22
The Effect of Altitude on Cognitive Performance and Mood States
Barbara Shukitt-Hale1 and Harris R. Lieberman
INTRODUCTION
High-altitude environments can be debilitating to unacclimatized individuals exposed to elevations above 3,000 m (9,843 ft) for periods ranging from several hours to days. Moderate hypoxia induces substantial alterations in physiological and psychological parameters within a few hours (Bahrke and Shukitt-Hale, 1993). Immediately upon ascent to high altitude, there is decreased blood oxygenation, which reduces the oxygen supply throughout the periphery and in the brain. With time the body compensates, at least in part, for the lack of oxygen with a variety of physiological responses and adjustments. In aggregate, this is termed acclimatization.
Adverse changes in mood states, as well as impairment in mental performance, occur during altitude exposure (Bahrke and Shukitt-Hale, 1993). Although numerous reports have been published concerning the physiological
alterations that occur under conditions of hypoxia, there have been few investigations of the mood and behavioral changes associated with altitude. This chapter discusses both field and laboratory studies conducted to assess changes in these parameters.
MOOD STATES
Observed behaviors and personal anecdotes suggest that the initial mood experienced at altitude is euphoria, followed by depression. With time, individuals may also become quarrelsome, irritable, anxious, and apathetic (Van Liere and Stickney, 1963). Unfortunately, although disturbances in emotional control have been noticed at altitude for decades, there are few quantitative studies assessing mood changes at altitude.
Shukitt and Banderet (1988) conducted one of the first systematic studies of mood changes at altitude. Self-rated moods were evaluated in 35 subjects using the Clyde Mood Scale. The Clyde Mood Scale (Clyde, 1963) consists of 48 adjectives rated on a 4-point scale: ''not at all," "a little," "quite a bit," and "extremely." These adjectives cluster into six mood factors: friendly, aggressive, clear thinking, sleepy, unhappy, and dizzy. Baseline values were determined at 200 m (656 ft); moods were then assessed for 2 days at 1,600 m (5,250 ft) in one group, or for 4 days at 4,300 m (14,110 ft, at the top of Pikes Peak) with a second group.
At 4,300 m (14,110 ft), moods differed from baseline (200 m [656 ft]) on the day of arrival (day 0) and differed even more after 1 day (Figure 22-1). Subjects became less friendly, less clear thinking, and dizzier. They also became sleepier and happier, while aggressiveness stayed the same (Figure 22-2). Only sleepiness changed at 1,600 m (5,250 ft), with the subjects becoming sleepier at this altitude compared to sea level. However, by day 2 after ascent to 4,300 m (14,110 ft), all changes had returned to baseline levels. Therefore, at 4,300 m (14,110 ft), the altered moods differed from baseline on the day of arrival (1 to 4 hours), differed even more after 1 day (18 to 28 hours), and returned to baseline by day 2 (42 to 52 hours). Mood states are thus adversely affected by both duration and level of altitude, and changes in mood states at altitude have a distinct and measurable time course.
In another study, Shukitt-Hale et al. (1990) evaluated mood states during a climb of Mount Sanford, Alaska to determine the extent to which mood was dependent not only on the altitude and rate of climb, but also on the length of stay and effort expended to reach the desired altitude. Self-rated moods were determined with the Profile of Mood States (POMS). This questionnaire (McNair et al., 1971) is a 65-adjective questionnaire, with the adjectives rated on a 5-point scale. It measures six mood factors: tension, depression, anger, vigor, fatigue, and confusion. Seven male volunteers from the U.S. Army were
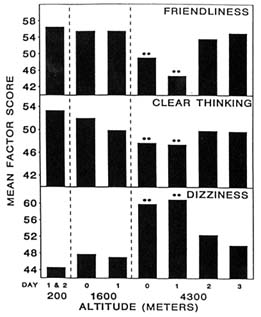
FIGURE 22-1 Factor scores for friendliness, clear thinking, and dizziness at 200; 1,600; and 4,300 m (656; 5,250; and 14,110 ft). ** indicate a significant difference (P < 0.01) from 200 m (656 ft). SOURCE: Shukitt and Banderet (1988), used with permission.
