CHAPTER 3
AQUATIC TOXICOLOGY AND BIOLOGICAL EFFECTS
INTRODUCTION
Difficult and time-sensitive decisions are made during an oil spill response to mitigate the environmental impacts of the spill. Under the appropriate conditions, these decisions may involve the use of dispersants, with a recognition that there are trade-offs associated with this and other response actions (see Chapters 5 and 6). Appropriate response decisions and tradeoff evaluations require the inclusion of sound scientific information. In particular, assessing the ecological and toxicological consequences of dispersant use relies in part on findings from laboratory studies. Since the publication in 2005 of the National Research Council (NRC) study Oil Spill Dispersants: Efficacy and Effects (NRC, 2005), more than 100 research papers and reviews have been published that specifically address topics of the toxicity of oil, dispersants, and dispersed oil mixtures to various biota. Much of the increase in research activity was driven by the requirements of the Natural Resource Damage Assessment (NRDA) and related research in the wake of the Deepwater Horizon (DWH) oil spill (Deepwater Horizon Natural Resource Damage Assessment Trustees, 2016; Jones, 2010). The large volumes of dispersants used during this spill, particularly at depth (~1,500 m), has given rise to a number of renewed questions regarding the toxicity of untreated oil, dispersants, and, dispersed oil mixtures (Deepwater Horizon Natural Resource Damage Assessment Trustees, 2016; Kujawinski et al., 2011). One of the key questions to be resolved is the potential for enhanced bioavailability and toxicity of oil when dispersants are used, balanced against a reduction of potential exposure to surface oil. The proper design of tests that address this question directly, and the use of a correct dose metric for mixtures of oil components that exert toxicity, are central to a scientifically valid assessment.
Comparisons of the conditions of various large-scale spills also may lead to varying conclusions regarding bioavailability and potential toxicity, especially as they relate to the use of dispersants. For example, the chronic release from the former Taylor Energy site at lease block MC-20 and the DWH oil spill represent extremes of acute versus long-term chronic releases of oil into the environment. DWH was a deep release, extending for 87 days and covering a maximum surface extent of about 28,200 km2 (MacDonald et al., 2015). The volume release rate was > 50,000 barrels
(bbl) per day, resulting in contamination of an extensive oil volume extending from the surface to 1,500 m. By contrast, the MC-20 release is ongoing since 2004 (Warren et al., 2014; 15 years at the writing of this document). According to a recent satellite remote sensing survey (Sun et al., 2018), the MC-20 release routinely generates a surface slick extending about 15 km2 (maximum spread of 1,900 km2) from the MC-20 location, with an estimated volume discharge of 48 to 1,700 bbl per day. Both scenarios would seem suited for long-term study of exposure toxicity, especially for sessile and demersal animals; but, while the toxic effects of DWH have been extensively reviewed, there are no such published results for Taylor.
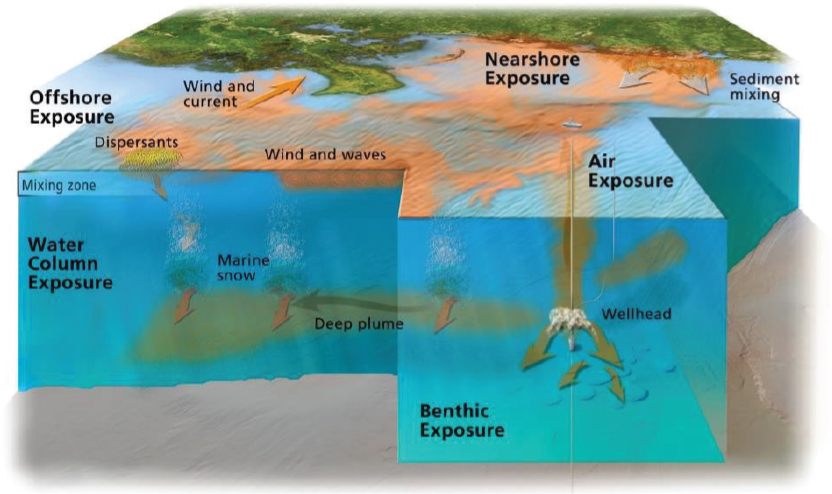
The case of the DWH oil spill illustrated the complex nature of a prolonged spill event as well as the potential for multiple pathways of exposure of biological resources to toxic oil compounds. These key considerations for assessing the toxicity of oil spills in relation to oil and dispersed oil are illustrated in Figure 3.1. Aquatic organisms were potentially exposed to physically- and chemically-dispersed oil due to the formation of deepwater plumes, to surface dispersed oil, and from rising and surfaced oil. Wind- and current-driven oil at the surface resulted in exposure of biological resources in offshore and coastal surface waters and, eventually, in estuaries and coastal barrier beaches. A secondary exposure of benthic ecosystems may have resulted from the formation of oiled marine snow (see Chapter 2), which eventually sank in the region around the wellhead, carrying with it dispersant residues and weathered oil components (Daly et al., 2016; Passow et al., 2012; Valentine et al., 2014). Volatile compounds from oil resulted in the elevated concentration of volatile organic compounds near the wellhead where air-breathing species (marine mammals and turtles) and response workers were potentially exposed. Adverse effects from oil can result from exposures to dissolved aqueous oil components, physical smothering from direct contact, and oil ingestion, inhalation, aspiration, and consumption of oiled prey (see Box 3.1), but these impacts vary by species, life stages, and behavior (e.g., seasonal migrations, benthic feeders).
This chapter synthesizes the state of knowledge on the toxicological effects of physically and chemically dispersed oil, particularly those published since the prior NRC report on dispersants (NRC, 2005), and with emphasis on studies emerging post-DWH. While the information synthesis presented in this chapter relies on recent knowledge, it is important to note that (1) continuous
deepwater releases such as the DWH oil spill are atypical and not representative of most spills; (2) when considering the toxicity associated with dispersant use, the baseline for comparison is untreated oil as opposed to a pristine environment; and (3) dispersants are not a viable response option for most spills in marine U.S. waters, as the vast majority of surface or deepwater oil spills are either too small or too close to shore for dispersant use because of the logistical complexity of a dispersant operation and potential harm to coastal resources and interactions with people (see Chapters 1 and 6).
This chapter first discusses the aquatic toxicity (absorption) and then the biological effects (direct contact, inhalation and aspiration, and ingestion) on exposed organisms. Because of the challenges in differentiating the biological effects of an oil spill with and without dispersant use under field conditions, the primary focus of this chapter is on aquatic toxicity as determined using laboratory tests. Finally, it provides context of toxicity in relation to observed field exposures and discusses potential future advances in the field of oil and dispersed oil toxicity testing. This chapter also identifies areas of uncertainty and outstanding questions, and it makes recommendations for additional studies that could fill critical gaps.
Despite the existence of recommended toxicity testing protocols developed by the Chemical Response to Oil Spills Ecological Effects Research Forum program (CROSERF; Aurand and Coelho, 2005), a review of the existing toxicity studies identified a wide variety in the methods used, including differences in media preparations, exposure methods, and chemical analyses. As
a result, there is no straightforward, consistent means of assessing the central question: whether exposure media containing chemically dispersed oil is more or less toxic than exposure media containing physically dispersed oil.
The committee evaluated two approaches to addressing this question. The first approach is based on the results of toxicity tests using the variable loading toxicity test design. As shown below, this test design can directly assess the difference in toxicity due to the presence of dispersants. The second approach is based on a combination of experimental data with a controlled design and modeling analysis. In particular, models have been developed that can predict the toxicity of oil of varying compositions to a wide variety of organisms (e.g., PETROTOX and Oiltox; as discussed below). This new capability permits a reassessment of the conclusions drawn from the previously available and newly published toxicity data. Finally, recommendations regarding research gaps, priorities for additional research, and “good practices” for the conduct of controlled exposure trials and in situ monitoring are provided.
The methods the committee recommends at the end of this chapter represent a paradigm shift away from developing toxicity tests that attempt to reproduce field exposure conditions. Instead, the committee recommends focusing effort on methods that consistently produce toxicity test results required for calibration and validation of toxicity models at environmentally realistic levels based on concentrations measured in samples from the field during a spill. The toxicity models are then used together with environmental fate models discussed in Chapters 2, 6, and 7 to evaluate the exposure and toxicity associated with various response options, in particular, the potential costs and benefits of dispersant use.
AQUATIC TOXICOLOGY
Research on the toxicity of oil and dispersed oil mixtures has used a wide variety of test biota (mostly invertebrates and fish) exposed to varying dispersant-to-oil ratios (DORs), under varying environmental conditions (e.g., laboratory, mesocosm, field) and varying physical conditions (e.g., temperature, salinity, etc.). Most of the previous studies have been single-exposure design, varying in exposure time and concentrations, measuring lethal and sublethal effects over experimental periods ranging from hours to weeks. Most often, the observation period has been on the order of several days, with the most common being 24-, 48-, and 96-hour experiments. The extrapolation of data from laboratory- or mesocosm-based studies requires calibrating the exposure duration and concentrations from these highly controlled studies to the mosaic of exposure conditions experienced in actual oil spills. This includes different exposure vectors (e.g., exposure to contaminated water, food, and sediments). Thus, when interpreting environmental impacts from actual spills, the baselines (e.g., background concentrations), environmental concentrations, and biodegradation of toxic compounds all are critical elements in estimating real-world toxicity.
A meta-analysis of laboratory studies on the toxicity of dispersants and of studies evaluating the toxicity of both physically and chemically dispersed oil was undertaken following the selection criteria summarized in Appendix F. This meta-analysis focused on studies published between 2005 and 2018, with studies published between 2005 and 2012 obtained from an existing data repository (Bejarano et al., 2016; NOAA ERD, 2015). While most of the data included in this meta-analysis build on the wealth of knowledge generated as a result of the DWH oil spill, data interpretations should not be taken as a reevaluation of damages as performed by the spill’s Natural Resource Trustees. The primary goal of this evaluation is to address the central question of whether exposure media containing chemically dispersed oil is more or less toxic than is exposure media containing physically dispersed oil, and to demonstrate challenges in interpreting toxicity data which were previously limited (NRC, 1989, 2005) by the availability of quantitative information. Box 3.2 includes definitions of toxicological terms and acronyms used in this chapter.
Toxicity of Dispersants in Aquatic Exposures
For as long as dispersants have been used, the toxicity of dispersants and dispersed oil has been the topic of controversy (NRC, 1989, 2005). Dispersant formulations have evolved over the last decades, and the current generation of commercially available dispersants (e.g., Dasic Slickgone, Finasol® OSR52, and Corexit® 9500) contain less harmful chemical constituents than older dispersants (NRC, 1989, 2005). Modern formulations contain nonaromatic hydrocarbons or water-miscible solvents (e.g., ethylene glycol or glycol ethers) and nonionic and/or anionic surfactants. Changes in dispersant formulations aimed at reducing toxicity warrant a brief overview of dispersant-only toxicity data. Such data are used for regulatory approvals to allow dispersant use and for evaluating the toxicity of oil-dispersant mixtures because the toxicity of dispersant alone must be considered.
With the exception of some of the data collected by CROSERF, most dispersant-only toxicity data generated since 2005 used constant static or static renewal tests. A total of 318 unique toxicity records from constant exposure experiments for 68 aquatic species (mostly marine species) were generated between 2015 and 2017 for nine dispersant formulations, with data for Corexit® 9500 accounting for nearly half of all toxicity data (reviewed in Bejarano, 2018). Early life stages, which are generally considered to be the most sensitive, accounted for 43% of all records followed by juveniles and adults (30% and 26%, respectively).
Figure 3.2 is a plot of the acute HC5 concentrations for nine dispersants (Bejarano, 2018; see Box 3.2 for definitions). The acute HC5s range from approximately 1 to 15 mg/L (ppm). While
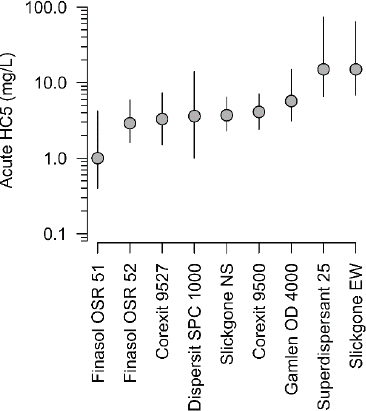
a much larger species diversity has been used in more recent toxicity testing (e.g., Echols et al., 2018), the documented HC5 has remained comparable to previously reported data (Barron et al., 2013; Bejarano, 2018; Bejarano et al., 2016). These HC5s are generally protective for the survival of the most sensitive species (Hemmer et al., 2010; Judson et al., 2010). While most dispersant-only toxicity data are for marine species, freshwater species appear to fall within the same range of sensitivity; thus, HC5 may also be protective for species in freshwater environments (Bejarano, 2018).
These HC5s are derived from dispersant-specific species sensitivity distributions (SSDs) that can be examined for the relative sensitivity of each aquatic species. For example, it is often assumed that shallow-water corals are one of the most sensitive taxonomic groups, but based on limited information for three species, their sensitivities fall within the middle to upper portions of the SSDs and are not grouped toward lower percentiles (Bejarano, 2018).
The data discussed above reflect constant concentration exposures. In some cases, declining concentration (i.e., spiked flow-through) tests with a half-life of ~2.5 hours have been conducted to mimic some field conditions (e.g., Aurand and Coelho, 2005; Clark et al., 2001). Analysis of these studies yielded an HC5 for Corexit® 9500 of 65.8 mg/L (95% confidence interval [CI]: 29.1-299 mg/L), which is 16 times higher than the acute HC5 resulting from 96-hour exposure: 4.1 mg/L (95% CI 2.4-7.1 mg/L). Furthermore, based on operational dispersant application rates (at least at the surface), dispersant-only concentrations are expected to range for minutes to several hours and between 3 and 10 mg/L depending on the characteristics of the application (NRC, 1989). These results, together with previous assessments (NRC, 1989, 2005), clearly point to the need to focus on the hazard posed by physically and chemically dispersed oil, not on the dispersants themselves. The key issue for dispersant use, as was underscored in the previous NRC dispersant studies (NRC, 1989, 2005), is whether the toxicity and exposure potential of untreated oil are less than or greater than that of dispersed oil under actual field conditions and in laboratory toxicity investigations.
Measuring the Toxicity of Oil
Determining the toxicity of oil from aquatic exposures for both physically and chemically dispersed oil is complicated not only by the idiosyncrasies of test conditions used in most studies
(Aurand and Coelho, 2005; Bejarano et al., 2014b; Coelho et al., 2013; NRC, 2005; Redman and Parkerton, 2015) but also by the complex composition and properties of oil. In particular, the following facts need to be considered:
- Separation of the aqueous phase from the bulk oil phase is difficult because oil is an immiscible liquid. Without a physical separation device such as a dialysis membrane, only the density differences can be employed to achieve the phase separation.
- Oil is a mixture of thousands of compounds of widely varying physical and chemical properties, and this complexity both within a single oil and especially across different types of oils influences the toxicity that results.
- Oil is only partially soluble in water. The different compounds dissolve in amounts that vary over five orders of magnitude. The actual amounts are determined by the component concentrations in the oil.
- Determining the dissolved component concentrations in the aqueous phase is difficult because of residual small oil microdroplets that remain in the aqueous phase due to incomplete separation of the aqueous phase from the bulk oil phase.
Because the appropriate interpretation of toxicity data hinges on the testing method used and the degree to which these factors have been appropriately addressed, testing protocols will be discussed first. Toxicity testing protocols consist of three main elements: media preparation, exposure, and chemical characterization. One of the more difficult to resolve aspects is media preparation, which is described in detail below.
Media Preparation
Preparation of WAFs
To evaluate the toxicity of a given oil it is necessary to prepare a test media for the organism exposure. The test medium is composed of a mixture of oil and seawater (simply referred to as “water” in the following discussion) at a specific concentration of oil (e.g., 100 mg oil/L). Mixing is provided for a long enough period of time to attain a reproducible suspension. When oil mixes with water, there is a quantity of liquid oil that remains in the aqueous phase after separation. This is unavoidable because the oil and water have similar densities and some energy must be introduced into the mixture to promote equilibration. Depending on the magnitude of mixing energy, the undissolved remaining oil is distributed in a range of oil droplet sizes, from millimeter to micrometer diameters. These remaining oil droplets are referred to collectively as microdroplets.1 After separation, the resulting aqueous phase is referred to as a water-accommodated fraction, or WAF (see Box 3.2). The WAF is a two-phase system: the oil phase, comprised of microdroplets, and the aqueous phase, comprised of water and the dissolved oil components.
Because the test media is comprised of two phases (oil and aqueous), both the mixing energy used (e.g., low, medium, or high) and the method for preparing the concentration series for the WAFs to be tested have the potential to influence the presence, concentration, and size of microdroplets. These definitions are used in the following section:
Dissolved Concentrations—the concentration of oil components in only the aqueous phase. This has also been referred to as “truly” or “freely” dissolved components.
