Summary
Food allergy, an adverse health effect arising from a specific immune response that occurs reproducibly on exposure to a given food, affects the health and quality of life of individuals and their caregivers across a range of dimensions. A food allergy can cause skin, respiratory, and gastrointestinal reactions. The quality of life of individuals with food allergy is diminished as their social interactions and routine life activities are affected. For some individuals, a food allergy can lead to severe allergic reactions and death. Food allergies typically develop within the first year of life but they can also develop later in life. Eight food groups are considered to be major allergens. These are milk, egg, peanut, tree nuts, wheat, soy, fish, and crustacean shellfish.
Questions persist about whether food allergy prevalence has been on the rise within the past two decades and why. The current data do not unequivocally support the occurrence of such a rise. Multiple hypotheses have been generated about potential genetic and environmental factors that lead to food allergies and a potential rise in food allergy cases. Concomitant with a widespread perception of an increase in prevalence, the public and other stakeholders frequently misinterpret a food allergy and its symptoms, how to differentiate a food allergy from other immune and gastrointestinal diseases, and what effective management and prevention approaches to use. For example, lactose intolerance symptoms can be misinterpreted as a food allergy, when in fact their physiological origin and management approaches are vastly different.
Food allergy is a complex disease at the molecular and cellular level and although much research data have accumulated, many fundamental
questions remain. For example, researchers are struggling to identify factors in utero and during the first year of life, such as the timing of introduction of solid foods or breastfeeding duration, that could lead to the onset of allergies. Overall, gaps in knowledge at the mechanistic level represent barriers to developing strategies for disease prevention and management. Not surprisingly then, recommendations by public health authorities or professional associations for preventing or managing a food allergy are limited by the scarce or inconsistent research findings. Although promising therapeutic approaches are being tested, no effective treatments currently exist for patients with food allergies.
In the absence of approved treatments, patients are advised to avoid the allergen, which can be very difficult, especially in some circumstances. For example, under the Food Allergen Labeling and Consumer Protection Act, food allergens must be listed in the ingredient list of a packaged food. Unfortunately, during production or manufacturing, cross-contamination may occur, resulting in the food product having a hidden allergen that does not appear on the label. In addition, food service establishments are not required to list food allergens, so an individual’s safety depends on clear communication and on employees’ knowledge of the allergen content of the food being served and on the establishment’s management practices. Even with the most stringent management practices, accidents, such as cross-contact events, can still occur when people with a food allergy eat outside of the home. Concerted efforts by policy makers, industry leaders, and others are necessary to bring about a safe environment for those with food allergy.
In summary, many stakeholders, including policy makers, the food industry, scientists, clinicians, and especially individuals with food allergy and their caregivers, are concerned about the misunderstandings, and the lack of effective treatment and clear approaches to prevent food allergy. This report collects and evaluates the scientific evidence on the prevalence, origins, diagnosis, prevention, and management of food allergy and makes recommendations to stakeholders to maximize safety and to increase research activities related to food allergy.
THE TASK AND COMMITTEE’S INTERPRETATION
An ad hoc committee of 15 experts was selected to respond to the statement of task (see Box S-1). The plan for the study included an advisory panel made up of nine parents of children with food allergy and one individual with food allergy. This panel was asked to present to the committee at public meetings; their testimonies were invaluable as examples of the challenges and burden of living with food allergies.
Given the misunderstandings related to food allergy, a first assignment
for the committee was to define the types of food allergies to address in this report. Food allergy, as opposed to a food intolerance, which does not have an immunologic component, arises from a specific immune response. Food allergy has two key classifications: immunoglobulin E (IgE)-mediated or non-IgE-mediated. The recommendations in this report focus on IgE-mediated food allergies, which have better defined underlying cellular mechanisms and physiological reactions. Other food-related diseases, such as celiac disease, food intolerances (e.g., lactose intolerance) are not covered. However, other non-IgE-mediated food allergies are mentioned when appropriate, particularly while discussing diagnostic methodologies. With a focus on the United States, many recommendations could apply in other countries.
