Appendix C
Risk Determinants Literature Search Strategy
Electronic literature searches of published systematic reviews (from 2010 to September 2015) and primary studies (from 2012 to September 2015) indexed in Medline, Cochrane Database of Systematic Reviews, EMBASE, and ISI Web of Science were conducted. For systematic reviews, a broad search was conducted to identify all systematic reviews with or without meta-analysis from 2010 onward related to food allergies or food sensitizations without restrictions to any interventions or exposures. For primary studies, search strategies in European Academy of Allergy & Clinical Immunology (EAACI) (de Silva et al., 2014) and Marrs et al. systematic reviews (Marrs et al., 2013) were adopted. The EAACI search strategies were developed to identify all randomized controlled trials, quasi-randomized controlled trials, controlled clinical trials, controlled before-and-after studies, interrupted time series studies, and prospective cohort studies that were primarily concerned with preventing sensitization to food(s) and/or the development of food allergy. The Marrs et al. search strategy was intended to capture any study designs describing food allergy or sensitization overall and to individual foods (milk, egg, peanut, tree nuts, fish, wheat, sesame, shellfish, and seafood) combined with search terms of factors that directly or indirectly influence microbial exposure (Marrs et al., 2013). All searches were restricted to human studies that were published in the English language from 2012 onward. Duplicate citations across databases were removed before screening. Medline searches conducted for this report for systematic reviews and individual studies are in Table C-1. Medline searches were used to develop the search strategies for the EMBASE and Web of Science databases.
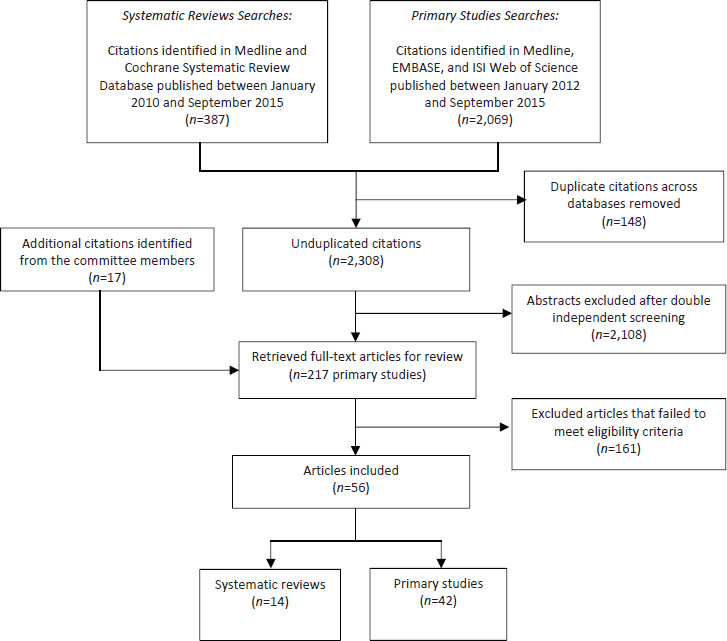
Abstrackr software (abstrackr.cebm.brown.edu), Endnote, and Microsoft Excel were used to manage the search outputs, screening, and data abstraction. After a training session to ensure understanding of the inclusion and exclusion criteria, title/abstract screening was conducted independently by two reviewers using a screening form that listed the inclusion and exclusion criteria and allowed selection of reasons for exclusion. A third reviewer reconciled the discrepant title/abstract selections. Full-text articles of all accepted title/abstracts were then retrieved and screened by one reviewer based on the study eligibility criteria. Second-level screening of full text articles was conducted by two reviewers and differences reconciled by a third reviewer. Boxes C-1 and C-2 list the study inclusion and exclusion criteria, respectively. Figure C-1 illustrates the study selection flow. Summary tables for the systematic reviews and studies selected for the evidence-based review are included in Tables C2-C6.
TABLE C-1 Medline Search Strategy to Identify Relevant Literature
| Search Number | Search Terms |
|---|---|
| a. Systematic Reviews Search Strategy | |
| 1 | exp food hypersensitivity/or exp egg hypersensitivity/or exp milk hypersensitivity/or exp nut hypersensitivity/or exp peanut hypersensitivity/or exp wheat hypersensitivity/ |
| 2 | (food$ adj2 (allergy$ or hypersensitivity)).mp. |
| 3 | ((milk or egg$ or shellfish or fish or nut$ or peanut$ or wheat or soybean$ or seasame or seafood$) adj1 (allerg$ or hypersensitivity or sensitization)).mp. |
| 4 | (sensitization or hypersensitivity).mp. |
| 5 | (food$ or diet$).mp. |
| 6 | 4 and 5 (13121) |
| 7 | 1 or 2 or 3 or 6 (15068) |
| 8 | (rat or rats or cow or cows or chicken? or horse or horses or mice or mouse or bovine or animal?).ti. |
| 9 | exp animals/not humans.sh. |
| 10 | 8 or 9 |
| 11 | 7 not 10 |
| 12 | MEDLINE.tw. |
| 13 | systematic review.tw. |
| 14 | meta analysis.pt. |
| 15 | or/12-14 |
| 16 | 11 and 15 |
| 17 | limit 16 to (English language and yr=“2010 -Current”) |
| b. Primary Studies: EAACI Search Strategy | |
| 1 | exp food hypersensitivity/or exp egg hypersensitivity/or exp milk hypersensitivity/or exp nut hypersensitivity/or exp peanut hypersensitivity/or exp wheat hypersensitivity/ |
| 2 | (food$ adj2 (allergy$ or hypersensitivity)).mp. |
| 3 | ((milk or egg$ or shellfish or fish or nut$ or peanut$ or wheat or soybean$ or seasame or seafood$) adj1 (allerg$ or hypersensitivity or sensitization)).mp. |
| 4 | (sensitization or hypersensitivity).mp. |
| 5 | (food$ or diet$).mp. |
| 6 | 4 and 5 (13121) |
| 7 | 1 or 2 or 3 or 6 (15068) |
| 8 | (rat or rats or cow or cows or chicken? or horse or horses or mice or mouse or bovine or animal?).ti. |
| Search Number | Search Terms |
|---|---|
| 9 | exp animals/not humans.sh. |
| 10 | 8 or 9 |
| 11 | 7 not 10 |
| 12 | randomized controlled trial.pt. |
| 13 | controlled clinical trial.pt. |
| 14 | randomized.ab. |
| 15 | placebo.ab. |
| 16 | clinical trials as topic.sh. |
| 17 | randomly.ab. |
| 18 | trial.ti. |
| 19 | or/16-22 |
| 20 | intervention?.ti. or (intervention? adj6 (clinician? or collaborat$ or community or complex or DESIGN$ or doctor? or educational or family doctor? or family physician? or family practitioner? or financial or GP or general practice? or hospital? or impact? or improv$ or individuali?e? or individuali?ing or interdisciplin$ or multicomponent or multi-component or multidisciplin$ or multidisciplin$ or multifacet$ or multi-facet$ or multimodal$ or multimodal$ or personali?e? or personali?ing or pharmacies or pharmacist? or pharmacy or physician? or practitioner? or prescrib$ or prescription? or primary care or professional$ or provider? or regulatory or regulatory or tailor$ or target$ or team$ or usual care)).ab. |
| 21 | (pre-intervention? or preintervention? or “pre intervention?” or postintervention? or postintervention? or “post intervention?”).ti,ab. |
| 22 | (hospital$ or patient?).hw. and (study or studies or care or health$ or practitioner? or provider? or physician? or nurse? or nursing or doctor?).ti,hw. |
| 23 | demonstration project?.ti,ab. |
| 24 | (pre-post or “pre test$” or pretest$ or posttest$ or “post test$” or (pre adj5 post)).ti,ab. |
| 25 | (pre-workshop or post-workshop or (before adj3 workshop) or (after adj3 workshop)).ti,ab. |
| 26 | trial.ti. or ((study adj3 aim?) or “our study”).ab. |
| 27 | (before adj10 (after or during)).ti,ab. |
| 28 | (“quasi-experiment$” or quasiexperiment$ or “quasi random$” or quasirandom$ or “quasi control$” or quasicontrol$ or ((quasi$ or experimental) adj3 (method$ or study or trial or design$))).ti,ab,hw. |
| 29 | (“time series” adj2 interrupt$).ti,ab,hw. |
| Search Number | Search Terms |
|---|---|
| 30 | (time points adj3 (over or multiple or three or four or five or six or seven or eight or nine or ten or eleven or twelve or month$ or hour? or day? or “more than”)).ab. |
| 31 | pilot.ti. |
| 32 | Pilot projects/ |
| 33 | (clinical trial or controlled clinical trial or multicenter study).pt. |
| 34 | (multicentre or multicenter or multi-centre or multi-center).ti. |
| 35 | random$.ti,ab. or controlled.ti. |
| 36 | (control adj3 (area or cohort? or compare? or condition or design or group? or intervention? or participant? or study)).ab. not (controlled clinical trial or randomized controlled trial).pt. |
| 37 | comment on.cm. or review.ti,pt. or randomized controlled trial.pt. |
| 38 | or/24-41 |
| 39 | exp cohort studies/ |
| 40 | cohort$.tw. |
| 41 | controlled clinical trial.pt. |
| 42 | epidemiologic methods/ |
| 43 | exp case-control studies/ |
| 44 | (case$ and control$).tw. |
| 45 | or/43-48 |
| 46 | 11 and 19 |
| 47 | 11 and 38 |
| 48 | 11 and 45 |
| 49 | or/46-48 |
| 50 | limit 49 to yr=“2012 -Current” |
| 51 | limit 50 to “review articles” |
| 52 | 50 not 51 |
| c. Primary Studies: Marrs et al. Search Strategy | |
| 1 | Measles/or measles.mp, |
| 2 | exp Mumps/or mumps.mp, |
| 3 | Whooping Cough/or whooping cough.mp, |
| 4 | exp Pneumonia/or pneumonia.mp, |
| 5 | exp Chickenpox/or chickenpox.mp, |
| 6 | hepatitis/or hepatitis a/or exp hepatitis b/ |
| Search Number | Search Terms |
|---|---|
| 7 | Hepatitis A/or exp Hepatitis B/ |
| 8 | hepatitis.mp, |
| 9 | exp Herpes Simplex/or herpes simplex.mp, |
| 10 | exp Rubella/or rubella.mp, |
| 11 | exp Helicobacter pylori/or helicobacter pylori.mp, |
| 12 | exp Tuberculosis/or tuberculosis.mp, |
| 13 | exp Mycobacterium bovis/ |
| 14 | exp Helminthiasis/ |
| 15 | helminthiasis.mp, |
| 16 | exp Helminths/ |
| 17 | helminths.mp, |
| 18 | exp Necator americanus/ |
| 19 | Necator americanus.mp, |
| 20 | exp Trichuris/or trichuris.mp, |
| 21 | exp Ascaris lumbricoides/or Ascaris lumbricoides.mp, |
| 22 | exp Schistosomiasis/or Schistosomiasis.mp, |
| 23 | exp Enterobius/ |
| 24 | enterobius vermicularis.mp, |
| 25 | exp Bacterial Infections/ |
| 26 | bacterial infection*.mp, |
| 27 | or/1-26 |
| 28 | hygiene/or skin care/ |
| 29 | hygiene.mp, |
| 30 | hygiene hypothesis.mp, |
| 31 | exp Anthroposophy/ |
| 32 | anthroposoph*.mp, |
| 33 | Child Day Care Centers/ |
| 34 | day care.mp, |
| 35 | Siblings/ |
| 36 | sibling*.mp, |
| 37 | Birth Order/ |
| 38 | birth order.mp, |
| 39 | nurser*.mp, |
| Search Number | Search Terms |
|---|---|
| 40 | agriculture/or animal husbandry/ |
| 41 | agriculture.mp, |
| 42 | farming.mp, |
| 43 | farms.mp, |
| 44 | farm.mp, |
| 45 | Animals, Domestic/ |
| 46 | pets.mp, |
| 47 | pet.mp, |
| 48 | Cats/ |
| 49 | cats.mp, |
| 50 | cat.mp, |
| 51 | Dogs/ |
| 52 | dog.mp, |
| 53 | dogs.mp, |
| 54 | exp Endotoxins/ |
| 55 | endotoxin*.mp, |
| 56 | exp Probiotics/ |
| 57 | probiotic*.mp, |
| 58 | lactobacillus.mp, |
| 59 | exp Lactobacillus/ |
| 60 | intestinal microflora.mp, |
| 61 | mycobacterium vaccae.mp, |
| 62 | Prebiotics/ |
| 63 | pre-biotic*.mp, |
| 64 | prebiotic*.mp, |
| 65 | pro-biotic*.mp, |
| 66 | exp Anti-Bacterial Agents/ |
| 67 | antibiotic*.mp, |
| 68 | Disinfectants/or disinfectant.mp, |
| 69 | vaccination.mp, |
| 70 | vaccinat*.mp, |
| 71 | unpasteuri* milk.mp, |
| 72 | unpasteuri* cow* milk.mp, |
| Search Number | Search Terms |
|---|---|
| 73 | pasteuri* milk.mp, |
| 74 | pasteuri* cow* milk.mp, |
| 75 | raw milk.mp, |
| 76 | raw cow* milk.mp, |
| 77 | unhomogeni* milk.mp, |
| 78 | unhomogeni* cow* milk.mp, |
| 79 | un-pasteuri* milk.mp, |
| 80 | un-homogeni* milk.mp, |
| 81 | or/28-80 |
| 82 | 27 or 81 |
| 83 | exp food hypersensitivity/or exp egg hypersensitivity/or exp milk hypersensitivity/or exp nut hypersensitivity/or exp peanut hypersensitivity/or exp wheat hypersensitivity/ |
| 84 | (food$ adj2 (allergy$ or hypersensitivity)).mp. |
| 85 | ((milk or egg$ or shellfish or fish or nut$ or peanut$ or wheat or soybean$ or seasame or seafood$) adj1 (allerg$ or hypersensitivity or sensitization)).mp, |
| 86 | (sensitization or hypersensitivity).mp, |
| 87 | (food$ or diet$).mp, |
| 88 | 86 and 87 |
| 89 | 83 or 84 or 85 or 88 |
| 90 | 88 and 89 |
| 91 | Cesarean Section/ |
| 92 | caesarian section.mp, |
| 93 | cesarian section.mp, |
| 94 | mode of delivery.mp, |
| 95 | microbiota.mp, |
