19
Assessment of Cellular Dysfunction During Physiologic Stress
Guy Miller1
THE TRANSITION FROM PHYSIOLOGIC TO CELLULAR ASSESSMENT
Organ Physiology Is Derived from Cell Biochemistry
The appropriate alimentation of an individual subjected to exogenous or endogenous stress ultimately calls for the conversion of basic food elements to cellular energy. The myriad of events that combine to regulate this process effectively is controlled at many levels of the organism. Various schemes have been employed to characterize such control mechanisms (Hochachka et al., 1992, 217–348). During homeostasis, anabolism and catabolism are coupled tightly throughout the various organ systems, and hence, an almost seamless cascade of events occurs—digestion, absorption, transport, cell uptake, and utilization—directed at supplying cells with vital macro-and micronutrients to
|
1 |
Guy Miller, Stanford University and Galileo Laboratories, Inc., Sunnyvale, CA 94086. gmiller@galileolabs.com |
maintain and promote cell function. How this complex balance behaves when subjected to stress is a matter of great importance.
Physiologic Reserve Is a Measure of Total Cell Mass Function
The concept of physiologic reserve often is used to connote the limits of function of a particular physiologic system when a stress is brought to bear upon that system. In such a manner, a tolerance to external stressors that are, in effect, compensated for by the physiologic reserve of the system, is observed. When a stress exceeds the capability of a particular organ system to compensate adequately, physiologic dysfunction ensues. Several factors influence the magnitude of the observed physiological derangement and include: the type of stress, the prior state of the system, and the duration of the stress interval.
Physiologic Reserve often Deteriorates Unnoticed
Alterations in physiologic function are oftentimes not apparent until a critical level of function is compromised. The generalized, nonlinear deterioration of physiologic performance as related to cell function results in a disparity between the (physiologic) output signal and the (cellular) signal generator (Figure 19-1). Hence, strategies to measure physiologic function (reserve) during stress should be directed at assessing both global organ function and cellular reserve. By this means, information is obtained on the current status of the system and expectations concerning performance potential.
Measure Cell Function to Treat Cell Dysfunction
With the ultimate goal of optimization of performance during acute stress, it would be preferable to monitor cellular parameters that were causally, not associatively, related to physiologic outcome. Two advantages are derived from monitoring underlying cell function. As detailed above, early indicators of total cell function will yield information concerning organ function and will serve as an early warning system for impending failure. Additionally, the monitoring of various parameters associated with cell function affords the opportunity to design and test nutritional strategies to treat selective aspects of cell function to optimize performance.
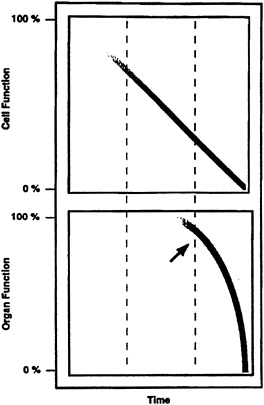
FIGURE 19-1 Organ physiology as a function of cell biochemistry. An idealized model of organ and cell function is expressed in which injury increases over a period of time. Various states are proposed that broadly reflect the severity of underlying physiologic performance as a function of progressive cellular dysfunction. Cell function can decrease by approximately one-third, with minimal decline in organ function (physiologic reserve). Further decreases in cell function are tolerated. It is often not until approximately two-thirds of total cell function are compromised that a change in traditional physiologic variables is appreciated (arrow). When a critical point is approached, attempts to improve outcome by optimizing physiologic function often fail.
CELLULAR PARAMETERS IN ACUTE CELL DYSFUNCTION
Cells Possess Multiple Responses to Stress
The cellular response to acute stress is dependent upon the cell type, the nature of the stress, and the environment of the cell. For example, intestinal epithelial cells respond differently to the effects of circulating epinephrine in comparison with myocardial myocytes. Additionally, the myocyte response depends upon the duration of epinephrine exposure (receptor downregulation). Lastly, the response of the enterocyte is dependent upon local autocrine mechanisms and is influenced by neighboring endothelial, leukocyte, and neuronal
action(s). These examples illustrate not only the range of responses to be understood, but the overall complexity of the system.
