20
Energy and Macronutrient Requirements for Work at High Altitudes
Reed W. Hoyt1 and Arnold Honig
INTRODUCTION
During military operations in the mountains foot soldiers are challenged with rugged terrain and long, physically demanding days. Hypoxia, cold temperatures, and restricted food and water availability can impose additional stresses. Developing operational field rations that meet nutritional demands under these conditions requires accurate estimates of soldier energy requirements. Energy requirements determine the total caloric contribution required from food and body energy stores. The specific fuel combusted to meet energy requirements depends primarily on the intensity and duration of work performed, macronutrient availability from rations and body energy stores, and environmental conditions (Ahlborg and Felig, 1982; Felig and Wahren, 1975; Stein et al., 1989; Young and Young, 1988).
It is increasingly evident that soldiers participating in field training exercises, particularly those in mountainous terrain, consistently have high rates of energy expenditure and limited dietary energy intakes (Hoyt et al., 1991, 1994a) (Table 20-1). Why are soldiers characteristically in negative energy balance? The following discussion will attempt to address this question and others posed by the Committee on Military Nutrition Research.
ENERGY BALANCE
Exercise Energy Expenditure
Duration of Activity
High rates of energy expenditure can be attributed in part to the large portion of the day that soldiers spend in physical activity. For example, monitoring of ambulatory activity showed that soldiers were active around 17.3 ± 0.2 h/d (mean ± SD) (n = 20) over the course of 11 days during a physically demanding winter military training course (Hoyt et al., 1991). Similarly, Special Operations Forces soldiers were active about 16 ± 2 h/d during a 6-d military field training exercise at 2,500 to 3,100 m (8,202 to 10,171 ft) elevation on Mount Rainier (elevation = 4,392 m [14,410 ft], Mount Rainier National Park, Wash.) (Hoyt et al., 1994a).
TABLE 20-1 Ration Consumption and Estimated Energy Expenditure of Soldiers in the Field
Work Intensity
Average daily multiples of basal metabolic rate (BMR) have been used to classify occupational work levels as light (1.55 × BMR), medium (1.78 × BMR), or heavy (2.10 × BMR) (FAO/WHO/UNU, 1985). Recent studies using the doubly labeled water method to measure total daily energy expenditure have found that the work level of soldiers commonly exceeds 2.10 × BMR. In a winter military training course, work levels averaged 4.03 ± 0.22 × BMR (7,131 ± 225 kcal/d) over the first 4 days when the soldiers were particularly active, with an overall average of 2.8 ± 0.2 × BMR (4,919 ± 190 kcal/d) for the entire 11-d study (Hoyt et al., 1991). Similarly, an average of 2.5 × BMR was expended over 7 days by soldiers training for jungle warfare (Forbes-Ewan et al., 1989), while the work level of Special Operations Forces soldiers on Mount Rainier was 3.2 ± 0.2 × BMR (4,558 ± 566 kcal/d) (Hoyt et al., 1994a).
By comparison, the work level of trained amateur cyclists studied over a 3.5-d period that included 2 days of heavy activity (two 4-to 5-hour bouts of exercise to exhaustion per day) averaged 2.6 × sleeping metabolic rate (Westerterp et al., 1988). Examples of extreme work rates (4.2 to 5.3 × BMR) have been reported for cyclists in the Tour de France (Westerterp et al., 1986) and in trained athletes in the laboratory (Stein et al., 1987). The predicted maximum rate of daily energy expenditure of ultra-long-distance runners is about 7.6 × BMR or 14,000 kcal/d (Davies and Thompson, 1979).
What is the Effect of Altitude Exposure on 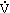 o2max and Physical Endurance?
o2max and Physical Endurance?
The cardiorespiratory responses to exercise at moderate altitudes are well documented (Fulco and Cymerman, 1988; Grover et al., 1986; Young and Young, 1988). Although variable among individuals, an approximate 10 percent decrease in maximal oxygen uptake (![]() o2max) occurs for every 1,000 m (3,280 ft) increase in elevation above 2,000 m (6,562 ft). The
o2max) occurs for every 1,000 m (3,280 ft) increase in elevation above 2,000 m (6,562 ft). The ![]() o2 at any given submaximal power output, however, remains unchanged from sea level to altitude. Consequently, exercise at altitude at a given power output requires a greater percentage of
o2 at any given submaximal power output, however, remains unchanged from sea level to altitude. Consequently, exercise at altitude at a given power output requires a greater percentage of ![]() o2max, that is, a higher relative exercise intensity.
o2max, that is, a higher relative exercise intensity.
