8
Military Schedules vs. Biological Clocks
R. S. Pozos1 D. E. Roberts A. C. Hackney and S. J. Feith
INTRODUCTION
During military operations, there is an inherent conflict between the internal body clock and operational schedules. Military operations require military personnel to dramatically change their daily schedules to accommodate prolonged activity at night and enforced inactivity during the day. Due to logistical challenges, food may not be delivered on a scheduled basis. Dehydration may be a constant concern. Military history is replete with reports of military personnel who were both sleep deprived, and malnourished and in very hostile thermal environments as they engaged hostile forces for prolonged periods of time.
Coexisting with this military schedule are the inborn biological ''clocks" that coordinate various physiological processes such as sleeping, eating, endocrine release, and motor activity. Long-term desynchronization of various physiological processes could result in disruption in the overall physiological
or mental performance of military personnel during military operations (Folkard et al., 1988).
Sleep is the physiological process that is most easily disrupted by military operations (Naitoh and Kelly, 1992). Transporting persons at night over various time zones is a classic way to affect the quality of sleep. The behavioral changes associated with sleep deprivation have been known since historical times. However the accompanying changes in core and skin temperature are still being elucidated. The focus of this paper will be to discuss the role that sleep has on thermal balance in a cold environment.
BIOLOGICAL CLOCKS: FROM MINUTES TO YEARS
Biological clocks have two major components: duration and activity. The manifestation of the clock is best demonstrated as a sine wave, which has amplitude, frequency characteristics, and duration. A biological clock for one physiological process may be synchronized with another clock resulting in two sine waves that may have the frequency, amplitude, or duration correlated with each other. Thus, a biological clock that controls body temperature will cause a variation in core temperature over a period of time. The oscillatory variation in core temperature of ± 1.5°C (2.7°F) results in a sine wave amplitude over that period of time. Biological clocks can range from minutes to years. The ultradian clocks are those that control physiological events that have a duration of a few minutes to several hours (Dean and Aschoff, 1981). An example of the expression of an ultradian clock is the release pattern of luteinizing hormone (Soper and Weick, 1980). Infradian cycles may range from 14 to 15 days and from 28 to 30 days and have been termed semilunar and lunar respectively (Neumann, 1989). In addition, there are rhythms that are regulated on an annual basis and have been called circannual. The best example of a circannual clock is the reproductive cycle of animals and associated activities such as milk production (Hastings, 1991).
The biological clocks that have received the most attention are the circadian oscillators which regulate and coordinate physiological processes on an approximately 24-h basis (Refinetti and Menaker, 1992). The expressions of these biological clocks in such behaviors as feeding, body temperature, and motor activity are independent of environmental cues and can be considered as independent oscillators (Inouye and Kawamura, 1979). However, under normal conditions the activity of the circadian oscillator is entrained or synchronized by various environmental cues. Of these external cues, the light/darkness cycle is the primary synchronizer for the circadian oscillators. This relationship between the light/darkness cycle and the 24-h circadian clock is very powerful since alteration in the light/darkness cycle influences multiple physiological processes that vary during a 24-h cycle. It was initially reported that the light/dark cycle was a poor synchronizer in humans; however, more
recent studies suggest that light is able to alter the sleep/wakefulness cycle if the intensity of the light is strong enough (Czeisler and Allen, 1980; Czeisler et al., 1986). In addition to the light/darkness cycle being a potent external signal, motor activity is another external cue that influences the circadian cycle. Recent studies in animals suggest that an increase in locomotive activity can shift the phase of the circadian cycle (Reebs and Mrolsovsky, 1989). The converse seems to hold as well in that induced inactivity can also induce phase shifts in the circadian cycle (Van Reeth et al., 1991).
While the light/darkness cycle and motor activity are just a few of many external cues that can alter circadian cycle, social factors may also play a role in synchronizing the clocks. Man is the only animal that habitually does not respond to his biological clocks. For example, during a military night operation, soldiers must be awake and may possibly engage in vigorous physical activity when normally they would be asleep.
DIFFERENT CIRCADIAN CLOCKS: SLEEP/WAKEFULNESS AND BODY TEMPERATURE
Both the sleep/wakefulness and body temperature systems have certain common features: (1) there are certain select areas (biological clock) of the central nervous system that control their expression, (2) sensory signals influence their activity, and (3) motor or effector signals are an expression of the biological clock. Each clock is controlled by a different area of the central nervous system; however, they interact with each other and influence other physiological systems.
