Richard Woychik, director of the National Institute of Environmental Health Sciences and the National Toxicology Program, noted that while there have been and will continue to be many exciting developments in genetics and genomics, “people can get sick because of environmental exposures, and it’s not just because of sequence variations in the genes they inherit from their parents that predispose them to disease.”
He said there have also been many exciting developments in environmental health sciences over the past several decades. Moreover, he said, gene–environment interactions make it necessary to look at both genomic and environmental factors associated with neurotoxicity.
For example, it has become increasingly clear that low-level chronic exposures and windows of exposure are critically important for human health, including for neurological disorders; that there are interactions between biological pathogens and environmental exposures; and that the microbiome plays an important role in how the body deals with environmental toxicants, said Woychik.
THE EXPOSOME
Given the fact that disease phenotypes are the product of both genetic and environmental effects, Christopher Wild introduced the concept of the “exposome” in 2005 to capture the totality of environmental exposures that impact human health, in much the same way as the genome captures
the totality of genetic effects that contribute to disease (Wild, 2005). Rappaport and Smith advanced the idea in an article in Science (Rappaport and Smith, 2010), and the National Academies of Sciences, Engineering, and Medicine Standing Committee on the Use of Emerging Science for Environmental Health Decisions convened a public meeting on the topic (Brown, 2012). As the idea of the exposome began to take hold, Gary Miller, vice dean for research and strategy innovation and professor of environmental health sciences at the Columbia University Mailman School of Public Health, said he began thinking about it in the context of the research he was doing at Emory University. Over the next few years, he and colleague Dean Jones developed the concept further (Miller and Jones, 2014). Miller noted that the exposome is not restricted to chemical exposures, but also integrates ecosystems, lifestyle, and social factors (Vermeulen et al., 2020) (see Figure 3-1).
Miller and colleagues developed a platform to measure the exposome in biological samples using high-resolution mass spectrometry and different
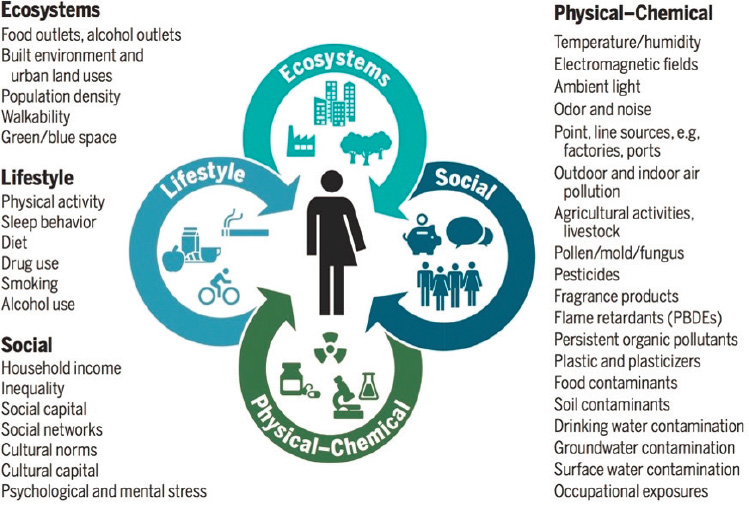
NOTE: PBDE = polybrominated diphenyl ether.
SOURCES: Presented by Gary Miller, June 25, 2020; Vermeulen et al., 2020.
separation techniques to enable measurement of thousands of exogenous chemicals and endogenous metabolites (Jones, 2016). The idea was to standardize these systems and quantify thousands of compounds in an untargeted manner to learn more about both known and unknown environmental chemicals. For example, in a study of plasma from Alzheimer’s disease (AD) patients and controls, three non-medication-related metabolites were identified that correlated reproducibly with AD biomarkers. One of them was an unknown halogenated compound that indicates an environmental origin and another (piperidine) a pepper derivative that indicates a dietary origin (Niedzwiecki et al., 2020).