Acclimatization to moderate altitudes dramatically increases endurance exercise capacity (time to exhaustion during sustained exercise at a constant exercise intensity) (Horstman et al., 1980; Maher et al., 1974), but it has little or no effect on ![]() o2max (Boutellier et al., 1990; Horstman et al., 1980; Young et al., 1982). Compared to sea level, cycle ergometer endurance time at 75 percent of
o2max (Boutellier et al., 1990; Horstman et al., 1980; Young et al., 1982). Compared to sea level, cycle ergometer endurance time at 75 percent of ![]() o2max increased 61 percent after 12 days at 4,300 m (14,110 ft) (Maher et al., 1974), while time to exhaustion with treadmill running at 85 percent of
o2max increased 61 percent after 12 days at 4,300 m (14,110 ft) (Maher et al., 1974), while time to exhaustion with treadmill running at 85 percent of ![]() o2max increased 41 percent after 16 days at 4,300 m (14,110 ft)
o2max increased 41 percent after 16 days at 4,300 m (14,110 ft)
(Horstman et al., 1980). The increase in endurance capacity with acclimatization to altitude appears to be related to improved oxygen delivery.
Resting Energy Expenditure
Does Altitude Exposure Increase Basal Energy Requirements?
Short-term measurements of gas exchange during several field studies suggest that acute hypoxia increases BMR and possibly even the energetic costs of exercise. The work of Schneider et al. (1924) suggested that oxygen consumption tended to increase after 3.5 to 8 hours of exposure to 400 mm Hg (about 4,900 m [16,076 ft]). Leulier (1954) found that 19 of 29 subjects exhibited increased BMR at an elevation of about 3,000 m (9,843 ft). Increased BMR was evident in 10 men and 2 women studied at sea level and during a 12-d sojourn at 1,850 m (6,070 ft) (Terzioglu and Aykut, 1954). Grover (1963) reported a small (5 percent) but significant increase in BMR in two women and four men with the transition from 1,580 m to 4,300 m (5,184 to 14,110 ft). Gill and Pugh (1964) found that six of eight expedition members showed a 10 percent increase above BMR values predicted for sea level. In a study by Surks et al. (1967), mean oxygen consumption in five young males increased significantly on the first day at altitude, remained significantly elevated on day 4, and then decreased progressively towards control values by day 8. Nair et al. (1971) reported that BMR increased significantly after 7 days at 3,353 m (11,000 ft), then returned to sea level values by day 14. This result is generally consistent with the findings of Meda (1955), who reported that no difference in metabolic rate was evident in seven adults (three males and four females) tested at sea level and after 7 to 17 days at 3,000 m (9,843 ft). In Hannon and Sudman's (1973) study of eight young women, exposure to an elevation of 4,300 m (14,110 ft) increased basal oxygen consumption, with the maximum increment at 36 hours. In a study of five males who were taken from 1,600 to 4,300 m (5,249 to 14,100 ft) (Huang et al., 1984), resting metabolic rate increased 16 percent on day 1 and remained elevated through the end of the experiment on day 5. In a recent study by Butterfield and coworkers (1992), BMR increased 27 percent on day 2, decreasing to +17 percent by day 10, in seven men taken to 4,300 m (14,110 ft) elevation. After 1 week at 3,650 m (11,975 ft) elevation, fasting resting metabolic rate and exercise metabolic rate in six volunteers, four males and two females, had increased significantly over sea-level values (Stock et al., 1978). Significant increases in oxygen uptake during standardized treadmill exercise were noted in a study of 27 young men after 4 to 9 days at 4,300 m (14,110 ft) (Johnson et al., 1971).
Although these field studies suggest that BMR increases with acute hypoxia, no consensus exists as to the magnitude of the effect. This conclusion is, in part, due to the very short duration of the gas exchange measurements
used to estimate BMR, the unavoidable lack of control in field studies, and the broad range of reported increases in BMR.
The effects of menstrual cycle on energy expenditure may contribute to variation in the effects of hypoxia on BMR. Significant (6 to 15 percent) increases in energy expenditure during pre- to postovulation are reported (Bisdee et al., 1989; Webb, 1986). Additionally, in a study of 10 women, a relatively large intrasubject coefficient of variation in resting energy expenditure was attributed in part to the effect of the menstrual cycle on energy expenditure (De Boer et al., 1987).
