Evolution of Brains and Behavior for Optimal Foraging: A Tale of Two Predators
![]()
Star-nosed moles and tentacled snakes have exceptional mechanosensory systems that illustrate a number of general features of nervous system organization and evolution. Star-nosed moles use the star for active touch—rapidly scanning the environment with the nasal rays. The star has the densest concentration of mechanoreceptors described for any mammal, with a central tactile fovea magnified in anatomically visible neocortical modules. The somatosensory system parallels visual system organization, illustrating general features of high-resolution sensory representations. Star-nosed moles are the fastest mammalian foragers, able to identify and eat small prey in 120 ms. Optimal foraging theory suggests that the star evolved for profitably exploiting small invertebrates in a competitive wetland environment. The tentacled snake’s facial appendages are superficially similar to the mole’s nasal rays, but they have a very different function. These snakes are fully aquatic and use tentacles for passive detection of nearby fish. Trigeminal afferents respond to water movements and project tentacle information to the tectum in alignment with vision, illustrating a general theme for the integration of different sensory modalities. Tentacled snakes act as rare enemies, taking advantage of fish C-start escape responses by startling fish toward their strike—often aiming for the future location of escaping fish. By turning
_____________
Department of Biological Sciences, Vanderbilt University, Nashville, TN 37235. E-mail: ken.catania@vanderbilt.edu.
fish escapes to their advantage, snakes increase strike success and reduce handling time with head-first captures. The latter may, in turn, prevent snakes from becoming prey when feeding. Findings in these two unusual predators emphasize the importance of a multidisciplinary approach for understanding the evolution of brains and behavior.
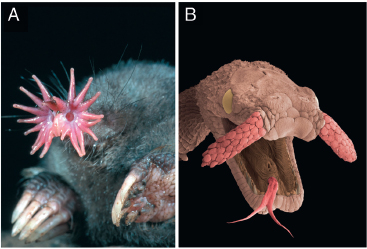
Star-nosed moles and tentacled snakes each have novel sensory appendages protruding from their faces. These appendages give both animals a unique appearance unparalleled among their peers—no other mammal or snake has comparable appendages (Fig. 13.1). However, there is more than the bizarre appearance of these animals to attract our attention. Extreme sensory specializations often reveal general principles of nervous system function and organization that are less obvious in other species (Hodgkin and Huxley, 1952; Carr and Konishi, 1990; Heiligenberg, 1991; Bass and Zakon, 2005; Kawasaki, 2009; Konishi, 2010; Nottebohm and Liu, 2010). More generally, extremes in morphology provide informative case studies in evolutionary biology. Indeed, Darwin (1859) devoted a special section of On the Origin of Species by Means of Natural Selection to “Organs of extreme perfection and complication.” One can argue whether these unusual species seem in some way perfected, but surprisingly, the complexity of the mole’s star has been cited as evidence of a divine creator (Weston and Wieland, 2003).
My goal is to review recent studies of these two species beginning with star-nosed moles, the species for which we have the most information from many years of study. The mole’s nose is exceptional not only in appearance but also in the high density of mechanoreceptors that covers the nasal rays and the complexity of the modular neocortical network that processes touch information from the star. These findings make the question of how and why the star evolved even more mysterious. However, expanding studies to include the mole’s habitat and behavior in the context of optimal foraging theory (Stephens and Krebs, 1986) strongly suggests a selective advantage (the ability to specialize on very small prey) that led to the evolution of the star as the highest resolution touch organ among mammals. Another extension of the research to include comparative and developmental studies provides compelling evidence for how the star evolved (Gould, 1977; Catania et al., 1999).
Recent investigations of aquatic tentacled snakes reveal a very different use for sensory appendages (Catania et al., 2010). Rather than serving active touch, the snake’s tentacles seem to act as fish-detecting motion sensors. However, the most interesting finding from the tentacled snake is its remarkable ability to use fish escape responses to its advantage (Catania, 2009, 2010).
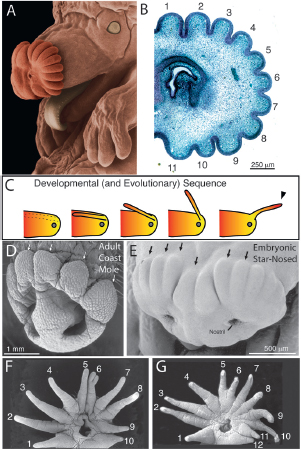
FIGURE 13.1 A star-nosed mole (Condylura cristata) and tentacled snake (Erpeton tentaculatus). (A) Star-nosed moles have large forelimbs, small eyes, and a nose ringed by 22 appendages or rays. (B) A colorized scanning electron micrograph shows the snake’s scaled tentacles. [Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
The details of how and why each species evolved appendages are very different, but the lessons from investigating their biology are similar. In each case, an integrative approach combining neurobiological, behavioral, and ecological facets is necessary to best understand the sensory system. In the spirit of such an approach, it is hoped that the reader will view Movies S1, S2, S3, S4, S5, and S6 of Supporting Information in Catania (2012) when reading the descriptions of behavior.