| 95 | 82 or 91 or 92 or 93 or 94 or 95 |
| 96 | 90 and 95 |
| 97 | limit 96 to “review articles” |
| 98 | 96 not 97 |
| 99 | limit 98 to yr=“2012 -Current” |
TABLE C-2a Microbial Exposure Hypothesis (Randomized Controlled Trials)
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Prebiotics/Probiotics | ||||
| Ivakhnenko and Nyankovskyy, 2013 | Randomized controlled trial (formula feeding) + 1 BF group (nonrandomized), Ukraine | Healthy, term newborns | 80 BF infants; 160 formula fed infants (80 formula enriched with the specific mixture of oligosaccharides; 80 standard formula) | 18 months |
NOTE: BF = breastfed; CI = confidence interval; GI = gastrointestinal; OFC = oral food challenge.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Allergic reactions to food (not defined) Allergic reactions to cow milk protein (not defined) GI symptoms of food allergy (not defined) | BF (group 1) versus formula enriched with oligosaccharides (scGOS/lcFOS; 9:1; 8 g/L) (group 2) versus standard formula (group 3) | Allergic reactions to food: 2/51 (3.92%) versus 3/62 (4.84%) versus 9/53 (16.98%); P<0.05 Allergic reactions to cow milk: 1/51 (1.96%) versus 2/62 (3.23%) versus 8/53 (15.09%); P<0.05 GI symptoms of food allergy: 1/51 (1.96%) versus 2/62 (3.23%) versus 7/53 (13.21%); P<0.05 |
51 (63.7%), 62 (77.5%), and 53 (66.3%) infants in groups 1, 2, and 3, respectively, completed the study. Analysis was done in completers only. Duration and exclusivity of BF were not measured. Food allergy not confirmed by OFC. |
TABLE C-2b Microbial Exposure Hypothesis (Observational Studies)
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Prebiotics/Probiotics | ||||
| Loo et al., 2014 | Long-term follow-up of a RCT, Singapore | Asian infants at risk for allergic disease | 226 | 3-5 years |
| West et al., 2013 | Long-term follow-up of an RCT, Sweden | Healthy, term infants with no prior allergic manifestations | 121 | 8-9 years |
| Route of Delivery | ||||
| McGowan et al., 2015 | Prospective cohort, Baltimore, Boston, New York City, St. Louis | Children from the Urban Environment and Childhood Asthma (URECA) study | 516 | 1-5 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Ever had food allergy (history of convincing symptoms of food allergy and the presence of IgE allergen) since year 3 | Intervention: Cow milk formula supplemented with probiotics (BL999 and LPR) from birth to age 6 months (N=117) Control: Cow milk formula supplemented without probiotics (N=109) |
RR=1.1 (0.1-17.0) | 245 infants were randomized; 220 (87%) completed 5-year follow-up. The analysis was done in 226 children (number of dropouts by groups was not reported). |
| IgE-associated food allergy | Intervention: Infant cereals with addition of probiotics (LF19 1 × 108 CFU per serving) from 4 to age 13 months (N=59) Control: Infant cereals without addition of probiotics (N=62) |
1.05 (0.14-7.73) | 171/179 randomized infants completed the trial; 121 children in the long-term follow-up. More children in the placebo group received antibiotics during intervention than probiotic group (32.3% versus 16.9%, P=0.05). Unadjusted analysis. |
| Food allergy (N=51) or sensitization (N=286): sIgE to milk, egg, peanut; clinical history | (1) Caesarean section (food allergy versus not allergic) (2) Caesarean section (food sensitized versus not sensitized) |
(1) 23.5% versus 31.6%; P=0.31 (2) 31.5% versus 30.9%; P=0.96 |
Unadjusted analysis. |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Peters et al., 2015 | Prospective cohort, Australia | Infants from the HealthNuts study | 5,276 | 1 year |
| Grimshaw et al., 2014 | Prospective nested case-control study, UK | Cases: all infants with food allergy by age of 2 years from the Prevalence of Infant Food Allergy (PIFA) study Controls: age-matched controls from the PIFA study | 123 (41 with food allergy; 82 controls) | 1-2 years |
| Luccioli et al., 2014 | Prospective cohort, US | Children who participated in the Infant Feeding Practices Study (IFPS) II | 1,363 | 6 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| IgE-mediated food allergy = positive OFC in the presence of positive test of sensitization (SPT ≥2 mm or sIgE ≥0.35 kua/L). Separate analysis for single egg allergy (9% of the cohort), multiple food allergies predominantly peanut (3% of the cohort), and multiple food allergies predominantly egg (2% of the cohort), comparing to no allergic disease at baseline. | Caesarean section versus vaginal birth | Single egg allergy: 1.02 (0.81-1.29) Multiple food allergies - peanut: 1.24 (0.86-1.78) Multiple food allergies - egg: 0.93 (0.56-1.60) |
5,142 infants underwent SPT to egg, peanut, or sesame and 1,089 infants were eligible for hospital assessment, of whom 908 participated in OFC. |
| Food allergy determined by SPT, physical exam, clinical history, sIgE, DBPCOFC | Birth by caesarean section (cases versus controls) | 31.7% versus 24.4%; P=0.255 | Unadjusted analysis except for pet ownership. |
| Physician-diagnosed food allergy as reported by parent | Caesarean section versus vaginal birth | 1.37 (0.84-2.21) |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Depner et al., 2013 | Prospective cohort, Austria, Finland, France, Germany, Switzerland | Children from the Protection against Allergy-Study in Rural Environments (PASTURE) birth cohort | 686 | Birth to 1 year |
| Pele et al., 2013 | Prospective cohort, France | Respondents to the 2-year follow-up FFQ of the PELAGIE mother–child cohort study | 1,487 | 2 years |
| Pyrhonen et al., 2013 | Retrospective cohort study, Finland | Children identified from the South Karelian Allergy Research Project (SKARP), a population-based study comprising all children of a given age range and living in the same province. | 3,181 | 1-4 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| sIgE to food allergens (hen egg, cow milk, peanut, hazlenut, carrot, wheat flour) | Caesarean section | 1.18 (0.69-2.03) | 793 (378 farm and 415 nonfarm) children were included in the analyses, of whom 686 were included in IgE to food allergens model. |
| Mother-reported food allergy in children (N=136): 37 had a medical diagnosis of cow milk allergy, 41 had a medical diagnosis of food allergy, and 22 of both, while 36 children had no doctor’s diagnosis | Cesarean section (yes versus no) | 8.7% versus 9.1%; P=0.10 | Nonrespondents (N=1,496) were younger at the birth of the child, less educated, and more likely to smoke. These factors were considered as covariates in the paper. Unadjusted analysis results only. |
| Physician-diagnosed allergic manifestations: positive specific IgE test, SPT, open food challenge (did not specify which foods) | Caesarean section | 1.15 (0.80-1.63) | Large nonresponse rate. |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Dowhower Karpa et al., 2012 | Retrospective case-control study, US | Cases: children visiting an allergy specialty clinic for a food allergy–related concern who were also born at the institution’s medical center. Age- and sex-matched controls: children visiting primary care practice who were also born at the institution’s medical center. | 99 case; 192 controls | No data |
| Antibiotics Use | ||||
| Grimshaw et al., 2014 | Prospective nested case-control study, UK | Cases: all infants with food allergy by age of 2 years from the PIFA study Controls: age-matched controls from the PIFA study | 123 (41 with food allergy; 82 controls) | 1-2 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| ICD-9-CM coding consistent with food-related allergic reactions and a confirmed presence of food allergies documented by either a positive serum specific IgE test or positive SPT | Caesarean (cases versus controls) | 32.2% versus 33.9%; P=0.79 | Retrospective chart review. Possible selection bias. Unadjusted analysis results only. |
| Food allergy determined by SPT, physical exam, clinical history, sIgE, double-blind placebo controlled food challenge | Maternal antibiotic use (cases versus controls) | No significant associations during or after pregnancy or while breastfeeding | Unadjusted analysis except for pet ownership. |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Metsala et al., 2013 | Prospective nested case-control study, Finland | Cases: infants who had received a special reimbursement for the cost of special infant formulas based on diagnosed cow milk allergy. Controls: randomly selected and matched for date of birth, sex, and the hospital district of birth. | 16,237 case-control pairs | 0-2 years |
| Dowhower Karpa et al., 2012 | Retrospective case-control study, US | Cases: children visiting an allergy specialty clinic for a food allergy-related concern who were also born at the institution’s medical center. Age-and-sex matched controls: children visiting primary care practice who were also born at the institution’s medical center. | 99 case; 192 controls | No data |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Pediatric certification of cow milk allergy based on clinical exam, symptoms, elimination diet, SPT, and elevated serum-specific IgE or open challenge test | (1) Maternal use of antibiotics before pregnancy (2) Maternal use of antibiotics during pregnancy (3) Child’s use of antibiotics from birth to 1 month |
(1) 1.26 (1.20-1.33) (2) 1.21 (1.14-1.28) (3) 1.71 (1.59-1.84) |
|
| ICD-9-CM coding consistent with food-related allergic reactions and a confirmed presence of food allergy documented by either a positive serum specific IgE test or positive SPT | (1) Neonatal antibiotics (cases versus controls) (2) Peripartum antibiotics (cases versus controls) |
(1) 16.2% versus 12.5%; P=0.39 (2) 28.3% versus 28.1%; P=1.0 |
Retrospective chart review. Possible selection bias. Unadjusted analysis results only. |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Exposure to Animals | ||||
| Martin et al., 2015 | Prospective cohort, Australia | Infants from the HealthNuts study | 4,453 (2,795 without eczema; 1,903 with eczema) | 1 year |
| Peters et al., 2015 | Prospective cohort, Australia | Infants from the HealthNuts study | 5,276 | 1 year |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| SPT or sIgE, OFC (egg white, peanut, sesame) or parent report of recent immediate-type reaction | (1) Pet dog among infants without eczema; among infants with eczema (2) Pet cat among infants without eczema; among infants with eczema |
(1) 0.9 (0.6-1.5); 0.7 (0.5-0.9) (2) 0.9 (0.5-1.6); 0.6 (0.4-0.8) |
Same cohort as Peters et al., 2015, but different analyses and outcome definitions. [Note: cesarean section results were not extracted for this study because for this factor the analysis was unadjusted.] |
| IgE-mediated food allergy = positive OFC in the presence of positive test of sensitization (SPT ≥2 mm or sIgE ≥0.35 kua/L). Separate analysis for single egg allergy (9% of the cohort), multiple food allergies, predominantly peanut (3% of the cohort), and multiple food allergies predominantly egg (2% of the cohort), compared to no allergic disease at baseline. | (1) Dogs allowed inside the home versus no dogs (2) Dogs outside only versus no dogs (3) Pet cats versus no dogs |