Sleep/Wakefulness
The complex activity of sleep/wakefulness varies so that on average there is approximately 8 hours of sleep and 16 hours of activity per day. However, this ratio varies depending on age, activity, and other factors. As is commonly observed, the sleep phase is associated with a decrease in activity which results in a decrease in metabolism and core temperature. This state of inactivity is further characterized by specific changes which can be measured by electroencephalogram (EEG), electromyogram (EMG), and electrooculogram (EOG), as well as endocrine changes. Based on these criteria, sleep can be divided into two distinct phases, nonrapid eye movement (NREM) and rapid eye movement (REM) sleep. During NREM sleep, there is an alteration in thermoregulation, which causes a decrease in core temperature (Glotzbach and Heller, 1993). During REM sleep, thermoregulation is further attenuated (Glotzbach and Heller, 1984). The neuroanatomical site for the sleep/wakefulness
clock is the suprachiasmatic nuclei (SCN) of the hypothalamus (Meijer and Rietveld, 1989). When the SCN is lesioned, sleep and wakefulness are distributed equally throughout the day (Ibuka and Kawamura, 1975), but normal oscillations of core temperature persist (Fuller et al., 1981). These data, reported from a number of different experiments, have led to the present model that states that the circadian clocks controlling sleep/wakefulness are separate from those that control body temperature.
The responses of the circadian clocks are dampened with aging. Numerous reports have demonstrated an alteration in both thermal and motor activity in aging animals (Martin et al., 1985).
The effect of sleep deprivation is of great concern to the military. Numerous studies have shown that sleep deprivation leads to impaired psychomotor performance, although physiological response to the cold is maintained (Fiorica et al., 1968). Sleep-deprived subjects experience periods of visual hallucinations, reduced vigilance, impaired balance, and impaired coordination (Opstad et al., 1978).
Thermal Regulation in the Cold
Normally, core body temperature varies no more than 1°C (0.9°F) per day (Guyton and Hall, 1996). Core temperature is controlled by nuclei in the preoptic anterior hypothalamus (POAH) (Boulant and Hardy, 1974; Wit and Wang, 1968). The POAH acts as a thermostat and maintains the core temperature at 37°C (98.6°F). Thermal sensory signals from both the periphery and the core are transmitted to the POAH. If there is a difference in temperatures between the periphery and the core, there will be an increase in motor tone, vasoconstriction, release of catecholamines, thyroid hormones, and other factors, resulting in a maintenance of core temperature. Hypothermia is clinically defined as a core body temperature of 35°C (95°F) or lower and is classified as either accidental (primary) or secondary. Primary hypothermia refers to a decrease in core temperature that is induced by an overwhelming environmental stress. Secondary hypothermia refers to a decrease in core temperature that is due to the fact that there is an underlying pathology which does not allow the thermally stressed individual to respond physiologically to the cold environment. The effects of a decrease in core temperature have been reported elsewhere but it should be noted that hypothermia has major effects on a person's cognition and higher mental functions, resulting in amnesia, apathy, dysarthria, impaired judgment, and maladaptive behavior (Danzl and Pozos, 1994). In contrast to the chronic exposure to cold that causes these changes, even acute exposure to cold environments will cause hallucinations without any other signs of hypothermia (Lloyd, 1983).
Sleep and Core Temperature
The interaction of sleep and the ambient environment can be considered from two points of view: (1) what are the changes in core temperature that occur during the REM and NREM sleep, and (2) what is the effect of ambient temperature on the subject during REM and NREM sleep. It has been commonly reported that changes in body temperature and metabolic rate at sleep onset in humans suggest a "decrease" in the regulation of core temperature. Decreases in brain temperature accompany the transition from wakefulness to NREM sleep (Glotzbach and Heller, 1993). The significance of this phenomenon is still debated. Among the more interesting hypotheses is that NREM sleep may be associated with a cooling of the brain. Experimental data do not support this contention since small animals in cold climates sleep as much as large animals in warm climates do.
During REM sleep, thermoregulation is inhibited in experimental animals. Parmeggiani and Rabini (1967) reported that cats who slept at low ambient temperatures ceased shivering at the onset of REM sleep. REM sleep is thought to be associated with brain warming, however since there is a decrease in thermoregulation during this period of time, this theory is also not well supported. Whatever the effect the various cycles of sleep have on thermoregulation, there is agreement that these various stages of sleep may play some role in possibly conserving energy as the animal sleeps.