___________________
1 As explained in Box 3.2 and the corresponding footnote, this definition is for the purposes of this chapter.
Total Concentrations—the sum of the concentration of oil components in the aqueous phase and the oil phase. It is the mass of oil contained in the aqueous phase and in the microdroplets per unit bulk volume of solution.
Method for Creating a Concentration Series: The Effects of Microdroplets
Two different methods have commonly been used to create exposure treatments for toxicity tests: variable loading and variable dilution (NRC, 2005). While the pros and cons of each of these methods have been extensively discussed (Aurand and Coelho, 2005; Barron and Ka’aihue, 2003; NRC, 2005; Redman and Parkerton, 2015; Singer et al., 2000, 2001), there does not appear to be a consensus among the scientific community on a preferred approach. In order to decide which design is preferable, a discussion is presented below that evaluates concentrations that result from variable loading and variable dilution experimental procedures.
Variable Loading
Variable loading toxicity tests consist of a series of individually prepared solutions at variable concentrations of oil (mg oil/L water) used to create a series of loadings, for example, 100, 50, 25, …, 0.25 mg oil/L. The loading level that results in a 50% mortality to the test organisms is known as the lethal loading 50 (LL50). It is found by fitting a dose response curve to the observed mortality versus loading. The discussion below focuses on the effect that the presence of microdroplets has on the resulting toxicity.
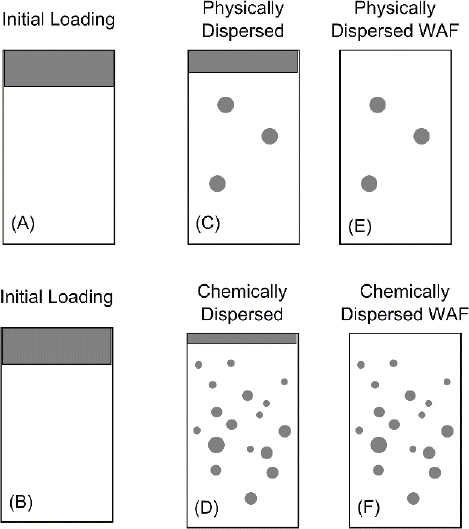
Consider a comparison of a physically dispersed LEWAF (see top row of Figure 3.3) and a chemically dispersed CEWAF oil (see bottom row of Figure 3.3). The initial state of the bulk oil-water system is shown in Figure 3.3A and B: oil floating on water. After physical (C) and chemical (D) dispersion, oil droplets are formed. Note that the chemically dispersed oil has a higher concentration of microdroplets. The aqueous phase is separated, resulting in the physically (E) and chemically (F) dispersed WAFs (LEWAF and CEWAF, respectively).
The mass of oil is the same in each vessel (C and D) before the solution is decanted from the bulk floating oil. Once the solutions are mixed for a sufficient time to achieve equilibrium, the dissolved concentrations of each component are the same in (C) and (D). The reason is that the mass of oil is equal in both vessels, and therefore the quantity of material that dissolves into the aqueous phase is the same. The critical point is that the state of disaggregation of the oil—that is, the concentration of microdroplets—does not affect the resulting dissolved concentration of oil components at equilibrium. It is controlled only by the mass of oil, which is the same in (C) and (D).
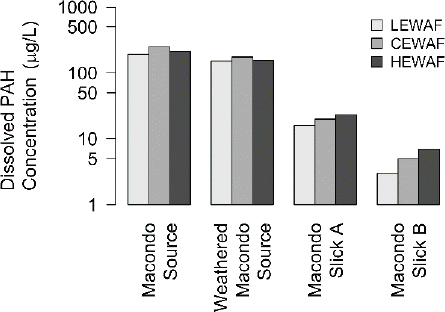
The WAFs are prepared by decanting the mixture of aqueous phase and microdroplets from the bulk oil to create the final LEWAF (E) and CEWAF (F) used for the exposure phase of the toxicity test. These two solutions, LEWAF (E) and CEWAF (F), have the same dissolved concentrations because a removal of a portion of the oil phase does not affect the equilibrium dissolved concentration. However, they have different total concentrations because the concentration of microdroplets is larger in the CEWAF. This difference has important consequences when the toxicities of these two solutions are compared in order to judge whether CEWAFs are more or less toxic than LEWAFs. The equality of dissolved concentrations is documented in a recent study by Forth et al. (2017) that compared total polycyclic aromatic hydrocarbon (PAH) dissolved concentrations from filtered samples of LEWAF, CEWAF, and HEWAF (see Figure 3.4). The remaining differences may be due to the degree to which filtering removes the microdroplets and the possibility that filtering alters the dissolved concentrations by sorption to the filter.
The difference in total concentrations between WAF and CEWAF can be used to test directly whether the differing concentrations of microdroplets change the toxicity of the WAFs. Figure 3.5 pres-
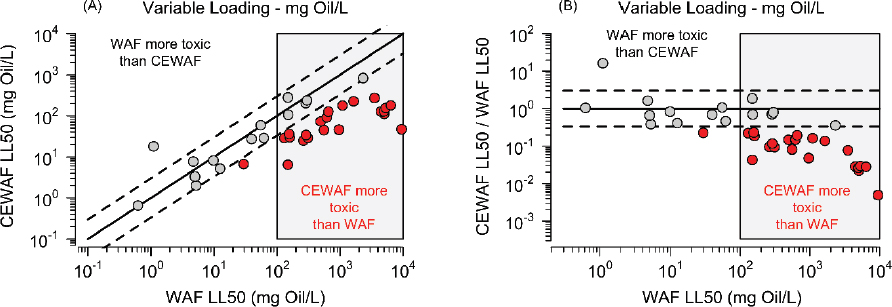
ents the available data from parallel variable loading experiments comparing CEWAFs (prepared with Corexit® 9500) to WAFs.
Figure 3.5A compares the LL50 lethal loading concentrations, the concentration of oil that causes 50% mortality for the CEWAF and WAF experiments. Figure 3.5B compares the ratio of CEWAF to WAF LL50s. In both cases, the solid line denotes equality of concentrations and the dashed lines are a factor of 1/3 and 3 variation. The red points denote experiments in which the CEWAF LL50 concentrations are less than the WAF LL50 by a factor of three, indicating that the presence of dispersant has increased the toxicity because a lower concentration causes 50% mortality. This analysis indicates that at lethal loadings less than approximately 100 mg oil/L, the addition of Corexit® 9500 does not affect toxicity. This is an important result because field measured concentrations during oil spills are typically well below this concentration. In contrast, for lethal loadings greater than approximately 100 mg oil/L, the CEWAF LL50 is lower than the WAF LL50 by at least a factor of 3. The difference is due either to the presence of more microdroplets in higher concentrations in the CEWAF relative to WAF, or that the concentration of Corexit® 9500 is high enough in the CEWAFs for it to be increasing toxicity. At 100 mg oil/L and the highest DOR used in these tests (1:20), the concentration of Corexit® 9500 would be 20 mg/L, which is above its acute HC5 (4.1 mg/L; see Figure 3.2). This suggests that at higher loading concentrations Corexit® 9500 could be contributing to the additional toxicity. However, a more detailed investigation is required in order to separate these effects more reliably.
The advantage of the variable loading method when creating test solutions is that it provides an initial answer to the central question posed in this chapter: whether exposure media containing chemically dispersed oil is more or less toxic than exposure media containing physically dispersed oil. Based on the committee’s current analysis of this limited data, the answer is that at loadings below approximately 100 mg oil/L, when the solutions are at equilibrium, the toxicity of the WAF is equivalent to the CEWAF. Note that the CEWAF solutions will reach equilibrium faster than WAF solutions due to the larger surface area of smaller microdroplets. However, at equilibrium, the addition of dispersant does not appear to increase the toxicity of the oil at loadings below approximately 100 mg oil/L. It should be pointed out that this is a first attempt at answering this central question with the data that are available and that can be used to provide an answer that is not confounded by other complications.
Variable Dilution
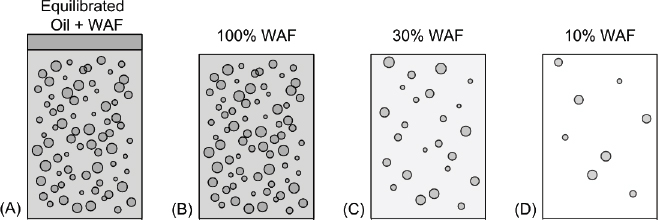
The majority of toxicity tests conducted since the DWH spill have been performed using variable dilution. In this method, rather than preparing individual solutions via the variable loading method described in the previous section, a single stock solution is prepared at a high oil loading (e.g., 1,000 mg oil/L) and a WAF is prepared. This single WAF solution is progressively diluted to create a series of different concentrations as depicted in Figure 3.6. The concentration of oil is equilibrated (see Figure 3.6A), and a series of WAFs is made by serially diluting the aqueous phase plus microdroplets (see Figures 3.6B, C, and D).
The reason why this method is thought to be attractive is that it is assumed that only the WAF produced initially (see Figure 3.6B) needs to be chemically analyzed, reducing the costs for analytical chemistry. The concentrations of all the dissolved components in the WAFs for the subsequent dilutions (see Figures 3.6C and D) are estimated by multiplying the measured concentrations in the original WAF (see Figure 3.6B) by the dilution factor.
However, when microdroplets are present, they invalidate the assumption that the dissolved concentrations can be estimated using the dilution factor (as also discussed in Chapter 2). The dissolved concentrations in (A) and (B) are the same because steady-state has been achieved and separating the WAF from the bulk oil does not disturb the dissolved concentrations. The diluted WAFs (C and D) have a smaller microdroplet and dissolved concentrations because new uncontamined water is added to (C) and (D). As the system approaches a new equilibrium, the dissolved concentrations increase and the component concentrations in the microdroplets decrease. The reason why is that when the WAF is initially diluted the dissolved concentrations of the oil components decrease. This disrupts the equilibrium between the microdroplet oil and aqueous phases in (B), which causes an additional dissolution of oil components from the microdroplets to the aqueous phase, as also described in Chapter 2. This increases the dissolved concentrations and decreases the microdroplet concentration and composition until equilibrium is again achieved. As a consequence, the dissolved concentration does not decrease in proportion to the dilution.
As shown subsequently, the difference between the dissolved concentrations estimated by dilution and the actual dissolved concentrations can exceed one to two orders of magnitudes depending on the concentration of microdroplets in the WAF. As a result, comparing parallel variable dilution experiments with or without a dispersant cannot be used to distinguish unambiguously the effect of the dispersant. In summary, the microdroplets serve as a buffer for delivering and maintaining hydrocarbons in the aqueous phase. They are a source of dissolved constituents that reach an equilibrium after each dilution. Therefore, the dissolved concentrations at equilibrium are higher than that expected by the dilution process alone.
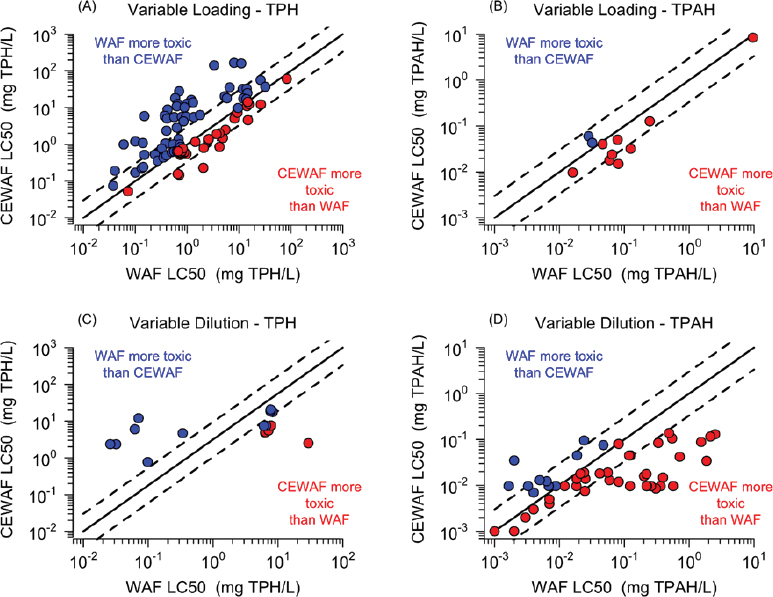
The problem can be seen in the comparison made in Figure 3.7. This comparison is analogous to that presented in Figure 3.5, where parallel variable loading CEWAFs and LEWAFs test results are cross plotted. However, instead of expressing toxicity in terms of LL50s, lethal concentrations (LC50s) are adopted as the exposure metric.
Because there are many individual oil components in the aqueous phase, a “concentration” needs to be defined. Two different concentrations are used to quantify the amount of oil components present in the WAF or CEWAF. Total petroleum hydrocarbons (TPHs) is the arithmetic sum of the concentrations (mg component/L) of either all the hydrocarbon components measured or determined analytically as a group. Total polycyclic aromatic hydrocarbons (TPAHs) are restricted to the arithmetic sum of only the aromatic hydrocarbons, and in some cases to only PAHs with two or more rings. The implicit assumption is that each component is equally toxic and that the arithmetic sum is the proper parameter or dose metric that correctly predicts the toxicity of the mixture. This is not a valid assumption because the toxicity of the various components of oil vary by up to four orders of magnitude. Their toxicity is discussed in more detail subsequently in the section “Toxicity of Mixtures.”
The top row of Figure 3.7 (A and B) compares the data from variable loading experiments where the LC50s are either TPH concentrations (A) or TPAH concentrations (B). The bottom row (C and D) compares the data from variable dilution experiments where the concentrations are either
(C) TPH or (D) TPAH. All the concentrations are from unfiltered samples. The solid line is 1:1 and the dashed lines are factors of 1/3 to 3 variation. Blue points denote CEWAF LC50 > WAF LC50 indicating that WAF is more toxic than CEWAF. Red points denote WAF LC50 > CEWAF LC50 indicating that CEWAF is more toxic than WAF.
For the variable loading experiments (top row), the TPH LC50s (A) scatter about the 1:1 line, with the WAF somewhat more toxic than the CEWAF. This appears to contradict the results from the variable loading experiments (see Figure 3.5) that used lethal loading LLC50s as the dose metric. However, depending on the analytical method used, the TPH measurement may include all the oil in the sample, including the residual bulk oil, and therefore be equal to the oil loading used in the test. Therefore, for the variable loading tests the TPH LC50 may approximate the LLC50s. One would expect that Figure 3.7A would give a similar result: no increased toxicity in CEWAFs relative to WAFs because all concentrations are < 100 mg oil/L. It is unclear why the data in Figure 3.7A indicate that the WAF is more toxic than the CEWAF.
Though based on a smaller number of test results, the comparison using TPAH LC50s (B) indicates the reverse. This suggests that TPH and TPAH concentrations are not directly related to the observed toxicity. They are arithmetic sums of concentrations that bear no relationship to the toxicity of the individual compounds in the sum and are unlikely to represent all the petroleum components that influence the toxicity. Because the components vary widely in toxicity, the arithmetic sum is not a precise aggregate representation of the cumulative toxicity present. This problem will be discussed later in this chapter, when the concept of toxic units (TUs) is introduced as the appropriate aggregate measure of the toxicity of the mixture.
The bottom row of Figure 3.7 compares the results for variable dilution experiments. The TPH comparison (C) suggests that WAFs are more toxic than CEWAFs, while the TPAH comparison (D) indicates the reverse. The data are widely scattered and contradictory. There are two problems with the variable dilution experiments. The first is in common with the variable loading experiment LC50s, namely, that TPH and TPAH are not sensible aggregates that adequately index toxicity. The second and apparently more serious problem is that the concentrations of each of the components are estimated from the dilution factor. As highlighted above, large errors result due to microdroplet dissolution. This point is discussed further later in the chapter. It is clear from these results that these toxicity data in their present form cannot be used to determine if exposure media containing chemically dispersed oil is more or less toxic than that containing physically dispersed oil. Variable dilution WAF and CEWAF tests can be objectively compared if the dissolved concentration and composition of the components are measured at each dilution step instead of being estimated from the dilution factor. For the variable loading experiments, the minimum chemistry required is the total oil and dispersant concentrations to ensure that the nominal quantities had in fact been added.
Some experiments have been performed where the samples were measured for total concentrations and then filtered to measure dissolved concentrations. The results are presented in Figure 3.8. The aggregate is TPAH50, the arithmetic sum of 50 PAHs. Four oils are included: Macondo source, artificially weathered Macondo oil, and field collected Macondo weathered oil—Slick A and Slick B. The left column (A, C, and E) compares the filtered to the unfiltered TPAH50 concentration. The right column (B, D, and F) plots the ratio of unfiltered to filtered concentrations versus the unfiltered concentration. The WAFs are LEWAF and MEWAF (A, B), CEWAF (C and D), and HEWAF (E and F). For LEWAFs and MEWAFs, there is no difference between filtered and unfiltered TPAH50 concentrations, indicating that the microdroplet concentration is small relative to the dissolved concentration. However, for CEWAF and especially for HEWAF, the difference is large with the ratio of unfiltered to filtered TPAH50 concentration in HEWAF reaching two orders of magnitude for Macondo source oil. Note that the data form unity slope lines for HEWAF for each oil as the concentration of unfiltered TPAH50 increases, indicating that the dissolved TPAH50 concentrations are remaining constant and the increases are due to increases in microdroplet concentration.
