23
Food Components and Other Treatments That May Enhance Mental Performance at High Altitudes and in the Cold
Harris R. Lieberman1 and Barbara Shukitt-Hale
INTRODUCTION
High-altitude exposure that results in hypobaric hypoxia can produce significant decrements in human mental performance (see Shukitt-Hale and Lieberman, Chapter 22 in this volume). Although these adverse effects are reduced by the acclimatization process, only partial adaptation occurs at extreme terrestrial altitudes. Furthermore, when soldiers are rapidly deployed to mountainous regions, their performance of required critical duties upon arrival, including offensive or defensive operations, may be impaired. The adverse effects of cold exposure on human behavior can also be significant and can exacerbate the physical consequences of exposure to extreme cold. Because mountainous regions are frequently cold, the adverse effects of both stressors on soldier performance—cold and high altitudes—may be additive. Cold and hypoxia are not the only stressors that soldiers experience. Other common
stressors include heat, psychological stress (which can be extraordinarily intense in combat operations), and sleep loss. Because soldiers are typically exposed to multistressor environments, the optimal treatment for any individual stressor might also have beneficial effects that generalize across other stressors. However, there is no reason to believe that if a treatment is effective in one environment it will also work in others.
Cold, heat, and hypoxia have different physiological effects. In the case of heat and cold, some of the effects, at least on the peripheral nervous system, are in opposition (Murphy and Redmond, 1975). Therefore, it may be likely that treatments for the adverse peripheral consequences of exposure to various stressors will not have common positive effects across stressors. In fact, treatments that have beneficial effects in one environment could even have adverse effects in other environments. It is also possible however, that enhancement of certain critical metabolic or physiological functions such as energy utilization or cardiovascular function would generalize across different stressors. Appropriate nutritional supplements might, in theory, be good candidates for producing beneficial effects in various environments and in situations in which multistressor exposure occurs. In addition, if there are common elements in the response of the central nervous system (CNS) to different types of stressors, modulation of these mechanisms could be a generic treatment for the adverse effects of stress. Treatments that are less stressor-specific might have significant advantages over strategies that are targeted at specific metabolic functions associated with individual stressors.
If it can be determined what underlying brain mechanisms are responsible for the adverse CNS response to stress, assuming there are some, it may be possible to find practical interventions—nutritional or other—to treat some of the adverse symptoms associated with the CNS components of the stress response. At the U.S. Army Research Institute of Environmental Medicine (USARIEM), a comprehensive program has been initiated to evaluate the neurochemical and behavioral effects of various stressors on brain function. One of the key objectives of this program is to specify which neurochemical changes are responsible for the adverse behavioral effects of stressors. With this basic information, researchers at USARIEM hope to determine how to treat adverse behavioral effects of stress appropriately by preventing or mitigating the underlying neurochemical deficits with nutrient and drug treatment. To date, two environmental stressors have been studied in some detail: cold and high altitude. Researchers hope to extend the research to various other stressors. In addition, two neurochemical systems have been studied that are believed to be intimately involved either in the response of the organism to stress (specifically the catecholaminergic system), or in critical behavioral functions such as learning and memory (specifically the cholinergic system).
STRESS AND CENTRAL CATECHOLAMINES
A great deal of evidence from the animal literature, and to a limited extent from the human clinical literature, supports the idea that there are common elements, across a variety of stressors, of the CNS response to acute stress (Gray, 1982). Furthermore, this response seems to have some common neurochemical substrates and involves, in particular, the activation of catecholaminergic systems (Lieberman, 1994; Murphy and Redmond, 1975; Stone 1975). An increase in the release of specific brain neurotransmitters may initially lead to an increased ability to cope, but if the stress is severe enough, these homeostatic regulatory mechanisms can be overwhelmed. When this situation occurs, the organism cannot effectively respond to the environment, and a syndrome characterized by lethargy and helplessness results (Gray, 1982; Maier and Seligman, 1976). A more detailed discussion of this hypothesis and the supporting evidence is provided by Wurtman and Lieberman (1989) and Lieberman (1994).