Hypoxia Is a Stress that Disrupts Cell Energy Metabolism and Affects Dysfunction
Hypoxia, a disruption in normal oxygen delivery to vital tissues, occupies a central role in causing organ dysfunction and death. Tissue hypoxia involving the splanchnic viscera is of particular interest to the nutritionist concerned with performance during stress in the presence or absence of extreme environmental conditions. Hypoxia-mediated splanchnic dysfunction can lead to nutrient malabsorption, gut dysfunction, bacterial translocation, and heat stroke. For a concise review of this topic, the reader is referred to a previous text issued by the Committee on Military Nutrition Research, Nutritional Needs in Hot Environments (IOM, 1993). To monitor and treat hypoxia-mediated gastrointestinal dysfunction most effectively, methodologies are being fashioned that address the underlying basis of the injury: disruptions in cell energy metabolism.
Measuring Cell Energy Metabolism
Cell function is dependent upon the delicate balance between anabolism and catabolism. This process is highly regulated at the cellular level. For many cell types, the bulk of energy is derived from processes involving oxygen. Hence, in the absence of oxygen, cell function is impaired. The time course for these events is dependent upon the particular cell type. For example, highly oxidative cells such as those comprising the brain, heart, and liver have a relatively poor tolerance to hypoxia, surviving on the order of minutes. In contrast, the nonoxidative red blood cells are relatively unaffected by extended periods without oxygen and can survive stored for months.
There are various ways to measure the function of the cellular components that comprise the energetic apparatus of the cell. The challenge is in integrating the events controlling substrate utilization, cell and tissue function, and whole animal function during both normal and pathological states. Because hypoxia has a rapid and profound effect on cell energy metabolism, measurements involving cell energetic function are useful tools with which to understand and monitor the effects of hypoxia. It should be kept in mind that a single or simple measurement of energy metabolism does not exist that yields exclusive and complete information as to the health or integrity of the system. With these limitations, what information can be obtained by direct or indirect measurements involving cell energy metabolism?
PRACTICAL ISSUES IN ASSESSING CELL FUNCTION
Studies on cell energy metabolism can be performed with in vivo or ex vivo preparations. There is a general trade-off between the ability to acquire mechanistic data and the complexity of the model system. While simplified cell-model systems often allow investigators to obtain more data, the extrapolation of these results to intact organ and animal systems is fraught with many limitations. Conversely, the limited data set derived from intact organ systems oftentimes does not allow investigators to obtain certain relevant mechanistic data. The following two sections detail the advantages and limitations associated with investigating hypoxic tissue dysfunction in ex vivo model systems. The data and interpretation are intended both to be informative for investigators with specific interests in the field of alimentation during hypoxic cell stress, as well as to discuss the general advantages and disadvantages of transitioning from organ-based to cell-based investigations.
Identification of the Correct Cell and Model
With the goal of extrapolating events from the cell to the whole organ level and the animal level, a careful choice of cell type and model is required. In general, investigators concerned with whole organ-animal function often employ cell culture techniques to achieve one of two end points: (1) the study of a distinct process or cellular component, or (2) the study of the cell as a miniaturized model of the often complex in vivo counterpart. It is towards the latter that this discussion is directed.
Cell Models of Tissue Hypoxia
Using a cell system to study the more complex in vivo system has many advantages; however, it comes with many potential pitfalls. All too often cellular mechanisms have been elucidated that bear little resemblance to the in vivo context. To avoid these problems, a thorough understanding of the tissue under examination and the choice of a cellular model system is needed.
Beyond the general problems associated with cellular model systems of complex diseases, the study of hypoxia imposes its own set of limitations on the choice of cell type. The cellular response(s) to hypoxia can be divided into fast and slow responses. Included in fast responses are rapid changes in metabolic flux that occur on the order of seconds to minutes. Slow responses, more prevalent in adaptation to chronic hypoxia, appear on the order of hours and would include, for example, the synthesis of inducible nonconstituent proteins such as heat shock protein 70 (De Maio, 1995).
Because hypoxia affects the method and amount of energy a cell produces, hypoxic conditions possess the ability to alter all energy-dependent functions. In
choosing a cell model to examine the fast responses associated with hypoxia, two general requirements exist: (1) the cell should derive its energy in a manner similar to the tissue of interest, and (2) the particular energy-dependent cell function of interest should possess a time-dependent decrement in function that closely parallels the modeled tissue. The following example illustrates these two points.
