Evolution of Columns, Modules, and Domains in the Neocortex of Primates
![]()
The specialized regions of neocortex of mammals, called areas, have been divided into smaller functional units called minicolumns, columns, modules, and domains. Here we describe some of these functional subdivisions of areas in primates and suggest when they emerged in mammalian evolution. We distinguish several types of these smaller subdivisions. Minicolumns, vertical arrays of neurons that are more densely interconnected with each other than with laterally neighboring neurons, are present in all cortical areas. Classic columns are defined by a repeating pattern of two or more types of cortex distinguished by having different inputs and neurons with different response properties. Sensory stimuli that continuously vary along a stimulus dimension may activate groups of neurons that vary continuously in location, producing “columns” without specific boundaries. Other groups or columns of cortical neurons are separated by narrow septa of fibers that reflect discontinuities in the receptor sheet. Larger regions of posterior parietal cortex and frontal motor cortex are parts of networks devoted to producing different sequences of movements. We distinguish these larger functionally distinct regions as domains. Columns of several types have evolved independently a number of times. Some of the columns found in primates likely emerged with the first primates, whereas others likely were present in earlier ancestors. The sizes and shapes of columns seem to depend on the balance of neuron activation patterns and molecular signals during development.
__________
Department of Psychology, Vanderbilt University, Nashville, TN 37240-7817. E-mail: jon.h.kaas@vanderbilt.edu.
Neocortex is an important part of the brain that varies in size from a small cap on the rest of the forebrain (Kaas, 2007) to approximately 80% of the brain in humans (Azevedo et al., 2009). The varied functions of neocortex depend on the cortical areas, the so-called “organs of the brain” (Brodmann, 1909) that are specialized for processing different inputs and providing different outputs. Cortical areas can be hard to define and identify, and their exact number in any species is uncertain. However, it is clear that the number of cortical areas varies across extant taxa, from approximately 20–30 or so to perhaps more than 200 in humans (Kaas and Preuss, 2008). Because the first mammals had little neocortex and likely few cortical areas, interest in the evolution of neocortex across the great radiation of mammals has largely focused on the issue of modifying and adding cortical areas. Some of the cortical areas proposed for primates are shown in Fig. 7.1. However, areas are often composed of smaller subdivisions, the cortical columns or modules, and these subdivisions within areas modify and expand the functions of areas. Thus, an understanding of how different types of neocortex evolved depends not only on determining the numbers and types of cortical areas that exist but also on the modifications of the internal organization of areas that occur in the various lines of evolution, including modifications in columnar organization. Here we review the types of columnar subdivisions of cortical areas that have been proposed (Hendrickson, 1985; Purves et al., 1992; Mountcastle, 1997; da Costa and Martin, 2010) and then consider how and when such modules might have evolved. The phyletic distributions of the types of columns in extant mammals allow one to infer when such columns evolved (Hennig, 1966; Striedter, 2005). Primates, rodents, tree shrews, and lagomorphs are all placed within the superorder Euarchontoglires. Thus, we are especially interested in how types of columns are distributed within the primate radiation, but also whether they are present in the closest relatives of primates. Because the shapes of columns are not always columnar, they also are called modules.