A DEVELOPMENTAL AND ECOLOGICAL PERSPECTIVE ON FOOD ALLERGY
For every individual, the risks and protections from food allergies vary over the life course, depending on individual genetic factors, biological development, exposures to allergens, and the contexts in which the individual lives (i.e., a developmental perspective). Before birth, a fetus interacts indirectly with systems because influences (e.g., diet) are mediated by maternal biological function. After birth, children continue to develop and they interact directly with numerous new systems, including peers, schools, social media, workplaces, and social contexts. But individuals are influenced by many additional systems beyond their proximal interactions, through cultural practices and governmental or nongovernmental policies or rules. The safety and well-being of individuals with potential food allergies, then, require recognition that risks and protections for public safety are spread across many systems, including food production and distribution systems, health care systems, and education systems (i.e., an ecological perspective). The committee developed a model to depict those important interactions (see Figure S-1).
THE ROADMAP TO SAFETY
In mapping the road to greater public safety regarding food allergy, in addition to the health care system, the committee selected the following settings for their relevance to the task at hand: food establishments, early care and education, schools, higher education, and the travel industry. These settings vary in policies and practices, and many improvements are both feasible and would likely contribute to preventing and managing severe allergic reactions and improving quality of life.
The committee’s roadmap to safety is multifaceted, involving many
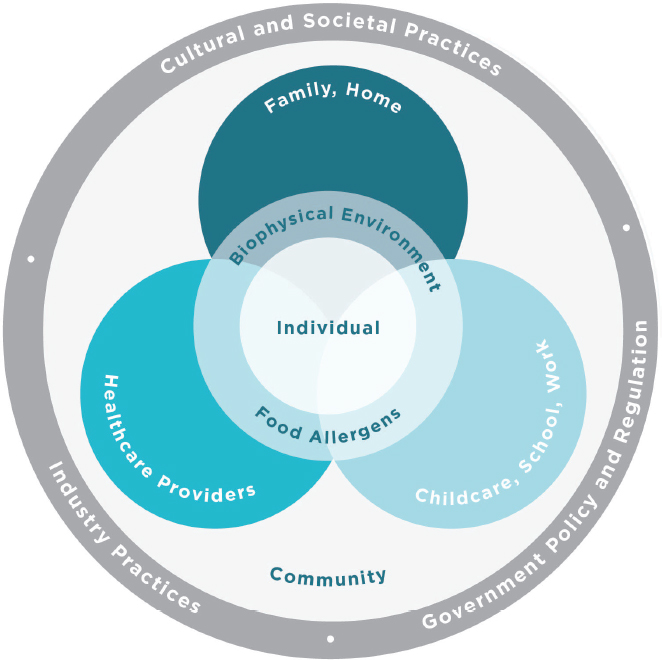
NOTES: Industry practices refers to all the manufacturing processes and allergen control plans followed during food production, distribution, preparation or cooking, and serving. They also refer to mandatory and voluntary labeling of food allergens and to recall procedures followed when a product is contaminated with a food allergen. Cultural and societal practices refer to the particular diets and foods of regions and countries. Biophysical environment refers to the external proximal environment (e.g., air) while Individual refers to all systems internal to a developing human, including genome, epigenome, proteome, metabolome, central nervous system, immune system, microbiomes, and many other self-regulatory systems involved in adaptation and sustaining life. Health care providers include the persons (e.g., physicians, dieticians) and the institutions that protect individual and public health. Child care, school, work includes all proximal settings that interact with an individual at different life stages. Finally, family, home refers to the system of people, relationships, routines, and practices occurring at home. Interactions (e.g., communication, physical contact) occur between and among all those systems and the individual to support (or not) food safety.
stakeholders and the following actions (see Figure S-2): (1) obtain accurate prevalence estimates, (2) use proper diagnostic methods and provide evidence-based health care, (3) identify evidence-based prevention approaches, (4) improve education and training, (5) implement improved policies and practices to prevent the occurrence of severe reactions, and (6) expand research programs. This section summarizes these actions and related recommendations.