SENSORY ORGANS AND INNERVATION OF THE STAR
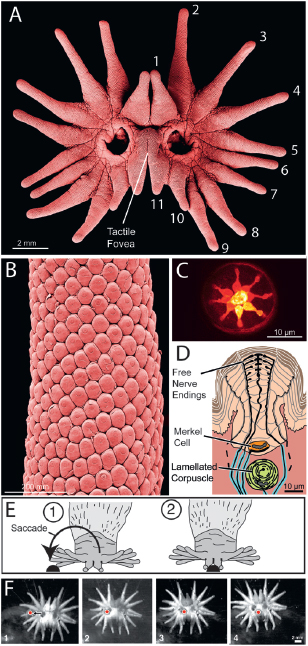
The star is a little over 1 cm across and composed of 22 epidermal appendages or rays. Thus, it is a skin surface and not a specialization for olfaction. The rays are numbered from 1 to 11, starting with the dorsal-most ray and ending ventrally with a small ray in front of the mouth (Fig. 13.2A). Each ray is covered with small domes called Eimer’s organs (Eimer, 1871; Van Vleck, 1965) (Fig. 13.2B). Such mechanosensory organs are found on the noses of most moles (Quilliam, 1966; Shibanai, 1988; Catania, 2000b) and are anatomically similar to small, domed push rods found on the snout of distantly related monotremes (Andres et al., 1991; Iggo et al., 1996; Manger and Pettigrew, 1996; Proske et al., 1998). In star-nosed moles, each organ is about 40–60 μm in diameter and has a small
FIGURE 13.2 The epidermis of the star. (A) A star under the scanning electron microscope showing the 22 rays. (B) Higher magnification showing Eimer’s organs covering a single ray. (C) Nerve endings labeled with DiI at the apex of an Eimer’s organ (confocal microscopy). (D) The internal organization of a single Eimer’s organ. (E) Schematic illustration of a saccadic star movement. (F) Frames from high-speed video illustrate a saccadic star movement to a small prey item (outlined in red).[Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
(15–20 μm) central disk on the outer surface. The disk is a single epidermal cell marking the top of a stack of cells that runs through the center. Each central cell column is associated with a Merkel cell–neurite complex at its base and a series of free nerve endings that travel through the column in a precise geometric ring pattern with a single nerve ending in the center (Fig. 13.2C and D). Directly below the cell column, a single lamellated corpuscle is located in the dermis.
This sensory unit is repeated 25,000 times on a typical star, providing a high concentration of mechanoreceptors. The mechanoreceptors are innervated by over 100,000 myelinated fibers (Catania and Kaas, 1997) carried by massive trigeminal nerves. The star has five times more mechanosensory afferents than the entire human hand (Vallbo and Johansson, 1984). Electrophysiological recordings from the nerves reveal tiny receptive fields on the star and show that Eimer’s organs are directionally sensitive and respond to the slightest deflection (Marasco and Catania, 2007).
BEHAVIOR REVEALS A HIGH-SPEED TACTILE FOVEA
Star-nosed moles repeatedly touch the star to objects and tunnel walls as they explore their underground habitat. This behavior is very rapid; a mole may touch the star to 10–13 different places per second as it searches for food (Catania and Remple, 2004; Catania, 2012, Supporting Information, Movie S1). Despite the extreme speed of these exploratory movements, slow-motion analysis of foraging behavior reveals a functional subdivision of the star into peripheral and central touch, much like visual systems with high-acuity foveas are subdivided (Catania and Remple, 2004). The mole’s tactile fovea consists of the paired 11th rays at the center of the star (Fig. 13.2A). Whenever moles touch something of interest with rays 1–10, they make a sudden movement of the star to position the 11th rays over the object for additional exploration [Fig. 13.2E and F; Catania (2012, Supporting Information, Movie S2, clips 1–3)]. These movements are similar to visual saccades in their form and time course (Carpenter, 1988; Catania and Remple, 2004).
STAR REPRESENTATION IN THE CNS
The segregated nature of the mole’s sensory rays suggested that there could be a corresponding modular representation within cortical and subcortical areas, which was found for the whiskers of rodents (Woolsey and Van der Loos, 1970; Van Der Loos, 1976; Ma, 1991). This is indeed the case; flattened sections of cortex processed for cytochrome oxidase reveal a complex series of septa and stripes corresponding to the nose representations in several somatosensory areas (Fig. 13.3). Electrophysiological
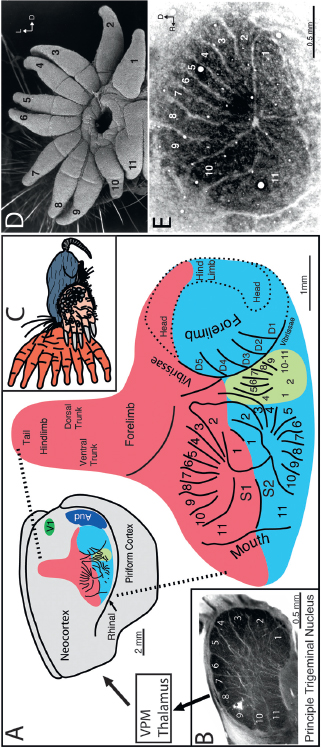
FIGURE 13.3 Mole somatosensory cortex. (A) The size and position of somatosensory areas with representations of different body parts labeled. (B) Anatomically visible star representation at the trigeminal level with each ray representation labeled. (C) A moleunculus showing the relative proportion of body parts represented in cortex. (D and E) The representation of the half star in primary somatosensory cortex with the 11 rays (rotated to match the cortex) under the scanning electron microscope (SEM) and the cortical representation in a cytochrome oxidase stain. Note the large representation of ray 11 (the tactile fovea) relative to its size on the star.