MINICOLUMNS
One of the defining features of neocortex is that it consists of layers and various sublayers of neurons specialized for different steps in processing; neurons in radial (vertical) arrays across the layers are more densely interconnected than neurons along the layers (Casagrande and Kaas, 1994; Nieuwenhuys, 1994b; Kaas, 2010). As a result, neurons in narrow vertical arrays share many response properties, especially the location of the receptor fields of neurons on the sensory receptor surface. This arrangement has great functional importance, and it is likely responsible for the impressive flexibility and powers of neocortex. Developmentally, minicolumns reflect
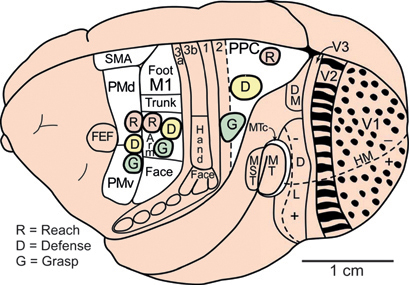
FIGURE 7.1 Some of the proposed cortical areas of primates shown on a dorsolateral view of the left cerebral hemisphere. Modular subdivisions of some of these areas are discussed in the text. Visual areas include the first, second, and third areas (V1, V2, V3), dorsomedial (DM or V3a) and dorsolateral visual areas (DL or V4), the middle temporal area (MT), the MT crescent (MTc), and the medial superior temporal (MST) area. The representation of the zero horizontal meridian (HM) divides the representation of the upper (+) and lower (-) visual hemifields. Motor areas include primary motor cortex (M1), ventral (PMv), and dorsal (PMd) premotor cortex, the supplementary motor area (SMA), and the frontal eye field (FEF). Somatosensory areas include the four areas of anterior parietal cortex (3a, 3b, 1, 2), with the region representing tactile inputs from the hand indicated in area 3b (S1). Modular subdivisions in V1 (dots) and V2 (bands) are shown in black (see text). Ovals mark the locations of proposed reach, defense, and grasp domains in motor and posterior parietal cortex (PPC). Based on Gharbawie et al. (2011a).
the radial migration of clones of excitatory neurons from progenitors in the ventricular and subventricular zones (Rakic, 1995b), as radially arranged sister neurons preferentially develop synapses with each other (Yu et al., 2009). These vertical arrays of interconnected neurons across the cortical layers have been called minicolumns (Mountcastle, 1957, 2003). Minicolumns are sometimes visible as vertical arrays of neurons separated somewhat by neuropil (Buxhoeveden and Casanova, 2002; DeFelipe et al., 2002). Minicolumns are thought to be 30–50 μm in diameter, although functional boundaries between them are not likely to be sharp owing to the spread of apical dendrites of pyramidal cells and the extents of axon arbors of cortical neurons and subcortical activating inputs. Because minicolumns are clearly visible in a number of cortical areas, and across mammalian species, including monotremes, they may have originated when the ancestors of all extant mammals with a cortex of six layers emerged.
Mountcastle (1957) introduced the concept of cortical columns after reporting that recordings along microelectrode trajectories tangential to the surface of somatosensory cortex encountered short sequences of neurons that responded either to light touch on the skin (superficial skin receptors) or touch with pressure (deep receptors). This grouping of cortical neurons according to how they respond to sensory stimuli led to the concept of a patchwork of alternating columns of neurons that extend across all cortical layers, with each type of column activated by a different somatosensory input. The subsequent evidence for such alternating patches of neurons activated by either deep or superficial receptors of the skin and deeper tissue has been limited, and they do not seem to exist in area 3b (S1 proper) of somatosensory cortex of monkeys. Instead, there is evidence for a modular arrangement of groups of neurons in layer 4 that responds to activation by inputs relayed from either slowly adapting or rapidly adapting cutaneous receptors of the skin (Sur et al., 1981, 1984). There is also evidence for at least a partial segregation of territories activated by slowly adapting and rapidly adapting receptors in area 1 of somatosensory cortex of monkeys (Friedman et al., 2004). However, given these limited observations, we can say little about the phyletic distribution of slowly adapting and rapidly adapting cortical columns, or their evolution, even in primates.
