More than 80,000 chemicals across multiple classes and types fall under the rubric of the Toxic Substances Control Act (TSCA),1 said Deborah Cory-Slechta, yet chemical exposures receive little attention due to several incorrect assumptions: (1) chemical exposures are less important than genes from a mechanistic perspective; (2) exposures are very low with small effect sizes and thus of little concern; (3) the Environmental Protection Agency (EPA), which is responsible for fulfilling the requirements of TSCA, is protecting the public, so there is little to worry about; and (4) so many chemicals exist that it would be nearly impossible to know where to start.
According to Cory-Slechta, these are all mistaken assumptions. With regard to genetics, she pointed out that in the 1990s, prominent biologists predicted that mapping of the entire human genome would reveal the causes of common and debilitating diseases, including common psychiatric disorders. However, what genetics has shown instead is that these disorders are usually caused by either rare mutations; interactions of multiple genes; or interactions with epigenetic, environmental, and microbial factors that exert their effects as early as during fetal development and across the entire life span (Berkowitz, 2020). (Gene–environment interactions are discussed further in Chapters 3–5.) Many of these risk factors, including environmental factors, affect the brain, she added.
__________________
1 For more information, go to https://www.epa.gov/laws-regulations/summary-toxic-substances-control-act (accessed August 12, 2020).
Moreover, said Cory-Slechta, humans are exposed to multiple chemicals through the air, from the soil through crops and meat, and from groundwater and surface water through drinking water and fish. EPA’s Toxic Release Inventory (TRI) Program2 records the release and management of certain toxic chemicals. It was established to provide a resource for researchers, policy makers, and the public. Maternal exposures may also result in exposure to the fetus during critical periods of development (Aylward et al., 2014) (see Figure 2-1). These include flame-retardant compounds, metals, pesticides, and herbicides.
Jennifer McPartland, senior scientist in the Health Program at the Environmental Defense Fund, added that for most chemicals in commerce, little information exists regarding potential hazards and extent of exposure, particularly as these factors relate to effects on the brain. Yet, she noted that new tools are available with the potential to fill these massive data gaps and complement some of the more traditional methods such as animal and epidemiological studies.
LEAD
The idea that exposures are too low to be of concern has also been repudiated, said Cory-Slechta. For example, people have been known since 2000 BC that lead—which she called “the poster child” of chemical exposures—has effects on the brain, yet not until 2012 did the Centers for Disease Control and Prevention decree that there is no safe level of lead exposure in the blood of children. Nonetheless, lead poisoning continues to kill children around the world, she said, noting that just in the past decade, several hundred children in Nigeria died from lead poisoning caused by metal smelting (Thurtle et al., 2014).
Adults are also susceptible to lead poisoning, said Cory-Slechta. Although the brain is the target organ for lead, it also accumulates in bone, where it will remain for decades, only to be released back into the bloodstream when the body requires extra calcium, such as during pregnancy and breast feeding. Once in the bloodstream, it can travel to the brain and other organs, and can also cross the placenta, exposing the fetus to toxic lead, she said.
Studies of lead poisoning have also repudiated the idea that effect sizes are too small to be of concern, said Cory-Slechta. A pooled analysis of eight research cohorts found an inverse relationship between blood lead level and IQ score and suggested there is “no evidence of a threshold for the adverse consequences of lead exposure” (Lanphear et al., 2005).
__________________
2 For more information, see https://www.epa.gov/toxics-release-inventory-tri-program (accessed August 11, 2020).
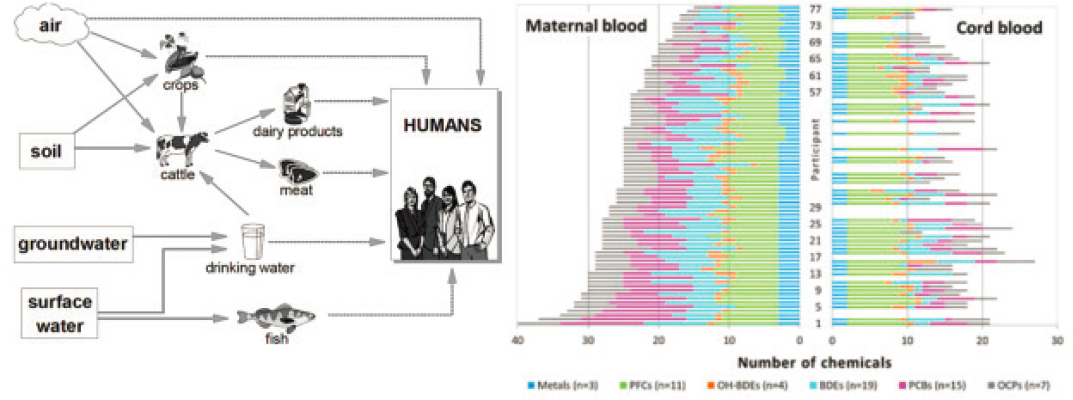
SOURCES: Presented by Deborah Cory-Slechta, June 25, 2020; European Commission, 2003 (left image); Morell-Frosch et al., 2016 (right image).