Central catecholaminergic stores are depleted by exposure to various types of stress, including both environmental and psychological stressors. Heat, cold, and electrical shock all produce significant increases in brain catecholaminergic activity (Stone, 1975). The neurotransmitter norepinephrine (NE) appears to be particularly critical in the brain's response to stress, although the other important brain catecholamine, dopamine, also appears to be involved. It is probably not coincidental that the classic performance-enhancing drugs are catecholamine (CA-) agonists that seem to have beneficial effects on a variety of stressors. The most widely recognized CA-agonist is the potent drug amphetamine, which also has beneficial effects on certain types of performance in unstressed organisms (Gray, 1982; Murphy and Redmond, 1975).
Tyrosine is a nutrient precursor of the catecholamines, and therefore, it has been studied as part of the Performance Enhancing Ration Component Program, a joint U.S. Army Research Institute of Environmental Medicine/Natick Research Development and Engineering Center program. Tyrosine appears, in certain situations, to enhance catecholaminergic function and to prevent certain stress-induced decrements in performance (Lieberman, 1994). Such beneficial effects were predicted because when catecholaminergic neurons are highly active (which occurs during exposure to many acute stressors), the amount of neurotransmitter they release increases if more tyrosine is made available (Wurtman et al., 1981). Because increased catecholamine release appears to be associated with an effective response of the organism to acute stress, the provision of supplemental tyrosine (precursor for the synthesis of catecholamines) might enhance the ability of the organism to respond appropriately to stress (Lehnert et al., 1984; for reviews see Lieberman, 1994; Owasoyo et al., 1992; Salter, 1989).
Since the mid-1980s, a series of animal and human studies have been conducted at USARIEM and elsewhere to examine this phenomenon, because
the results could have practical implications for treatment of stress-induced decrements of mental performance and even ration design (Lieberman, 1994). As part of this program, the authors have employed various techniques to assess changes in animal brain function and in animal and human behavior associated with acute stress. Furthermore, the authors have studied the effects of tyrosine administration on neurotransmitter release and behavior in acutely stressful scenarios.
Microdialysis is one of the techniques used at USARIEM to examine the effects of various stressors on brain function. This technique permits continuous monitoring of neurotransmitter release in vivo (Ungerstedt et al., 1982) by sampling extracellular fluid from a small brain region to assess neurotransmitter and metabolite levels. A specially manufactured guide cannula is implanted stereotaxically onto the skull of an anesthetized animal and above a specific brain region of interest. After the animal has recovered from the effects of the surgery and anesthesia, a microdialysis probe can be inserted through the cannula into the predesignated brain region. The probe consists of two separate inflow and outflow tubes connected at the end by a closed loop of dialysis membrane. The tip of the probe is typically 1 to 3 mm in length. When the probe is inserted into the brain, it is perfused with artificial cerebrospinal fluid (CSF) at a flow rate of 2 µl/min. Although no fluid is added or removed from the brain, by perfusing the probe with a solution that does not contain brain metabolites, a concentration gradient is established across the dialysis membrane. Therefore, brain metabolites move from the brain extracellular space across the dialysis membrane into the artificial CSF due to this gradient. The collected artificial CSF is then assayed for the concentration of the brain neurotransmitter(s) and metabolites of interest. It typically takes 10 to 20 minutes to collect a sample large enough for analysis. For assay of brain catecholamines, high performance liquid chromatography with electrochemical detection (Lehnert et al., 1994) is the method of choice.
Using this technique, researchers at USARIEM have assessed NE release during exposure of rats to cold water stress and to restraint stress and have reported substantial changes in levels of hippocampal NE (Luo et al., 1993). Microdialysis guide cannulae have been implanted in Fischer rats to assess the release of NE in the hippocampus. This brain region plays a key role in learning and memory function. In addition, NE collected in the hippocampus is released only by afferents from the locus coeruleus, the only brain region providing noradrenergic input to the hippocampus. The locus coeruleus is a very small but critical region of the brain, which plays an important role in regulating aspects of the acute response of animals to stressors (Stone, 1975). This midbrain region provides noradrenergic innervation to large areas of the brain, including the cerebral cortex, and probably plays an important role in the regulation of arousal level and attention (Gray, 1982).