In a collaboration with Richard Mayeux, chair of neurology at Columbia University, Miller is using the mass spectrometry approach to evaluate longitudinal blood samples from the multiethnic cohort enrolled in the Washington Heights-Inwood Columbia Aging Project (WHICAP) study. Interestingly, while genome-wide association studies (GWASs) have failed to pick up differences among ethnicities, high-resolution mass spectrometry identified ethnicity-related differences in metabolites. These differences help reveal why certain ethnic groups are at increased risk of AD (Vardarajan et al., 2020).
The exposomic approach has also shown promise as a tool for testing hypotheses in animal models, said Miller. For example, colleagues in his lab have exposed C. elegans to different mixtures and combinations of compounds and then looked at thousands of metabolomic features using mass spectrometry. Agreeing that more animal studies combining exposures and neurosciences are needed, Brenda Eskenazi, the Jennifer and Brian Maxwell Professor Emeritus of Maternal and Child Health and Epidemiology and director of the Center for Environmental Research and Children’s Health at the University of California, Berkeley, noted that aligning animal and in vitro studies to human studies is also critical.
The beauty of high-resolution mass spectrometry approach for studying the exposome is that it works with multiple biological matrixes as well as in dust, water, and other environmental samples, said Miller. Thus, it provides the field with a tool that facilitates a systematic, comprehensive, and unbiased assessment of environmental contributors to multiple conditions, including neurodegenerative, neuropsychiatric, and neurodevelopmental disorders.
GENETIC FACTORS
Gene–environment interactions are implicated in many neurodevelopmental and neurodegenerative disorders, including autism, Parkinson’s disease (PD), and AD. For example, over the past few years, hundreds of gene mutations have been linked to autism, said Mark Zylka, director of
the Neuroscience Center at the University of North Carolina at Chapel Hill (O’Roak et al., 2014; Pinto et al., 2014; Stessman et al., 2017). However, he said, heritability studies clearly show that environmental influence and environmental risks also play a role. The importance of this lies in the fact that unlike inherited mutations that cannot be avoided, if a chemical exposure can be identified, there is the potential to minimize it or eliminate it during critical periods of brain development and thus to eliminate environmentally induced forms of autism.
Moreover, he said many environmental toxicants target the same molecular pathways that are affected by de novo gene mutations linked to autism. Beate Ritz added that genetic variation in many different biological pathways may also influence susceptibility to exposure from pesticides and increase the risk of developing neurodegenerative diseases (see Figure 3-2).
Among the top pathways linked through genetics to autism that are also known to be affected by chemicals in the environment are the Wnt/β catenin pathway, genes associated with synaptic function, and genes associated with neuroinflammation. If a chemical targets one of those pathways, it may be able to increase the risk of autism, said Zylka. His lab and others are now using high-throughput screens and transcriptomic approaches to try to define chemicals that target these specific pathways, he said.
Multiple factors also contribute to the etiology of PD, said J. Timothy Greenamyre, Love Family Professor and vice-chair of neurology, chief of movement disorders, and director of the Pittsburgh Institute for Neurodegenerative Diseases at the University of Pittsburgh. At one end of the spectrum are monogenic causes. Despite the fact that about 20 genes have been implicated, monogenic causes together account for only about 10 percent of cases, said Greenamyre. At the other end of the spectrum are clearly defined environmental or toxicant exposures, including pesticides and industrial solvents that have been associated with PD. For most people with PD, Greenamyre believes the disease arises from their composite genetic makeup of resistance and susceptibility genes combined with a lifetime of environmental exposures.
