4
Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline1
In the presentations on the B Vitamins and Choline Report (IOM, 1998a), Steven H. Zeisel of the University of North Carolina–Chapel Hill (former member of the Panel on B Vitamins and Choline) addressed research on thiamin, riboflavin, niacin, pantothenic acid, and choline.2 Dr. Patrick Stover of Cornell University covered folate, vitamins B12 and B6, and biotin. Both presenters addressed lack of progress and new developments since 1997–1998, and they suggested future topics for research.
DISCUSSION OF RESEARCH RECOMMENDATIONS: THIAMIN, RIBOFLAVIN, NIACIN, PANTOTHENIC ACID, AND CHOLINE
Presenter: Steven H. Zeisel
This presentation highlighted major new developments, pointed out that genetic polymorphism and epigenetics merit special attention, and addressed major research gaps and the progress made.
Major New Developments
Genetic Polymorphisms
New science in genetic polymorphisms suggests that the requirements for some of the nutrients vary, in part dependent on whether individuals possess single nucleotide polymorphisms (SNPs). SNPs are deoxyribonucleic acid (DNA) sequence variations that occur when a single nucleotide (A, T, C, or G) in the genome sequence is altered. Such genetic polymorphisms are not rare.
In setting Recommended Dietary Allowances (RDAs), the Panel on B Vitamins and Choline assumed that it had to cover only 95 percent of the population and that it did not need to address genetic mutations. For some of these nutrients, however, more than half the population has at least one genetic polymorphism. Thus, genetic mutations merit attention.
Epigenetics
The area of epigenetics has developed tremendously since the B Vitamins and Choline Report was written. Clearly, some of the nutrients in this group, specifically the methyl donors and biotin, are part of the process of epigenetic modification. Considerable data point to the need to consider whether gene expression is being changed by changes made to the promoter regions of genes or to the histones that surround genes.
Major Research Gaps and Progress Made
Major research gaps identified in the report included (1) human information on the requirements of children, pregnant and lactating women, and the elderly; (2) other indicators that could be used to assess function; and (3) adverse effects of high doses of the nutrient. A summary follows of progress made with regard to thiamin, riboflavin, niacin, pantothenic acid, and choline.
Thiamin
Little has been done to meet the research needs pertaining to thiamin.
Riboflavin
Since 1998, at least one large population study (O’Brien et al., 2001) on intake reports that a sizable segment of the population (12.5 percent of men and 20.6 percent of women) had intakes below the Estimated Average Requirement (EAR) in Ireland.
Hustad and coworkers (2000) show that riboflavin is inversely associated with homocysteine concentration in blood. The authors propose that individuals who have a SNP in the flavin-requiring enzyme methylene tetrahydrofolate reductase (MTHFR) are sensitive to riboflavin concentrations. With higher intakes of riboflavin, the homocysteine concentration in their blood decreases.
Flavin-adenine dinucleotide (FAD)-dependent glutathione reductases have been used as the markers for riboflavin sufficiency in setting the EAR for riboflavin. Some work suggests a new functional measure for riboflavin status. In particular, a clinical trial (Jacques et al., 2005) reports a reduction of age-related lens opacification in humans treated with riboflavin supplements. Cataract production was an end point that was not fully considered when setting the EAR for riboflavin, but it probably is of interest in the consideration of revisions to riboflavin requirements.
Niacin
Research gaps Research gaps particular to niacin include (1) increased niacin requirements secondary to oxidant exposure, (2) the identification of a better method for determining niacin status other than the urinary excretion currently used, and (3) improvement of nutrient databases to differentiate the forms of niacin—specifically the naturally occurring niacin content of foods and niacin added as a fortificant.
Progress made Some progress has been made in addressing niacin research gaps since 1998:
-
A toxicology panel was convened; in 2005, it reported on the toxicity and potential toxicity of higher dose niacin (Cosmetic Ingredient Review Expert Panel, 2005). That report discusses possible end points and markers, and it would be useful in reconsidering Tolerable Upper Intake Levels (ULs) for niacin.
-
High and low affinity nicotinic acid receptors have been identified and cloned (Wise et al., 2003).
-
A G-protein-coupled receptor, HM74A, has been shown to mediate skin flushing (a side effect of high niacin intake) (Benyo et al., 2005). The G-protein-coupled receptor might be used to define niacin status and the niacin requirement in terms of another functional end point—namely, the activation or inactivation of that receptor.