Energy Intake
Field Ration Consumption at Sea Level
Soldiers normally, perhaps innately, do not consume enough food to meet the energy demands of strenuous field training exercises, regardless of the type of field ration they are consuming (Table 20-1). Voluntary consumption of currently available field rations rarely exceeds 3,000 kcal/d, with food wastage averaging about a third of the total calories available. The only instance in which physically active soldiers have maintained energy balance while in the field was when they were provided hot A Rations at regularly scheduled meal times (Rose and Carlson, 1986).
Inadequate food intake in the field has been ascribed to poor ration palatability, menu boredom, inability to work on a full stomach, lack of water, decreased appetite due to increased exercise, lack of specific meal periods and time to prepare meals, anxiety due to field conditions, and intentional dieting (Popper et al., 1989; Rose and Carlson, 1986). In animals, it is thought that anorexia (reduced food intake even when food is readily available) may improve survival and competitive success during important activities such as defense against predators, seeking shelter from bad weather, migrating, and courtship (King and Murphy, 1985; Mrosovsky and Sherry, 1980). The universality of the voluntary anorexia seen in soldiers during field exercises suggests that a similar innate process may be occurring. Although it is difficult to quantify the adaptive value of anorexia to soldiers operating in a field environment, it is possible that anorexia helps soldiers adapt to demanding situations. A decrease in food intake may benefit soldiers by limiting postmeal impairments in the ability to maintain attention and react quickly (Smith and Miles, 1986). A decrease in the need to carry, prepare, and eat rations would decrease the energetic cost of load carriage and increase the amount of time and resources available for more important military tasks.
Does Altitude Exposure Influence Appetite?
Under resting conditions in rats, acute exposure to moderate and well-tolerated, high-altitude hypoxia inhibits not only food and salt appetite but also thirst, probably by independent but still not fully understood mechanisms (Behm et al., 1984, 1989; Fregly et al., 1976; Schnakenberg et al., 1971, 1973). Food and water intake are inhibited only in the first 1 to 4 days of well-tolerated hypoxia, whereas salt intake can be inhibited for a longer time. This picture might be modified by physical exercise and during extreme or badly tolerated hypoxia where the general aversion to food, water, and salt intake may simply reflect altitude illness rather than an adaptive physiologic mechanism. The reduced appetite and thirst with acute exposure to moderate, well-tolerated altitudes appears to support physiologic hypohydration and adaptation to acute hypoxia (Hoyt and Honig, 1996). The primary adaptive value of anorexia in the first days at high altitudes may be to reduce dietary salt intake, with the reduction in caloric intake as a secondary effect.
Is There Any Significant Effect of the Type of Ration on the Total Calories Consumed or the Percentage of Calories as Carbohydrate?
No effect of ration type on total caloric consumption was evident in a study of 28 Marines during a strenuous, 11-d cold-weather field exercise at 2,200 to 2,550 m (7,218 to 8,366 ft) elevation (Hoyt et al., 1991; Morgan et al., 1988) (Table 20-1). Energy expenditure, measured by the doubly labeled water method, was 4,919 ± 190 kcal/d (mean ± SD, n = 23). Three different ration types were issued: the standard Meal, Ready-to-Eat (MRE: 5,192 kcal/d, 644 g carbohydrate); the Ration, Cold Weather (RCW: 4,470 kcal/d, 661 g carbohydrate); and the Ration, Lightweight (RLW: 4,219 kcal/d, 400 g carbohydrate). Mean daily caloric intakes (MRE: 3,217 ± 285 kcal/d, n = 8; RCW: 2,892 ± 326 kcal/d, n = 10; RLW: 3,205 ± 433 kcal/d, n = 10) and the amount of carbohydrate consumed (MRE: 367 ± 34 g/d; RCW: 410 ± 47 g/d; RLW: 345 ± 47 g/d) did not differ significantly among the three ration groups. The percent contribution of carbohydrates to total calories consumed ranged from 43 go 57 percent (MRE: 46 percent; RCW: 57 percent; RLW: 43 percent). Neglecting modest body carbohydrate reserves, the percent contribution of dietary carbohydrate to total energy expenditure was only 28 to 33 percent. Significant differences in carbohydrate consumption between the MREs, RLWs, and the higher-carbohydrate RCW ration may have been evident if a larger group of volunteers had been studied. In any case, the RCW ration barely met the recommended minimum carbohydrate consumption of 400 g/d (IOM, 1992). A carbohydrate supplement is probably needed to meet the carbohydrate demands of physically active soldiers.