Wnt/β Catenin Signaling Pathway
At least 19 percent of all genes linked to autism are in the Wnt signaling pathway, said Zylka (Bernier et al., 2014; Packer, 2018). One of the most commonly mutated genes in idiopathic autism is the chromodomain helicase DNA-binding protein 8 (CHD8). CHD8 regulates transcription by altering chromatin structure (Thompson et al., 2008) and has been associated with cortical overgrowth and enlarged brains, which is a common phenotype in many individuals with autism, said Zylka (Bernier et al., 2014). The antiepileptic drug valproate also activates Wnt signaling, and
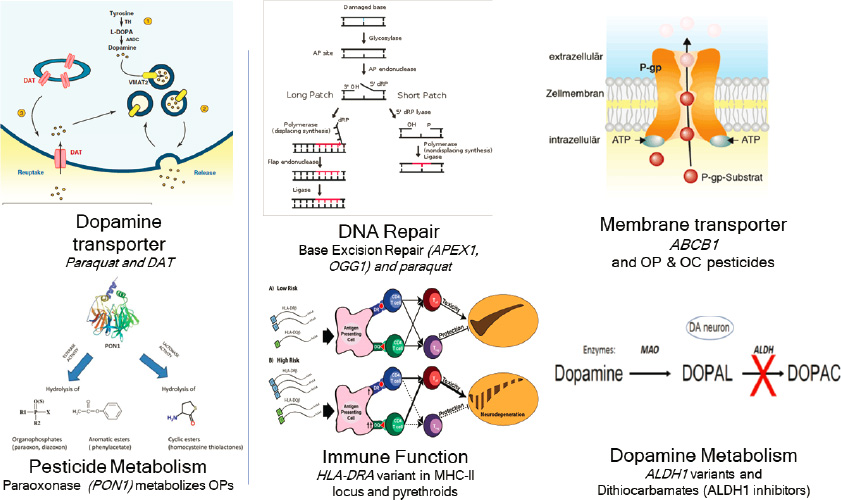
NOTES: Different pesticides have been linked to neurodegenerative disorders through multiple genetic mechanisms, including DNA repair, immune function, dopamine metabolism, and membrane transport; and by genetic variants in the dopamine transporter or enzymes that metabolize pesticides. ABCB1 = ATP binding cassette subfamily B member 1; ALDH1 = aldehyde dehydrogenase I; APEX1 = apurinic/apyrimidinic endodeoxyribonuclease 1; DAT = dopamine active transporter; HLA-DRA = major histocompatibility complex, class II, DR alpha; MHC-II = major histocompatibility complex II; OC = organochlorine; OGG1 = 8-oxoguanine glycosylase; OP = organophosphate.
SOURCE: Presented by Beate Ritz, June 25, 2020.
maternal use of the drug has been linked to an elevated risk of autism as well as increased brain growth (Christensen et al., 2013). Valproate thus represents an epidemiologically verified environmental risk, said Zylka. His lab is now using a collection of human neural progenitor cells, mouse cortical neurons, and other types of cells to examine how valproate and other chemicals affect signaling pathways in neural and other cells with common genetic variants associated with autism.
Synaptic Function
Autism-linked genes associated with synaptic function also are beginning to reveal clues about environmental risks, according to Zylka. Mutations in one of these genes, SCN2A, which is a voltage-gated sodium channel, have been shown to dampen or eliminate the function of the channel (Ben-Shalom et al., 2017; De Rubeis et al., 2014). Similarly, agricultural pesticides called pyrethroids, which have been linked epidemiologically to increased risk of both autism and attention-deficit hyperactivity disorder (ADHD), also inactivate this channel (Furlong et al., 2017; Shelton et al., 2014).
One of the leading mechanisms by which lead exposure impairs brain function is through its effects on synaptic function. According to Tomas Guilarte, dean of the Robert Stempel College of Public Health & Social Work at Florida International University, lead is a potent and selective non-competitive antagonist of N-methyl-d-aspartate (NMDA) receptors. Expression of NMDA receptors in the hippocampus is critical for learning and memory, said Guilarte. By inhibiting the NMDA receptor, lead exposure impairs synaptic plasticity and long-term potentiation, resulting in deficits in cognitive function (Nihei et al., 2000). In hippocampal neuron culture, this effect can be mitigated or reversed by exogenous addition of brain derived neurotrophic factor (BDNF) (Neal et al., 2010). A BDNF mimetic called 7,8-dihydroxyflavone, which is abundant in the human diet, has also been shown to reverse the effects of lead exposure on synaptic function (Zhang et al., 2018).