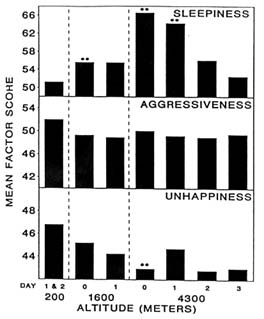
FIGURE 22-2 Factor scores for sleepiness, aggressiveness, and unhappiness at 200; 1,600; and 4,300 m (656; 5,250; and 14,110 ft). ** indicate a significant difference (P < 0.01) from 200 m 656 ft). SOURCE: Shukitt and Banderet (1988), used with permission.
tested over a period of 7 days during a climb to 3,630 m (11,909 ft). The subjects were tested five times,twice at 2,225 m (7,300 ft); then at 2,530 m (8,300 ft); 3,080 m (10,105 ft); and 3,630 m (11,909 ft).
Two mood states were found to be adversely affected over time by the changes in altitude. At 3,080 m (10,105 ft) and 3,630 m (11,909 ft), the subjects were less vigorous and more fatigued (Figure 22-3). These changes were different from those on day 2 at 2,225 m (7,300 ft). The fewer adverse effects seen on day 2 at 2,225 m (7,300 ft) show that some degree of acclimatization may have taken place from day 1 to day 2 at this altitude.
It is interesting to note from this study that even though the climb to the moderate altitude of 3,630 m (11,909 ft) was relatively slow (less than 300 m/d [984 ft/d]), adverse changes were still evident. Exercise promotes a greater demand for oxygen, so a climber can become more hypoxic under these conditions. These data imply that factors other than just the level of altitude can affect mood states, such as physical exertion (or exercise) associated with a climb.
COGNITIVE PERFORMANCE
As with mood changes, the research on cognitive functioning at altitude is somewhat limited. Most investigations have shown decrements in cognitive
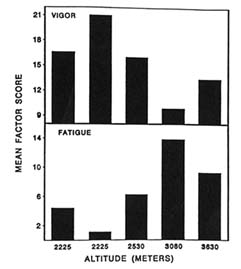
FIGURE 22-3 Profile of Mood States (POMS) factor scores for vigor and fatigue at 2,225; 2,225; 2,530; 3,080; and 3,630 m (7,300; 7,300; 8,300; 10,105; and 11,909 ft). SOURCE: Shukitt-Hale et al. (1990), used with permission.
performance beginning at an altitude of 3,000 m (9,843 ft) (Bahrke and Shukitt-Hale, 1993; Tune, 1964). Interestingly, a few studies have even reported that some of the changes in performance after exposures to extreme altitudes persist for up to a year or longer after return to lower altitudes, although much debate surrounds this issue (Bahrke and Shukitt-Hale, 1993).
High altitude produces substantial impairments in a number of cognitive performances. Changes in psychomotor performance, mental skills, reaction time, vigilance, memory, and logical reasoning have all been measured at altitudes above 3,000 m (9,843 ft) (Bahrke and Shukitt-Hale, 1993). Cognitive performance is usually more vulnerable to altitude than psychomotor performance, and it has been suggested that complex tasks are typically affected before simple tasks (Cudaback, 1984). Impaired performance at altitude can manifest itself in increased errors, slowing of performance, or a combination of these factors (Banderet and Burse, 1991).
Because human cognitive function is sensitive to changes in oxygen availability, exposure to hypoxia should produce a continuum of effects as altitude level and duration increase. One study (Lieberman et al., 1991) evaluated the behavioral effects of hypoxia as a function of time of exposure and altitude level with various standardized tests of cognitive performance. Twenty-three males were tested in an altitude chamber during a 4.5-h exposure to hypobaric hypoxia. The tests (Banderet and Lieberman, 1989) were administered using paper and pencils as well as lap-top computers. All subjects were exposed to two levels of hypoxia, the equivalent of 4,206 m (13,800 ft) and 4,725 m (15,500 ft), as well as a near sea level control condition (549 m [1,800 ft]). During exposure, tests were given from one to three times. Prior to simulated altitude exposure, subjects practiced all tests extensively to insure that performance was stable and near-maximal.