What is the Ideal Macronutrient Composition for a High-Altitude Diet?
Under resting conditions during the first 1 to 3 days at well-tolerated altitudes, subjects should eat a diet poor in salt and rich in carbohydrates. Drinking water should be freely consumed to foster physiologic natriuresis. Advantages of a carbohydrate-rich diet include oxidative end products, carbon dioxide and water, which have little impact on renal function, and an energy yield per liter of oxygen higher than that of fat (Lusk, 1928). After the initial period of adaptation to hypoxia, subjects should resume eating a normal, sea-level diet. Thus pure water, salt-free fluid, tea, or coffee should be readily available and consumption encouraged or even required.
Sources of Metabolic Fuel
Body Fat and Carbohydrate Reserves
Although soldiers normally do not consume enough food to meet energy demands in the field, they generally have substantial body fat available to meet the deficit. For example, a typical young male soldier weighing 74 kg has approximately 13.5 kg of body fat (Fitzgerald et al., 1986). This is equivalent to about 81,000 kcal, assuming that body fat energy density is 7,700 kcal/kg and that a minimum of 4 percent body fat or about 3 kg of fat is needed for normal physiological function (Friedl et al., 1994). This fat energy reserve, which constitutes approximately 98 percent of the body's energy reserve (Sahlin, 1986), is enough energy to meet a 2,000 kcal energy deficit per day for over a month.
In contrast to the large fat energy reserves, body carbohydrate reserves constitute only around 2 percent of the body's energy reserves and are readily depleted in the absence of adequate dietary carbohydrate intake (Sahlin, 1986). If the total daily energy expenditure of a soldier during a mountain operation is 4,000 kcal/d with an average respiratory exchange ratio of 0.85, a carbohydrate combustion of about 500 g/d would be expected. However, carbohydrate intake in the field is typically around 300 g/d (Table 20-1) (Hoyt et al., 1991, 1994a), far short of the minimum 400 g per soldier per day needed for a reasonable rate of glycogen resynthesis (IOM, 1992). When carbohydrate reserves are depleted there is a switch to a fat-predominant fuel metabolism characterized by decreased physical performance (Costill, 1988; Phinney et al., 1983) and loss of lean body mass (Askew et al., 1987b). Easily consumed carbohydrate beverages (Askew et al., 1987a) or high-carbohydrate supplements (Edwards et al., 1989) are needed to boost carbohydrate intake to the recommended minimum of 400 g per soldier per day (IOM, 1992).
Does Altitude Exposure Alter Metabolic Fuel Sources for Work at High Altitudes?
The high rates of energy expenditure and large energy deficits during field training exercises at altitude (Hoyt et al., 1991, 1994a) can result in an acute shift from a carbohydrate- to a fat-predominant fuel metabolism (Felig and Wahren, 1975; Stein et al., 1989). Body fat reserves meet fat energy needs, but carbohydrate deficits shift fuel metabolism from carbohydrate to fat. The time course of this transition depends on the subject's ![]() o2max, physical activity, and diet (Ahlborg and Felig, 1982; Felig and Wahren, 1975; Stein et al., 1989). Exercise- or diet-induced transitions to a fat-predominant metabolism are usually associated with muscle glycogen depletion and a reduction in maximum sustainable exercise intensity (Hultman, 1967; Phinney et al., 1980). In contrast, exercise after altitude acclimatization is associated with greater endurance exercise capacity, less muscle glycogen utilization (Young et al., 1982), and less muscle lactate accumulation (Green et al., 1989) than exercise of the same relative intensity at sea level.
o2max, physical activity, and diet (Ahlborg and Felig, 1982; Felig and Wahren, 1975; Stein et al., 1989). Exercise- or diet-induced transitions to a fat-predominant metabolism are usually associated with muscle glycogen depletion and a reduction in maximum sustainable exercise intensity (Hultman, 1967; Phinney et al., 1980). In contrast, exercise after altitude acclimatization is associated with greater endurance exercise capacity, less muscle glycogen utilization (Young et al., 1982), and less muscle lactate accumulation (Green et al., 1989) than exercise of the same relative intensity at sea level.
Can Specific Fuel Requirements Be Quantified?
