2
Macronutrient Requirements
Women are often under the impression that they are eating for two when they become pregnant. This mindset can be particularly problematic, given the prevalence of overweight and obesity. Coupled with this are changing dietary patterns in the population, which has implications for the quantity and quality of macronutrients consumed. Moderated by Patrick Catalano, professor and vice chair of obstetrics and gynecology at the Tufts University School of Medicine and maternal–fetal medicine specialist at the Tufts Medical Center, the first session of the workshop explored the evidence that has emerged regarding macronutrient requirements during pregnancy and lactation and their relationships with maternal health and infant growth. Highlights from the session presentations are presented in Box 2-1.
DIETARY FAT, FATTY ACIDS, AND THE SPECIFIC HEALTH EFFECTS OF OMEGA-3 FATTY ACIDS IN PREGNANCY AND LACTATION
Maria Makrides, theme leader for SAHMRI Women and Kids at South Australian Health and Medical Research Institute, opened her remarks by explaining that although the omega-3 long-chain polyunsaturated fatty acids (LCPUFAs) docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) can be oxidized for energy, research has largely focused on their bioactive roles. These roles include being a part of cell membranes, cell signaling, and inflammation resolution. Over the course of her presentation, she reviewed relationships between maternal omega-3 LCPUFA supplemen-
tation and various perinatal outcomes, including developmental outcomes, allergies, asthma, childhood growth, and prematurity.
Maternal Depression and Childhood Neurodevelopment
In the DHA to Optimize Mother Infant Outcomes (DOMInO) Study, nearly 2,400 pregnant Australian women less than 20 weeks gestation with
singleton pregnancies were randomized to receive either fish oil capsules containing 800 mg of DHA per day or control capsules containing a blend of vegetable oils with no DHA (Makrides et al., 2010). No statistically or clinically significant differences in postnatal depression were found between the two groups of women at either 6 weeks or 6 months postpartum. Differences between the groups in the developmental and intelligence quotients of the children of the women at 18 months, 4 years, and 7 years of age did not reach statistical or clinical significance (Gould et al., 2017; Makrides et al., 2010). Makrides said:
Given that most of those babies were born at term, who were largely breastfed with milk containing about 0.2 percent of DHA, it really does strongly suggest the current guidance is adequate and does not require extra supplementation in otherwise healthy populations to improve neurodevelopmental outcomes for children.
She also mentioned that this finding was consistent with recent systematic reviews.
Risk of Allergy and Asthma in Children
Makrides described a theory that posits that higher omega-3 fatty acid intake can decrease the conversion of the omega-6 fatty acid arachidonic acid to prostaglandin E2 (a pro-inflammatory compound). Decreases in prostaglandin E2 production would then reduce the synthesis of Th2 type cytokines and IgE antibodies (i.e., typical of an allergic inflammatory response). Conceptually, this theory could have implications for atopic responses, such as allergy and asthma.
Makrides contrasted the two largest studies that assessed the relationship between maternal omega-3 supplementation and allergy and asthma outcomes. The DOMInO Study followed children at high risk for IgE allergy. The prevalence of children with IgE-associated allergies was slightly reduced at 1 year of age among those whose mothers received DHA supplements during pregnancy; however, there was no effect in the prevalence of asthma or respiratory illness, nor a sustained effect in IgE or any other allergies at 3 or 6 years of age (Best et al., 2016; Palmer et al., 2012, 2013). A Danish study, called the Copenhagen Prospective Study on Asthma in Childhood (COPSAC), provided slightly different findings (Bisgaard et al., 2016). In the trial, 736 pregnant women were randomized to receive either EPA-rich fish oil capsules containing 2.4 g of omega-3 LCPUFA or control capsules containing olive oil but no omega-3 LCPUFA. Although there was no effect on eczema, children of women who were supplemented with the omega-3 LCPUFA had reductions in persistent wheeze or asthma over
the first 5 years of their lives, as compared to children of women who were in the control group. Makrides suggested that differences in study design might have contributed to the differences in findings across the two trials. For instance, the DOMInO Study assessed children at high hereditary risk and provided a lower dose of EPA as compared to COPSAC.