LRRK2
The most common autosomal dominant form of PD is caused by mutations in a gene called leucine-rich repeat kinase 2 (LRRK2) (Li et al., 2014). Many mutations in LRRK2 that result in increased activity of the kinase have been associated with PD; however, Greenamyre said other factors may also interact with LRRK2 and influence an individual’s risk of developing PD. His laboratory has been exploring the role of LRRK2 in idiopathic
PD (iPD), the vast majority of cases in which the cause is unknown. Using an assay they developed, they demonstrated increased LRRK2 activity in postmortem brain tissue from patients with iPD (Di Maio et al., 2018); and reproduced this in an animal model of PD in which exposure to a common pesticide called rotenone replicates the key pathological and behavioral characteristics of PD (Betarbet et al., 2000). Greenamyre and colleagues hypothesize that rotenone produces reactive oxygen species, including superoxide, causing oxidative stress, which activates LRRK2 (Di Maio et al., 2018). In human PD and in the animal model, LRRK2 activity is also associated with endolysosomal deficits, which result in accumulation of disease-associated proteins such as α-synuclein (Rocha et al., 2020).
Greenamyre’s lab went on to explore whether LRRK2 activation is a common feature of PD-associated environmental toxicants, such as trichloroethylene (TCE) (see Figure 3-3). TCE, rotenone, another pesticide called paraquat, and many other environmental toxicants are known to cause oxidative stress and mitochondrial dysfunction (Tanner et al., 2011). Investigators in Greenamyre’s lab have shown that TCE exposure induces LRRK2 activity in dopaminergic neurons and that an inhibitor of LRRK2 attenuates neurodegeneration in the rotenone model of PD (De Miranda and Greenamyre, 2020). This suggests that LRRK2 inhibitors may be useful not only for individuals with LRRK2 mutations, but those with iPD as well.
EPIGENETICS
Epigenetics emerged in the early 2000s to help explain how environmental conditions can alter the expression of genes and how, for example, early life events can cause disorders with onset later in life, according to Marisa Bartolomei, Perelman Professor of Cell and Developmental Biology and co-director of the Penn Epigenetics Institute at the University of Pennsylvania. Bartolomei and colleagues have been investigating how bisphenol A (BPA)—an estrogen mimetic used in manufacturing many types of food and beverage containers—may reprogram the adult mouse brain through epigenetic modification of genes. During early development, genes are particularly vulnerable to environmental perturbations that lead to changes in the levels of DNA methylation, histone modifications, and other chemical alterations to genes, resulting in dysregulation of gene function, said Bartolomei. One group of genes that is protected from this genome-wide methylation is imprinted genes. These are genes expressed in a monoallelic parent-of-origin-specific manner (Bartolomei, 2009). They are essential for fetal growth and also are involved postnatally in regulating behavior, metabolism, and other processes, said Bartolomei (Kalish et al., 2014). Dys-
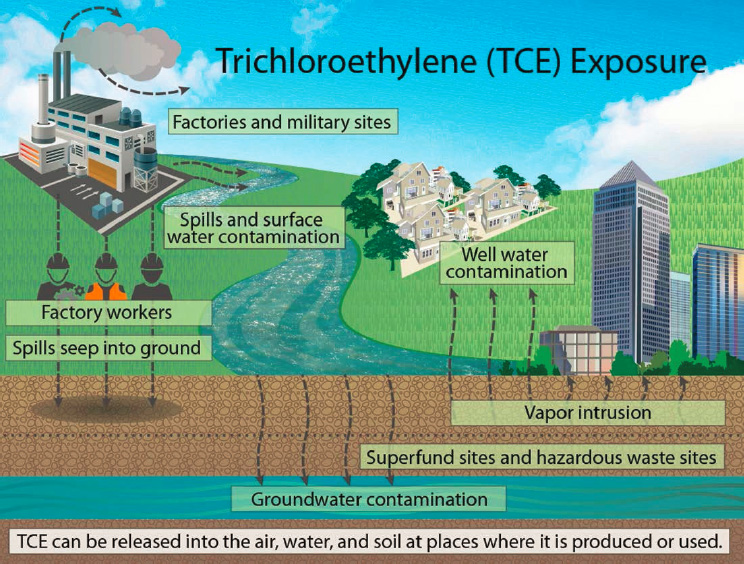
SOURCES: Presented by J. Timothy Greenamyre, June 25, 2020; HHS, 2016.