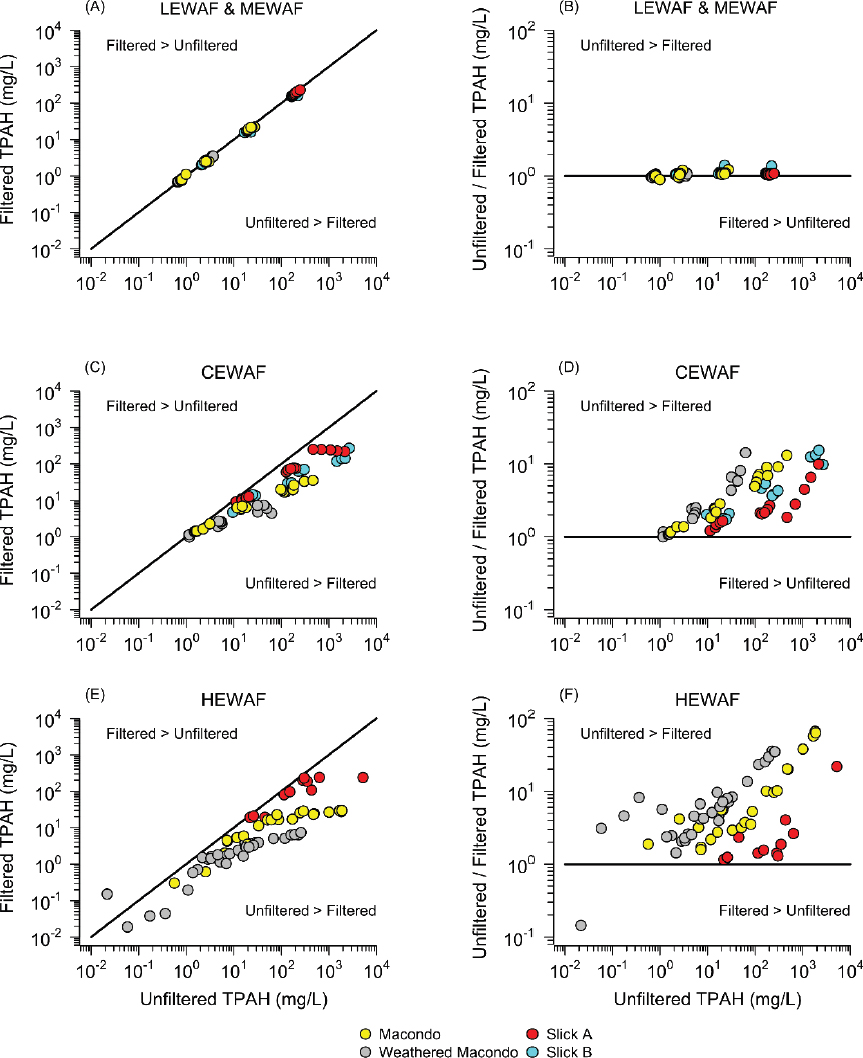
These data indicate that the unfiltered total concentrations can differ from the filtered dissolved concentrations by one order of magnitude for CEWAFs and up to two orders of magnitude for HEWAFs. Because this ratio is varying widely, it is not possible to distinguish between the effects of dissolved components and microdroplets. Furthermore, PAH composition would also vary, further modulating toxicity, a point that is discussed in detail later in the chapter.
Variable loading tests directly address the question of whether the toxicity of physically and chemically dispersed oil differs. The comparison between the two is direct, because the dissolved concentrations at equilibrium are equal in both the LEWAF and the CEWAF for the same oil loading. The only difference is the amount and degree of aggregation of the undissolved droplet oil and the added exposure to dispersant. Thus, a comparison of LL50s derived using the variable loading method with and without dispersant allows the effect of dispersant on oil toxicity to be elucidated as illustrated previously in Figure 3.5.
As demonstrated above, interpretations based on total concentration, which include microdroplets, complicate the problem of understanding the toxicity of WAF versus CEWAF; the relationship between measured concentrations in the laboratory and field samples; and interpretations of observed toxicity. These and related conclusions (e.g., Redman and Parkerton, 2015; Sandoval et al., 2017) collectively emphasize the need to address the issue of microdroplets such that adequate interpretations of toxicity data may be made in the future. Estimation of toxicity values from variable dilution preparations should only be made based on analyses on filtered samples at each dilution and not solely on unfiltered samples in WAF stock, as is often the case for standard toxicity testing.
As an example of the difficulty of interpreting data, a study with early life stages of mahi-mahi found that the acute toxicity of CEWAF was higher than in HEWAF when comparisons were based on TPAH total concentrations, while their toxicities were comparable based on dissolved concentrations of TPAHs and 3-ring PAHs (Esbaugh et al., 2016). However, this study found higher lethality of weathered oil (Slick A) compared to source oil on the basis of either total or dissolved PAH, but not on the basis of dissolved 3-ring PAHs. These findings provide an example of how different metrics of toxicity could lead to divergent conclusions on the toxicity of different oils, pointing to the importance of hydrocarbon composition in the exposure media (see discussion on TUs below). These results are consistent with a test conducted with red drum embryos showing that cardiotoxic phenotypes of weathered oil prepared under LEWAF and HEWAF mixing energies were similar when expressed based on estimated dissolved TPAH concentrations (Morris et al., 2018).
The above discussions are not meant to suggest that oil microdroplets be removed from toxicity testing, as they may enhance the uptake of bioavailable PAHs through adhesion to outer embryo tissues (e.g., Carls et al., 2008; Redman et al., 2014; Sørhus et al., 2015). Instead, efforts should focus on understanding the influence of microdroplets on toxicity test results where the dissolved concentrations are measured and the appropriate TU dose metric is employed to quantify the toxicity of all the dissolved components. The additional toxicity, if any, can then be tentatively ascribed to the factors other than the dissolved concentrations, including microdroplets.
From an experimental point of view, the discussion above describes the practical advantages of the variable loading preparation method. An outstanding issue raised by the National Research Council (NRC, 2005) remains unresolved, however: namely, a conclusive demonstration of which method more appropriately simulates the complex time variable and compositional variation characteristic of conditions during a spill and spill response. While this question remains unanswered, an argument could be made that in order to allow laboratory to field extrapolations of toxicity data, predictive models would need to be employed. This could only be achieved, though, by using toxicity data from tests that address the issues introduced by the preparation method. A clear difference between the findings of the previous study (NRC, 2005) and this updated report is that empirical data are now available to support the technical basis of the variable loading method. This method
produces data that can be used directly to answer the question whether exposure media containing chemically dispersed oil is more or less toxic than is exposure media containing physically dispersed oil. It can also be used to evaluate the performance of the available toxicity models that are used to predict toxic impacts in field exposures. Thus, the finding presented here may facilitate discussions toward consensus on a preferred preparation method among the scientific community. Given the different approaches used by various investigators (as discussed above) the benefits and limitations of various approaches are summarized in Table 3.1. This table provides an evaluation of various solution preparation methods, other exposure media preparations (e.g., contaminated sediments), the exposure regime/tempo, and options for quantifying the chemistry of toxic exposures.
MODELING THE TOXICITY OF OIL
In addition to its comparisons of media test preparation protocols, exposure durations, and chemistry approaches, the committee also considered the strengths and weaknesses of three approaches that have been used to quantify the toxicity of water-oil mixtures and arguments that have been made to support the use of TUs. The TU approach as advocated in this chapter implicitly assumes that (1) the toxic compounds can be identified; (2) their toxicity as individual compounds is known; and (3) if toxicity is then estimated, it is under a baseline minimal toxicity that does not account for the known specific mechanisms of PAHs and heterocyclic aromatics. All other dose metrics have the same requirements—that is, only the identified components with known concentrations can be added together into TPAH and TPH. Dose metrics based on known specific mechanisms need to be applicable to the evaluation of the toxicity of oil-water mixtures. A test of the utility of the target lipid model (TLM) and TUs for the prediction of chronic TUs has been published (Redman et al., 2017). A comparison of predicted and observed “no effect loadings” (Redman et al., 2017, Figure 5) and the observed dose responses versus chronic TUs (Redman et al., 2017, Figure 6) demonstrates the extent to which the TLM-TU model incorporated into PETROTOX correctly reproduces observed toxicity.
Understanding the toxicity of oil is complicated due to the nature of oil and its behavior in WAFs. The problems outlined above can only be addressed quantitatively using a model of the toxicity of oil that explicitly includes the processes that influence the distribution of components between the oil and aqueous phases and includes an appropriate aggregate dose metric that deals appropriately with the fact that individual components have widely differing toxicity. The next sections outline the present state of development of models of oil toxicity. The topics covered are
- The toxicity of the individual petroleum hydrocarbons in an aqueous solution that make up the components of the oil;
- The toxicity of a mixture of these compounds in an aqueous solution; and
- The relationship between the oil composition and the dissolved aqueous concentrations of the compounds that results when oil and water mix.
Toxicity of Hydrocarbons
The aquatic toxicity of hydrocarbons has been studied extensively. They exert acute toxicity via a narcotic mode of action. Studies have reported the relationship between the toxicity of individual narcotic chemicals and their octanol-water partition coefficient KOW (Di Toro et al., 2000; McGrath and Di Toro, 2009; Van Leeuwen et al., 1992; Veith et al., 1983). The TLM was developed to predict the toxicity of PAHs in particular and narcotic chemicals in general (Di Toro et al., 2000; McGrath and Di Toro, 2009). It is applicable to a wide variety of aquatic species with only one parameter required for each species. The TLM was initially calibrated using a large collection of mortality
TABLE 3.1 Benefits and Limitations of Various Methods for the Preparation of Water Accommodated Fractions (WAFs) and Other Media Preparation for Toxicity Testing of Oil in Water
| Solution Preparation Protocols | Benefits | Limitations | |
|---|---|---|---|
| LEWAFa Low-energy WAF | Deionized water added to aspirator bottle; stir bar set at 300 rpm to prevent any vortexa | Simulates passive dosing, method results in few dispersed droplets | May not account for realistic mixing energies, particularly resulting from a blowout |
| MEWAF Mid-energy WAF | Solution derived from vortex mixing (20%-25%) using magnetic stir bar with 18-24 hrs mixing and 2-8 hrs settlement period | Realistic mixing energies in terms of field-level extrapolation | Likely more variability in mixing in field applications than implied from laboratory protocol |
| HEWAFa High-energy WAF | Solution derived using the lowest speed with a food blender for a 30 sec blend and 1 hr settlement period | Results in dispersion of many (most) microdroplets | Unrealistic in terms of real-world mixing energies |
| SHEWAFb,c Super-high-energy WAF | Solution derived from the lowest blending speed using a food blender with 120 sec blend and 1 hr settle | Results in dispersion of many (most) microdroplets | Unrealistic in terms of real-world mixing energies |
| Variable dilution method | Solution derived from a single oil and water mixture (stock solution) from which dilutions are prepared | Standard stock solution prepared once at initiation of experiments | Dissolution of oil components from microdroplets results in changing concentrations of dissolved oil components |
| Variable loading method | Individual test solutions are prepared using variable concentrations (loadings) of oil | Multiple test solutions are prepared instead of one stock solution | This dissolved concentration is the same with and without dispersant added |
| Variable dispersant: oil ratios | Range of dispersant oil ratios used 1:10 to 1:100 dispersant-to-oil ratio to reflect both surface and subsurface use | Allows optimization of dispersant application | Subject to microdroplet induced changes in ratios if used with variable dilution method |
| Other Exposure Media Preparation | Benefits | Limitations | |
| Oil emulsionb | Solution derived from a low blending speed using a food blender with 30 sec blend and used in its entirety without settlement | Allows interpretation of weathering oil | Complicated and rapidly changing weathering scenarios |
| Oiled sedimentb | Sediments mixed with oil for 30 minutes at a moderate speed using either a large food preparation mixer or concrete mixer | Allows more complete interpretation of exposure vectors | Oiled sediments may be more layered than mixed laboratory preparations, subject to bioturbation and wave/current action in shallow waters |
| Sediment-derived WAFb | Supernatant derived by adding water to contaminated sediments then either stirring for 10 seconds followed by 2 hrs settle, or by shaking at 300 rpm for 6 hrs with 12 hrs settle | Simulates secondary water-based contamination from sedimented oil | Difficult to relate the cause of toxicity to oil composition and sediment properties |
| Solution Preparation Protocols | Benefits | Limitations | |
|---|---|---|---|
| Exposure Regime | Benefits | Limitations | |
| Static renewal or nonrenewal | Refers to a constant exposure in which there is test solution renewal at regular time intervals (typically 24 hrs) with fresh test solution made at the same concentration or there is no test solution renewal | Constant exposure concentrations allow the construction of dose-response curves | Unrealistic assumptions of constant concentrations from actual oil spills |
| Co-stressors | Influence of co-stressors investigated (i.e., UV light, temperature, dissolved oxygen | Real-world oil exposures are complicated by these co-varying stressors that may be additive or multiplicative | Complicated to understand and requires factoral designs for experiments |
| Chemistry | Benefits | Limitations | |
| Total PAH (TPAH or ∑50PAH) concentration | Arithmetic sum of PAHs, including alkyl homologues by GC/MS/SIM, individually quantified and generally comprised of 50+ PAH compounds (MC 252 QAPP, 2011) | Quantifies exposure to arguably the most toxic/carcinogenic constituents of oil | Does not weight the relative toxicity of various PAH compounds |
| Total petroleum hydrocarbon (TPH) concentration | Measures the total hydrocarbon concentration using a solvent extraction and quantification using 1-D GC-FID that typically sums from a lower cutoff up to typically C38 carbon number | Measures all TPH concentrations up to the very heavy components. If all the oil is measured before separation, this is equivalent to the oil loading. | No distinction between the variations in toxic components and additionally the highly weathered components that are not toxic |
| Chemical partitioning (dissolved/particulate phases) | Pass test solution through 2 stacked GF (0.3 µm) filtersd,e,f or glass wool and 1µm GF/F and 0.7 GF/F2 under gentle vacuum and analysis of filtrate and non-filtered test solutions | Removes dissolved droplets (depending on size) from test media thereby allowing interpretation of toxicity to dissolved oil | Microdroplets are a reality of oil spills and contribute to the overall exposure burden |
| Quantitation of droplets | Recommend analysis of size and distribution | Allows complete interpretation of oil in both droplet and dissolved manifestations | Difficult to understand the relative contributions of droplets and dissolved components in mixtures |
NOTE: Modified from original table proposed by Mitchelmore et al., 2020.
data and subsequently has been extended to include a wider range of organisms from the initial 33 species to 79 species (McGrath et al., 2018). The TLM predictions of the acute LC50 data of single monoaromatic hydrocarbons (MAHs) and PAH compounds for various species are generally within a factor of 2 to 3 of the observed toxicity over a concentration range of approximately 10 µg/L to in excess of 100 mg/L (McGrath and Di Toro, 2009, Figure 1).
Chronic criteria based on sublethal endpoints have also been developed that protect the organism from a lifetime exposure (i.e., development as measured by growth and weight at maturity and reproductive success). The parameter that has been used historically to estimate the chronic criteria is the acute-to-chronic ratio (ACR; Stephen et al., 1985). The chronic criteria are computed by dividing the acute criteria by the ACR. Following this approach, ACRs for individual petroleum hydrocarbons have been developed, resulting in a probability distribution of species geometric average ACRs (McGrath et al., 2018). These updated ACRs facilitate extrapolation of acute to chronic toxicity values.
Modern chronic criteria, first utilized by the European regulatory agencies, are based on the HC5 concentration (Aldenberg and Slob, 1993), which is established by treating the variations in species sensitivity and the ACR as probability distributions and computing the concentration that protects 95% of the tested species. The use of the probability distributions of both the SSD and the ACR accounts for the variation in the SSD and the ACR for the tested species. Based on the updated ACRs, chronic TLM-derived HC5s for baseline narcotics, MAHs, and PAHs were developed, confirming that these values are protective of species for which chronic data are available (McGrath et al., 2018).
Toxicity of Mixtures: Toxic Units
The soluble oil components contribute to aquatic toxicity to variable degrees, and a model is needed to quantify the toxicity of mixtures. For components that exert their toxicity through a narcotic mode of action, the use of TUs has been shown to correctly predict observed toxicities (Hermens, 1989).