Size and Childhood Growth
To explore the relationship between omega-3 fatty acid intake and size and childhood growth, Makrides again compared results from the DOMInO Study and COPSAC. Rates of prematurity in the DOMInO Study were similar to the Australian national averages, and effects on birth weight were likely related to length of pregnancy, she noted. Maternal omega-3 supplementation during pregnancy did not lead to long-term effects on childhood growth. Children from both the intervention and control groups had similar body mass index (BMI) z-scores, with 22 percent exceeding the 90th percentile—similar to the Australian national average. In contrast, COPSAC found prematurity rates were lower and birth weights were higher than the Danish average in its study participants; again, birth weights were thought to be a function of pregnancy length. Modest effects on long-term childhood growth were reported. BMI z-scores were modestly higher in children of supplemented mothers compared with the control in COPSAC, but only approximately 10 percent of children had BMI z-scores above the 90th percentile. “We concluded that it might be too difficult to identify any modest growth effect in the long term in heterogeneous populations that are exposed to obesogenic environments,” said Makrides.
Prematurity
With approximately 15 million babies born preterm every year, prematurity is a global public health issue. In the DOMInO Study, maternal omega-3 supplementation reduced early preterm birth (< 34 weeks gestation) and increased postterm inductions and postterm, prelabor Cesarean sections (Makrides et al., 2010). These results suggest the intervention was influencing gestational length, shifting the distribution to the right, said Makrides. It was estimated that 87 women would need to be treated to prevent one early preterm birth, but only 25 women would need to be treated to cause one postterm induction or Cesarean section.
In 2018, Makrides and colleagues updated a Cochrane review that explored the relationship between omega-3 LCPUFA and pre- and postterm birth (Middleton et al., 2018). The systematic review included 70 randomized controlled trials with a wide variety of omega-3 LCPUFA doses and reflected nearly 20,000 women, primarily with singleton pregnancies.
Omega-3 LCPUFA interventions led to a 42 percent reduction in preterm births before 34 weeks gestation (based on 11 trials) and an 11 percent reduction in preterm births before 37 weeks gestation (based on 25 trials). The review also found that LCPUFA interventions resulted in a 61 percent increase in births after 42 weeks (based on 6 trials).
A recent blinded randomized controlled trial, Omega-3 Fats to Reduce the Incidence of Prematurity (ORIP), has further investigated this relationship in a population-wide approach (Makrides et al., 2019). Study participants, which included 5,544 women with singleton or multiple pregnancies, were randomized to either the omega-3 supplement or the control group. The intervention began before 20 weeks gestation and continued only until 34 weeks gestation, in an effort to prevent postterm obstetric interventions. The trial found that preterm births, postterm labor or Cesarean section, or gestational age did not differ between groups. When the Cochrane review was updated with these results and those of a study from China, the findings regarding risk reduction in preterm birth among women with singleton pregnancies remained, but it was not apparent for women pregnant with multiples.
Makrides and her colleagues performed a secondary analysis on the ORIP data using total omega-3 fatty acid, which is the biomarker with the strongest relationship to omega-3 status (Simmonds et al., 2020). Her group found reduction in early preterm birth was associated with higher baseline omega-3 status among control group participants, which Makrides noted was consistent with findings from epidemiological studies. Women who had a baseline omega-3 fatty acid status of less than or equal to 4.1 percent of total fatty acids and were supplemented experienced a 77 percent reduction in early preterm birth. Supplementing women who had a higher baseline omega-3 fatty acid status, however, may increase risk for early preterm birth. In an analysis of data from COPSAC, the benefits of supplementation were seen in women with baseline status of 2.7 percent fatty acids of DHA and EPA. Makrides stated that the findings indicate that women who have a replete or high omega-3 fatty acid status (4.9 percent or higher) should not receive supplementation. “This actually implies a narrower window of opportunity than we envisioned, especially in a population where prenatal vitamin and mineral supplements with DHA is relatively common,” she said. Makrides commented that a replete status can be achieved by regular, long-term consumption of fatty fish or an omnivorous diet with modest supplementation.