recordings reveal three maps of the contralateral star in lateral cortex (Catania and Kaas, 1995; Catania, 2000a). Each map can be seen as a separate series of stripes representing the nasal rays. The most distinctive area corresponds to the primary somatosensory representation (S1) of the star. The secondary somatosensory area (S2) also contains a large star representation. A third smaller star representation is located just caudal to S2. Injections of neuroanatomical tracers show that S1 is topographically interconnected with the corresponding ray representations in S2 and S3, forming a cortical processing network (Catania and Kaas, 2001). Finally, recent investigation of the principal trigeminal sensory nucleus (Catania et al., 2011) reveals a large, visible representation of the star consisting of 11 modules that bulge out of the brainstem (Fig. 13.3B). The mole’s principal nucleus is proportionally much larger than the corresponding nucleus in rodents (Ashwell et al., 2006).
Four features of the neocortex highlight the specialized nature of star-nosed mole brains. First and most obviously, a large proportion of somatosensory cortex is devoted to the star. This example of extreme cortical magnification is schematically illustrated in Fig. 13.3C. Second, star-nosed moles are the only species with three anatomically visible cortical representations of a single sensory surface. Third, within S1, the 11th foveal appendage is greatly overrepresented relative to its size, the number of sensory organs on its surface, and the number of nerve fibers that supply the ray. This finding parallels the way that visual systems are organized (Azzopardi and Cowey, 1993) and suggests a general organizational framework for the evolution of high-resolution sensory systems [Suga et al. (1975) and Azzopardi and Cowey (1993) discuss bats]. Fourth, star-nosed moles have an extra cortical representation of the nose compared with other moles and shrews (Suga et al., 1987). This finding suggests that star-nosed moles have added a cortical area to their processing network.
OPTIMAL FORAGING AND THE FUNCTION OF THE STAR
Having outlined the unusual and specialized nature of the mole’s somatosensory system, it seems natural to wonder why such a structure evolved. It is not enough to suggest that they simply have a very well-developed sense of touch, because many other moles are touch specialists. What can star-nosed moles do that other moles cannot? A likely answer comes from considering the star-nosed mole’s behavior and environment in the context of optimal foraging theory.
Competition in the Swamp
Star-nosed moles are the only mole species that lives in the muddy soil of wetlands. Unlike typical mole habitats where soil is dense and stable,
tunnels in wetlands tend to be shallow, ephemeral, and interspersed with grassy runways and leaf litter. As a result, the tunnels are accessible to diverse mammals that also feed on the many invertebrates in the nutrient-rich soil. For example, when live-trapping star-nosed moles, we usually capture a greater number of other insectivores (Catania, 2012, Supporting Information, Fig. S1) that share the same tunnels (mostly shrews). Thus, star-nosed moles have substantial competition for prey.
A second feature of star-nosed mole habitats is the small size of the prey compared with more terrestrial settings. Wetlands are a rich source of small invertebrates (Anderson and Smith, 2000). Our preliminary comparisons of invertebrates around the wetland tunnels of star-nosed moles and more terrestrial tunnels of eastern moles (Scalopus aquaticus) found the wetland prey to be an average of 20 times smaller than the prey in the drier habitat (Catania, 2012, Supporting Information, Fig. S1). This finding is consistent with gut content studies of star-nosed moles, which show that they eat large numbers of these small invertebrates (Hamilton, 1931).
Prey Profitability and Star-Nosed Mole Behavior
The former considerations suggest that star-nosed moles live in a competitive environment with diverse prey. With these observations in mind, it is useful to turn briefly to mathematical models of predator behavior for additional clues to answer why the star might have evolved. Foraging theory (Stephens and Krebs, 1986) provides a framework for predicting how predators may behave, assuming that the goal is to maximize the rate of energy gained while foraging. In this paradigm, the rate of energy intake (R) is equivalent to E/(Ts + Th), where E is the energy gained from a prey item, Ts is the time spent searching for prey, and Th is the time spent handling prey (handling time includes pursuit, capture, and consumption of prey). A key variable in considering which prey items should be included in the optimal diet is prey profitability (P). Prey profitability is simply the ratio of energy gained (E) to handling time (Th). Prey profitability has the general form of the equation y = 1/x, with y (profitability) approaching infinity as x (handling time) approaches zero (Fig. 13.4A). In this formulation (often called the prey model) [Stephens and Krebs (1986) have a full treatment], the optimal diet is obtained by adding prey items to the diet if (based on their profitability) they increase the average rate of energy intake or alternatively, rejecting prey items if they decrease the average rate of energy intake while foraging.