The TU concentration for a specific compound is defined as the ratio of the dissolved aqueous concentration of the compound, CW, to the toxic concentration, either LC50 or HC5 of that compound, as shown in Equation 1.

For a mixture of N compounds with concentrations CW,1, CW,2, …, CW,N, the predicted TU concentration for the mixture TUT is the sum of the individual TU concentrations, as shown in Equation 2.
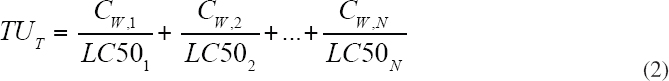
If the total TU concentration TUT = 1, the mixture is predicted to cause a 50% effect if LC50s are used for the effect concentration in Equations 1 or 2. Equation 2 has been validated using data from a number of studies, using mixtures of up to 50 compounds (Hermens, 1989). The toxicity of mixtures of PAHs has also been tested, and the predictions based on TU additivity have been shown
to be within the usual range of a factor of two or three, consistent with single chemical toxicity predictions (Di Toro and McGrath, 2000; McGrath et al., 2005; Redman et al., 2012b).
Modeling the Toxicity of Oil—PETROTOX
The PETROTOX model has been developed to predict the aquatic toxicity of water in contact with oil (Redman et al., 2012b). It uses the TLM to predict the toxicity of the individual oil components and the TU mixture model to predict the toxicity of the mixture. An earlier model Oiltox (French-McCay, 2002) also used the TLM and TUs as the basis for predicting the toxicity.
PETROTOX is comprised of four parts:
- The composition of the oil being evaluated is specified as a mixture of a number of blocks. For each block, a chemical is selected whose physical and chemical properties are similar to the concentration of the chemicals found in that block. This block approximation is designed to be compatible with the two-dimensional gas chromatographic (GC×GC) method that is used to quantify the composition of the oil (Reddy et al., 2012). For the Macondo oil, there are 318 non-zero block concentrations that comprise the input. PETROTOX includes both a high and a low resolution blocking scheme (e.g., aromatic versus aliphatic), which makes it applicable to a lower resolution analysis of the oil.
- The model for the WAF determines the dissolved concentrations using Raoult’s Law and the composition of the oil phase. The equilibrium composition is computed for both the oil and the aqueous phase (Di Toro et al., 2007).
- The TLM is used to compute the LC50 for acute toxicity or for chronic toxicity using the ACR, as described above, for each of the dissolved concentrations.
- The TU model (Equation 2) adds the TUs to produce the total TUT for that oil.
PETROTOX predictions have been compared to experimentally determined toxicity of various oil samples using LL50 concentrations obtained from variable loading tests (Redman et al., 2017). The types of petroleum products that have been tested are naphtha, kerosene, gas oils, heavy fuel oils, distillate aromatic extracts, residual aromatic extracts, and other residual classes. Organisms tested are fish, zooplankton, and algae. The algae and daphnia predictions are generally within a factor of three. The predicted LL50 for fish is smaller than the observed LL50. This has been attributed to the loss of dissolved hydrocarbons during the test procedures (Redman et al., 2017).
PETROTOX is used below to:
- Predict the toxicity of various oils;
- Examine the utility of TPH and TPAH as dose metrics for the mixture of petroleum components in WAFs; and
- Evaluate how the toxicities of different oils vary with oil loading and microdroplet concentrations.
Toxicity of Various Oils
PETROTOX can be used to predict the toxicity of oils for which the detailed oil composition obtained by using the comprehensive GC×GC method is available (Reddy et al., 2012). The required acute and chronic HC5 critical body burden required for the calculation are from McGrath et al. (2018). Figure 3.9 presents the toxicity of 10 oils, ranging from fresh to highly weathered samples. The acute HC5s TUs of the WAFs are plotted against oil loading. At the highest loading (1,000 mg oil/L), the WAFs of the least toxic oils—for example, Macondo Slick A and Slick B—are saturated
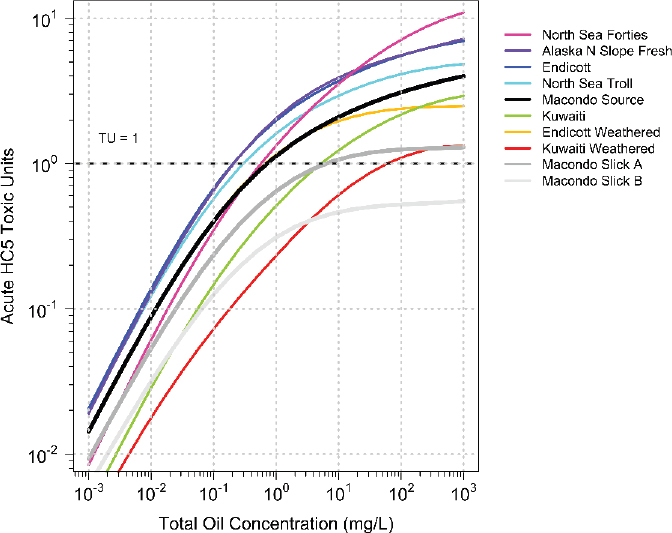
and have reached their highest toxicity, whereas the toxicity of the WAFs of the most toxic oils (e.g., North Sea Forties) would increase at higher loadings, as indicated by the upward curves of the lines. As the oil loading decreases the acute HC5 TU concentrations—and therefore the toxicity—all decrease, but not in proportion to the reduction in loading until the loadings decrease below approximately 1.0 mg oil/L. There are significant differences among the oils between the rate of decline and the toxicity. Slick B oil, the most weathered, never reaches a level of TU = 1. At TU = 1, the loading for the various oils ranges from approximately 100 mg oil/L to nearly 0.1 mg oil/L, a difference of almost three orders of magnitude. This shows that there are significant differences due to the oil composition, and that for the same oil (e.g., Macondo, Endicott), toxicity decreases with weathering. Therefore, if toxicity predictions are required to be representative of a specific fresh or weathered oil, its composition must be known or approximated by an oil of known composition.
Dose Metrics: Toxic Unit, TPH, and TPAH
In order to quantify the toxicity of a mixture, a dose metric is necessary to establish the dose response. As discussed above, the dose metric for mixtures of the hydrocarbons in oil that correctly predicts the observed dose response in many experiments is the TU (see Equation 2). Unfortunately, this is not the metric that is normally used in either toxicity tests or in reporting field observations. The most common dose metric is TPAH, which is the arithmetic sum of the concentrations in weight units (e.g., µg TPAH/L). A more complete sum is TPH that includes all the hydrocarbons that are resolved using the analytical method employed. Equations 3 and 4 define TPAH and TPH, respectively. A comparison to the defining equation for TUs (Equation 2) illustrates the differences in these dose metrics.
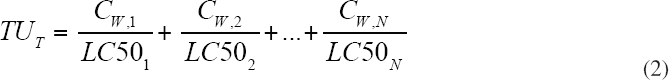
![]()
![]()
The TUT (Equation 2) weights the concentration of each petroleum hydrocarbon CW by its effect concentration LC50, using the dissolved concentrations. TPAH (Equation 3) and (Equation 4) are the sums of the aqueous concentrations without regard to their individual toxicity, and they are usually the sum of the dissolved and microdroplets concentrations. In addition, TPAH only considers the PAHs in the mixture.
It is clear from Equations 2-4 that these three dose metrics are incompatible if they are used to compare oils of differing composition. Consider the example presented in Table 3.2. Two example oils, labeled “Neat” and “Weathered,” are assumed to be composed of equitoxic mixtures of the PAHs listed. The neat and weathered oil composition results in one TU. Therefore, both oils result in 50% mortality and are equally toxic. The TPAH concentrations, however, are different because the two least toxic PAHs with the highest concentrations are not in the weathered oil. Therefore, if only the TPAH concentrations were used as the dose metric, the weathered oil has a lower TPAH concentration at 50% mortality (TPAH LC50 = 14.4 µg/L) than does the neat oil (TPAH LC50 = 49.4 µg/L) and therefore would be judged to be the more toxic oil. Using the TU dose metric, both of these oils would be judged to be equitoxic. This problem has been addressed in an investigation of the relative toxicity of neat and weathered oils. It demonstrates that the use of TPAH leads to the incorrect conclusion that weathered oils are more toxic than neat oil is (Di Toro et al., 2007). What follows is a more detailed evaluation that uses the PETROTOX model to examine the extent that TPAH values correlate to TUs, and it can therefore be used as a surrogate for TUs.
TPAH and TPH can be used if the fractional composition of the dissolved components in the aqueous phase is not changing. For this situation, the TPAH and TPH would be proportional to the TU concentrations. Note that for cases where the dissolved concentrations of the TPAH and TPH are explicitly measured, the computation of the TU concentration is straightforward because the acute LC50 and chronic EC20 are available for many species (Di Toro and McGrath, 2000; Di Toro et al., 2000; McGrath et al., 2004), and the acute and chronic HC5s are also available if the species of interest is not available (McGrath et al., 2018).
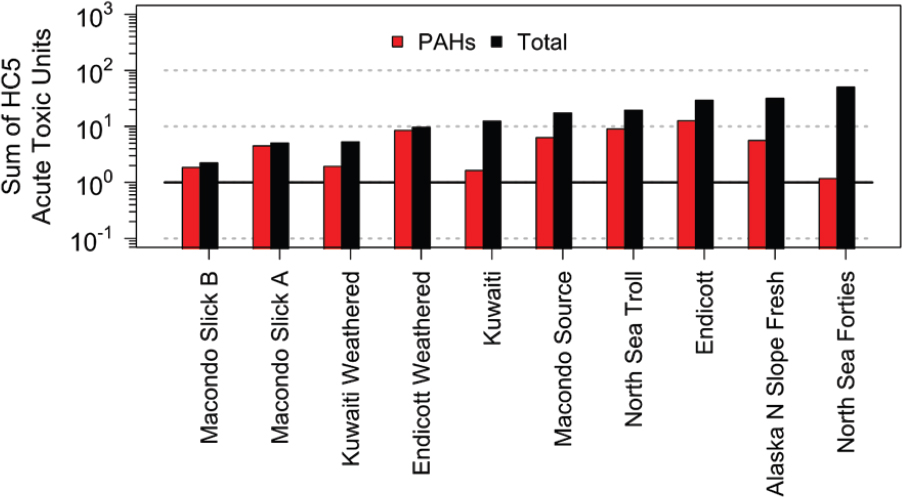
Another problem with TPAH is that it does not capture all the toxic components. The comparison below considers the toxicity of an aqueous phase WAF in equilibrium with a large enough oil loading to ensure that the oil composition is not significantly altered by the dissolution of the oil components into the aqueous phase once equilibrium has been achieved. The resulting saturated aqueous phase has the highest dissolved concentrations that can exist for this oil. Figure 3.10 presents the results of a PETROTOX simulation for the 10 oil samples presented in Figure 3.9, for which the detailed composition is available at an oil loading = 10 g oil/L that saturates the aqueous phase. The figure presents both the sum of the acute HC5 TUs due to dissolved PAHs only and the total TU that considers all the dissolved hydrocarbons.
The results indicate that acute HC5 TU concentrations for saturated WAFs vary from ~5 to ~50 TUs, whereas the TUs due to total PAHs vary from ~1 to ~10 TUs. It is clear that for several of the oils, the PAHs are not the major source of TUs and, therefore, the toxicity of the WAF. Therefore, using TPAH as a dose metric would miss a significant fraction of the toxic components for the
TABLE 3.2 Comparison of Total PAH and Toxic Unit Dose Metrics
| Neat Oil | |||||
| Chemical | log Kow | HC5 (uM) | HC5 (ug/L) | EqiTox (ug/L) | EqiTox (TU) |
| Naphthalene | 3.256 | 1.033 | 132 | 22.000 | 0.167 |
| Acenaphthylene | 3.436 | 0.7 | 107 | 17.833 | 0.167 |
| Acenaphthene | 3.878 | 0.269 | 41.5 | 6.917 | 0.167 |
| Anthracene | 4.546 | 0.0633 | 11.3 | 1.883 | 0.167 |
| Pyrene | 5.126 | 0.018 | 3.64 | 0.607 | 0.167 |
| Chrysene | 5.782 | 0.00434 | 0.99 | 0.165 | 0.167 |
| TPAH | TU | ||||
| Sum | 49.405 | 1.000 | |||
| Weathered Oil | |||||
| Chemical | log Kow | HC5 (uM) | HC5 (ug/L) | EqiTox (ug/L) | EqiTox (TU) |
| Acenaphthene | 3.878 | 0.269 | 41.5 | 10.375 | 0.250 |
| Anthracene | 4.546 | 0.0633 | 11.3 | 2.825 | 0.250 |
| Pyrene | 5.126 | 0.018 | 3.64 | 0.910 | 0.250 |
| Chrysene | 5.782 | 0.00434 | 0.99 | 0.248 | 0.250 |
| TPAH | TU | ||||
| Sum | 14.358 | 1.000 | |||
majority of these oils, in addition to not having the appropriate normalization that is included in derivation of TUs (Equation 1). For these two reasons, the use of TPAH as a dose metric is not scientifically defensible when applied across oils or different weathering states of the same oil.
Effect of Variable Oil Concentrations
The previous section examined the toxicity of oil of a saturated WAF. In this section, the toxicity of Macondo oil is computed as it is diluted from oil loadings that produce a saturated WAF (104 mg oil/L) to a very diluted concentration of 10–3 mg oil/L = 1 µg oil/L. Figure 3.11A shows the resulting TPH and TPAH dissolved concentrations as well as the concentration of undissolved remaining oil.
Even at the lowest total oil concentration (1 µg oil/L), a liquid oil phase (“remaining oil”) persists because not all the oil components are soluble. As the amount of added oil increases, both TPH and TPAH start to approach the saturated concentrations.
Figure 3.11B presents the acute HC5 TUs due to PAHs (TPAH-TU) and the total acute HC5 TU concentrations versus oil concentration. The dashed line is a unity slope line. At low oil concentrations of 1 to 10 µg oil/L, the increase in TU concentration is almost in proportion to the increase in loading concentrations (compare to the unity slope line). However, at higher concentrations, both the TU and the TPAH-TU curves begin to plateau as the samples approach saturation. As saturation is approached, the TU and TPAH-TU curves start to diverge, indicating that the TPAH-TUs represent an increasingly smaller fraction of the total TUs present in the mixture.
In practice, the concentrations of TPH and TPAH are used as dose metrics. Their validity and utility can be judged by comparing TPH and TPAH concentrations to the TU concentrations as a ratio. If the TPH and TPAH are proportional to TUs then these concentrations can serve as dose metrics that are proportional to the TU concentration. Figure 3.12 presents the results.
At low total oil concentrations (i.e., < 10 µg/L = 10–2 mg/L), both TPH/TU and TPAH/TU ratios are reasonably constant and therefore can be used as dose metrics for this oil. However, as the oil concentration increases, the ratios start to increase. For TPH and TPAH, the ratios climb by approximately two and 1.5 orders of magnitude, respectively, as saturation is approached. Therefore, for total oil concentrations of > 100 µg/L = 10–1 mg/L, TPH and TPAH are no longer representative of the TU concentration. They are not a constant multiple of the actual TU concentration. Their use as dose metrics for toxicity tests at different total oil concentrations can lead to incorrect
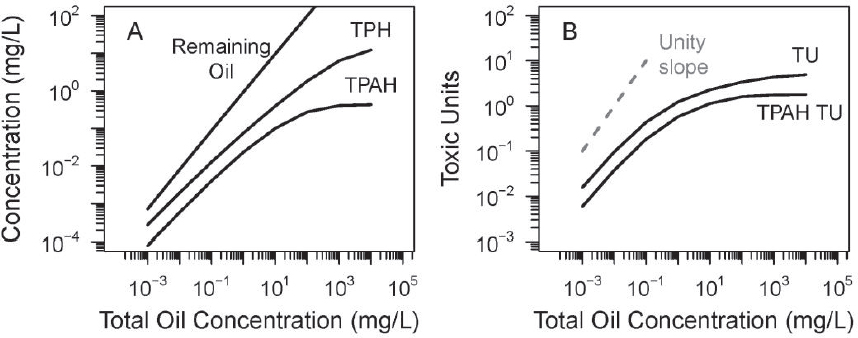
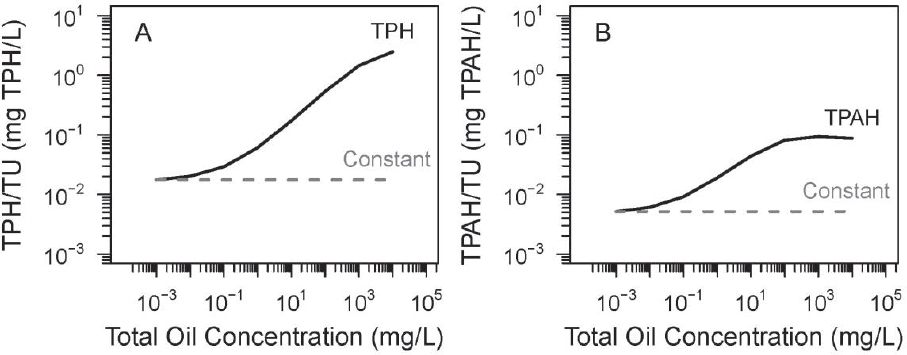
conclusions. Also, because these ratios change with different oils, using TPH and TPAH as a dose metric to compare the toxicity of different oils would likely be misleading. Unfortunately, this is the range at which most toxicity tests are conducted, as seen in the results of PETROTOX simulations of the TU concentrations of the 10 oils examined in Figure 3.9.