(1) Single egg allergy: 0.76 (0.56-1.05) Multiple food allergies - peanut: 0.40 (0.21-0.73) Multiple food allergies - egg: 0.59 (0.26-1.34) (2) Single egg allergy: 1.56 (1.10-2.21) Multiple food allergies - peanut: 0.82 (0.44-1.54) Multiple food allergies - egg: 0.39 (0.13-1.18) (3) Single egg allergy: 0.80 (0.57-1.12) Multiple food allergies - peanut: 0.83 (0.47-1.47) Multiple food allergies - egg: 0.86 (0.38-1.91) |
5,142 infants underwent SPT to egg, peanut or sesame and 1,089 infants were eligible for hospital assessment, of whom 908 participated in OFC. |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Grimshaw et al., 2014 | Prospective nested case-control study, UK | Cases: all infants with food allergy by age of 2 years from the PIFA study Controls: age-matched controls from the PIFA study | 123 (41 with food allergy; 82 controls) | 1-2 years |
| Stelmach et al., 2014 | Prospective cohort, Poland | Children from the Polish Mother and Child Cohort Study (REPRO_ PL cohort) | 501 | 1-2 years |
| Depner et al., 2013 | Prospective cohort, Austria, Finland, France, Germany, Switzerland | Children from the Protection against PASTURE birth cohort | 686 | Birth to 1 year |
| Goldberg et al., 2013 | Prospective case-cohort study, Israel | Cases: IgEcow milk allergy children identified from a cohort study (Katz, 2010) Controls: healthy children randomly chosen from the cohort | 66 cases 156 controls | 2-3 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Food allergy determined by SPT, physical exam, clinical history, sIgE, DBPCOFC | Pet ownership (yes versus no) | 1.275 (0.49-3.33) | |
| Food allergy ever diagnosed by doctor according to international guidelines | Pets at home during pregnancy (yes versus no) | 1.48 (1.02-2.16) | Frequency of cleaning was not associated with food allergy and was dropped out from multivariate model. |
| sIgE to food allergens (hen egg, cow milk, peanut, hazlenut, carrot, wheat flour) | (1) Early contact with sheep, goats, hares (2) Farming |
(1) 0.92 (0.75-1.13) (2) 2.11 (1.33-3.34) |
793 (378 farm and 415 nonfarm) children were included in the analyses, of whom 686 were included in IgE to food allergens model. |
| IgE-mediated cow milk allergy defined by a suggestive history of an immediate response, a positive SPT response, and, in most cases, a positive challenge result to cow milk protein | Pets in home (cases versus controls) | 26.2% versus 30.1%; P=0.72 | Unadjusted analysis. |
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Pele et al., 2013 | Prospective cohort, France | Respondents to the 2-year follow-up FFQ of the PELAGIE mother–child cohort study | 1,487 | 2 years |
| Koplin et al., 2012 | Prospective cohort, Australia | Infants from the HealthNuts study | 4,963 | 1 year |
NOTE: CI = confidence interval; DBPCOFC = double-blind, placebo-controlled oral food challenge; FFQ = food frequency questionnaire; IgE = immunoglobulin E; OFC = oral food challenge; RAST = radioallergosorbent test; RR = relative risk; sIgE = food-specific serum IgE; SPT = skin prick test; UK = United Kingdom; US = United States.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Mother-reported food allergy in children (N=136): 37 had a medical diagnosis of cow milk allergy, 41 a medical diagnosis of food allergy, and 22 of both, while 36 children had no doctor’s diagnosis | Farm animal contact (yes versus no) | 8.9% versus 9.1%; P=0.88 | Nonrespondents (N=1,496) were younger at the birth of the child, less educated, and more likely to smoke. These factors were considered as covariates in the paper. Unadjusted analysis results only. |
| IgE-mediated egg allergy: Allergic on formal egg challenge or previous history of clear reaction to egg occurring within 1 month of a positive SPT or RAST | (1) Dog outside only versus no dog (2) Dog allowed inside versus no dog (3) Cat outside only versus no cat (4) Cat allowed inside versus no cat |
(1) 1.09 (0.75-1.57) (2) 0.72 (0.52-0.99) (3) 0.93 (0.49-1.77) (4) 0.75 (0.52-1.09) |
Same cohort as Peters et al., 2015 but different analyses and outcome definitions. |
TABLE C-3a Allergen Avoidance Hypothesis (Randomized Controlled Trials)
| Author Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Breastfeeding | ||||
| Ivakhnenko and Nyankovskyy, 2013 | Randomized controlled trial (formula feeding) + 1 BF group (nonrandomized), Ukraine | Healthy, term newborns | 80 BF infants; 160 formula fed infants (80 formula enriched with the specific mixture of oligosaccharides; 80 standard formula) | 18 months |
| Infant Formula | ||||
| Lowe et al., 2011 | RCT, Australia | Infants with a family history of allergic disease | 620 | 6, 12, and 24 months |
NOTE: BF = breastfed; CI = confidence interval; GI = gastrointestinal; pHWF = partially hydrolyzed whey formula.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcom Definitione | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Allergic reactions to food (not defined) Allergic reactions to cow milk protein (no defined) GI symptoms of food allergy (not defined) | BF (group 1) versus formula enriched with oligosaccharides t (scGOS/lcFOS; 9:1; 8 g/L) (group 2) versus standard formula (group 3) | Allergic reactions to food: 2/51 (3.92%) versus 3/62 (4.84%) versus 9/53 (16.98%); P<0.05 Allergic reactions to cow milk: 1/51 (1.96%) versus 2/62 (3.23%) versus 8/53 (15.09%); P<0.05 GI symptoms of food allergy: 1/51 (1.96%) versus 2/62 (3.23%) versus 7/53 (13.21%); P<0.05 |
51 (63.7%), 62 (77.5%), and 53 (66.3%) infants in groups 1, 2, and 3, respectively, completed the study. Analysis was done in completers only. Duration and exclusivity of BF were not measured. Food allergy not confirmed by OFC. |
| Food reaction, SPT (milk, egg, peanut) | Soy-based formula, pHWF, or cow milk formula at cessation of breastfeeding | Positive SPT to cow milk within first 2 years: pHWF versus CMF: 0.79 (0.35-1.77) Soy formula versus CMF: 0.78 (0.32-1.92) Any food reaction: pHWF versus CMF: 0.95 (0.51-1.75) Soy formula versus CMF: 1.21 (0.67-2.19) |
TABLE C-3b Allergen Avoidance (Observational Studies)
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Maternal Intake During Pregnancy and Lactation | ||||
| Bunyavanich et al., 2014 | Prospective cohort, US | Mother-child pairs in the Project Viva prebirth cohort recruited from a large multidisciplinary practice | 1,277 mother–child pairs | 7.9 years (mean) |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Food allergy to peanut, milk, wheat, egg, and/or soy based on sIgE to the particular food and EpiPen prescribed. Food allergy to peanut was more specifically defined by parent report of convincing symptoms of a peanut allergic reaction (history of peanut allergy AND a cutaneous, respiratory, cardiovascular, gastrointestinal and/or anaphylactic symptom following peanut ingestion). | Maternal intake (total servings per day as measured by FFQ) during first and second trimester of: (1) peanut (2) milk (3) wheat (4) egg (5) soy Intake reported as z-scores |
First trimester (1) 0.53 (0.30-0.94) (2) 0.90 (0.50-1.62) (3) 1.26 (0.75-2.12) (4) 0.76 (0.28-2.08) (5) 0.61 (0.16-2.31) Second trimester (1) 0.88 (0.61-1.27) (2) 1.47 (0.91-2.37) (3) 1.07 (0.62-1.85) (4) 0.77 (0.28-2.15) (5) 1.18 (0.95-1.48) |
All ORs are adjusted for child age, sex, breastfeeding history, parental atopy, and maternal education. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Frazier et al., 2014 | Prospective cohort, US | Boys and girls (born between 1990 and 1994) participating in the Growing Up Today Study 2 (GUTS2) and their mothers. (These are children of women in the Nurse’s Health Study II.) |
8,205 mother–child pairs | Unclear |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Peanut or tree nut (walnut, almond, pistachio, cashew, pecan, hazelnut, macadamia, and Brazil nut) allergy in offspring based on maternal confirmation of food allergy diagnosis, review of physical copies of laboratory results of testing (SPT, sIgE, OFC) by two board-certified pediatricians, and confirmation of food allergy in writing from the child’s treating physician | Peripregnancy maternal consumption of peanuts or tree nuts: (1) <1 serving/month (2) 1-3 servings/month (3) 1-4 servings/week (4) ≥5 servings/week |
Multivariable OR (1) reference group (2) 0.90 (0.55-1.48) (3) 0.65 (0.43-0.97) (4) 0.58 (0.34-0.99) Ptrend=0.04 |
The dietary questionnaires were not specific for the actual dates of the pregnancy but were chosen as the one completed closest to the child’s date of birth. Only 45% of the dietary questionnaires were completed during the pregnancy; 76% were within 1 year of the pregnancy. Multivariable models control for continuous maternal age, maternal history of non-nut food allergy, maternal allergic rhinitis, eczema, or asthma, and season at child’s birth (spring or summer versus fall or winter). |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Mother-reported food allergy in children (N=136): 37 had a medical diagnosis of cow milk allergy, 41 a medical diagnosis of food allergy, and 22 of both, while 36 children had no doctor’s diagnosis | (1) Maternal pre-pregnancy consumption of fish (<1 time/month versus 1-4 times/month) (2) Maternal pre-pregnancy consumption of fish (<1 time/month versus ≥2 times/week) (3) Maternal pre-pregnancy consumption of shellfish (<1 time/month versus ≥1 time/month) All exposures measured by FFQ |
(1) 1.27 (0.72-2.24) (2) 1.48 (0.80-2.76) (3) 1.62 (1.11-2.37) |
Nonrespondent mothers (N=1,496) were younger at the birth of the child, less educated, and more likely to smoke than the participants (N=1,500). These factors were considered as covariates in the paper. ORs adjusted for: mother’s age, maternal education, folic acid supplementation, familial history of asthma/allergy, child’s sex, small-for-gestational age, infant’s method of feeding, day care attendance, postnatal exposure to tobacco, and child’s age at follow-up. |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Food allergy (N=51) or sensitization (N=286): sIgE to milk, egg, peanut; clinical history | (1) Ever BF | (1) Food allergy versus no food allergy: 35/51 (68.8%) versus 193/377 (52.9%); P=0.05 Food sensitization versus no food sensitization: 161/286 (58.3%) versus 121/230 (53.8%); P=0.35 | Of the 609 children initially enrolled, 516 (85%) were included. Unadjusted analysis. |
| (2) BF at 3 months | (2) Food allergy versus no food allergy: 16/51 (32.7%) versus 76/377 (22.8%); P=0.18 Food sensitization versus no food sensitization: 64/286 (25.1%) versus 48/230 (23.4%); P=0.76 |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Peters et al., 2015 | Prospective cohort, Australia | Infants from the HealthNuts study | 5,276 | 1 year |