-
Results of numerous studies have been published on the pharmacological use of niacin for the reduction of hyperlipidemia. Two are especially notable: (1) the Atherosclerotic Disease Multiple Intervention Trial (ADMIT), a randomized controlled trial in patients with diabetes and peripheral artery disease; and (2) the Arterial Biology for the Investigation for the Treatment Effects of Reducing Cholesterol (ARBITER) trial, a double-blind, placebo-controlled style of extended-release niacin. With regard to the DRIs, both of those trials probably are most useful for defining human exposure and potential toxicity at high doses. However, they might also be useful in defining peripheral vascular disease function as an end point for niacin optimization.
Pantothenic Acid
Although lack of pantothenic acid has been seen to cause deficiency in humans very rarely, this vitamin is a widely used cofactor. Very little information was available about pantothenic acid in 1998, and more recent information has been provided by only two studies—both of which address assays for pantothenic acid in beverages, vitamins, and foods. Improved assay methods might help to improve the estimates of pantothenic acid content in food composition databases.
Choline
In 1998, insufficient data were available to set an EAR for choline, so Adequate Intakes (AIs) were set instead. Major research gaps particular to choline included its roles in chronic disease, bioavailability, metabolic effects, and interrelationships with vitamins B12 and B6 and with methionine and folate metabolism. Choline has three functions: methyl
donation, lipid formation, and formation of acetyl choline. Could methyl donors spare the entire requirement for choline or not?
All the studies that had been done before 1998 used male subjects. Because data on the choline content of foods were very sparse, the panel had little information on the choline content of the food supply or the extent of human exposure to choline. Limited data were available concerning high pharmacological doses of choline and their side effects; these data were used to establish a UL.
Depletion–repletion study findings New human data obtained from a 92-day depletion–repletion study (da Costa et al., 2006a,b) demonstrate that estrogen induces the endogenous synthesis of choline. Eighty percent of men and postmenopausal women developed liver and muscle dysfunction when deprived of choline, but only 44 percent of premenopausal women did.
At baseline, 60 individuals were given 550 mg of choline per 70 kg of body weight daily for 10 days (see Figure 4-1). Individuals then were depleted (given less than 50 mg/per 70 kg of body weight daily) until they developed signs of deficiency or for a maximum of 42 days. Fifty-five percent of women with estrogen did not need any choline for 42 days. Although their blood concentrations decreased, they did not develop other signs of choline depletion. Individuals who developed organ dysfunction were given graded doses of choline until they reverted to normal. Liver dysfunction (i.e., fatty liver by mass resonance imaging) was the common sign of depletion.
A subgroup of the individuals developed muscle dysfunction, as identified by creatine phosphokinase leakage into the plasma, which resolved with the refeeding of choline. This outcome affected 10 percent of the population, including men and one postmenopausal woman. Other research indicates that some men require substantially more than the current AI for choline (da Costa et al., 2006a). Potential functional markers for choline include muscle damage, lymphocyte apoptosis, and elevated homocysteine after a methionine load.
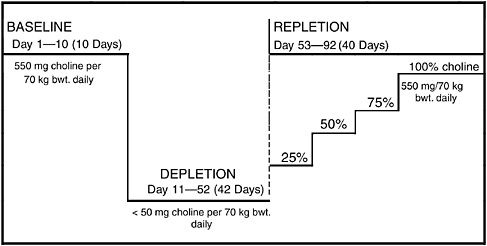
FIGURE 4-1 Study design for choline depletion–repletion trial. The trial included 31 healthy men and 35 health women. The daily baseline diet also contains 400 dietary folate equivalents (DFE) and the depletion and repletion diets have 100 DFE. On Day 11, subjects are randomized to 100 DFE (placebo) or 400 µg folic acid for the remainder of the study.
NOTE: bwt. = body weight.
SOURCE: da Costa et al. (2006b). Reproduced with permission from The American Journal of Clinical Nutrition.
The finding that premenopausal women are less likely to develop organ dysfunction when fed a low choline diet led to the examination of the gene that encodes for the enzyme that allows the liver to make the choline moiety. That gene has a number of estrogen-response elements in its promoter, and it is induced by estrogen. Further investigation revealed that individuals who develop organ dysfunction when deprived of choline have a greatly increased risk if they have one or two of several genetic polymorphisms in genes of choline or folate metabolism. The study found a polymorphism in phosphatidylethanolamine-N-methyltransferase (PEMT), the gene responsible for endogenous biosynthesis of the choline moiety.