The Caco-2 cell is a human colonic tumor derived cell line frequently employed to study various aspects of enterocyte function. Caco-2 cells derive the bulk of their adenosine 5'-triphosphate (ATP) requirements from glycolysis, that is, substrate level phosphorylation. In contrast, intestinal function is highly dependent upon ATP-derived from oxidative phosphorylation. While removal of oxygen results in substantial intestinal (tissue) death on the order of hours, the time response for the Caco-2 cells is on the order of days (Personal communication, M. Fink, Harvard Beth Israel Hospital, Boston, Mass., 1995). The poor energy dependency and time response makes the Caco-2 cells a suboptimal model in which to study the rapid response to hypoxia.
Measurements of the Cellular Response to Hypoxia
Investigators concerned with bioenergetics have developed many techniques with which to assess cellular energy metabolism. A partial list includes the assessment of cellular energy charge by nuclear magnetic resonance spectroscopy, high-pressure liquid chromatography, or enzymatic techniques; the measurement of substrate flux; the assessment of mitochondrial function by measurement of ATP synthesis, membrane gradients, or potentials; and the examination of specific cell functions closely linked to energy supply, such as second messenger-mediated events. The choice of a particular technique is guided in part by the process under study. To examine the effects of hypoxia on splanchnic function, it was of interest to derive a measurement that was convenient and yielded generalized data on global cell energy function.
Proton Flux to Measure Rapid Responses to Change in Cell Energy Metabolism
Cell proton production and excretion are linked closely to energy metabolism. As a first approximation, protons are derived from two metabolic pathways: glycolysis and the tricarboxylic acid (TCA) cycle. Protons produced in glycolysis are derived from the oxidation of glyceraldehyde 3-phosphate to 3-phosphoglycerate. These protons often are associated with the acid proton component of lactic acid. Additional protons are produced by the oxidation of substrates to produce carbon dioxide within the TCA cycle (CO2 + H2O → H2CO3 → HCO3- + H+). These protons leave the cell by one of several mechanisms (Figure 19-2).
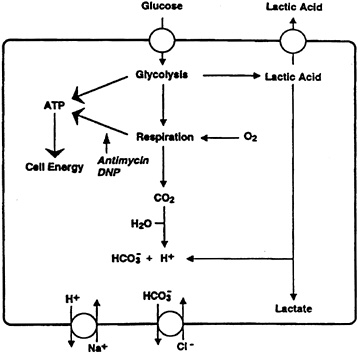
FIGURE 19-2 Cellular proton production. Protons are generated through various metabolic pathways. In this simplified scheme, protons that arise from glycolysis and respiration are shown. Protons exit the cell by one of several mechanisms, including the sodiumproton antiporter, monocarboxylate transporter, diffusion of a neutral weak acid, and diffusion of carbon dioxide-carbonic acid. Adenosine 5'-triphosphate (ATP) generated by substrate level phosphorylation and oxidative phosphorylation provides the chemical energy to power the cell. Antimycin and 2,4-dinitrophenol (DNP) inhibit the respiratory component of ATP synthesis.
Measurements of extracellular acidification can be employed to assess substrate flux through the TCA cycle or the glycolytic pathway indirectly (McConnell et al., 1992). In effect, the extracellular acidification rate can be thought of as a reflection of cellular metabolic rate expressed as proton production per minute. Changes in metabolic rate thus serve as a convenient proxy for assessing cellular energy metabolism on a more global scale.
APPLICATION: UNDERSTANDING SPLANCHNIC DYSFUNCTION DURING ISCHEMIC-HYPOXIC STRESS
Stress states arising from various conditions can lead to a reduction in blood flow to the splanchnic viscera (Gutierrez and Brown, 1996). When blood flow is reduced for critical periods of time, tissue injury can ensue, leading to cell death
and/or dysfunction. With reference to the intestinal barrier function, such a sequence of events has been implicated in setting off a cascade of events associated with various systemic inflammatory responses and/or sepsis (Brinkmann et al., 1996).
Healthy and infirm individuals can be at risk for hypoperfusion of the gut. (For a detailed discussion of gut dysfunction in the critically ill, the reader is referred to the attached reference list.) Within the population of healthy individuals, splanchnic hypoperfusion has been implicated in heat stroke and bowel dysfunction secondary to dehydration and extreme stress conditions (IOM, 1993). In instances where blood flow is not promptly restored, splanchnic hypoperfusion can result in signs and symptoms ranging from generalized cramping and diarrhea to epithelial sloughing, intestinal hemorrhage, bowel perforation, and death.
To understand and design cell-based nutritional strategies to ameliorate the effects of splanchnic hypoperfusion, it is of interest to study this process with a cell-based model.