The specific macronutrient requirements of physically active soldiers cannot be quantified without detailed and accurate information on the intensity and duration of work performed. Little information of this kind is currently available. However, recent findings suggest new approaches to this problem.
First, a new ambulatory monitor has been developed and validated that accurately estimates the metabolic cost of human locomotion from total body weight and the time during each stride that a single foot contacts the ground (Hoyt et al., 1994b; Kram and Taylor, 1990; Taylor, 1985). This approach is based on the fact that the rate of metabolic energy expenditure during walking or running is primarily determined by the cost of supporting body weight and the rate at which this force is generated (Kram and Taylor, 1990). Thus, the rate of force generation can be estimated as total body weight divided by the time during each stride that a single foot was in contact with the ground (Hoyt et al., 1994b; Kram and Taylor, 1990).
A second, related finding is that soldiers performing similar tasks in mountainous terrain have energy expenditures proportional to their total weight (Hoyt et al., 1994a). Differences in fat-free mass and total weight among the subjects explained 89 and 95 percent of the variance in total daily energy expenditure. This variance is consistent with previous studies showing a correlation between total daily energy expenditure and fat-free mass or total weight in subjects with similar activity patterns (Cunningham, 1991; Schoeller and Van Santen, 1982).
These and other findings suggest that energy expenditure patterns of soldiers during field exercises can be accurately estimated from a soldier's total weight, foot contact time, and the nature of the terrain (footing, grade) (Hoyt et al., 1994b; Pandolf et al., 1977). Field studies are planned to test whether macronutrient requirements of physically active soldiers can be quantified using this approach.
AUTHORS' CONCLUSIONS AND RECOMMENDATIONS
Soldiers participating in field training exercises, particularly those in mountainous terrain, are characteristically in negative energy balance. This energy deficit is attributable to the large portion of the day spent in physical activity, heavy work loads, increased basal energy requirements at altitude, and limited consumption of field rations. Food and salt intake also appear to be inhibited by the physiologic processes associated with adaptation to altitude. Although substantial body fat reserves are available to meet the fat energy deficit, carbohydrate intake falls short of the minimum 400 g per soldier per day recommended by the Committee on Military Nutrition Research (IOM, 1992).
The following recommendations are made:
- Formally incorporate easily consumed liquid and/or solid carbohydrate supplements into the military ration system.
- Investigate whether restricting sodium intake while promoting carbohydrate and water consumption facilitates adaptation to acute hypoxia.
- Test whether macronutrient requirements in the field can be accurately estimated from a soldier's total weight, speed of locomotion, and nature of the terrain.
REFERENCES
Ahlborg, G., and P. Felig 1982 Lactate and glucose exchange across the forearm, legs and splanchnic bed during and after prolonged leg exercise. J. Clin. Invest. 69:45–54.
Askew, E.W., J.R. Claybaugh, G.M. Hashiro, W.S. Stokes, A. Sato, and S.A. Cucinell 1987a Mauna Kea III: Metabolic effects of dietary carbohydrate supplementation during exercise at 4,100 m altitude. Technical Report T12-87. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine
Askew, E.W., I. Munro, M.A. Sharp, S. Siegel, R. Popper, M. Rose, R.W. Hoyt, J.W. Martin, K. Reynolds, H.R. Lieberman, D. Engell, and C.P. Shaw 1987b Nutritional status and physical and mental performance of Special Operations soldiers consuming the Ration, Lightweight or the Meal, Ready-to-Eat military field ration during a 30-day field training exercise. Technical Report T7-87. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine .
Behm, R., A. Honig, M. Griethe, M. Schmidt, and P. Schneider 1984 Sustained suppression of voluntary sodium intake of spontaneously hypertensive rats (SHR) in hypobaric hypoxia. Biomed. Biochem. Acta 43:975–985.
Behm, R., B. Gerber, J-O. Habeck, C. Huckstorf, and K. Ruckborn 1989 Effect of hypobaric hypoxia and almitrine on voluntary salt and water intake in carotid body denervated spontaneously hypertensive rats. Biomed. Biochem. Acta 48:689–695.
Bisdee, J.T., W.P.T. James, and M.A. Shaw 1989 Changes in energy expenditure during the menstrual cycle. Br. J. Nutr. 61:187–199.
Boutellier, U., D. Dériaz, P.E. Di Prampero, and P. Cerretelli 1990 Aerobic performance at altitude: Effects of acclimatization and hematocrit with reference to training. Int. J. Sports Med. 11(suppl. 1):S21–S26.