The rats tested in this study were treated with either 400 mg of tyrosine per kg body weight, administered intraperitoneally (i.p.), or with a saline
placebo treatment (Luo et al., 1993) (Figure 23-1). Simply exposing the saline-treated control animals to 10 minutes of restraint stress caused a massive increase in the release of NE, greater than 100 percent, as assessed by in vivo microdialysis (Figure 23-1). However, an additional stressor, cold, begun 30 minutes into the experiment, caused no additional increase in NE release. Treatment with tyrosine increased the release of NE over 500 percent above baseline levels, 20 minutes after the beginning of exposure to stress. Higher rates of NE release were maintained in tyrosine-treated animals throughout the exposure to the multiple stressors. It appears that by supplying additional neurotransmitter precursor to the brain by peripheral administration of tyrosine, which readily crosses the blood-brain barrier, substantial additional NE is released during exposure to acute stress.
Changes in the release of particular brain neurotransmitters, while of theoretical interest, are by themselves difficult to interpret. Because an increase in neurotransmitter release could either have positive or negative behavioral consequences, it is essential to study the behavioral correlates of neurochemical phenomena. Several laboratories have conducted such studies (for a review see Lieberman, 1994), including the authors'. In particular, this lab has attempted to determine whether tyrosine administration has beneficial behavioral effects when animals or humans are exposed to various stressors, alone and in combination.
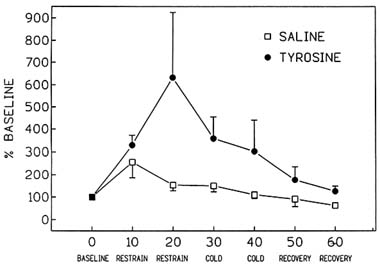
FIGURE 23-1 Effect of immobilization stress alone and in combination with cold stress on hippocampal norepinephrine release in tyrosine- (400 mg/kg; intraperitoneally [i.p.]) and placebo-treated rats. Testing conditions, in 10-min intervals, are specified on the x-axis. SOURCE: Adapted from Luo et al. (1993).
In a series of animal studies, the authors have attempted to develop a paradigm that consistently detects the effects of tyrosine on stress-related coping behavior. The test employed, the Porsolt swim test, was previously reported to be sensitive to other catecholaminergic agonists and has been used as a screening test to help in the selection of new antidepressant drugs (Porsolt et al., 1978). The Porsolt swim test was also reported to detect the effects of various neurotransmitter precursors, including tyrosine, in relatively nonstressed animals (Gibson et al., 1982). The test is simple to administer; a rat is placed in a glass beaker filled with water for several minutes, and the amount of time it spends actively attempting to escape is assessed. An animal who is actively attempting to escape is judged to be more appropriately responding to the environment than one who is inactive and helpless. Of course, other behavioral tests are necessary to confirm and extend the results of the Porsolt test, but it has usually proven to be a reliable indicator of the potency and selectivity of many experimental treatments (Porsolt et al., 1978). Furthermore, the authors have found it to be consistently sensitive to the effects of tyrosine on animals exposed to acute cold stress (Luo et al., 1992; Rauch and Lieberman, 1990). In one study, tyrosine, when administered in a dose of 400 mg/kg, significantly increased the amount of time animals were active in the Porsolt swim test following a brief exposure to cold water stress (Rauch and Lieberman, 1990). More recently, the authors found that tyrosine in lower doses (200 mg/kg) also improved performance in the Porsolt test. As would be predicted, tyrosine is less potent when administered in this dose than the higher dose (Luo et al., 1992) (Figure 23-2).