Variations in Cell Culture Lines
All enteric cells are not alike. Similarly, cells derived from the same organ and even the same (sub)population can display marked dissimilarities within cell culture conditions. For example, the susceptibility of two liver parenchymal cell types to the effects of hypoxia can differ markedly. WIF-B cells, a hepatoma-derived hybrid line, suffer near complete injury as evidenced by extensive membrane leakage of intracellular lactate dehydrogenase (LDH) after 3- to 4-h exposure to conditions modeling the effect(s) of hypoxia (Shanks et al., 1994) (Table 19-1). Hep-G2 cells, also a liver cell line obtained from a human hepatocellular carcinoma, exhibited a different pattern of injury. LDH leakage was less
TABLE 19-1 Antimycin-Mediated Cell Death
extensive in the Hep-G2 cell line, suggesting either 100 percent injury for approximately 50 percent of one or more subpopulations, or a sublethal, approximately 50 percent injury dispersed over the entire cell population(s). In contrast, the intestinal derived Caco-2 cell line exhibits minimal enzyme leakage under similar conditions (Personal communication, M. Fink, Harvard Beth Israel Hospital, Boston, Mass., 1995).
Irrespective of the mechanism of differential cell death, the data demonstrate one limitation associated with cellular model systems: variability. The properties exhibited by the WIF-B cell line closely approximate in vivo events (Unpublished data, C. Kasserra, Galileo Laboratories, Inc., Sunnyvale, Calif., 1997). The Hep-G2 cells display moderate susceptibility to the effects of hypoxia, while the Caco-2 cells are almost completely resistant to hypoxic conditions. This variation in response severely affects outcome measurements. For this reason, careful consideration must be given to choosing a cell system in which to investigate the effects of hypoxia. The following general statement can be made: To maximize the utility of a given cell model, the cell type should display a pattern of and susceptibility to injury similar to that observed in the organ or tissue of interest.
Metabolic Rate Changes during Hypoxia
The dependency of the choice of cell line on variations in hypoxia-mediated LDH leakage was described in the previous section. Measurements such as enzyme (LDH) leakage assays yield information that is mostly associative, not causally related, to cell injury or death. To derive a more detailed understanding of the causal events associated with hypoxia-mediated cell injury, it was of interest to examine the effects of hypoxia on cellular energy metabolism. The specific aims included: (1) the feasibility of using proton excretion measurements to study cell-based model systems of hypoxia and (2) the use of such model systems to gain an understanding of cell function during hypoxia.
Glycolytic Cells May Upregulate Glycolysis during Hypoxia
Caco-2 cells derive the bulk of their energy supply from glycolysis and thus are relatively insensitive to the effects of agents such as dinitrophenol or antimycin that disrupt (mitochondrial) oxidative phosphorylation (Personal communication, M. Fink, Harvard Beth Israel Hospital, Boston, Mass., 1995) (see Figure 19-2). To understand the effects of hypoxic conditions more thoroughly, corresponding changes in proton flux were recorded for Caco-2 cells incubated in the presence of dinitrophenol (DNP) (Figure 19-3). Proton excretion rate increased on exposure to DNP. Removal of DNP resulted in reequilibration of the metabolic rate to baseline values, demonstrating that this process was reversible.
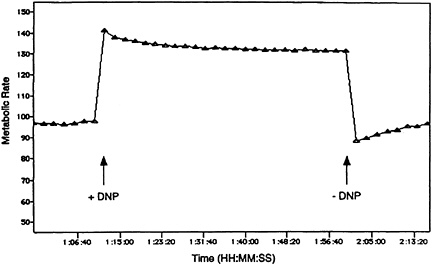
FIGURE 19-3 Effects of dinitrophenol (DNP) on metabolic rate of Caco-2 cells. Studies employing standard cell culture techniques were performed on Caco-2 cells. Cells were grown in Dulbecco's modified Eagle's medium (DMEM) plus 10 percent fetal bovine serum medium on microporous polycarbonate transwells and studied 24 hours post-seeding. Glycolytic and tricarboxylic acid (TCA) flux were assessed by quantifying proton production using a pH-sensitive silicon sensor interfaced to a 3-µL flow chamber (Owicki and Parce, 1992). Cell perfusate contained balanced salt solution, 10 mM glucose, and 100 µM DNP where indicated. Metabolic rate (-µvolt × sec-1) is expressed as a percentage of basal activity obtained in balanced salt solution plus 10 mM glucose (100%).