Future Directions
Makrides concluded her presentation by highlighting areas in which there remain unanswered questions. She noted that further exploration
into the usefulness of screening and monitoring omega-3 fatty acid status is needed. Determining if markers such as dietary intake or demographic characteristics can be used to predict status would also be of value, she said. Makrides also suggested additional work on omega-3 intake in women pregnant with multiples is warranted.
DISCUSSION WITH MARIA MAKRIDES
After her presentation, Makrides answered questions from the audience. Topics raised included additional analyses of the prematurity data and approaches to meeting dietary omega-3 needs.
Additional Analyses of the Prematurity Data
Catalano opened the discussion with Makrides by asking her if the finding regarding the relationship between omega-3 supplementation and prematurity was mediated through anti-inflammatory mechanisms. Cautioning that there are many causes of preterm birth and that omega-3 status is not the only contributor, Makrides thought that inflammation likely plays a role. An unidentified audience member questioned whether trans fatty acids could be interacting with omega-3 fatty acids. Trans fatty acid intake in Australia and concentrations in the blood are both very low, remarked Makrides. She offered that her group has investigated the possibility that arachidonic acid and linoleic acid, both omega-6 fatty acids, compete with omega-3 fatty acids, but neither fatty acid was related to the preterm birth outcomes. Leslie Myatt of Oregon Health & Science University asked if Makrides’s group was able to determine which type of preterm birth was being affected by omega-3 supplementation. Makrides said, based on the ORIP data, that omega-3 supplementation appears to reduce risk for both spontaneous early preterm birth and iatrogenic early preterm birth. Myatt also wondered if the preterm and neurocognitive outcomes had been stratified by sex, as they are known to be sexually dimorphic. Makrides indicated that neither outcome had shown a difference by sex.
Approaches to Meeting Dietary Omega-3 Needs
Leanne Redman of the Pennington Biomedical Research Center wondered what proportion of pregnant women are meeting omega-3 LCPUFA intake requirements and how intake can be assessed in a clinical setting. Makrides stated that various organizations recommend women consume 200–400 mg/day omega-3 fatty acids to be replete, and that most women do not achieve this intake level without some level of supplementation or use of fortified foods, unless they consume at least one fatty fish meal
per week. Regarding assessing omega-3 intake, she noted that dietary fish intake is modestly associated with omega-3 status.
A member of the webcast audience asked if the current 2015–2020 Dietary Guidelines for Americans recommendation of 8–12 oz of fish per week is sufficient. Makrides thought it would likely lead to total omega-3 status above 4.1 percent.
PROTEIN AND AMINO ACID REQUIREMENTS IN PREGNANCY AND LACTATION:
NEW EVIDENCE CHALLENGES CURRENT RECOMMENDATIONS
To provide context for his remarks, Rajavel Elango, associate professor in the Department of Pediatrics in the School of Population and Public Health at The University of British Columbia, explained that protein is needed throughout the life span for maintenance and during developmental stages for growth. Protein requirements have been defined as
the lowest level of dietary protein intake that will balance the losses of nitrogen from the body, and thus maintain the body protein mass, in persons at energy balance with modest levels of physical activity, plus, in children or in pregnant or lactating women, the needs associated with the deposition of tissues or secretion of milk at rates consistent with good health. (FAO/WHO/UNU, 2007)
Existing Dietary Reference Intakes for Adequate Protein Intake
Dietary Reference Intakes (DRIs) for protein were established in 2005 (IOM, 2005). Acknowledging that the evidence available at the time was limited, Elango described how the factorial method was used to derive the DRI recommendations for pregnant and lactating women. For pregnant women, the Estimated Average Requirement (EAR) for protein was set at 0.88 g/kg/day and the Recommended Dietary Allowance (RDA) was set at 1.1 g/kg/day. The EAR reflects the estimate that 0.22 g/kg/day of protein is needed for growth and tissue deposition during pregnancy, plus 0.66 g/kg/day of protein needed for maintenance; the RDA for pregnancy assumes a 24 percent coefficient of variation. The protein EAR and the RDA for lactating women are 1.05 and 1.3 g/kg/day, respectively. The EAR incorporates an estimate of 0.39 g/kg/day for milk production sufficient for the first 6 months of an infant’s life, along with the protein maintenance estimate of 0.66 g/kg/day.