It is commonly observed that when individuals have a prolonged period of sleeplessness they complain of being cold. The explanation for this phenomenon may be that the sleep wake cycle has a 33-h period whereas core temperature has a 24-h period and the two may become desynchronized. The physiological mechanisms for maintaining normal core temperatures can be divided into two major categories: heat dissipating such as peripheral vasodilation and heat productive such as increase in metabolism. In humans in whom the sleep/wake cycle and core temperature have been desynchronized (loss of coupling), control of the peripheral vasculature is coupled with the sleep/wake cycle and is independent of the core body temperature (Czeisler et al., 1980). As a result, the sleep deprived individual may experience peripheral vascular constriction during the day, giving the sensation of peripheral coolness.
External Temperatures and Sleep
It is commonly observed that individuals can adapt to sleeping in various thermal environments. Initially, a person who is not accustomed to sleeping in the cold will experience a decrease in the amount of total sleeping time. However, once the individual has become acclimated to the cold there will be no decrease in the time spent in REM sleep (Palca et al., 1986). Although high
intensity exercise has been reported to increase delta sleep, a component of NREM sleep, and to have no effect on REM sleep (Horne and Moore, 1985), additional studies suggest that such a definitive effect of exercise on various components of sleep may not exist (Horne and Staff, 1983). Due to the complexity of the interaction between sleep and the thermoregulatory response to environmental temperatures, there is no model that is able to coalesce the data from various experiments.
Sleep and Nutrition
The interaction between sleep and nutrition should be addressed since sleep may play a role in altering the rate of metabolism. In experimental animals, it has been shown that the amount and type of food ingested and the duration of subsequent sleep are highly correlated (Danguir et al., 1979). A number of studies suggest that ingestion of carbohydrates, which triggers the release of insulin, is associated with an increase in the duration of NREM, whereas ingestion of protein triggers the release of somatostatin to cause an increase in REM sleep (Danguir, 1996). The importance of these studies is that they suggest that sleep is not merely a process that is regulated by various levels of light and darkness or exercise, but that nutrition may also play a role in modifying the sleep cycle. Complicating this picture is the fact that diet-induced thermogenesis is dependent on the time at which the food is ingested. In a study in which human subjects were fed the same diet during different phases of the sleep/wakefulness cycle, the authors reported different thermogenic responses (Romon et al., 1993).
THE REAL WORLD: MILITARY OPERATIONS AND TRAINING
The above studies demonstrate that there is a complex relationship between sleep/wakefulness and thermoregulation. These data should not be construed to suggest that persons who are sleeping in cold environments might inadvertently slip into hypothermia. Individuals who are physically fit and who are not suffering from some disease or taking medication are able to react to the environmental stressors that occur during a cold night. In the event that the subject is: (1) physically exhausted; (2) energy depleted (malnourished); (3) sleep deprived; and (4) in constant exposure to a cold environment, it is very probable that he/she will suffer a drop in core temperature leading to secondary hypothermia and death (Pugh, 1966). It should be emphasized that, before this point is reached, the higher mental dysfunctions that are associated with sleep deprivation and/or cold stress will be evident. The actual cause of death of a hypothermic person is usually lack of sleep, insufficient food, and improper clothing which leads to a drop in core temperature. This triggers
lapses in judgment that lead to further drops in core temperature and ultimately to death (Pugh, 1966). A classical historical example of the interplay between sleep deprivation, exhaustion, and constant exposure to the cold is the Finnish Campaign against the Russians in World War II. The Finnish Army did not let the Russians sleep, interrupted their supply of food and water, and patiently waited while the cold sapped the physical and mental stamina of the Russians. When the Finns attacked, the Russians were unable to mount an effective counterattack (Jarvinen, 1944). This same tactic was basically applied by the Russians when they repulsed the German invasion in World War II (Jarvinen, 1944).