The women who have this SNP in the PEMT promoter have increased susceptibility to choline deficiency (which is characterized by fatty liver, muscle and liver damage) with an observed risk 25 times that of controls. This huge increase in observed risk made it possible to identify the risk with only 60 subjects in this clinical study. A SNP in the gene for methyl-tetrahydrofolate dehydrogenase 1 (MTHFD1) also in-
creased observed risk for developing choline deficiency (Kohlmeier et al., 2005).
Of particular note is that the SNPs related to choline deficiency are not rare. For example, 18 percent of subjects were homozygous for the PEMT SNP, and more than half the population has one allele for this SNP.
Food composition data and epidemiologic findings Several epidemiologic studies have examined associations of choline intake with health-related outcomes. These studies were possible because new food composition data on choline are available in tables from the U.S. Department of Agriculture (USDA).
In a case-control study, Shaw and coworkers (2004) found that women in the lowest quartile for dietary choline intake who also have a low dietary intake of folate have four times the risk of having a baby with a neural tube defect than mothers who are in the highest quartile of choline intake. More recently, Shaw and colleagues (2006) reported that the risk for cleft lip and palate in the high choline intake group is half that of the low choline intake group.
A large epidemiologic study reported an inverse relationship between dietary choline intake and plasma total homocysteine concentration (Cho et al., 2006). An intervention trial demonstrated that treatment with betaine, which is a choline metabolite, reduces the homocysteine concentration in humans (Melse-Boonstra et al., 2005).
Concluding Remarks
Dr. Zeisel ended his presentation by suggesting that future DRI panels address the roles of genetic polymorphisms when revising DRIs and that researchers consider conducting studies to determine whether there is a differential response, based on SNPs, to classical depletion– repletion.
DISCUSSION OF RESEARCH RECOMMENDATIONS
Presenter: Patrick J. Stover
Biotin
The one major gap listed for biotin was a serious lack of data to determine an EAR. The gap exists because biotin deficiency is extremely rare. Data suggest that humans obtain most of their biotin from microflora in the gut.
Among the new discoveries is the finding that biotin modifies histones (Stanley et al., 2001). It has been known for years that biotin affects the expression of enzymes in carbohydrate metabolism; the modification of histones and chromatin by biotin is an epigenetic signature that probably accounts for these findings. This finding also was confirmed with the identification of holocarboxylase synthase deficiency. This deficiency results in decreased histone biotinylation (Narang et al., 2004) and in neurological development and/or metabolic abnormalities that can be resolved with pharmacological levels of biotin. These genomic effects may provide new indicators for setting an EAR for biotin.
Vitamin B6
Major research gaps listed for vitamin B6 (pyridoxine) include better indicators for the requirement and information about genetic variation, chronic disease prevention, interaction with other vitamins, and the needs of children, the elderly, and pregnant and lactating women. Relatively little progress has been made.
Perhaps the biggest recent discovery was the confirmation that the plasma vitamin B6 concentration falls dramatically in inflammation (Chiang et al., 2005b; Gori et al., 2006; Younes-Mhenni et al., 2004). Vitamin B6 concentration is inversely correlated with C-reactive protein (CRP) and interleukin-6 (IL-6) in a number of disorders, including sickle cell, Crohn's disease, and rheumatoid arthritis. Pyridoxine supplementation has been shown to correct the deficiencies in some of these states, but not the inflammation (Chiang, 2005a).
These findings may have bearing on the UL. The loss of vitamin B6 in inflammation is not due to urinary excretion; rather the vitamin B6 is
sequestered somewhere in the body. The lowering of plasma vitamin B6 concentration may be a protective homeostatic response to inflammation. If demonstrated, this may indicate the potential for unidentified adverse effects resulting from pyridoxine supplementation.
Folate
Progress in Filling Research Gaps
A summary of progress in filling research gaps on folate appears below. Folic acid is a synthetic form of folate that is not normally found in foods—a form that can be converted to a natural form of folate once ingested.
Effects of folic acid fortification The mandatory fortification of enriched cereal grains with folic acid, which began in the United States in the late 1990s, has provided the opportunity for comprehensive analysis of risks and benefits of folic acid fortification. Good data are available on the substantial reduction of the incidence of neural tube defects that occurred following this mandatory fortification. Investigators are still examining data on effects of the fortification on conditions such as vascular disease, cancer, cognition outcomes, and dementia.