AGRICULTURAL CHEMICALS: HERBICIDES, PESTICIDES, INSECTICIDES, AND FUNGICIDES
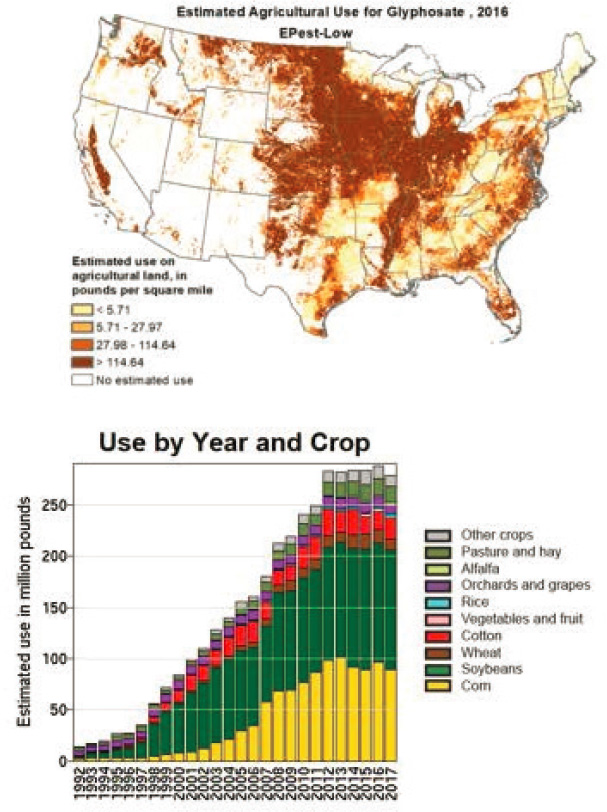
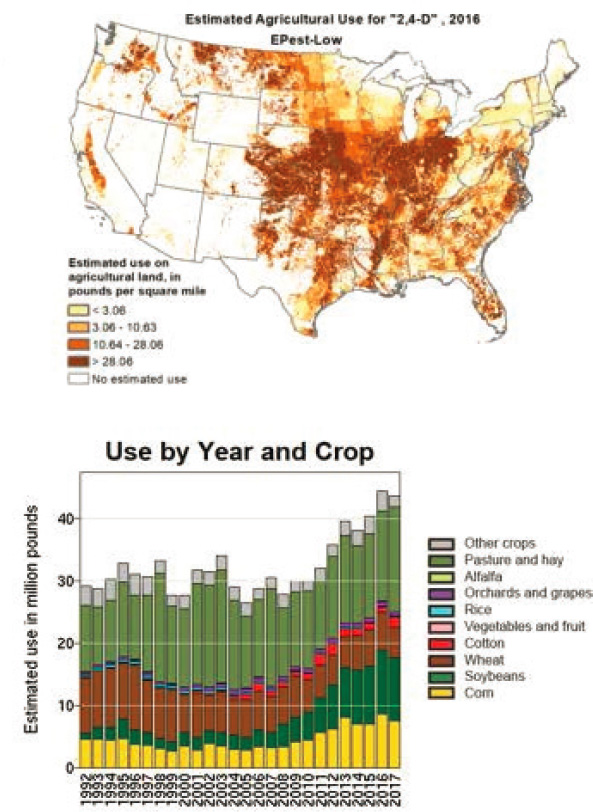
Exposure to agricultural chemicals has also increased markedly in recent years, said Cory-Slechta. Data found on the U.S. Geological Survey (USGS) website illustrates the heavy use of three herbicides—atrazine, glyphosate (known as Roundup), and 2-4-D (see Figure 2-2). In animal models glyphosates have been linked to oxidative stress, neurotransmitter alterations, and depressive-like behaviors (Cattani et al., 2017).
Pesticides represent “a unique environmental factor,” said Jason Richardson, professor of toxicology and associate dean for research in the Robert Stempel College of Public Health & Social Work at Florida International University. They are among the only chemicals intentionally put into the environment with the intention to kill by targeting circuits in the nervous system in insects that are also present in humans (Richardson et al., 2019). Moreover, they are particularly dangerous for children because many child- and toddler-specific behaviors such as playing close to the ground and putting hands to mouth may increase exposure, but also because children have immature physiological detoxification systems and are in a critical period of brain development (NRC, 1993), added Richardson.