A key factor in any instance of hypothermia is not only sleep deprivation but also body weight loss. Without the metabolic substrates that are required to maintain normal core temperature, the elaborate circadian cycles will not be able to maintain normal human performance in a cold environment. In a recent study, Hackney et al. (1995) studied the changes in lean body mass of military personnel engaging in mountain operations. Fifteen U.S. Navy and Marine Corps personnel were part of a Marine Corps expedition to climb Mount Denali, Alaska. The total expedition took 13.5 days during which time the subjects were able to eat and drink rations ad libitum. The subjects carried their own equipment (backpacks [23 to 28 kg] and sleds [33 to 50 kg]). Weather conditions varied from -30° to 17°C (-22° to 63°F). During the ascent, the group was caught in a snow storm and became separated. Four men were able to make the ascent successfully. Body weight was significantly less following the expedition (post = 76.6 ± 1.5 kg vs. pre = 81 ± 1.5 kg). Those who were able to complete the climb had lost more weight than those who were forced to stay in their tents due to climatic conditions. The men reported that they slept when they could. This example is used to emphasize that many times during training or real military operations, the challenges facing the personnel are multiple: lack of food, unanticipated events, and sleep deprivation (Hackney et al., 1995). Sleep deprivation by itself would not be lethal; however, when combined with a decrease in body fat, and thus, the body's inability to resist the cold challenge—then hypothermia and frost bite can occur.
In military situations, men have slept on the cold ground and have not died from hypothermia. The classic studies by Hammel (1964) demonstrate that "primitive men" are able to sleep nude in cold environments that would be unacceptable to European men accustomed to sleeping in a warm microenvironment. He studied groups living in various parts of the world and noted that when the subjects were sleeping there were diverse physiological responses to the cold environment. His subjects included Australian aborigines, bushmen of the Kalahari Desert, Alacaluf Indians of Tierra del Fuego, Arctic Indians, Andean Indians, Eskimos, and Lapps. In addition, he was able to show that Europeans were able to adjust to the cold after being exposed for a few weeks and becoming cold "hardy" (Hammel, 1964). This practice
continues in that modern explorers who are attempting to cross the South or North Pole sleep outdoors during the winter months in northern Minnesota to get their bodies adjusted to the anticipated cold (Personal communication, W. Steger, Steger Inc., Duluth, Minn., 1990).
Technology to the Rescue: Antiquity to the Present
Man's ability to withstand the challenges of a cold environment have ultimately evolved into modifications of the immediate environment. Although physiological adjustments have been reported by Hammel and many others, mankind is ingenious in finding ways to survive and sleep in cold environments. Initially, mankind was able to modify the environment by living in caves, discovering fire, and using animal furs for protection. Presently, sleeping bags have become a key component in the military's ability to conduct cold weather operations. In a recent study, Roberts et al. (Unpublished manuscript, "Physiological evaluation of sleeping bag cover," D. E. Roberts, J. E. Reading, S. J. Feith, and R. S. Pozos, Naval Health Research Center, San Diego, 1995) studied the rectal and peripheral temperatures of Marines who slept at -29°C (-20°F) in a variety of sleeping bags that were lightweight and constructed of synthetic material. Subjects were outfitted with skin and rectal thermocouples while they slept in a cold chamber. Subjects wore "long underwear," caps, and socks. Rectal temperatures fell approximately 1°C (1.8°F) over the duration of the entire night (Figure 8-1). Weighted skin temperatures also fell approximately 1°C (1.8°F), but this figure is misleading since it does not reflect the actual digit temperatures (Figure 8-2). Thus, as new advances in textiles continue, the military and recreational communities can continue to have adequate protection while sleeping in cold climates that otherwise might be lethal.
The Challenge: Field Studies in Bosnia
Although a large amount of data has been accumulated from studies that investigate various complex interrelationships among sleep, nutrition, physical activity, and skin and core temperature, most of these studies occur in laboratory settings. Although helpful, these studies do not shed light on the overall physiological adjustments that occur during field conditions. With the advent of miniaturized recording equipment, it should be possible to record skin temperatures and physical activity from NATO military forces in Bosnia. In addition, as more women enter the military, it is imperative to study their responses in cold operations. Most studies that have been conducted on human subjects have evaluated male responses primarily. There is a paucity of information about the physiological responses of female personnel who have
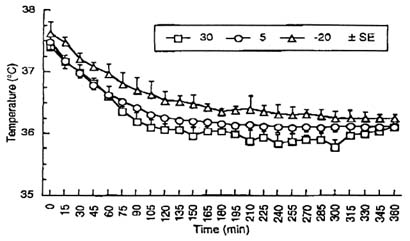
FIGURE 8-1 Rectal temperature (± SE) for 14 male subjects sleeping in military sleeping bags for 6 hours. Sleeping bags are temperature specific for temperatures: 30°, 5°, and -20°C (86°, 41°, and -4°F). SOURCE: Roberts et al. (Unpublished manuscript, "Physiological evaluation of sleeping bag cover," D. E. Roberts, J. E. Reading, S. J. Feith, and R. S. Pozos, Naval Health Research Center, San Diego, 1995).