Cognitive performance was significantly impaired on 8 out of the 10 performance measures. Even on relatively simple performance tasks—such as simple and choice reaction time—as well as complex tests of cognition—such as the addition test—impairments occurred in a graded manner. The number of hits on the Bakan vigilance task (Banderet and Lieberman, 1989) decreased with increasing hypoxia (or simulated altitude) (Figure 22-4). Additionally, simple reaction time increased as a function of increased hypoxia (Figure 22-5) as did the percent errors on the four-choice reaction time test. The number of correct responses on the addition (Figure 22-6), coding (Figure 22-7), number comparison (Figure 22-8), pattern recognition, and Tower of Hanoi optimal tests decreased with increasing simulated altitude and time at altitude. Therefore, adverse changes in cognitive performance increased with higher altitudes and longer durations.
As part of this study, mood states were also evaluated at the equivalent of 4,206 (13,800 ft) and 4,725 m (15,500 ft), using the POMS and the Clyde Mood Scale. Data from the POMS showed increased anger, confusion, fatigue, tension, and vigor, while data from the Clyde Mood Scale showed increased
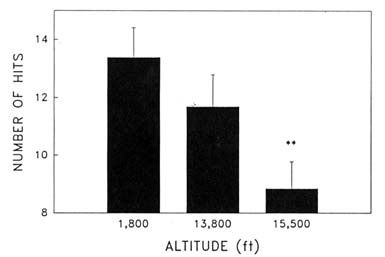
FIGURE 22-4 Effects of simulated altitude (549; 4,206; and 4,725 m [1,800; 13,800; and 15,500 ft]) on number of hits on the Bakan vigilance task. ** indicate a significant difference (P < 0.01) from 549 m (1,800 ft). SOURCE: Lieberman et al. (1991), used with permission.
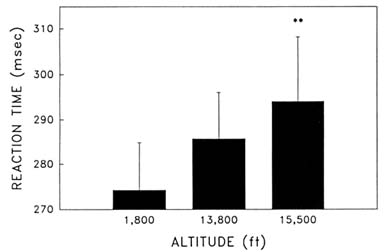
FIGURE 22-5 Effects of simulated altitude (549; 4,206; and 4,725 m [1,800; 13,800; and 15,500 ft] on reaction time on the simple reaction time task. ** indicate a significant difference (P < 0.01) from 549 m (1,800 ft). SOURCE: Lieberman et al. (1991), used with permission.
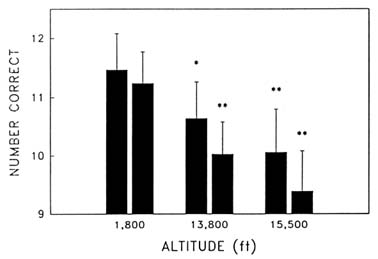
FIGURE 22-6 Effects of simulated altitude (549; 4,206; and 4,725 m [1,800; 13,800; and 15,500 ft]) and time of exposure on number correct for the addition task at two time points. Asterisks indicate a significant difference (*, P < 0.05; **, P < 0.01) from 549 m (1,800 ft). SOURCE: Lieberman et al. (1991), used with permission.
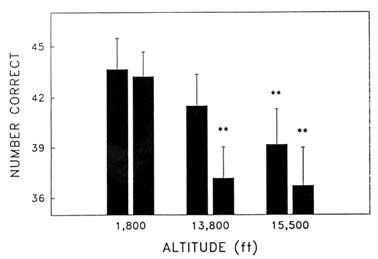
FIGURE 22-7 Effects of simulated altitude (549; 4,206; and 4,725 m [1,800; 13,800; and 15,500 ft]) and time of exposure on number correct for the coding task at two time points. ** indicate a significant difference (P < 0.01) from 549 m (1,800 ft)
SOURCE: Lieberman et al. (1991), used with permission.