| Grimshaw et al., 2013 | Prospective nested case-control study, UK | Cases: all infants with food allergy by age of 2 years from the Prevalence of Infant Food Allergy (PIFA) study Controls: age-matched controls from the PIFA study | 123 (41 with food allergy; 82 controls) | 1-2 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| IgE-mediated food allergy = positive OFC in the presence of positive test of sensitization (SPT ≥2 mm or sIgE ≥0.35 kua/L). Separate analysis for single egg allergy (9% of the cohort), multiple food allergies predominantly peanut (3% of the cohort), and multiple food allergies predominantly egg (2% of the cohort), comparing to no allergic disease at baseline | Duration of BF (up to 12 months) | Single egg allergy: 1.02 (0.99-1.04) Multiple food allergy (predominantly peanut): 1.00 (0.96-1.05) Multiple food allergy (predominantly egg): 1.17 (1.09-1.24) | 5,142 infants underwent SPT to egg, peanut, or sesame and 1,089 infants were eligible for hospital assessment, of whom 908 participated in OFC. Multinomial logistic regression was used to determine risk factors for each class, also weighted for posterior probabilities of class membership. Three separate multivariable models were fitted for the three categories of risk factors (parental, infant, and environmental). |
| Food allergy determined by SPT, physical exam, clinical history, sIgE, DBPCOFC | (1) BF duration, median weeks (2) Exclusive BF, median weeks (3) % BF initiation |
(1) Cases versus controls: 21.0 (3.0-30.5) versus 24.0 (7.0-31.0); P=0.295 (2) Cases versus controls: 5.0 (2.8-16.3) versus 8.5 (4.0-15.0); P=0.933 (3) Cases versus controls: 92.7% versus 96.3%; P=0.21 |
Only age adjusted (matching factor). |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Liao et al., 2014 | Prospective cohort, Taiwan | Infants ≥37 weeks from the The Prediction of Allergy in Taiwanese Children (PATCH) cohort | 258 (238, 226, 217, 210, and 198 completed 6, 12, 18, 24, and 36 months of follow-ups) | 6, 12, 18, 24, and 36 months |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| sIgE antibody included a mix of six common allergens: Dermatophagoides pteronyssinus (Dp) Dermatophagoides farinae (Df), egg white, cow milk, Cladosporium herbarum (Hormodendrum), and wheat. Participants were characterized as atopic or been sensitized if any of the sIgE level was greater than 0.35 IU/ml. | (1) Exclusive BF ≥4 versus <4 months, (2) Partial BF |
(1) Cow milk sensitization at 6, 12, 18, 24, 36 months: 1.0 (0.3, 3.3); 0.2 (0.07-0.5; 0.2 (0.07-0.5); 0.3 (0.1-0.7); 0.6 (0.2-1.7) Egg sensitization at 6, 12, 18, 24, 36 months: 1.3 (0.5-3.5); 1.4 (0.5-3.7); 1.6 (0.7-3.8); 1.6 (0.7-3.7); 0.7 (0.2-2.0) (2) Cow milk sensitization: There was a trend of reduced risk for cow milk protein sensitization as duration of partial breastfeeding was increased; the result was not statistically significant |
Of the original 258 neonates, blood samples and questionnaires were available from 238 infants at the age of 6 months. 226, 217, 210, and 198 children completed 12, 18, 24 and 36 months of follow-ups, respectively. Unadjusted analysis only. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Luccioli et al., 2014 | Prospective cohort, US | Children who participated in the Infant Feeding Practices Study (IFPS) II | 1,363 (823 high-risk group) | 6 years |
| Mailhol et al. 2014 | Cross-sectional study, France | Children (0 to 18 years of age) with atopic dermatitis seen consecutively at multidisciplinary clinics from May 2002 to December 2008 | 386 | 0 to 18 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Total pFA (all children with a current physician diagnosis of food allergy at age 6 years). (N=89, 7%) New pFA (subset of children with physician diagnosis of food allergy at age 6 years but with no diagnosis before 1 year of age) (N=71, 5.2%) High-risk pFA (subset of children with pFA at age 6 years and report of any of the following atopic risk factors: family history of food allergy, family history of other atopy, or eczema before age 1 year) |
Exclusive BF duration 1-3 months, ≥4 months versus 0 months (reference group) | Total pFA: Exclusive BF 1-3 month = 0.72 (0.42-1.23) Exclusive BF ≥4 months = 0.69 (0.36-1.29) New pFA: Exclusive BF 1-3 month = 0.78 (0.43-1.38) Exclusive BF ≥4 months = 0.51 (0.24-1.03) High risk pFA: Exclusive BF 1-3 month = 0.81 (0.42-1.51) Exclusive BF ≥4 months = 0.58 (0.26-1.25) |
Adjusted for mother’s education, race, income, child’s gender, parity, type of delivery, family history of food allergy, family history of other atopy, reported eczema before age 1 year, maternal tobacco smoke, other tobacco smoke exposure in home, complementary food introduction by infant age. |
| SPT. Positive (histamine 10 mg/mL [Stallergenes, Antony, France]) and negative controls and fresh foods or commercial extracts in the case of food items with histamine-releasing properties were used | Exclusive BF yes versus no | 1.8 (0.9-3.5) | Among the 386 evaluated children, food allergy was diagnosed in 69 children, of whom 26 children had a reaction to more than one food item. Duration of exclusive BF was not measured. Note: exclusive BF was dropped out in the final model. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Stelmach et al., 2014 | Prospective cohort, Poland | Children from the Polish Mother and Child Cohort Study (REPRO_ PL cohort) | 501 | 1-2 years |
NOTE: BF = breastfed; CI = confidence interval; FFQ = food frequency questionnaire; IgE = immunoglobulin E; OFC = oral food challenge; OR = odds ratio; pFA = probable food allergy; RCT = randomized controlled trial; sIgE = food-specific serum IgE; SPT = skin prick test; UK = United Kingdom; US = United States.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Food allergy ever diagnosed by doctor according to international guidelines | Duration of BF (up to 12 months) | 0.88 (0.82-0.95) | A stepwise forward procedure was then used to select variables. |
TABLE C-4a Dual Antigen Hypothesis (Randomized Controlled Trials)
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Timing of Introduction of Solid Foods and Infant Feeding | ||||
| DuToit et al., 2016 | RCT, UK (follow-up to primary trial [DuToit et al., 2015]) | Children, median age 61.3 months, who had completed the primary trial. Half were in the peanut-avoidance group; the other half were in the peanut-consumption group. | 628 | 72 months |
| Perkin et al., 2016 | RCT, UK | Exclusively breastfed infants age 3 months in the general population | 1,303 | 3 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| DBPCOFC to peanut | 12 months of peanut avoidance (peanut-avoidance versus peanut-consumption group based on primary trial) | Prevalence of peanut allergy at 72 months: Peanut-avoidance group: 18.6% Peanut-consumption group: 4.8% P<0.001 |
|
| DBPCOFC (peanut, cooked egg, cow milk, sesame, whitefish and wheat) | Early Introduction Group (EIG): early introduction of 6, allergenic foods Or Standard Introduction Group (SIG): exclusive BF to ~6 months of age. After 6 months, the consumption of allergenic foods was allowed according to parental discretion. |
Intention to Treat Analysis
Per Protocol analysis
|
Low adherence to the protocol in the EIG (42.8%). |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Clinical history, SPT, OFC, DBPCOFC, sIgE (peanut) 530 had negative SPT at baseline 98 had positive SPT at baseline |
Peanut intake (avoidance versus consumption) | Prevalence of peanut allergy at 60 months: SPT Negative Group 13.7% avoidance group 1.9% consumption group (P<0.001) (86.1% relative reduction in the prevalence of peanut allergy) SPT Positive Group 35.3% avoidance group 10.6% consumption group (P=0.004) (70.0% relative reduction in the prevalence of peanut allergy) No significant between-group difference in the incidence of serious adverse events |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Palmer et al., 2013 | RCT, Australia | Singleton term infants with symptoms of moderate-to-severe eczema. Infants who had begun solids before 4 months of age or who had any previous known direct ingestion of egg were excluded | 86 49 egg group 37 rice group (control) |
12 months |
NOTE: CI = confidence interval; EIG = Early Introduction Group; IgE = immunoglobulin E; OFC = oral food challenge; SIG = Standard Introduction Group; sIgE = food-specific serum IgE; SPT = skin prick test; UK = United Kingdom; US = United States.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| IgE-mediated egg allergy, as defined based on the results of an observed pasteurized raw egg challenge and SPT | 1 teaspoon of pasteurized raw whole egg powder versus rice powder (control) daily from 4 to 8 months of age Cooked egg was introduced to both groups after an observed feed at 8 months |
RR: 0.65 (0.38-1.11) | At 4 months of age, before any known egg ingestion, 36% (24/67) of infants already had egg-specific IgE levels of greater than 0.35 kilounits of antibody (kUA)/L. Egg-specific IgG4 levels were significantly (P<0.001) greater in the egg group at both 8 and 12 months. |
TABLE C-4b Antigen Exposure Hypothesis (Observational Studies)
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Diet Diversity | ||||
| Grimshaw et al., 2014 | Nested case control, UK | Infants from the PIFA Study who had been diagnosed as having a food allergy and their 2 age-matched controls | 41 cases 82 controls |
2 years |
| Roduit et al., 2014 | Prospective cohort, Europe | Children from rural areas in five European countries | 856 | Up to age 6 years |
NOTE: CI = confidence interval; DBPCOFC = double-blind, placebo-controlled oral food challenge; sIgE = food-specific serum IgE; SPT = skin prick testing; UK = United Kingdom.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Parent report, physical exam, SPT, sIgE, exclusion diet, DBPCOFC | Dietary patterns during first year of life | Scores were significantly different between the food allergic and control infants (P=0.002) for component 1 (diet high in fruits and vegetables) | Early infant feeding patterns did not have an association with the later development of food allergy. Children who did not have a food allergy by the age of 2 years had a dietary pattern in later infancy characterized by higher intake of fruits, vegetables, and home-prepared foods as compared to children who had a food allergy. Unadjusted analysis only. |
| Parent report of doctor diagnosis; sIgE (hen egg, cow milk, peanut, hazelnut, carrot, and wheat flour) | Food diversity during first year of life (1) 0-3 items (2) 4-5 items (3) 6 items (ref) (4) diversity score continuous |
(1) 4.43 1.62-12.10 (2) 1.85 1.02-3.35 (3) 1 (4) 0.70 (0.57-0.86), |
Adjusted for center, farmer, parents with allergy, sex, breastfeeding, siblings, and maternal education. |
TABLE C-5 Nutritional Immunomodulation Hypothesis (Observational Studies)
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Lipids/Omega-3 Fatty Acids (see systematic reviews below) | ||||
| Vitamin D | ||||
| Koplin et al., 2016 | Prospective cohort, Australia | Infants participating in the HealthNuts study | 5,276 | 1 year |