Obtain Accurate Prevalence Estimates
To prioritize food allergy as a public health concern and ensure that adequate resources are directed at the issue, the extent of the problem must be defined. No study in the United States has been conducted in a systematic manner, with sufficient sample size, and in various populations to determine the true prevalence of food allergy. Because of the low quality of data, particularly the use of self-reported data instead of the gold standard oral food challenge (OFC)1 method, the true prevalence of food allergy is likely overestimated in most published studies.
The committee recommends that the Centers for Disease Control and Prevention obtain prevalence estimates on food allergy in a systematic and statistically sound manner.
Prevalence should be assessed in a systematic fashion in a sufficiently large population, with consideration given to using stratified sampling for cost-efficiency, with frequency-weighting used to obtain population-wide estimates. Prevalence estimates should be conducted in both children and adults and in groups defined by race, ethnicity, and socioeconomic status to determine differences in diagnosis and prevalence within these subgroups. To support population risk assessments, the committee also recommends that the dietary intake history of those reporting food allergy be compared to those who do not, particularly for the specific foods of interest.
Although a new study design (or the use of other data surveillance systems) is possible, the National Health and Nutrition Examination Survey (NHANES) is a feasible option to systematically examine the prevalence of food allergy by collecting data on
__________________
1 Oral food challenge is a feeding test that involves gradual, medically supervised ingestion of increasingly larger doses of the food being tested as a possible food allergen. The test is positive when the individual experiences food allergy symptoms, such as skin, respiratory, and gastrointestinal reactions.
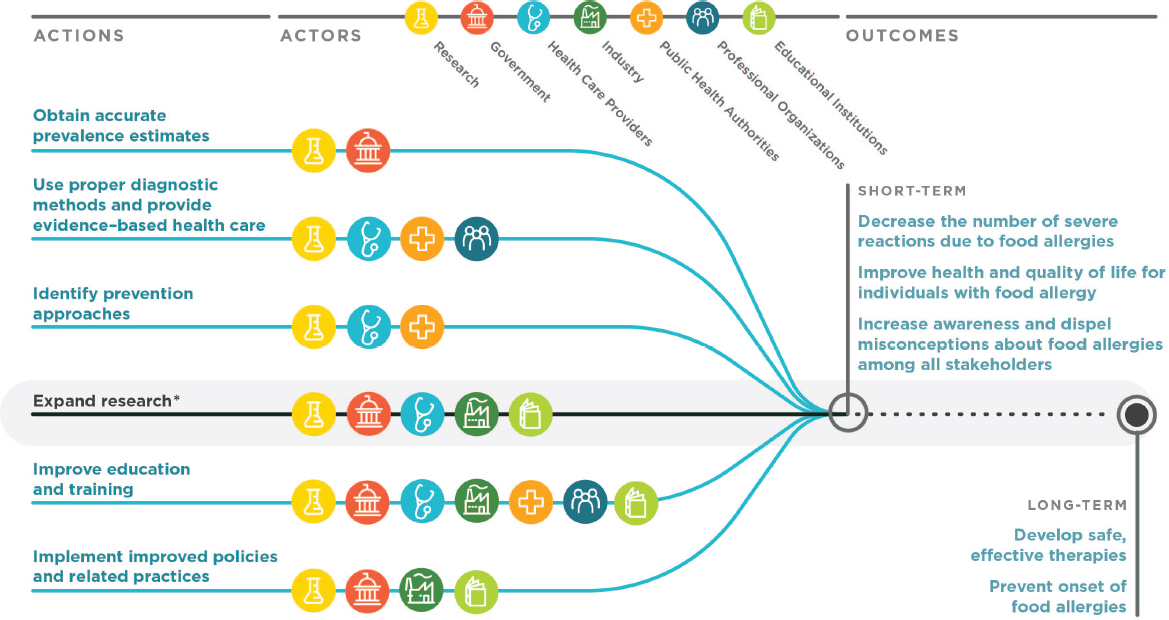
* Research is needed to achieve all other actions and to reach the short- and long-term goals (see Chapter 9 for all specific areas of research). The actors represent the primary stakeholders that will be involved in implementing the actions.
self-reported food allergies, food-specific immunoglobulin E concentrations, food-specific skin prick test results, and oral food challenge results.2 Specific suggestions for use of NHANES (or other data surveillance systems), such as oversampling of young children (<6 years) as an important group, are included in Chapter 3.