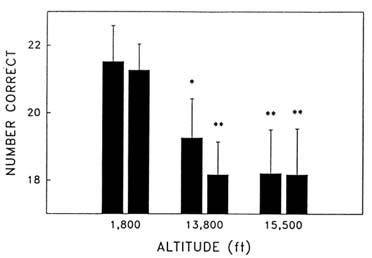
FIGURE 22-8 Effects of simulated altitude (549; 4,206; and 4,725 m [1,800; 13,800; and 15,500 ft]) and time of exposure on number correct for the number comparison task at two time points. Asterisks indicate a significant difference (*, P < 0.05; **, P < 0.01) from 549 m (1,800 ft). SOURCE: Lieberman et al. (1991), used with permission.
sleepiness and dizziness as well as decreased friendliness. Again, the changes observed were a function of both exposure duration and altitude level. A more detailed discussion of these results can be found in Shukitt-Hale et al. (Unpublished manuscript, "Elevation-dependent symptom, mood, and performance changes produced by exposure to hypobaric hypoxia," B. Shukitt-Hale, L. E. Banderet, and H. R. Lieberman, 1996).
TIME COURSE OF EFFECTS
Because adverse changes in mood and cognitive performance occur at altitude, one interesting question is, What is the time course of these effects, and are they related to increases in the incidence of acute mountain sickness (AMS)? The number, severity, rapidity of onset, and duration of symptom, mood, and performance changes vary from person to person and are related to both level of altitude and rate of ascent (Hansen et al., 1967; Shukitt-Hale et al., 1991a). It is usually assumed that individuals afflicted with AMS will be more susceptible to changes in these other parameters. However, previous studies have suggested that their time courses are different (Banderet and Burse, 1991).
AMS symptoms begin to appear after 6 hours, increase from 6 to 24 hours, and reach maximum severity after 30 to 40 hours of exposure (Carson et al., 1969; Hackett, 1980). The time courses of other symptoms and mood states appear similar to that of AMS symptomatology, although these changes are not as well documented (Shukitt and Banderet, 1988). The time course of performance impairments, however, appears somewhat different. One study (Banderet et al., 1986) found decreased levels of performance on all seven tasks administered 1 or 6 hours after ascent to 4,600 m (15,092 ft). At 14 or 19 hours, only four tasks were still impaired, and by 38 or 42 hours, only two were still impaired (Figure 22-9). Therefore, changes in performance were most severe at 1 to 6 hours, the time when AMS symptoms are only beginning to appear. Additionally, performance changes return to baseline when AMS symptoms are most severe (after 30 to 40 hours).
Shukitt-Hale et al. (1991a) conducted a study to determine whether individuals afflicted with initial symptoms of AMS would be more susceptible to adverse changes in other symptoms, moods, and performance, since they may be differentially affected at this point in time. The AMS-Cerebral (AMS-C) factor of the Environmental Symptoms Questionnaire (ESQ) was chosen as the best measure of altitude sickness, since it is a standard index of the degree of illness. Other measures were chosen to assess symptoms, moods, and performance.
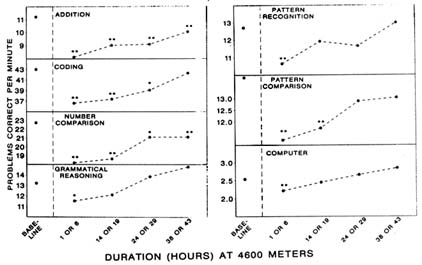
FIGURE 22-9 Performance on cognitive tasks at simulated 4,600 m (15,092 ft) after varied durations of altitude exposure. Performance impairments, significantly different from baseline, are indicated with asterisks above each data point.
SOURCE: Banderet et al. (1986), used with permission.
Twenty subjects were evaluated on 11 symptom, 13 mood, and 14 cognitive-motor performance measures after exposure in the hypobaric chamber to simulated altitudes of 4,725 m (15,500 ft) for 5 to 7 hours. The AMS-C score was significantly correlated with the composite measures of symptoms (r = 0.90), moods (r = 0.77), and performance (r = 0.59) (Figure 22-10). Individuals afflicted with AMS appear to be more susceptible to changes in symptoms, then moods, and finally performance at an altitude of 4,725 m (15,500 ft) for 5 to 7 hours, since the time courses are different. Therefore, it is important to measure a variety of parameters during altitude studies because they are not affected similarly.