Butterfield, G.E., J. Gates, S. Fleming, G.A. Brooks, J.R. Sutton, and J.T. Reeves 1992 Increased energy intake minimizes weight loss in men at high altitude . J. Appl. Physiol. 72:1741–1748.
Costill, D.L. 1988 Carbohydrates for exercise: Dietary demands for optimal performance. Int. J. Sports Med. 9:1–18.
Cunningham, J.J. 1991 Body composition as a determinant of energy expenditure: A synthetic review and a proposed general equation. Am. J. Clin. Nutr. 54:963–969.
Davies, C.T.M., and M.W. Thompson 1979 Aerobic performance of female marathon and male ultramarathon athletes. Eur. J. Appl. Physiol. 41:233–245.
De Boer, J.O., A.J.H. van Es, J.E. Vogt, J.M.A. van Raiij, and J.G.A.J. Haustvast 1987 Reproducibility of 24 h expenditure measurements using a human whole body indirect calorimeter. Br. J. Nutr. 57:201–209.
Edwards, J.S.A., D.E. Roberts, T.E. Morgan, and L.S. Lester 1989 An evaluation of the nutritional intake and acceptability of the Meal, Ready-to-Eat consumed with and without a supplemental pack in a cold environment. Technical Report T18-89. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
FAO/WHO/UNU (Food and Agriculture Organization/World Health Organization/United Nations University) 1985 Energy and Protein Requirements. Report of a Joint FAO/WHO/UNU Expert Consultation. WHO Technical Report Series No. 724. Geneva: World Health Organization.
Felig, P., and J. Wahren 1975 Fuel homeostasis in exercise. N. Engl. J. Med. 293:1078–1084.
Fitzgerald, P.I., J.A. Vogel, W.L. Daniels, J.E. Dziados, M.A. Teves, R.P. Mello, and P.J. Reich 1986 The body composition project: A summary report and descriptive data. Technical Report T5-87. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
Forbes-Ewan, C.H., B.L.L. Morrissey, G.C. Gregg, and D.R. Waters 1989 Use of doubly labeled water technique in soldiers training for jungle warfare. J. Appl. Physiol. 67:14–18.
Fregly, M.J., E.L. Nelson, and P.E. Tyler 1976 Water exchange in rats exposed to cold, hypoxia, and both combined. Aviat. Space Environ. Med. 47:600–607.
Friedl, K.E., R.J. Moore, L.E. Martinez-Loez, J.A. Vogel, E.W. Vogel, L.J. Marchitelli, R.W. Hoyt, and C.C. Gordon 1994 Lower limit of body fat in health active men. J. Appl. Physiol. 77(2):933–940.
Fulco, C.S., and A. Cymerman 1988 Human performance and acute hypoxia. Pp. 467–495 in Human Performance Physiology and Environmental Medicine at Terrestrial Extremes, K.B. Pandolf, M.N. Sawka, and R.R. Gonzalez, eds. Indianapolis, Ind.: Benchmark Press.
Gill, M.B., and L.G.C.E. Pugh 1964 Basal metabolism and respiration in men living at 5,800 m (19,000 ft). J. Appl. Physiol. 19:949–954.
Green, H.J., J.R. Sutton, P.M. Young, A. Cymerman, and C.S. Houston 1989 Operation Everest II: Muscle energetics during maximal exhaustive exercise. J. Appl. Physiol. 66:142–150.
Grover, R.F. 1963 Basal oxygen uptake of man at high altitude. J. Appl. Physiol. 18:909–912.
Grover, R.F, J.V. Weil, and J.T. Reeves 1986 Cardiovascular adaptation to exercise at high altitude. Pp. 269–302 in Exercise and Sport Sciences Reviews, K.B. Pandolf, ed. New York: Macmillan.
Hannon, J.P., and D.M. Sudman 1973 Basal metabolic and cardiovascular function of women during altitude acclimatization. J. Appl. Physiol. 34:471–477.
Horstman, D., R. Weiskopf, and R.E. Jackson 1980 Work capacity during a 3-week sojourn at 4,300 m: Effects of relative polycythemia. J. Appl. Physiol. 49:311–318.
Hoyt, R.W., and A. Honig 1996 Body fluid and energy metabolism at high altitude. Pp. 1277–1289 in Handbook of Physiology, Section 4: Environmental Physiology, C.M. Blatteis and M.J. Fregly, eds. New York: Oxford University Press for the American Physiological Society.
