5
Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids1
Presentations on the Dietary Reference Intakes (DRI) Antioxidants Report (IOM, 2000b) were given by Susan Taylor Mayne of Yale University School of Medicine, who served on the panel for the report, and by John N. Hathcock of the Council for Responsible Nutrition, who was asked to provide a fresh perspective. Dr. Mayne provided an overview of progress made and addressed emerging or persistent questions. (Vitamin C was not covered because of time limitations.) She also addressed consistency of the research recommendations with public health concerns, methods to address research recommendations in future DRIs, and the use of Adequate Intakes (AIs). Dr. Hathcock’s presentation focused on the DRI process.
DISCUSSION OF RESEARCH RECOMMENDATIONS: VITAMIN C, VITAMIN E, SELENIUM, AND CAROTENOIDS
Presenter: Susan Taylor Mayne
Overview
The DRI Antioxidant Report (IOM, 2000b) contains Estimated Average Requirements (EARs), Recommended Dietary Allowances (RDAs), and Tolerable Upper Intake Levels (ULs) for vitamin C, vita-
min E, and selenium for each life-stage group above age 12 months. In each case, criteria were based on a specific function and not on the prevention of chronic disease. The report also addresses beta-carotene and the carotenoids; but no EARs, RDAs, or ULs were established for any of those substances.
Selenium
Progress Made
Considerable research has been conducted on selenium in the past 6 years. In basic science, for example, 25 genes have been identified that code for selenoproteins. Characterization of these genes is now in progress and is expected to yield useful functional information and clarification of biochemical mechanisms.
Selenoprotein P is now considered a highly promising biomarker for selenium status. It could be considered along with selenium-dependent glutathione peroxidase (the current functional indicator) by a future DRI panel.
Disease prevention was and continues to be a particular area of interest related to the antioxidant nutrients. The ongoing Selenium and Vitamin E Chemoprevention Trial (SELECT), a very large cancer prevention clinical trial involving 35,000 men, will provide much needed clinical data on possible roles of selenium in disease prevention and on adverse effects that may occur with selenium supplementation. The basis for SELECT comes from the Selenium Skin Cancer Prevention Trial (Clark et al., 1996), which studied skin cancer prevention resulting from 4.5 years of supplementation with 200 µg/day of selenium in the form of selenium-enriched yeast. No reduction in second skin cancers occurred; but, unexpectedly, fewer cancers of the prostate, lung, and colorectum were noted in the group that received selenium supplements. The SELECT trial is designed to try to replicate this provocative finding. Figure 5-1 depicts the SELECT study design.
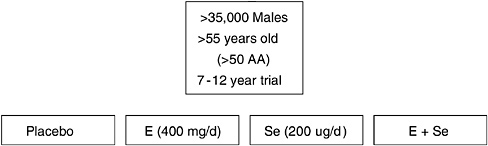
FIGURE 5-1 Overview of Selenium and Vitamin E Chemoprevention Trial (SELECT).
A subgroup analysis of the Nutritional Prevention of Cancer Trial (Duffield-Lillico et al., 2002) addressed efficacy of the selenium supplementation as a function of baseline selenium status. Although the investigators found reductions in incident cancer among subjects in the first and second tertiles of selenium intake at entry (both of which represented low selenium intakes because participants for this study were recruited from low-selenium regions of the United States), there was a 20 percent increase in the risk of total cancer among subjects in the highest tertile of selenium intake. This finding of greater efficacy in persons with lower nutrient status at baseline is supportive of results of some other antioxidant nutrient trials, indicating that baseline nutrient status may be important in determining the efficacy of supplementation with that nutrient.
Although SELECT will provide a wealth of data, a future DRI panel will face challenges in interpreting the results if no benefits are observed. Concerns have been raised about the dose of vitamin E (higher than that used in the Alpha-Tocopherol Beta-Carotene (ATBC) Trial; see further discussion), the form of selenium being used, the timing and duration of the intervention, and lifestyle factors and baseline nutritional status that may modify the effects.
Questions for Future Research
Dr. Mayne raised the following questions for future research concerning selenium:
-
Could selenoprotein P be used as the basis for setting the EAR and RDA for selenium?
-
What health risks are associated with marginal selenium intake?
-
Does supplemental selenium benefit only those with inadequate status?
-
What mechanisms are responsible for selenium's health effects? Evidence indicates that there are many possible mechanisms in addition to antioxidant function.
A persistent research gap that applies to all the antioxidant nutrients concerns the nutrient needs of children. Little or no data are available on the nutrient requirements of children or infants.
Vitamin E
Despite the considerable interest in roles of vitamin E in the prevention of chronic disease, the DRI panel on antioxidants determined that the research base was insufficient to allow the use of a chronic disease end point as a functional indicator for setting the EAR and RDA for vitamin E. Instead, the functional indicator selected for those DRI values was the prevention of hydrogen peroxide-induced hemolysis.