More can be said about the blob and interblob surround organization of primary visual cortex (V1) in primates (Fig. 7.2). All primates seem to have a pattern of cytochrome oxidase (CO)-rich blobs (reflecting high metabolic activity) within interblob surrounds of lower CO levels (Horton, 1984; Horton and Hedley-Whyte, 1984; Preuss and Kaas, 1996). Neurons in the blobs respond to color, are less selective for stimulus orientation, and have higher firing rates than neurons between the blobs (Livingstone and Hubel, 1984; Hendrickson, 1985; Felleman, 2008; Lu and Roe, 2008; Economides et al., 2011). However, blobs and interblob regions are found not only in primates with trichromatic or dichromatic color systems but also in nocturnal primates with only one functional type of cone in the retina (Wikler and Rakic, 1990). The blobs and interblobs are also distinguished by different patterns of inputs from the visual thalamus, intrinsic connections, and connections with other visual areas (Livingstone and Hubel, 1984; Casagrande and Kaas, 1994). In macaque monkeys, most of these connections are well developed in newborns (Barone et al., 1996; Baldwin et al., 2012). The segregation of groups of neurons by differences in response characteristics that are mediated by differences in activating inputs fits the classic definition of cortical columns, although the blobs and interblobs do not occupy equal territories, and the interblob territory is continuous. The blob and surround pattern evolved in the immediate
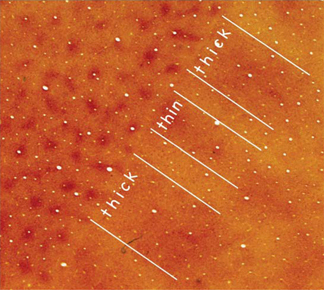
FIGURE 7.2 Anatomically defined columns in visual cortex of primates. Sections of primary visual cortex (V1) and the adjoining second visual area (V2) of a macaque monkey have been cut parallel to the brain surface and processed for CO, a marker of neurons with high metabolic requirements. The brain sections provide a “surface view” of parts of V1 and V2. In V1, there is a pattern of CO-rich “blobs” (also called “puffs” or “patches”) surrounded by cortex that expresses less CO, the interblob territory. In V2 an alternating pattern of CO-dark bands, separated by CO-light bands, cross the width of V2. The CO-dark bands are of two types, thick and thin. Thus, there are three types of bank-like structures in V2 that can be anatomically distinguished. Because the CO blobs and interblobs, as well as the CO-dense thick, thin, and interbands have neurons that differ in response properties, they can be considered classic columns. A pattern of CO-dense and CO-light bands is also present in the third cortical visual area, V3, along the outer border (on the right) of V2. Compare with Fig. 7.1.
ancestors of primates, or in archaic primates, given that none of the close relatives of primates, tree shrews, rodents, and lagomorphs have blobs in V1.
Classic columns are also found in the second visual area, V2, of most primates, where V2 is characterized by a repeating series of CO-dense thick stripes and CO-dense thin stripes separated by CO-pale interstripes. These band-like stripes cross the narrow width of V2, and they seem to exist in all anthropoid primates (Kaas, 2003). The three types of stripes differ in anatomical connections and have neurons with different response properties. The stripes and differences in connections are apparent in newborn macaques (Barone et al., 1996; Baldwin et al., 2012). Although the CO-dense stripes are not consistently distinguishable as thick or thin, they can be identified by functional differences, with neurons in the thick stripes sensitive to binocular disparities and stimulus orientation, the neurons in the thin stripes sensitive to luminance and color, and neurons
in pale stripes sensitive to stimulus orientation (Hubel and Livingstone, 1987; Livingstone and Hubel, 1988; Lu and Roe, 2007; Felleman, 2008; Kaskan et al., 2009). The thick stripes project to visual area MT, whereas the other bands project to DL (V4). In prosimian primates, CO stripes in V2 are only weakly apparent, and such stripes are not present in V2 of tree shrews and rodents (Kaas, 2003). Thus, aspects of the stripe pattern may have evolved in early primates, whereas such stripes became fully developed as anthropoid primates emerged.
Although the V1 blob and interblob regions, as well as the V2 stripes, do not look like cylindrical pillars, they otherwise conform to the expectations of classic cortical columns. Other such classic columns undoubtedly exist (Mountcastle, 1997), but they largely remain to be explored. One such example is in the MT crescent, MTc, a visual area that forms a belt around the middle temporal visual area, MT (Fig. 7.1). This poorly understood visual area is composed of a series of CO-dense puffs in a single row, like beads on a string in a belt of CO-pale tissue (Kaas and Morel, 1993). The significance of these puffs and surrounds in MTc, which have different connections with other visual areas, remains to be determined.