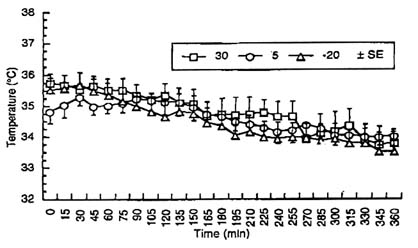
FIGURE 8-2 Mean-weighted skin temperature (± SE) for 14 male subjects sleeping in military sleeping bags for 6 hours. Sleeping bags are temperature specific for temperatures: 30°, 5°, and -20°C (86°, 41°, and -4°F). SOURCE: Roberts et al. (Unpublished manuscript, "Physiological evaluation of sleeping bag cover," D. E. Roberts, J. E. Reading, S. J. Feith, and R. S. Pozos, Naval Health Research Center, San Diego, 1995).
been sleep deprived and are participating in cold weather operations. Although gathering data in the field is a challenging situation, these data could assist in the development of the appropriate boots, tents, food, etc., to minimize cold stress and cold injury.
AUTHORS' CONCLUSION
There will always be a conflict between the demands of military operations and the biological clocks that coordinate various physiological functioning in military personnel. In general, the human body is robust enough to handle these challenging changes by utilizing behavioral and physiological responses. Research results from strictly controlled laboratory settings will assist in the development of possible biological/pharmacological agents that may alter circadian and other biological clocks to enhance human performance in adverse environmental settings. Until that time, modifications of man's immediate environment such as clothing and sleeping bags will be the major way the military will be able to conduct long-term cold weather operations. The evaluation of such gear in the field is required to quantitate the effects of the interplay of various biological clocks on human performance.
REFERENCES
Boulant, J.A., and J.D. Hardy 1974 The effect of spinal and skin temperatures on the firing rate and thermosensitivity of preoptic neurons. J. Physiol. 240:639–660.
Czeisler, A., and J.S. Allan 1980 Rapid phase shifting in humans requires bright light [abstract]. Chronobiologica 14:167.
Czeisler, C.A., E.I. Weitzman, M.C. Moore-Ede, J.C. Zimmerman, and R.I. Knauer 1980 Human sleep: Its duration and structure depends on its circadian phase . Science 210:1264–1267.
Czeisler, C.A., J.S. Allan, S.G. Strogatz, J.M. Ronda, C.D.R. Sanchez, W.O. Freitag, G.S. Richardson, and R.E. Kronauer 1986 Bright light resets the human circadian pacemaker independent of the timing of the sleep-wake cycle. Science 233:667–671.
Daan, S., and J. Aschoff 1981 Short-term rhythms in activity. Pp. 491–498 in Biological Rhythms, J. Aschoff, ed. New York: Plenum.
Danguir, J. 1996 The relationship between food and sleep. Pp. 1375–1387 in Handbook of Physiology, Environmental Physiology, M.J. Fregly and C.M. Blatteis, eds. New York: Oxford University Press.
Danguir, J., H. Gerard, and S. Nicolaidis 1979 Relations between sleep and feeding patterns in the rat. J. Comp. Physiol. Psychol. 93:820–830.
Danzl, D.F., and R.S. Pozos 1994 Accidental hypothermia. New Engl. J. Med. 331:1756–1760.
Fiorica, V., E.A. Higgins, P.F. Iampietro, M.T. Lategola, and A.W. Davis 1968 Physiological response of men during sleep deprivation. J. Appl. Physiol. 24(2):167–176.
Folkard, S., D.S. Minors, and J.M. Waterhouse 1988 Chronobiology and shift work: Current issues and trends. Chronobiologica 12:31–54.
Fuller, C.A., R. Lydic, F.M. Sulzman, H.E. Albers, B. Tepper, and M.C. Moore-Ede 1981 Circadian rhythm of body temperature persist after suprachiasmatic lesions in the squirrel monkey. Am. J. Physiol. 241:R385–R391.
Glotzbach, S.F., and H.C. Heller 1984 Changes in the thermal characteristics of hypothalamic neurons during sleep and wakefulness. Brain Res. 309:17–26.
1993 Temperature regulation. Pp. 260–275 in Principles and Practice of Sleep Medicine, 2nd ed., M.H. Kryer, T. Roth, and W.C. Dement. Philadelphia: W.B. Saunders Co.