Elango characterized the estimates for growth, tissue deposition, and lactation as being fairly well done, but he called into question the estimate of protein needed for maintenance. The 0.66 g/kg/day value was estimated
from a meta-analysis that used a linear regression model to fit data from nitrogen balance studies conducted in young men (IOM, 2005; Rand et al., 2003). Elango contended that the data would better fit with a two-phase linear regression model, which results in a higher estimate of nitrogen balance (Humayun et al., 2007). He further noted that the 2005 DRI report acknowledged that nitrogen balance studies were no longer considered the gold standard and alternative methods for determining protein adequacy were needed.
New Estimates of Protein Needs
A new methodology, called indicator amino acid oxidation, was developed to overcome the limitations assessing protein needs using nitrogen balance studies. The concept—which Elango attributed to Paul Pencharz, Ronald Ball, and colleagues—uses stable isotopes to trace the utilization of a labeled amino acid by the body. In this approach, oxidation of the indicator amino acid decreases with increasing intake until it reaches a plateau, or breakpoint, which Elango noted as representing the EAR (Elango et al., 2008) (see Figure 2-1). Based on indicator amino acid oxidation, the protein recommendations for adult males were estimated to be 30 percent higher than the EAR and the RDA values that were established in 2005 (Humayun et al., 2007). The methodology, which measures isotopes in the breath in a noninvasive manner, has also been used to assess protein requirements during pregnancy. Breakpoints were estimated to be 1.2 and 1.52 g/kg/day during early pregnancy (11–20 weeks gestation) and late pregnancy (30–38 weeks gestation), respectively (Stephens et al., 2015). Elango explained that the indicator amino acid oxidation data provides evidence that protein needs increase over the course of pregnancy and are higher than the existing, static DRI values, but still fall within the Acceptable Macronutrient Distribution Range (Stephens et al., 2015). Underscoring that the recommendations do not reflect a maximum intake, he further noted that the estimates are based on consumption of high-quality protein. Elango said there is a need to understand the protein demands of lactation, which have yet to be directly explored.
Elango and colleagues assessed the protein intake of 212 healthy pregnant women from Vancouver, Canada (Stephens et al., 2014). Intake was close to the protein requirements his group had estimated using the indicator amino acid oxidation method, with median protein intake of 1.5 and 1.3 g/kg/day for women at approximately 16 and 36 weeks gestation, respectively. Showing that there was normal pregnancy weight gain and healthy infant birth weight, Elango suggested that this level of protein intake did not appear to have adverse effects.
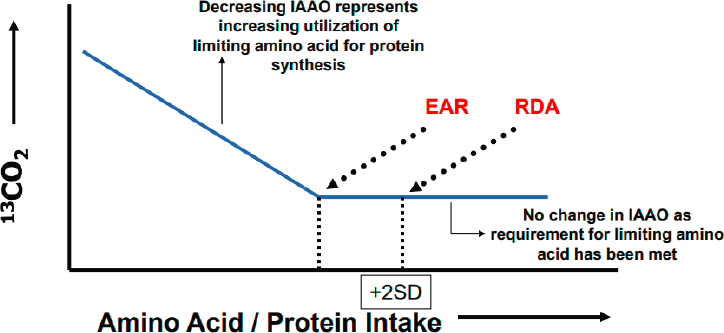
NOTE: EAR = Estimated Average Requirement; IAAO = indicator amino acid oxidation; RDA = Recommended Dietary Allowance; SD = standard deviation.
SOURCES: Presented by Rajavel Elango. Concept from Elango et al., 2008.