BEHAVIOR AND NEUROCHEMISTRY
Because mood and cognitive performance are adversely affected by hypoxia, the question becomes, What is responsible for these effects? The central mechanisms responsible for the effects of hypoxia on behavior and cognitive
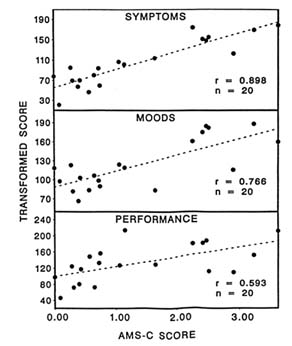
FIGURE 22-10 Correlation with acute mountain sickness-cerebral (AMS-C) score for symptoms, moods, and performance at simulated 4,725 m (15,500 ft). SOURCE: Shukitt-Hale et al. (1991a), used with permission.
performance are not known. It is likely that the direct effects of mild transient hypoxia on the brain are variations in the level of specific neurotransmitters. Because the synthesis of several neurotransmitters is oxygen dependent, abnormalities of neurotransmitter metabolism may mediate the early functional changes due to acute hypoxia (Gibson and Duffy, 1981). It is likely that some of the behavioral decrements caused by hypoxia are attributable to changes in neurotransmitter utilization and concentration (Freeman and Gibson, 1988).
The cholinergic system is particularly vulnerable to hypoxia, and it appears that acetylcholine (ACh) is the neurotransmitter primarily affected (Gibson and Duffy, 1981). ACh is a neurotransmitter involved in the regulation of learning and memory processes (Freeman and Gibson, 1988). The rates of synthesis of other neurotransmitters (e.g., dopamine, serotonin, and the amino acids) are also sensitive to hypoxia, but perhaps less so than the rate of ACh synthesis (Freeman et al., 1986). To determine the effect and influence of acetylcholine on learning and memory performance at altitude, this laboratory undertook a variety of studies using the rat as an experimental model (Shukitt-Hale et al., 1991b, 1993a, 1993b, 1994, in press).
The first series of studies sought to determine the effects of various levels of hypobaric hypoxia on spatial memory in rats and to develop a model to test drug and nutrient interventions. Although deficits in human cognitive performance are well established, few studies have measured cognitive changes in animals exposed to hypoxia.
The Morris water maze (MWM) was used to assess behavior in these studies (Shukitt-Hale et al., 1993b, 1994). The MWM is a standardized test of spatial learning and memory that has been shown to be sensitive to decrements in hippocampal cholinergic function. The maze is a circular black pool filled with water, in which a circular black escape platform is located. The platform is kept below the surface of the water so that the rat must use distal cues to locate it. Numerous extramaze cues were located on the walls of the altitude chamber. Accurate navigation is rewarded with escape from the water onto the platform. The MWM does not rely on food as a reward like other tests of spatial memory (e.g., radial maze or T-maze). Because food consumption is suppressed under hypoxia (discussed below), this is an advantage of using the MWM to assess learning at altitude.
Rats were tested at 2 and 6 hours while exposed to a range of simulated altitudes: sea level, 5,500 m (18,045 ft); 5,950 m (19,521 ft); and 6,400 m (20,997 ft). At the beginning of each trial the rat was gently immersed in the water so that it was facing and touching the wall of the pool. Each rat was allowed 120 seconds to escape onto the platform (Trial 1: termed reference or long-term memory). If the rat failed to escape within this time, it was guided to the platform. Once the rat reached the platform, it remained there for 10 seconds. Trials were given in pairs, with the second trial using the same starting location as the first (Trial 2: termed working or short-term memory).
Pairs of trials were given every 20 minutes until all four starting locations had been used.