Guyton, A.C., and J.E. Hall 1996 Textbook of Medical Physiology, 9th ed., pp. 911–912. Philadelphia: W.B. Saunders Co.
Hackney, A.C., S. Feith, R.S. Pozos, and J. Seale 1995 Effects of high altitude and cold exposure on resting thyroid hormone concentrations. Aviat. Space Environ. Med. 66:325–329.
Hammel, H.T. 1964 Terrestrial animals in cold: Recent studies of primitive man. Pp. 413–434 in Handbook of Physiology, Section 4: Adaptation to the Environment, D.B. Dill, E.F. Adolph, and C.G. Wilber, eds. Washington, D.C.: American Physiological Society.
Hastings, M.H. 1991 Neuroendocrine rhythms. Pharmacol. Ther. 50:35–71.
Horne, J.A., and V.J. Moore 1985 Sleep EEG effects of exercise with and without additional body cooling. EEG Clin. Neurophysiol. 60:33–38.
Horne, J.A., and L.H. Staff 1983 Exercise and sleep: Body-heating effects. Sleep 6:36–46.
Ibuka, N., and H. Kawamura 1975 Loss of circadian rhythm in sleep-wakefulness cycle in the rat by suprachiasmatic nucleus lesions. Brain Res. 96:76–81.
Inouye, S.I.T., and H. Kawamura 1979 Persistence of circadian rhythmicity in a mammalian hypothalamic island containing the suprachiasmatic nucleus. Proc. Natl. Acad. Sci. 76:5962–5966.
Jarvinen, Y.A. 1944 Finnish and Russian Tactics in the Winter War. Helsinki: Werner Söderström, D.Y.
Lloyd, E. 1983 Hallucinations in hypothermia cold stress and their neurochemical basis. Pp. 40–42 in Environment, Drugs and Thermoregulation, P. Lomax and E. Schönbaum, eds. New York: Karger.
Martin, J.R., A. Fuchs, R. Bender, and J. Harting 1985 Altered light-dark activity difference with aging in two rat strains. J. Gerontol. 44:2–7.
Meijer, J.H., and W.J. Rietveld 1989 The suprachiasmatic circadian pacemaker in rodents. Physiol. Rev. 69:671–707.
Naitoh, P., and T.L. Kelly 1992 Sleep management user's guide for special operations personnel. Naval Health Research Center Technical Report No. 92-28. San Diego, Calif.: Naval Health Research Center.
Neumann, D. 1989 Circadian components of semilunar and lunar timing mechanisms. J. Biol. Rhythms 4:285–294.
Opstad, P., R. Erwenger, M. Nummestad, and N. Raabe 1978 Performance, mood and clinical symptoms in men exposed to prolonged, severe physiological work and sleep deprivation. Aviat. Space Environ. Med. 49:1065–1073.
Palca, J.W., J.M. Walkelr, and R.J. Berger 1986 Thermoregulation, metabolism and stages of sleep in cold exposed men. J. Physiol. 61:940–947.
Parmeggiani, P.L., and C. Rabini 1967 Shivering and panting during sleep. Brain Res. 6:789–791.
Pugh, L.G.C.E. 1966 Accidental hypothermia in walkers, climbers, and campers: Report to medical commission on accident prevention. Brit. Med. J. 1:123–129.
Reebs, S., and N. Mrolsovsky 1989 Effects of induced wheel running on the circadian activity rhythms of Syrian hamsters: Entrainment and phase response curve. J. Biol. Rhythms 4:39–48.
Refinetti, R., and M. Menaker 1992 The circadian rhythm of body temperature. Physiol. Behav. 51:613–637.
Romon, M., J.L. Edme, L.C. Boulenguez, J.L. Lescroart, and P. Frimat 1993 Circadian variation of diet induced thermogenesis. Am. J. Clin. Nutr. 57:476–480.
Soper, B.D., and R.F. Weick 1980 Hypothalamic and extrahypothalamic medication of pulsatile discharges of luteinizing hormone in the ovariectomized rat. Endocrinology 106:348–355.
Van Reeth, O., D. Hinch, J.M. Tecco, and F.W. Turek 1991 The effects of short periods of immobilization on the hamster circadian clock. Brain Res. 545:208–214.
Wit, A., and S.C. Wang 1968 Temperature sensitive neurons in preoptic/anterior hypothalamic region: Effects of increasing ambient temperature. Am. J. Physiol. 215:1151–1159.