The authors have also seen beneficial effects of tyrosine using behavioral tasks that require more cognitive performance than the Porsolt swim test. For example, a recently conducted series of studies examined some of the neurochemical, behavioral, and morphologic consequences of acute and chronic exposure to hypobaric hypoxia (Shukitt-Hale and Lieberman, Chapter 22 in this volume; Shukitt-Hale et al., 1993, 1994, in press a, in press b). Unfortunately, the authors were unable to assess noradrenergic function during exposure to hypobaric hypoxia because NE release fell to undetectable levels soon after the start of exposure. However, it was possible to study cholinergic function using microdialysis. When rats were exposed to a simulated altitude of 18,000 ft (5,486 m), hippocampal acetylcholine levels significantly decreased (Shukitt-Hale et al., 1993) (Figure 23-3). Exposure to an altitude of 19,500 ft (5,944 m) also adversely affected learning and memory (Shukitt-Hale et al., 1994). However, upon administering a drug (the calcium channel blocker, nimodipine), which restored acetylcholine to normal levels (Figure 23-3), performance on a test of learning and memory (the Morris water maze [MWM]) was not restored (Unpublished observations, B. Shukitt-Hale and H. R. Lieberman, U.S. Army Research Institute of Environmental Medicine, Natick, Mass., 1994).
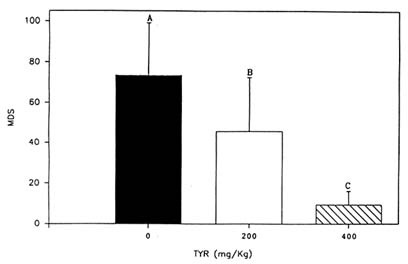
FIGURE 23-2 The effect of two doses of tyrosine (TYR) (200 and 400 mg/kg; i.p.) on duration of immobility in the Porsolt swim test. Prior to testing, core body temperature was lowered to 86°F (30°C). These data demonstrate that the effects of tyrosine on cold-stressed animals are dose dependent. Values are expressed as mean difference scores (MDS), which equal hypothermia immobility time minus normothermia immobility time. A lower score indicates better performance. Treatment means that are labeled with different letters are significantly different. Results are expressed as mean + SEM.
SOURCE: Adapted from Lieberman (1994) and Luo et al. (1992).
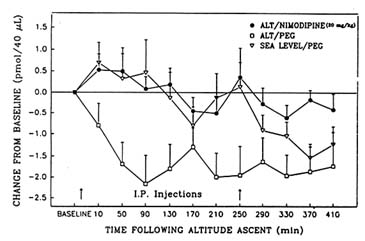
FIGURE 23-3 Changes in extracellular hippocampal acetylcholine levels under three conditions: ALT/NIMODIPINE, a simulated altitude of 18,000 ft (5,486 m) and treatment with the drug nimodipine (20 mg/kg); ALT/PEG, an altitude of 18,000 ft and treatment with a placebo; SEA LEVEL/PEG, sea level altitude and treatment with a placebo. Arrows above the x-axis indicate times of drug or placebo injection. SOURCE: Adapted from Shukitt-Hale et al. (1993).
The authors have also conducted a study in which tyrosine was administered twice (400 mg/kg on each occasion) to rats exposed to hypobaric hypoxia, with the result that it significantly improved spatial learning performance on the MWM (Lieberman et al., 1992; Shukitt-Hale et al., in press b) (Figure 23-4). These studies support the conclusion that, at least for some of the behavioral symptoms of hypobaric hypoxia, the cholinergic system may play a less-important role or may be more difficult to modify than the catecholaminergic system. That beneficial effects of tyrosine were detectable by very different behavioral tests, such as the MWM and the Porsolt swim test, is encouraging with regard to the generalization of tyrosine's effects. Furthermore, that tyrosine's effects are present when animals are exposed to very different types of stress also supports its potentially comprehensive effects.