Protein Interventions During Pregnancy
Elango indicated that much of the available evidence on high protein intake during pregnancy is observational, and trials that have been conducted show different results depending on the macronutrient composition and total energy content of the intervention. Questions about the safety of high protein intake during pregnancy were raised in the 1980s, after a trial reported that a high-protein supplement (approximately 34 percent of the supplement’s energy came from protein) led to higher weight gain and higher neonatal death (Rush et al., 1980), but Elango emphasized that the results did not reach statistical significance. Data from the Danish National Birth Cohort indicate a possible U-shaped relationship between protein intake and low birth weight; women whose total energy intake from protein was less than 10 percent, or more than 20 percent, were at increased risk for having a low birth weight infant, noted Elango. A trial conducted in Gambia reported that women given a supplement of 1,017 kcal/day and 22 g/day of protein reported a 39–42 percent reduction in low birth weight infants (Ceesay et al., 1997). A meta-analysis by Imdad and Bhutta (2012) found that balanced protein-energy supplements (< 25 percent of the supplement’s energy coming from protein) led to a 32 percent reduction in the prevalence of low birth weight. The Women and Their Children’s Health (WaTCH) Study, an Australian cohort, found that pregnant women may achieve the largest variety of nutrients across food groups when they have moderate protein intake (Blumfield et al., 2012). Further underscoring
the importance of overall dietary context, Elango noted that the Montreal Diet Dispensary case series provided early evidence that food-based interventions, nutritional counseling, and other health benefits during pregnancy can be beneficial to the infants (i.e., normal birth weight) (Higgins et al., 1989; King, 2000).
Changes in Amino Acid Needs During Pregnancy
The body’s requirements for protein stems from its need for individual amino acids. The Food and Agriculture Organization of the United Nations (FAO) and the World Health Organization (WHO) have recommended that dietary amino acids be considered individual nutrients and requirements should be established for each (FAO/WHO, 2013). Acknowledging not all protein sources have the same amino acid profiles, Elango noted that plant-based proteins can be limited in certain amino acids. In the study of dietary intake among pregnant women in Vancouver, Canada, his group found that protein sources ranged in quality, with dairy being the leading source both among vegetarian and nonvegetarian participants during early and late pregnancy (Stephens et al., 2014). His group has begun to assess what the requirements for individual amino acids are across pregnancy. “While protein needs are increased early on, not all amino acids are simultaneously increased,” remarked Elango. The need for the amino acid lysine, for instance, is not particularly elevated during early pregnancy, but it increases in late-stage pregnancy (Payne et al., 2018). The need for phenylalanine, in contrast, rises in early pregnancy (Ennis et al., 2019). Investigation into the needs of other amino acid requirements during pregnancy are currently under way.
Applying his remarks to dietary patterns, Elango highlighted three common diets and their implications for protein intake. He indicated that plant-based diets have been shown to be safe and result in normal pregnancies, but he expressed concern for omnivorous women who switch to a plant-based diet during pregnancy and do not account for protein quality. Two recent systematic reviews have assessed the safety of consuming vegetarian and vegan diets during pregnancy (Piccolli et al., 2015; Tan et al., 2019), but neither could arrive at conclusive statements about harms or benefits owing to the limited number of observational studies with large heterogeneity. Elango characterized the ketogenic diet as being perfectly normal for the clinical management of seizures during pregnancy. However, he noted that under normal circumstances glucose and amino acids are the primary sources of fuel for a fetus, and said, “You cannot switch it out and consider ketones to be a source for the fetus in the long run.” Elango indicated that the safety of the ketogenic diet for healthy pregnant women is unknown, and caution needs to be exercised.
CARBOHYDRATES AND ENERGY REQUIREMENTS
Leanne Redman, professor at the Pennington Biomedical Research Center within the Louisiana State University system, set the stage for her remarks by describing some basic characteristics of carbohydrates. Used for energy by virtually all cells of the body, particularly the brain, carbohydrates consist of saccharide (sugar) units. The simple carbohydrates include monosaccharides (one sugar unit) and disaccharides (two sugar units), whereas complex carbohydrates include starches and polysaccharides. The number of saccharide subunits a carbohydrate contains determines its rate of digestion and absorption, as well as the rise in blood glucose concentrations after a meal.