Hoyt, R.W., T.E. Jones, T.P. Stein, G. McAninch, H.R. Lieberman, E.W. Askew, and A. Cymerman 1991 Doubly labeled water measurement of human energy expenditure during strenuous exercise. J. Appl. Physiol. 71:16–22.
Hoyt, R.W., T.E. Jones, C.J. Baker-Fulco, D.A. Schoeller, R.B. Schoene, R.S. Schwartz, E.W. Askew, and A. Cymerman 1994a Doubly labeled water measurement of human energy expenditure during exercise at high altitude. Am. J. Physiol. 266:R966–R971.
Hoyt, R.W., J.J. Knapik, J.F. Lanza, B.H. Jones, and J.S. Staab 1994b Ambulatory foot contact monitor to estimate the metabolic cost of human locomotion. J. Appl. Physiol. 76:1818–1822.
Huang, S.Y., J.K. Alexander, R.F. Grover, J.T. Maher, R.E. McCullough, R.G. McCullough, L.G. Moore, J.V. Weil, J.B. Sampson, and J.T. Reeves 1984 Increased metabolism contributes to increased resting ventilation at high altitude. Respir. Physiol. 57:377–385.
Hultman, E. 1967 Physiological role of muscle glycogen in man, with special reference to exercise. Cric. Res. 20–21(suppl. 1):99–114.
IOM (Institute of Medicine) 1992 Letter report: Calorie-dense rations. In Committee on Military Nutrition Research Activity Report 1986–1992. A report of the Committee on Military Nutrition Research, Food and Nutrition Board. Washington, D.C.: National Academy Press.
Johnson, H.L., C.F. Consolazio, T.A. Daws, and H.J. Krzywicki 1971 Increased energy requirements of man after abrupt altitude exposure. Nutr. Rep. Int. 4:77–82.
Jones, T.E., R.W. Hoyt, C.J. Baker, C.B. Hintlian, P.S. Walczak, R.A. Kluter, C.P. Shaw, D. Shilling, and E.W. Askew 1990 Voluntary consumption of a liquid carbohydrate supplement by Special Operations Forces during a high altitude cold weather field training exercise. Technical Report T20-90. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
King, J.R., and M.E. Murphy 1985 Periods of nutritional stress in annual cycles of endotherms: Fact or fiction? Am. Zool. 25:955–964.
Kram, R., and C.R. Taylor 1990 Energetics of running: A new perspective. Nature 346(6281):265–267.
Leulier, J. 1954 Le métabolisme de bas et les dépenses d'oxygene en haute montagne. Concours Med. 76:4865–4866.
Lusk, G. 1928 The Elements of the Science of Nutrition, 4th ed. New York: Academic Press. Reprint, New York: Johnson Reprint Corp., 1976.
Maher, J.T., L.G. Jones, and L.H. Hartley 1974 Effects of high-altitude exposure on submaximal endurance capacity of men. J. Appl. Physiol. 37:895–898.
Meda, di E. 1955 Dispendio di base dell'uomo in montagna. Arch. di E. Fisiolo. 55:144–151.
Morgan, T.E., L.A. Hodgess, D. Schilling, R.W. Hoyt, E.J. Iwanyk, G. McAninch, T.C. Wells, V.S. Hubbard, and E.W. Askew 1988 A comparison of the Meal, Ready-to-Eat, Ration, Cold Weather, and Ration, Lightweight nutrient intakes during moderate altitude cold weather field training operations. Technical Report T5-99. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
Mrosovsky, N., and D.F. Sherry 1980 Animal anorexias. Science 207:837–842.
Nair, C.S., M.S. Malhotra, and P.M. Gopinath 1971 Effect of altitude and cold acclimatization on the basal metabolism in man. Aerosp. Med. 42:1056–1059.
Pandolf, K.B., B. Givoni, and R.F. Goldman 1977 Predicting energy expenditure with loads while standing or walking very slowly. J. Appl. Physiol. 43:577–581.
Phinney, S.D., E.S. Horton, E.A.H. Sims, J.S. Hanson, E. Danforth, Jr., and B.M. LaGrange 1980 Capacity for moderate exercise in obese subjects after adaptation to a hypocaloric, ketogenic diet. J. Clin. Invest. 66:1152–1161.