The results of these measurements for star-nosed moles are astounding (Catania and Remple, 2005); star-nosed moles have the shortest handling time documented for any mammal when consuming small prey (Catania, 2012, Supporting Information, Movie S2, clips 4 and 5). In a
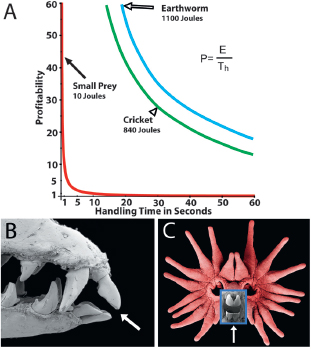
FIGURE 13.4 Profitability of prey. (A) A graph showing the profitability of prey relative to handling time for three different sizes corresponding to different amounts of energy. The red line represents profitability for small (10 J) prey vs. handling time, which was used in mole feeding experiments (Catania and Remple, 2005). For most handling times, small prey items are minimally profitable. However, star-nosed moles have very short (average of 227 ms) handling time, making small prey profitable (filled arrow). Larger prey items are much more profitable overall (green and blue lines) for similar handling times, but they take much longer to handle (open arrowhead and open arrow; 20–30 s), making them similar to small prey in profitability. (B) Unusual front teeth in star-nosed moles (arrow). (C) These teeth are located directly behind the tactile fovea and are used for rapidly picking up small prey (Catania, 2012, Supporting Information, Movie S2 shows tooth use). [Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
laboratory setting, they were able to identify a prey item (small earthworm segments of 10 J energy content), make a saccadic movement to the tactile fovea, and then consume the prey in as little as 120 ms (Catania and Remple, 2005). The average handling time for small prey was 227 ms. When profitability for small prey is plotted relative to handling time, the value for star-nosed moles is surprisingly large (Fig. 13.4A, red line), corresponding to a position high on the vertical asymptote. It seems that star-
nosed moles have come as close as possible to zero handling time. This latter conclusion is supported by the frequent occurrence of double takes when food is first contacted (Catania and Remple, 2005). In these cases, moles contact the prey but briefly move in the wrong direction before foveating to the item (Catania, 2012, Supporting Information, Movie S2, clips 2 and 3), suggesting that nervous system processing of touch lags behind the rapid star movements.
To put short handling time in context, it is important to consider profitability for larger prey items. This profitability is illustrated for a cricket (840 J) and a large earthworm segment (1,100 J) by the green and blue lines, respectively, in Fig. 13.4A. These latter plots of prey profitability vs. handling time dwarf the plot for small 10 J prey. It is telling that star-nosed moles seldom eat chitinous crickets, whereas short-tailed shrews (Blarina brevicauda) eat these crickets and other insects. The handling time for a short-tailed shrew to consume a cricket is roughly 30 s (Catania and Remple, 2005). Remarkably, this time could make small prey items more profitable to star-nosed moles than much larger insects are to competing insectivores.
These considerations suggest that star-nosed moles should include small prey in their diet. However, there is additional evidence to support this interpretation. The front teeth of star-nosed moles are unique among mammals (Fig. 13.4B). They are tiny, with a refined shape that requires the union of two upper teeth and four lower teeth across the midline to form what appears to be a small beak. This tweezer-like structure is located directly behind the somatosensory fovea (Fig. 13.4C), and it is used to efficiently pluck small prey from the substrate (Catania, 2012, Supporting Information, Movie S2, clip 4). The behavioral sequence is closely integrated with star movements, such that the 11th foveal appendages spread apart to accommodate the small teeth. The tiny, specialized teeth are strong evidence of a long evolutionary history of star-nosed moles feeding on small prey. However, it is also important to note that star-nosed moles have larger back teeth for eating larger prey items, especially soft-bodied earthworms (Fig. 13.4B). This finding is consistent with optimal foraging theory, which predicts that large prey items are also profitable (Fig. 13.4A). The conclusion is that star-nosed moles can include small prey items in a broader diet of invertebrates.
Function of the Star
A number of facets of the mole’s behavior and environment suggest that it is adapted to rapidly locate small prey, presumably providing a resource that is difficult for other competing species to exploit. For example, profitability would be many times lower for a competitor that handled
small prey for even 0.5 s longer than a star-nosed mole (Fig. 13.4A, red line). Observations of eastern moles (S. aquaticus) presented with arrays of small prey (Catania, 2012, Supporting Information, Movie S2, clip 6) clearly show that star-nosed moles are more efficient at exploiting this resource. Thus, numerous small Eimer’s organs, modified teeth integrated with the star, and many CNS specializations seem to be adaptations that help to reduce handling time (Th) such that small prey can be exploited.
There is an additional component of the sensory system that can be interpreted in light of optimal foraging theory—the large size of the star compared with the nose of other moles (which also have Eimer’s organs). The expanded surface area means that star-nosed moles contact a large area with each touch, and this area, in turn, reduces time searching compared with the time searching by moles with a smaller array of sensors (Catania and Remple, 2005). Time searching (Ts) is the other part of the denominator used to calculate R, and therefore, minimizing both Th and Ts maximizes the rate of energy intake. Put another way, profitable small prey items are only useful if they can be taken in large numbers, and for that to occur, prey must be located. Thus, optimal foraging theory suggests both the behavior and anatomy of the star-nosed are admirably adaptive.