Concentration of Microdroplets
The previous sections analyzed the behavior of the dissolved concentrations in an oil-water mixture. This section examines the effects of the presence of microdroplets in the WAF.
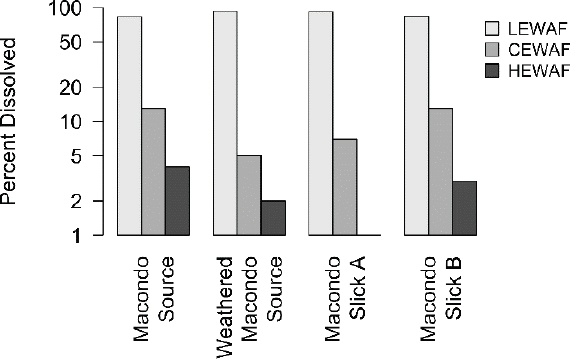
There is a large variation in the concentration of droplets in various WAFs, which is the result of varying mixing energies and the use of dispersants. A recent report (Forth et al., 2017b) examined the LEWAF, CEWAF, and HEWAF prepared from four oils collected from the DWH oil spill. Figure 3.13 presents the percent dissolved in the three WAFs.
In LEWAF preparations, the percent dissolved PAH concentrations comprise the large majority (> 90%) of the total dissolved oil concentrations resulting from the minimization of oil droplet interference by mixing the exposure media without promoting microdroplet formations. In contrast, the percent dissolved for CEWAF and HEWAF are a smaller percentage of the total dissolved concentrations (below ~20% and ~5%, respectively), indicating that the majority of the oil in these dispersed WAFs is not dissolved. This is a critical issue because these concentrations are usually incorrectly assumed to be bioavailable. Therefore, a careful consideration of the effect of the microdroplets on the dissolved concentrations and toxicity is required.
The concentration of microdroplets is difficult to measure directly, but it can be inferred from the measured concentration of essentially insoluble components of the oil (Redman et al., 2012a).
PETROTOX is used to predict the concentration of dissolved oil and the composition of the remaining oil phase that is expected to be present for an oil of known composition and concentration in the WAF. The concentration of microdroplets is estimated to reproduce the measured concentrations. For the cases investigated, the concentration of microdroplets ranged from ~50 to ~500 µg oil/L (Redman et al., 2012a).
Effect of Microdroplets on Dissolved Concentrations in Variable Dilution Media
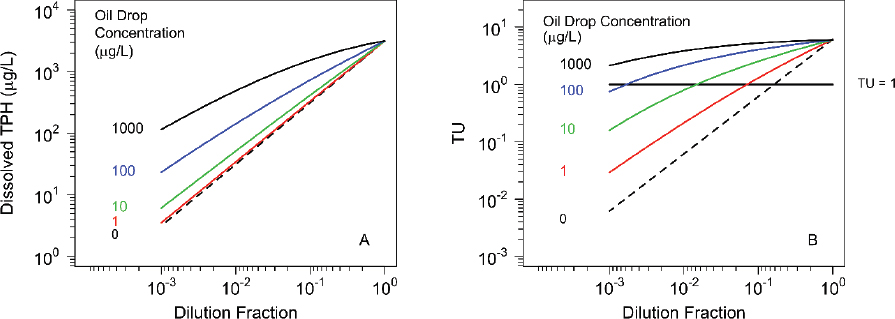
The failure of the variable dilution toxicity test to determine whether the addition of dispersant increased or decreased the toxicity of the exposure media containing oil (as shown in Figure 3.7) is attributed to the influence of microdroplets. The magnitude of the departure from the concentration estimates using dilution is remarkably large (as shown in Figure 3.14), a PETROTOX simulation of a variable dilution experiment.
Without microdroplets (dashed line) the TPH concentration (see Figure 3.14A) decreases linearly with dilution: for instance, a 10-fold decrease in TPH for a 10-fold decrease in total oil, corresponding to a 10-fold dilution. For small microdroplet concentrations = 1.0 and 10.0 µg/L (red and green lines), the decrease is almost linear, although there is a contribution to the TPH concentration from the dissolution of the microdroplets. At higher midrodroplet concentrations = 100 and 1,000 µg/L (blue and black lines), the contribution of microdroplet dissolution is substantial, causing an increase in excess of one order of magnitude. Recall that the range in microdroplet concentrations found in LEWAF toxicity tests ranged from ~50 to ~500 µg/L.
The contribution of microdroplets dissolution to acute HC5 TUs (see Figure 3.14b) is even more extreme. The dilution to achieve TU = 1 varies as the microdroplet concentration (MD) increases. For MD = 0 the dilution D0 = 0.133. For microdroplet concentrations of MD = 1, 10, and 100 µg/L the dilutions at TU =1 are D = 0.071, 0.016, and 0.0018. The problem with using a variable dilution toxicity test to determine whether adding dispersants increases toxicity is that the dissolution of microdoplets increases the toxicity dramatically. There may or may not be additional toxicity due to the dispersant, but the effect of microdroplet dissolution is so large that the additional toxicity may not be detected.
The flaw with the variable dilution test design is that dilution is used as a surrogate for the dose metric, and in the presence of microdroplets, the actual TUs in the aqueous phase will vary systematically for a given oil type, but not among oils, thereby hindering comparisons across multiple studies (e.g., meta-analyses). It is for this reason that variable loading tests are
the recommended test design (e.g., OEDC, 2002) for evaluating the effect of dispersant on the toxicity of oil. The dose metric is unambiguous: that is, the concentration of oil added to the aqueous phase. The quantity of oil actually added needs to be verified by measurement such as a comprehensive TPH measurement that includes all the oil in the sample (see Table 3.1). Tests reporting only nominal oil concentrations are not acceptable.
Effect of Exposure Time
Exposure time, the duration of time that the organism is exposed to the chemical, is important in determining the effect of that exposure. The progressive decrease in the 24-hour, 48-hour, and 96-hour LC50 concentrations commonly found attests to the usual finding that increasing the length of exposure results in higher lethality for a given concentration. The designation of acute versus chronic toxicity testing differentiates between shorter and longer exposure times.
The variations in exposure times and concentrations can be extreme during oil spills. Therefore, an understanding of the effect of exposure time is an important component in determining the effect. The French-McCay OilToxEx model (French-McCay, 2002) includes a consideration of the effect of exposure time (see Equation 5):
![]()
where is the LC50 as t → ∞ and ϵ is the elimination rate constant. ln(ε ) is a decreasing function of log KOW and an increasing function of temperature. A more recent analysis examined the effect of organism life history characteristics and found large species-specific differences for ϵ. For fish, however, ϵ is slightly dependent on log KOW, and for invertebrates ϵ is invariant with respect to organism weight, temperature, and log KOW (Redman, 2018). A comparison of these two models is presented in Figure 3.15 for four PAHs.
Both models behave similarly. There is a dramatic variation in toxicity for exposure times less than or greater than 24 hours. As a point of reference, under noncontinuous oil releases in the open ocean, both physically and chemically dispersed oil are subject to rapid dilution, resulting in concentrations declining rapidly over time, typically within 4 hours (reviewed in Bejarano et al., 2014b). The horizontal lines in Figure 3.15 are the aqueous solubility of the compounds. If the
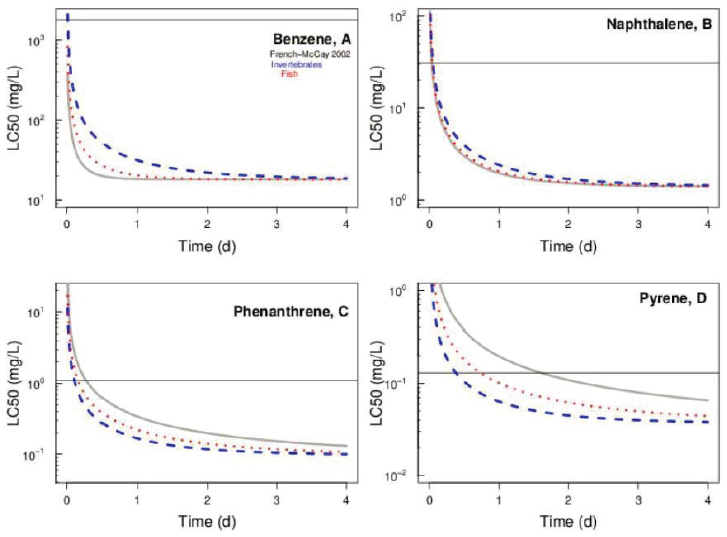
model LC50 exceeds the aqueous solubility, the prediction would be that no mortality is observed for that duration of exposure. It is clear that the effect of exposure time is a necessary component of evaluating the toxicity in field situations where the patchy distribution of the oil results in variable durations of exposure. Toxicity tests that are designed to augment the available data for shorter exposure periods could be used to estimate HC5s for exposure times less than the available 96-hour HC5.
Phototoxicity
The fact that exposure to solar radiation increases the toxicity of certain PAHs in mammalian species has been known for more than 80 years (Findlay, 1928). For aquatic species, comprehensive reviews are available (Arfsten et al., 1996; Barron, 2017; Diamond, 2003; Giesy et al., 2013), and a mechanistic explanation of the phototoxicity pathway is provided in the previous NRC report (NRC, 2005). Briefly, the mechanism is referred to as photosensitization. The PAHs that partition into the organism absorb light energy and are converted to an excited state that decays and causes tissue damage within the organism (Little et al., 2000).
Exposure to ultraviolet also causes PAHs to photodegrade. The extent of photo degradation and the toxicity of the photodegradation products need to be considered as well. The research in this area has focused mainly on photodegradation (Bacosa et al., 2015; Huba et al., 2016; Maki et al., 2001; Shankar et al., 2015; Vergeynst et al., 2019). Determining the toxicity of the degradation products may require a more advanced model than the TLM for the narcosis mode of action as discussed below.
An example of the increase in toxicity of four PAHs to Daphnia magna exposed to simulated solar radiation is shown in Figure 3.16A, which presents the LC50s in the absence and presence of simulated sunlight.
For certain PAHs with a large overlap of the absorbance and solar radiation spectrum (fluoranthane and anthracene), the increase in toxicity can approach 100-fold. For other PAHs, the increase is less: for example, chrysene increases by a factor of 10. For other compounds, such as phenanthrene, there is no observed increase. It is interesting to note that while anthracene and phenanthrene are at the opposite end of the degree of phototoxicity, they are isomers with the same number (3) of aromatic rings as well as the same toxicity in the absence of solar radiation. The different molecular structure, namely, how the rings are fused, is responsible for the difference in the absorbance spectra and the consequent remarkable difference in phototoxicity.
In order to be able to predict the toxicity of a WAF, it is necessary to predict the phototoxicity of each component. A number of models of PAH phototoxicity have been proposed. Table 3.3 presents the models and the mechanisms that are included in the formulations. The models consider, in varying degrees, the absorbance spectra of PAHs, the spectral distribution of the incident radiation, the length of time of exposure, and the sensitivity of the species in question.
A model that addresses each of these processes is the Phototoxic Target Lipid Model (Marzooghi et al., 2017), which is based on the TLM used in PETROTOX. It computes the ratio of the phototoxic LC50 (PLC50) to the TLM LC50 as a function of the spectral absorbance of the PAH and the spectral distribution of the incident light exposure. The model has two constants that apply to all PAHs and organisms. It has been calibrated using the acute mortality LC50s and also the lethal time to death (LT50), the length of time of exposure required to cause 50% mortality at a specific concentration. The calibration dataset consists of 333 observations for 20 individual PAHs and 15 test species that include arthropods, fishes, amphibians, annelids, mollusks, and algae. The exposures are simulated solar and various UV light sources. The exposure times vary from less than 1 hour to 100 hours. The resulting LC50 concentrations range from less than 0.1 µg/L to greater that 10 mg/L. The root mean square errors of prediction for log (LC50) and log (LT50) are 0.473 and 0.382, respectively.
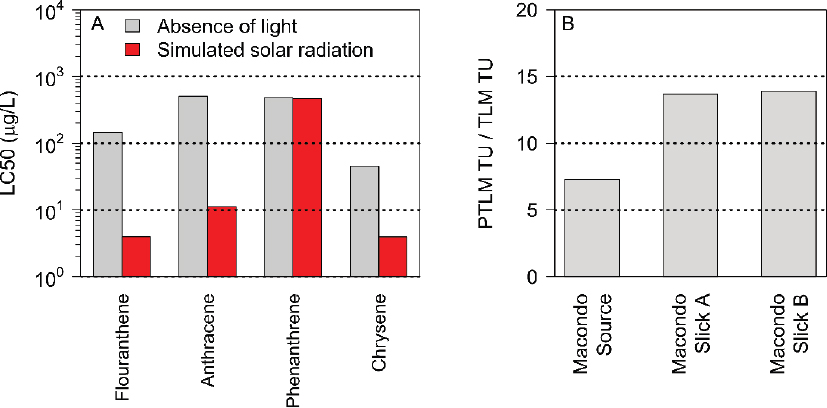
TABLE 3.3 Characteristics of Available Phototoxicity Models
| PAH Phototoxicity Model | PAH Absorbance Spectra Used | Incidence Light Spectra | Duration of Light Exposure Used | Quantity Predicted | BCF Used | Method to Deal with Species Sensitivitya | Method to Deal with Chemical Potencyb | Number of PAHs Considered | Number of Species Considered |
|---|---|---|---|---|---|---|---|---|---|
| Morgan and Warshawsky, 1977 | √ | UV 366 nm | √ | ANIc Rate | √ | RPAg | 28 | 1 | |
| Newsted and Giesy, 1987 | √ | UV | √ | QSARc | √ | RPAg | 20 | 1 | |
| Mekenyan et al., 1994 | UV | QSARd | HOMO-LUMO gap | 20 | 1 | ||||
| Oris and Giesy, 1985 | UV | √ | LT50 | 1 | 1 | ||||
| Ankley et al., 1995 | UVA | √ | LT50 | √ | 3i | 1 | |||
| Krylov et al., 1997 | √ | UV+VIS | QSARe | 16 | 1 | ||||
| Grote et al., 2005a | √ | UV+VIS | √ | LC50 | √ | RPEh | 11 | 1 | |
| Sellin Jeffries et al., 2013 | UVA | √ | LT50 | RPAf | RPAf | 18 | 4 | ||
| Marzooghi et al., 2016 | √ | UV+VIS | √ | LC50 and LT50 | √ | TLM Critical Lipid Conc. | TLM Lipid-H2O Partition Coef. | 20 | 15 |
NOTE: ANI = average number of Artemia salina nauplii immobilized; HOMO-LUMO gap = difference in energy between the highest occupied molecular orbital and the lowest unoccupied molecular orbital; LC50 = lethal concentration at 50% mortality; LT50 = lethal time at 50% mortality; RPA = relative phototoxic activity; UV = ultraviolet; VIS = visible.
aFactor accounting for the difference between organism sensitivities.
bChemical toxicity potency accounting for differing chemicals properties.
cQuantitative structure–activity relationship (QSAR) model developed to predict the LT50 using the empirical physiochemical parameters.
dQSAR to predict LT50 based on HOMO-LUMO gap.
eQSAR to calculate photosensitization constant and a photomodification constant.
fRelative phototoxic activity (RPA) = (LT50PAHi)1/(LT50Anthracene)1.
gPotency (rate of mortality per unit time per unit of absorbed irradiance) relative to that of the reference compound (benzo[b]anthracene).
hRelative phototoxic efficacy calculated as coefficients for each PAH.
iRegression analyses for each PAH separately. Chemical potency correction not performed.
SOURCE: Adopted from Marzooghi and Di Toro, 2017.
The Phototoxic Target Lipid Model has been validated by predicting the PLC50s for four species exposed to artificial sunlight for 12 compounds, including alkylated PAHs and dibenzothiophene in single compound tests, and for binary and ternary mixtures of pyrene, anthracene, and fluoranthene using TU addition as the mixture model (Marzooghi et al., 2018). It uses the same framework as the TLM to compute the TU concentration. Using this framework, the Phototoxic Target Lipid Model was used to predict the toxicity of WAFs of neat and naturally weathered Macondo crude oil samples (Source, Slick A, and Slick B) to three Gulf of Mexico species. The root mean square errors of prediction were comparable to the calibration dataset.