| Baek et al., 2014 | Cross-sectional, Korea | Children with atopic dermatitis or suspected food allergy, who had not been on vitamin supplementation for at least 1 month before the study | 226 | 3-24 months |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| SPT, sIgE, OFC | Serum 25(OH)D3 ≤50 nmol/L = vitamin D insufficiency 51-74 nmol/L = intermediate vitamin D ≥75 nmol/L = high vitamin D |
Infants with GG genotype (insufficient versus intermediate): 6.0 (0.9-38.9) Infants with GT/TT phenotypes (insufficient versus intermediate): 0.7 (0.2-2.0) Infants with GG genotype (high versus intermediate): 4.0 (1.3-12.9) |
Adjusted for infants’ consumption of egg and formula use and parents’ country of birth and used a seasonally adjusted measure of serum 25(OH)D3. |
| History of acute reaction + sIgE ≥0.35 kU/L or >95% predictive decision points | Serum 25(OH)D3 Deficiency: <20ng mL Insufficiency: 20-29ng/mL Sufficiency: ≥30ng/mL |
Deficient versus sufficient Food allergens: 5.0 (1.8-14.1) Milk: 10.4 (3.3-32.7) Wheat: 4.2 (1.1-15.8) |
Vitamin D deficiency increased the risk of sensitization to food allergens, especially to milk and wheat. The Scoring Atopic Dermatitis index was independently related to 25(OH)D levels after adjusting for the level of sensitization. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Norizoe et al., 2014 | Longitudinal Study, Japan | Infants with facial eczema and their mothers | 164 | 3-24 months |
| Wawro et al., 2014 | Cross-sectional, Germany | Samples from two German birth cohort studies | 2,815 | 10 years |
| Allen et al., 2013 | Cross-sectional, Australia | Infants from HealthNuts population-based cohort | 5,276 | 1 year |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| Doctor-diagnosed allergic incidents, including atopic dermatitis, food allergy with or without being positive for IgE food allergens, or wheeze or asthma with or without being positive for IgE inhaled allergens | Maternal vitamin D (800 IU/day) supplement | RR=3.42 (1.02-11.77) | Vitamin D supplementation may not decrease the severity of infantile eczema at 3 months of age, but may rather increase the risk of later food allergy up to 2 years of age. Unadjusted analysis only. |
| sIgE >0.35 kU/l | Serum 25(OH)D Q1: <57.9 (nmol/L) Q2: 57.9- <71.5 Q3: 71.5- <87.8 Q4: ≥87.8 Continuous variable |
1 0.91 (0.67-1.25) 1.25 (0.93-1.69) 1.30 (0.97-1.75) 1.07 (1.02-1.11) |
Lifetime prevalence also was significantly related to vitamin D status. |
| OFCs + SPT/sIgE ≥0.35 kU/L | Vitamin D ≥50 nmol/L (insufficiency) (1) All infants (2) Infants with one or both parents born overseas (3) Infants with both parents born in Australia |
Any food allergy versus none: (1) 1.29 (0.51-3.25) (2) 0.39 (0.08-1.76) (3) 3.08 (1.10-8.59) Peanut allergy versus none (infants with both parents born in Australia): 11.51 (2.01-65.79) Egg allergy versus none (infants with both parents born in Australia): 3.79 (1.19-12.08) |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Liu et al., 2013 | Prospective longitudinal cohort study, US | Children in the Boston Birth cohort | 460 | 0-3 years |
| Weisse et al., 2013 | Prospective longitudinal cohort study, Germany | Mother–child pairs from the Lifestyle and environmental factors and their Influence on Newborns Allergy risk (LINA) cohort study | 378 | First 2 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| sIgE >0.35 kU/l | Vitamin D (ng/ml) cord blood/postnatal ≥11/≥30 (reference) ≥11/<30 <11/≥30 <11/<30 |
All children: 1 0.73 (0.42-1.29) 0.90 (0.54-1.51) 2.03 (1.02-4.04) Children with C allele of rs2243250: 1 0.52 (0.23–1.18) 1.26 (0.65–2.43) 3.23 (1.37-7.60) |
There was no association between low vitamin D status and food sensitization at any single time point alone. Adjusted for a child’s sex and ancestry proportion, breastfeeding, postnatal maternal smoking, household income, and maternal age. |
| Parental report of a doctor diagnosis. tIgE levels >0.7 kU/l in cord blood and >3.8 kU/l at age of 1 or 2 yrs, or sIgE >0.35 kU/l | Maternal vitamin D Median = 22.2 ng/ml (55.41 nmol/ml) Cord blood vitamin D Median = 10.95 ng/ml (27.33 nmol/ml) |
1st year of life: 1.27 (0.67–2.40) 2nd year of life: 3.66 (1.36–9.87) 2-year lifetime period: 1.91 (1.09–3.37) 1st year of life: 0.92 (0.45–1.85) 2nd year of life: 4.65 (1.50–14.48) 2-year lifetime period: 1.70 (0.92-3.14) |
Adjusted for sex, number of siblings, increased cord blood tIgE levels, family atopy history, cotinine levels during pregnancy, breastfeeding, UV intensity at birth and vitamin D supplementation within the first year of life. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Jones et al., 2012 | Prospective longitudinal cohort study, Australia | High-risk infants | 231 | 12 months |
| Liu et al., 2011 | Prospective longitudinal birth cohort, US | Mother-infant pairs in the Boston Birth Cohort | 649 children | Around 2 years |
| Lipids/Omega-3 Fatty Acids (see systematic reviews below) | ||||
| Folate | ||||
| Okupa et al., 2013 | Cohort study, US | Children at high risk of developing asthma and allergic disease | 138 | 2, 4, 6, and 8 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| SPT History of immediate symptoms + SPT | Cord blood vitamin D <50 nmol/L versus ≥75 nmol/L (reference) |
Risk of allergen sensitization: 1.0 (0.9-1.01) Risk of developing IgE-mediated food allergy: 1.00 (0.99-1.02) |
Adjusted for season of birth, pets in the home, infant sex, maternal age, maternal education, and ethnicity. |
| sIgE (milk, egg white, peanut, soy, shrimp, walnut, cod fish, and wheat) | Cord blood plasma total 25(OH)D concentrations (<11 ng/ml = deficiency) | Vitamin D deficient versus not deficient (reference) Any food sensitization: 1.16 (0.83-1.63) Egg sensitization: 0.84 (0.56-1.27) Milk sensitization: 1.15 (0.76-1.73) Peanut sensitization: 1.06 (0.64-1.75) |
|
| Allergic sensitization (sIgE to milk/egg/peanut for years 1 to 3 and egg/peanut for years 5+) | Plasma folate levels | High versus low folate levels at or before age 6 years): 8% versus 26% (P=0.02) | Unadjusted analysis only. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Dunstan et al., 2012 | Prospective cohort, Australia | Pregnant women (healthy nonsmokers with uncomplicated term pregnancies) | 628 women 484 infants |
12 months |
| Magdelijns et al., 2011 | Prospective birth cohort, the Netherlands | Children in the KOALA birth cohort | 2,834 | 2 years |
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| IgE-mediated food allergy was defined as a history of immediate symptoms following contact and/or ingestion and a positive SPT to the implicated food Allergic sensitization (milk, peanut, whole egg) was assessed by SPT at 1 year of age |
Tertiles of maternal folate intake from supplements (FFQ during 3rd trimester) (1) <200 mg/day (2) 200-499 mg/day (3) >500 mg/day Tertiles of cord blood folate at delivery (1) <50.3 nmol/l (2) 50.3-75.1 nmol/l (3) >75.1 nmol/l |
Folate intake: IgE-mediated food allergy (1) reference group (2) 1.4 (0.7-3.0) (3) 1.1 (0.5-2.4) Sensitized to food allergens (1) reference group (2) 1.3 (0.7-2.3) (3) 1.1 (0.6-2.0) Cord blood folate: IgE-mediated food allergy (1) 1.7 (0.5-5.6) (2) reference group (3) 2.6 (0.9-8.1) Sensitized to food allergens (1) 2.2 (0.9-5.6) (2) reference group (3) 1.1 (0.5-2.4) All ORs adjusted for maternal allergy and infant postnatal diet |
Also looked at maternal intake of folate from food, but found no differences in the maternal dietary folate intakes of infants with any allergic outcomes. Because of the focus on allergy in the primary study, sensitized allergic mothers were over represented (70.6%). All ORs adjusted for maternal allergy and infant postnatal diet. |
| sIgE (hen egg, cow milk, peanut, and aeroallergens) | Folic acid supplement use during pregnancy (measured as quintiles of intracellular folate status during 3rd trimester) | Folic acid supplement use versus no use (reference) Increased sIgE: 1.06 (0.67-1.68) |
Allergic sensitization was to both food allergens and aeroallergens. |
| Author, Year | Study Design, Country | Population | N | Age When Outcome Was Ascertained |
|---|---|---|---|---|
| Other Nutrient Intakes | ||||
| West et al., 2012 | Prospective cohort, Australia | Pregnancy cohort with a family history of allergic rhinitis, asthma, eczema, food or other allergy | 300 mother–infant pairs | 12 months |
NOTE: CI = confidence interval; FFQ = food frequency questionnaire; IgE = immunoglobulin E; OFC = oral food challenge; OR = odds ratio; RCT = randomized controlled trial; RR = relative risk; sIgE = food-specific serum IgE; SPT = skin prick test; tIgE = total IgE; UK = United Kingdom; US = United States; UV = ultraviolet.
a Bold indicates statistical significance at P<0.05. Results were reported as odds ratio (95% confidence interval) unless otherwise noted. Adjusted results were extracted in the summary table unless otherwise noted.
| Food Allergy or Sensitization Outcome Definition | Exposure | Odds Ratioa (95% CI) of Food Allergy | Comments |
|---|---|---|---|
| IgE-mediated food allergy was defined as a history of immediate symptoms after contact with and/or ingestion and a positive SPT at 12 months (milk, egg, peanut) | Quartiles of daily maternal dietary and total intakes during pregnancy (1) b-carotene (2) vitamin C (3) vitamin E (4) copper (5) zinc Quartiles Q1 lowest (reference) Q2 Q3 Q4 highest Measured by semiquantitative FFQ administered after 28 weels gestation |
(1) b-carotene 0.40 (0.12-1.32) 1.16 (0.43-3.11) 0.38 (0.11-1.27) Ptrend=0.2 (2) Vitamin C 0.22 (0.06-0.78) 0.75 (0.27-2.06) 0.46 (0.16-1.36) Ptrend=0.1 (3) Vitamin E 0.96 (0.32-2.84) 0.86 (0.29-2.54) 0.57 (0.19-1.72) Ptrend=0.8 (4) copper 0.60 (0.22-1.60) 0.40 (0.13-1.22) 0.38 (0.11-0.95) Ptrend=0.2 (5) zinc 0.67 (0.22-2.03) 1.28 (0.46-3.53) 0.52 (0.16-1.73) Ptrend=0.4 |
Adjusted for maternal education, paternal history of allergic disease, birth weight, and exposure to furred pets at home; all were included in the multiple logistic regression model. |
TABLE C-6 Systematic Review Summaries
| Author, year | Best et al., 2016 | ||
| Aims/Key questions | To develop a clearer understanding of the effect to the developing fetus, before commencement of the progression of atopy (“atopic march”) and establishment of allergic disease symptoms. | ||
| Study eligibility criteria |
Inclusion criteria:
|
||
| Literature search dates or year range | Inception to July 30, 2015 | ||
| Number of food allergy studies included | 3 RCTs (based on SPT) | ||
| Synthesis methods | Summary tables and meta-analysis | ||
| Key findings |
Three RCTs with sensitization outcomes at age 12 months were combined in meta-analysis. Definitions of sensitization were inconsistent. Overall risk of bias of the three RCTs was low to moderate. One RCT was rated high risk of bias for incomplete outcome data reporting and one was rated high risk of bias for selective reporting.