Hypobaric hypoxia adversely affected performance in the Morris water maze in an elevation-dependent fashion; as the simulated altitude increased above 5,500 m (18,045 ft), the decrement in performance also increased. Exposure to simulated altitudes of 5,950 and 6,400 m (19,521 and 20,997 ft) produced deficits in both the reference (Figure 22-11) and working memory (Figure 22-12) components of this task as indicated by the significant increase in latency. Also at these altitudes, path lengths increased and swim speeds decreased compared to sea level values. Performance at 6,400 m (20,997 ft) was inferior to that at 5,950 m (19,521 ft), while at 5,500 m (18,054 ft) there were no differences from sea level.
These results agree with human studies that demonstrate elevation-dependent impairments in spatial memory performance during exposure to hypobaric hypoxia. It is important to establish that these decrements in performance exist in animals because numerous treatment strategies can then be evaluated. Also the theory that these deficits may be attributable to changes in hippocampal cholinergic function or other central neurotransmitter systems can be more easily tested. A later chapter (see Lieberman and Shukitt-Hale, Chapter 23 in this volume) will discuss the neurochemical studies done to explore this hypothesis.
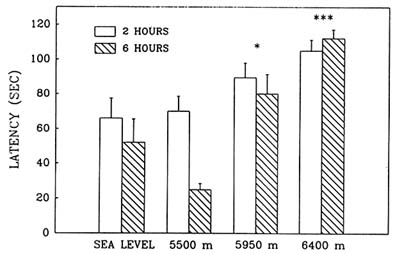
FIGURE 22-11 Effect of simulated altitude (5,500; 5,950; and 6,400 m [18,045; 19,521; and 20,997 ft]) exposure on Morris water maze performance (latency) for Trial 1-reference memory. Asterisks indicate significant group differences between sea level and altitude (*, P < 0.05; **, P < 0.01; ***, P < 0.001).
SOURCE: Shukitt-Hale et al. (1993b), used with permission.
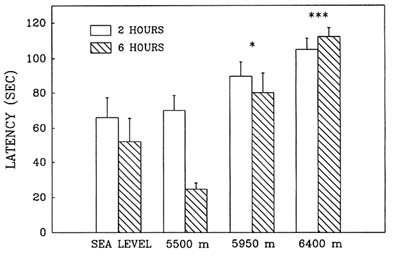
FIGURE 22-12 Effect of simulated altitude (5,500; 5,950; and 6,400 m [18,045; 19,521; and 20,997 ft]) exposure on Morris water maze performance (latency) for Trial 2—working memory. Asterisks indicate significant group differences between sea level and altitude (*, P < 0.05; **, P < 0.01; ***, P < 0.001). SOURCE: Shukitt-Hale et al. (1993b), used with permission.
NUTRITION
High-altitude conditions cause anorexia, a self-induced starvation (Banderet and Burse, 1991). When this laboratory evaluated the effect of prolonged exposure to hypobaric hypoxia in the rat (Shukitt-Hale et al., 1991b, in press), one portion of the study evaluated the effect of long-term exposure to hypoxia on growth and feeding rates. Weight loss and food consumption were measured in three different groups of rats (termed ''morphology" or "behavior" groups) after a simulated 4-d exposure to sea level; 5,500 m (18,045 ft); or 6,400 m (20,997 ft).
Long-term exposure to simulated altitude had an adverse effect on weight. Rats at altitude lost significantly more weight than did animals at sea level (Figure 22-13). They also consumed significantly less food than rats at sea level (Figure 22-14). These data show that rats exposed to prolonged altitude only consumed approximately one-quarter of what a normal rat would eat.
These results corroborate previous findings that animals and humans lose body weight when exposed to low oxygen conditions, due to reduced daily food intake and perhaps other factors. Rose et al. (1988) found similar results
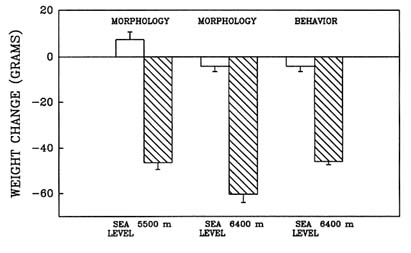
FIGURE 22-13 The effect of simulated altitude (5,500 or 6,400 m [18,045 or 20,997 ft]) on weight loss in three groups of rats.