Caco-2 cells synthesize ATP by both oxidative phosphorylation and substrate level phosphorylation. One possible explanation for the increased proton flux during DNP treatment is an increase in glycolytic flux to compensate for a decrement in ATP derived from oxidative phosphorylation. Indirect evidence for such a mechanism is derived from the known effects of variations in cellular energy charge (EC = ATP + 0.5 ADP/ATP + ADP + AMP)2 on allosteric regulation of glycolytic enzymes, for example, phosphofructokinase-1 (Atkinson, 1977, 85–107).
Oxidative Cells Decrease Metabolic Rate during Hypoxia
In contrast to Caco-2 cells, proton flux studies performed on primary rat liver hepatocytes treated with DNP had a different outcome (cf., Figures 19-3 and 19-4). The initial response recorded for hepatocytes paralleled that observed for the Caco-2 cells, namely an increase in metabolic rate. The increase in proton flux was transient and declined over the next several minutes, equal to an
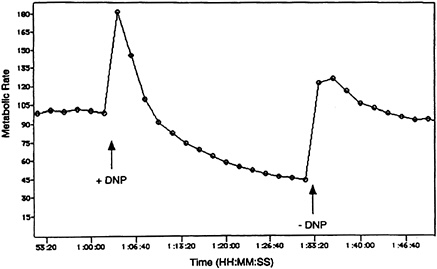
FIGURE 19-4 Effects of dinitrophenol (DNP) on metabolic rate of primary rat liver hepatocyte cells. Assessment of metabolic rate on hepatocyte cells was performed as described in the legend to Figure 19-3.
approximately 45 percent reduction in metabolic rate. Removal of DNP from the cell perfusate resulted in an approximately 20 percent overshoot of the baseline metabolic rate, followed by a trend towards reequilibration to baseline value after approximately 10 minutes.
Highly oxidative cells such as hepatocytes derive a significant portion of ATP from oxidative phosphorylation. While the effects of hypoxia-mediated ATP depletion have been studied extensively in numerous cell and organ models, an adequate explanation linking ATP depletion to a causal sequence of events leading to cell death has yet to be advanced. Proton flux data obtained from isolated hepatocytes suggest that dinitrophenol-mediated uncoupling of the mitochondrial proton gradient from ATP synthesis results in a marked reduction in net proton flux. Whether this is brought about by substrate (ATP) limitation or as a result of the triggering of a sequence of events to modulate energy consumption, is unknown. Further studies are underway to define how metabolic rate varies as a function of a graded depletion of ATP.
APPLICATIONS: FUTURE QUESTIONS TO BE ADDRESSED
The cell-based experiments described above only barely begin to address many pertinent questions of interest to nutritionists examining function during stress. The following section contains a partial list of questions whose solution(s) may be facilitated by methodologies employing cell-based assessment of cellular energy metabolism.
Substrate Flux during Hypoxic Stress
Is there a preferential use of substrates during hypoxic stress? If certain substrates are used to the exclusion of others, a nutritional formulation could be developed to potentiate beneficial metabolic pathways.
Cell Hibernation
How does the cell acutely adapt to hypoxic conditions, that is, conditions in which energy demand exceeds supply? Preliminary data suggest that the cell responds with an initial increase in metabolic rate followed by a decrease. What are the events that mediate these processes?
Metabolic Cofactors
What are the effects of metabolic cofactors such as vitamin E, glutathione, and trace minerals on conferring resistance to hypoxic stress? The antioxidants as a group have been examined extensively for nutritional efficacy during various stress circumstances. While their general role continues to be somewhat controversial, less is known about antioxidants as nutritional adjuncts during acute hypoxic stress.
Enteric Function
Will performance and outcome be improved by strategies that maintain splanchnic function during acute stress? Maintaining gut and hepatic function is critical to maintaining systemic homeostasis. The deterioration of either of these organ systems has been implicated as the causal mechanism of heat stroke and other conditions. Will strategies that are designed to use cell-based systems extrapolate to intact organ systems, and if not, how can investigators improve such model systems?
REFERENCES
Atkinson, D. 1977 Cellular Energy Metabolism and Its Regulation. New York: Academic Press.