Use Proper Diagnostic Methods and Provide Evidence-Based Health Care
No simple diagnostic tests exist for food allergy, and the selection and interpretation of tests depend upon the nature of the disorder and the individual medical history. The OFC carries risk and expense and is underused. The medical history and other test results (e.g., skin prick test) can suggest the likelihood of a food allergy, but in some cases an OFC is needed to confirm the presence of a clinical disease.
The committee recommends that physicians use evidence-based, standardized procedures as the basis for food allergy diagnosis and avoid nonstandardized and unproven procedures (e.g., applied kinesiology, immunoglobulin G panels, electrodermal testing). When food allergy is suspected, a patient should be evaluated by a physician who has the training and experience to select and interpret appropriate diagnostic tests.
Although this process often may include an initial evaluation by a primary physician, it is important that those with suspected food allergy be diagnosed appropriately, which is likely to involve referral to or consultation with a physician specialist who can diagnose, comprehensively evaluate, and manage the food allergy.
Food allergy evaluation procedures include a medical history and physical examination, and also may include a food-specific skin prick test, food-specific serum immunoglobulin E test, diagnostic food elimination diet, and OFC. Selection of the specific tests needs to be individualized based on the medical history of each patient. Health care providers trained in food allergy, leaders of health care facilities, and health care payor groups can facilitate the appropriate use of OFCs, including personnel, facilities, and safety guards, so that physicians are not deterred from performing
__________________
2 The gold standard OFC is an expensive method and must be administered in a clinic and under supervision of a trained physician. The testing sequence, therefore, is meant to lead to a population sample that is enriched with individuals reporting food allergy and that minimizes cost and effort.
the types of diagnostic testing that are appropriate for the patient’s diagnosis and care.
Identify Evidence-Based Prevention Approaches
Although many factors have been postulated to contribute to the onset of food allergy, strong evidence is lacking about any association, mainly due to methodological limitations and variations in study designs. The strongest data derive from recent studies supporting the dual allergen exposure hypothesis, which proposes that a food allergy may occur through exposure to low doses of allergen through damaged skin (such as in eczema) followed by oral exposure to these allergens through consumption early in infancy. The hypothesis proposes that the practice of delaying the introduction of allergens may have contributed to the presumptive rise in food allergy prevalence.
The committee recommends that public health authorities and clinical practice guidelines include consistent, clear, and evidence-based advice for families and health care providers, including dietitians, about the potential benefits of introducing allergenic foods (e.g., peanut products, egg, dairy, and wheat) in the first year of life to infants, when an infant is developmentally ready (around 6 months of age), but not before 4 months of age, particularly to those at high risk of allergy. Guidelines also should include information about the circumstances in which health care providers should advise their patients about the safest way to introduce in their diet peanut products (and/or other foods, as determined by the results of ongoing research).
Improve Education and Training
Public Health Authorities, Health Care Providers, and Their Patients and Caregivers
The committee generally supports current guidelines and U.S. practice parameters for food allergy management and emphasizes those areas where improvements would lead to significant changes in the quality of life of patients and their caregivers, such as training and education.
The committee recommends that the Centers for Disease Control and Prevention work with other public health authorities to plan and initiate a public health campaign for the general public, individuals with food allergy, and all relevant stakeholders to increase
awareness and empathy as well as to dispel misconceptions about food allergy and its management.
For example, as part of that campaign and taking advantage of the popularity of digital media among the public, particularly children and adolescents, public health authorities could develop effective media engagement programs. To plan for this campaign and develop media programs, public health authorities could conduct formative research with all potential audiences.