The predicted increase in phototoxicity for each of the three Macondo oils, a factor of approximately 6 to approximately 14, is presented in Figure 3.16B as the ratio of phototoxic PAH TU to narcotic PAH TU at saturation. The weathered oils exhibit the largest increase, which is due to the larger percentages of phototoxic PAHs in the weathered oils. This would increase their toxicity so that more weathered Slick A would be approximately as toxic as the fresher Macondo source oil (see Figure 3.9). This result also indicates that a comprehensive model is required for the prediction of the toxicity in the presence of solar radiation.
The use of dispersants can affect the concentration and composition of the oil that rises to the surface where solar radiation can increase toxicity. A number of factors need to be considered. For an application of dispersant to a surface spill, the dispersant would increase the dissolved concentrations. The dispersed oil droplets would be mixed into the top few meters, and PAHs would dissolve from the droplets to increase the aqueous concentrations. Oil droplets may also reduce UV attenuation in the water column, potentially reducing phototoxicity. Thus, while the volume of water potential impacted by photoxicity may be increased in the presence of surface dispersant application, the resulting risks are unclear.
For the subsea dispersant injection, the reduction in droplet size distribution may accelerate the rate of dissolution during droplet rise to the surface and therefore may have affected the concentrations of phototoxic PAHs at the surface. However, without an evaluation that can produce the concentrations of the phototoxic compounds that would result with or without dispersant use, the magnitude of the resulting changes in toxicity cannot be determined. For both these scenarios the resulting risk could be explored using coupled fate and effect models.
It should be noted that the tools to do such an evaluation are available. Models that predict the dissolved concentrations of various hydrocarbon blocks have been employed (see Chapter 2). The required toxicity models—PETROTOX and the phototoxic TLM—are also available. However, the effect of photo degradation of PAHs has not yet been included in the fate models. The PAHs that efficiently absorb incident light are also the PAHs that are susceptible to photo degradation. This is an important mechanism that also needs to be included in the fate models. The toxicity of the degradation products also needs to be included in the toxicity models.
A number of recent publications report phototoxic effects, including the effect of dispersants on photo toxicity (Alloy et al., 2017; Barron, 2017; Barron et al., 2018; Bridges et al., 2018; Finch and Stubblefield, 2016; Finch et al., 2016, 2017a,b, 2018; Nordborg et al., 2018; Overmans et al., 2018; Salvo et al., 2016). These tests cannot be evaluated to determine the effect of dispersants on phototoxicity of oil unless the concentrations of the dissolved components were measured at all the dilutions or unless the experiments were variable loading tests with measured total oil added (see Table 3.1). It is the same problem that limits the utility of the variable dilution toxicity tests. Additionally, only selective PAHs are phototoxic and their dissolved concentrations are required in order to confirm the cause of the observed toxicity.
Sediment Toxicity
Evaluating the toxicity of chemicals in sediments is complicated by the large variations in bioavailability to sediment-dwelling organisms due to variations in sediment composition. While not addressing oil contamination per se, in a classic paper by Adams et al. (1985) the observed LC50 (mortality) and EC50 (growth reduction) concentrations of the insecticide Kepone to the sediment-dwelling organism Chironomus tentans was < 1 µg/g for a sediment with low organic carbon concentration, fOC = 0.09% by weight. By contrast, for a sediment with fOC = 1.5% organic carbon, the EC50 and LC50 are approximately 7 and 10 µg/g, respectively. The high organic carbon sediment (fOC = 12%) exhibits still higher LC50 and EC50 values on a total sediment Kepone concentration basis, 35 and 37 µg/g, respectively (Adams et al., 1985). As a consequence, the dose metric employed for sediment toxicity tests must account for this.
The Kepone experiment provided two critical findings. The first was the observation that for these toxicity tests the pore water LC50 or EC50 varied only by approximately a factor of two for the three sediments, whereas the sediment LC50 or EC50 had an almost 40-fold range in Kepone concentrations. This pointed out the importance of the pore water concentration as a dose metric.
The second insight was the relationship of the pore water LC/EC50s and the LC/EC50s obtained from water-only exposures to the same organism. The pore water LC50s are 19 to 30 µg/L, and the water-only exposure LC50 is 26 µg/L. The pore water EC50s are 17 to 49 µg/L, and the water-only EC50 is 16 µg/L (Adams et al., 1985). This result suggested that if the concentration in the pore water equaled the LC50 or EC50 concentration for that organism in a conventional water-only exposure, the sediment would exhibit 50% mortality or 50% growth reduction. This observation suggested that the LC50s and EC50s obtained from conventional water-only exposures could be utilized to predict sediment toxicity.
The importance of pore water concentrations as a dose metric has prompted the development of methods to sample sediment pore waters directly (e.g., Arp et al., 2015). Until recently, however, methods for efficient and trustworthy sampling of sediment pore water were not available. The usual sediment sampling methods determined the chemical concentration in the total sediment. This included the chemical in the pore water and the chemical associated with the sediment particles and, if present, the chemical in condensed phases: for example, as a liquid or a solid. Two classes of models have been developed to evaluate the relationship between chemical concentrations in sediment and benthic organism toxicity (Wenning, 2005).
The first, referred to as empirical models, are based on large datasets of paired total chemical concentrations in the sediment and measured sediment toxicity. Various statistical methods have been employed to establish threshold and median concentrations for various chemicals (Long, 2006; Long et al., 1995, 1998).
The second, referred to as mechanistic models, deal with the bioavailability problem by utilizing models that predict the pore water concentration from the bulk sediment concentration and the relevant sediment properties (Di Toro et al., 1991). A comprehensive review and a comparison of the empirical and mechanistic models is available (Wenning, 2005).
Equilibrium Partitioning Model of Sediment Toxicity
The equilibrium partitioning (EqP) model was developed to establish sediment quality criteria (Di Toro et al., 1991). The EqP model assumes that the dose delivered from the pore water and the sediment solids exposure are equal if the pore water and sediment solids concentrations are in thermodynamic equilibrium. In this case, the chemical potential is equal in both phases and the compound in each phase exerts the same chemical binding strength toward the organism. Sediment criteria derived using the EqP model used the water-only aquatic life criteria concentration as the
pore water concentration and predicted the bulk sediment concentration that would result for a sediment with a specific composition.
The TLM model was developed for the U.S. Environmental Protection Agency (EPA) sediment criteria development effort for PAH mixtures to predict the pore water concentrations for use in the EqP model (Burgess et al., 2012; Di Toro and McGrath, 2000; EPA, 2003). The model has been validated for individual PAHs in laboratory spiked sediments and also in PAH contaminated field collected sediments (Redman et al., 2014). It has been applied by EPA in the initial evaluation of sediment toxicity for the DWH oil spill (EPA, 2010, 2016a,b).
The application of the EqP-derived sediment toxicity concentrations requires a chronic exposure concentration. The reason is that sediment-dwelling organisms are exposed for either their partial or full life cycle. Therefore, the chronic criteria are appropriate. In the original EPA guidance documents (EPA, 2003), the chronic water quality criterion was employed. For application to this investigation, the chronic HC5 concentration can be employed. Figure 3.17 presents the chronic HC5 oil concentrations. Comparing Figure 3.17 (chronic HC5) to Figure 3.9 (acute HC5) illustrates the difference in concentrations for protection of acute exposures and mortality endpoints as well as chronic exposures and sublethal endpoints. The EPA sediment toxicity guidelines employ the latter.
Macondo Slick A and Slick B concentrations at TU = 1 can be used to provide the basis for a comparison to observed concentrations in the field, as shown next.
Comparison to Field Collected Data
Sediment Data
An example of TPAH concentrations in sediments is shown in Figure 3.18, for series of sediment cores collected during the DWH sampling cruises (Stout and Payne, 2016a).
The concentration units are mg TPAH/kg sediment, the concentration of TPAH per kg dry weight of sediment. An approximate comparison can be made using the TPAH concentration corresponding to chronic HC5 TUs = 1. The approximation employed is detailed in Di Toro and
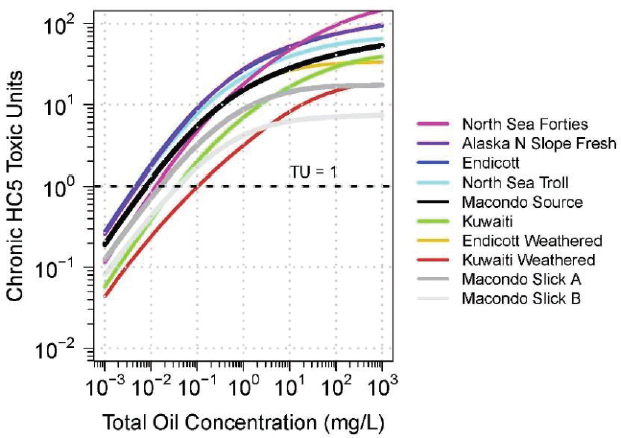
McGrath (2000). The required sediment concentration is the organic carbon normalized concentration (mg TPAH/kg sediment organic carbon.) In order to make the comparison, the fraction organic carbon in the sediment is required. The shaded box in Figure 3.18 enclose the TPAH sediment concentrations (mg TPAH/kg sediment) that are within the fraction organic carbon spanning the 5th and 95th concentrations (0.00208 and 0.0428 gOC/g sediment) found in sediments from productive regions areas (Seiter et al., 2004, Figure 2C). As a comparison, the Long and Morgan effects range low (ERL) empirical sediment criteria for the sum of TPAHs (Long et al., 1995) are quite close to the TPAH chronic HC5 concentration. For fOC = 0.01 gOC/g sediment, the ERL = 4.64 and the chronic HC5 = 5.32 mg TPAH /kg sediment. Most of the sediment concentrations within 1.6 km of the discharge and the surface samples at all distances sampled are in the uncertain range. It is not possible to make the comparison to the chronic HC5 for these data. This comparison highlights the importance of expressing sediment concentration as organic carbon normalized concentrations (EPA, 2003). For a more complete analysis, the following also need to be considered: the TPH that are present in addition to the TPAH; other sediment phases that sorb PAHs such as black carbon (Ghosh, 2007; Lohmann et al., 2005), and the extent that the sorption is reversible (e.g., Fu, 1994).
Water Column Data
A selection of water column field data is presented in Figure 3.19 (Boehm et al., 2016). The concentration units are mg TPAH/L. A conversion of these concentrations to TUs is necessary for the same reason that it is required for the sediment data. The conversion requires the individual PAH concentrations and an estimate of the concentration of microdroplets in order to obtain the dissolved PAH concentrations. The necessary acute and chronic HC5 concentrations are available for the calculation of the TU concentration (see Figures 3.9 and 3.17, respectively). The choice for using the acute HC5 for the water column and the chronic HC5 for sediments is made because there are no duration-specific criteria and this choice is conventional. As pointed out above, the effect of exposure time is substantial.
The acute and chronic HC5 concentrations for MASS oil are approximately 1,000 and 10 mg oil/L. These correspond to approximately dissolved TPAH concentrations of 10 and 0.5 mg TPAH/L
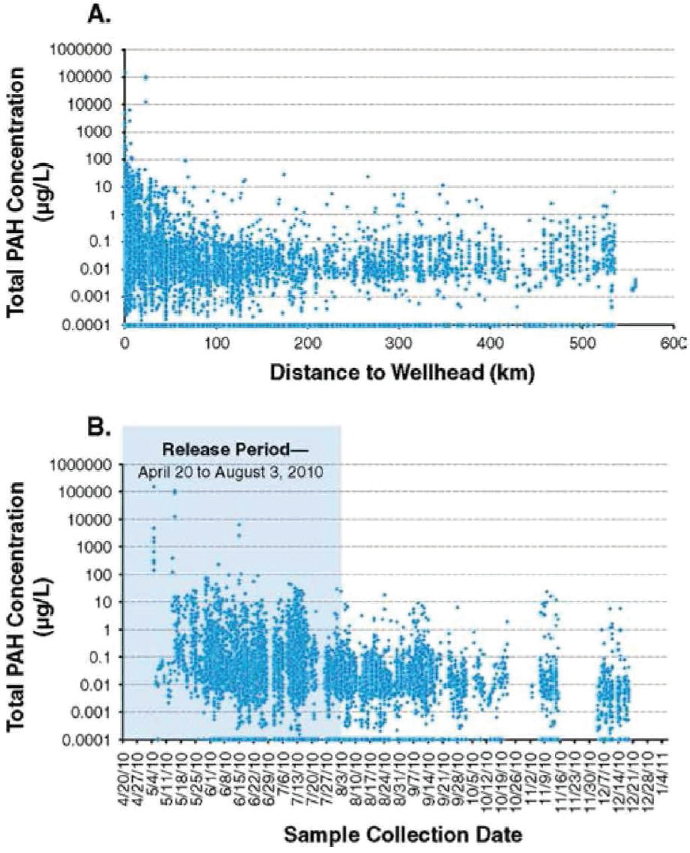
(see Figure 3.11A). During the release period (see Figure 3.19B), most of the observed TPAH concentrations are below the acute HC5 (10 mg TPAH/L), and they bracket the dissolved chronic HC5 (0.5 mg TPAH/L). Because the TPAH concentrations in the field data include the microdroplet contribution, which could be significant (see Figure 3.11A), it would need to be subtracted from the observed concentrations in the field in order to judge the extent of toxicity. This illustrates the need to make the appropriate measurements of dissolved oil components so that a direct estimate of TU concentration can be made.
The above analyses are presented in order to highlight the problems that are encountered if a direct comparison of field TPAH concentrations and acute and chronic HC5 TPAH are compared. The field data from the DWH could be used to calibrate and validate exposure predictions using fate models that incorporate both surface and subsurface dispersant at the rates and timing applied during the response. Such exercises are necessary in order to understand quantitatively the effect that the use of dispersants could have on water column and sediment toxicity. In the case of the DHW, the use of dispersants potentially increased the mass of oil reaching the sediments through the formation of oiled marine snow (see Chapter 2). Therefore, a reevaluation and reinterpretation of the field data from the DWH are required in order to understand the magnitude and extent of toxicity.
Evaluating the effect that the use of dispersants had on the resulting water column and sediment toxicity can only be made using models that compute the changes in fate and the resulting toxicity with and without dispersant addition. For example, the presence of dispersants potentially increased the mass of oil reaching the sediments through the formation of oiled marine snow (see Chapter 2), which increases the rate of deposition to sediments with the combination of oil, dispersants, and clay minerals forming oil-mineral aggregates. In the case of the DWH and Ixtoc I blowouts, the addition of large quantities of dispersants apparently increased the intensity of oiled marine snow, resulting in elevated toxicity to benthic organisms (Daly et al., 2016; Vonk et al., 2015).
A PATH FORWARD FOR AQUATIC TOXICITY TESTING
Test Standardization
As discussed in the sections above, the specific parameters used to prepare WAF or CEWAF (i.e., mixing energy, preparation method) have an effect on the composition of hydrocarbons in the exposure media, thus influencing their toxicity. Consequently, the comparability and reproducibility of toxicity data, as well as their practical application to spill situations, require the consistent use of standardized test procedures. While this is not a novel concept (e.g., CROSERF; Aurand and Coelho, 2005; NRC, 2005), the lack of adherence to existing toxicity testing methods continues to be an issue. As a result, recent efforts have further advocated for standardization of test procedures (Bejarano et al., 2014b; Coelho et al., 2013; Hodson et al., 2019; Redman and Parkerton, 2015). The test procedures originally proposed by CROSERF sought to provide a consistent framework for generating and interpreting toxicity test results. Although the initial framework proved to be comprehensive, modifications were proposed more than a decade ago in the previous NRC report (2005), and NRDA researchers have since suggested more modifications (Forth et al., 2017a; Krasnec et al., 2016).
This current evaluation of the toxicological effects has again emphasized the need to update the CROSERF protocols based on an increased understanding of exposure and uptake and to align with current state-of-the-art technology for solution preparation and analysis. Examples of proposed updates include:
- Conduct (and report) detailed chemical characterization of the source oil that is used in the toxicity test.
- Examine the relevance of mixing energy of WAFs to generate useful data that can be used to validate toxicity models, and also identify a single mixing energy that can be used to prepare WAFs and CEWAFs (to allow for dataset comparison).
- Eliminate the use of HEWAF preparations that generate excessive microdroplets unless there is conclusive evidence to justify that these high concentrations occur in the real world.
- Standardize toxicity test dispersant-to-oil ratios for CEWAFs to align with operational use (e.g., 1:20 for surface dispersant application and 1:100 for SSDI).
- Expand research to include all dispersants likely to be employed in the global dispersant stockpile (not just Corexit®).
- Reserve declining concentration exposure regimes such as spiked exposures for specific situations when appropriate because the chemical exposure from most spills is constantly changing and is exceedingly difficult to quantify. Instead, consider a few constant exposure periods that could equate to short exposures resulting from using dispersants on a batch spill (few hours) to longer exposures that could equate to arctic, cold weather, or prolonged subsea exposure.