HOW DID THE STAR EVOLVE?
Having described the function of the star and by extension, the likely selective pressures that led to its evolution, there remains the question of how it evolved. The star is a biological novelty consisting of many appendages, and it might be expected to have evolved by redeployment of conserved developmental mechanisms for appendage formation. Although we do not yet have evidence for genetic patterning mechanisms, the unusual morphology of the developing star seems to tell the story of its evolution (Catania et al., 1999). When the star first begins to emerge in embryos, it appears as if the rays are folded backward on the snout (Fig. 13.5A). However, sections through the developing nose (Fig. 13.5B) show that each nascent ray is simply a swelling of the epidermis with no underlying cell layer to form the bottom portion. Later in development (Catania et al., 1999), a second layer of epidermis extends below the nascent rays to form the bottom wall, and the rays become backward-facing cylinders embedded in epidermis of the face. Shortly after birth, these cylinders emerge, break free, and bend forward to form the adult star. To summarize, the rays develop in place as backward-facing cylinders that later detach and rotate forward (Fig. 13.5C). As a consequence, the tip of each ray is derived from tissue more caudal than the base of each ray, because each ray reverses its orientation during development—an unprecedented mechanism for appendage formation.
FIGURE 13.5 Development reveals evolution of the star. (A) An embryonic star-nosed mole showing the nascent rays. (B) A section of the snout (same stage as A) reveals the rays as swelling or waves in the epidermis with no underlying structure to form a complete cylinder. Later in development, a second layer of epidermis forms under these epidermal waves to form backward-facing cylinders. (C) The developmental sequence illustrated schematically for a single ray. The ray forms in place facing backward and then emerges from the side of the face to bend forward. The tip (arrowhead) is thus formed by caudal snout tissue (orange). (D) An adult coast mole showing extensions of Eimer’s organs attached to the side of the face, which was hypothesized for ancestral star-nosed moles. (E) An early embryonic star-nosed mole nose looks strikingly similar to an adult coast mole nose. (F and G) Congenitally abnormal mole noses with (F) fewer or (G) greater numbers of rays. [Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
Why would such an apparently poorly engineered developmental sequence exist (Jacob, 1977)? Perhaps star-nosed moles evolved from an ancestor with strips of sensory organs on its snout that later raised up and
bent forward over many generations. In the absence of additional evidence, this hypothesis would have to remain very tentative. However, the discovery of a mole with just such an intermediate stage of sensory organs on its (adult) nose provides powerful support for this suggestion. The coast mole (Scapanus orarius) has a series of short strips of Eimer’s organs that extend caudally on the snout. The adult coast mole nose has a striking resemblance to an early embryonic star-nosed mole nose (Fig. 13.5D and E). Of course, the coast mole is not the ancestor of the star-nosed mole, but the existence of this protostar in a living species strongly suggests that such an ancestor to the star-nosed mole existed. A similarity between the adult, ancestral anatomy and an extant embryonic form was predicted by Gould (1977) for developmental sequences that have been built upon with evolutionary changes occurring primarily at the terminal stages of development (Gould, 1977).The result is partial recapitulation of an evolutionary sequence during development (Gould, 1977; Northcutt, 1990).
The most obvious difference between the morphology of adult coast mole sensory swellings and embryonic star-nosed mole swellings is the greater number on the latter. This difference is not hard to account for, because sudden duplications of rays could readily occur. Such meristic changes are common in evolution (Raff, 1996). In fact, we commonly find star-nosed moles with congenitally abnormal noses (Catania et al., 1999). Approximately 5% of star-nosed moles have either greater or fewer than the usual 22 rays (Fig. 13.5F and G). This finding is a high rate of abnormality, much greater than for the tetrapod limb (Castilla et al., 1996; Zguricas et al., 1998). Darwin (1859) predicted this kind of variability in On the Origin of Species by Means of Natural Selection, stating that “in those cases in which the modification has been comparatively recent and extraordinarily great . . . we ought to find the generative variability, as it might be called, still present to a high degree” (Darwin, 1859). Thus, far from being inexplicable (Weston and Wieland, 2003), star-nosed moles provide strong support for basic evolutionary principles, including Darwin’s predictions for rates of variation, Gould’s (1977) theories of the relationship between ontogeny and phylogeny, and the “tinkering” nature of evolution (Jacob, 1977), which often produces new and unusual solutions to old developmental problems.
FISHING SNAKE
At first glance, the rays of the star-nosed mole and the tentacles of the tentacled snake seem superficially similar. Both are flexible extensions of the epidermis on the front of the face. However, the behavior of the two species and the function of their appendages are very different. Star-nosed moles are active explorers that move the rays in a flurry of
motion as they travel through their tunnels or forage in shallow water. In contrast, tentacled snakes are sit-and-wait predators (Fig. 13.6). They are fully aquatic and never leave the water, and they feed exclusively on fish. When hunting, the snake adopts a J-shaped posture (Fig. 13.6A) and waits for fish to enter the concave area formed by the bend of its neck and head. When fish are in this favorable position, the snake strikes explosively and typically reaches the position of the fish in about 25 ms (Catania, 2009). Given this hunting strategy, it seems reasonable to hypothesize that the tentacles function as fish detectors. This idea and others have been suggested for over a century, but only recently, experiments have been conducted to investigate various possibilities. The function of the tentacles was explored using a multifaceted approach that included anatomical investigation of their internal and external anatomy, electrophysiological recordings from the trigeminal afferents and the optic tectum, and behavioral observation based on slow motion analysis of high-speed video recordings under visible or infrared lighting (Catania et al., 2010).