Glycemic Index
Foods have been categorized and characterized based on the extent to which they raise blood glucose concentrations, using a system called the glycemic index (GI). Developed in the early 1980s, the GI represents the change in blood glucose concentrations after a person consumes 50 g of a single carbohydrate compared to the response in blood glucose after ingesting 50 g of pure glucose. High GI foods (i.e., GI > 1) tend to be monosaccharides, processed sugars, or refined sugars. By comparison, low GI foods (i.e., GI < 1) tend to be complex carbohydrates. “For chronic disease, it is not necessarily the amount of carbohydrates in the diet that is of concern or of importance. It is more the composition of these carbohydrates relative to their changes in postprandial glucose response,” stated Redman.
Dietary Reference Intakes for Carbohydrates and Total Fiber Requirements
Brain glucose utilization is considered the gold standard for measuring carbohydrate requirements and is fairly consistent over the life span, said Redman. The 2005 DRIs established estimates of carbohydrate needs based on this measure (IOM, 2005). For nonpregnant women, the carbohydrate EAR of 100 g/day was derived considering the average size of the female brain and average rate of brain glucose utilization after accounting for the amount of glucose that can be derived from protein turnover or from glycerol. The carbohydrate EAR for pregnant women of 135 g/day accounts for both maternal and fetal brain glucose utilization. The carbohydrate EAR of 160 g/day for lactating women was estimated based on maternal brain glucose utilization and on breast milk lactose composition. The RDAs for each of these life stages account for estimation error, using a coefficient of variation of 15 percent.
Turning to fiber, Redman noted that it tends to be found in low GI foods and has been linked to a range of health benefits, including delayed gastric emptying, improved insulin sensitivity, and prolonging satiation (or the feeling of being full). The 2005 DRIs established Adequate Intakes (AIs) for fiber based on epidemiological evidence that fiber intake reduces risk of coronary heart disease and some cancers and plays a role in weight management. Remarking that none of the outcomes used to derive the AI values were specific to pregnancy, Redman noted the total fiber AI for pregnancy was established at 28 g/day (14 g/1,000 kcals total energy intake).
Existing Recommendations Related to Added Sugars
Redman continued by discussing recommendations related to added sugars. She stated that the 2015–2020 Dietary Guidelines for Americans brought the attention to added sugar, which it defined as any sugars or syrups added during the processing of foods. The 2015–2020 Dietary Guidelines for Americans recommends that less than 10 percent of total energy intake should come from added sugars (HHS/USDA, 2015). She further noted that although the Academy of Nutrition and Dietetics did not provide quantitative recommendations for added-sugar intake during pregnancy, the organization did recommend that women reduce their intake (Procter and Campbell, 2014).
Carbohydrate Intake of Pregnant Women
Characterizing the available evidence on carbohydrate intake of pregnant women as sparse, Redman drew on a 2012 systematic review of 90 studies of more than 126,000 women (Blumfield et al., 2012). Mean carbohydrate intake among pregnant women was nearly 270 g/day. Although this intake exceeds the carbohydrate RDA, it fell within the Acceptable Macronutrient Distribution Range. Carbohydrate intake among pregnant women was found to be highest in the United States and Canada and lowest in Japan. Based on three studies, the systematic review reported that pregnant women in developed countries consume 50 g/day added sugars (Blumfield et al., 2012). A recent analysis of data from the National Health and Nutrition Examination Survey found that pregnant women in the United States consume approximately 85 g/day added sugar, which was nearly 15 percent of their total energy intake and higher than added-sugar intake among nonpregnant women (Cioffi et al., 2018). Redman emphasized that a substantial portion of added-sugar intake among pregnant women, approximately 39 percent, came from sugar-sweetened beverages. With respect to fiber, the 2012 systematic review found that pregnant women consumed markedly less than the AI, with a mean fiber intake of
approximately 19 g/day (Blumfield et al., 2012); fiber intake was not significantly different by socioeconomic status or stage of pregnancy.