Unbounded Columns That Represent Sectors of a Continuous Stimulus Dimension
Several cortical areas have repeating representations of stimulus orientations for different portions of the visual field (Hubel and Wiesel, 1963). Most notably, primary visual cortex of primates, carnivores, and tree shrews have repeating “pinwheel” patterns of cortex, in which stimulus orientation is systematically represented from vertical to horizontal lines and edges and back again (Bonhoeffer and Grinvald, 1991; Fitzpatrick, 1996; Kaschube et al., 2010). Groups of neurons most sensitive to one stimulus orientation or another can be selectively activated, the activity pattern optically imaged, and regions of cortex sensitive to different orientations color coded to produce colorful illustrations of arrays of orientation “columns.” These “columns” differ from classic columns in that they have no borders because the orientations of stimuli change continuously without disruption. Thus, the illustrated “borders” between orientation columns are arbitrary. In addition, all “orientation columns” are selective for the same stimulus features, and thus these columns are not of the classic type, which are segregated by different classes of activating inputs. However, each entire array of orientation-selective neurons, the pinwheel for a given location in the visual field, can be considered as a larger domain or hypercolumn (Hubel and Wiesel, 1972, 1977). Orientation hypercolumns are widespread in visual cortex of primates: they also have been identified in V2 stripes, V3, V4 (DL), and MT (Kaskan et
al., 2009, 2010; Tanigawa et al., 2010). Neurons in orientation-selective hypercolumns may be divided for each orientation column into halves, preferring one or the other direction of motion perpendicular to the preferred orientation (Kaskan et al., 2010). The grouping of neurons by their preferences for stimulus orientation seems to be a trait that emerged first in V1 in the common ancestors of tree shrews and primates, because tree shrews also have orientation hypercolumns. However, the more distant relatives, rodents and rabbits, have orientation-selective neurons in visual cortex but not orientation-selective columns (Kaschube et al., 2010). Carnivores have independently evolved orientation hypercolumns in V1. Possibly, the presence of orientation hypercolumns in V1 is a prerequisite for the evolution of such hypercolumns in other visual areas, as found in primates. Orientation hypercolumns have not been reported for areas of extrastriate cortex of tree shrews. Thus, the extrastriate hypercolumns for stimulus orientation may have emerged with the first primates.
Other proposed modules of V2 in primates include subregions of thin stripes selective for different hues (Xiao et al., 2003; Roe, 2004). These hue-selective subregions are not classic columns because they are not separated by columns that are most sensitive to another stimulus feature, and they have arbitrary boundaries.
There is only limited evidence for the existence of classic columns in auditory cortex. All mammals seem to have primary cortical auditory areas that represent the receptors of the cochlea in a linear manner so that neurons are arranged in one dimension across a cortical area from being most sensitive to low-frequency sounds on one end, to high-frequency sounds on the other (Kaas, 2011). Thus, there are no modular divisions based on sound frequency, although isofrequency bands with arbitrary borders have been described. However, bands of primary auditory cortex where neurons that are excited from both ears (EE bands) alternate with bands of cortex with neurons that are excited by the contralateral ear and inhibited by the ipsilateral ear (EI bands) have been reported for cats (Merzenich and Kaas, 1980). The EE and EI bands extend across the isofrequency contours. Because EE and EI bands have neurons of differing functional properties, they qualify as classic columns (although shaped like bands). Such bands have not been identified in auditory cortex of primates.