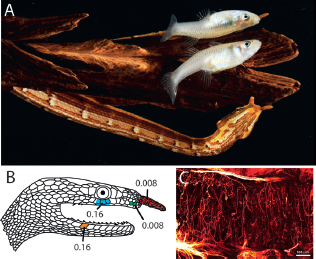
FIGURE 13.6 Tentacled snake hunting posture and sensory appendages. (A) The characteristic J-shaped hunting position for this sit-and-wait predator. (B) Examples of single-unit receptive fields for trigeminal afferents and the lowest forces (grams) that produced action potentials. (C) The dense network of fibers that traverse the center of the tentacle. [Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
APPENDAGES OF TENTACLED SNAKES ARE SENSITIVE MECHANOSENSORS
Two branches of the trigeminal nerve innervate each tentacle. Confocal microscopy of fluorescently (DiI) labeled fibers reveals a dense array of fine-nerve terminals that cross the middle of each tentacle orthogonal to the long axis (Fig. 13.6C). The fibers are poorly placed for detecting details of stimuli that compress the epidermis (in contrast to fibers in Eimer’s organs) but are well positioned for detecting movement of the entire tentacle. Electrophysiological recordings from trigeminal afferents confirm this suggestion (Catania et al., 2010). The tentacles are sensitive to the slightest deflection caused by the finest calibrated von Frey hairs (Fig. 13.6B). When the snake’s head is submerged in water, tentacle afferents also respond strongly to movement of a nearby vibrating sphere used to simulate moving fish. The tentacles are not responsive to electric fields, and there is no evidence of electroreceptors or chemoreceptors on their surface (Catania et al., 2010).
TACTILE AND VISUAL RESPONSES IN THE TECTUM
As would be expected, the snake’s optic tectum is highly responsive to visual stimuli (Catania et al., 2010). Receptive fields for neurons in the superficial layers of the tectum form a visuotopic map of the contralateral eye, with superior fields represented dorsally, inferior fields represented laterally, nasal fields represented rostrally, and temporal fields represented caudally. Compared with vision, tactile responses in the tectum are less refined with larger receptive fields and weaker responses. Nevertheless, the overall topography of the somatosensory representation is in approximate register with the overlying visual representation, suggesting that mechanosensory and visual cues are integrated in this region (Catania et al., 2010).
TENTACLED SNAKE BEHAVIOR
To further explore the function of the tentacles, snakes were filmed under visible or infrared illumination (the latter is used to control for vision). The results under lighted conditions showed that fish seldom approach the tentacles, and therefore, a function as lures seems unlikely. This conclusion is also supported by the observation that snakes seldom strike at fish directly adjacent to their tentacles, probably because it is not possible to generate sufficient striking momentum over short distances. Under 950-nm wavelength illumination (Catania, 2012, Supporting Information, Movie S3), which they cannot see (Catania et al., 2010), tentacled
snakes are still able to strike and capture fish. However, they strike less often in the absence of eyesight and are less accurate. Overall, the results suggest that the main function of the tentacles is to aid in the localization of fish when eyesight is reduced at night or in murky water, thus allowing prey capture in a much wider range of conditions than for vision alone. However, a number of observations suggest that tentacled snakes rely most heavily on visual cues to guide their strikes when possible (Catania, 2009, 2010; Catania et al., 2010). This finding should not be too surprising, and it does not detract from the usefulness of the tentacles. For example, barn owls are renowned for their hearing but have acute vision that guides their attacks when available. Similarly, a pit viper can easily strike visible targets, but this ability does not detract from the use of the infrared-detecting pits for hunting warm-blooded prey at night or in underground burrows. Thus, many species have adaptations that importantly supplement more dominant visual systems.