The committee recommends that public health authorities, such as the National Institutes of Health and the World Health Organization, and professional organizations, such as the American Academy of Pediatrics; the American Academy of Allergy, Asthma & Immunology; American Academy of Family Physicians; and the Academy of Nutrition and Dietetics, regularly update guidelines on diagnosis, prevention, and management of food allergy based on strong scientific evidence, as emerging scientific data become available.
For example, current evidence is insufficient to associate any of the following behaviors with prevention of food allergy: food allergen avoidance diets for pregnant or lactating women, prolonged allergen avoidance in infancy, vaginal delivery, breastfeeding, infant formulas containing extensively or partially hydrolyzed protein, and supplementation with specific nutrients (e.g., vitamin D, folate, fatty acids) in children or adults.
The committee recommends that medical schools as well as residency and fellowship programs and other relevant schools include training for health care providers in the management of food allergy and anaphylaxis. Health care providers, including dietitians and mental health professionals, also should receive training on approaches to counseling patients and their caregivers. Counseling training is envisioned to be provided, in part, by professional organizations through various means, including the Internet.
The following elements of food allergy training are appropriate for all health care providers, including emergency medical technicians, emergency room staff, nurses, dietitians, and others:
- Emergency management. This includes training to recognize and manage an anaphylaxis emergency, such as the use
-
of intramuscular epinephrine as a first line of emergency management for episodes of anaphylaxis.
- Counseling on food allergy management and anaphylaxis. This includes identifying food allergies as well as managing and treating them in various settings (e.g., home, school, restaurants), as well as emergency management of anaphylaxis.
As appropriate, physicians and other health care providers also may receive training to provide the following:
- Nutrition counseling. This includes discussion of safe and nutritionally adequate avoidance diets to individuals with food allergies, particularly children and their caregivers. The training also could include offering referral to a dietitian when needed and as part of reimbursable care. In addition, dietitians may receive training in providing individualized dietary advice to people with food allergy and their caregivers.
- Psychosocial counseling. This includes identifying and discussing with patients and caregivers psychosocial concerns (e.g., bullying), validation of feelings, and balancing management with participation in daily activities. Training also could include offering referral to a mental health professional when needed and as part of reimbursable care. In addition, mental health professionals may receive training in counseling individuals with food allergy and their caregivers.
The committee recommends that health care providers counsel patients and their caregivers on food allergy following the most recent food allergy guidelines and emphasizing the need to take age-appropriate responsibility for managing their food allergy. Counseling is particularly important for those at high risk of food allergy and severe food allergy reactions, such as adolescents, young adults, and those with both food allergy and asthma.
The committee recommends that health care providers and others use intramuscular epinephrine (adrenaline) in all infants, children, and adults as a first line of emergency management for episodes of food allergy anaphylaxis. The Food and Drug Administration should evaluate the need for, and, if indicated, industry should
develop an auto-injector with 0.075 mg epinephrine specifically designed for use in infants.
Current auto-injectors have 0.15 mg or 0.30 mg epinephrine, which is not suitable for infants. Consensus is currently lacking on first aid management using available auto-injectors when managing infants. A dose of 0.075 mg from an auto-injector could fill this gap. Labeling the auto-injectors in a standard manner to differentiate doses also could be beneficial.
Training First Responders and First Aiders
Food anaphylaxis can occur in any setting, and proper emergency management can be life-saving. The public, particularly first responders and first aid personnel, need to be prepared to assist with food-related severe reactions. Overall, food allergy anaphylaxis is not included in training curricula of organizations that offer certifications on emergency training or specialized training for professionals, such as pediatric specialization for early care and education providers.
The committee recommends that organizations, such as the American Red Cross or the National Safety Council, who provide emergency training (e.g., first aid training, basic life support) to the general public and to first responders and first aid personnel in various professions and workplaces, include food allergy and anaphylaxis management in their curricula.