regulation of imprinted genes is associated with neurobehavioral disorders such as Prader Willi and Angelman syndromes, she said.
Human exposure to BPA is widespread and has been linked to behavioral abnormalities as well as cancer, metabolic disorders, and infertility (Susiarjo et al., 2013). In a 2005 study of urine samples collected in the Third National Health and Nutrition Examination Survey (NHANES III), BPA was detected in 95 percent of samples (Calafat et al., 2005). In 2007, Dolinoy and colleagues showed that exposure of pregnant mice to BPA resulted in a change in the color of their coats. These changes resulted from epigenetic changes—hypomethylation or loss of methylation—in a specific genetic locus, a retrotransposon called the intracisternal A particle (Dolinoy et al., 2007).
Bartolomei and colleagues were interested in learning how BPA affected imprinted genes. They exposed female mice to BPA at two physiologically relevant doses of BPA (lower and higher doses) both before mating (preoocyte development) and during pregnancy (earliest stage of embryonic development); then analyzed embryonic and placental tissue from pregnant mice euthanized at two time points. Genetic analyses of the tissue revealed that maternal BPA exposure during late stages of oocyte development and early embryonic development disrupted genomic imprinting in embryos and placentas of several genes associated with human imprinting disorders. Genome-wide methylation levels in the placenta, but not the embryo, was also reduced, said Bartolomei (Susiarjo et al., 2013).
They also assessed multigenerational behavioral and metabolic consequences of BPA exposure by allowing mice to be born, continuing to expose mothers to BPA through weaning, then weaning mice with no further BPA exposure and examining behavior and metabolism phenotypes in F1 (first generation) and F2 (second generation) mice. The F1 offspring of BPA-exposed mice exhibited male-specific, depressive-like behavior, as well as metabolic and skeletal phenotypes, said Bartolomei. She added that the metabolism phenotypes were multigenerational, but the behavioral phenotype was not transmitted to subsequent generations (Xin et al., 2018).
Cellular Mosaicism
Richard Woychik said there has been interest at the National Institutes of Health in looking at cellular mosaicism, where a person may have genetically different sets of cells in their bodies. This can be driven by activation of transposable elements through epigenetic activation or inactivation, he said, suggesting there might be an environmental trigger for this process. Zylka noted that mosaicism has been demonstrated in some people with idiopathic PD or AD and may be confined to the brain or another small region of the body. Mosaicism has also been seen in autism and epilepsy, he said.
CONCURRENT RISK FACTORS: STRESS AND POVERTY
Devon Payne-Sturges, associate professor at the Maryland Institute for Applied Environmental Health, added that the issue of cumulative exposures is particularly relevant in terms of environmental justice. In addition to multiple chemical exposures, the social context in which these exposures occur is important, she said, adding that environmental and social stressors have been shown to have similar biological targets and possible synergistic
effects. Indeed, said David Bellinger, disadvantaged children suffer disproportionately from neurotoxicants like lead. Not only are they more highly exposed to neurotoxicants, but the same magnitude of exposure has a greater adverse impact on them compared to more advantaged children, he said. For more on how social stressors may impact neurological disorders, see the section on autism and ADHD in Chapter 4. For more about research that is needed to better understand these interactions, see Chapter 5.
This page intentionally left blank.