TURNING THE TABLES ON FISH
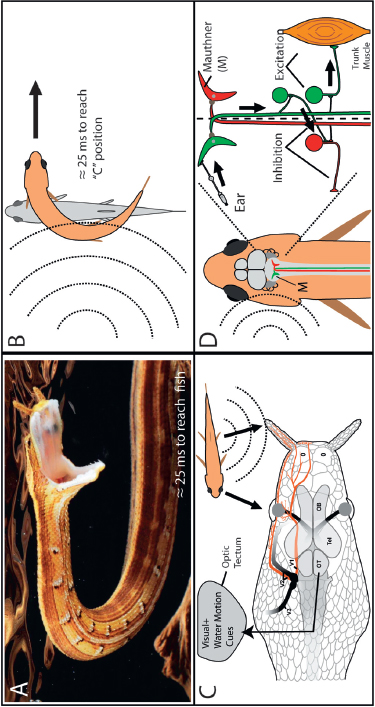
Although tentacled snakes can make an explosive strike with remarkable speed (Smith et al., 2002; Catania, 2009), fish are expert escape artists with a well-studied neural circuitry that mediates high-speed evasion of predators (Zottoli, 1977; Eaton and Hackett, 1984; Faber et al., 1989; Korn and Faber, 2005). The fish C-start escape response has an onset latency of only about 7 ms from the detection of a water disturbance and begins with a C-shaped bend of the body followed by propulsion away from the predator. The C-start is mediated by two giant Mauthner cells (neurons), one cell on each side of the brainstem. Turning away from an approaching predator is important, and the decision about which direction to turn (which Mauthner cell fires first) occurs in the first few milliseconds after detecting a threatening stimulus. Within 25 ms, the fish is in mid C-start and primed to swim farther away. However, it takes a snake only about 25 ms to reach a fish when striking. Thus, the snake’s strike and the fish’s C-start have been consistently pitted against each other for the long evolutionary history of this predator–prey interaction. Fig. 13.7 outlines the senses and behaviors used by snakes and fish in this competition.
In adapting to this long-standing challenge, tentacled snakes have evolved a remarkable strategy to take advantage of the specialized escape circuitry of fish (Catania, 2009). Recall that tentacled snakes prefer to strike at fish that have entered the concave area formed by the J-shaped hunting posture. Just before the strike, the snake feints with its body, which is on the opposite side of the fish relative to the snake’s jaws. As a result, fish usually (~80% of the time) turn away from the snake’s body and thus toward the approaching jaws—sometimes swimming
FIGURE 13.7 Summary of the sensory systems and behavior in the predator–prey interaction between snakes and fish. (A) A striking tentacled snake takes roughly 25 ms to reach a nearby fish (Catania, 2012, Supporting Information, Movie S4). (B) A directional C-start escape response occurs 7–8 ms after sound detection, and within 25 ms, the C-shaped posture is attained. (C) Visual and mechanosensory information converges in the snake’s optic tectum. (D) The Mauthner circuitry mediating fish escape responses. When a threatening sound occurs on the left side, the left Mauthner neuron usually fires first (green), projecting to the right side of the body and stimulating right trunk muscles while inhibiting left muscles (red) to cause a right turn away from the stimulus. [Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
straight into the snake’s mouth (Catania, 2012, Supporting Information, Movie S4). Hydrophone recordings correlated with video recordings confirm that a pressure wave is generated by the snake’s initial body feint (Catania, 2009). These results explain an early description in the work by Cornellissen (1970) that fish are partially swallowed during strikes and the later observation in the work by Murphy (2007) that some fish disappeared completely in a single video frame when strikes were filmed at 30 frames/s, suggesting minimal or even nonexistent handling times (Murphy, 2007). Swimming into the mouth of your adversary certainly reduces handling time.
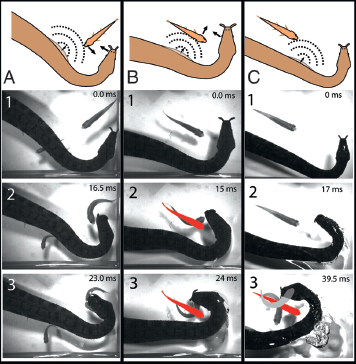
Startling fish toward the strike is an impressive adaptation, but this strategy is only feasible when fish are oriented roughly parallel to the snake’s jaws (Fig. 13.8A). When fish are oriented at a right angle to the jaws, the C-start can translate fish to one side or the other but not directly toward the strike. In these cases, the snake uses an even more surprising strategy—it feints with its body and aims for the far side of the fish, which is the most likely future position of the head (Fig. 13.8B; Catania, 2012, Supporting Information, Movie S5). Most fish turn away from the body feint, often placing their head directly into the snake’s oncoming jaws. Because the strike is ballistic and does not make use of visual feedback, these attacks require a prediction of future fish behavior. The latter is clear from the speed of the strike, which begins before the C-start, and the fact that snakes retract their eyes when they strike (Catania, 2009). It can also be shown by examining trials when no C-start (or opposite C-starts) occurred (Catania, 2009) and snakes usually struck to the most likely (but incorrect) future location of the moving head (Fig. 13.8C).
The snake’s strategy of startling fish toward the strike has the obvious benefit of improving capture success. However, it also has the added advantage that most fish are caught head-first and often partly swallowed, greatly reducing handling time (Murphy, 2007). When this time was measured explicitly by manually presenting fish either head- or tail-first, the former allowed for much shorter swallowing (handling) times. Because tentacled snakes are cryptic sit-and-wait predators, the more quickly they can swallow prey, the more likely they are to remain camouflaged to other nearby fish, thus indirectly increasing their foraging efficiency. In addition, tentacled snakes often exhibit a tail-wiggling behavior when swallowing large fish (when handling time is long). This behavior may distract the snake’s own predators, which would presumably attack the wiggling tail, allowing for escape. This subtle but important behavioral adaptation suggests that snakes are in danger of becoming prey themselves when movement (e.g., swallowing fish) breaks their camouflage. Thus, reduced handling time likely has benefits related to both long-term foraging efficiency and short-term survival.