The authors have also conducted several studies to determine whether tyrosine protects humans from the adverse effects of the combined stressors of cold and high altitudes. In two separate but similar studies, tyrosine given orally in doses of 85 to 170 mg/kg protected humans from some of the adverse effects of these stressors on symptoms, mood, and performance (Banderet and Lieberman, 1989; Lieberman et al., 1990). In these studies, mood, performance, and symptoms associated with cold and hypoxia were partially mitigated
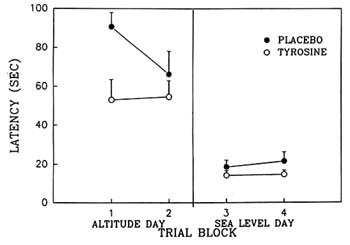
FIGURE 23-4 Effect of tyrosine and placebo on working (short-term) memory as assessed by the Morris water maze. Rats were exposed to hypobaric hypoxia (equivalent to 19,500 ft [5,944 m]) or sea-level conditions. Four hundred mg/kg of tyrosine was administered i.p. on two occasions: 1.5 hours and 5.5 hours after the start of exposure to hypoxia. Hypoxia clearly impaired performance on this task, and tyrosine reduced the decrements in performance as assessed by latency to locate the hidden platform on the first trial block. There were no residual effects of tyrosine on the next day (sea-level day). Results are expressed as mean + SEM. Tyrosine significantly different from placebo (p < 0.04). SOURCE: Adapted from Lieberman et al. (1992).
in many individuals who were treated with tyrosine. Research conducted by the Navy Medical Research Laboratory in humans and animals exposed to cold stress have confirmed and extended these findings (Ahlers et al., 1994; Shurtleff et al; 1992).
STRESS AND THE CHOLINERGIC SYSTEM
As noted above, the cholinergic system is considered to be especially critical with regard to regulation of certain types of learning and memory. To better understand the neurochemical and behavioral consequences of stress on this key brain system, this lab has studied the effects of several stressors on brain cholinergic function and behavior. As discussed above, hypobaric hypoxia was observed to cause substantial reductions in the release of acetylcholine in the rat hippocampus (Figure 23-3 above) and to impair learning and memory (Shukitt-Hale et al., 1993, 1994). The authors have also studied the effects of cold stress and restraint stress on hippocampal acetylcholine and choline levels. As shown in Figure 23-5, restraint stress causes significant reductions in acetylcholine release in the rat hippocampus. The combination of restraint and cold stress causes a somewhat larger reduction in acetylcholine release then restraint alone (Lieberman et al., 1994; Stillman et al., 1993). In addition, when only restraint stress is employed, there is a
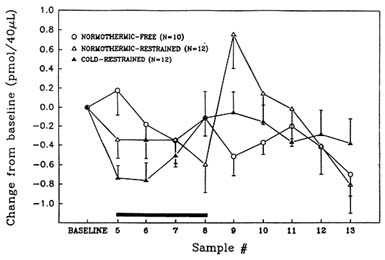
FIGURE 23-5 Effects of restraint stress alone and in combination with cold stress on hippocampal acetylcholine levels. An unrestrained normothermic group was also tested. Samples were taken every 20 minutes. Bar denotes duration of acute stress exposure for cold-restraint and normothermic-restraint conditions. SOURCE: Adapted from Lieberman et al. (1994) and Stillman et al. (1993).
large burst of acetylcholine release following removal from stress. This phenomenon does not occur in animals exposed to the more severe combination of both stressors. When learning and memory performance are assessed with the eight-arm radial maze after exposure to these stressors, animals exposed to the combination of stressors are the most severely impaired (Lieberman et al., 1994; Stillman et al., 1994) (Figure 23-6). These results support the idea that the surge in acetylcholine release among animals that are only exposed to a single stressor may contribute to behavioral recovery from stress exposure.