Modules Representing Separated Parts of Sensory Surfaces
Another type of module, one that also would not qualify as a classic column, concerns separations of groups of neurons in somatotopic maps of the body surface, or retinotopic maps of the two eyes, in areas of cortex. The best-known example is the rows and columns of “barrels” in primary
somatosensory cortex of rats and mice, where a barrel-like structure represents each of the large sensory whiskers on the side of the face (Woolsey et al., 1975). The digits and pads of the feet also relate to separated groups of neurons (Dawson and Killackey, 1987).
The many studies of the “barrel field” of mice and rats have revealed that differences in neural activity are important in the formation of barrels, such that the number of barrels varies with the number of facial whiskers. Molecular factors also alter the formation of barrels, as revealed in mutant mice (Erzurumlu and Kind, 2001). Such segregations of cortical neurons by body part are found in primary somatosensory cortex of many species, but are perhaps most apparent in the somatosensory cortex of the star-nosed mole, where the highly innervated tactile rays of the nose are each separately represented in three areas of somatosensory cortex (Catania and Kaas, 1996). In primary somatosensory cortex of New World and Old World monkeys (Jain et al., 1998; Qi and Kaas, 2004), and possibly other anthropoid primates, the representations of the digits are separated from each other by narrow cell-poor septa, with a more conspicuous septum separating the representation of digit 1 (thumb) from that of the face. Such separated representations of digits in area 3b of primates are variable and have not been described in prosimian primates. Septa that separate representations of digits are more apparent in macaque monkeys than in New World owl monkeys and squirrel monkeys.
It could be argued that the narrow septal regions that separate the cortical barrels, bands, and other modules related to body parts do have neurons that differ in connections, such as having corpus callosum connections, and thus there is an alteration of functional types of columns in the classical sense. However, the septa are cell-poor, narrow regions that are primarily there to reflect disruptions of the receptor sheet. Yet, these narrow septa may be opportunistically occupied by late-developing sources of input. Because the septa that form module borders reflect junctions in neuron activity patterns during sensory activation, these septa are most apparent early in sensory hierarchies where short response latencies to sensory stimuli are maintained.
The retina of each eye is a continuous sensory surface, except for the nerve head and a narrow septum corresponding to the nerve head, which disrupts layers of the lateral geniculate nucleus that receive projections from the contralateral eye (Kaas et al., 1973). In cortex, the ocular dominance “columns” in primary visual cortex of primates fall into the category of modules based on disruptions of the sensory surface, because the retina of the two eyes have independent activity patterns prenatally. Thus, the afferents from the hemiretina of each eye terminate in separate layers in the lateral geniculate nucleus of the visual thalamus, and then these layers project in retinotopically matched patterns to primary visual
cortex to either congruently overlap or to separate locally in variable patchy-to-banding patterns in layer IV while maintaining some level of retinotopy, depending on species (Florence and Kaas, 1992; Horton and Adams, 2005). Ocular dominance columns, first revealed in microelectrode recordings (Hubel and Wiesel, 1968), and axon termination patterns from layers of the lateral geniculate nucleus (Wiesel et al., 1974) can also be demonstrated by differences in activity levels after blocking activity from one eye (Horton, 1984; Takahata et al., 2009a). The segregation of eye-related afferents is very weak in some primates, such as nocturnal prosimian galagos and owl monkeys (Kaskan et al., 2007; Takahata et al., 2011), and highly variable patterns exist in New World monkeys, even across individuals within a species (Horton and Adams, 2005). Ocular dominance patterns may reflect a high degree of segregation of thalamic afferents in layer 4 of primary visual cortex, as in Old World monkeys (Fig. 7.3), apes, and humans, or reflect such a low level of separation that they are anatomically cryptic and only revealed by relative differences in
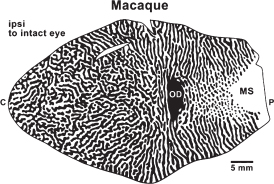
FIGURE 7.3 Ocular dominance columns (bands) in a flat surface view of primary visual cortex (V1) of an Old World macaque monkey as reflected by distribution of terminations of lateral geniculate axons related to each eye in cortical layer 4. Regions of black receive inputs from the ipsilateral eye, including the region of the optic disk of the retina that produces a gap in the projection of the hemiretina of the contralateral eye (OD in cortex). The monocular segment (MS) of V1 is activated by the monocular segment of the contralateral visual hemifield that is seen only by the contralateral eye. Foveal and central vision is represented to the left, and the extreme of peripheral vision is represented to the right. The ocular dominance bands break up into a dot-and-surround pattern in the part of V1 that represents peripheral vision as the inputs from the contralateral eye (white) become proportionately greater, and form the larger surrounds. Modified from Florence and Kaas (1992).
