a compromise between maximizing the three-dimensional surface area of photosynthesizing thallus or leaf surface per square meter of seabed and the extent to which crowding does not impede CO2 exchange.
- Fate of the fixed carbon. If seagrass or algal decomposition is allowed to occur within the management area, then the net Ωa change may be zero. A harvesting protocol can help increase efficacy (Mongin et al., 2016a), as can hydrodynamic settings where detritus is transported into deep water (Krause-Jensen and Duarte, 2016). If a harvesting strategy is part of an algal farm, then social and economic benefits can promote sustainability and upscaling.
- Geographical, oceanographic, ecological, geomorphological, and meteorological setting. A large number of processes will need to be aligned for biological OA management to be possible. Coral reefs directly exposed to a strong flow of oceanic water cannot be managed for OA locally. From a planning perspective, strategies that start by identifying natural OA refugia (Manzello et al., 2012) are likely to have higher efficacy than attempting to create such an alignment of factors by design or via engineering.
Current Feasibility
The main barrier to successful local OA management using seagrasses and macroalgae is the alignment of environmental factors that can transfer the benefits of OA reversal by the primary producers to the coral reef with maximum efficacy. Inshore settings where seagrasses are already elevating Ωa naturally above levels of the open ocean demonstrate feasibility and readiness (Manzello et al., 2012). In other settings, feasibility needs to be addressed with a more general question: Is the setting such that the water body “treated” by the primary producers flows over the coral reef consistently? If so, does the Ωa increase in that water body more than exceed the Ωa decline caused by the open-ocean OA influence and by reef calcification and metabolism?
Dynamic modeling allows for the formal design and quantitative testing of the efficacy of a local OA management solution. Specifically, linked biogeochemical and hydrodynamic models resolve local and potentially regional oceanographic drivers and their interaction with the benthic ecosystems (Mongin et al., 2016b; Schiller et al., 2014).
Potential Scale
While ocean acidification is driven predominantly by atmosphere-seawater exchange of CO2 across the global oceans, the role of benthic processes and coastal influences increases as one moves into shallow water and toward coastlines (Duarte et al., 2013). In shallow water, and especially coastal environments, the carbonate chemistry of seawater is
modified by benthic biogeochemical processes operating over multiple spatial and temporal scales (Albright et al., 2015, 2016; Takeshita et al., 2018).
The extent to which the local OA mitigation potential of either group of primary producers can be realized at scale depends on a number of regional and local factors. Most critically, does the per-area drawdown of CO2 by the seagrass meadow or macroalgal bed match the required Ωa increase on the coral reef after accounting for differences in the areas of primary producers and coral reef? If so, then local OA mitigation has the potential to work at any scale. OA mitigation can also potentially operate at the scale of meters by managing mixed coral-algal communities, for example in embayments or in areas with high seawater residence times.
Risks
Seagrass meadows are vulnerable to ocean warming and cyclones (Waycott et al., 2009) whereas macroalgae are generally more resilient (Diaz-Pulido et al., 2007). Furthermore, seagrass meadows have high conservation value (Cullen-Unsworth and Unsworth, 2016), whereas macroalgae are generally a competitor and indicator of reef decline (Bruno et al., 2009; Hughes et al., 1999; McClanahan et al., 2002; Mumby et al., 2007). This represents a dilemma from a local OA management perspective: On the one hand, the use of seagrasses in local OA management would serve two conservation objectives, but their climate vulnerability could jeopardize long-term OA management plans. On the other hand, macroalgae are relatively climate-hardy and can generate economic benefits if harvested, but they represent a risk to coral reefs. This risk is likely to be exacerbated under ocean acidification because it boosts the competitive dominance of some macroalgae over corals (Diaz-Pulido et al., 2011). Additionally, unless macroalgae are removed from the system, they will lead to reacidification and renutrification when they decompose, and under crowded conditions can lead to night-time anoxia (Greg Rau, presentation to committee, 2018).
Limitations
The efficacy of seagrasses and macroalgae as a local OA management tool is foremost limited by geographic setting, reef type, and oceanography (Mcleod et al., 2013). A small oceanic reef flushed by strong ocean currents will have minimal capacity for local OA mitigation. Conversely, a coral reef in a shallow embayment where water has high residence time or a reef in a coastal zone near dense seagrass beds will have the greatest scope. A key regional or local factor co-determining OA management
efficacy is water quality, for two reasons. First, the growth rate of macroalgae is nutrient (but not carbon) limited (e.g., Schaffelke and Klumpp, 1998). Therefore, the scope for local OA mitigation by means of macroalgal management is likely to be greatest in coastal, nutrient-rich coral reef waters. In reef systems such as the Great Barrier Reef, where inshore waters are influenced by land-use runoff, Ωa is already suppressed relative to that of the open ocean (Mongin et al., 2016b; Uthicke et al., 2014). Second, coastal shallow-water environments are the primary habitat for seagrasses (Grech and Coles, 2010). Although macroalgae are a ubiquitous component of coral reefs (McCook, 1999), their local abundance is driven primarily by a combination of grazing pressure (Mumby and Steneck, 2008) and nutrient supply (Burkepile et al., 2013).
The modeling study by Mongin et al. (2016a) illustrates the local OA management challenge on offshore reefs using reef macroalgae. They used a spatiotemporal model of water transport patterns to identify locations and harvesting schedules for macroalgal farms that maximize local Ωa increase on Heron Island Reef. They showed that an optimal spatial configuration of farms, seaweed density, and harvesting protocol would elevate Ωa by 0.1 over a 24 km2 reef area. While this result appears insignificant in the context of open-ocean OA projections exceeding one or two Ωa units (Cao and Caldeira, 2008), it is important to note that Mongin et al. (2016a) used small hypothetical algal farms (1.9 km2) to counteract OA in a much larger downstream area where calcification and respiration are driving Ωa in the opposite direction. Because Heron Reef is under strong oceanic influence and has high abundance of herbivores, the expected capacity for OA mitigation in this setting using macroalgae would be close to zero, so results from Mongin et al. (2016a) show surprising efficacy.
Infrastructure
Introduction of seagrass meadows and macroalgal beds relies on infrastructure commonly used for restoration efforts, including laboratories for propagule cultivation, and boats and divers for field transplants. Use of rhizomes has been found to be more successful than seeds or seedlings for restoration of seagrasses, along with the use of weights or staples to keep them in place (Katwijk et al., 2016). Macroalgae can be planted along submerged ropes to control their placement (Chung et al., 2013).




