Progress Made
Over the past few years, much research attention has been given to vitamin E with regard to basic science progress (ongoing stable isotope studies, for example, will greatly illuminate understanding of metabolism and kinetics for vitamin E), chronic disease studies (especially coronary heart disease prevention studies in high-risk populations), and vitamin E supplementation trials for cancer prevention (primarily among high-risk populations).
The most promising data to come out of the large clinical trials on vitamin E is the possibility of a protective effect of vitamin E against prostate cancer (Virtamo et al., 2003). An unexpected 30 percent reduction in prostate cancer incidence occurred in the ATBC Trial, a lung cancer prevention trial conducted in Finland. In this trial, smokers were randomized to receive vitamin E (50 mg/day) or placebo. Since this was a lung cancer prevention trial rather than a prostate cancer prevention trial and all the subjects in the trial were smoking at entry, questions remain: Is vitamin E protective against prostate cancer? Is the effect limited to smokers only?
Additional evidence suggests that vitamin E may reduce the risk of prostate cancer in smokers only. An observational cohort study (Chan et al., 1999) found that vitamin E supplement use was not related to overall prostate cancer risk. However, among the current smokers or recent quitters in the study, an inverse association was suggested, particularly for metastatic or fatal prostate cancer. The relative risk was 0.44, which was nearly statistically significant. Similarly, in the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial, supplemental vitamin E was not associated with the overall risk of prostate cancer. With both higher dose and longer duration of vitamin E supplementation, however, a statistically significant decrease in the risk of advanced prostate cancer was observed among current and recent smokers (Kirsh et al., 2006).
Continuing Research Gaps
A continuing research gap relates to the limited information about vitamin E in food composition databases. Data still are not widely available on the alpha-tocopherol, gamma-tocopherol, and delta-tocopherol contents of foods. Moreover, evidence is needed on the biological activity of various vitamin E forms in humans.
Dr. Mayne raised a new question in terms of research challenges; namely, When conducting prevention trials, is it more informative to base the research design on unexpected results from previous trials or to base them on findings from observational epidemiology? SELECT will provide data pertinent to this question.
Beta-Carotene and the Carotenoids
The panel for the DRI Antioxidant Report considered a number of possible functional indicators for beta-carotene and the carotenoids: vitamin A equivalency, markers of antioxidant activity, modulation of gap junctional communication, immune functions, and relationship to chronic diseases such as cancer, coronary heart disease, macular degeneration, and cataract formation. Vitamin A equivalency was addressed by the DRI panel on micronutrients.
Progress Made
Uneven progress has been made. Functional indicators such as gap junctional communication, immune modulation, and antioxidant markers still remain unvalidated as being predictive of a disease outcome. However, dermal carotenoid concentration, measured using noninvasive techniques, is a possible new status indicator.
Although some prevention trial results were available to the panel before publication of the DRI Antioxidants Report in 2000, more recent analyses have identified subgroups that apparently benefit and subgroups that are harmed by supplementation. For example, one of the trials (Baron et al., 2003) found that beta-carotene significantly reduced the risk of colorectal adenoma recurrence in nonsmokers and nondrinkers, and significantly increased it in smokers and drinkers. The Supplémentation en Vitamines et Minéraux Antioxydants (SUVIMAX) trial of many different nutrients, including antioxidant nutrients, found that men benefited and women did not with regard to cancer incidence (the primary outcome studied).
Considerable progress has been made in research related to macular degeneration. The Age-Related Eye Disease Study (AREDS), a randomized clinical trial using antioxidant nutrients and zinc, found that this nutrient combination significantly reduced the risk of advanced macular degeneration among persons who already had some macular changes (Age-Related Eye Disease Study Research Group, 2001). AREDS II will be the first large controlled trial of supplemental lutein and zeaxanthin— carotenoids—for which very little human supplementation data are available. The trial also will include testing of intake of long-chain omega-3 fatty acids.
Critically needed methodologic work has been conducted on the measurement of macular pigment, a possible new status indicator. The new methods (heterochromatic flicker photometry and Raman spectroscopy) have been used in studies that look at determinants (e.g., diet, genetics, and adiposity) of macular pigment. The predictive ability of macular pigment for future disease risk is still unknown—a causal link has not yet been established.
Continuing Research Gaps
Continuing research gaps include the following:
-
The dose-dependence of carotenoid effects on health
-
Effects of polymorphisms in antioxidant-related or regulated genes on the efficacy of antioxidant nutrients
-
The role of oxidative stress in chronic disease
-
Predictive value of markers of oxidative stress for clinical end points
-
Understanding subgroup effects, especially smoking, which are evident throughout the antioxidant nutrient literature
Development and validation of biomarkers of oxidative stress are still lacking. The prevention of artifact is a big challenge in this work. Moreover, it is necessary to characterize the intra- and intersubject variability of these markers before their use can be explored in population studies.