Training Food Industry Personnel
The committee found deficiencies in the knowledge of food industry personnel, including poor communication within the establishment, staff failure to prevent cross-contact, and lack of knowledge about hidden ingredients.
The committee recommends that food industry leaders provide the necessary resources for integrating food allergy training (e.g., food allergen identification and preventive controls, effective risk communication with customers) into existing general food safety and customer service training for employees at all levels and stages in the food industry, as appropriate, encompassing processing, retail food and grocery stores, restaurants, and other food service venues.
Training for employees could be offered through, for example, supporting conferences, workshops, or webinars to share best practices related to allergen preventive controls, food allergen risk communication, and other food allergen safety topics. State health departments could develop a certification process for allergy awareness and management in restaurants modeled after the letter grading system that rates their food safety performance.
Implement Improved Policies and Practices to Prevent the Occurrence of Severe Reactions
Policies Regarding Labeling of Packaged Foods
The food processing industry and the federal government have an essential role in informing individuals at risk of food allergy about the presence of allergens in foods. There are two types of allergen labeling: (1) mandatory, when the allergen is added as an ingredient; (2) voluntary, when the allergen might be inadvertently in the food as a result of cross-contact.
The list of major allergens to be labeled in food packages, which has been adopted by many countries, has not been reviewed since it was developed by the Codex Alimentarius Commission (CAC) in 1999. Also, some U.S. labeling policies are not effective in informing consumers about the risks from food allergens.
In terms of voluntary labeling, unintentional allergens at levels that could cause a reaction can be identified on the labels of packaged foods using precautionary allergen labels (PALs) with wording such as “X may be present.” Currently, PALs bear no relationship to risk. To improve the labeling of unintentional allergens, the Allergen Bureau of Australia and New Zealand has developed the VITAL® (Voluntary Incidental Trace Allergen Labeling) program, which is based on risk assessment principles.
The committee recommends that the Codex Alimentarius Commission and public health authorities in individual countries decide on a periodic basis about which allergenic foods should be included in their priority lists based on scientific and clinical evidence of regional prevalence and severity of food allergies as well as allergen potency.
For example, in the United States, some foods listed by the Food and Drug Administration as tree nuts (i.e., beech nut, butternut, chestnut, chinquapin, coconut, gingko nut, hickory nut, lichee nut, pili nut, shea nut) could be removed from the current priority
list based on the paucity of data or low frequency of allergic reactions. In addition, evidence of the allergy prevalence and reaction severity to sesame seeds may warrant their inclusion on the priority allergen list in the United States.
The committee recommends that the Food and Drug Administration makes its decisions about labeling exemptions for ingredients derived from priority allergenic sources based on a quantitative risk assessment framework.
A quantitative risk assessment is based on knowledge of the detectable level of protein, its presence in the ingredient, exposure levels to the ingredient, and threshold dose-distributions for individuals allergic to the food.
The committee recommends that the food manufacturing industry, the Food and Drug Administration (FDA), and the U.S. Department of Agriculture (USDA) work cooperatively to replace the Precautionary Allergen Labeling system for low-level allergen contaminants with a new risk-based labeling approach, such as the VITAL program used in Australia and New Zealand.
To meet this risk-based approach, the following three steps are recommended:
- The FDA and the USDA should establish Reference Doses (thresholds) for allergenic foods, where possible. The committee concludes that at this time, sufficient data exist on milk, egg, peanut, certain tree nuts (i.e., cashew, walnut, hazelnut), wheat, soybean, fish, and crustacean shellfish (shrimp) to establish Reference Doses. The FDA and the USDA should review the Reference Doses periodically, with particular attention to the remaining tree nuts for which data to establish Reference Doses are not currently available (i.e., almond, Brazil nut, macadamia nut, and pine nut).