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | N | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | N | |
| Likelihood of publication bias assessed? | Y (the authors noted that the risk of publication bias cannot be excluded because only published studies were included in meta-analysis) | |
| Conflict of interest (COI) stated? | Y/N (COI of the systematic review authors was provided but not provided for included studies) | |
| Author, year | Boyle et al., 2016 | |
| Aims/Key questions | To determine whether feeding infants with hydrolyzed formula reduces their risk of allergic or autoimmune disease. | |
| Study eligibility criteria |
Interventions and comparators:
|
|
| Literature search dates or year range | The Cochrane Library (2013, issue 7), EMBASE (1947 to July 2013), LILACS (1982 to July 2013), Medline (1946 to July 2013), and Web of Science (1970 to July 2013). Searches run on July 25, 2013, and rerun on April 17, 2015. | |
| Number of food allergy studies included | 13 | |
| Synthesis methods | Narrative text and meta-analysis | |
| Key findings | There was no significant difference in risk of “any food allergy” with partially (pooled RR: 1.73; 95% CI: 0.79-; I2=42%) or extensively (pooled RR 0.86; 95% CI: 0.26-2.82; I2=42%) hydrolyzed formula compared with standard formula at age 0-4 years, nor for extensively hydrolyzed formula at age 5-14 years. [Note that number of studies included in each meta-analysis was not reported in text.] No difference was found in food allergy to cow milk, egg, or (partially hydrolyzed formula only) peanut. Direct comparison of the two formulas (egg allergy) and casein versus whey dominant extensively hydrolyzed formula showed no significant difference in risk of food allergy. [Note: no other details reported in text.] There was no significant difference in risk of allergic sensitization to cow milk with partially (pooled RR: 1.30; 95% CI: 0.65-2.60; I2=0%; seven studies) or extensively (pooled RR: 0.77; 95% CI: 0.09-6.73; I2=77%; three studies) hydrolyzed formula, and no significant difference between groups for risk of allergic sensitization to “any allergen” or raised total IgE level. The strength of evidence was graded as moderate for partially hydrolyzed formula, and as very low for exclusively hydrolyzed formula. | |
| Limitations | Many studies of allergic outcomes included in this review had unclear or high risk of bias and evidence of conflict of interest, often because of inadequate methods of randomization and treatment allocation (selection bias) and support of the study or investigators from manufacturers of hydrolyzed formula. In many cases study participants were infants with early full formula feeding, so findings might not be applicable to populations with more typical feeding patterns. | |
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y – Appendix 1 | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Partially – List of excluded studies was not reported | |
| Characteristics of included provided? | Y – Characteristics of included studies are summarized in tables A and B in appendix 3. | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y – GRADE approach | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | Y | |
| Conflict of interest stated? | Y | |
| Author, year | Cuello-Carcia et al., 2016 | |
| Aims/Key questions | To provide evidence-based recommendations about the use of prebiotic supplements for the primary prevention of allergies. | |
| Study eligibility criteria |
According to the evidence profiles table, the study eligibility criteria can be assumed to be:
|
|
| Literature search dates or year range | Up to January 2015, with an update on July 29, 2015 | |
| Number of food allergy studies included | 1 | |
| Synthesis methods | GRADE approach | |
| Key findings | Outcome | Without prebiotics (per 1,000) | With prebiotics (per 1,000) | Difference (95% CI) (per 1,000) | Relative effect (95% CI) | Certainty of the evidence (GRADE) |
| Food allergy | 170 | 48 (14 to 170) | 122 fewer (0 to 156 fewer) | RR 0.28 (0.08 to 1.00) | VERY LOW |
| Conclusions and research needs | The guideline panel determined that there is a low certainty of a net benefit from using prebiotics in infants. Based on the body of available evidence, it is likely that prebiotic supplementation in infants reduces the risk of developing recurrent wheezing and possibly also the development of food allergy. There is very low certainty prebiotics have an effect on other outcomes. However, because of low certainty of evidence or no published information about other outcomes, the fact that the authors did not find evidence of an effect on these outcomes does not imply that such an effect does not exist. | |
| Limitations | This publication is a guideline paper. Although the guideline appears to be based on a systematic review, the methods of systematic review were not fully reported in this publication. No other source or citation to the systematic review was found. | |
| AMSTAR rating | ||
| An a priori design? | Yes | |
| Duplicate study selection and data extraction? | Not reported | |
| Comprehensive literature search? | Yes | |
| Status of the publication as an inclusion criterion? | Not reported | |
| List of studies (included and excluded) provided? | Partially – Excluded studies were not reported | |
| Characteristics of included provided? | Yes | |
| Scientific quality of the included studies assessed and reported? | Yes | |
| Scientific quality used in formulating conclusions? | Yes | |
| Methods used to combine the findings appropriate? | Yes | |
| Likelihood of publication bias assessed? | No | |
| Conflict of interest stated? | Yes | |
| Author, year | Newberry et al., 2016 | |
| Aims/Key questions | To update a prior systematic review on the effects of omega-3 fatty acids (n-3 fatty acids) on maternal and child health and to assess the evidence for their effects on, and associations with, additional outcomes. | |
Key Question 2: Fetal/childhood exposures – What is the influence of maternal intakes of n-3 fatty acids or the n-3 fatty acid content of maternal breast milk (with or without knowledge of maternal intake of n-3 fatty acids) or n-3 fatty acid-supplemented infant formula or intakes of n-3 fatty acids from sources other than maternal breast milk or supplemented infant formula on the following outcomes in term or preterm human infants?
|
||
| Study eligibility criteria |
Populations of interest:
|
|
| Interventions of interest: | ||
|
||
| Literature search dates or year range | Update searches were from the year 2000. For the newly added topics (e.g., allergies), the authors “reference mined” articles that they identified to determine whether any studies conducted and published before 2000 should be obtained and included. | |
| Number of food allergy studies included | 3 RCTs | |
| Synthesis methods | Narrative text and meta-analysis | |
| Key findings |
All three RCTs recruited pregnant women whose infants were at high risk of atopy (e.g., parent diagnosis of allergy, or sibling has diagnosed or suspected allergy).
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y/N (The search strategy was not designed to specifically for food allergies outcomes because “food allergies” were not one of the pre-specified outcomes of interest) | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | N (only published studies were included) | |
| Conflict of interest stated? | Y | |
| Author, year | Cuello-Carcia et al., 2015 | |
| Aims/Key questions |
To synthesize the evidence supporting use of probiotics to prevent allergies and inform World Allergy Organization guidelines on probiotic use. Three key questions of this systematic review are:
|
|
| Study eligibility criteria |
|
|
| Literature search dates or year range | From inception to December 2014 | |
| Number of food allergy studies included | 6 RCTs [Note: some RCTs contributed data for more than one meta-analysis.] | |
| Synthesis methods | Narrative text, meta-analysis, and GRADE | |
| Key findings |
|
|
| Outcomes | No. of participants (studies) | Strengtd of evidence (GRADE) | Relative effect, RR (95% CI) | Anticipated absolute effects | ||
| Risk witd no probiotics | Risk difference with probiotics | |||||
| Probiotics versus no probiotics in pregnant women for prevention of allergy in their children (indirect evidence) | ||||||
| Food allergy, follow-up: range, 12-24 months | 355 (3 RCTs) | •ooo Very low | 1.08 (0.73-1.59) | Study popuation | ||
| Note: 1.49 (0.58, 3.81) in the forest plot | 39 per 1,000 | 3 more per 1,000 (11 fewer to 23 more) | ||||
| Probiotics compared with no probiotics in breastfeeding women to prevent allergies in their children (indirect) | ||||||
| Food allergy, follow-up: range, 12-24 months | 167 (2 RCTs) | •ooo Very low | 1.7 (0.58-4.96) | Study population | ||
| 59 per 1,000 | 41 more per 1,000 (25 fewer to 233 more) | |||||
| Probiotics compared with no probiotics in infants to prevent allergies (direct) | ||||||
| Food allergy, follow-up: range, 6-24 months | 349 (3 RCTs) | •ooo Very low | 0.88 (0.55-1.43) | Study population | ||
| 167 per 1,000 | 20 fewer per 1,000 (75 fewer to 72 more) | |||||
| Outcomes | No. of participants (studies) | Strengtd of evidence (GRADE) | Relative effect, RR (95% CI) | Anticipated absolute effects | |
| Risk witd no probiotics | Risk difference with probiotics | ||||
| Probiotics compared with no probiotics in infants to prevent allergies (indirect) | |||||
| Food allergy, follow-up: range, 6-24 months | 295 (2 RCTs) | •ooo Very low | 1 (0.25-3.91) | Study population | |
| 27 per 1,000 | 0 fewer per 1,000 (20 fewer to 79 more) | ||||
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y – Table E3 and online repository | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | N | |
| Conflict of interest stated? | Y | |
| Author, year | Gunaratne et al., 2015 | |
| Aims/Key questions | To assess the effect of n-3 LCPUFA supplementation in pregnant and/or breastfeeding women on allergy outcomes (food allergy, atopic dermatitis [eczema], allergic rhinitis [hay fever] and asthma/wheeze) in their children. | |
| Study eligibility criteria |
Inclusion criteria:
|
|
| Literature search dates or year range | Inception to August 2014 | |
| Number of food allergy studies included | Five | |
| Synthesis methods | Narrative text and meta-analysis | |
| Key findings |
Three of the five trials had high risk of bias for incomplete outcome data (attrition bias) and/or selective reporting bias.