The National Health and Nutrition Examination Survey (NHANES) has been documenting human exposure levels to pesticides and other environmental chemicals, said Beate Ritz, professor of epidemiology, environmental health sciences, and neurology at the University of California, Los Angeles. The NHANES 1999–2000 reported the presence of metabolites of organophosphate pesticides in more than half of the urine samples collected and chorpyrifos in more than 90 percent (Barr et al., 2004); the NHANES 2015–2016 reported that about half of the U.S. population aged 3 to 59 had urinary levels of neonicotinoid insecticides, which studies suggest may have neurodevelopmental toxicity (Kagawa and Nagao, 2018; Ospina et al., 2019).
INDUSTRIAL CHEMICALS AND FLAME-RETARDANT CHEMICALS
Statistics compiled by the Autism and Developmental Disabilities Monitoring Network3 indicate that between 2000 and 2016, the prevalence of autism has tripled, according to Tracey Woodruff. The incidence of other neurodevelopmental disorders and attention-deficit hyperactivity disorder (ADHD) has also increased during this time, she said. Some of these
__________________
3 For more information, see https://www.cdc.gov/ncbddd/autism/addm.html (accessed October 15, 2020).
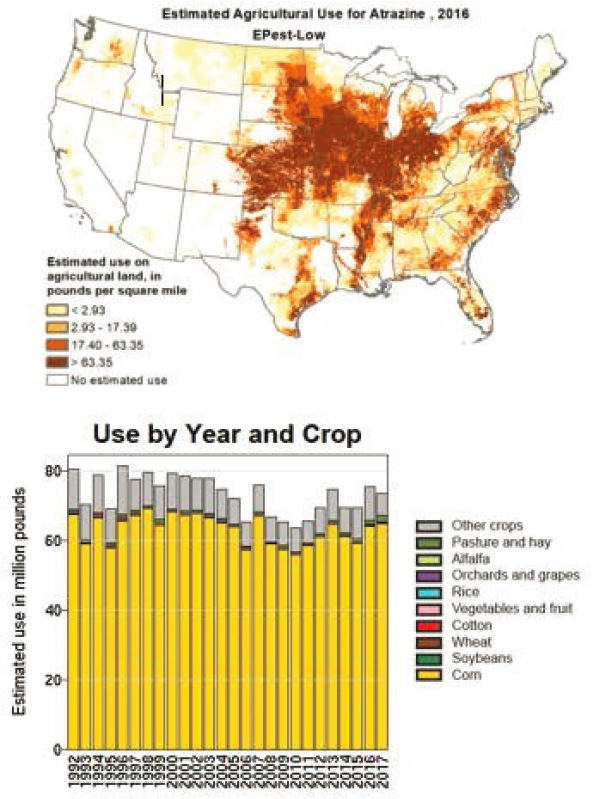
SOURCES: Presented by Deborah Cory-Slechta, June 25, 2020; USGS, 2020.
increases may be due to better diagnosis and awareness, but other factors are almost certainly contributing, particularly because genetics are unlikely to be changing substantially during this short time period, said Woodruff. What has changed, she said, is chemical production, which has increased more than 15-fold since the 1940s. Among the chemicals being produced at these high rates are industrial chemicals including perfluorinated chemicals such as Teflon, flame retardants used in consumer products such as mattresses and couches, plasticizers like phthalates, and chemicals used in food packaging, such as bisphenol A.
These chemicals are particularly problematic during critical periods of fetal brain development when they can affect different types of brain health effects that can result in effects on the brain that may not be apparent until childhood or later in life, said Woodruff. For example, David Bellinger, professor of neurology, psychology, and environmental health at Boston Children’s Hospital, Harvard Medical School, and the Harvard T.H. Chan School of Public Health, and colleagues estimated the contribution of several risk factors to IQ loss in a population of 25.5 million children. They concluded that just three environmental chemical exposures—lead, organophosphate pesticides, and methyl mercury—are together responsible for greater IQ loss than medical conditions such as preterm birth, neurodevelopmental disorders such as autism and ADHD, and socioeconomic and nutrition-related factors such as iron deficiency and non-organic failure to thrive (Bellinger, 2012).
Woodruff and colleagues studied the risks posed by flame-retardant chemicals such as polybrominated diphenyl ethers (PBDEs). The chemical structure of PBDE chemicals bears a strong resemblance to the chemical structure of thyroid hormone, said Woodruff. This has raised concerns that PBDEs can disrupt the normal function of thyroid hormone, including the important role this hormone plays in brain development. Studies by Woodruff find that pregnant women across the United States are ubiquitously exposed to PBDEs during pregnancy and pregnant women in California have had the highest levels compared to pregnant women worldwide (Woodruff et al., 2011; Zota et al., 2011). In human embryonic stem cell model of neurogenesis, PBDEs affect cytotoxicity and the function of neural progenitor cells and cause global changes in the expression of genes involved in thyroid hormone signaling, central nervous system development, oxidative stress, and cell cycle targets (Chen et al., 2019). Moreover, said Woodruff, these effects occur at relatively low exposure levels. A systematic review of human studies has also shown there is sufficient evidence that PBDE exposure is linked to reduced IQ (Lam et al., 2017).