- Expand chemical characterization to include as many individual constituents as possible (> 50+ PAHs), including all the hydrocarbons, based on latest analytical capabilities.
- Analyze unfiltered and filtered water samples or passive sampling of the dissolved concentration of each test solution to account for relative contribution of microdroplets.
- If the variable loading method is adopted, develop and standardize analytical protocols that focus on dissolved oil exposures (e.g., filtering or passive sampling).
One potential mechanism to make these modifications to CROSERF is to create a working group of toxicologists, modelers, and resource trustees to ensure that these modifications will support future needs for science to support the operational response community. Future modifications should emphasize the need to generate toxicity data that will inform, validate, or improve toxicity models. Ultimately, having better toxicity models will help response decision makers and response personnel make informed decisions about dispersant use based on the best available information.
Passive Dosing and Passive Samplers
More sophisticated systems have emerged in recent years to generate toxicity data using continuous exposures in a flow-through system (Nordtug et al., 2011). This system enables the quantification of the relative contribution of both dissolved oil fractions and oil droplets to the overall short- or long-term toxicity to aquatic species. For example, this flow-through test system was used to assess the toxicity of physically and chemically dispersed oil to cod larvae (Hansen et al., 2019). This study provided additional evidence on the limitations of total PAH as an exposure metric in oil toxicity, further demonstrating the limited role that oil droplets play in driving toxicity. These systems are also promising as they may provide the necessary link between laboratory and field measurements and observations.
An alternate approach for preparing exposure media and understanding the toxicity of WAF and CEWAF is the use of passive dosing techniques. In recent years, passive dosing approaches have been developed to generate and maintain stable aqueous concentrations of hydrophobic chemicals, including hydrocarbons. Passive dosing commonly employs silicone polydimethylsiloxane (PDMS) tubes. PDMS tubes are loaded with a test solution and directly immersed in water allowing the continuous partitioning of freely dissolved material through the permeable membrane into
the aqueous exposure media. This approach alleviates the interference introduced in toxicity data by oil microdroplets while controlling for compound losses due to volatilization. When placed in test systems, PDMS tubes loaded with known oil concentrations (i.e., WAF or CEWAF) serve as a passive dosing source of dissolved oil in toxicity tests. Such experiments have been successfully carried out with single hydrocarbon compounds, hydrocarbon mixtures, and crude oils under acute and chronic exposures (Butler et al., 2013, 2016; Letinski et al., 2014; Redman et al., 2018; Renegar et al., 2017a). Furthermore, one study demonstrated that passive dosing produces comparable exposures of dissolved oil as WAF generated with the CROSEF method (Bera et al., 2018).
Similarly, the use of solid-phase microextraction (SPME) polymer fibers as passive samplers have also been proposed as cost-effective tools in the quantification of freely dissolved chemicals (Leslie et al., 2002; Mayer et al., 2014; Verbruggen et al., 2000). Because SPMEs serve as a surrogate hydrophobic phase of lipids, dissolved constituents in an aqueous exposure media would partition to SPMEs simulating bioconcentration and providing a quantifiable dose metric. Analytical chemical characterization of SPMEs could then be interpreted to represent bioavailable dissolved concentrations. SPMEs coated with PDMS have been tested and used with hydrocarbon mixtures and crude oils (Letinski et al., 2014; Parkerton et al., 2000; Redman et al., 2018), but their use in toxicity testing carries nuances (i.e., equilibrium with the exposure media and negligible depletion of dissolved-phase concentrations) that need to be carefully considered (Redman and Parkerton, 2015). While the use of passive dosing and sampling approaches is promising, further standardization of test procedures is needed to ensure their inter-laboratory comparability and reproducibility.
While understanding the consequences of oil spills (with or without dispersant use) on exposed populations is of high importance, these cannot be made solely based on results from laboratory studies. Such assessments involve the integration of complex biological and ecological knowledge, including effect responses (e.g., laboratory and field exposures) at different levels of biological organization, life-history parameters and population structure, environmental drivers of population dynamics, etc. Related efforts have been undertaken (e.g., Fodrie et al., 2014; Gallaway et al., 2017), suggesting that effect responses (sublethal or lethal) on individuals may not necessarily translate into population-level impacts. These and related studies point to the need for better integrating aquatic toxicology studies with knowledge on other factors that determine population viability.
BIOLOGICAL EFFECTS
The exposure of aquatic species to the toxic fractions in oil under field conditions depends on the rate at which petroleum hydrocarbons partition and dilute into the water column, with a greater petroleum hydrocarbon exposure potentially resulting from the use of chemical dispersants, although the combination of dilution, dispersion, and biodegradation serves to reduce aqueous concentrations significantly and rapidly. Slow moving or immobile aquatic species and life stages that are entrained within water masses containing physically and/or chemically dispersed oil may be at greater risk of exposure to dissolved oil fractions. Even when entrained within a water mass, physical processes and water column mixing dilute dissolved oil fractions, making exposures variable over time. As a result, one of the greatest limitations in understanding the potential impacts related to dispersant use in open waters is the lack of data derived from exposure conditions that capture the environmental realism of most oil spills (Aurand and Coelho, 2005; Bejarano et al., 2014b; Clark et al., 2001). Even greater uncertainties and data limitations exist when understanding potential impacts from subsea dispersant injection. This is due in part to the difficulty of working with deepwater species as they require special conditions, including high pressure, low temperatures, and darkness.
Despite these limitations, toxicity data from controlled laboratory studies provide conservative estimates of potential impacts. Exposure of aquatic species—particularly early life stages—to physically and chemically dispersed oil can lead to lethal and ecologically important sublethal
impacts, but the onset of these impacts depends on several factors, among them concentrations of dissolved hydrocarbon fractions, exposure duration, and species/life stage sensitivity to oil. A growing body of literature (e.g., Carls et al., 1999; Esbaugh et al., 2016; Heintz et al., 1999; Incardona et al., 2004, 2011, 2013; Mager et al., 2014) has shown that under controlled laboratory conditions, fish embryos exposed for several hours post hatch to low PAH concentration (in the low µg/L) may develop gross abnormalities, with permanent impacts potentially causing reduced survival later in life (Incardona et al., 2013).
In their recent review The Toxicity to Fish Embryos of PAH in Crude and Refined Oils, Hodson et al. (2017) present a series of conclusions that can be analyzed and compared using the TLM.
- “3-5-ringed nonsubstituted, alkylated, hydroxylated, and heterocyclic PAH[s] cause effects on fish embryos that closely resemble those of crude and refined oils;
- Alkyl PAH[s] are the predominant congeners in crude and refined oils. Their toxicities must be considered in assessments of the ecological risks and impacts of oil spills;
- The embryo toxicity of PAH[s] increases predictably with an increasing [of] the number of rings, alkyl carbons, and Kow, indicating that water–lipid partitioning controls exposure and tissue dose; . . . .”
The TLM toxicity prediction is the sum of the TUs of all the components of oils, in particular alkylated, hydroxylated, and heterocyclic PAHs. Predicted toxicity increases as KOW increases.
- “There are significant differences in embryotoxicity among PAH, which can be associated with the pattern of alkyl substitution, but not with Kow; . . . .”
This level of molecular detail is not part of the original TLM. It could be investigated using the more recent TLM model that uses additional molecular properties (e.g., molecular polarizability, dipole-dipole and dipole-induced dipole interaction, and hydrogen bonding) to predict the toxicity from molecular structure (Boone and Di Toro, 2019; Kipka and Di Toro, 2009).
- “Chronic EC50s for individual PAH[s] range from 0.3 to 100 [µg/L].”
A recent reevaluation of the TLM that expands the chronic toxicity database results in chronic HC5s ranges that are quite close: from approximately 0.085 µg/L to 200 µg/L for PAHs with logKOW = 6.5 to 2.5 (McGrath et al., 2018).
- “Because crude oil includes some PAH[s] that induce CYP1A enzymes, all PAH[s] in oil will be subject to higher rates of oxygenation, even if they are not inducers;
- The potentiation and antagonism of CYP1A metabolism and toxicity of PAH[s] in mixtures suggest that measured toxicities of single PAH[s] are conservative and may not be a sound basis for predicting the impacts of mixed PAH[s] from an oil spill; . . . .”
This is a molecular level interaction that can be addressed with the modern models that use molecular properties to assess toxicity as discussed above.
- “Because mixture interactions are not well studied, TPAH concentrations 0.1 µg/L following oil spills should be considered hazardous.”
The use of TPAH concentrations as the dose metric is not consistent with the large variation in toxicity of the individual PAHs. The only currently available experimentally validated mixture model is TUs. If a more reliable mixture model is developed and validated, then it can be used.
When reviewing these laboratory studies, the reader is cautioned to carefully consider how the exposure media were prepared (especially with respect to presence of microdroplets) as well as how the chemistry was conducted and reported. Also, the environmental relevance of these species and life stages, in the context of the specific marine ecosystem, merits consideration. One of the reasons that dispersant application is generally not considered close to shore is to prevent the introduction of dispersed oil into shallow and/or nearshore environments (e.g., in the vicinity of many fish nursery habitats). Dispersant use in marine offshore environments involves trade-off decisions (see Chapter 6), with the recognition that early life stages (generally assumed to be more sensitive to oil) and entrained species may be at increased risk of exposure.
As a point of reference, 75% of water samples collected during the DWH oil spill had TPAH concentrations (sum of 50 parent and alkylated PAHs) of < 1 µg/L, though water samples in the vicinity of the wellhead had concentrations > 1,000 µg/L (Boehm et al., 2016; see Figure 3.19). Concentrations in excess of 1 µg TPAH/L were generally within 20 km of the wellhead at depths of 1,000-1,200 m and in the top 3 m of the water column under surface oil (Boehm et al., 2016). Field assessments of dispersant effectiveness found average TPAH concentrations in the top 1 m of the water column of 10.5 µg/L following “very effective” surface dispersant applications, which rapidly and substantially diluted in less than an hour (Bejarano et al., 2013). This type of rapid dilution is frequently overlooked in laboratory studies.
Many studies have been conducted that examine the sensitivity of cold water species to dispersed oil. Most of these studies have used crude oil or individual polycyclic aromatic compounds and have exposed copepods and fish larvae (e.g., Baussant et al., 2009; Christiansen et al., 1996; Grenvald et al., 2013; Hansen et al., 2011; Hjorth and Nielsen, 2011; Ingebritsen et al., 2000; Jensen and Carroll, 2010; Jensen et al., 2008; Perkins et al., 2005; Skadsheim et al., 2009). Studies looking at both physically and chemically dispersed oil have demonstrated that the toxicities are essentially the same provided that field relevant concentrations are used (e.g., Gardiner et al., 2013; Hansen et al., 2012; McFarlin et al., 2011). These studies further showed that while dispersants temporarily increase the bioavailability of oil, the acute toxicity from the dispersants resulted only at much higher concentrations than would be expected in the water column following an appropriate application of dispersant.
It can be challenging to obtain regionally specific toxicity data because of practical limitations associated with testing Arctic species in standard laboratory test. Therefore, several studies have considered the possible applicability of temperate species toxicity data to Arctic species (Bejarano et al., 2017; de Hoop et al., 2011; Gardiner et al., 2013; Olsen et al., 2016). Studies suggest that cold water species have similar sensitivity as do temperate species to petroleum-related compounds based on acute effects. The Norwegian Research Council reached a similar conclusion based on reviews of research conducted over a 10-year period on the long-term impacts of the oil and gas sector on the environment (Norwegian Research Council, 2012). According to multiple studies, however, cold water species may take longer to exhibit effects from hydrocarbon exposures (Chapman and Riddle, 2005; Gardiner et al., 2013; Hansen et al., 2013; Olsen et al., 2011). Increased response time of cold water species may be attributed to morphological and physiological adaptations for cold water survival that may impact toxic responses, such as increased lipid stores and decreased metabolism (de Hoop et al., 2011). Although similar comparative studies are still needed for deepwater species, limited empirical data suggest that their sensitivity is also comparable and within the range of sensitivity of temperate and shallow-water species (e.g., Frometa et al., 2017; Knap et al., 2017; McConville et al., 2018).
In addition to liquid oil released at depth, gas also escapes. The high pressure at depth increases the solubility and therefore the exposure of organisms to low molecular weight dissolved hydrocarbons (e.g., methane, ethane, etc.). However, the increased pressure also decreases the toxicity of these compounds. The required toxicity pressure corrections have been developed for use in the TLM (Paquin et al., 2018).
Apart from the direct impacts of oil and dispersed oil on mortality of various species and life stages, there are a wide variety of sublethal effects (e.g., physiological, immune, structural, and behavioral responses) that can lead to negative outcomes. These include alterations of vital rates (e.g., growth, fecundity), compromised immune functions, and other stressors, leading, in some cases, to susceptibility to other pathologies. Oil spill effects on growth rates of larvae (e.g., Hernandez et al., 2016) and adult fishes (e.g., Herdter et al., 2017) have been well documented in the literature. What is unclear is the role that dispersed oil plays in these changes.
Despite the relatively low oil concentrations observed in field-based assessments of DWH, particularly at some distance from the wellhead, recent research has also emphasized that even very low concentrations (single digits to < 1 µg/L) of oil exposure can have severe sublethal impacts resulting in impairment of cardiac function (Brette et al., 2014; Incardona et al., 2014); larval developmental anomalies; reduced physiological performance; compromised sensory systems and behavior; impacts on microbiomes; altered immune function; DNA damage and oxidative stress; compromised reproduction; and other serious effects (see Grosell et al., in press, for a review of recent literature). These symptoms can be significant for the individual, but if a sufficient fraction of the extant population is exposed, it can result in lower overall fitness, in reductions in recruitment (survival of young), and in reduced population viability. This may be particularly important with respect to long-lived animals, those already reduced because of other factors, or populations with limited geographic ranges.
Few studies have evaluated such factors based on laboratory control of exposures, although some studies exist. These vital rates are important to populations because the overall fitness of an animal population is determined by the number of viable offspring it produced. In the case of fishes, fecundity is both a logarithmic function of size (length) and a linear function of body weight. Thus, if growth rates of adults are suppressed, this will lead directly to lower overall egg production. Furthermore, if those larvae produced have poor condition and survival, this too will result in lower production and thus lower fitness of the population. Studies showing sublethal impacts, including cardiac dysfunction (Incardona et al., 2014), behavioral impairment (Stieglitz et al., 2016), and other negative physiological effects (Whitehead et al., 2011), can lead to mortality (especially for larvae). They may also lead to impaired population fitness. It is thus important to consider sublethal impacts, including those directly related to population vital rates, when considering the effects of toxic exposures from the population life cycle perspective.
For more than 20 years, there has been concern that if oil spills (and other toxic substances) result in genetic mutations, these could be passed along through subsequent generations, resulting in decreased fitness of populations in perpetuity (Cronin and Bickham, 1998). Surprisingly, little research on the heritability of mutations due to oil exposure has been conducted following the study of White et al. (1999). Several studies have, however, identified genomic and transcriptomic changes in various organisms (e.g., Whitehead et al., 2011; Xu et al., 2016), including fishes, although the phenotypic consequences of genomic modifications resulting from oil exposure have not been obvious in many cases. This merits priority for future research because of the potential long-term consequences for populations and ecosystems.
Sensitive Habitats
In recent years, literature reviews have been published on the impacts of oil spills on shoreline or nearshore habitats (Bejarano and Michel, 2016; Duke, 2016; Michel and Rutherford, 2014; Turner and Renegar, 2017). In general, the scale of impacts and speed of recovery varies depending on the spill size and the magnitude of response actions or treatment intensity. However, because of their specific focus, some of these reviews have included only limited discussions related to dispersants. While spills of opportunity have provided valuable information on the impacts of oil
on sensitive habitats, most have not involved the use of dispersants. Thus, the lack of comparative field studies on the impacts of treated versus untreated oil has restricted the understanding of the hazard posed by dispersant use. One exception has been the 1984 Tropical Oil Pollution Investigations in Coastal Systems (TROPICS) field study (reviewed in NRC, 2005). The TROPICS is the longest monitored field study. It has been monitored for 25-plus years to assess the impacts of chemically dispersed oil on nearshore shallow-water (< 1 m depth) habitats (Ballou et al., 1989). This field study simulated maximum exposure from a single dispersant application for exposures of intertidal mangroves, subtidal seagrass, and shallow-water corals to Prudhoe Bay oil released in boom-enclosed areas (900 m2) and chemically dispersed with Corexit® 9527. Monitoring within 2 years post exposure found declines in the abundance of corals and associated fauna as well as reduced coral growth rate in one species in the chemically dispersed area (Ballou et al., 1989), with complete recovery after 10 years (DeMicco et al., 2011). In contrast, oil penetrated the substrate at the non-dispersed site, serving as a source of hydrocarbons to adjacent habitats (DeMicco et al., 2011). As a direct result of hydrocarbon leaching from the substrate, seagrass beds of Thalassia testudinum at the non-dispersed area had a 58% decrease in coverage and slower growth rates compared to the chemically dispersed area (Baca and Getter, 1984; DeMicco et al., 2011). Other field studies have also documented higher oil persistence in nearshore sediments of untreated oil areas compared to areas treated with dispersants (reviewed in NRC, 1989, 2005; see also Blackall and Sergy, 1981; Gilfillan et al., 1986, for details). For example, the 3-year investigation of the Baffin Island Oil Spill Project showed lower incorporation of petroleum hydrocarbons in Arctic subtidal sediments following an experimental release near the bottom of chemically dispersed (Corexit® 9527) Lagomedio crude oil than did a release of untreated oil (Boehm et al., 1987).