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | Y | |
| Conflict of interest stated? | Y | |
| Author, year | de Silva et al., 2014 | |
| Aims/Key questions | This systematic review is one of the series of systematic reviews for developing EAACI Guidelines for Food Allergy and Anaphylaxis. This systematic review examined ways to prevent the development of food allergy in children and adults. | |
| Study eligibility criteria |
|
|
| Literature search dates or year range | The following databases were searched: Cochrane Library, MEDLINE, Embase, CINAHL, ISI Web of Science, TRIP Database, and Clinicaltrials.gov from inception to September 30, 2012 | |
| Number of food allergy studies included | 74 | |
| Synthesis methods | Narrative synthesis and summary tables | |
| Key findings |
Table 1 summarized key evidence about prevention strategies:
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y (protocol published elsewhere) | |
| Status of the publication as an inclusion criterion? | Y (protocol published elsewhere) | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | N | |
| Conflict of interest stated? | Y | |
| Author, year | Kong et al., 2014 | |
| Aims/Key questions | To investigate the preventive effect of probiotics on pediatric food allergy. | |
| Study eligibility criteria |
|
|
| Literature search dates or year range | Last search conducted September 30, 2013 | |
| Number of food allergy studies included | 10 | |
| Synthesis methods | Meta-analysis | |
| Key findings |
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | N – Only one database used | |
| Status of the publication as an inclusion criterion? | N | |
| List of studies (included and excluded) provided? | N – Excluded studies not provided | |
| Characteristics of included provided? | N | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | Y | |
| Conflict of interest stated? | N – COI not provided for either the review of the included studies | |
| Author, year | Marrs et al., 2013 | |
| Aims/Key Questions | To systematically review the evidence on the associations between microbial exposure and food allergies. | |
| Study eligibility criteria |
Inclusion criteria:
|
|
| Literature search dates or year range | Medline from inception to July 2012 | |
| Number of food allergy studies included | 46 | |
| Synthesis methods |
|
|
| Key findings |
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Not reported | |
| Comprehensive literature search? | N (Medline only) | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | No but available on request | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | N | |
| Conflict of interest stated? | Y/N – COI of the systematic review authors were reported but COI not provided for the included studies | |
| Author, year | Kramer and Kakuma, 2012 | |
| Aims/Key questions | To assess the effects of prescribing an antigen avoidance diet during pregnancy or lactation, or both, on maternal and infant nutrition and on the prevention or treatment of atopic disease in the child; positive SPTs to dietary antigens; and cord blood levels of IgE (a predictor of subsequent atopic disease). | |
| Study eligibility criteria |
Inclusion criteria:
|
|
| Literature search dates or year range | Unspecified; search conducted on July 6, 2012 | |
| Number of food allergy studies included | Three (based on SPT results) | |
| Synthesis methods | Summary tables and meta-analysis | |
| Key findings |
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | Y | |
| Conflict of interest stated? | Y/N (funding sources of included studies were not reported but the systematic review authors reported no conflict of interest) | |
| Author, year | Fisher et al., 2011 | |
| Aims/Key questions | To determine whether specific oral tolerance induction is more effective than avoidance in inducing tolerance in children ages 0 to 18 years who have IgE-mediated food allergy. | |
| Study eligibility criteria |
Inclusion criteria:
|
|
| Literature search dates or year range | 1950 to July 2009 | |
| Number of food allergy studies included | Three | |
| Synthesis methods | Summary tables and meta-analysis | |
| Key findings |
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | N (only one author conducted the search and one author reviewed the studies) | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | N | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | N | |
| Conflict of interest stated? | Y/N (COI of the systematic review authors was provided but not provided for included studies) | |
| Author, year | Klemens et al., 2011 | |
| Aims/Key questions | To determine if n-3 PUFA supplementation during pregnancy and lactation reduces risk for childhood allergic disease. | |
| Study eligibility criteria |
Inclusion criteria:
|
|
| Literature search dates or year range | 1950 to October 2010 | |
| Number of food allergy studies included | Three | |
| Synthesis methods | Summary tables, meta-analysis and narrative synthesis | |
| Key findings |
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | N | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combine the findings appropriate? | Y | |
| Likelihood of publication bias assessed? | Y | |
| Conflict of interest stated? | Y/N (COI of the systematic review authors was provided but not provided for included studies) | |
| Author, year | Osborn and Sinn, 2006 | |
| Aims/Key questions |
|
|
| Study eligibility criteria |
Inclusion criteria:
|
|
| Literature search dates or year range | Inception to March 2006 | |
| Number of food allergy studies included | Five | |
| Synthesis methods | Meta-analysis, summary table, narrative text | |
| Key findings |
For Comparison 01 Early short-term feeding: Hydrolyzed formula versus human milk feeding among low-risk infants:
|
|
For Comparison 03 Early short-term feeding: Hydrolyzed formula versus cow milk formula:
|
||
For Comparison 04 Prolonged feeding: Hydrolyzed formula versus cow milk formula:
|
||
For Comparison 07 Prolonged feeding: Extensively hydrolyzed formula versus cow milk formula:
|
||
For Comparison 08 Prolonged feeding: Partially hydrolyzed formula versus cow milk formula:
|
||
For Comparison 09 Prolonged feeding: Extensively hydrolyzed formula versus partially hydrolyzed formula:
|
||
| Key findings (continued) |
For Comparison 11 Prolonged feeding: Hydrolyzed formula versus cow milk formula: Allergy/intolerance confirmed by test:
|
|
For Comparison 14 Prolonged feeding: Partially hydrolyzed whey formula versus cow milk formula:
|
||
For Comparison 15 Prolonged feeding: Partially hydrolyzed casein containing formula versus cow milk formula:
|
||
For Comparison 17 Prolonged feeding: Extensively hydrolyzed casein containing formula versus cow milk formula:
|
||
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | N | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | Y | |
| Methods used to combie the findings appropriate? | N (many “meta-analysis” had one study) | |
| Likelihood of publication bias assessed? | N | |
| Conflict of interest stated? | Y | |
| Author, year | Kramer and Kakuma, 2004 | |
| Aims/Key questions | To assess the effects on child health, growth, and development, and on maternal health, of exclusive breastfeeding for 6 months versus exclusive breastfeeding for 3 to 4 months with mixed breastfeeding (introduction of complementary liquid or solid foods with continued breastfeeding) thereafter through 6 months. | |
| Study eligibility criteria |
Inclusion criteria:
|
|
| Study eligibility criteria (continued) |
|
|
| Literature search dates or year range | Inception to June 15, 2011 | |
| Number of food allergy studies included | One cohort study | |
| Synthesis methods | Narrative synthesis; meta-analysis (N/A for food allergy outcome because only one study was included) | |
| Key findings |
|
|
| Limitations |
|
|
| AMSTAR rating | ||
| An a priori design? | Y | |
| Duplicate study selection and data extraction? | Y | |
| Comprehensive literature search? | Y | |
| Status of the publication as an inclusion criterion? | Y | |
| List of studies (included and excluded) provided? | Y | |
| Characteristics of included provided? | Y | |
| Scientific quality of the included studies assessed and reported? | Y | |
| Scientific quality used in formulating conclusions? | N | |
| Methods used to combine the findings appropriate? | N (many “meta-analysis” only has 1 study) | |
| Likelihood of publication bias assessed? | N | |
| Conflict of interest stated? | N | |
NOTE: ALA = alpha-linolenic acid; CDC = Centers for Disease Control and Prevention; CI = confidence interval; DBPCOFC = double-blind, placebo-controlled oral food challenge; DHA = docosahexaenoic acid; DPA = docosapentaenoic acid; EAACI = European Academy of Allergy & Clinical Immunology; EPA = eicosapentaenoic acid; IgE = immunoglobulin E; IU = international units; LCPUFA, long-chain polyunsaturated fatty acid; N/A = not available; OR = odds ratio; PUFA = polyunsaturated fatty acid; RAST = radioallergosorbent; RCT = randomized controlled trial; RR = relative risk; SDA = stearidonic acid; SPT = skin prick test; tIgE = total IgE; WHO = World Health Organization.
REFERENCES
Allen, K. J., J. J. Koplin, A. L. Ponsonby, L. C. Gurrin, M. Wake, P. Vuillermin, P. Martin, M. Matheson, A. Lowe, M. Robinson, D. Tey, N. J. Osborne, T. Dang, H. T. Tina Tan, L. Thiele, D. Anderson, H. Czech, J. Sanjeevan, G. Zurzolo, T. Dwyer, M. L. Tang, D. Hill, and S. C. Dharmage. 2013. Vitamin D insufficiency is associated with challenge-proven food allergy in infants. J Allergy Clin Immunol 131(4):1109-1116, 1116.
Baek, J. H., Y. H. Shin, I. H. Chung, H. J. Kim, E. G. Yoo, J. W. Yoon, H. M. Jee, Y. E. Chang, and M. Y. Han. 2014. The link between serum vitamin D level, sensitization to food allergens, and the severity of atopic dermatitis in infancy. J Pediatr 165(4):849-854.
Best, K. P., M. Gold, D. Kennedy, J. Martin, and M. Makrides. 2016. Omega-3 long-chain PUFA intake during pregnancy and allergic disease outcomes in the offspring: A systematic review and meta-analysis of observational studies and randomized controlled trials. Am J Clin Nutr 103(1):128-143.
Boyle, R. J., D. Ierodiakonou, T. Khan, J. Chivinge, Z. Robinson, N. Geoghegan, K. Jarrold, T. Afxentiou, T. Reeves, S. Cunha, M. Trivella, V. Garcia-Larsen, and J. Leonardi-Bee. 2016. Hydrolysed formula and risk of allergic or autoimmune disease: Systematic review and meta-analysis. BMJ 352:i974.
Bunyavanich, S., S. L. Rifas-Shiman, T. A. Platts-Mills, L. Workman, J. E. Sordillo, C. A. Camargo, Jr., M. W. Gillman, D. R. Gold, and A. A. Litonjua. 2014. Peanut, milk, and wheat intake during pregnancy is associated with reduced allergy and asthma in children. J Allergy Clin Immunol 133(5):1373-1382.
Cuello-Garcia, C. A., J. L. Brozek, A. Fiocchi, R. Pawankar, J. J. Yepes-Nunez, L. Terracciano, S. Gandhi, A. Agarwal, Y. Zhang, and H. J. Schunemann. 2015. Probiotics for the prevention of allergy: A systematic review and meta-analysis of randomized controlled trials. J Allergy Clin Immunol 136(4):952-961.
Cuello-Garcia, C. A., A. Fiocchi, R. Pawankar, J. J. Yepes-Nunez, G. P. Morgano, Y. Zhang, K. Ahn, S. Al-Hammadi, A. Agarwal, S. Gandhi, K. Beyer, W. Burks, G. W. Canonica, M. Ebisawa, R. Kamenwa, B. W. Lee, H. Li, S. Prescott, J. J. Riva, L. Rosenwasser, H. Sampson, M. Spigler, L. Terracciano, A. Vereda, S. Waserman, H. J. Schunemann, and J. L. Brozek. 2016. World Allergy Organization-McMaster University Guidelines for Allergic Disease Prevention (GLAD-P): Prebiotics. World Allergy Organ J 9:10.
de Silva, D., M. Geromi, S. Halken, A. Host, S. S. Panesar, A. Muraro, T. Werfel, K. Hoffmann-Sommergruber, G. Roberts, V. Cardona, A. E. Dubois, L. K. Poulsen, R. Van Ree, B. Vlieg-Boerstra, I. Agache, K. Grimshaw, L. O’Mahony, C. Venter, S. H. Arshad, and A. Sheikh. 2014. Primary prevention of food allergy in children and adults: Systematic review. Allergy 69(5):581-589.
Depner, M., M. J. Ege, J. Genuneit, J. Pekkanen, M. Roponen, M. R. Hirvonen, J. C. Dalphin, V. Kaulek, S. Krauss-Etschmann, J. Riedler, C. Braun-Fahrlander, C. Roduit, R. Lauener, P. I. Pfefferle, J. Weber, and E. von Mutius. 2013. Atopic sensitization in the first year of life. J Allergy Clin Immunol 131(3):781-788.
Dowhower Karpa, K., I. M. Paul, J. A. Leckie, S. Shung, N. Carkaci-Salli, K. E. Vrana, D. Mauger, T. Fausnight, and J. Poger. 2012. A retrospective chart review to identify perinatal factors associated with food allergies. Nutr J 11:87.
Du Toit, G., G. Roberts, P. H. Sayre, H. T. Bahnson, S. Radulovic, A. F. Santos, H. A. Brough, D. Phippard, M. Basting, M. Feeney, V. Turcanu, M. L. Sever, M. Gomez Lorenzo, M. Plaut, and G. Lack. 2015. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med 372(9):803-813.
Du Toit, G., P. H. Sayre, G. Roberts, M. L. Sever, K. Lawson, H. T. Bahnson, H. A. Brough, A. F. Santos, K. M. Harris, S. Radulovic, M. Basting, V. Turcanu, M. Plaut, G. Lack. 2016. Effect of avoidance on peanut allergy after early peanut consumption. N Engl J Med 374:1435-1443.
Dunstan, J., C. West, S. McCarthy, J. Metcalfe, S. Meldrum, W. Oddy, M. Tulic, N. D’Vaz, and S. Prescott. 2012. The relationship between maternal folate status in pregnancy, cord blood folate levels, and allergic outcomes in early childhood. Allergy 67(1):50-57.
Fisher, H. R., G. du Toit, and G. Lack. 2011. Specific oral tolerance induction in food allergic children: Is oral desensitisation more effective than allergen avoidance?: A meta-analysis of published RCTs. Arch Dis Child 96(3):259-264.
Frazier, A. L., C. A. Camargo, Jr., S. Malspeis, W. C. Willett, and M. C. Young. 2014. Prospective study of peripregnancy consumption of peanuts or tree nuts by mothers and the risk of peanut or tree nut allergy in their offspring. JAMA Pediatr 168(2):156-162.
Goldberg, M., E. Eisenberg, A. Elizur, N. Rajuan, M. Rachmiel, A. Cohen, G. Zadik-Mnuhin, and Y. Katz. 2013. Role of parental atopy in cow’s milk allergy: A population-based study. Ann Allergy Asthma Immunol 110(4):279-283.
Grimshaw, K. E., J. Maskell, E. M. Oliver, R. C. Morris, K. D. Foote, E. N. Mills, G. Roberts, and B. M. Margetts. 2013. Introduction of complementary foods and the relationship to food allergy. Pediatrics 132(6):e1529-e1538.
Grimshaw, K. E. C., J. Maskell, E. M. Oliver, R. C. G. Morris, K. D. Foote, E. N. C. Mills, B. M. Margetts, and G. Roberts. 2014. Diet and food allergy development during infancy: Birth cohort study findings using prospective food diary data. Journal of Allergy and Clinical Immunology 133(2):511-519.
Gunaratne, A. W., M. Makrides, and C. T. Collins. 2015. Maternal prenatal and/or postnatal n-3 long chain polyunsaturated fatty acids (LCPUFA) supplementation for preventing allergies in early childhood. Cochrane Database Syst Rev 7:CD010085.