Genetic variants New genetic variants have been identified that affect carbon metabolism and that are risk factors for various disease outcomes. In addition to the MTHFR polymorphism known before 1998, researchers have identified another risk factor for neural tube defects, the MTHFD1 polymorphism, which was mentioned by Dr. Zeisel in relation to choline deficiency. Although these two human polymorphisms in folate metabolism are associated with increased risk for neural tube defects and alter one-carbon metabolism, they make up only a small fraction of the total genetic component that poses risk for neural tube defects.
Concern had been raised about whether the EARs for folate cover individuals with MTHFR polymorphism. This type of polymorphism is common—homozygotes for this population represent about 20 percent of the Caucasian and Asian populations. A recent depletion–repletion study (Guinotte et al., 2003) demonstrated that the current RDA for folate is
adequate for young women for all three MTHFR phenotypes, suggesting that there is not a need for individualized requirements.
Potential indicator of folate requirement Preliminary data from the Third National Health and Nutrition Examination Survey (NHANES III) indicate that a useful cut point appears when homocysteine concentration is graphed against serum folate. Thus, a serum folate concentration of 10 nmol/L could serve as a useful indicator for the folate requirement.
Bioavailability Relatively little research has been conducted on the bioavailability of folate. Despite several studies, the interaction of folate with vitamin B12 deficiency remains to be determined. The concern is that adequate folate or high intake of folate may exacerbate the neurological symptoms and neurodegeneration associated with vitamin B12 deficiency, but this notion has yet to be proven or dismissed.
Folate requirements for infants and other special groups Most of the work related to the role of folate in the differentiation of development is still descriptive, not mechanistic. Little progress has been made in determining whether folate requirements vary by trimester in pregnancy or in obtaining data specific to folate requirements for children, elderly persons, and women of reproductive age.
Interactions with other nutrients Considerable data are available now regarding interactions of folate with certain other B vitamins and choline, both of which affect methylation status—especially the methylation of DNA and histones, which has effects on gene expression and stem cell programming.
Measurement of folate In analytical methodology, the biggest advancements have been made in the area of developing mass spectrometry techniques to measure folate concentration in serum.
New Concern
Among persons who have high intakes of folic acid, the unmetabolized form may appear in serum (Kelly et al., 1997; Troen et al., 2006). Seventy-eight percent of fasting adult participants in one study exhibited the presence of a substantial concentration of unmetabolized folic acid in
serum. Concern has been raised about this—with regard both to children and adults. Folic acid itself is not a biologically active form of the vitamin, and it can have inhibitory properties. Troen and colleagues (2006) showed reduced natural killer cell cytotoxicity in women with folic acid present in the plasma.
Vitamin B12
Major gaps related to vitamin B12 included
-
its role in vascular disease;
-
the impact of genetic variation;
-
requirements in the elderly (one in six individuals over the age of 65 years is believed to be at risk for deficiency of vitamin B12);
-
the effect of folate on progression of clinical symptoms of vitamin B12 deficiency, especially neurological outcomes;
-
methods to detect status;
-
indicators, especially in the elderly and vegans who have a propensity to become vitamin B12 deficient; and
-
the efficacy of vitamin B12 fortification, should such fortification be initiated.
A polymorphism in transcarbolamine 2 has been found to affect indicators of blood B12 status, namely holotranscobalamin (holoTC concentration) and homocysteine concentration. Some progress has occurred in developing methods to determine vitamin B12 status: a holoTC kit is now available and may be an improvement, and other diagnostic kits are being developed.
Better indicators may also be available. In particular, the serum B12 value required to maintain hematological status (estimated earlier to be 150 pmol/L of serum B12) is consistent with cut points for the log of homocysteine and the log of methyl-malonic acid versus serum B12 that occur near 150 pmol/L of serum B12 (Unpublished data, Jacob Selhub, Tufts University, 2006).
Future Perspectives
In terms of future perspectives, Dr. Stover focused on three areas: (1) disease and pathology outcomes, (2) genetic variation and requirements, and (3) fetal/stem cell programming.
Disease and Pathology Outcomes
MTHFR polymorphism inhibits the remethylation cycle, which may increase the homocysteine concentration of the blood, decrease methylation potential, and alter chromatin structure and gene expression. These alterations in biochemistry present both benefits and risks. For example, in utero risk for neural tube defects increases, as does the risk for miscarriage; but homozygous carriers of that polymorphism have a 70 percent reduction in risk for colon cancer compared to people who carry the more prevalent allele. This raises the question of whether a given nutrient intake level presents opposing benefits and risks for different diseases.
An increasing literature raises concern that a high folate status actually may present risk for colon cancer. That is, folate may be preventive in the initiation stage of cancers, but it may accelerate cellular transformation once the cancer is initiated. Thus, great care is needed in choosing outcomes on which to base the DRIs.