Phinney, S.D., B.R. Bistrian, R.R. Wolfe, and G.L. Blackburn 1983 The human metabolic response to chronic ketosis without caloric restriction: Physical and biochemical adaptation. Metabolism 32:757–768.
Popper, R., G. Smits, G., H.L. Meiselman, and E. Hirsch 1989 Eating in combat: A survey of U.S. Marines. Milit. Med. 154:619–623.
Rose, M.S., and D.E. Carlson 1986 Effects of A Ration meals on body weight during sustained field operations. Technical Report T/2-87. Natick, Mass.: U.S. Army Research Institute of Environmental Medicine.
Sahlin, K. 1986 Metabolic changes limiting muscle performance. Pp. 323–344 in Biochemistry of Exercise VI, B. Saltin, ed. International Series on Sports Sciences, vol. 16. Champaign, Ill.: Human Kinetics Publishers.
Schnakenberg, D.D., L.F. Krabill, and P.C. Weiser 1971 The anorexic effect of high altitude on weight gain, nitrogen retention and body composition of rats. J. Nutr. 101:787–796.
Schnakenberg, D.D., P.M.B. Leung, R.A. Freedland, and Q.R. Rogers. 1973 Separation of hypophagic and hypodipsic responses to acute hypoxia exposure [abstract]. Fed. Proc. 32:362.
Schneider, E.C., D. Truesdell, and R.W. Clarke 1924 Oxygen consumption in men during short exposures to low barometric pressures. Am. J. Physiol. 70:283–299.
Schoeller, D.A., and E. Van Santen 1982 Measurement of energy expenditure in humans by doubly labeled water method. J. Appl. Physiol. 53:955–959.
Smith, A.P., and C. Miles 1986 Effects of lunch on selective and sustained attention. Neuropsychobiology 16:117–120.
Stein, T.P., R.W. Hoyt, R.G. Settle, M. O'Toole, and W.D.B. Hiller 1987 Determination of energy expenditure during heavy exercise, normal daily activity, and sleep using the doubly-labeled-water (2H2180) method . Am. J. Clin. Nutr. 45:534–539.
Stein, T.P., R.W. Hoyt, M. O'Toole, M.J. Leskiw, M.D. Schluter, R.R. Wolfe, and W.D.B. Hiller 1989 Protein and energy metabolism during prolonged exercise in trained athletes. Int. J. Sports Med. 10:311–316.
Stock, M.J., N.G. Norgan, A. Ferro-Luzzi, and E. Evans 1978 Effect of altitude on dietary-induced thermogenesis at rest and during light exercise in man. J. Appl. Physiol. 45:345–349.
Surks, M.I., H.J. Beckwitt, and C.A. Chidsey 1967 Changes in plasma thyroxine concentration and metabolism, catecholamine excretion and basal oxygen consumption in man during acute exposure to high altitude. J. Clin. Endocrinol. 27:789–799.
Taylor, C.R. 1985 Force development during sustained locomotion: A determinant of gait, speed and metabolic power. J. Exp. Biol. 115:253–262.
Terzioglu, M., and R. Aykut 1954 Variations in basal metabolic rate at 1.85 km altitude. J. Appl. Physiol. 7:329–332.
Webb, P. 1986 24–hour energy expenditure and the menstrual cycle. Am. J. Clin. Nutr. 44:614–619.
Westerterp, K.R., W.H.M. Saris, M. Van Es, and F. ten Hoor 1986 Use of the doubly labeled water technique in humans during sustained heavy exercise. J. Appl. Physiol. 61:2162–2167.
Westerterp, K.R., F. Brouns, W.H.M. Saris, and F. ten Hoor 1988 Comparison of doubly labeled water with respirometry at low and high-activity levels. J. Appl. Physiol. 65:53–56.
Young, A.J., and P.M. Young 1988 Human acclimatization to high terrestrial altitude. Pp. 467–495 in Human Performance Physiology and Environmental Medicine at Terrestrial Extremes, K.B. Pandolf, M.N. Sawka, and R.R. Gonzalez, eds. Indianapolis, Ind.: Benchmark Press.
Young, A.J., W.J. Evans, A. Cymerman, K.B. Pandolf, J.J. Knapik, and J.T. Maher 1982 Sparing effect of chronic high-altitude exposure on muscle glycogen utilization. J. Appl. Physiol. 52:857–862.
| This page in the original is blank. |