AUTHORS' CONCLUSIONS
As noted above, exposure to different stressors, particularly those as physiologically different in their consequences as hypoxia and cold stress, may require different types of treatment. However, if there are common CNS elements to the response of organisms to stress, common treatment strategies may be useful for a wide range of dissimilar stressors. For soldiers who are exposed to multiple stressors, a common approach to treating the adverse effects of stress would be particularly appropriate. Use of catecholaminergic agonists like tyrosine appear to be one such strategy with considerable promise. This laboratory and other research groups have reported in both animal
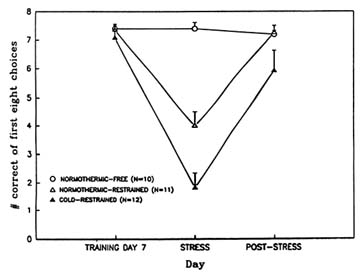
FIGURE 23-6 Effects of restraint stress alone and in combination with cold stress on learning as assessed with the eight-arm radial maze. A control, unstressed group was also tested. SOURCE: Adapted from Lieberman et al. (1994) and Stillman et al. (1993).
and human studies that tyrosine, a food constituent, has beneficial effects under conditions of hypoxia and cold, both separately and in combination. However, considerable basic research must be conducted to specify further the neurochemical mechanisms underlying various types of stress. In addition, behavioral studies that assess performance and symptoms associated with particular stressors and a variety of different treatment strategies are necessary. Whenever possible, such applied studies should be based on an understanding of the underlying neurochemical mechanisms responsible for the adverse effects of each stressor. In addition to tyrosine, certain other food constituents, particularly caffeine, should also be considered for their potential to treat some of the adverse effects of cold or altitude exposure.
REFERENCES
Ahlers, S.T., J.R. Thomas, J. Schrot, and D. Shurtleff 1994 Tyrosine and glucose modulation of cognitive deficits. Pp. 301–320 in Food Components to Enhance Performance, An Evaluation of Potential Performance-Enhancing Food Components for Operational Rations, B. Marriott, ed. A report of the Committee on Military Nutrition Research, Food and Nutrition Board, Institute of Medicine. Washington, D.C.: National Academy Press.
Banderet, L.E., and H.R. Lieberman 1989 Treatment with tyrosine, a neurotransmitter precursor, reduces environmental stress in humans. Brain Res. Bull. 22:759–762.
Gibson, C.J., S.M. Deikel, S.N. Young, and Y.M. Binik 1982 Behavioral and biochemical effects of tryptophan, tyrosine and phenylalanine in mice. Psychopharmacology 76:118–121.
Gray, J.A. 1982 The Neuropsychology of Anxiety: An Inquiry into the Functions of the Septo-hippocampal System. Oxford: Clarendon Press.
Lehnert, H.R., D.K. Reinstein, B.W. Strowbridge, and R.J. Wurtman 1984 Neurochemical and behavioral consequences of acute, uncontrollable stress: Effects of dietary tyrosine. Brain Res. 303:215–223.
Lieberman, H.R. 1994 Tyrosine and stress: Human and animal studies. Pp. 277–299 in Food Components to Enhance Performance, An Evaluation of Potential Performance-Enhancing Food Components for Operational Rations, B. Marriott, ed. A report of the Committee on Military Nutrition Research, Food and Nutrition Board, Institute of Medicine. Washington, D.C.: National Academy Press.
Lieberman, H.R., L.E. Banderet, and B. Shukitt-Hale 1990 Tyrosine protects humans from the adverse effects of acute exposure to hypoxia and cold. Abstr. Soc. Neuro. 16:272.
Lieberman, H.R., B. Shukitt-Hale, S. Luo, J.A. Devine, and J.F. Glenn 1992 Tyrosine reduces the adverse effect of hypobaric hypoxia on spatial working memory of the rat. Abstr. Soc. Neuro. 18(1):715.
Lieberman, H.R., M.J. Stillman, B. Shukitt-Hale, R.L. Galli, and A. Levy 1994 Acute cold and restraint stress alters hippocampal cholinergic neurotransmission and memory [abstract]. P. 59 in First World Congress on Stress Abstracts.
Luo, S., L. Villamil, C.J. Watkins, and H.R. Lieberman 1992 Further evidence tyrosine reverses behavioral deficits caused by cold exposure. Abstr. Soc. Neuro. 18(1):716.
Luo, S., E.T.S. Li, and H.R. Lieberman 1993 Tyrosine increases hypothermia-induced norepinephrine (NE) release in rat hippocampus assessed by in vivo microdialysis. Abstr. Soc. Neuro. 19:1452.