The DRI Process
DRI Process as Related to Antioxidant Nutrients
Process questions specific to the antioxidant nutrients appear in Box 5-1. With regard to question 1, many nutrients and other food components that have antioxidant function were not addressed by the first panel
|
BOX 5-1 DRI Process Questions Related to the Antioxidant Nutrients
|
on antioxidants. This applies, for example, to many of the phytochemcals. Question 4 was considered by the first panel on antioxidants, for example, with regard to lycopene.
General Aspects of the Dietary Reference Intake Process
According to Dr. Mayne, a major issue is how to incorporate health promotion into the DRI process. The lack of functional indicators that are associated with the promotion of optimal health limited the ability of the panel on antioxidants to address this. There is a lack of congruity between the public health priority (which is the prevention of chronic diseases) and the current DRI process (which says that a functional indicator is needed to set an EAR or AI). Because of this, all the EARs in the DRI Antioxidant Report are based upon functional indicators, and none are linked to any chronic disease prevention end points. With this in mind, Dr. Mayne raised the question, “Should the concept of a range of nutrient intake be reconsidered, or is there some other method to incorporate information about disease prevention (including subgroup-specific information) into the DRI process? Dr. Mayne invited other speakers to address this issue.
DIETARY REFERENCE INTAKES FOR ANTIOXIDANT NUTRIENTS: GENERAL AND SPECIFIC FRESH PERSPECTIVES
Presenter: John N. Hathcock
The DRI Process
This presentation provided perspectives that relate mainly to the DRI process and included a number of suggestions, which are summarized below.
-
Rather than setting the RDA based on a 10 percent or 20 percent coefficient of variation (about two standard deviations higher than the EAR), consider setting it at four standard deviations above the EAR. This would cover the entire population but would not be near the UL. For vitamin E, for example, the values would be as follows:
-
EAR, 12 mg for adult males
-
Current RDA, 15 mg
-
Proposed RDA (EAR plus four standard deviations), 18 mg
-
-
Could risk assessment methodology be used to evaluate the lower end rather than just the upper end of nutrient risk? Figure 5-2 depicts the concept. Dr. Hathcock suggested that examination of the potential advantages and disadvantages of implementing this concept be undertaken by an expert national committee.
-
Can the UL concept be expanded to include a value for nutrients for which an adverse effect or a toxicity has not been clearly established, such as vitamin B12? To date, no UL has been set for such nutrients. The lack of a UL might be interpreted in either of two ways: (1) there is no UL because of a lack of adequate data, or (2) there is almost no evidence whatever of toxicity despite ample data. Dr. Hathcock suggested that a value called the Highest Observed Intake in a recent Food and Agriculture Organization and World Health Organization report (FAO/WHO, 2005) (a value that he calls an Observed Safe Level) would be a useful addition.
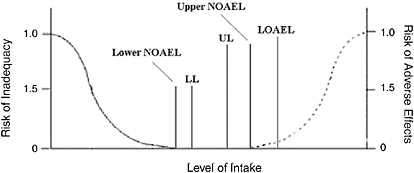
FIGURE 5-2 Hypothetical curve depicting symmetrical risk assessment.
NOTE: NOAEL = No Observed Adverse Effect Level, LL = Lower Level, UL = Tolerable Upper Intake Level, LOAEL = Lowest Observed Adverse Effect Level
-
Can the process of setting ULs be made more systematic—one that consistently uses a decision tree approach?
-
Are separate UL values needed for different forms of a nutrient? Examples include alpha- and gamma-vitamin E and forms of selenium such as selenized yeast, sodium selenide, sodium selenate, and sodium methionine.
-
Can risk and benefit curves be prepared for lutein, zeaxanthin, lycopene, and selenium chemical species and modulators for use in setting DRIs?
Other Concerns
In addition, Dr. Hathcock pointed out concerns he had regarding the meta-analysis of high-dosage vitamin E supplementation conducted by Miller and colleagues (2005) (concerns that one of the coauthors rebutted during the discussion period). Dr. Hathcock also raised questions about the advisability of terminating a randomized-controlled trial when clear benefit is seen for a secondary rather than a primary end point (as was the case in the study of selenium supplementation by Clark et al. [1996]). In particular, if a secondary end point is one that automatically has to be retested, is it appropriate to use it as the basis for stopping a study early? In short, Dr. Hathcock stated that there is a need to examine the statistical methods applied to multiple clinical trials and to multiple end points.
DISCUSSION
Discussion centered on concerns related to setting the EAR for vitamin E based on vitamin E concentrations and hydrogen peroxide-induced hemolysis—even though such hemolysis is not a clinical problem. This was the indicator for which data were available; there were insufficient data about other indicators and chronic disease end points that had been considered by the panel. For points raised about how this relates to appropriateness of the model, see the discussion section in Chapter 13.