SOURCE: Shukitt-Hale et al. (1991b), used with permission.
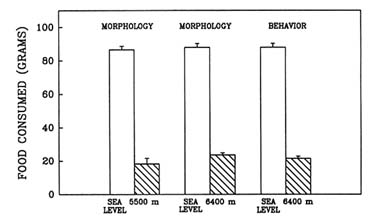
FIGURE 22-14 The effect of simulated altitude (5,500 or 6,400 m [18,045 or 20,997 ft]) on food consumption in three groups of rats.
SOURCE: Shukitt-Hale et al. (1991b), used with permission.
during their investigation of six men fed a palatable diet during a 40-d simulated ascent of Mount Everest to 8,850 m (29,035 ft) (Operation Everest II) (Houston et al., 1987). Prolonged exposure to increasing hypoxia was associated with a reduction in body weight, particularly due to a decrease in carbohydrate preference, despite access to ample varieties and quantities of food. This study (Rose et al., 1988) suggested that hypobaric hypoxia rather than the combined stresses of the mountain environment can be sufficient cause for the weight loss and decreased food consumption reported by major mountain expeditions.
AUTHORS' CONCLUSIONS AND RECOMMENDATIONS
The studies reviewed here show that altitude produces adverse alterations in human mood states, behavior, and cognitive functioning. Human performance, including many critical behavioral functions such as memory, reasoning, and vigilance are significantly impaired initially. Behavioral impairments caused by ascent to high altitude can thus degrade military operations, since both rate of performance and judgment are affected. If the elevation is high enough, complete acclimatization is not possible. The adverse behavioral consequences of hypoxia are dependent on both the level of altitude and the duration at altitude, and they have distinct and measurable time courses. Limited research seems to indicate that some performance decrements induced by extreme altitudes may persist for up to a year or longer after individuals return to lower elevations. These issues need to be considered when military operations at altitude are planned and undertaken.
The neurochemical basis for mood and performance changes needs to be further explored since behavioral decrements at altitude may be attributable to changes in neurochemistry. Strategies, including drugs and nutrients, to facilitate psychological coping and improve performance and functioning in high-altitude environments need to continue to be evaluated. Both animal and human models need to be developed to test the efficacy of these strategies. These treatment strategies may one day be useful in high-altitude military operations to prevent performance decrements and mood changes caused by this environment.
REFERENCES
Bahrke, M.S., and B. Shukitt-Hale 1993 Effects of altitude on mood, behaviour, and cognitive functioning: A review. Sports Med. 16:97–125.
Banderet, L.E., and H.R. Lieberman 1989 Treatment with tyrosine, a neurotransmitter precursor, reduces environmental stress in humans. Brain Res. Bull. 22:759–762.
Banderet, L.E., and R.L. Burse 1991 Effects of high terrestrial altitude on military performance. Pp. 233–254 in The Handbook of Military Psychology, R. Gal and D. Mangelsdorff, eds. New York: Wiley.
Banderet, L.E., B.L. Shukitt, E.A. Crohn, R.L. Burse, D.E. Roberts, and A. Cymerman 1986 Effects of various environmental stressors on cognitive performance. Pp. 592–597 in Proceedings of the 28th Annual Conference of the Military Testing Association. Mystic, Conn.: U.S. Coast Guard Academy.
Carson, R.P., W.O. Evans, J.L. Shields, and J.P. Hannon 1969 Symptomatology, pathophysiology, and treatment of acute mountain sickness. Fed. Proc. 28:1085–1091.
Clyde, D.J. 1963 Manual for the Clyde Mood Scale. Coral Gable, Fla.: Biometric Laboratory, University of Miami.
Cudaback, D.D. 1984 Four-km altitude effects on performance and health. Publications of the Astronomical Society of the Pacific 96:463–477.
Freeman, G.B., and G.E. Gibson 1988 Dopamine, acetylcholine, and glutamate interactions in aging: Behavioral and neurochemical correlates. Ann. N.Y. Acad. Sci. 515:191–202.