Genetic Variation and Requirements
With regard to genetic variation and requirements, it will be important to assess whether or not to individualize dietary requirements to subgroups that have a specific genetic polymorphism. In doing so, it is necessary to consider both the penetrance of the polymorphism (that is, the probability of expressing a phenotype from the given genotype at a given time) and its prevalence (which is a measure of the proportion of persons in the population with a certain SNP at a given time).
Evidence suggests that few SNPs are likely to be sufficiently penetrant to warrant genotype-specific recommendations. One basis for this position is that most of the SNPs known that affect indicators of status, at least in B vitamin metabolism, also are risk factors for miscarriage. The current experience indicates that genotypes that confer large differences in nutrient utilization and hence nutritional requirements will probably be
lost in utero. However, some have suggested that the use of supplemental folate reduces rates of human spontaneous abortion. That is, given that folate apparently can prevent neural tube defects, it might rescue some of these miscarriages. The desirability of increasing disease alleles in the population is an open question and probably warrants some attention.
SNP–SNP interactions are likely to be more penetrant than most SNPs in isolation, but they have a lower prevalence. In general, as the penetrance of an SNP or SNP–SNP interaction increases, in terms of the effects of genetics on an indicator of status or outcome, the prevalence of that genetic variation decreases.
Examples of gene–gene interactions from Dr. Stover’s laboratory involve cytoplasmic serine hydroxymethyltransferase (cSHMT) and 5-MTHFR. Results from the National Aging Study cohort analysis (Lim et al., 2005) show that neither the MTHFR polymorphism or the cSHMT polymorphism is very penetrant in terms of the risk of cardiovascular disease. However, cardiovascular disease risk increases if cSHMT polymorphism occurs among heterozygotes for the MTHFR polymorphism. Moreover, for homozygotes with both of the polymorphisms, the risk increases substantially. Thus, gene–gene interactions are important, but they have low prevalence.
Dr. Stover suggested the development of a framework for establishing impacts and cut-off points for genetic variation that addresses two major points: (1) How prevalent do these polymorphisms have to be to warrant genotype specific recommendations? and (2) What is the penetrance—that is, what is the genetic contribution of variation compared to the overall variation in requirement—and is that penetrance sufficient to warrant the use of genotypic-specific results to develop nutrient recommendations for subgroups?
Fetal and Stem Cell Programming
Dr. Stover emphasized the importance of investigating fetal and stem cell programming, which first received attention by the nutrition community related to Barker’s fetal origins of adult disease hypothesis. This hypothesis suggests that fetal environmental exposures, especially nutrition, act in utero or in the neonate to program risk for adult health outcomes.
Evidence from mouse models suggests that folate can program gene expression (Cooney et al., 2002; Dolinoy et al., 2006; Morgan et al.,
1999; Waterland and Jirtle, 2003) A number of mouse models involve a genetic deletion that results either in nonviable embryos or some sort of a severe phenotype that can be rescued with nutrition. Thus, care is warranted related to the possibility that epigenetic effects modify genetic insults that exist in the human population. Dr. Stover believes that certain reprogramming events probably are common, can be modified by nutrition, and influence a number of folate-associated diseases.
Concluding Remarks
In conclusion, Dr. Stover emphasized the need to consider genetic variation, determine relevant parameters, and determine whether or not individual recommendations for B vitamin requirements are needed. Furthermore, there is a need for better understanding of epigenetic effects. In investigating new indicators for both the EAR and UL, a disease prevention approach could involve targeting the molecular antecedents of disease. Promising ones might include molecular antecedents for cancer and for other diseases with mutation rates that can be measured in the population. However, since this approach probably would result in increasing the RDA, attention also would need to be given to new concerns for ULs, including epigenetic effects and genetic rescue.
DISCUSSION
A number of comments made during the discussion relate to the DRI paradigm. Those comments appear at the end of Chapter 13, “Wrap-Up.”
Dr. Appel asked about homocysteine as a surrogate for folate intake. In particular, findings in recently published clinical trials indicated that decreases in serum homocysteine concentration were not accompanied by a reduction in adverse health outcomes. In some cases, there was the appearance of potential harm. Considering this, how could homocysteine serve as a useful surrogate for folate intake? Dr. Stover responded that those findings were from secondary prevention trials and that primary prevention trials are needed because that probably is where homocysteine is exerting its effects.