Evidence Relating Carbohydrate Intake to Pregnancy Outcomes
Relationships between carbohydrate intake and pregnancy outcomes can be difficult to discern owing to differences in the proportion and quality of carbohydrates tested, stated Redman. One study reported that pregnant women who consume a low carbohydrate dietary pattern are at increased risk for gestational diabetes (Bao et al., 2014). Other studies have found a reduced risk of gestational diabetes when pregnant women consume the Dietary Approaches to Stop Hypertension (DASH) dietary pattern (which is low in added sugars and high in fruits and vegetables). Intake of the DASH dietary pattern has also been linked to reduced risk of subsequently developing type 2 diabetes among women who experienced gestational diabetes, as well as reduced risk of preterm birth, noted Redman.
Regarding a low GI diet, a recent Cochrane review did not find benefits related to gestational diabetes, gestational age, macrosomia, or various other pregnancy outcomes (Tieu et al., 2017). The authors of the systematic review concluded that the quality of the evidence was low to very low. Given this conclusion, Redman suggested it is premature to draw definitive conclusions about the relationships between low GI diets and pregnancy outcomes. She did, however, point to a pilot study that found a high carbohydrate, low GI diet lowered fasting blood glucose levels and reduced insulin resistance among women with gestational diabetes (Hernandez et al., 2016). Similar findings were reported in a recent systematic review, which found that women with gestational diabetes who consumed a low GI diet experienced reductions in fasting glucose levels, in need for medication, and in macrosomia (Yamamoto et al., 2018).
Energy Requirements During Pregnancy
Energy requirements increase during pregnancy to support maternal tissue development and fetal growth, but requirements vary by a woman’s prepregnancy weight status, physical activity level, and stage of pregnancy. Redman said:
Part of the variance here in energy requirements is not only the growth and development of maternal tissues, but we know that with excess weight gain … there is a tendency for the women to gain excess fat, which would then influence the calculations of energy requirements if we include these women in these calculations.
Total energy expenditure begins to increase at 13 weeks gestation in a linear fashion related to weight gain, resulting in an approximately 400 kcal/day increase by the end of the third trimester. All but 75 kcals/day of this increased total energy expenditure can be attributed to gestation weight gain. Redman added that physical activity levels tend to decrease during pregnancy.
The 2005 DRIs established the Estimated Energy Requirement (EER) (IOM, 2005). For nonpregnant women, the EER equation accounts for age, weight, height, and physical activity level. Women in their first trimester have no additional energy requirements. Additional energy expenditure and energy deposition are accounted for in the EER equations for the second and third trimesters, resulting in an increase of 340 and 452 additional kcals/day, respectively (see Box 2-2). A 2009 Institute of Medicine and National Research Council report on pregnancy weight gain upheld these estimates, but it also noted that energy needs may be lower for women with overweight or obesity (IOM and NRC, 2009). A recent study has found that women with obesity may not need to increase their energy intake during pregnancy, and they may even need a slight reduction in intake, to achieve the recommended weight gain, because of mobilization of their fat stores (Most et al., 2019).
Redman indicated that new, simpler equations were needed that reflect new evidence on the energy needs of women with obesity. In their current form, the equations do not consider variation in gestational weight gain or prepregnancy weight status. She also noted that the equations include data from women who had excessive gestational weight gain.
DISCUSSION WITH RAJAVEL ELANGO AND LEANNE REDMAN
Following Redman’s presentation, she and Elango participated in a discussion with the audience. Speakers explored factors affecting protein requirements, considerations related to the DRIs, and dietary intake to meet protein needs.
Factors Affecting Protein Requirements
To begin the discussion, Catalano asked Elango whether protein and amino acid needs are affected by a woman’s prepregnancy metabolic status. Elango confirmed its importance and said, “I do not think pregnant women who are already well nourished who started off in a fairly nourished state need additional protein beyond their normal consumption patterns.” Building on this concept, Erica Gunderson of the Kaiser Permanente Northern California Division of Research wondered if Elango had related the protein requirements to lean body mass, given how it affects metabolic state. He replied that his group has not explored this yet, and that they primarily study healthy pregnant women and adults who have normal body composition.