FIGURE 13.8 Frames captured from high-speed video illustrate tentacled snake strikes. (A) In this trial, a fish is oriented roughly parallel to the jaws. The snake startles the fish to its strike and into its mouth (Catania, 2012, Supporting Information, Movie S4). (B) In this trial, a fish is at an approximately right angle to the jaws. The snake startles the fish and strikes to the future location of the head (Catania, 2012, Supporting Information, Movie S5). The red outline shows the original position of the fish. (C) In this trial, the snake fails to elicit a C-start, and instead, the fish responds to the snake’s moving head, turning to the body (Catania, 2009). Nevertheless, the snake aimed for the approximate future location of the fish’s head had it responded to the body feint (gray; reflected C-start). Small numbers show the number of milliseconds from the first movement of the snake. [Note: Figure can be viewed in color in the PDF version of this volume on the National Academies Press website, www.nap.edu.]
BORN KNOWING
The observation that tentacled snakes can startle fish and predict their future movements raises the question of whether this strategy is an innate ability or learned through a lifetime of striking at escaping fish. This question was addressed in laboratory-born snakes that had never experienced live fish. To prevent the naïve snakes from learning during the trials, snakes were placed in a chamber above fish, separated by a
thin transparency sheet. The transparency sheet (instead of glass) was chosen to minimize the distance between the fish and snake. However, it also had the unexpected benefit of allowing the pressure wave generated by the snake’s body feint to startle underlying fish. As a result, snake movements could be observed in relationship to escaping fish, although snakes could never contact fish (when the flexible barrier was replaced by glass, fish did not respond to the purely visual stimulus of striking snakes) (Catania, 2010).
The results clearly show that tentacled snakes are born with the ability to make predictive strikes (Catania, 2012, Supporting Information, Movie S6). The surprising ability of naïve snakes to predict the future behavior of their prey is a testament to the long evolutionary history of this predator–prey interaction. It is an example of selection acting on innate behavior over the course of evolution in contrast to learning, which selects behavior during an animal’s lifetime. In this sense, tentacled snakes fall on the extreme nature side of the nature vs. nurture continuum, at least for striking behavior. Tentacled snakes also provide a compelling example of the rare enemy effect as outlined in the work by Dawkins (1982). In this scenario, a predator may take advantage of a trait that is usually adaptive. Because tentacled snakes are less common than a host of other predators, the best bet for a fish is to turn away from a water disturbance. It is an unlucky fish that encounters a tentacled snake and makes a wrong turn.
CONCLUSIONS
Specialized sensory systems are inherently interesting to biologists because they represent extremes in the process of evolution. However, they are more than curiosities—they represent a challenge and an opportunity. The challenge is to understand how they function and why they evolved in the context of their environment. The opportunity comes in the form of more general insights into biological processes that may be derived from this understanding. Star-nosed moles and tentacled snakes provide examples of these dual perspectives. Star-nosed moles conveniently show principles of mammalian brain organization owing to the multiple anatomically visible maps of the star in the neocortex. However, they also provide clues to more general biological principles such as theories of predator diet selection or the relationship between development and evolution. Tentacled snakes similarly show how information from different senses is integrated in the tectum and the importance of multiple cues for detecting environmental stimuli. However, the most surprising finding for this species is their ability to startle fish toward strikes, thus taking advantage of the neural circuitry that mediates obligatory fish escape responses. Tentacled snakes provide a concrete example of the
rare enemy effect, which suggests that uncommon predators may tap into prey behavior that is usually adaptive. These various discoveries in two divergent species illustrate the necessity of integrating neurobiological, behavioral, and ecological approaches to best understand adaptations.
ACKNOWLEDGMENTS
This research was supported by National Science Foundation Grant 0844743 (to K.C.C.) and a MacArthur Award.
![]()
PHYLOGENY OF HUMAN BRAINS AND HUMAN MINDS
The chapters in Part IV address the question of human uniqueness in brain organization and behavior. In Chapter 14, Todd Preuss focuses on molecular genetic differences between human brains and the brains of our closest relatives. Particular emphasis is given to the role of foxP2, which has, at times, been called the human language gene. Not surprisingly, the true story of foxP2 is more complex, because as Preuss puts it, “we are trying to relate a multifunctional gene to a complex, high-level phenotype.” To deal with this complexity, Preuss suggests that we need a better understanding not of single-gene variation, but of variation in many genes and, particularly, brain development. Preuss also notes that human brains mature more slowly than the brains of other species, which would explain why brain metabolic activity is surprisingly high and structural plasticity unusually protracted in humans. Particularly interesting is the observation that some patterns of gene expression in the prefrontal cortex of humans are seen only during development in other species. The mechanisms underlying this heterochrony as well as their functional sequelae remain unclear. However, childhood is well known to be more protracted in humans than in other apes.
Lizabeth Romanski reviews in Chapter 15 the anatomical and physiological organization of the ventrolateral prefrontal cortex (vlPFC) of macaque monkeys. This cortical region is of special interest because its homolog in humans includes several language-related areas (e.g., Broca’s area). In a key experiment, Romanski and her colleagues took movies of vocalizing monkeys, separated them into audio and visual streams, and