Although field studies in nearshore areas have provided valuable information, it is important to note that at least in the United States, dispersants are not preauthorized for use in shallow waters (generally < 10 m depth, or < 3 nautical miles from the shoreline), and that best management practices are in place to minimize impacts of response actions on sensitive habitats. Deepwater benthos in offshore marine environments may be exposed to oil by the transport of organic and inorganic particles from the upper layers of the water column via particle formation and sinking through a number of processes (see Chapter 2). However, the role of particles in transferring oil to deeper waters with and without the use of dispersants is not well understood.
Wildlife
Surface or subsurface dispersant use results in small oil droplets with larger surface-to-volume ratios that enhance the dissolution of soluble and semi-volatile compounds, resulting in lower concentrations of airborne volatiles and enhancing safety for response workers (Curd, 2011; Gros et al., 2017; see Chapters 2, 4, and 7 for details). This also potentially reduces impacts to air breathing wildlife at the water surface. Despite this potential reduction in exposure to volatile compounds, the hazard posed by dispersant use to wildlife under field conditions is not fully understood because it is difficult to differentiate the impacts of chemically dispersed oil from those of physically dispersed oil. Most of the current knowledge on oil spill impacts to wildlife has been generated through controlled laboratory exposures or from real-world incidents that did not involve the use of dispersants.
It is generally well known that cetaceans could be susceptible to the inhalation of volatile oil fractions and to the inhalation and aspiration of oil droplets at the water surface, which could cause tissue damage along the respiratory tract and lungs, resulting in inflammation of airways, lung disease, and pneumonia (Engelhardt, 1983; Geraci and Aubin, 1988; Schwacke et al., 2013; Takeshita et al., 2017). Inhaled or aspired oil could result in prolonged exposures to lung tissue or in the absorption of hydrocarbons into the bloodstream during long dives. There is evidence from
laboratory studies that dispersant use to treat oil slicks causes aerolization of small oil droplets (Afshar-Mohajer et al., 2018) at the water surface-air interface where cetaceans breathe. Although these droplets could be aspired into the blowhole of cetaceans, the degree of exposure and the implications on their health are not fully understood and require further investigation. While this exposure pathway is a source of concern, best management practices are in place during surface dispersant applications to minimize direct exposure to marine mammals, which would reduce the likelihood of exposure to aerosolized oil droplets. These practices include having trained wildlife observers confirming the absence of cetaceans, birds, and turtles within 1 km of aerial dispersant operations.
Compared to cetaceans, oils spills pose a greater fouling hazard to furred marine mammals, marine sea turtles, waterfowl, and diving birds, because they spend large amounts of time at the water surface. Thus, effective chemical dispersion of surface slicks may decrease oil concentration and thickness, potentially reducing the risk of exposure to these wildlife. Sea turtles could be exposed to oil at the water surface via inhalation of volatile fractions and ingestion of oil mistaken as food, which could cause skin irritation and lesions as well as alteration of respiration and diving patterns (Albers and Loughlin, 2003; Curd, 2011; Lutcavage et al., 1995; Lutz and Lutcavage, 1989; NRC, 2013). For example, following the Ixtoc I and DWH oil spills, large numbers of sea turtles were found to have oil in their oral and nasal cavities and in their digestive tracts (Hall et al., 1983; Mitchelmore et al., 2017). A study on the impacts of chemically dispersed oil on sea turtle embryos resulted in no adverse impacts (Van Meter et al., 2006). Commonly reported impacts of oil spills on birds are associated with dermal exposure and fouling, which reduces buoyancy, water repellency, and insulation provided by feathers and leads to disruptions in thermoregulation, causing hypothermia (Duerr et al., 2011; Jenssen, 1994; Jenssen and Ekker, 1991; NRC, 2005; O’Hara and Morandin, 2010; Whitmer et al., 2017). Laboratory studies have found that chemically dispersed oil alters the structure and geometry of common murre (Uria aalge) feathers, causing a disruption of waterproofing properties (Duerr et al., 2011). A related study also found that both physically and chemically dispersed oil reduced waterproofing of this same species in a dose-dependent manner (Whitmer et al., 2017). Although the same study found negative impacts on waterproofing following direct exposure to Corexit® 9500A alone, best management practices are in place during surface dispersant applications to minimize direct dispersant spraying of wildlife. Direct application of undiluted Corexit® 9500 to mallard (Anas platyrhyncos) eggs also led to embryotoxicity (i.e., reduced hatching success, altered development) (Wooten et al., 2012), though this direct exposure pathway is unlikely as dispersants are not intentionally applied to adult birds or developing eggs.
Given the relatively limited information, it is clear that studies are needed to address the uncertainties associated with the impacts of chemically dispersed oil relative to floating oil and physically dispersed oil on wildlife. Although a similar conclusion was reached by NRC previously (NRC, 1989, 2005), relatively few studies have been conducted since those recommendations were made. The current state of science is to use oil thickness as the dose metric. See French-McCay (2016) for a review of studies used to establish screening thresholds for oil thickness to wildlife and shore habitats. It is unclear, however, whether this is the correct dose metric, whether the suggested thresholds are correct, and whether the same threshold applies for chemically and physically dispersed oil. In addition, other exposure pathways (e.g., inhalation of vapors or oil aerosols, ingestion of contaminated diet) are not considered.
APPLICATION TO THE CONTEXT OF FIELD EXPOSURES
When considering dispersant use for a spill response scenario, it is important to acknowledge the hazards to aquatic resources due to the toxicity of the oil itself. Unmitigated floating oil slicks pose significant hazards to wildlife, especially animals that rely on the surface of the ocean to rest,
feed, or breathe air. During higher sea states, untreated oil will be naturally dispersed, resulting in elevated hydrocarbon concentrations even without the use of dispersants. So, the decision to use dispersants must account for the risks posed by untreated oil as compared to the risk of chemically dispersing the oil (see Chapter 6). One important consideration is the extent and speed of habitat and population recovery after initial impact. Another important consideration is the broad range of sensitivity among species—and life stage sensitivity differences within a single species—so proper identification of the species (and life stages) present in any area where dispersant use is being considered is needed to make sound decisions (see Chapter 6).
It is also important to recognize that ongoing scientific research plays an important role in increasing our understanding of environmental challenges in the context of potential dispersant use. Because each spill has its own challenges and environmental settings, the practical application of scientific knowledge, especially from toxicity studies, may not be direct because most of the available data may not represent typical environmental field conditions. Disparities arise from the fact that the hypotheses being tested by scientists in the laboratory may not always align with the scientific needs of spill responders in the field. Generally, exposure concentrations under laboratory conditions are held relatively constant for a prolonged period to ensure that dosing is sufficiently high to elicit a toxicological response. In contrast, more representative test protocols for operational decisions are those from exposures that allow for water mixing and dilution during the exposure period (e.g., Aurand and Coelho, 2005; Bejarano et al., 2014b; Clark et al., 2001). As a result, what is toxic under controlled laboratory conditions, even those from more representative, declining exposures, does not necessarily translate into similar effects under field conditions.
New scientific information has been generated through the NRDA process from multiple prior oil spills. To support injury assessments and damage quantification, NRDA generates information using reproducible standard scientific approaches, which often involve toxicity testing under controlled laboratory conditions. However, some of the same constraints previously described may also apply to data developed under the NRDA protocols. Despite these challenges, scientific knowledge from laboratory exposures is valuable because such knowledge answers important questions on how aquatic organisms respond to oil exposures. Furthermore, laboratory toxicity studies have facilitated the development of models that provide a scientific link between laboratory and field exposures and effects.
The development of the models described in this chapter relies on the toxicity data generated from toxicity tests. Rather than developing toxicity tests that attempt to simulate the exposure and duration in field exposures, the committee recommends that toxicity tests be designed to calibrate and validate the toxicity models at environmentally realistic concentrations. The toxicity models would be used together with environmental fate models discussed in Chapters 6 and 7 to evaluate the exposure and toxicity associated with various response options, in particular, the potential costs and benefits of dispersant use.
In order to evaluate the impact of dispersant use, it is important to understand the complexity of exposures that generally occur under field conditions. In the water column, the toxicity of physically or chemically dispersed oil relates to these four factors:
- Concentration exceeding known acute or chronic toxicity thresholds for the specific oil;
- Duration of exposure above toxic thresholds;
- Spatial and temporal distribution of marine life; and
- Species sensitivity to oil exposure above the acute or chronic toxicity thresholds.
When examining the expected initial concentrations of dispersed oil, it is important that both lethal and sublethal (e.g., impairment in growth, reproduction, respiration rates) effects are considered. Laboratory tests can identify species thresholds both for mortality and for these
other serious sublethal effects. Figures 3.9 and 3.17 present examples of acute and chronic HC5 concentration, which accounts for species sensitivity by setting the effect concentration that is protective of 95% of the tested species using the SSD.
The use of these laboratory results to assess the potential risks of dispersed oil to marine life provides an incomplete understanding of the potential effects in the ocean, because laboratory data rarely approximate field exposure. Open ocean field experiments conducted in the North Sea showed rapid dilution of dispersed oil concentrations following dispersant application (see Box 6.1). The rapid dilution of dispersed oil is also documented in a literature review (Bejarano et al., 2014b) that included field measurements from extensive studies during the DWH spill (OSAT, 2010), in which less than 1% of water samples analyzed exceeded aquatic toxicity benchmarks. Furthermore, many mobile marine organisms may display an avoidance behavior, not remaining in a region with continual exposure to dispersed oil. As a result of the different exposure durations, a direct comparison between laboratory and field effects is problematic (Aurand and Coelho, 2005; Bejarano et al., 2014b; Clark et al., 2001; IPIECA, 2015). For these reasons, it is logical to calibrate a coupled exposure and effects model under laboratory exposure conditions using a given oil to predict toxicity under different exposure conditions for other oils.
Despite the limitations of extrapolating laboratory to field effects, efforts have been made to find practical applications of the substantial toxicity data produced over several decades. The Coastal Response Research Center sponsored a project in 2013 to make toxicity data for dispersants, oil, and dispersed oil more readily available by centralizing toxicity data. The end result is a data compilation that provides a quantitative basis for a more thorough assessment of hazard concentrations (Bejarano et al., 2016; NOAA ERD, 2015). This new tool has been incorporated into the NOAA Chemical Aquatic Fate and Effects database, allowing users to quickly develop SSDs to improve hazard estimates during oil spill response activities and exercises. The database enables users to filter information that specifically relates to a particular oil or dispersant, which allows decision makers to rapidly access past research and apply it in a meaningful way. From a practical perspective, the use of SSDs is advantageous because:
- SSDs provide potentially useful information to stakeholders involved in oil spill response decision making.
- Even when the SSDs are based on standard laboratory exposures rather than real-world exposure regimes, they can provide scientifically defensible benchmarks for dispersant use decisions.
- In the absence of toxicity data for species in extreme environments—such as Arctic and deepwater—adjustments to the SSD can be made to predict the change that might result to the HC5.
Finally, in recent years there has been dramatic expansion of toxicity research well beyond the organismal unit into areas of metabolomics, genetics, species interactions, and ecosystem-level responses (Deepwater Horizon Natural Resource Damage Assessment Trustees, 2016; Tarnecki and Patterson, 2015). More recent studies are also advancing the understanding of the effects of oil and dispersed oil exposure when compounded by other environmental stressors, such as UV radiation, temperature, salinity, etc. While these advances in science offer new insights into oil toxicology, the scientific community should remain vigilant about appropriate interpretation of these data and meaningfully communicating appropriate results to the operational response community.
FINDINGS AND RECOMMENDATIONS
Finding: The use of total petroleum hydrocarbons (TPHs) and total polycyclic aromatic hydrocarbons (TPAHs) as dose metrics are not sufficiently predictive of observed toxicity of complex mixtures of oil and dispersed oil (see Figure 3.12). The use of toxic units (TUs) is the only presently available, scientifically sound dose metric.
Finding: Toxicity tests using variable dilution cannot be used to determine if exposure media containing dispersed oil is more or less toxic than is the exposure media containing untreated oil, because (1) dilution changes the concentration of microdroplets in the water-accommodated fractions (WAFs), (2) dilution changes the concentrations of dissolved components in the WAFs, and (3) the concentration dissolved components cannot be estimated from the dilution. By contrast, toxicity tests using the variable loading design can be used to make this determination because the dissolved concentration of the oil components in the WAFs are the same at the same oil loading with or without dispersants.
Finding: Data from high-energy WAF (HEWAF) experiments are difficult to interpret because of the creation of excess microdroplets relative to the dissolved concentrations (see Figure 3.13).
Finding: Data from recent variable loading toxicity tests indicate that dispersed oil is not more toxic than is untreated oil at concentrations below approximately 100 mg oil/L. At concentrations above approximately 100 mg oil/L, the presence of dispersants contributes to increased toxicity (see Figure 3.5). At oil loadings much lower than approximately 100 mg/L, the toxicity hazard posed by dispersed oil does not come from the dispersant itself because the concentrations of dispersants in the toxicity tests are below the acute Hazard Concentration 5% (HC5) (see Figure 3.2).
Finding: The acute and chronic TUs of saturated WAFs for a range of oils, and the chronic and acute HC5, vary over an order of magnitude depending on the composition of the oil (see Figures 3.9 and 3.17).
Finding: The use of passive dosing shows promise for generating exposure media without microdroplets, thereby enabling toxicity testing of dissolved components only. Passive sampling approaches, such as solid-phase microextraction (SPME), show promise for rapid field and laboratory sampling as well. Further standardization of these testing procedures would ensure their interlaboratory comparability and reproducibility.
Finding: Phototoxicity models exist but have not been utilized for determining the contribution of phototoxicity to the overall toxicity of oil in the field.
Recommendation: The use of toxic units should be integrated into revised toxicity testing standards, evaluation criteria for models, and response option risk analysis. This represents a paradigm shift away from developing toxicity tests that attempt to reproduce field exposure conditions and toward developing a consistent means of using toxicity metrics such as HC5 and LC50 for toxicity models used with fate and transport models to compare the exposure and toxicity of various response options, including dispersants.
Recommendation: Recent advances in predictive toxicity models under both lab and field conditions should be incorporated into user accessible tools. The availability of such tools
would facilitate further calibration, validation, and/or refinement as well as support decision making. Future models could include the inclusion of environmental factors (UV light, pressure) and the role of both photosensitization and photomodification on predicted oil toxicity.
Recommendation: Models that simulate the distribution and toxicity of spilled oil need to be validated by comparing their predictions for the same test conditions. Organizations that fund model development should ensure that models, such as PETROTOX and Oiltox, are enhanced and validated through interlab comparisons. As new models evolve for more refined toxicity prediction (e.g., models of phototoxicity), the same should be done. If a future field trial is planned, further validation of these models should be incorporated into the design.
Recommendation: Modify CROSERF protocols so that future toxicity testing data are geared toward informing and validating toxicity models: media preparation, exposure regimes, chemical characterization of exposure media and test species after exposure, and reporting of dose metrics.
Recommendation: Consideration should be given to choosing a number of standardized oil compositions for use as inputs to fate and toxicity model runs. Characterizing these oils into a manageable number of hydrocarbon blocks—consistent with acute and chronic toxicity, phototoxicity, and other more specialized properties—would also be useful.
Recommendation: Funding agencies, research consortia, and other sponsoring groups should require that research teams use standardized toxicity testing methods, such as those developed by the Chemical Response to Oil Spills Ecological Effects Research Forum (CROSERF) program, and analytical chemistry protocols to fully characterize hydrocarbon composition and concentrations in the exposure media. For testing the effect of dispersant, the variable loading test design is recommended.
Recommendation: Improve standardization of oil characterization and composition to provide consistent input that could be coupled with fate and effect models, including lower resolution “fit for purpose” analyses able to quantify oil pseudo-components applicable to trajectory oil spill modeling.
Recommendation: More broadly test the use of passive sampling devices, such as SPME fibers, for real-time field monitoring of dissolved oil exposures and possible toxicity prediction.
















