Maier, S.F., and M.E.P. Seligman 1976 Learned helplessness: Theory and evidence. J. Exp. Psychol. 105:3.
Murphy, D.L., and D.E. Redmond 1975 The catecholamines: Possible role in affect, mood and emotional behavior in man and animals. Pp. 73–117 in Catecholamines and Behavior, A.J. Freidhoff, ed. New York: Plenum Press.
Owasoyo, J.O., D.F. Neri, and J.G. Lamberth 1992 Tyrosine and its potential use as a countermeasure to performance decrement in military sustained operations. Aviat. Space Environ. Med. 63:364–369.
Porsolt, R.D., G. Anton, N. Blavet, and M. Jalfre 1978 Forced swimming in rats: Hypothermia, immobility and the effects of imipramine. Eur. J. Pharmacol. 47:379–391.
Rauch, T.M., and H.R. Lieberman 1990 Tyrosine pretreatment reverses hypothermia induced behavioral depression. Brain Res. Bull. 24:147–150.
Salter, C. 1989 Dietary tyrosine as an aid to stress resistance among troops. Milit. Med. 154:144–146.
Shukitt-Hale, B., M.J. Stillman, A. Levy, J.A. Devine, and H.R. Lieberman 1993 Nimodipine prevents the in vivo decrease in hippocampal extracellular acetylcholine produced by hypobaric hypoxia. Brain Res. 621:291–295.
Shukitt-Hale, B., M.J. Stillman, D.I. Welch, A. Levy, J.A. Devine, and H.R. Lieberman 1994 Hypobaric hypoxia impairs spatial memory in an elevation-dependent fashion. Behav. Neural Biol. 62:244–252.
Shukitt-Hale, B., T. Kadar, B.E. Marlowe, M.J. Stillman, R.L. Galli, A. Levy, J.A. Devine, and H.R. Lieberman In Morphological alterations in the hippocampus following hypobaric hypoxia. In Human Press a and Experimental Toxicology.
Shukitt-Hale, B., M.J. Stillman, and H.R. Lieberman In Tyrosine Administration prevents hypoxia-induced decrements in learning and memory. Press b In Physiol. Behav.
Shurtleff, D., J.R. Thomas, J. Schrot, K. Kowalski, R. Harford, M.O. Thornton, P.A. Shea, and M. Malik 1992 Tyrosine reverses a cold-stress-induced memory deficit in humans. Abstr. Soc. Neuro. 18(2):1210.
Stillman, M.J., B. Shukitt-Hale, A. Levy, and H.R. Lieberman 1993 Cold stress exposure alters hippocampal cholinergic neurotransmission. Abstr. Soc. Neuro. 19:1451.
Stillman, M.J., B. Shukitt-Hale, A. Levy, H.E. Modrow, and H.R. Lieberman 1994 Differential effects of acute stress exposure on radial arm maze performance. Abstr. Soc. Neuro. 20:1443.
Stone, E.A. 1975 Stress and catecholamines. Pp. 31–71 in Catecholamines and Behavior, A.J. Freidhoff, ed. New York: Plenum Press.
Ungerstedt, U., C. Forster, M. Herrera-Marschitz, I. Hoffman, U. Jungnelius, U. Tossman, and T. Zetterstrom 1982 Brain dialysis: A new in vivo technique for studying neurotransmitter release and metabolism. Neurosci. Lett. 10(suppl.):S493.
Wurtman, R.J., and H.R. Lieberman 1989 Use of tyrosine and other nutrients to protect against stress and to enhance and sustain performance. Pp. 67–77 in Stress: Neurochemical and Humoral Mechanisms, vol. 1, G.R. Van Loon, R. Kvetnansky, R. McCarty, and J. Axelrod, eds. New York: Gordon and Breach.
Wurtman, R.J., F. Hefti, and E. Melamed 1981 Precursor control of neurotransmitter synthesis. Pharmacol. Rev. 32:315–335
| This page in the original is blank. |