neural responses to each eye as revealed in optical imaging experiments (Kaskan et al., 2007) or the expression pattern of activity-dependent genes (Takahata et al., 2009b). Ocular dominance “columns” are absent in the closest relatives of primates, tree shrews, rodents, and rabbits, and thus are a feature of visual cortex that evolved in early primates but became more pronounced in Old World monkeys, apes, and humans. Obvious ocular dominance columns have evolved independently in carnivores (Anderson et al., 1988), and they likely exist in other taxa.
Domains: Larger Functional Divisions of Cortical Areas
Primary motor cortex and dorsal and ventral premotor areas are widely recognized as valid cortical areas, and each of these areas has a somatotopic representation of small movements of body parts that are revealed by brief trains of near-threshold pulses of electrical current. However, cortical motor areas representing major body parts, such as the forelimb, have a locally fractured somatotopy so that different movement zones, roughly the size of minicolumns, are mixed and repeated (Fig. 7.4). Thus, the forelimb region mixes zones for digit, wrist, elbow, and shoulder movements in a puzzling arrangement (Gould, 1986; Donoghue et al., 1992; Qi et al., 2000) that is unlike that of primary sensory representations, which closely reflect the organization of the sensory sheet. However, the somatosensory representation of tactile projections to the cerebellar cortex forms a fractured representation of the body surface (Shambes et al., 1978), much like the representations in motor cortex. The explanation for these adjoining patches of cerebellar cortex devoted to various nonadjacent body parts was that neurons in groups of such patches could interact to form “action-involved structures” for directing movement patterns.
It has long been known that longer trains of electrical pulses at higher current levels evoke more complex movement sequences from motor cortex than do short trains at threshold levels (Leyton and Sherrington, 1917). More recently, Graziano et al. (2002) have used longer (0.5 s) trains of electrical pulses to define different regions or domains (Fig. 7.1) in motor cortex where different ethologically relevant movement can be evoked (climbing, reaching, grasping, defense of the head, hand-to-mouth). Matching movement domains have been identified in posterior parietal cortex (Cooke et al., 2003; Stepniewska et al., 2005; Gharbawie et al., 2011a,b). In primary motor cortex, several different domains for functionally distinct movement patterns are found in separate parts of the forelimb representation, perhaps offering some explanation for the mosaic of minicolumns for different but related small movements and muscle twitches that are revealed by short trains of pulses at threshold levels of stimulating current (Fig. 7.4). Thus, circuits within a domain may evoke
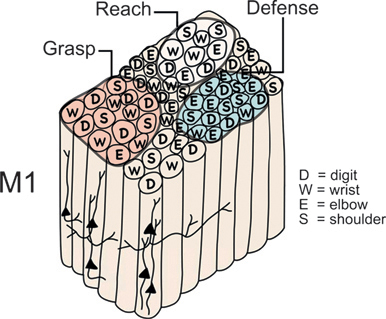
FIGURE 7.4 Proposed functional organization of the hand–forearm segment of primary motor cortex (M1) in monkeys and other primates. Although M1 has an overall somatotopy, the local somatotopy is fractured to form a mosaic of radial rows of neurons that evoke small, specific movements when electrically stimulated with brief trains of electrical pulses at threshold levels of current. Thus, neuron arrays or minicolumns for digit movement may adjoin those for wrist, elbow, or shoulder movements. Subsets of these minicolumns seem to be grouped to function in the production of more complex, ethologically relevant movement sequences, such as grasping, reaching, or defending the head against a blow. We refer to these larger divisions of motor, premotor, and posterior parietal cortex (Fig. 7.1) as domains (Gharbawie et al., 2011a).
sequences of movements involving the different body parts represented within the domain.