- Once Reference Doses are established, a food product would carry an advisory label (e.g., “peanut may be present”) only in situations when ingesting the product would expose the individual to a level above the Reference Dose for that allergen. The FDA should restrict the number of allowable advisory labels to one phrase. Because this labeling is voluntary, the product should clearly inform the
-
consumer, through labeling as appropriate, as to whether a risk-based approach (such as VITAL) has been followed for each specific product. The FDA and the USDA should educate health care providers and consumers about the meaning of such a food allergy advisory statement.
- The FDA and the USDA, together with the food industry and the analytical testing industry, should develop and validate detection methods and sampling plans for the various food allergens for which Reference Doses are established. A common unit of reporting also should be established, such as parts per million of protein from the allergenic source, so that comparisons can be made between methods and between levels in the food and clinical threshold values.
Policies at Specific Settings
The FDA Food Code provides advice from the FDA for uniform systems and practices that address the safety of food sold in food establishments. The 2013 FDA Food Code includes provisions on preventing food allergic reactions but it has not been adopted by all states.
The committee recommends that all state, local, and tribal governmental agencies adopt the 2013 Food and Drug Administration Food Code, which includes provisions for food establishments on preventing food allergic reactions. Working in collaboration with other stakeholders, the agencies also should propose that the next Food Code requires that the person in charge in food establishments pass an accredited food safety certification program that includes basic food allergy management in order to decrease or prevent the risk of food allergen exposure. In addition, agencies should develop guidance on effective approaches to inform consumers with food allergies in food service establishments.
Guidance on effective approaches to inform consumers with food allergens in food service establishments could include menu designations of allergens and posters, and other forms of displaying information about food allergens in food establishments.
The CDC Voluntary Guidelines for Managing Food Allergies in Schools and Early Care and Education Programs (the CDC Food Allergy Guidelines) includes essential management approaches, such as preparing for food allergy emergencies, but they have not been implemented widely in all schools. Higher education institutions do not have similar guidelines.
Although reports of severe reactions while flying are rare, accidents can occur and improving policies and practices might prevent them. In response to its task, the committee developed specific recommendations for ways to assure that appropriate guidance and education are in place to create a safe public environment for individuals with food allergy. In doing so, the committee recognized that its task did not include recommendations for therapeutic intervention or clinical management of food allergies.
The committee recommends that, within the next year, relevant federal agencies (e.g., the Food and Drug Administration [FDA], the Centers for Disease Control and Prevention [CDC], the Federal Aviation Administration) convene a special task force that includes participants from the medical community, food companies, and advocacy stakeholder groups to establish and implement policy guidelines to:
- Assure emergency epinephrine capabilities are in place for children and adults in public venues, including schools, early care and education facilities, and on-board airlines;
- Provide standardized food allergy and anaphylaxis first aid training (e.g., identification of major food allergens, signs and symptoms of allergic reactions, and emergency treatment protocols) to appropriate school and university health staff, early care and education providers, and on-board flight crews; and
- Implement education standards for responding to and managing food allergy emergencies in schools and early care and education facilities (e.g., CDC Food Allergy Guidelines), and on airlines.
The committee recommends that the FDA continue to work together with other relevant federal, state, and local agencies to develop and implement labeling policies specific to allergenic ingredients in packaged and prepared foods that are distributed through airlines and other public venues, including schools and early care and education facilities.
Expand Research Programs
The committee lists research needs in areas of mechanisms of action, better diagnostic tools, effective educational approaches, and evidence-based guidelines for all stakeholders, and prospective and clinical trials to support or refute current hypotheses on the development of food allergies. In addition, although the committee did not review emerging therapeutic
approaches to cure food allergies, it included development of effective and safe therapies as a key long-term goal. The details of the research needs are in Chapter 9.
The committee envisions that this report will reach many stakeholders, including consumers, patients, health care providers, school leaders, food manufacturers, and food establishment managers, and serve as guidance for future understanding and management of food allergies. The committee also has confidence that the recommendations in this report, if implemented, will stimulate progress in the understanding of food allergies, reduce further uptakes in prevalence, and improve the quality of life of those with this chronic disease and their caregivers.


