Considerations Related to Protein and Carbohydrate Intake Recommendations
Several audience members wanted to know more about the underestimation of the existing DRIs for protein adequacy. Regarding the reanalysis of the nitrogen balance data that were used to estimate the existing protein EARs for adults, Johanna Dwyer of the National Institutes of Health’s Office of Dietary Supplements asked if the data had been fit with splines. Noting that the publication included justification for why splines were not used, Elango mentioned that the two-phase approach that was selected leads to the identification of a breakpoint, which reflects the average requirement. Dwyer also wanted to know more about how the indicator amino acid oxidation methodology compared to the nitrogen balance approach. Elango described the two techniques as being conceptually similar. Both approaches look for a response to protein intake ranging from low to high. Nitrogen balance studies evaluate retention versus excretion,
whereas the indicator amino acid oxidation methodology only focuses on oxidation to reflect whole body protein synthesis, explained Elango. Noting that oxidation is a rapid process, and protein synthesis is a good indicator of the body’s needs, Elango stated that the indicator amino acid oxidation approach is a shorter, acute model. From a practical perspective, Redman wondered if increasing the protein DRIs for adequacy would affect birth outcomes. Elango emphasized that protein intake is one of a wide range of factors that affect birth outcomes, so he did not expect significant changes in a well-nourished population. He did, however, raise concerns regarding major shifts in dietary protein quality and amounts (e.g., a sudden shift to a plant-based diet), suggesting that pregnancy was not the time to make such dramatic dietary changes.
A question about placental needs over the course of pregnancy revealed differences in the methodologies for assessing carbohydrate and protein requirements. Redman reminded the audience that the current DRIs for carbohydrate needs were derived using brain glucose utilization estimates; she was unaware of any stable isotope studies that had been conducted in pregnant women to assess whole body oxidation of carbohydrates. By contrast, Elango stated that the benefit of the indicator amino acid oxidation approach was that it accounted for maternal, fetal, and placental protein use. He noted that the contributions of each component to protein needs, however, cannot be determined.
Two questions were raised regarding the practical implications of the existing recommendations. In reflecting on the 2015–2020 Dietary Guidelines for Americans recommendation, Barbara Laraia of the University of California, Berkeley, was concerned that 10 percent of energy from added sugars would be a sizable portion of a pregnant woman’s carbohydrate requirements, and she wanted to know how best to convey this recommendation. Agreeing that the added-sugar recommendation appeared liberal, Redman note that the 2015–2020 Dietary Guidelines for Americans provides an explanation for how it arrived at this value. Nevertheless, Redman thought more stringent added-sugar recommendations were likely needed, given their purported detrimental health effects. “We know that added sugars shouldn’t be there at all. The value should be zero really,” Redman said, although she was unclear how to best to establish added-sugar recommendations. Later, Redman posed a question to Elango, as to whether protein recommendations should be trimester specific or whether they should be static for ease of use. Elango was supportive of the trimester-specific recommendations, an approach that is used by WHO.
Plant-Based Dietary Intake to Meet Protein Needs
In response to a comment made earlier in the session, Anna Maria Siega-Riz of the University of Massachusetts Amherst asked Elango to discuss his concerns regarding pregnant women shifting to a plant-based diet, given Redman’s presentation that such dietary patterns have been associated with beneficial outcomes. Elango clarified that there is no evidence to suggest properly planned plant-based diets are harmful during pregnancy, but he indicated that making such recommendations can have broader implications that should be considered moving forward (e.g., climate change, sustainability).
A member of the webcast viewing audience wondered if shifts toward plant-based beverages affect protein intake. Acknowledging that our food supply is ever evolving, Elango explained that the protein content of plant-based beverages tends to be significantly lower than cow milk. Those that contain protein supplements may still not achieve the composition of cow milk. “Similar to how it is almost impossible to mimic human milk, I think it is also very hard to achieve the status of bovine milk, as well, with any of those [products],” stated Elango.
Focusing on vegetarian and vegan pregnant women, an unidentified audience member asked if there are specific foods that should be consumed to meet the changing needs for specific amino acids. Elango cautioned that more work needs to be done before such recommendations could be made. Underscoring the importance of a diet with a variety of protein sources, Elango indicated that there are likely minimal consequences of the increased amino acid needs for well-nourished women—his concern is for vulnerable women.
This page intentionally left blank.


