Functionally matched domains for at least some of the complex movement patterns of primary motor cortex also exist in premotor cortex and in posterior parietal cortex. The domains in posterior parietal cortex may be parts of larger cortical areas. The domains in frontal and posterior parietal cortex have similar spatial arrangements in prosimian galagos, two species of New World monkeys, and Old World macaque monkeys, and there is indirect evidence for them in humans (Kaas et al., 2011). Thus, they likely exist in all primates. Such domains for complex movements may also exist in motor cortex of the relatives of primates, tree shrews and rodents, where M1 also has a fractured somatotopy (Remple et al., 2007; Cooke et al., 2011). However, posterior parietal cortex is no more than a narrow strip of cortex in tree shrews and rodents and is unlikely to contain a series of primate-like domains.
Other areas of cortex may also have larger functionally distinct regions within cortical areas. For example, some of the face-selective and object-
selective regions of temporal cortex in macaque monkeys and humans resemble domains (Tsao et al., 2003, 2008a; Pinsk et al., 2005; Rajimehr et al., 2009). Likewise, the large visual area termed V4 or DL has been divided into large regions or domains of neurons that are either color selective or orientation selective (Tanigawa et al., 2010), although these large regions might also be considered separate cortical areas (Cusick and Kaas, 1988; Stepniewska and Kaas, 1996).
How Do Columns and Modules Emerge in Development?
A number of factors likely contribute to the functional organization of cortex, but at the modular level, activity-dependent selection of coactive afferents together with cellular signals that are position dependent probably are two of the most important variables (Erzurumlu and Kind, 2001; Sur and Leamey, 2001; Kaas and Catania, 2002). There is considerable evidence to support this conclusion, but some of the most impressive evidence comes from studies that created three-eyed frogs (Constantine-Paton and Law, 1978; Katz and Constantine-Paton, 1988). In frogs, each optic tectum normally receives inputs from only the contralateral eye, but when a third eye is added experimentally to one side of the head during embryonic development, both eyes on that side compete for territory in the same contralateral optic tectum. The projections from each of these eyes respond to molecular signals that tend to produce the same retinotopic pattern in the optic tectum, but local groups of tectal neurons favor inputs from one eye or the other. The result is that the afferents from the two eyes form alternating bands or stripes that resemble the ocular dominance bands in cats and anthropoid primates. The borders between these bands in the optic tectum and visual cortex correspond to locations where abrupt differences in activity patterns occur, and they do not develop or they degrade when activity is blocked (Cline et al., 1987). Obviously, the ability to form ocular dominance bands did not evolve via natural selection in the optic tectum of frogs for some future function. Instead, the developmental factors that produced these columns were present for other reasons that are not clear but apparently are widely important in nervous system development (Katz and Constantine-Paton, 1988). The capacity for module formation seems to be inherent in all cortical tissue, as well as in other tissue such as the optic tectum or superior colliculus, where inputs of different activation patterns compete for location with an overall global map. Thus, ocular dominance bands and other configurations, as well as orientation modules and other types of columns, including those based on discontinuities of the receptor sheet, have emerged independently in several lines of mammalian evolution. For some of these types of modules, asking what they do (Horton and Adams, 2005) may be the wrong
question. Instead, we might ask, what else is achieved in neural tissue by the mix of activity-dependent and position-dependent factors that select and group synaptic contacts when these factors coexist at particular developmental times? Purves et al. (1992) have suggested that some of the columns that have been described in cortex are “by-products” of synaptic development. If so, what is the product?