Ivakhnenko, O., and S. Nyankovskyy. 2013. Effect of the specific infant formula mixture of oligosaccharides on local immunity and development of allergic and infectious disease in young children: Randomized study. Pediatria Polska 88(5):398-404.
Jones, A. P., D. Palmer, G. Zhang, and S. L. Prescott. 2012. Cord blood 25-hydroxyvitamin D3 and allergic disease during infancy. Pediatrics 130(5):e1128-e1135.
Katz, Y., N. Rajuan, M. R. Goldberg, E. Eisenberg, E. Heyman, A. Cohen, and M. Leshno. 2010. Early exposure to cow’s milk protein is protective against IgE-mediated cow’s milk protein allergy. J Allergy Clin Immunol 126(1):77-82.
Klemens, C. M., D. R. Berman, and E. L. Mozurkewich. 2011. The effect of perinatal omega-3 fatty acid supplementation on inflammatory markers and allergic diseases: A systematic review. BJOG 118(8):916-925.
Kong, X. Y., Y. Yang, J. Guan, and R. Z. Wang. 2014. Probiotics’ preventive effect on pediatric food allergy: A meta-analysis of randomized controlled trials. Chin Med Sci J 29(3):144-147.
Koplin, J. J., S. C. Dharmage, A. L. Ponsonby, M. L. Tang, A. J. Lowe, L. C. Gurrin, N. J. Osborne, P. E. Martin, M. N. Robinson, M. Wake, D. J. Hill, and K. J. Allen. 2012. Environmental and demographic risk factors for egg allergy in a population-based study of infants. Allergy 67(11):1415-1422.
Koplin, J. J., N. H. Suaini, P. Vuillermin, J. A. Ellis, M. Panjari, A. L. Ponsonby, R. L. Peters, M. C. Matheson, D. Martino, T. Dang, N. J. Osborne, P. Martin, A. Lowe, L. C. Gurrin, M. L. Tang, M. Wake, T. Dwyer, J. Hopper, S. C. Dharmage, K. J. Allen. 2016. Polymorphisms affecting vitamin D-binding protein modify the relationship between serum vitamin D (25[OH]D3) and food allergy. J Allergy Clin Immunol 137(2):500-506.
Kramer, M. S., and R. Kakuma. 2004. The optimal duration of exclusive breastfeeding: A systematic review. Adv Exp Med Biol 554:63-77.
Kramer, M. S., and R. Kakuma. 2012. Maternal dietary antigen avoidance during pregnancy or lactation, or both, for preventing or treating atopic disease in the child. Cochrane Database Syst Rev 9:CD000133.
Liao, S. L., S. H. Lai, K. W. Yeh, Y. L. Huang, T. C. Yao, M. H. Tsai, M. C. Hua, and J. L. Huang. 2014. Exclusive breastfeeding is associated with reduced cow’s milk sensitization in early childhood. Pediatr Allergy Immunol 25(5):456-461.
Liu, X., G. Wang, X. Hong, D. Wang, H. J. Tsai, S. Zhang, L. Arguelles, R. Kumar, H. Wang, R. Liu, Y. Zhou, C. Pearson, K. Ortiz, R. Schleimer, P. G. Holt, J. Pongracic, H. E. Price, C. Langman, and X. Wang. 2011. Gene-vitamin D interactions on food sensitization: A prospective birth cohort study. Allergy 66(11):1442-1448.
Liu, X., L. Arguelles, Y. Zhou, G. Wang, Q. Chen, H. J. Tsai, X. Hong, R. Liu, H. E. Price, C. Pearson, S. Apollon, N. Cruz, R. Schleimer, C. B. Langman, J. A. Pongracic, and X. Wang. 2013. Longitudinal trajectory of vitamin D status from birth to early childhood in the development of food sensitization. Pediatr Res 74(3):321-326.
Loo, E. X., G. V. Llanora, Q. Lu, M. M. Aw, B. W. Lee, and L. P. Shek. 2014. Supplementation with probiotics in the first 6 months of life did not protect against eczema and allergy in at-risk Asian infants: A 5-year follow-up. Int Arch Allergy Immunol 163(1):25-28.
Lowe, A. J., C. S. Hosking, C. M. Bennett, K. J. Allen, C. Axelrad, J. B. Carlin, M. J. Abramson, S. C. Dharmage, and D. J. Hill. 2011. Effect of a partially hydrolyzed whey infant formula at weaning on risk of allergic disease in high-risk children: A randomized controlled trial. J Allergy Clin Immunol 128(2):360-365.
Luccioli, S., Y. Zhang, L. Verrill, M. Ramos-Valle, and E. Kwegyir-Afful. 2014. Infant feeding practices and reported food allergies at 6 years of age. Pediatrics 134(Suppl 1):S21-S28.
Magdelijns, F. J., M. Mommers, J. Penders, L. Smits, and C. Thijs. 2011. Folic acid use in pregnancy and the development of atopy, asthma, and lung function in childhood. Pediatrics 128(1):e135-e144.
Mailhol, C., F. Giordano-Labadie, V. Lauwers-Cances, A. Ammoury, C. Paul, and F. Rance. 2014. Point prevalence and risk factors for food allergy in a cohort of 386 children with atopic dermatitis attending a multidisciplinary dermatology/paediatric allergy clinic. Eur J Dermatol 24(1):63-69.
Marrs, T., K. D. Bruce, K. Logan, D. W. Rivett, M. R. Perkin, G. Lack, and C. Flohr. 2013. Is there an association between microbial exposure and food allergy? A systematic review. Pediatr Allergy Immunol 24(4):311-320.
Martin, P. E., J. K. Eckert, J. J. Koplin, A. J. Lowe, L. C. Gurrin, S. C. Dharmage, P. Vuillermin, M. L. Tang, A. L. Ponsonby, M. Matheson, D. J. Hill, and K. J. Allen. 2015. Which infants with eczema are at risk of food allergy? Results from a population-based cohort. Clin Exp Allergy 45(1):255-264.
McGowan, E. C., G. R. Bloomberg, P. J. Gergen, C. M. Visness, K. F. Jaffee, M. Sandel, G. O’Connor, M. Kattan, J. Gern, and R. A. Wood. 2015. Influence of early-life exposures on food sensitization and food allergy in an inner-city birth cohort. J Allergy Clin Immunol 135(1):171-178.
Metsala, J., A. Lundqvist, L. J. Virta, M. Kaila, M. Gissler, and S. M. Virtanen. 2013. Mother’s and offspring’s use of antibiotics and infant allergy to cow’s milk. Epidemiology 24(2):303-309.
Newberry, S. J., M. Chung, M. Booth, M. A. Maglione, A. M. Tang, C. E. O’Hanlon, D. D. Wang, A. Okunogbe, C. Huang, A. Motala, M. Timmer, W. Dudley, R. Shanman, T. R. Coker, and P. G. Shekelle. 2016. Omega-3 fatty acids and maternal and child health: An updated systematic review. Evidence Report/Technology Assessment No. 224. AHRQ Publication No. 16-E003-EF. Rockville, MD: Agency for Healthcare Research and Quality.
Norizoe, C., N. Akiyama, T. Segawa, H. Tachimoto, H. Mezawa, H. Ida, and M. Urashima. 2014. Increased food allergy and vitamin D: Randomized, double-blind, placebo-controlled trial. Pediatr Int 56(1):6-12.
Okupa, A. Y., R. F. Lemanske, Jr., D. J. Jackson, M. D. Evans, R. A. Wood, and E. C. Matsui. 2013. Early-life folate levels are associated with incident allergic sensitization. J Allergy Clin Immunol 131(1):226-228.
Osborn, D. A., and J. Sinn. 2006. Formulas containing hydrolysed protein for prevention of allergy and food intolerance in infants. Cochrane Database Syst Rev(4):CD003664.
Palmer, D. J., J. Metcalfe, M. Makrides, M. S. Gold, P. Quinn, C. E. West, R. Loh, and S. L. Prescott. 2013. Early regular egg exposure in infants with eczema: A randomized controlled trial. J Allergy Clin Immunol 132(2):387-392.
Pele, F., E. Bajeux, H. Gendron, C. Monfort, F. Rouget, L. Multigner, J. F. Viel, and S. Cordier. 2013. Maternal fish and shellfish consumption and wheeze, eczema and food allergy at age two: A prospective cohort study in Brittany, France. Environ Health 12:102.
Perkin, M. R., K. Logan, A. Tseng, B. Raji, S. Ayis, J. Peacock, H. Brough, T. Marrs, S. Radulovic, J. Craven, C. Flohr, G. Lack, and EAT Study Team. 2016. Randomized trial of introduction of allergenic foods in breast-fed infants. N Engl J Med 374(18):1733-1743.
Peters, R., K. Allen, S. Dharmage, C. Lodge, J. Koplin, A. Ponsonby, M. Wake, A. Lowe, M. Tang, M. Matheson, and L. Gurrin. 2015. Differential factors associated with challenge-proven food allergy phenotypes in a population cohort of infants: A latent class analysis. Clin Exp Allergy 45(5):953-963.
Pyrhonen, K., S. Nayha, L. Hiltunen, and E. Laara. 2013. Caesarean section and allergic manifestations: Insufficient evidence of association found in population-based study of children aged 1 to 4 years. Acta Paediatr 102(10):982-989.
Roduit, C., R. Frei, M. Depner, B. Schaub, G. Loss, J. Genuneit, P. Pfefferle, A. Hyvarinen, A. M. Karvonen, J. Riedler, J. C. Dalphin, J. Pekkanen, E. von Mutius, C. Braun-Fahrlander, and R. Lauener. 2014. Increased food diversity in the first year of life is inversely associated with allergic diseases. J Allergy Clin Immunol 133(4):1056-1064.
Stelmach, I., M. Bobrowska-Korzeniowska, K. Smejda, P. Majak, J. Jerzynska, W. Stelmach, K. Polanska, W. Sobala, J. Krysicka, and W. Hanke. 2014. Risk factors for the development of atopic dermatitis and early wheeze. Allergy Asthma Proc 35(5):382-389.
von Berg, A., B. Filipiak-Pittroff, U. Kramer, B. Hoffmann, E. Link, C. Beckmann, U. Hoffmann, D. Reinhardt, A. Grubl, J. Heinrich, H. E. Wichmann, C. P. Bauer, S. Koletzko, D. Berdel, and G. I. s. group. 2013. Allergies in high-risk schoolchildren after early intervention with cow’s milk protein hydrolysates: 10-year results from the German Infant Nutritional Intervention (GINI) study. J Allergy Clin Immunol 131(6):1565-1573.
Wawro, N., J. Heinrich, E. Thiering, J. Kratzsch, B. Schaaf, B. Hoffmann, I. Lehmann, C. P. Bauer, S. Koletzko, A. von Berg, D. Berdel, and J. Linseisen. 2014. Serum 25(OH)D concentrations and atopic diseases at age 10: Results from the GINIplus and LISAplus birth cohort studies. BMC Pediatr 14:286.
Weisse, K., S. Winkler, F. Hirche, G. Herberth, D. Hinz, M. Bauer, S. Roder, U. Rolle-Kampczyk, M. von Bergen, S. Olek, U. Sack, T. Richter, U. Diez, M. Borte, G. I. Stangl, and I. Lehmann. 2013. Maternal and newborn vitamin D status and its impact on food allergy development in the German LINA cohort study. Allergy 68(2):220-228.
West, C. E., J. Dunstan, S. McCarthy, J. Metcalfe, N. D’Vaz, S. Meldrum, W. H. Oddy, M. K. Tulic, and S. L. Prescott. 2012. Associations between maternal antioxidant intakes in pregnancy and infant allergic outcomes. Nutrients 4(11):1747-1758.
West, C. E., M. L. Hammarstrom, and O. Hernell. 2013. Probiotics in primary prevention of allergic disease—follow-up at 8-9 years of age. Allergy 68(8):1015-1020.