Freeman, G.B., P. Nielsen, and G.E. Gibson 1986 Monoamine neurotransmitter metabolism and locomotor activity during chemical hypoxia. J. Neurochem. 46:733–738.
Gibson, G.E., and T.E. Duffy 1981 Impaired synthesis of acetylcholine by mild hypoxic hypoxia or nitrous oxide. J. Neurochem. 36:28–33.
Hackett, P.H. 1980 Acute mountain sickness—the clinical approach. Adv. Cardiol. 27:6–10.
Hansen, J.E., C.W. Harris, and W.O. Evans 1967 Influence of elevation of origin, rate of ascent and a physical conditioning program on symptoms of acute mountain sickness. Milit. Med. 132:585–592.
Houston, C.S., J.R. Sutton, A. Cymerman, and J.T. Reeves 1987 Operation Everest II: Man at extreme altitude. J. Appl. Physiol. 63:877–882.
Lieberman, H.R., B. Shukitt-Hale, L.E. Banderet, and J.F. Glenn 1991 Exposure to hypobaric hypoxia: A method for the comparison of neuropsychologic tests in humans. Abstr. Soc. Neuro. 17:1233.
McNair, D.M., M. Lorr, and L.F. Droppleman 1971 Profile of Mood States Manual. San Diego, Calif.: Educational and Industrial Testing Service.
Rose, M.S., C.S. Houston, C.S. Fulco, G. Coates, J.R. Sutton, and A. Cymerman 1988 Operation Everest II: Nutrition and body composition. J. Appl. Physiol. 65:2545–2551.
Shukitt, B.L., and L.E. Banderet 1988 Moods states at 1600 and 4300 m terrestrial altitude. Aviat. Space Environ. Med. 59:530–532.
Shukitt-Hale, B., T.M. Rauch, and R. Foutch 1990 Altitude symptomatology and mood states during a climb to 3630 m. Aviat. Space Environ. Med. 61:225–228.
Shukitt-Hale, B., L.E. Banderet, and H.R. Lieberman 1991a Relationships between symptoms, moods, and performance, and acute mountain sickness at 4700 meters. Aviat. Space Environ. Med. 62:865–869.
Shukitt-Hale, B., T. Kadar, A. Levy, M.J. Stillman, J.A. Devine, and H.R. Lieberman 1991b Morphology and muscarinic receptor density in the rat brain following exposure to hypobaric hypoxia. Abstr. Soc. Neuro. 17:1532.
Shukitt-Hale, B., M.J. Stillman, A. Levy, J.A. Devine, and H.R. Lieberman 1993a Nimodipine prevents the in vivo decrease in hippocampal extracellular acetylcholine produced by hypobaric hypoxia. Brain Res. 621:291–295.
Shukitt-Hale, B., M.J. Stillman, B.E. Marlowe, D.I. Welch, R.L. Galli, J.A. Devine, A. Levy, and H.R. Lieberman 1993b Hypobaric hypoxia adversely affects spatial memory in an exposure-dependent fashion. Abstr. Soc. Neuro. 19:363.
Shukitt-Hale, B., M.J. Stillman, D.I. Welch, A. Levy, J.A. Devine, and H.R. Lieberman 1994 Hypobaric hypoxia impairs spatial memory in an elevation-dependent fashion. Behav. Neural Biol. 62:244–252.
Shukitt-Hale, B., T. Kadar, B.E. Marlowe, M.J. Stillman, R.L. Galli, A. Levy, J.A. Devine, and H.R. Lieberman In Press: Morphological alterations in the hippocampus following hypobaric hypoxia. Hum. Exp. Toxicol.
Tune, G.S. 1964 Psychological effects of hypoxia: A review of certain literature from the period 1950 to 1963. Percept. Mot. Skills 19:551–562.
Van Liere, E.J., and J.C. Stickney 1963 Hypoxia. Chicago: University of Chicago Press.
| This page in the original is blank. |


