Brinkmann, A., C.F. Wolf, D. Berger, E. Kneitinger, B. Neumeister, M. Büchler, P. Radermacher, W. Seeling, and M. Georgieff 1996 Perioperative endotoxemia and bacterial translocation during major abdominal surgery: Evidence for the protective effect of endogenous prostacyclin. Crit. Care Med. 24:1293–1301.
De Maio, A. 1995 The heat shock response. New Horizons 3:198-207.
Gutierrez, G., and S. Brown 1996 Gastrointestinal tonometry: A monitor of regional dysoxia. New Horizons 4:413–419.
Hochachka, P.W., P.L. Lutz, T. Sick, M. Rosenthal, and G. van den Thillart, eds. 1992 Surviving Hypoxia: Mechanisms of Control and Adaptation. Tokyo: CRC Press.
IOM (Institute of Medicine) 1993 Nutritional Needs in Hot Environments, Applications for Military Personnel in Field Operations, B.M. Marriott, ed. A report of the Committee on Military Nutrition Research, Food and Nutrition Board. Washington, D.C.: National Academy Press.
McConnell, H.M., J.C. Owicki, J.W. Parce, D.L. Miller, G.T. Baxter, H.G. Wade, and S. Pitchford 1992 The cytosensor microphysiometer: Biological applications of silicon technology. Science 257:1906–1912.
Owicki, J.C., and J.W. Parce 1992 Biosensors based on the energy metabolism of living cells: The physical chemistry and cell biology of extracellular acidification. Biosens. Bioelectron. 7:255–272.
Shanks, M.R., D. Cassio, O. Lecoq, and A.L. Hubbard 1994 An improved polarized rat hepatoma hybrid cell line. J. Cell. Science 107:813–825.
DISCUSSION
DONALD McCORMICK: Fascinating. Can you use similar electrodes at the organelle level?
GUY MILLER: Yes. We have reconfigured the microphysiometer to assess cellular redox state. Specifically, we incubate cells in the presence of a redox active compound such as menadione. Menadione is taken up by the cell and couples into electron transport and undergoes a reduction reaction. Reduced menadione diffuses out of the cell and reduces an extracellular Fe(III)->Fe(II) redox couple that in turn donates an electron to the sensor producing a current.
There are several limitations to this approach that include redox and chemical perturbations affected by menadione. While I believe this to be a useful technology, improvements will have to be made to allow less cellular alterations during measurement intervals, as well as improvements in the time response of the system.
ARTHUR ANDERSON: Are the liver cells that you are growing in this system grown on a biomatrix? Or are they by themselves because liver cells will differentiate on a biomatrix, but they will not differentiate as single-cell isolates on a flat surface?
GUY MILLER: The WIF-B cells grow directly on standard 24- or 96-well cell culture plates. Your question infers the difficulty of working with primary-de-
rived hepatocytes with specific requirements for a collagen matrix or other suitable support. One of several unique properties of the WIF-B cells is ease of propagation and convenience relative to primary cell harvesting. For your reference, these cells form a pseudo-organized liver architecture containing bile canaliculi.
DENNIS BIER: I found this very intriguing because I think it is a new, expanded way to look at sodium metabolism. I am not sure you can do some of these things in other ways. For example, people have used specifically tritiated labeled glucose molecules for years to look at the appearance rate of tritiated water to measure various steps in glycolysis outside the cell.
One can look at the TCA [tricarboxylic acid] cycle activity by looking at the rearrangement of carbons and oxaloacetate, either radio-or isotopically labeled, and that can be done outside the cell as well.
Now, people have not looked at 10-s intervals because they have not felt that that was particularly important. Maybe it is, but there are other ways to approach this. I am not sure that it should not be combined in some way with what you are doing.
GUY MILLER: I think that the idea of combining several metabolic assessment modalities to simultaneously assess both cellular and tissue metabolic variables is a good one. As I understand from Harris Lieberman, one issue to be addressed by the CMNR during this session is to examine undertaking such activities. It would be my opinion that a concerted multidisciplinary effort needs to be undertaken in the area of acute metabolic derangements during circumstances of oxygen limitation, i.e., extreme exertion or illness. While we understand a great deal about particular metabolic transformations employing single enzyme experiments, we have just begun to decipher the processes governing the dynamics of metabolic control. New technologies and approaches will also be required that take advantage of our growing understanding of functional genomics. How these methodologies are fashioned together to enhance our understanding of relevant nutritional issues will be a challenging and vital task in forging the next set of advances.