In the early 1970s, California began requiring the use of flame-retardant chemicals in polyurethane foam applications, but banned the use of PBDEs in 2003 as production of these chemicals began to be phased out nationally. Woodruff’s research subsequently showed that the levels of PBDEs in pregnant women declined by 40 percent between 2008–2009 and 2011–2012 (Zota et al., 2013). Unfortunately, said Woodruff, PBDEs have been replaced with other flame-retardant chemicals, like organophosphate flame retardants, that may also be as toxic—resulting in regrettable substitutions.
AIR POLLUTION
In the past 10 years, air pollution has emerged as an important neurotoxicant, said Cory-Slechta. Air pollution consists of a complex mixture of particles and gases that come together to generate secondary pollutants. The particles, which occur in sizes ranging from ultrafine (< 100 nanometers in diameter) to fine (≤ 2.5 microns in diameter) to coarse (≤ 10 microns in diameter), adsorb many different contaminants such as trace elements, metals, and organic compounds, said Cory-Slechta. Ultrafine particles (UFPs) are the most reactive and are able to bypass the blood–brain barrier via uptake by the olfactory and trigeminal nerves into the brain. Dissolved, they can also cross the placenta to reach the fetal bloodstream, added Cory-Slechta. UFPs are so small that after birth they can get into the deepest areas of the lung directly into the blood, where they can bypass macrophages and travel via the vagal nerve directly to the brain stem. Yet, while EPA regulates fine and coarse particles, exposure to UFPs is not regulated.
Once air pollutants get into the body, they trigger biological responses such as increased oxidative stress and inflammation in the brain and periphery, which may contribute to the accumulation of neuropathology in the brain and the development of cerebrovascular disease, said Andrew Petkus, assistant professor of clinical neuropsychology at the University of Southern California.
ASSESSING THE RISK OF EXPOSURES
Regulatory agencies have developed risk assessment paradigms such as the four-step risk assessment process used by EPA, which characterizes the risk posed by a certain chemical by first identifying health problems caused by the particular pollutant and then assessing dose–response and extent of exposure over time, said Cory-Slechta (see Figure 2-3). Risk management decisions, however, consider not just the risk characterization, but economic and political considerations as well, she said. Furthermore, this paradigm considers just one chemical at a time, when in reality humans are exposed to multiple different chemicals simultaneously.
Consideration of concurrent and sequential exposures is necessary to establish a framework for interaction effects of exposures—both genetic and environmental, said Cory-Slechta. Moreover, these exposures occur in different social and economic contexts, she said. For example, exposure to air pollution is nearly always highest in communities of color and low socioeconomic status (Cushing et al., 2015; Johnson et al., 2016; Mullen et al., 2020). Lead exposure also occurs in the context of the stress of pov-

SOURCES: Presented by Deborah Cory-Slechta, June 25, 2020; EPA, 2020; NRC, 1983.
erty, where by comparison to higher socioeconomic status communities, residents may be exposed to violence, neighborhood crime, poor education, resource and economic deprivation, inadequate prenatal care, and other stressors, said Cory-Slechta. This is important, she said, because most of EPA’s Toxic Release Inventory (TRI) facilities are located in areas with the highest percentage of non-white populations.
According to Cory-Slechta, these various stressors and risk factors may actually affect the same biological targets. For example, both lead exposure and prenatal stress are known to act on the hypothalamic-pituitary-adrenal axis and the mesocorticolimbic systems of the brain. Therefore, it is imperative when studying chemical exposures in animal models to provide realistic simulations of human environmental conditions, she said.
Cory-Slechta and colleagues have, nonetheless, learned a great deal about the combined effects of exposure to lead and prenatal stress by studying behavior and neurochemistry in animal models (Cory-Slechta et al., 2012; Virgolini et al., 2008). The interaction of lead and prenatal stress increased the rate of both behavioral and neurochemical effects in a way that would not have been seen if the two exposures were studied individually, said Cory-Slechta. It is likely, she said, that even when exposures affect the same system, they may do so through different mechanisms. The implication of that scenario is that homeostatic mechanisms that might have reregulated one component of a system may not be sufficient if multiple chemicals are acting on different components of the system.
This page intentionally left blank.












